Removal Performance and Mechanism of Iron–Phosphorus-Based Composite Biochar for Pb(II) and Sb(III) from Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of Modified Biochar
2.3. Characterization and Analysis
2.4. Adsorption Experiment
3. Results
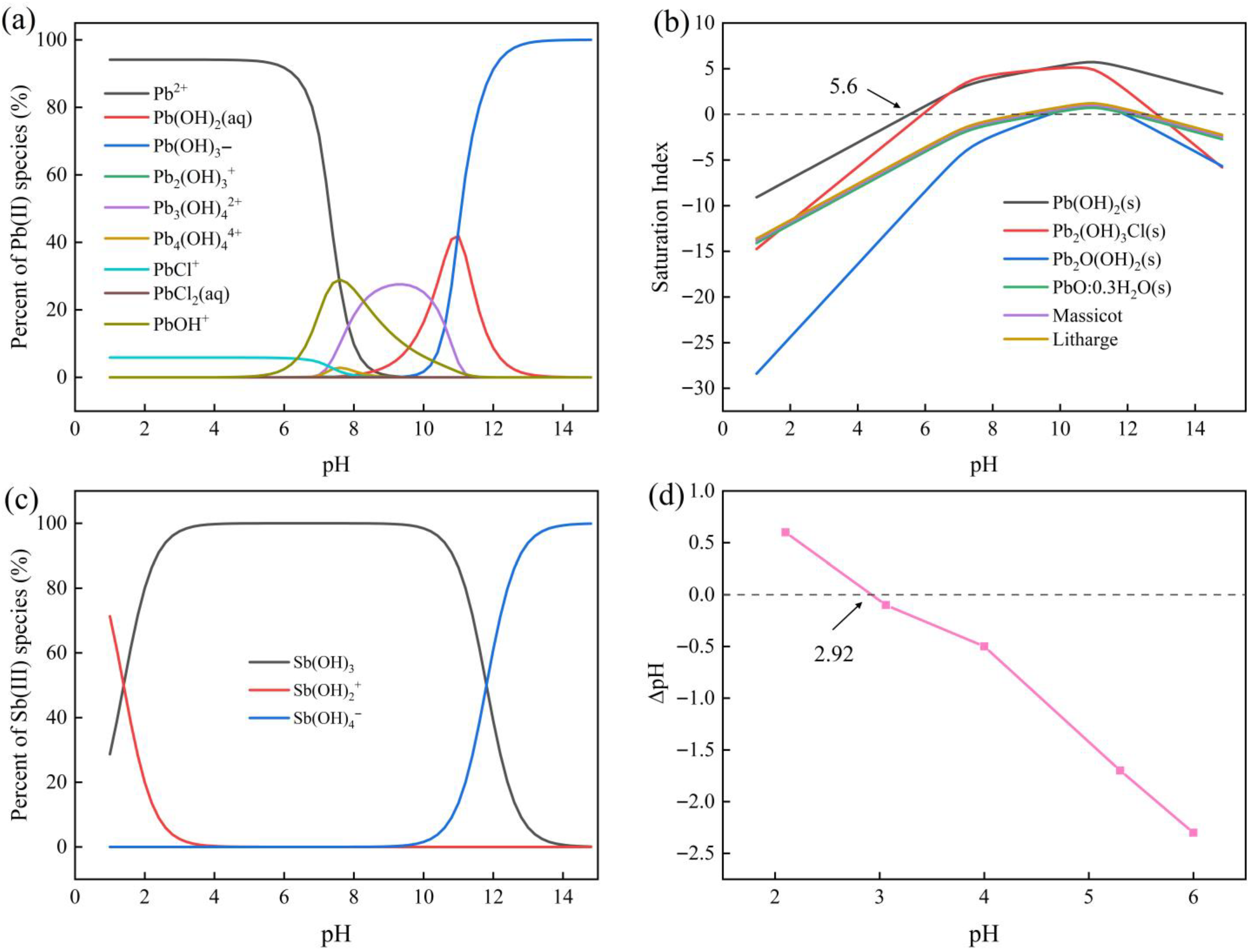
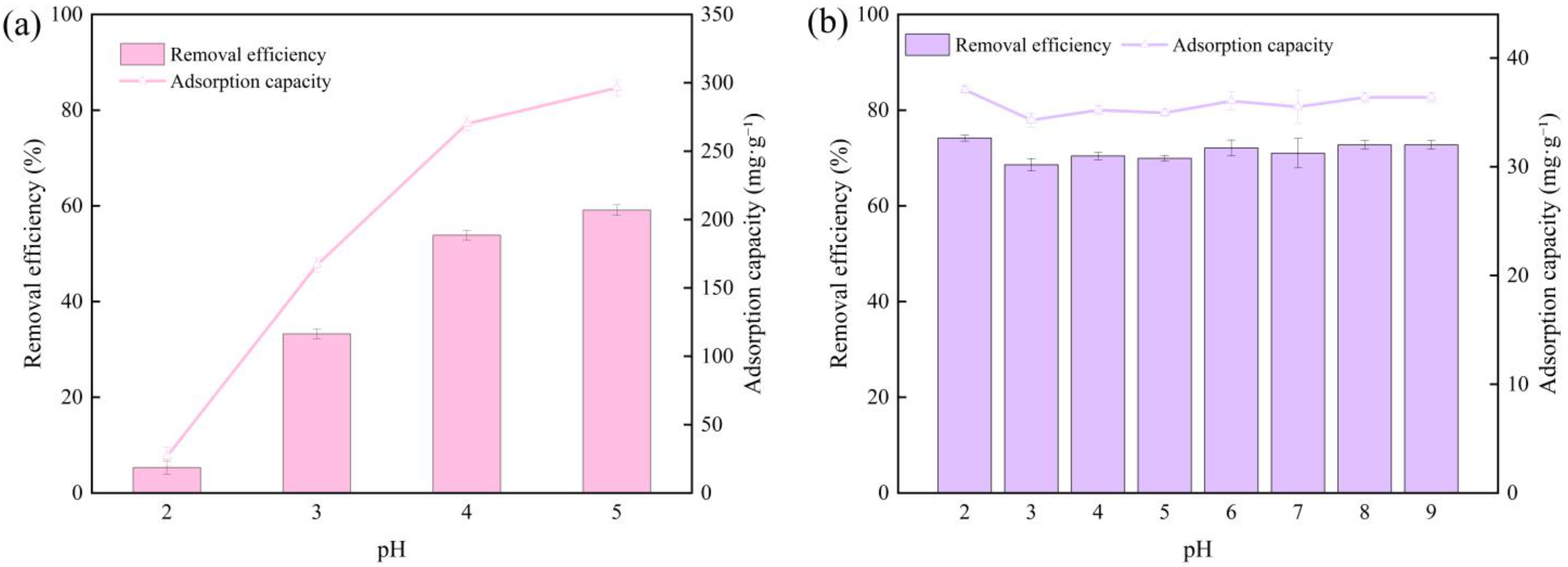
3.1. Effect of Solution pH on Adsorption
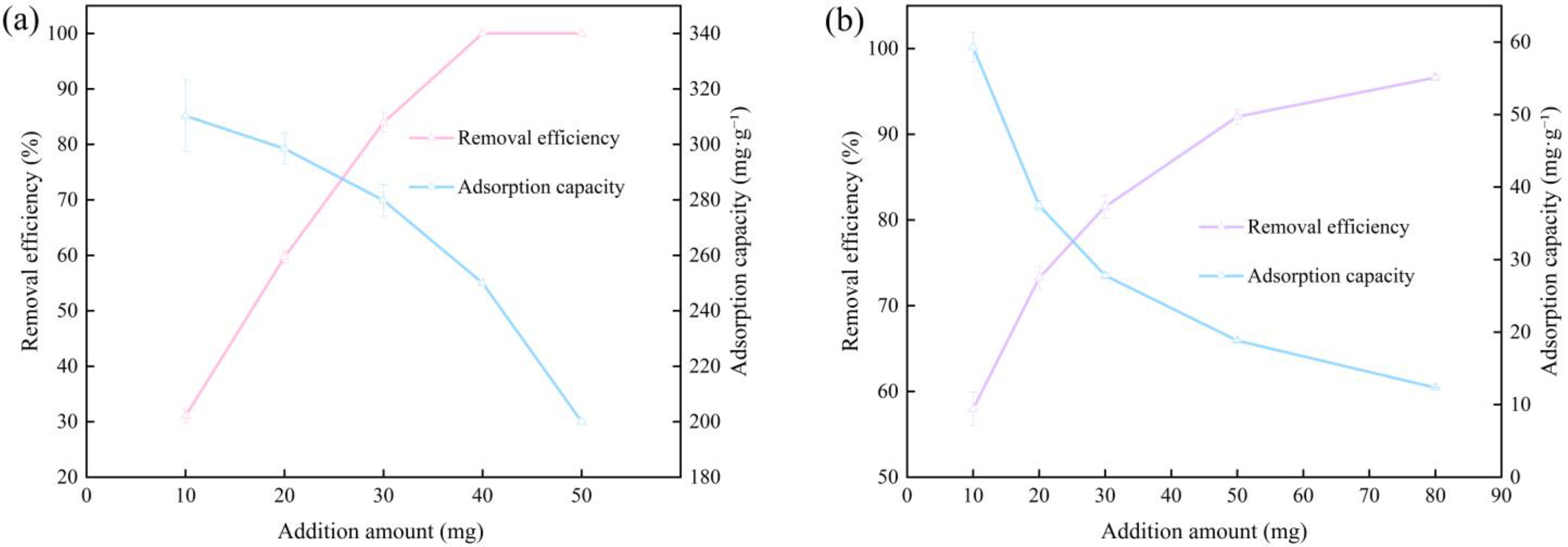
3.2. Effect of Dosage of FPBC on Adsorption
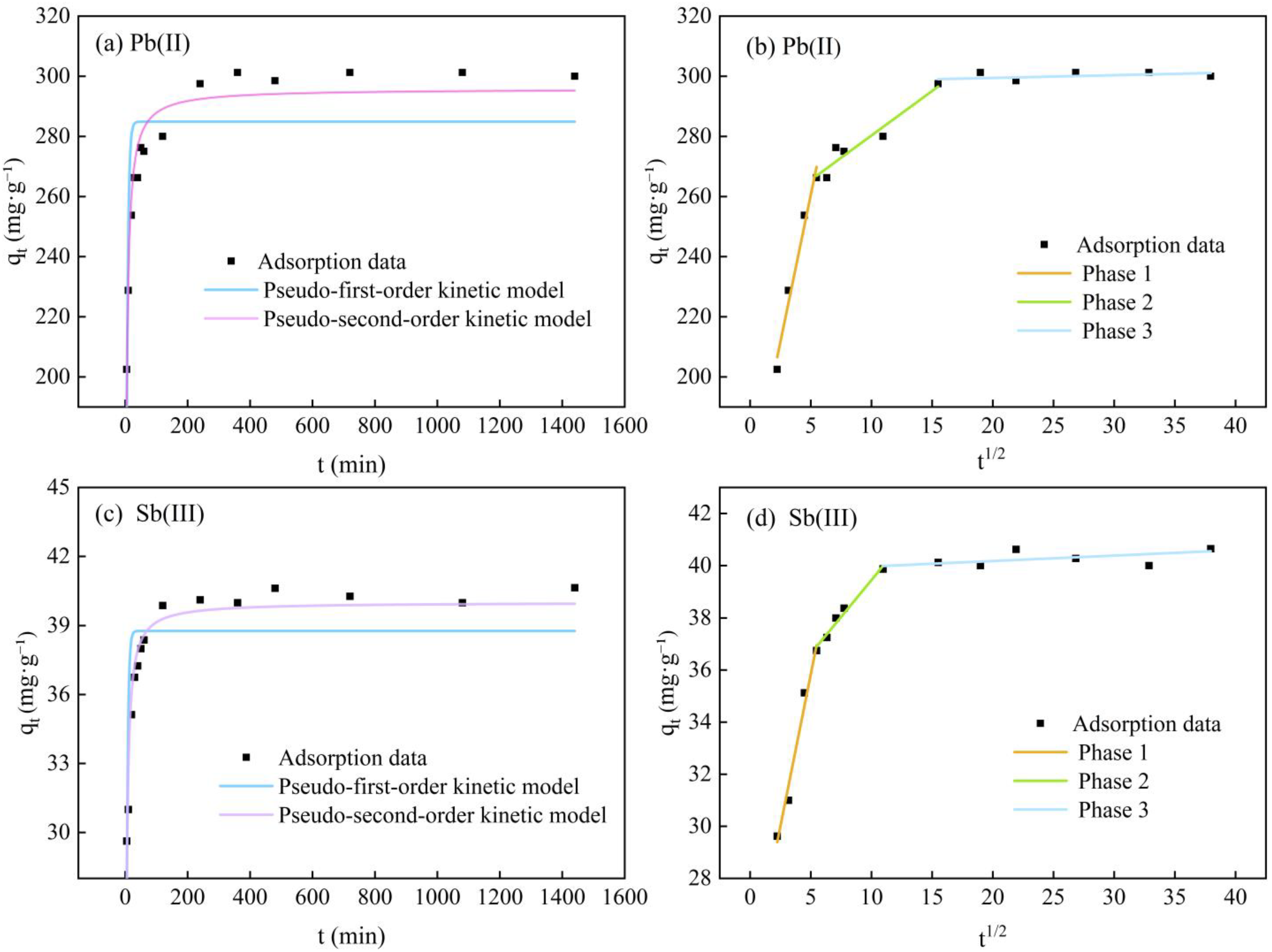
3.3. Adsorption Kinetic
3.4. Adsorption Isotherm
3.5. Thermodynamics Analysis
3.6. Investigation of Adsorption Performance in Binary Systems of Heavy Metals
3.7. Comparison with Other Reported Materials
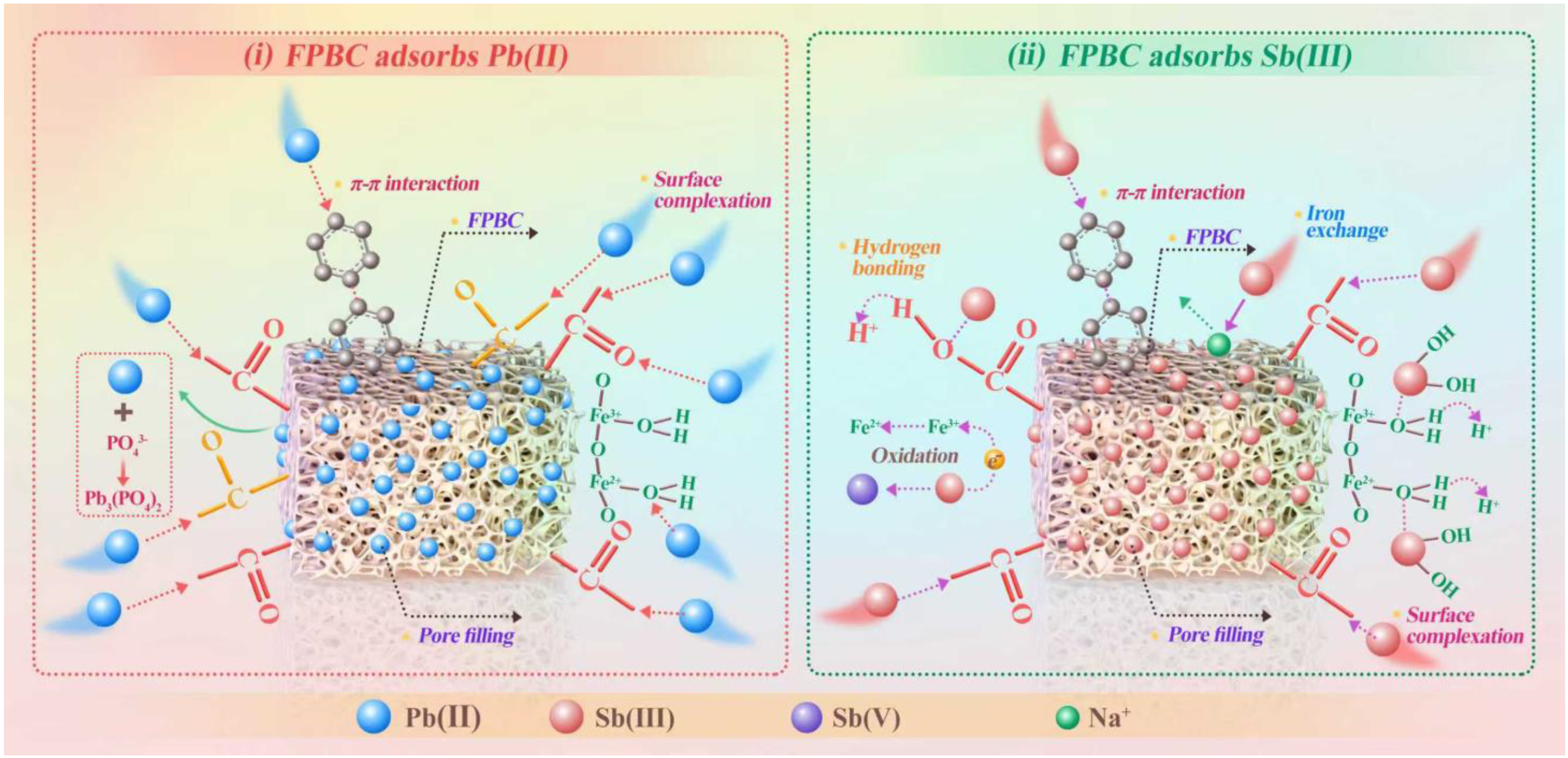
3.8. Adsorption Mechanism
3.8.1. SEM-EDS
3.8.2. BET
3.8.3. XRD
3.8.4. FTIR
3.8.5. XPS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ettler, V.; Tejnecký, V.; Mihaljevič, M.; Šebek, O.; Zuna, M.; Vaněk, A. Antimony mobility in lead smelter-polluted soils. Geoderma 2009, 155, 409–418. [Google Scholar] [CrossRef]
- Sun, X.; Sun, M.; Chao, Y.; Shang, X.; Wang, H.; Pan, H.; Yang, Q.; Lou, Y.; Zhuge, Y. Effects of lead pollution on soil microbial community diversity and biomass and on invertase activity. Soil Ecol. Lett. 2022, 5, 118–127. [Google Scholar] [CrossRef]
- Gupta, M.; Dwivedi, V.; Kumar, S.; Patel, A.; Niazi, P.; Yadav, V.K. Lead toxicity in plants: Mechanistic insights into toxicity, physiological responses of plants and mitigation strategies. Plant Signal. Behav. 2024, 19, 2365576. [Google Scholar] [CrossRef]
- Dragana, J.; Katarina, B.; Đurđica, M.; Stefan, M.; Danijela, Đ.; Zorica, B.; Buha, D.A. Exploring the endocrine disrupting potential of lead through benchmark modelling—Study in humans. Environ. Pollut. 2022, 316, 120428. [Google Scholar] [CrossRef]
- Nahid, K.; Nahid, A.; Sadegh, H.M.; Mansour, S.; Kazem, N.; Vahide, O. Blood lead level monitoring related to environmental exposure in the general Iranian population: A systematic review and meta-analysis. Environ. Sci. Pollut. Res. Int. 2021, 28, 32210–32223. [Google Scholar] [CrossRef]
- Lianhua, L.; Xavier, S.T.R.; Mengchang, H.; Wei, O.; Qingwen, Z.; Miguel, H.Y.C.; Cristhian, C.O. Antimony pollution threatens soils and riverine habitats across China: An analysis of antimony concentrations, changes, and risks. Crit. Rev. Environ. Sci. Technol. 2024, 54, 797–816. [Google Scholar] [CrossRef]
- Zheng, J.; Ohata, M.; Furuta, N. Studies on the speciation of inorganic and organic antimony compounds in airborne particulate matter by HPLC-ICP-MS. Analyst 2000, 125, 1025–1028. [Google Scholar] [CrossRef]
