“Green” Fluorescent–Plasmonic Carbon-Based Nanocomposites with Controlled Performance for Mild Laser Hyperthermia
Abstract
:1. Introduction
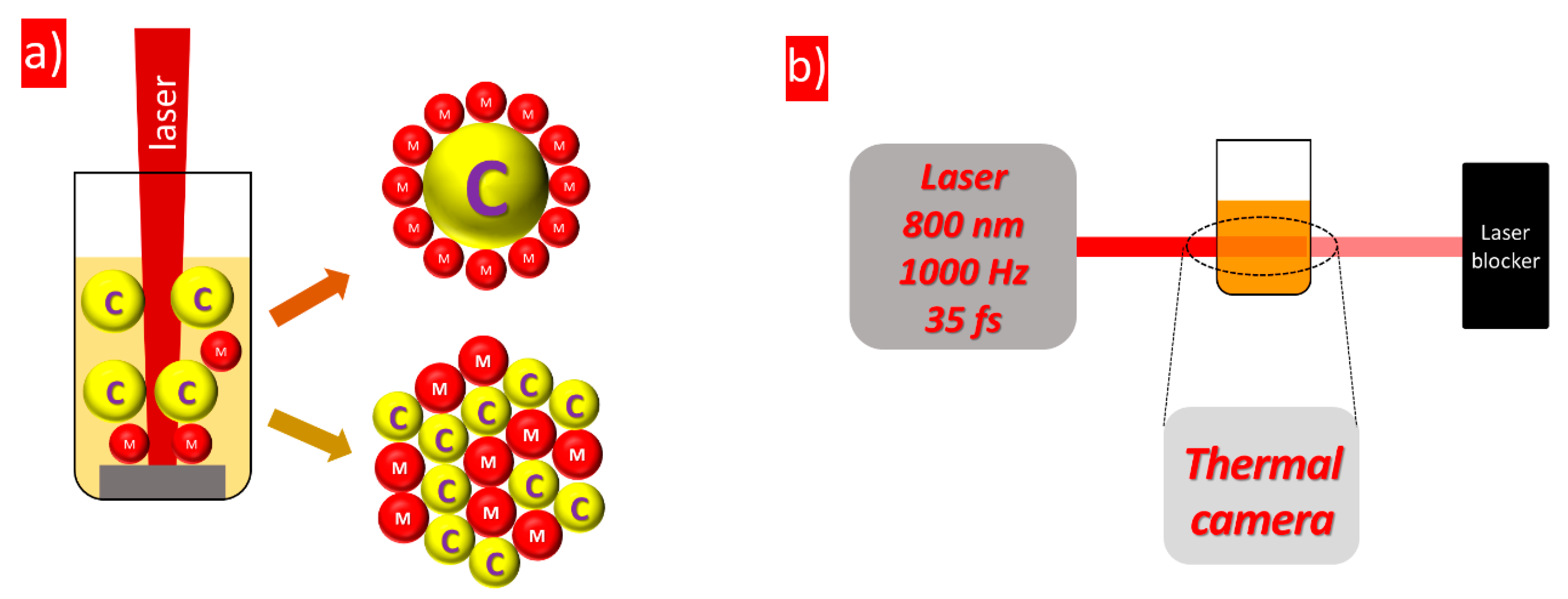
2. Experimental Section/Methods
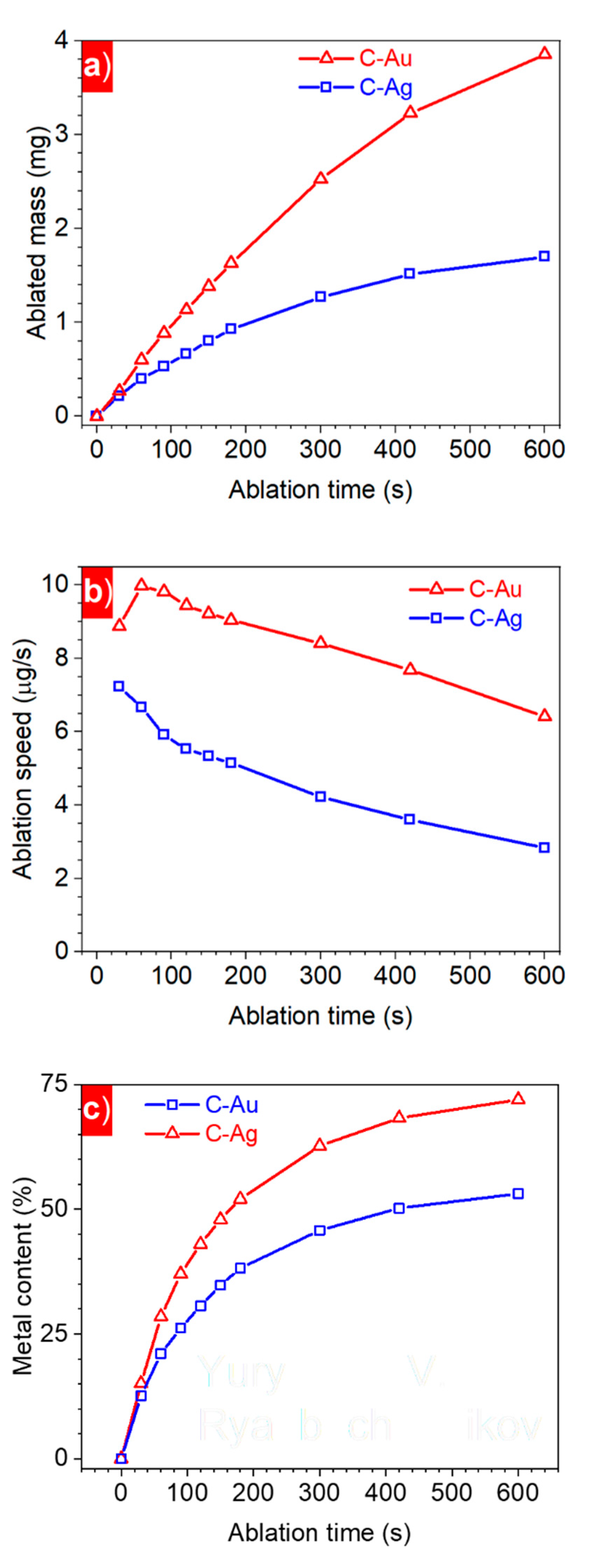
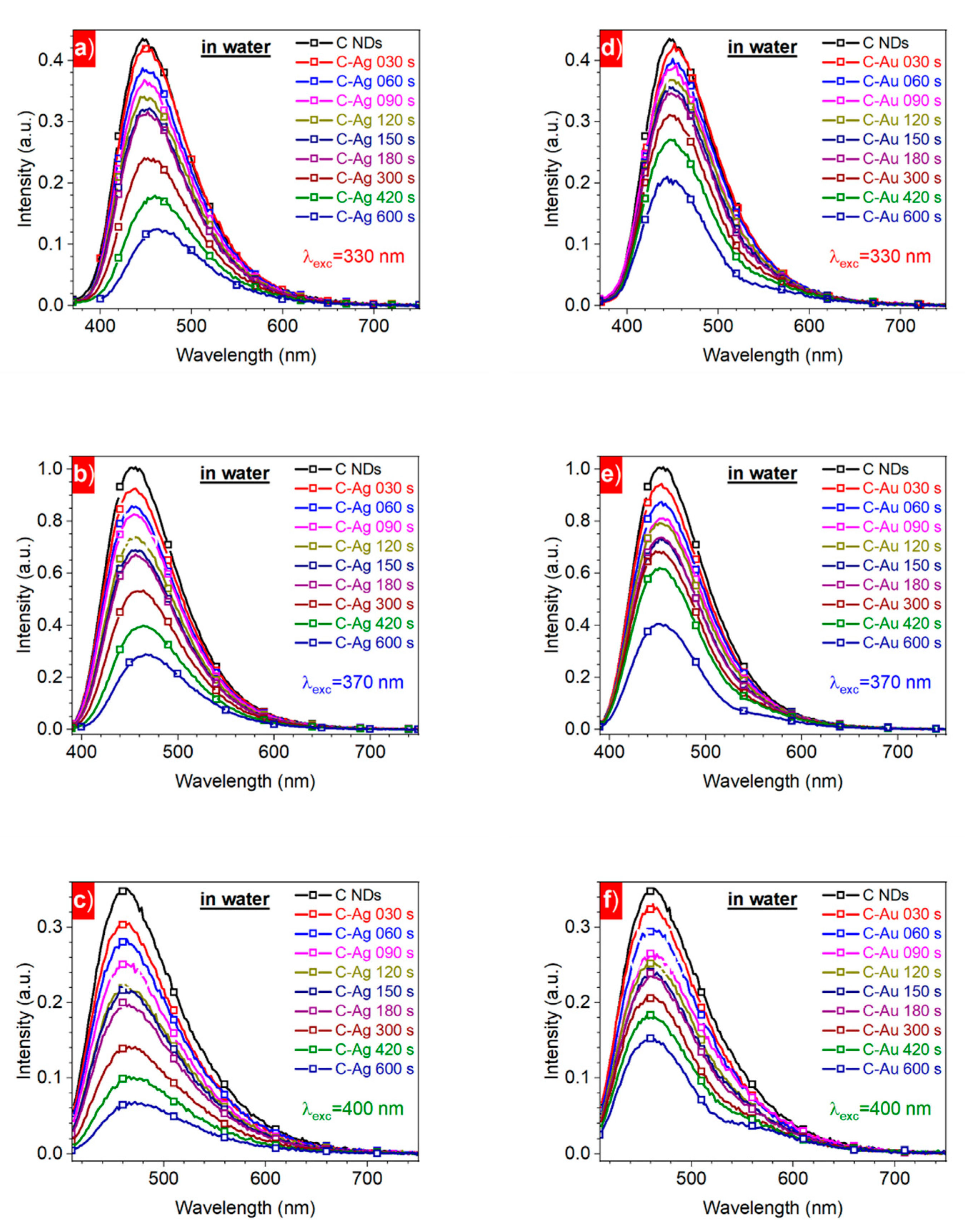
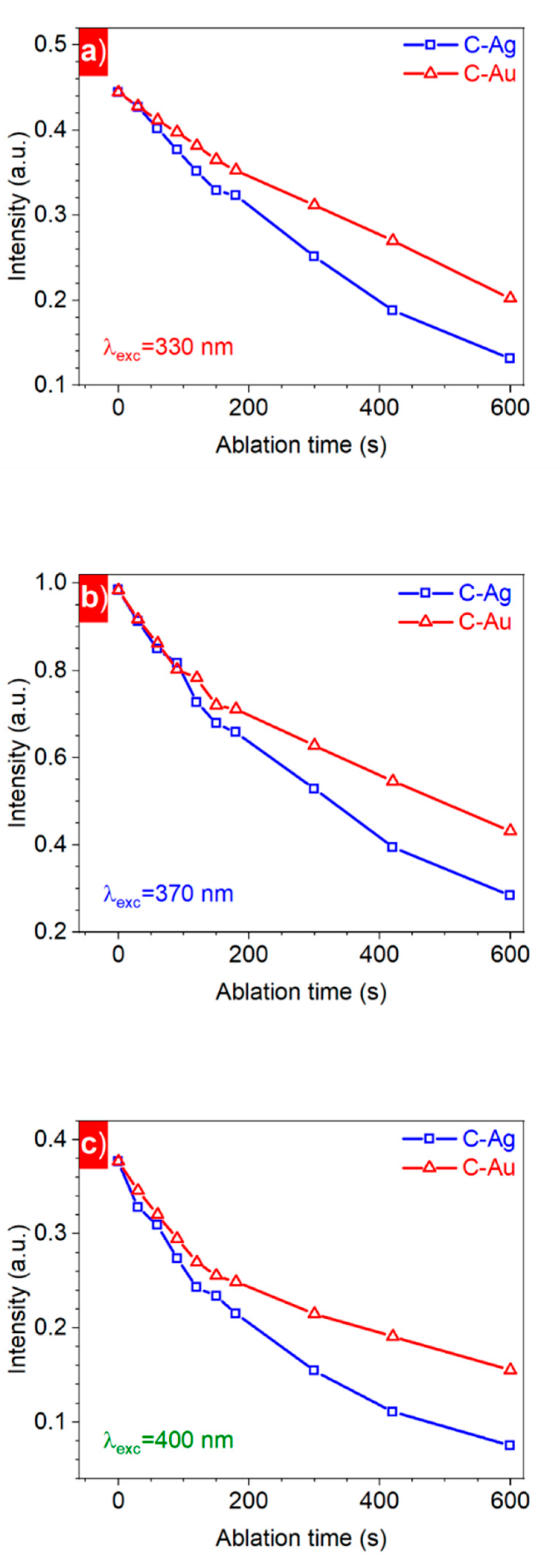
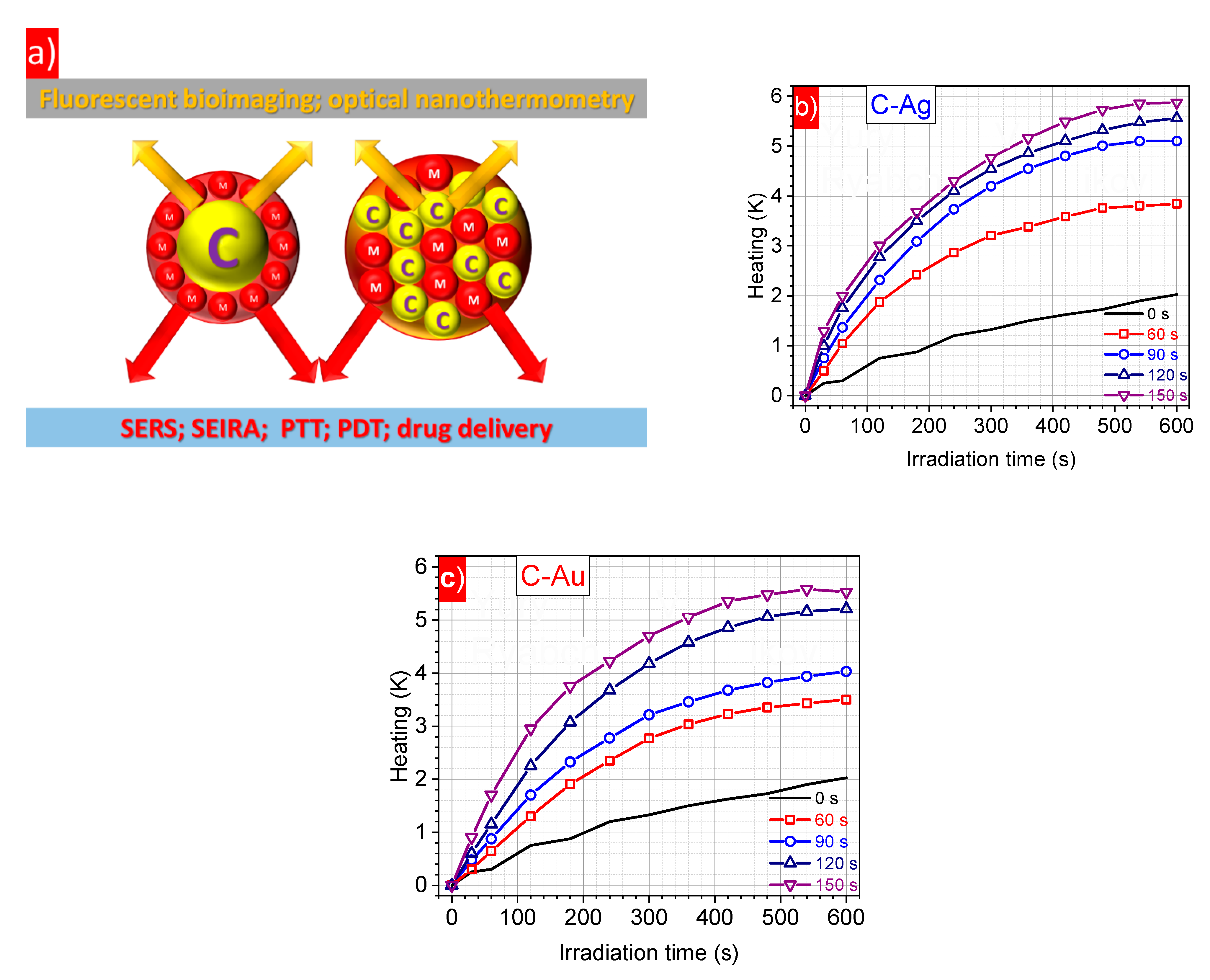
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Naghdi, M.; Ghovvati, M.; Rabiee, N.; Ahmadi, S.; Abbariki, N.; Sojdeh, S.; Ojaghi, A.; Bagherzadeh, M.; Akhavan, O.; Sharifi, E.; et al. Magnetic nanocomposites for biomedical applications. Adv. Colloid Interface 2022, 308, 102771. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, P.; Li, Y.; Hou, Y.; Yin, C.; Wang, Z.; Liao, Z.; Fu, X.; Li, M.; Fan, C.; et al. Light-Activated Gold–Selenium Core–Shell Nanocomposites with NIR-II Photoacoustic Imaging Performances for Heart-Targeted Repair. ACS Nano 2022, 16, 18667–18681. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, S.; Shahid, S.; Ashiq, K.; Alwadai, N.; Javed, M.; Iqbal, S.; Fatima, U.; Zaman, S.; Sarwar, M.N.; Alshammari, F.H.; et al. Controlled growth of nanocomposite thin layer based on Zn-Doped MgO nanoparticles through Sol-Gel technique for biosensor applications. Inorg. Chem. Commun. 2022, 142, 109702. [Google Scholar] [CrossRef]
- Ge, X.; Wong, R.; Anisa, A.; Ma, S. Recent development of metal-organic framework nanocomposites for biomedical applications. Biomaterials 2022, 281, 121322. [Google Scholar] [CrossRef] [PubMed]
- Morone, M.V.; Dell’Annunziata, F.; Giugliano, R.; Chianese, A.; De Filippis, A.; Rinaldi, L.; Gambardella, U.; Franci, G.; Galdiero, M.; Morone, A. Pulsed laser ablation of magnetic nanoparticles as a novel antibacterial strategy against gram positive bacteria. Appl. Surf. Sci. Adv. 2022, 7, 100213. [Google Scholar] [CrossRef]
- Kucherik, A.O.; Ryabchikov, Y.V.; Kutrovskaya, S.V.; Al-Kattan, A.; Arakelyan, S.M.; Itina, T.E.; Kabashin, A.V. Cavitation-Free Continuous-Wave Laser Ablation from a Solid Target to Synthesize Low-Size-Dispersed Gold Nanoparticles. ChemPhysChem 2017, 18, 1185–1191. [Google Scholar] [CrossRef]
