Effects of Performance and Task Duration on Mental Workload during Working Memory Task
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
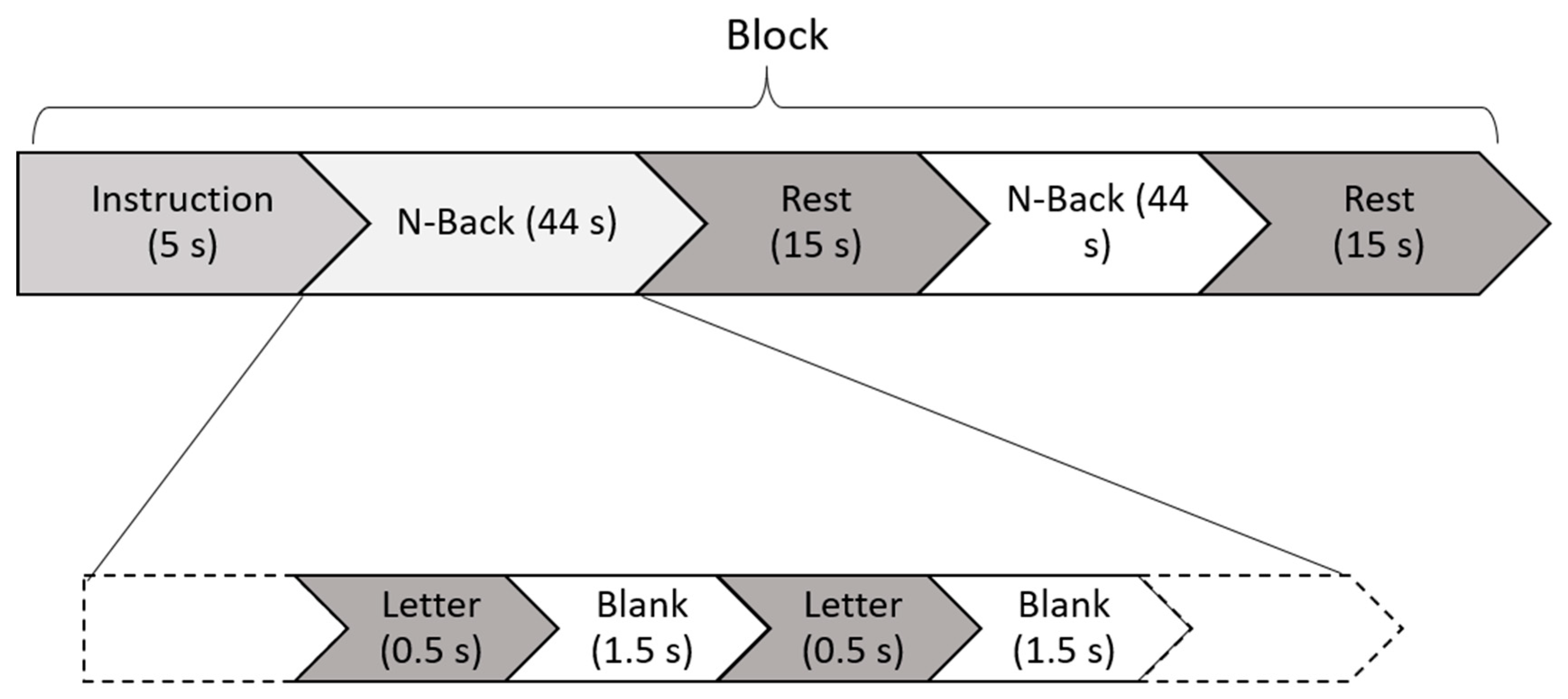
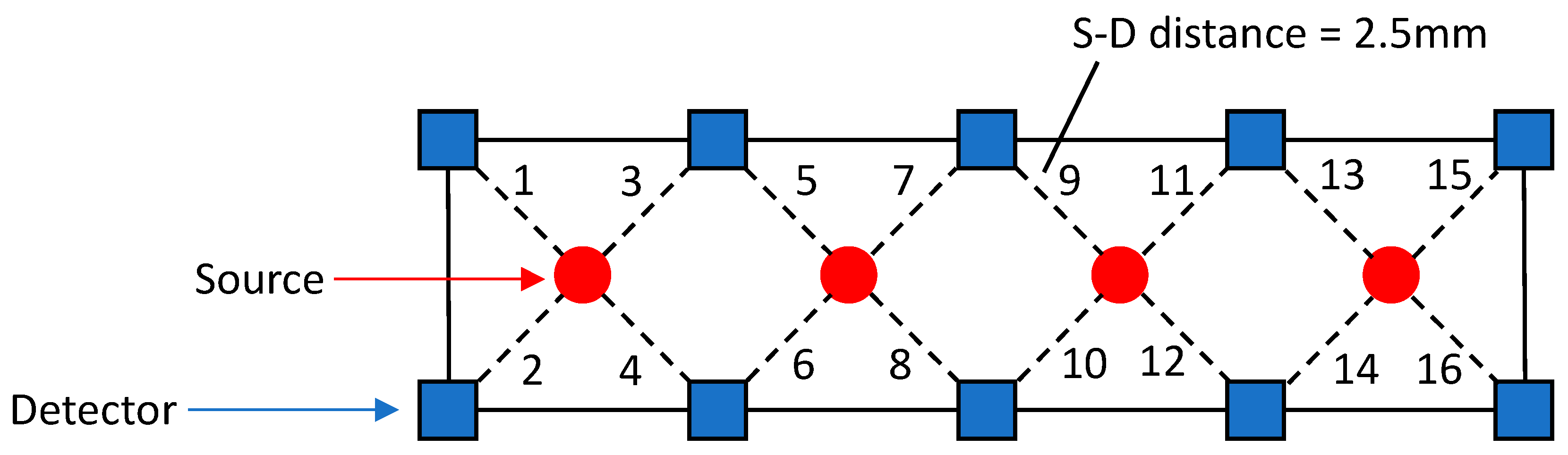
2.2. Task and Experimental Design
2.3. NIRS Data Analysis
2.4. Behavioral Data Analysis
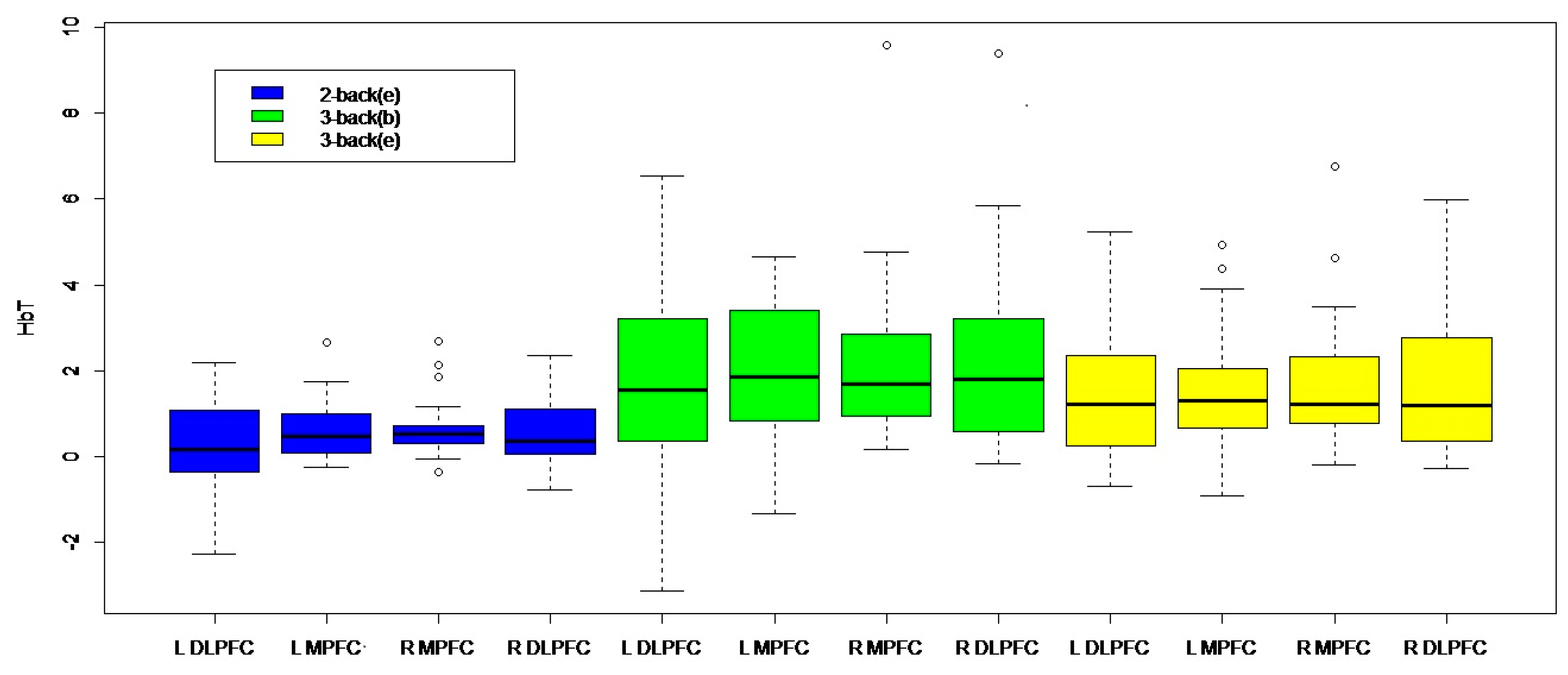
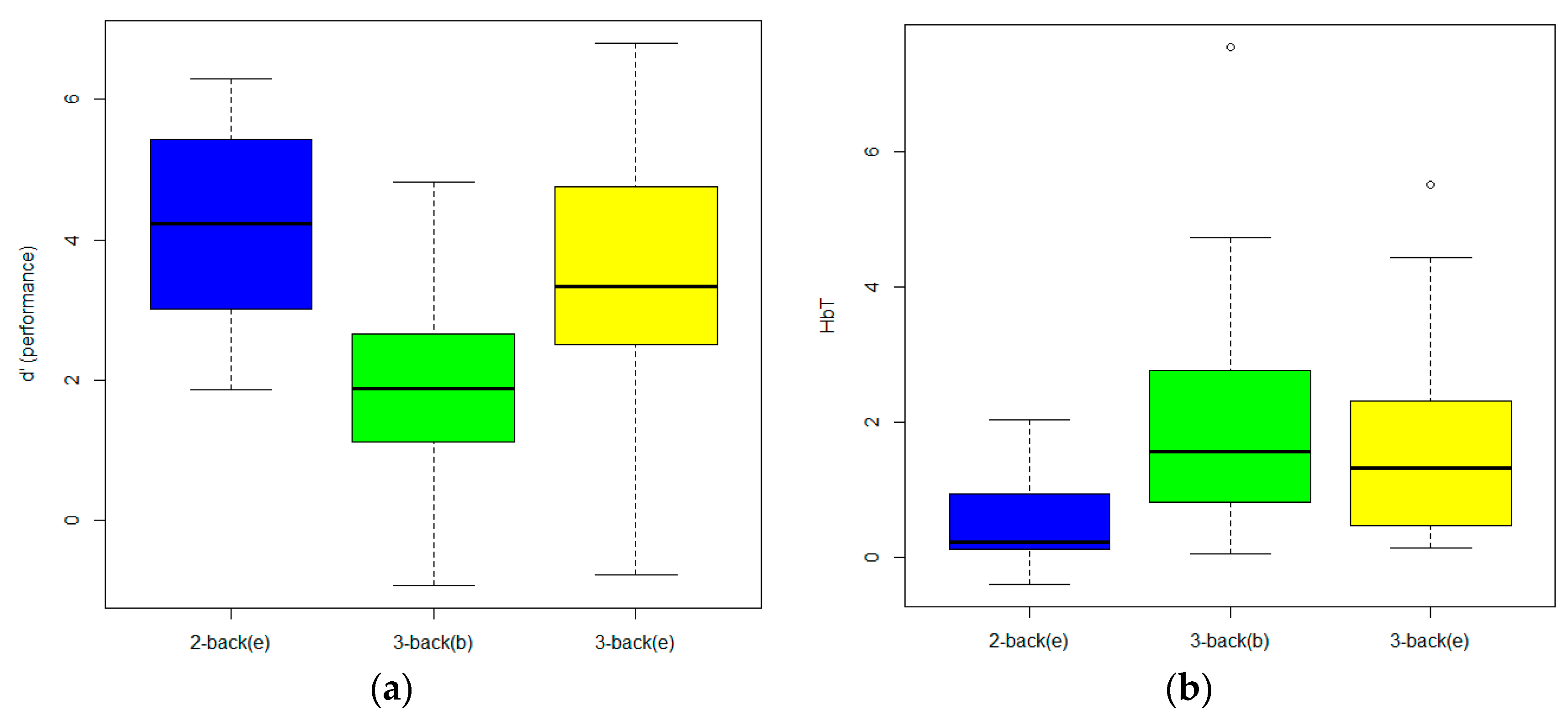
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baddeley, A. Working memory: Looking back and looking forward. Nat. Rev. Neurosci. 2003, 4, 829–839. [Google Scholar] [CrossRef] [PubMed]
- Baddeley, A. Working Memory: Theories, Models, and Controversies. Annu. Rev. Psychol. 2012, 63, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Possin, K.L.; Filoteo, J.V.; Song, D.D.; Salmon, D.P. Spatial and Object Working Memory Deficits in Parkinson’s Disease are Due to Impairment in Different Underlying Processes. Neuropsychol. 2008, 22, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Brunoni, A.R.; Vanderhasselt, M.-A. Working memory improvement with non-invasive brain stimulation of the dorsolateral prefrontal cortex: A systematic review and meta-analysis. Brain Cogn. 2014, 86, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Klingberg, T.; Fernell, E.; Olesen, P.J.; Johnson, M.; Gustafsson, P.; Dahlström, K.; Gillberg, C.G.; Forssberg, H.; Westerberg, H. Computerized Training of Working Memory in Children With ADHD-A Randomized, Controlled Trial. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Barendse, E.M.; Hendriks, M.P.; Jansen, J.F.; Backes, W.H.; Hofman, P.A.; Thoonen, G.; Kessels, R.P.; Aldenkamp, A.P. Working memory deficits in high-functioning adolescents with autism spectrum disorders: Neuropsychological and neuroimaging correlates. J. Neurodev. Disord. 2013, 5, 14. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.A.; Parsa, K.; Geiger, S.; Zaragoza, R.; Kermanian, R.; Miguel, H.; Dashtestani, H.; Chowdhry, F.A.; Smith, E.; Aram, S.; et al. Exploring the role of task performance and learning style on prefrontal hemodynamics during a working memory task. PLoS ONE 2018, 13, e0198257. [Google Scholar] [CrossRef] [PubMed]