- Zubair, M.; Ihsanullah, I.; Aziz, H.A.; Ahmad, M.A.; Al-Harthi, M.A. Sustainable wastewater treatment by biochar/layered double hydroxide composites: Progress, challenges, and outlook. Bioresour. Technol. 2020, 319, 124128. [Google Scholar] [CrossRef]
- Meng, K.; Dong, Y.; Liu, J.; Xie, J.; Jin, Q.; Lu, Y.; Lin, H. Advances in selective heavy metal removal from water using biochar: A comprehensive review of mechanisms and modifications. J. Environ. Chem. Eng. 2025, 13, 116099. [Google Scholar] [CrossRef]
- Yang, Z.; Liang, L.; Yang, W.; Shi, W.; Tong, Y.; Chai, L.; Gao, S.; Liao, Q. Simultaneous immobilization of cadmium and lead in contaminated soils by hybrid bio-nanocomposites of fungal hyphae and nano-hydroxyapatites. Environ. Sci. Pollut. Res. Int. 2018, 25, 11970–11980. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Q.; Yan, Y.; Jiang, X.; Shen, J. Development of nano-hydroxyapatite@biochar composite from low-cost precursors for enhanced immobilization of lead and cadmium. J. Hazard. Mater. 2025, 500, 140535. [Google Scholar] [CrossRef]
- Xu, W.; Wang, H.; Liu, R.; Zhao, X.; Qu, J. The mechanism of antimony(III) removal and its reactions on the surfaces of Fe–Mn Binary Oxide. J. Colloid Interface Sci. 2011, 363, 320–326. [Google Scholar] [CrossRef]
- Shan, C.; Ma, Z.; Tong, M. Efficient removal of trace antimony(III) through adsorption by hematite modified magnetic nanoparticles. J. Hazard. Mater. 2014, 268, 229–236. [Google Scholar] [CrossRef]
- Alessia, C.; Silvano, M.; Vincenzo, F. Removal of Lead, Copper, Zinc and Cadmium from Water Using Phosphate Rock. Acta Geol. Sin.-Engl. Ed. 2010, 82, 1223–1228. [Google Scholar] [CrossRef]
- Wang, C.; Zhou, Y.; Yu, F.; Zhu, X.; Dong, M.; Li, Q. Recovery of phosphate from aqueous solution by modified biochar with concentrated seawater and its potential application as fertilizer. J. Environ. Chem. Eng. 2024, 12, 112646. [Google Scholar] [CrossRef]
- Kang, D.H.P.; Chen, M.; Ogunseitan, O.A. Potential environmental and human health impacts of rechargeable lithium batteries in electronic waste. Environ. Sci. Technol. 2013, 47, 5495–5503. [Google Scholar] [CrossRef] [PubMed]
- Kallitsis, E.; Korre, A.; Kelsall, G.H. Life cycle assessment of recycling options for automotive Li-ion battery packs. J. Clean. Prod. 2022, 371, 133636. [Google Scholar] [CrossRef]
- Li, H.; Xing, S.; Liu, Y.; Li, F.; Guo, H.; Kuang, G. Recovery of Lithium, Iron, and Phosphorus from Spent LiFePO4 Batteries Using Stoichiometric Sulfuric Acid Leaching System. ACS Sustain. Chem. Eng. 2017, 5, 8017–8024. [Google Scholar] [CrossRef]