- Bucharskaya, A.B.; Khlebtsov, N.G.; Khlebtsov, B.N.; Maslyakova, G.N.; Maslyakova, G.N.; Navolokin, N.A.; Genin, V.D.; Genina, E.A.; Tuchin, V.V. Photothermal and Photodynamic Therapy of Tumors with Plasmonic Nanoparticles: Challenges and Prospects. Materials 2022, 15, 1606. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V. Size Modification of Optically Active Contamination-Free Silicon Nanoparticles with Paramagnetic Defects by Their Fast Synthesis and Dissolution. Phys. Status Solidi A 2019, 216, A1800685. [Google Scholar] [CrossRef]
- Aikens, C.M.; Alloyeau, D.; Amendola, V.; Amiens, C.; Andreazza, P.; Bakker, J.M.; Baletto, F.; Barcikowski, S.; Barrabés, N.; Bowker, M.; et al. Nanoalloy magnetic and optical properties, applications and structures: General discussion. Faraday Discuss. 2023, 242, 389–417. [Google Scholar] [CrossRef]
- Basagni, A.; Torresan, V.; Marzola, P.; Fernandez van Raap, M.B.; Nodari, L.; Amendola, V. Structural evolution under physical and chemical stimuli of metastable Au–Fe nanoalloys obtained by laser ablation in liquid. Faraday Discuss. 2023, 242, 286–300. [Google Scholar] [CrossRef]
- Torresan, V.; Forrer, D.; Guadagnini, A.; Badocco, D.; Pastore, P.; Casarin, M.; Selloni, A.; Coral, D.; Ceolin, M.; Fernández van Raap, M.B.; et al. 4D Multimodal Nanomedicines Made of Nonequilibrium Au–Fe Alloy Nanoparticles. ACS Nano 2020, 14, 12840–12853. [Google Scholar] [CrossRef] [PubMed]
- Johny, J.; Prymak, O.; Kamp, M.; Calvo, F.; Kim, S.H.; Tymoczko, A.; El-Zoka, A.; Rehbock, C.; Schürmann, U.; Gault, B.; et al. Multidimensional thermally-induced transformation of nest-structured complex Au-Fe nanoalloys towards equilibrium. Nano Res. 2022, 15, 581–592. [Google Scholar] [CrossRef]
- Gurbatov, S.O.; Puzikov, V.; Storozhenko, D.; Modin, E.; Mitsai, E.; Cherepakhin, A.; Shevlyagin, A.; Gerasimenko, A.V.; Kulinich, S.A.; Kuchmizhak, A.A. Multigram-Scale Production of Hybrid Au-Si Nanomaterial by Laser Ablation in Liquid (LAL) for Temperature-Feedback Optical Nanosensing, Light-to-Heat Conversion, and Anticounterfeit Labeling. ACS Appl. Mater. Interfaces 2023, 5, 3336–3347. [Google Scholar] [CrossRef] [PubMed]