- Owen, A.M.; McMillan, K.M.; Laird, A.R.; Bullmore, E.; Bullmore, E. N-back working memory paradigm: A meta-analysis of normative functional neuroimaging studies. Hum. Brain Mapp. 2005, 25, 46–59. [Google Scholar] [CrossRef]
- Curtis, C.E.; D’Esposito, M. Persistent activity in the prefrontal cortex during working memory. Trends Cogn. Sci. 2003, 7, 415–423. [Google Scholar] [CrossRef]
- Amyot, F.; Zimmermann, T.; Riley, J.D.; Kainerstorfer, J.M.; Chernomordik, V.; Najafizadeh, L.; Wassermann, E.M.; Gandjbakhche, A.H. Normative database of judgment of complexity task with functional near infrared spectroscopy – Application for TBI. NeuroImage 2012, 60, 879–883. [Google Scholar] [CrossRef]
- Karamzadeh, N.; Amyot, F.; Kenney, K.; Anderson, A.; Chowdhry, F.; Dashtestani, H.; Wassermann, E.M.; Chernomordik, V.; Boccara, C.; Wegman, E.; et al. A machine learning approach to identify functional biomarkers in human prefrontal cortex for individuals with traumatic brain injury using functional near-infrared spectroscopy. Brain Behav. 2016, 6, e00541. [Google Scholar] [CrossRef] [PubMed]
- Izzetoglu, M.; Bunce, S.C.; Izzetoglu, K.; Onaral, B.; Pourrezaei, A.K. Functional brain imaging using near-infrared technology. IEEE Eng. Med. Boil. Mag. 2007, 26, 38–46. [Google Scholar] [CrossRef]
- Strangman, G.; Boas, D.A.; Sutton, J.P. Non-invasive neuroimaging using near-infrared light. Boil. Psychiatry 2002, 52, 679–693. [Google Scholar] [CrossRef]
- Anderson, A.A.; Smith, E.; Chowdhry, F.A.; Thurm, A.; Condy, E.; Swineford, L.; Manwaring, S.S.; Amyot, F.; Matthews, D.; Gandjbakhche, A.H. Prefrontal Hemodynamics in Toddlers at Rest: A Pilot Study of Developmental Variability. Front. Mol. Neurosci. 