- Li, X.; Zhang, J.; Song, D.; Song, J.; Zhang, L. Direct regeneration of recycled cathode material mixture from scrapped LiFePO4 batteries. J. Power Sources 2017, 345, 78–84. [Google Scholar] [CrossRef]
- Bian, D.; Sun, Y.; Li, S.; Tian, Y.; Yang, Z.; Fan, X.; Zhang, W. A novel process to recycle spent LiFePO4 for synthesizing LiFePO4/C hierarchical microflowers. Electrochim. Acta 2016, 190, 134–140. [Google Scholar] [CrossRef]
- Xu, L.; Chen, C.; Huo, J.-B.; Chen, X.; Yang, J.-C.E.; Fu, M.-L. Iron hydroxyphosphate composites derived from waste lithium-ion batteries for lead adsorption and Fenton-like catalytic degradation of methylene blue. Environ. Technol. Innov. 2019, 16, 100504. [Google Scholar] [CrossRef]
- Wu, D.Y.; Pan, X.C.; Liu, Z.Q.; Rao, S.; Zhang, K.F. Study on Adsorption Performance of Spent Lithium Iron Phosphate Oxygen Pressure Leaching Residue for Heavy Metals. Nonferrous Met. (Extr. Metall.) 2021, 117–122. [Google Scholar]
- Tran, H.N.; Wang, Y.-F.; You, S.-J.; Chao, H.-P. Insights into the mechanism of cationic dye adsorption on activated charcoal: The importance of π–π interactions. Process Saf. Environ. Prot. 2017, 107, 168–180. [Google Scholar] [CrossRef]
- Kosmulski, M. Surface Charging and Points of Zero Charge; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Liu, L.; Yue, T.; Liu, R.; Lin, H.; Wang, D.; Li, B. Efficient absorptive removal of Cd(II) in aqueous solution by biochar derived from sewage sludge and calcium sulfate. Bioresour. Technol. 2021, 336, 125333. [Google Scholar] [CrossRef]
- Chen, H.; Gao, Y.; Fang, Z.; Li, J.; Pillai, S.C.; Song, H.; Sun, C.; Bolan, N.; Yang, X.; Vithanage, M.; et al. Investigating the electron-scale adsorption mechanisms using DFT calculations and experimental studies in self-assembly magnetic biochar gel incorporated with graphene nanosheets for enhanced Sb(III) removal. Chem. Eng. J. 2024, 487, 150740. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, J.; Liu, X.; Bai, S.; Lin, H.; Wang, D. Reduction and removal of As(V) in aqueous solution by biochar derived from nano zero-valent-iron (nZVI) and sewage sludge. Chemosphere 2021, 277, 130273. [Google Scholar] [CrossRef]
- Long, Y.; Jiang, J.; Hu, J.; Hu, X.; Yang, Q.; Zhou, S. Removal of Pb(II) from aqueous solution by hydroxyapatite/carbon composite: Preparation and adsorption behavior. Colloids Surf. A Physicochem. Eng. Asp. 2019, 577, 471–479. [Google Scholar] [CrossRef]