- Gurbatov, S.; Puzikov, V.; Modin, E.; Shevlyagin, A.; Gerasimenko, A.; Mitsai, E.; Kulinich, S.A.; Kuchmizhak, A. Ag-Decorated Si Microspheres Produced by Laser Ablation in Liquid: All-in-One Temperature-Feedback SERS-Based Platform for Nanosensing. Materials 2022, 15, 8091. [Google Scholar] [CrossRef]
- Elsayed, K.A.; Alomari, M.; Drmosh, Q.A.; Manda, A.A.; Haladu, S.A.; Alade, I.O. Anticancer Activity of TiO2/Au Nanocomposite Prepared by Laser Ablation Technique on Breast and Cervical Cancers. Opt. Laser Technol. 2022, 149, 107828. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V. Facile Laser Synthesis of Multimodal Composite Silicon/Gold Nanoparticles with Variable Chemical Composition. J. Nanopart. Res. 2019, 21, 85. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Behrends, J. Expedient Paramagnetic Properties of Surfactant-Free Plasmonic Silicon-Based Nanoparticles. Opt. Quantum Electron. 2020, 52, 177. [Google Scholar] [CrossRef]
- Saraeva, I.N.; Luong, N.V.; Kudryashov, S.I.; Rudenko, A.A.; Khmelnitskiy, R.A.; Shakhmin, A.L.; Kharin, A.Y.; Ionin, A.A.; Zayarny, D.A.; Tung, D.H.; et al. Laser synthesis of colloidal Si@Au and Si@Ag nanoparticles in water via plasma-assisted reduction. J. Photochem. Photobiol. A 2018, 360, 125–131. [Google Scholar] [CrossRef]
- Kutrovskaya, S.; Arakelian, S.; Kucherik, A.; Osipov, A.; Evlyukhin, A.; Kavokin, A.V. The Synthesis of Hybrid Gold-Silicon Nano Particles in a Liquid. Sci. Rep. 2017, 7, 10284. [Google Scholar] [CrossRef]
- Parola, S.; Julián-López, B.; Carlos, L.D.; Sanchez, C. Optical Properties of Hybrid Organic-Inorganic Materials and their Applications. Adv. Funct. Mater. 2016, 26, 6506–6544. [Google Scholar] [CrossRef]
- Li, X.; Zhang, F.; Zhao, D. Lab on upconversion nanoparticles: Optical properties and applications engineering via designed nanostructure. Chem. Soc. Rev. 2015, 44, 1346–1378. [Google Scholar] [CrossRef] [PubMed]
