Syntheses and Characterization of Two Dicyanamide Compounds Containing Monovalent Cations: Hg2[N(CN)2]2 and Tl[N(CN)2]
Abstract
1. Introduction
2. Results and Discussion
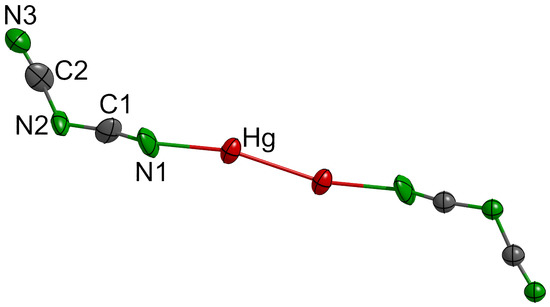
2.1. Structural Description and Discussion
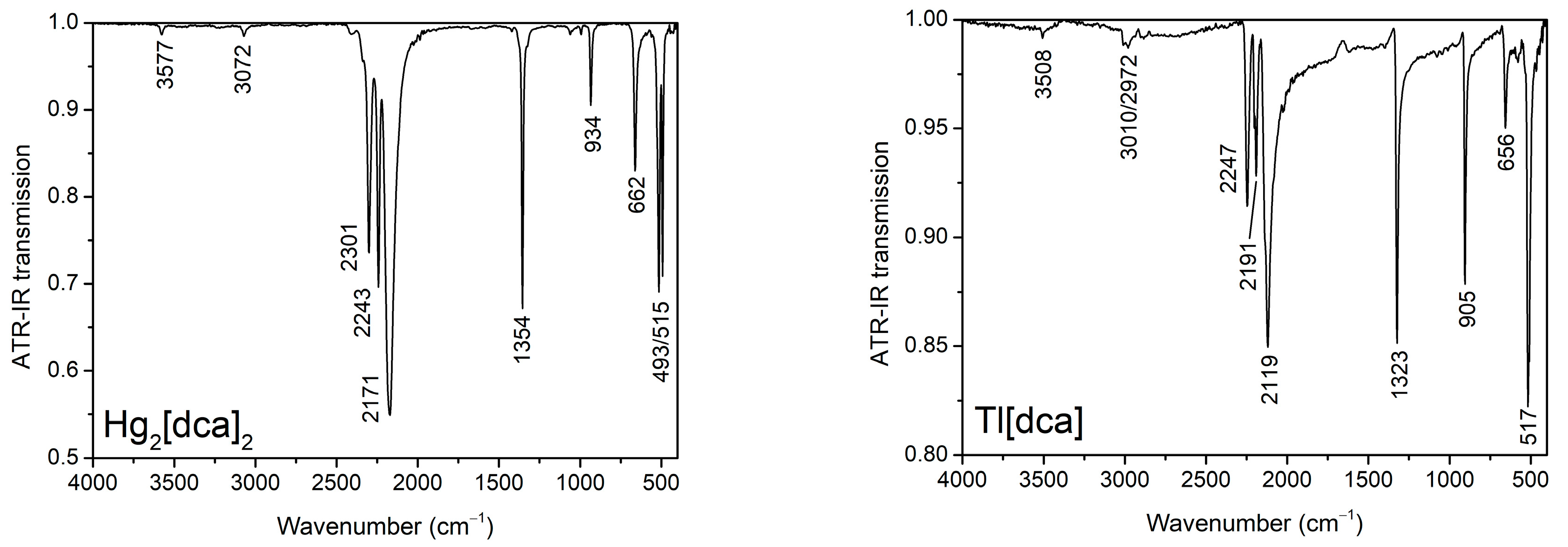
2.2. IR-Spectra
3. Materials and Methods
3.1. Syntheses
3.2. Single Crystal Diffraction
3.3. Infrared Spectra
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Scholz, T.; Görne, A.L.; Dronskowski, R. Itinerant nitrides and salt-like guanidinates—The diversity of solid-state nitrogen chemistry. Prog. Solid State Chem. 2018, 51, 1–18. [Google Scholar] [CrossRef]
- Sougrati, M.T.; Arayamparambil, J.J.; Liu, X.; Mann, M.; Slabon, A.; Stievano, L.; Dronskowski, R. Carbodiimides as energy materials: Which directions for a reasonable future? J. Chem. Soc. Dalton Trans. 2018, 47, 10827–10832. [Google Scholar] [CrossRef] [PubMed]
- Sougrati, M.T.; Darwiche, A.; Liu, X.; Mahmoud, A.; Hermann, R.P.; Jouen, S.; Monconduit, L.; Dronskowski, R.; Stievano, L. Transition-Metal Carbodiimides as Molecular Negative Electrode Materials for Lithium- and Sodium-Ion Batteries with Excellent Cycling Properties. Angew. Chem. Int. Ed. 2016, 55, 5090–5095. [Google Scholar] [CrossRef] [PubMed]
- Jürgens, B.; Höppe, H.A.; Irran, E.; Schnick, W. Transformation of Ammonium Dicyanamide into Dicyandiamide in the Solid. Inorg. Chem. 2002, 41, 4849–4851. [Google Scholar] [CrossRef] [PubMed]
- Reckeweg, O.; DiSalvo, F.J.; Schulz, A.; Blaschkowski, B.; Jagiella, S.; Schleid, T. Synthesis, Crystal Structure, and Vibrational Spectra of the Anhydrous Lithium Dicyanamide Li[N(CN)2]. Z. Anorg. Allg. Chem. 2014, 640, 851–855. [Google Scholar] [CrossRef]
- Starynowicz, P. Structure of caesium dicyanamide. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1991, 47, 2198–2199. [Google Scholar] [CrossRef]
- Irran, E.; Jürgens, B.; Schnick, W. Trimerization of alkali dicyanamides M[N(CN)2] and formation of tricyanomelaminates M3[C6N9] (M = K, Rb) in the melt: Crystal structure determination of three polymorphs of K[N(CN)2], two of Rb[N(CN)2], and one of K3[C6N9] and Rb3[C6N9] from X-ray powder diffractometry. Chem. Eur. J. 2001, 7, 5372–5381. [Google Scholar] [PubMed]