- Wei, S.; Tao, Y.; Ma, M.; Tong, W.; Bi, F.; Wang, L.; Qu, J.; Zhang, Y. One-step microwave-assisted synthesis of MgO-modified magnetic biochar for enhanced removal of lead and phosphate from wastewater: Performance and mechanisms. Sep. Purif. Technol. 2025, 354, 128936. [Google Scholar] [CrossRef]
- Apiratikul, R.; Pavasant, P. Sorption of Cu2+, Cd2+, and Pb2+ using modified zeolite from coal fly ash. Chem. Eng. J. 2008, 144, 245–258. [Google Scholar] [CrossRef]
- Park, J.-H.; Ok, Y.S.; Kim, S.-H.; Cho, J.-S.; Heo, J.-S.; Delaune, R.D.; Seo, D.-C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.B.; Ma, Y.B.; Chen, L.; Xian, K. Adsorption of aqueous Cd2+, Pb2+, Cu2+ ions by nano-hydroxyapatite: Single- and multi-metal competitive adsorption study. Geochem. J. 2010, 44, 233–239. [Google Scholar] [CrossRef]
- Zhang, Y.; Xia, M.; Wang, F.; Ma, J. Experimental and theoretical study on the adsorption mechanism of Amino trimethylphosphate (ATMP) functionalized hydroxyapatite on Pb(II) and Cd(II). Colloids Surf. A Physicochem. Eng. Asp. 2021, 626, 127029. [Google Scholar] [CrossRef]
- Chen, J.; Tang, C.; Li, X.; Sun, J.; Liu, Y.; Huang, W.; Wang, A.; Lu, Y. Preparation and Modification of Rape Straw Biochar and Its Adsorption Characteristics for Methylene Blue in Water. Water 2022, 14, 3761. [Google Scholar] [CrossRef]
- Yalasangi, V.; Mayilswamy, N.; Kandasubramanian, B. Biochar-derived adsorbents for removal of Rhodamine B from wastewater. Bioresour. Technol. Rep. 2024, 28, 101987. [Google Scholar] [CrossRef]
- Xianhong, D.; Wenbin, W.; Shuangqin, T.; Yi, H.; Shujie, W.; Bifang, Z.; Kai, X.; Ziqin, Z.; Lihong, T. Composite adsorbents of aminated chitosan @ZIF-8 MOF for simultaneous efficient removal of Cu(II) and Congo Red: Batch experiments and DFT calculations. Chem. Eng. J. 2024, 479, 147634. [Google Scholar] [CrossRef]
- Verma, M.; Borah, R.; Kumar, A.; Chae, S.-H.; Pan, S.-Y.; Kumar, V.; Vlaskin, M.S.; Kim, H. Capturing of inorganic and organic pollutants simultaneously from complex wastewater using recyclable magnetically chitosan functionalized with EDTA adsorbent. Process Saf. Environ. Prot. 2022, 167, 56–66. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, H.; Yang, J.; Zhang, L.; Cao, B.; Liu, L.; Gong, W. Removal of cadmium and lead from aqueous solutions using iron phosphate-modified pollen microspheres as adsorbents. Rev. Adv. Mater. Sci. 2021, 60, 365–376. [Google Scholar] [CrossRef]
- Tuyiringire, D.; Liu, X.; Zheng, Q.; Wang, S.; Zhang, W.; Bi, F.; Zhang, Y.; Wang, Y.; Qu, J.; Zhang, Y. Ball-milled phosphate/micro zero-valent iron/biochar for lead and cadmium removal and stabilization in water and soil: Performance, mechanisms, and environmental applications. Sep. Purif. Technol. 2025, 362, 131895. [Google Scholar] [CrossRef]