- Belogorokhov, I.A.; Ryabchikov, Y.V.; Tikhonov, E.V.; Pushkarev, V.E.; Breusova, M.O.; Tomilova, L.G.; Khokhlov, D.R. Photoluminescence in Semiconductor Structures Based on Butyl–Substituted Erbium Phtalocyanine Complexes. Semiconductors 2008, 42, 321–324. [Google Scholar] [CrossRef]
- Konstantinova, E.A.; Demin, V.A.; Vorontzov, A.S.; Ryabchikov, Y.V.; Belogorokhov, I.A.; Osminkina, L.A.; Forsh, P.A.; Kashkarov, P.K.; Timoshenko, V.Y. Electron Paramagnetic Resonance and Photoluminescence Study of Si Nanocrystals—Photosensitizers of Singlet Oxygen Molecules. J. Non-Cryst. Solids 2006, 352, 1156–1159. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Vorontsov, A.S.; Osminkina, L.A.; Timoshenko, V.Y.; Kashkarov, P.K. Dependence of the Singlet Oxygen Photosensitization Efficiency on Morphology of Porous Silicon. Phys. Status Solidi A 2007, 204, 1271–1275. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Gongalskiy, M.B.; Osminkina, L.A.; Timoshenko, V.Y. Photosensitized Generation of Singlet Oxygen in Powders and Aqueous Suspensions of Silicon Nanocrystals. Semiconductors 2011, 45, 1059–1063. [Google Scholar] [CrossRef]
- Zabotnov, S.; Golovan, L.; Kurakina, D.; Khilov, A.; Sokolovskaya, O.; Skobelkina, A.; Kashaev, F.; Kashkarov, P.; Sergeeva, E.; Kirillin, M. Designing Silicon Nanoparticles for Optical Bioimaging. FiO+LS 2020, paper FTh2D.2. [Google Scholar] [CrossRef]
- Zabotnov, S.V.; Skobelkina, A.V.; Sergeeva, E.A.; Kurakina, D.A.; Khilov, A.V.; Kashaev, F.V.; Kaminskaya, T.P.; Presnov, D.E.; Agrba, P.D.; Shuleiko, D.V.; et al. Nanoparticles Produced via Laser Ablation of Porous Silicon and Silicon Nanowires for Optical Bioimaging. Sensors 2020, 20, 4874. [Google Scholar] [CrossRef]
- Zabotnov, S.V.; Kashaev, F.V.; Shuleiko, D.V.; Gongalsky, M.B.; Golovan, L.A.; Kashkarov, P.K.; Loginova, D.A.; Agrba, P.D.; Sergeeva, E.A.; Kirillin, M.Y. Silicon nanoparticles as contrast agents in the methods of optical biomedical diagnostics. Quantum Electron. 2017, 47, 638. [Google Scholar] [CrossRef]