2017, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Sassaroli, A.; Frederick, B.D.; Tong, Y.; Renshaw, P.F.; Fantini, S. Spatially weighted BOLD signal for comparison of functional magnetic resonance imaging and near-infrared imaging of the brain. NeuroImage 2006, 33, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Huppert, T.J.; Hoge, R.D.; Diamond, S.G.; Franceschini, M.A.; Boas, D.A. A temporal comparison of BOLD, ASL, and NIRS hemodynamic responses to motor stimuli in adult humans. Neuroimage 2006, 29, 368–382. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Bray, S.; Bryant, D.M.; Glover, G.H.; Reiss, A.L. A quantitative comparison of NIRS and fMRI across multiple cognitive tasks. Neuroimage 2011, 54, 2808–2821. [Google Scholar] [CrossRef]
- Sato, H.; Yahata, N.; Funane, T.; Takizawa, R.; Katura, T.; Atsumori, H.; Nishimura, Y.; Kinoshita, A.; Kiguchi, M.; Koizumi, H.; et al. A NIRS—fMRI investigation of prefrontal cortex activity during a working memory task. NeuroImage 2013, 83, 158–173. [Google Scholar] [CrossRef]
- Pinti, P.; Tachtsidis, I.; Hamilton, A.; Hirsch, J.; Aichelburg, C.; Gilbert, S.; Burgess, P.W.; Pinti, P. The present and future use of functional near-infrared spectroscopy (fNIRS) for cognitive neuroscience. Ann. New York Acad. Sci. 2018. [Google Scholar] [CrossRef]
- Wilcox, T.; Biondi, M. fNIRS in the developmental sciences. Wiley Interdiscip. Rev. Cogn. Sci. 2015, 6, 263–283. [Google Scholar] [CrossRef]
- Cutini, S.; Moro, S.B.; Bisconti, S. Functional near Infrared Optical Imaging in Cognitive Neuroscience: An Introductory Review. J. Near Infrared Spectrosc. 2012, 20, 75–92. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Thiers, A.; Hamacher, D.; Schega, L. Functional near-infrared spectroscopy in movement science: A systematic review on cortical activity in postural and walking tasks. Neurophotonics 2017, 4, 41403. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, L.; Yücel, M.A.; Dehaes, M.; Cooper, R.J.; Perdue, K.L.; Selb, J.; Huppert, T.J.; Hoge, R.D.; Boas, D.A. Quantification of the cortical contribution to the NIRS signal over the motor cortex using concurrent NIRS-fMRI measurements. Neuroimage 2012, 59, 3933–3940. [Google Scholar] [CrossRef] [PubMed]