- Jürgens, B.; Irran, E.; Schneider, J.; Schnick, W. Trimerization of NaC2N3 to Na3C6N9 in the Solid: Ab Initio Crystal Structure Determination of Two Polymorphs of NaC2N3 and of Na3C6N9 from X-ray Powder Diffractometry. Inorg. Chem. 2000, 39, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Jürgens, B.; Irran, E.; Schnick, W. Syntheses, Vibrational Spectroscopy, and Crystal Structure Determination from X-Ray Powder Diffraction Data of Alkaline Earth Dicyanamides M[N(CN)2]2 with M = Mg, Ca, Sr, and Ba. J. Solid State Chem. 2001, 157, 241–249. [Google Scholar] [CrossRef]
- Manson, J.L.; Kmety, C.R.; Epstein, A.J.; Miller, J.S. Spontaneous Magnetization in the M[N(CN)2]2 (M = Cr, Mn) Weak Ferromagnets. Inorg. Chem. 1999, 38, 2552–2553. [Google Scholar] [CrossRef]
- Manson, J.L.; Kmety, C.R.; Huang, Q.-Z.; Lynn, J.W.; Bendele, G.M.; Pagola, S.; Stephens, P.W.; Liable-Sands, L.M.; Rheingold, A.L.; Epstein, A.J.; et al. Structure and Magnetic Ordering of MII[N(CN)2]2 (M = Co, Ni). Chem. Mater. 1998, 10, 2552–2560. [Google Scholar] [CrossRef]
- Reckeweg, O.; Dinnebier, R.E.; Schulz, A.; Blaschkowski, B.; Schneck, C.; Schleid, T. About the air- and water-stable copper(I) dicyanamide: Synthesis, crystal structure, vibrational spectra and DSC/TG analysis of Cu[N(CN)2]. Z. Naturforsch. B Chem. Sci. 2017, 72, 159–165. [Google Scholar] [CrossRef]
- Hodgson, S.A.; Hunt, S.J.; Sørensen, T.J.; Thompson, A.L.; Reynolds, E.M.; Faulkner, S.; Goodwin, A.L. Anomalous Thermal Expansion and Luminescence Thermochromism in Silver(I) Dicyanamide. Eur. J. Inorg. Chem. 2016, 2016, 4378–4381. [Google Scholar] [CrossRef]
- Reckeweg, O.; Schulz, A.; Schneck, C.; Lissner, F.; Schleid, T. Syntheses, single-crystal structures, vibrational spectra and DSC/TG analyses of orthorhombic and trigonal Ag[N(CN)2]. Z. Naturforsch. B Chem. Sci. 2016, 71, 827–834. [Google Scholar] [CrossRef]
- Manson, J.L.; Lee, D.W.; Rheingold, A.L.; Miller, J.S. Buckled-layered Structure of Zinc Dicyanamide, ZnII[N(CN)2]2. Inorg. Chem. 1998, 37, 5966–5967. [Google Scholar] [CrossRef] [PubMed]
- Jürgens, B.; Irran, E.; Höppe, H.A.; Schnick, W. Phase Transition of a Dicyanamide with Rutile-like Structure: Syntheses and Crystal Structures of α- and β-Cd[N(CN)2]2. Z. Anorg. Allg. Chem. 2004, 630, 219–223. [Google Scholar] [CrossRef]
- Jürgens, B.; Irran, E.; Schnick, W. Synthesis and characterization of the rare-earth dicyanamides Ln[N(CN)2]3 with Ln = La, Ce, Pr, Nd, Sm, and Eu. J. Solid State Chem. 2005, 178, 72–78. [Google Scholar] [CrossRef]