- Kirillin, M.Y.; Sergeeva, E.A.; Agrba, P.D.; Krainov, A.D.; Ezhov, A.A.; Shuleiko, D.V.; Kashkarov, P.K.; Zabotnov, S.V. Laser-ablated silicon nanoparticles: Optical properties and perspectives in optical coherence tomography. Laser Phys. 2015, 25, 075604. [Google Scholar] [CrossRef]
- Konstantinova, E.A.; Ryabchikov, Y.V.; Osminkina, L.A.; Vorontsov, A.S.; Kashkarov, P.K. Effect of Adsorption of the Donor and Acceptor Molecules at the Surface of Porous Silicon on the Recombination Properties of Silicon Nanocrystals. Semiconductors 2004, 38, 1344–1349. [Google Scholar] [CrossRef]
- Lai, W.C.; Chakravarty, S.; Zou, Y.; Chen, R.T. Silicon nano-membrane based photonic crystal microcavities for high sensitivity bio-sensing. Opt. Lett. 2012, 37, 1208–1210. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Alekseev, S.A.; Lysenko, V.; Bremond, G.; Bluet, J.M. Photoluminescence thermometry with alkyl–terminated silicon nanoparticles dispersed in low–polar liquids. Phys. Status Solidi RRL 2013, 7, 414–417. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Lysenko, V.; Nychyporuk, T.J. Enhanced Thermal Sensitivity of Silicon Nanoparticles Embedded in (nano–Ag)/SiNx for Luminescent Thermometry. Phys. Chem. C 2014, 118, 12515–12519. [Google Scholar] [CrossRef]
- Gurbatov, S.O.; Puzikov, V.; Cherepakhin, A.; Mitsai, E.; Tarasenka, N.; Shevlyagin, A.; Sergeev, A.; Kulinich, S.A.; Kuchmizhak, A.A. Hybrid Au@Si microspheres produced via laser irradiation in liquid for nonlinear photonics. Opt. Laser Technol. 2022, 147, 107666. [Google Scholar] [CrossRef]
- Ming, T.; Dietzek-Ivanšić, B.; Lu, X.; Zuo, X.; Sivakov, V. Silicon Nanowires Decorated with Silver Nanoparticles for Photoassisted Hydrogen Generation. ACS Appl. Energy Mater. 2022, 5, 7466. [Google Scholar] [CrossRef]
- Ming, T.; Turishchev, S.; Schleusener, A.; Parinova, E.; Koyuda, D.; Chuvenkova, O.; Schulz, M.; Dietzek, B.; Sivakov, V. Silicon Suboxides as Driving Force for Efficient Light-Enhanced Hydrogen Generation on Silicon Nanowires. Small 2021, 17, 2007650. [Google Scholar] [CrossRef]