- Lara, A.H.; Wallis, J.D. The Role of Prefrontal Cortex in Working Memory: A Mini Review. Front. Syst. Neurosci. 2015, 9, 829. [Google Scholar] [CrossRef] [PubMed]
- Barbey, A.K.; Koenigs, M.; Grafman, J. Dorsolateral prefrontal contributions to human working memory. Cortex 2013, 49, 1195–1205. [Google Scholar] [CrossRef] [PubMed]
- Owen, A.M.; Iddon, J.L.; Hodges, J.R.; Summers, B.A.; Robbins, T.W. Spatial and non-spatial working memory at different stages of Parkinson’s disease. Neuropsychologia 1997, 35, 519–532. [Google Scholar] [CrossRef]
- Euston, D.R.; Gruber, A.J.; McNaughton, B.L. The Role of Medial Prefrontal Cortex in Memory and Decision Making. Neuron 2012, 76, 1057–1070. [Google Scholar] [CrossRef]
- Rushworth, M.F.; Noonan, M.P.; Boorman, E.D.; Walton, M.E.; Behrens, T.E. Frontal Cortex and Reward-Guided Learning and Decision-Making. Neuron 2011, 70, 1054–1069. [Google Scholar] [CrossRef]
- Smith, R.; Lane, R.D.; Alkozei, A.; Bao, J.; Smith, C.; Sanova, A.; Nettles, M.; Killgore, W.D.S. The role of medial prefrontal cortex in the working memory maintenance of one’s own emotional responses. Sci. Rep. 2018, 8, 3460. [Google Scholar] [CrossRef]
- Wang, S.; Gwizdka, J.; Chaovalitwongse, W.A. Using wireless EEG signals to assess memory workload in the nn-back task. IEEE Trans. Hum. Mach. Syst. 2015, 46, 424–435. [Google Scholar] [CrossRef]
- Fishburn, F.A.; Norr, M.E.; Medvedev, A.V.; Vaidya, C.J. Sensitivity of fNIRS to cognitive state and load. Front. Hum. Neurosci. 2014, 8, 76. [Google Scholar] [CrossRef] [PubMed]
- Morrison, B.A.; Chein, J.M. Does working memory training work? The promise and challenges of enhancing cognition by training working memory. Psychon. Bull. Rev. 2011, 18, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Buschkuehl, M.; Hernandez-Garcia, L.; Jaeggi, S.M.; Bernard, J.A.; Jonides, J. Neural Effects of Short-Term Training on Working Memory. Cogn. Affect. Behav. Neurosci. 