- Nag, A.; Schmidt, P.J.; Schnick, W. Synthesis and Characterization of Tb[N(CN)2]3·2H2O and Eu[N(CN)2]3·2H2O: Two New Luminescent Rare-Earth Dicyanamides. Chem. Mater. 2006, 18, 5738–5745. [Google Scholar] [CrossRef]
- Nag, A.; Schnick, W. Synthesis, Crystal Structure and Thermal Behavior of Gadolinium Dicyanamide Dihydrate Gd[N(CN)2]3·2H2O. Z. Anorg. Allg. Chem. 2006, 632, 609–614. [Google Scholar] [CrossRef]
- Reckeweg, O.; Wakabayashi, R.H.; DiSalvo, F.J.; Schulz, A.; Schneck, C.; Schleid, T. About alkali metal dicyanamides: Syntheses, single-crystal structure determination, DSC/TG and vibrational spectra of KCs[N(CN)2]2 and NaRb2[N(CN)2]3·H2O. Z. Naturforsch. B Chem. Sci. 2015, 70, 365–372. [Google Scholar] [CrossRef]
- Reckeweg, O.; DiSalvo, F.J. Synthesis and single-crystal structure of the pseudo-ternary compounds LiA[N(CN)2]2 (A = K or Rb). Z. Naturforsch. B Chem. Sci. 2016, 71, 157–160. [Google Scholar] [CrossRef]
- Jürgens, B.; Milius, W.; Morys, P.; Schnick, W. Trimerisierung von Dicyanamid-Ionen C2N3‒ im Festkörper—Synthesen, Kristallstrukturen und Eigenschaften von NaCs2(C2N3)3 und Na3C6N9·3H2O. Z. Anorg. Allg. Chem. 1998, 624, 91–97. [Google Scholar] [CrossRef]
- Mann, M.; Reckeweg, O.; Dronskowski, R. Synthesis and Characterization of the New Dicyanamide LiCs2[N(CN)2]3. Inorganics 2018, 6, 108. [Google Scholar] [CrossRef]
- Madelung, W.; Kern, E. Über Dicyanamid. Justus Liebigs Ann. Chem. 1922, 427, 1–26. [Google Scholar] [CrossRef]
- Kuhn, M.; Mecke, R. IR-Spektroskopische Untersuchungen am Dicyanamid-Anion, [N(CN)2]‒. Chem. Ber. 1961, 94, 3010–3015. [Google Scholar] [CrossRef]
- Dorm, E. Studies on the Crystal Chemistry of the Mercurous Ion and of Mercurous Salts; Stockholm Univ.: Stockholm, Sweden, 1970; pp. 1–25. [Google Scholar]
- Reckeweg, O.; Simon, A. Azide und Cyanamide—Ähnlich und doch anders/Azides and Cyanamides – Similar and Yet Different. Z. Naturforsch. B Chem. Sci. 2003, 58, 1097–1104. [Google Scholar] [CrossRef]
- Biltz, W. Raumchemie der festen Stoffe; Verlag von Leopold Voss: Leipzig, Germany, 1934. [Google Scholar]
- Stork, L.; Liu, X.; Fokwa, B.P.T.; Dronskowski, R. Crystal Structure Determination of Thallium Carbodiimide, Tl2NCN. Z. Anorg. Allg. Chem. 2007, 633, 1339–1342. [Google Scholar] [CrossRef]
- X-Area Integrate; 1.71.0.0; Stoe & Cie GmbH: Darmstadt, Germany, 2016.