- Mussabek, G.; Alekseev, S.A.; Manilov, A.I.; Tutashkonko, S.; Nychyporuk, T.; Shabdan, Y.; Amirkhanova, G.; Litvinenko, S.V.; Skryshevsky, V.A.; Lysenko, V. Kinetics of Hydrogen Generation from Oxidation of Hydrogenated Silicon Nanocrystals in Aqueous Solutions. Nanomaterials 2020, 10, 1413. [Google Scholar] [CrossRef]
- Hu, J.; Lu, Q.; Wu, C.; Liu, M.; Li, H.; Zhang, Y.; Yao, S. Synthesis of Fluorescent and Water-Dispersed Germanium Nanoparticles and Their Cellular Imaging Applications. Langmuir 2018, 34, 8932–8938. [Google Scholar] [CrossRef]
- Mederos, M.; Mestanza, S.N.M.; Lang, R.; Doi, I.; Diniz, J.A. Germanium nanoparticles grown at different deposition times for memory device applications. Thin Solid Films 2016, 611, 39–45. [Google Scholar] [CrossRef]
- Vaughn, D.D.; Schaak, R.E. Synthesis, properties and applications of colloidal germanium and germanium-based nanomaterials. Chem. Soc. Rev. 2013, 42, 2861–2879. [Google Scholar] [CrossRef]
- Baker, S.N.; Baker, G.A. Luminescent Carbon Nanodots: Emergent Nanolights. Angew. Chem. Int. Ed. 2010, 49, 6726–6744. [Google Scholar] [CrossRef]
- Vasin, A.; Kysil, D.; Rusavsky, A.; Isaieva, O.; Zaderko, A.; Nazarov, A.; Lysenko, V. Synthesis and Luminescent Properties of Carbon Nanodots Dispersed in Nanostructured Silicas. Nanomaterials 2012, 11, 3267. [Google Scholar] [CrossRef]
- Kasouni, A.; Chatzimitakos, T.; Stalikas, C. Bioimaging Applications of Carbon Nanodots: A Review. C 2019, 5, 19. [Google Scholar] [CrossRef]
- Lopes, J.L.; Martins, M.J.; Nogueira, H.I.S.; Estrada, A.C.; Trindade, T. Carbon-based heterogeneous photocatalysts for water cleaning technologies: A review. Environ. Chem. Lett. 2021, 19, 643–668. [Google Scholar] [CrossRef]
- Dubyk, K.; Borisova, T.; Paliienko, K.; Krisanova, N.; Isaiev, M.; Alekseev, S.; Skryshevsky, V.; Lysenko, V.; Geloen, A. Bio-distribution of Carbon Nanoparticles Studied by Photoacoustic Measurements. Nanoscale Res. Lett. 2022, 17, 127. [Google Scholar] [CrossRef]
- Liang, S.X.; Zhang, L.C.; Reichenberger, S.; Barcikowski, S. Design and perspective of amorphous metal nanoparticles from laser synthesis and processing. Phys. Chem. Chem. Phys. 2021, 23, 11121–11154. [Google Scholar] [CrossRef]