2014, 14, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, S.; Grahn, J.; Hampshire, A.; Mobbs, D.; Dalgleish, T. Training the Emotional Brain: Improving Affective Control through Emotional Working Memory Training. J. Neurosci. 2013, 33, 5301–5311. [Google Scholar] [CrossRef] [PubMed]
- Chein, J.M.; Schneider, W. Neuroimaging studies of practice-related change: fMRI and meta-analytic evidence of a domain-general control network for learning. Cogn. Brain Res. 2005, 25, 607–623. [Google Scholar] [CrossRef] [PubMed]
- Thompson, T.W.; Waskom, M.L.; Gabrieli, J.D.E. Intensive Working Memory Training Produces Functional Changes in Large-Scale Fronto-Parietal Networks. J. Cogn. Neurosci. 2016, 28, 575–588. [Google Scholar] [CrossRef] [PubMed]
- Au, J.; Sheehan, E.; Tsai, N.; Duncan, G.J.; Buschkuehl, M.; Jaeggi, S.M. Improving fluid intelligence with training on working memory: A meta-analysis. Psychon. Bull. Rev. 2015, 22, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Herff, C.; Heger, D.; Fortmann, O.; Hennrich, J.; Putze, F.; Schultz, T. Mental workload during n-back task—Quantified in the prefrontal cortex using fNIRS. Front. Hum. Neurosci. 2014, 7, 935. [Google Scholar] [CrossRef] [PubMed]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Müller, N. Applications of Functional Near-Infrared Spectroscopy (fNIRS) Neuroimaging in Exercise–Cognition Science: A Systematic, Methodology-Focused Review. J. Clin. Med. 2018, 7, 466. [Google Scholar] [CrossRef]
- Kuruvilla, M.S.; Green, J.R.; Ayaz, H.; Murman, D.L. Neural correlates of cognitive decline in ALS: An fNIRS study of the prefrontal cortex. Cogn. Neurosci. 2013, 4, 115–121. [Google Scholar] [CrossRef]
- Ayaz, H.; Shewokis, P.A.; Curtin, A.; Izzetoglu, M.; Izzetoglu, K.; Onaral, B. Using MazeSuite and functional near infrared spectroscopy to study learning in spatial navigation. JoVE 2011, 56, e3443. [Google Scholar] [CrossRef] [PubMed]