- X-Area X-Red; 1.63.2.0; Stoe & Cie GmbH: Darmstadt, Germany, 2017.
- Palatinus, L.; Chapuis, G. SUPERFLIP– a computer program for the solution of crystal structures by charge flipping in arbitrary dimensions. J. Appl. Crystallogr. 2007, 40, 786–790. [Google Scholar] [CrossRef]
- Petříček, V.; Dušek, M.; Palatinus, L. Crystallographic Computing System JANA2006: General features. Z. Kristallogr. 2014, 229, 345–352. [Google Scholar] [CrossRef]
- SADABS; 2014/5; Bruker AXS Inc.: Madison, WI, USA, 2014.
- XPREP; 2014/5; Bruker AXS Inc.: Madison, WI, USA, 2014.
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
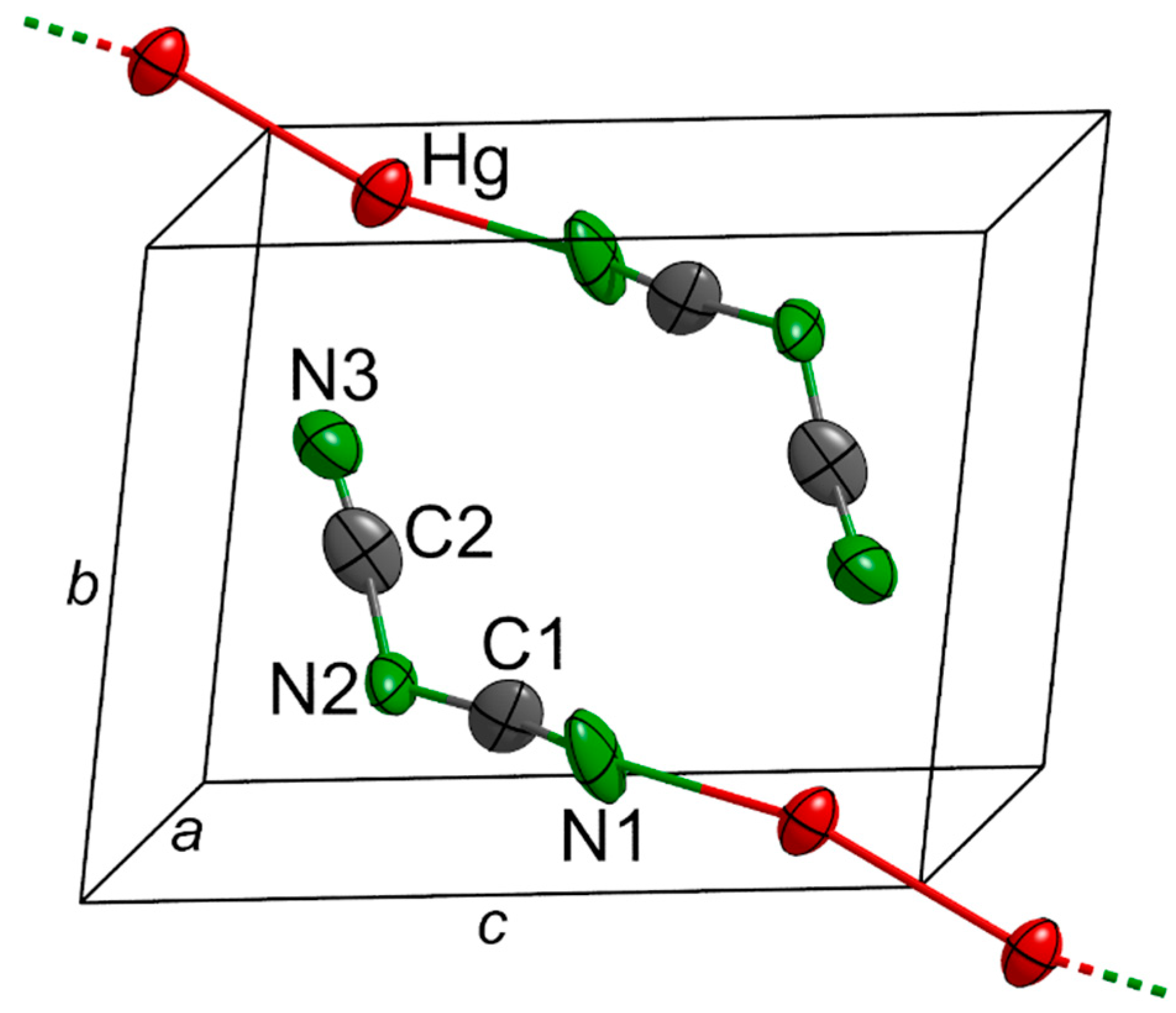
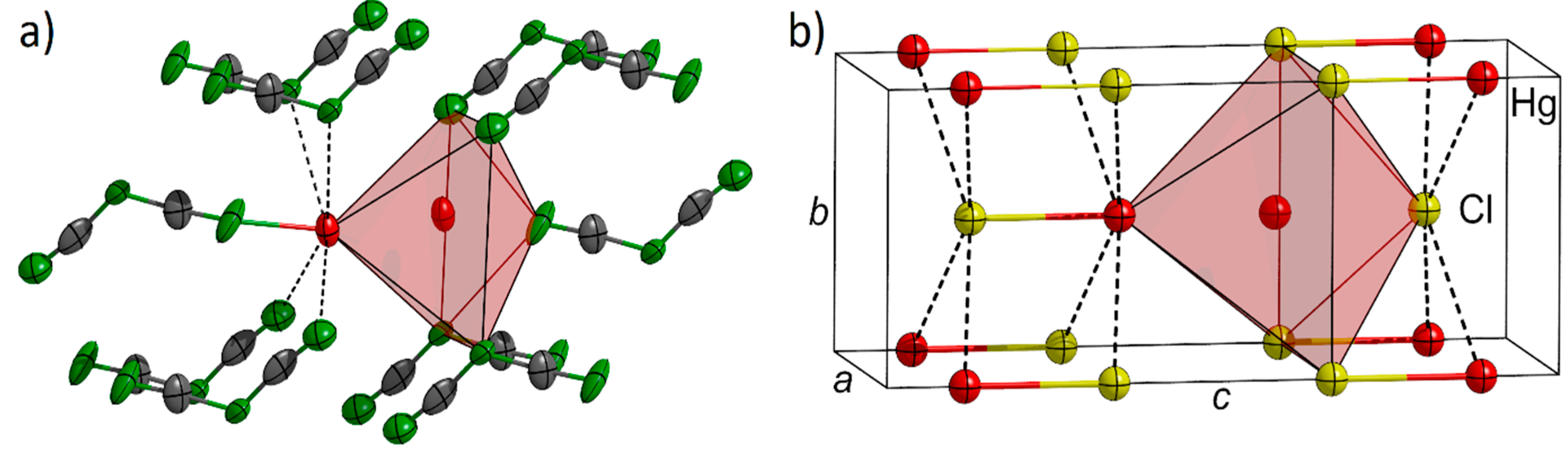

| Hg2[dca]2 | (°) | Tl[dca] | (°) |
|---|---|---|---|
| ∡(N1–C1–N2) | 171.6(14) | ∡(N1–C1–N2) | 172.0(6) |
| ∡(N2–C2–N3) | 173.2(13) | ∡(N2–C2–N3) | 172.6(6) |
| ∡(C1–N2–C2) | 119.4(10) | ∡(C1–N2–C2) | 120.6(5) |
| (Å) | (Å) | ||
| N1–C1 | 1.148(14) | N1–C1 | 1.148(8) |
| C1–N2 | 1.325(13) | C1–N2 | 1.316(8) |
| N2–C2 | 1.317(16) | N2–C2 | 1.319(7) |
| C2–N3 | 1.108(16) | C2–N3 | 1.148(7) |
| Hg–Hg | 2.518(3) | Tl–Tl (2×) | 3.6153(7) |
| Hg–N1 | 2.166(10) | Tl–Tl (2×) | 4.5129(7) |
| Hg–N2 | 3.412(11) | Tl–N1 (2×) | 3.089(4) |
| Hg–N2 | 3.084(9) | Tl–N1 (2×) | 3.082(4) |
| Hg–N3 | 2.612(12) | Tl–N2 (2×) | 3.053(5) |
| Hg–N3 | 2.834(10) | Tl–N3 (2×) | 2.870(4) |
| M[dca] | Cell Volume (Å3), Z | T (K) | Molar Volume (cm3·mol−1) | M+ Volume (cm3·mol−1) [28] | [dca]− Volume (cm3·mol−1) |
|---|---|---|---|---|---|
| NH4[dca] [4] | 428.32, 4 | 200 | 64.5 | 19.5 | 45.0 |
| Li[dca] [5] | 313.84, 4 | 170 | 47.2 | 1.5 | 45.7 |
| Na[dca] [8] | 345.25, 4 | 293 | 52.0 | 6.5 | 45.5 |