- Fan, G.; Xu, X.; Huang, Y.; Zhu, X.; Chen, X. The Sorption of Lead and Cadmium by Hydroxyapatite-Biochar Nanocomposite from Aqueous Solution. Int. J. Environ. Res. 2024, 19, 37. [Google Scholar] [CrossRef]
- Nan, L.; Zhang, Y.; Liu, M.; Zhao, L.; Zhu, Y.; Zhang, X. Application of Magnetic Aquatic Plant Biochar for Efficient Removal of Antimony from Water: Adsorption Properties and Mechanism. Separations 2024, 12, 2. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, X.; Ju, N.; Jia, H.; Sun, Z.; Liang, J.; Guo, R.; Niu, D.; Sun, H.-B. High capacity adsorption of antimony in biomass-based composite and its consequential utilization as battery anode. J. Environ. Sci. 2023, 126, 211–221. [Google Scholar] [CrossRef]
- Yang, B.; Fan, W.; Yanyan, G. Oxidation and adsorption of antimony(iii) from surface water using novel Al2O3-supported Fe-Mn binary oxide nanoparticles: Effectiveness, dynamic quantitative mechanisms, and life cycle analysis. Environ. Sci.-Nano 2020, 7, 3047–3061. [Google Scholar] [CrossRef]
- Teng, F.; Zhang, Y.; Wang, D.; Shen, M.; Hu, D. Iron-modified rice husk hydrochar and its immobilization effect for Pb and Sb in contaminated soil. J. Hazard. Mater. 2020, 398, 122977. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 2013, 57, 603–619. [Google Scholar] [CrossRef]
- Luo, F.; Feng, X.; Li, Y.; Zheng, G.; Zhou, A.; Xie, P.; Wang, Z.; Tao, T.; Long, X.; Wan, J. Magnetic amino-functionalized lanthanum metal-organic framework for selective phosphate removal from water. Colloids Surf. A Physicochem. Eng. Asp. 2021, 611, 125906. [Google Scholar] [CrossRef]
- Kabekkodu, S.N.; Dosen, A.; Blanton, T.N. PDF-5+: A comprehensive Powder Diffraction FileTM for materials characterization. Powder Diffr. 2024, 39, 47–59. [Google Scholar] [CrossRef]
- Hannan, F.; Huang, Q.; Farooq, M.A.; Ayyaz, A.; Ma, J.; Zhang, N.; Ali, B.; Deyett, E.; Zhou, W.; Islam, F. Organic and inorganic amendments for the remediation of nickel contaminated soil and its improvement on Brassica napus growth and oxidative defense. J. Hazard. Mater. 2021, 416, 125921. [Google Scholar] [CrossRef] [PubMed]
- Kwaśniak-Kominek, M.; Matusik, J.; Bajda, T.; Manecki, M.; Rakovan, J.; Marchlewski, T.; Szala, B. Fourier transform infrared spectroscopic study of hydroxylpyromorphite Pb10(PO4)6OH2–hydroxylmimetite Pb10(AsO4)6(OH)2 solid solution series. Polyhedron 2015, 99, 103–111. [Google Scholar] [CrossRef]