- Dashtestani, H.; Zaragoza, R.; Kermanian, R.; Knutson, K.M.; Halem, M.; Casey, A.; Karamzadeh, N.S.; Anderson, A.A.; Boccara, A.C.; Gandjbakhche, A. The role of prefrontal cortex in a moral judgment task using functional near-infrared spectroscopy. Brain Behav. 2018, 8, e01116. [Google Scholar] [CrossRef] [PubMed]
- Dashtestani, H.; Zaragoza, R.; Pirsiavash, H.; Knutson, K.M.; Kermanian, R.; Cui, J.; Harrison, J.D.; Halem, M.; Gandjbakhche, A. Canonical correlation analysis of brain prefrontal activity measured by functional near infra-red spectroscopy (fNIRS) during a moral judgment task. Behav. Brain Res. 2019, 359, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Dashtestani, H.; Zaragoza, R.; Kermanian, R.; Condy, E.; Anderson, A.; Chowdhry, F.; Karamzadeh, N.S.; Miguel, H.; Aram, S.; Gandjbakhche, A. The Quest for Functional Biomarkers in the Prefrontal Cortex Using Functional Near-Infrared Spectroscopy (fNIRS). Neurophotonics Biomed. Spectrosc. 2019, 123–136. [Google Scholar]
- Sassaroli, A.; Fantini, S. Comment on the modified Beer-Lambert law for scattering media. Phys. Med. Boil. 2004, 49, 255. [Google Scholar] [CrossRef]
- Pierro, M.L.; Sassaroli, A.; Bergethon, P.R.; Ehrenberg, B.L.; Fantini, S. Phase-amplitude investigation of spontaneous low-frequency oscillations of cerebral hemodynamics with near-infrared spectroscopy: A sleep study in human subjects. NeuroImage 2012, 63, 1571–1584. [Google Scholar] [CrossRef] [PubMed]
- Brigadoi, S.; Cooper, R.J. How short is short? Optimum source–detector distance for short-separation channels in functional near-infrared spectroscopy. Neurophotonics 2015, 2, 25005. [Google Scholar] [CrossRef]
- Funane, T.; Atsumori, H.; Katura, T.; Obata, A.N.; Sato, H.; Tanikawa, Y.; Okada, E.; Kiguchi, M. Quantitative evaluation of deep and shallow tissue layers’ contribution to fNIRS signal using multi-distance optodes and independent component analysis. NeuroImage 2014, 85, 150–165. [Google Scholar] [CrossRef]