| α-K[dca] [7] | 390.31, 4 | 293 | 58.7 | 16 | 42.7 |
| β-K[dca] [7] | 416.78, 4 | 293 | 62.7 | 16 | 46.7 |
| γ-K[dca] [7] | 415.56, 4 | 293 | 62.5 | 16 | 46.5 |
| α-Rb[dca] [7] | 433.4, 4 | 293 | 65.2 | 20 | 45.2 |
| β-Rb[dca] [7] | 1778.31, 16 | 293 | 66.9 | 20 | 46.9 |
| Cs[dca] [6] | 919.74, 8 | 299 | 69.2 | 26 | 43.2 |
| Cu[dca] [12] | 332.21, 4 | 248 | 50.0 | 5 | 45.0 |
| Ag[dca], o [14] | 349.24, 4 | 293 | 52.6 | 9 | 43.6 |
| Ag[dca], tr [14] | 256.36, 3 | 293 | 51.4 | 9 | 42.4 |
| Hg2[dca]2 | 186.84, 1 | 100 | 112.4 | 16 | 40.2 |
| Tl[dca] | 401.60, 4 | 100 | 60.4 | 18.5 | 41.9 |
| Vibration | ν(Hg2[dca]2) | ν(Tl[dca]) |
|---|---|---|
| σas(N–C≡N) | 493/515 | 517 |
| σs(N–C≡N) | 662 | 656 |
| νs(N–C) | 934 | 905 |
| νas(N–C) | 1354 | 1323 |
| νas(N≡C) | 2171 | 2119 |
| νas(N–C) + νs(N–C) | 2243 | 2191 |
| νs(N≡C) | 2301 | 2247 |
| νas(N≡C) + νs(N–C) | 3072 | 3010/2972 |
| νs(N≡C) + νas(N–C) | 3577 | 3508 |
| Chemical Formula | Hg2[dca]2 | Tl[dca] |
|---|---|---|
| Formula weight (g∙mol−1) | 533.3 | 270.42 |
| Crystal system | triclinic | orthorhombic |
| Space group | P (no. 2) | Pbcm (no. 57) |
| Temperature (K) | 100(2) | 100(2) |
| a (Å) | 3.7089(5) | 8.5770(17) |
| b (Å) | 6.4098(6) | 6.4756(13) |
| c (Å) | 8.150(6) | 7.2306(14) |
| α (°) | 81.575(6) | 90 |
| β (°) | 80.379(7) | 90 |
| γ (°) | 80.195(7) | 90 |
| V (Å3) | 186.84(14) | 401.60(14) |
| Z | 1 | 4 |
| Radiation, λ (Å) | Mo Kα, 0.71073 | Mo Kα, 0.71073 |
| μ (mm−1) | 41.09 | 40.022 |
| Crystal shape and color | Colorless block | Colorless block |
| Crystal size (mm3) | 0.17 × 0.08 × 0.02 | 0.05 × 0.04 × 0.02 |
| ρcalcd (g∙cm−3) | 4.739 | 4.473 |