- Zhang, D.; Gökce, B.; Barcikowski, S. Laser Synthesis and Processing of Colloids: Fundamentals and Applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef]
- Zhang, J.; Chaker, M.; Ma, D.J. Pulsed laser ablation based synthesis of colloidal metal nanoparticles for catalytic applications. Colloid Interf. Sci. 2017, 489, 138–149. [Google Scholar] [CrossRef]
- Dittrich, S.; Streubel, R.; McDonnell, C.; Huber, H.P.; Barcikowski, S.; Gökce, B. Comparison of the productivity and ablation efficiency of different laser classes for laser ablation of gold in water and air. Appl. Phys. A 2019, 125, 432. [Google Scholar] [CrossRef]
- Saraeva, I.N.; Kudryashov, S.I.; Lednev, V.N.; Makarov, S.V.; Pershin, S.M.; Rudenko, A.A.; Zayarny, D.A.; Ionin, A.A. Single- and multishot femtosecond laser ablation of silicon and silver in air and liquid environments: Plume dynamics and surface modification. Appl. Surf. Sci. 2019, 476, 576–586. [Google Scholar] [CrossRef]
- Petrikaite, V.; Skapas, M.; Stankevičius, E. Generation of gold and silver nanoparticles using laser ablation of thin bimetallic films and bulk targets in water. Opt. Mater. 2023, 137, 113535. [Google Scholar] [CrossRef]
- Singh, M.R.; Guo, J.; Fanizza, E.; Dubey, M.J. Anomalous Photoluminescence Quenching in Metallic Nanohybrids. Phys. Chem. C 2019, 123, 10013–10020. [Google Scholar] [CrossRef]
- Asberg, P.; Nilsson, P.; Inganäs, O.J. Fluorescence quenching and excitation transfer between semiconducting and metallic organic layers. Appl. Phys. 2004, 96, 3140–3147. [Google Scholar] [CrossRef]
- Andsager, D.; Hilliard, J.; Hetrick, J.M.; AbuHassan, L.H.; Plisch, M.; Nayfeh, M.H. Quenching of porous silicon photoluminescence by deposition of metal adsorbates. J. Appl. Phys. 1993, 74, 4783–4785. [Google Scholar] [CrossRef]
- Kavalaraki, A.; Spyratou, E.; Kouri, M.A.; Efstathopoulos, E.P. Gold Nanoparticles as Contrast Agents in Ophthalmic Imaging. Optics 2023, 4, 74–99. [Google Scholar] [CrossRef]