- Mancini, D.M.; Bolinger, L.; Li, H.; Kendrick, K.; Chance, B.; Wilson, J.R. Validation of near-infrared spectroscopy in humans. J. Appl. Physiol. 1994, 77, 2740–2747. [Google Scholar] [CrossRef]
- Olsen, W. Triangulation in social research: Qualitative and quantitative methods can really be mixed. Dev. Sociol. 2004, 20, 103–118. [Google Scholar]
- Miller, E.K.; Cohen, J.D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 2001, 24, 167–202. [Google Scholar] [CrossRef]
- Brigadoi, S.; Ceccherini, L.; Cutini, S.; Scarpa, F.; Scatturin Pi Selb, J.; Gagnon, L.; Boas, D.A.; Cooper, R.J. Motion artifacts in functional near-infrared spectroscopy: A comparison of motion correction techniques applied to real cognitive data. NeuroImage 2014, 85, 181–191. [Google Scholar] [CrossRef]
- Di Lorenzo, R.; Pirazzoli, L.; Blasi, A.; Bulgarelli, C.; Hakuno, Y.; Minagawa, Y.; Brigadoi, S. Recommendations for motion correction of infant fNIRS data applicable to data sets acquired with a variety of experimental designs and acquisition systems. NeuroImage 2019, 200, 511–527. [Google Scholar] [CrossRef]
- Fantini, S.; Frederick, B.; Sassaroli, A. Perspective: Prospects of non-invasive sensing of the human brain with diffuse optical imaging. APL Photon 2018, 3, 110901. [Google Scholar] [CrossRef]
- Metz, A.J.; Klein, S.D.; Scholkmann, F.; Wolf, U. Continuous coloured light altered human brain haemodynamics and oxygenation assessed by systemic physiology augmented functional near-infrared spectroscopy. Sci. Rep. 2017, 7, 10027. [Google Scholar] [CrossRef]
- Pinti, P.; Ceccherini, L.; Cutini, S.; Scarpa, F.; Scatturin Pi Selb, J.; Gagnon, L.; Boas, D.A.; Cooper, R.J. Current status and issues regarding pre-processing of fNIRS neuroimaging data: An investigation of diverse signal filtering methods within a General Linear Model framework. Front. Hum. Neurosci. 2018, 12, 505. [Google Scholar] [CrossRef]