| Diffractometer | STOE STADIVARI with Hybrid Pixel Counting Detector | Bruker AXS Enraf-Nonius with KappaCCD Detector |
| Absorption correction | Gaussian Integration, STOE X-RED | Gaussian Integration, SADABS 2014/15 |
| Tmin, Tmax | 0.0672, 0.6730 | 0.15236, 0.46282 |
| No. of measured, independent and observed [I > 3σ(I)] reflections | 3671 | 8880 |
| 1263 | 816 | |
| 1154 | 549 | |
| Robs | 4.68 | 1.87 |
| wR2obs | 10.88 | 3.94 |
| Rall | 4.92 | 3.25 |
| wR2all | 10.91 | 4.46 |
| GOFobs | 3.60 | 1.09 |
| No. of parameters, restraints | 55, 0 | 36, 0 |
| Atom | x | y | z | Ueq |
|---|---|---|---|---|
| Hg | 0.96205(10) | 0.90300(6) | 0.14632(4) | 0.01463(12) |
| N1 | 0.923(3) | 0.805(2) | 0.4137(12) | 0.024(3) |
| N2 | 0.619(3) | 0.7427(15) | 0.7062(10) | 0.015(2) |
| N3 | 0.406(3) | 0.4138(17) | 0.8391(12) | 0.020(3) |
| C1 | 0.765(3) | 0.7693(19) | 0.5460(13) | 0.016(3) |
| C2 | 0.510(3) | 0.559(2) | 0.7710(14) | 0.018(3) |
| Atom | U11 | U22 | U33 | U23 | U13 | U12 |
|---|---|---|---|---|---|---|
| Hg | 0.01303(19) | 0.0173(2) | 0.01206(19) | −0.00301(13) | 0.00010(12) | 0.00189(13) |
| N1 | 0.026(5) | 0.038(6) | 0.011(4) | −0.014(4) | 0.005(3) | −0.012(4) |
| N2 | 0.023(4) | 0.012(4) | 0.009(3) | 0.001(3) | −0.001(3) | −0.004(3) |
| N3 | 0.025(5) | 0.018(5) | 0.018(4) | −0.005(4) | −0.002(3) | −0.006(3) |
| C1 | 0.008(4) | 0.022(5) | 0.018(4) | −0.005(4) | 0.000(3) | 0.000(4) |
| C2 | 0.006(4) | 0.027(6) | 0.020(5) | −0.002(4) | 0.004(3) | −0.010(4) |
| Atom | Site | x | y | z | Ueq |
|---|---|---|---|---|---|
| Tl | 4c | 0.31674(2) | ¼ | ½ | 0.01287(6) |
| N1 | 4d | 0.5898(6) | 0.3835(8) | ¾ | 0.0183(10) |
| N2 | 4d | 0.7848(7) | 0.1054(9) | ¾ | 0.0218(12) |
| N3 | 4d | 0.0707(6) | 0.1391(8) | ¾ | 0.0194(10) |