- Ain, Q.U.; Zhang, H.; Yaseen, M.; Rasheed, U.; Liu, K.; Subhan, S.; Tong, Z. Facile fabrication of hydroxyapatite-magnetite-bentonite composite for efficient adsorption of Pb(II), Cd(II), and crystal violet from aqueous solution. J. Clean. Prod. 2020, 247, 119088. [Google Scholar] [CrossRef]
- Cheng, C.; Zhang, J.; Mu, Y.; Gao, J.; Feng, Y.; Liu, H.; Guo, Z.; Zhang, C. Preparation and evaluation of activated carbon with different polycondensed phosphorus oxyacids (H3PO4, H4P2O7, H6P4O13 and C6H18O24P6) activation employing mushroom roots as precursor. J. Anal. Appl. Pyrolysis 2014, 108, 41–46. [Google Scholar] [CrossRef]
- Wang, Z.; Liu, G.; Zheng, H.; Li, F.; Ngo, H.H.; Guo, W.; Liu, C.; Chen, L.; Xing, B. Investigating the mechanisms of biochar’s removal of lead from solution. Bioresour. Technol. 2015, 177, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Zhang, W.; Yang, Y.; Huang, X.; Wang, S.; Qiu, R. Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar. Water Res. 2011, 46, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Ni, Q.; Lin, Q.; Khan, K.Y.; Li, T.; Khan, M.B.; He, Z.; Yang, X. Simultaneous sorption and catalytic oxidation of trivalent antimony by Canna indica derived biochars. Environ. Pollut. 2017, 229, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.L.; Shen, J.J.; Li, Z.C.; Lv, C.W.; He, J. Kinetics mechanisms of Sb(III)adsorbed by three synthesized Schwertmannites. Environ. Chem. 2024, 43, 4182–4194. [Google Scholar]
- Tice, L.; Hersey, J.; Karla, S.; Sorci, M.; Plawsky, J.; Giglia, S.; Belfort, G. Impact of pH, ionic strength, and operating conditions on capture mechanisms in nanoparticle filtration. J. Membr. Sci. 2024, 706, 122926. [Google Scholar] [CrossRef]
- Lin, J.-Y.; Kim, M.; Li, D.; Kim, H.; Huang, C.-P. The removal of phosphate by thermally treated red mud from water: The effect of surface chemistry on phosphate immobilization. Chemosphere 2020, 247, 125867. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, Y.; Zhou, J.; Guo, J.; Ren, J.; Zhou, F. Efficient removal of lead from aqueous solution by urea-functionalized magnetic biochar: Preparation, characterization and mechanism study. J. Taiwan Inst. Chem. Eng. 2018, 91, 457–467. [Google Scholar] [CrossRef]
- Zhang, M.; Song, L.; Jiang, H.; Li, S.; Shao, Y.; Yang, J.; Li, J. Biomass based hydrogel as an adsorbent for the fast removal of heavy metal ions from aqueous solutions. J. Mater. Chem. A 2017, 5, 3434–3446. [Google Scholar] [CrossRef]