- Scholkmann, F.; Wolf, M. General equation for the differential pathlength factor of the frontal human head depending on wavelength and age. J. Biomed. Opt. 2013, 18, 105004. [Google Scholar] [CrossRef]
- Scholkmann, F.; Hafner, T.; Metz, A.J.; Wolf, M.; Wolf, U. Effect of short-term colored-light exposure on cerebral hemodynamics and oxygenation, and systemic physiological activity. Neurophotonics 2017, 4, 1. [Google Scholar] [CrossRef]
- Tachtsidis, I.; Scholkmann, F. False positives and false negatives in functional near-infrared spectroscopy: Issues, challenges, and the way forward. Neurophotonics 2016, 3, 30401. [Google Scholar] [CrossRef]
- Fairclough, S.H.; Venables, L.; Tattersall, A. The influence of task demand and learning on the psychophysiological response. Int. J. Psychophysiol. 2005, 56, 171–184. [Google Scholar] [CrossRef]
- Helton, W.S.; Russell, P.N. Working memory load and the vigilance decrement. Exp. Brain Res. 2011, 212, 429–437. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khaksari, K.; Condy, E.; Millerhagen, J.B.; Anderson, A.A.; Dashtestani, H.; Gandjbakhche, A.H. Effects of Performance and Task Duration on Mental Workload during Working Memory Task. Photonics 2019, 6, 94. https://doi.org/10.3390/photonics6030094
Khaksari K, Condy E, Millerhagen JB, Anderson AA, Dashtestani H, Gandjbakhche AH. Effects of Performance and Task Duration on Mental Workload during Working Memory Task. Photonics. 2019; 6(3):94. https://doi.org/10.3390/photonics6030094
Chicago/Turabian StyleKhaksari, Kosar, Emma Condy, John B. Millerhagen, Afrouz A. Anderson, Hadis Dashtestani, and Amir H. Gandjbakhche. 2019. "Effects of Performance and Task Duration on Mental Workload during Working Memory Task" Photonics 6, no. 3: 94. https://doi.org/10.3390/photonics6030094
APA StyleKhaksari, K., Condy, E., Millerhagen, J. B., Anderson, A. A., Dashtestani, H., & Gandjbakhche, A. H. (2019). Effects of Performance and Task Duration on Mental Workload during Working Memory Task. Photonics, 6(3), 94. https://doi.org/10.3390/photonics6030094