| C1 | 4d | 0.6880(5) | 0.2630(11) | ¾ | 0.0144(10) |
| C2 | 4d | 0.9369(7) | 0.1356(9) | ¾ | 0.0150(10) |
| Atom | U11 | U22 | U33 | U23 | U13 | U12 |
|---|---|---|---|---|---|---|
| Tl | 0.01236(9) | 0.01241(10) | 0.01384(9) | −0.00016(13) | 0 | 0 |
| N1 | 0.0111(19) | 0.013(2) | 0.031(3) | 0 | 0 | −0.002(2) |
| N2 | 0.012(2) | 0.010(2) | 0.043(4) | 0 | 0 | −0.003(2) |
| N3 | 0.015(2) | 0.012(2) | 0.031(3) | 0 | 0 | 0.0007(19) |
| C1 | 0.012(2) | 0.009(3) | 0.022(3) | 0 | 0 | −0.003(3) |
| C2 | 0.017(3) | 0.008(2) | 0.020(3) | 0 | 0 | −0.002(2) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mann, M.; Reckeweg, O.; Nöthling, N.; Goddard, R.; Dronskowski, R. Syntheses and Characterization of Two Dicyanamide Compounds Containing Monovalent Cations: Hg2[N(CN)2]2 and Tl[N(CN)2]. Inorganics 2018, 6, 135. https://doi.org/10.3390/inorganics6040135
Mann M, Reckeweg O, Nöthling N, Goddard R, Dronskowski R. Syntheses and Characterization of Two Dicyanamide Compounds Containing Monovalent Cations: Hg2[N(CN)2]2 and Tl[N(CN)2]. Inorganics. 2018; 6(4):135. https://doi.org/10.3390/inorganics6040135
Chicago/Turabian StyleMann, Markus, Olaf Reckeweg, Nils Nöthling, Richard Goddard, and Richard Dronskowski. 2018. "Syntheses and Characterization of Two Dicyanamide Compounds Containing Monovalent Cations: Hg2[N(CN)2]2 and Tl[N(CN)2]" Inorganics 6, no. 4: 135. https://doi.org/10.3390/inorganics6040135
APA StyleMann, M., Reckeweg, O., Nöthling, N., Goddard, R., & Dronskowski, R. (2018). Syntheses and Characterization of Two Dicyanamide Compounds Containing Monovalent Cations: Hg2[N(CN)2]2 and Tl[N(CN)2]. Inorganics, 6(4), 135. https://doi.org/10.3390/inorganics6040135