- Zou, J.P.; Liu, H.L.; Luo, J.; Xing, Q.J.; Du, H.M.; Jiang, X.H.; Luo, X.B.; Luo, S.L.; Suib, S.L. Three-Dimensional Reduced Graphene Oxide Coupled with Mn3O4 for Highly Efficient Removal of Sb(III) and Sb(V) from Water. ACS Appl. Mater. Interfaces 2016, 8, 18140–18149. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, S.; Lv, J.; Nie, Y.; Shi, W. High-Efficiency Adsorption of Cadmium from Water and Soil by Iron and Phosphorus Co-Doped Biochar Modified Geopolymer Composite: Performance and Mechanisms. Water Air Soil Pollut. 2026, 237, 388. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, J.; Huang, X.; Wang, W.; Sun, P.; Li, Y.; Han, J. Functionalized biochar-supported magnetic MnFe2O4 nanocomposite for the removal of Pb(II) and Cd(II). RSC Adv. 2019, 9, 365–376. [Google Scholar] [CrossRef] [PubMed]
- HJ 776-2015; Water Quality—Determination of 32 Elements—Inductively Coupled Plasma Optical Emission Spectrometry. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2015.







| Elements | Fe | P | Li | Al | |
|---|---|---|---|---|---|
| Samples | |||||
| Leachate/mg | 882.81 | 364.06 | 101.25 | 33.75 | |
| Filtrate/mg | 2.42 | 88.21 | 99.75 | 0.14 | |
| Loading efficiency/% | 99.73 | 75.77 | 1.48 | 99.58 | |
| Model | Parameter | FPBC-Pb(II) | FPBC-Sb(III) |
|---|---|---|---|
| Pseudo-first-order kinetic model | qt (mg·g−1) | 284.89 | 38.77 |
| K1 (min−1) | 0.21 | 0.23 | |
| R2 | 0.65861 | 0.62260 | |
| Pseudo-second-order kinetic model | qt (mg·g−1) | 295.79 | 40.01 |
| K2 (g·mg−1·min−1) | 0.01 | 0.01 | |
| R2 | 0.93654 | 0.92507 |
| Phase | Parameter | FPBC-Pb(II) | FPBC-Sb(III) |
|---|---|---|---|
| Phase 1 | K1 (mg·g−1·min−0.5) | 19.54 | 2.33 |
| C1 | 162.85 | 24.17 | |
| RC1 | 54.06 | 60.41 | |
| R2 | 0.96317 | 0.97045 | |
| Phase 2 | K2 (mg·g−1·min−0.5) | 2.96 | 0.57 |
| C2 | 250.67 | 33.80 | |
| RC2 | 83.21 | 84.48 | |
| R2 | 0.91657 | 0.96984 | |
| Phase 3 | K3 (mg·g−1·min−0.5) | 0.09 | 0.02 |
| C3 | 297.64 | 39.76 | |
| RC3 | 98.80 | 99.38 | |
| R2 | 0.22766 | 0.22108 |
| Model | Parameter | FPBC-Pb(II) | ||
|---|---|---|---|---|
| 30 °C | 40 °C | 50 °C | ||
| Langmuir | Qmax (mg·g−1) | 312.54 | 321.22 | 326.28 |
| KL (L·mg−1) | 0.92 | 1.05 | 1.32 | |
| R2 | 0.97284 | 0.96184 | 0.93920 | |
| Freundlich | KF [(mg1−1/n·L1/n)·g−1] | 174.00 | 180.74 | 188.53 |
| 1/n | 0.13 | 0.12 | 0.12 | |
| R2 | 0.84145 | 0.83809 | 0.81631 | |
| D-R | Qmax (mg·g−1) | 300.78 | 310.12 | 318.19 |
| E (kJ·mol−1) | 1.54 | 1.71 | 1.96 | |
| R2 | 0.89981 | 0.9052 | 0.93196 | |
| Model | Parameter | FPBC-Sb(III) | ||
|---|---|---|---|---|
| 30 °C | 40 °C | 50 °C | ||
| Langmuir | Qmax (mg·g−1) | 219.20 | 224.18 | 236.61 |
| KL (L·mg−1) | 0.02 | 0.03 | 0.03 | |
| R2 | 0.96726 | 0.99236 | 0.99653 | |
| Freundlich | KF [(mg1−1/n·L1/n)·g−1] | 31.07 | 45.08 | 53.13 |
| 1/n | 0.32 | 0.27 | 0.24 | |
| R2 | 0.94635 | 0.92389 | 0.95877 | |