- Alavi, M. Bacteria and fungi as major bio-sources to fabricate silver nanoparticles with antibacterial activities. Expert Rev. Anti-Infect. Ther. 2022, 20, 897–906. [Google Scholar] [CrossRef]
- Zagaynova, E.V.; Shirmanova, M.V.; Kirillin, M.Y.; Khlebtsov, B.N.; Orlova, A.G.; Balalaeva, I.V.; Sirotkina, M.A.; Bugrova, M.L.; Agrba, P.D.; Kamensky, V.A. Contrasting properties of gold nanoparticles for optical coherence tomography: Phantom, in vivo studies and Monte Carlo simulation. Phys. Med. Biol. 2008, 53, 4995. [Google Scholar] [CrossRef]
- Mirsaeidi, A.M.; Yousefi, F.J. Viscosity, thermal conductivity and density of carbon quantum dots nanofluids: An experimental investigation and development of new correlation function and ANN modeling. Therm. Anal. Calorim. 2021, 143, 351–361. [Google Scholar] [CrossRef]
- van Rhoon, G.C.; Franckena, M.; ten Hagen, T.L.M. A moderate thermal dose is sufficient for effective free and TSL based thermochemotherapy. Adv. Drug Deliv. Rev. 2020, 163–164, 145–156. [Google Scholar] [CrossRef]
- Roti Roti, J.L. Cellular responses to hyperthermia (40–46 °C): Cell killing and molecular events. Int. J. Hyperth. 2008, 24, 3–15. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryabchikov, Y.V.; Zaderko, A. “Green” Fluorescent–Plasmonic Carbon-Based Nanocomposites with Controlled Performance for Mild Laser Hyperthermia. Photonics 2023, 10, 1229. https://doi.org/10.3390/photonics10111229
Ryabchikov YV, Zaderko A. “Green” Fluorescent–Plasmonic Carbon-Based Nanocomposites with Controlled Performance for Mild Laser Hyperthermia. Photonics. 2023; 10(11):1229. https://doi.org/10.3390/photonics10111229
Chicago/Turabian StyleRyabchikov, Yury V., and Alexander Zaderko. 2023. "“Green” Fluorescent–Plasmonic Carbon-Based Nanocomposites with Controlled Performance for Mild Laser Hyperthermia" Photonics 10, no. 11: 1229. https://doi.org/10.3390/photonics10111229
APA StyleRyabchikov, Y. V., & Zaderko, A. (2023). “Green” Fluorescent–Plasmonic Carbon-Based Nanocomposites with Controlled Performance for Mild Laser Hyperthermia. Photonics, 10(11), 1229. https://doi.org/10.3390/photonics10111229