| Temkin | KT (L·mg−1) | 39.78 | 37.33 | 35.47 |
| b (J·mol−1) | 0.44 | 0.79 | 1.24 | |
| R2 | 0.95701 | 0.97035 | 0.93160 | |
| Samples | ΔH | ΔS | ΔG (kJ·mol−1) | ||
|---|---|---|---|---|---|
| (kJ·mol−1) | (kJ·mol−1·K−1) | 30 °C | 40 °C | 50 °C | |
| Pb(II) | 4.19 | 0.03 | −3.3 | −3.56 | −3.79 |
| Sb(III) | 28.03 | 0.10 | −3.55 | −4.38 | −5.63 |
| Adsorbents | Samples | pH | Equilibrium Time (h) | Qmax (mg·g−1) |
|---|---|---|---|---|
| Pollen@FePO4 [38] | Pb(II) | 5.92 | 0.5 | 61.35 |
| P-mZVIBC500 [39] | 7 | 1 | 243.57 | |
| HAP/BC [40] | 4.5 | 2 | 826.25 | |
| LMBC [41] | Sb(III) | 8 | 12 | 26.07 |
| Fe3O4-CS/EDTA [42] | 3 | 4–5 | 657.1 | |
| Fe–Mn@Al2O3 [43] | 6.4 | 4 | 272.2 | |
| HC12.5-180 [44] | Pb(II) | 6–7 | 12 | 64.69 |
| Sb(V) | 3–8 | 12 | 91.54 | |
| This work | Pb(II) | 5 | 4 | 312.54 |
| Sb(III) | 2 | 2 | 219.20 |
| Elements | FPBC | FPBC-Pb(II) | FPBC-Sb(III) | |||
|---|---|---|---|---|---|---|
| Atomic % | Weight % | Atomic % | Weight % | Atomic % | Weight % | |
| C | 41.68 | 26.57 | 46.97 | 16.29 | 42.03 | 20.59 |
| O | 39.67 | 33.68 | 31.66 | 14.63 | 35.76 | 23.34 |
| Na | 4.42 | 5.40 | 0.17 | 0.12 | 0.16 | 0.15 |
| P | 5.92 | 9.73 | 5.97 | 5.34 | 7.44 | 9.40 |
| Fe | 8.31 | 24.62 | 6.30 | 10.16 | 9.69 | 22.07 |
| Pb | — | — | 8.93 | 53.46 | — | — |
| Sb | — | — | — | — | 4.92 | 24.45 |
| Total | 100 | 100 | 100 | 100 | 100 | 100 |
| Materials | Special Surface Area | Total Pore Volume | Average Pore Diameter |
|---|---|---|---|
| m2·g−1 | cm3·g−1 | nm | |
| BC | 1219.73 | 0.99 | 6.69 |
| FPBC | 388.35 | 0.38 | 10.19 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ren, T.; Zhu, H.; Zhu, Z.; Tan, J.; Qin, Q. Removal Performance and Mechanism of Iron–Phosphorus-Based Composite Biochar for Pb(II) and Sb(III) from Water. Separations 2026, 13, 104. https://doi.org/10.3390/separations13040104
Ren T, Zhu H, Zhu Z, Tan J, Qin Q. Removal Performance and Mechanism of Iron–Phosphorus-Based Composite Biochar for Pb(II) and Sb(III) from Water. Separations. 2026; 13(4):104. https://doi.org/10.3390/separations13040104
Chicago/Turabian StyleRen, Tingting, Hongxiang Zhu, Zongqiang Zhu, Jian Tan, and Qiqi Qin. 2026. "Removal Performance and Mechanism of Iron–Phosphorus-Based Composite Biochar for Pb(II) and Sb(III) from Water" Separations 13, no. 4: 104. https://doi.org/10.3390/separations13040104
APA StyleRen, T., Zhu, H., Zhu, Z., Tan, J., & Qin, Q. (2026). Removal Performance and Mechanism of Iron–Phosphorus-Based Composite Biochar for Pb(II) and Sb(III) from Water. Separations, 13(4), 104. https://doi.org/10.3390/separations13040104






