1. Introduction
Vanadium is a strategically important metal, widely utilized in the production of alloy steels and catalytic materials, and is often referred to as the “monosodium glutamate of industry” due to its performance-enhancing role [
1,
2]. Vanadium-bearing shale represents a significant alternative resource for vanadium supply, complementing the more conventional source of vanadium titano-magnetite [
3]. Driven by sustained demand from the steel industry and the rapid expansion of emerging energy storage technologies such as vanadium redox flow batteries [
4], the global demand for vanadium has exhibited a marked upward trend. Consequently, the development of clean and efficient methods for vanadium extraction from vanadium-bearing shale carries substantial practical and strategic significance.
Oxalic acid is a naturally occurring organic acid with strong reducing and complexing properties, widely employed in hydrometallurgy for the leaching and separation of metal ions, such as in iron recovery from red mud [
5,
6,
7]. It serves as an efficient and environmentally benign leaching agent in the extraction of vanadium from vanadium-bearing shale [
8]. Compared to conventional strong acid systems, oxalic acid not only effectively dissolves and liberates vanadium but also significantly mitigates adverse environmental impacts. Studies have demonstrated that oxalic acid enables high-efficiency vanadium leaching from vanadium-bearing shale, whether through direct leaching or roasting-leaching processes. Moreover, iron can be selectively precipitated as ferrous oxalate (FeC
2O
4), facilitating effective separation of vanadium and iron [
9,
10]. However, due to the isomorphic substitution of vanadium within the muscovite crystal lattice in vanadium-bearing shale [
11,
12], aluminum ions are concurrently leached as impurities. This leads to a complex oxalic acid leachate containing multiple metallic species, with particularly high concentrations of aluminum, which interferes with the selective recovery of vanadium [
13]. Therefore, efficient vanadium separation requires the deep removal of aluminum ions from the leachate.
Solvent extraction is an efficient technique for the selective separation of target metals from complex multielement aqueous solutions [
14,
15,
16]. By selecting appropriate extractants and tuning the conditions of the aqueous phase, efficient separation of the target metal ion—whether present as a cation or anionic complex—from coexisting impurity ions can be achieved [
17,
18,
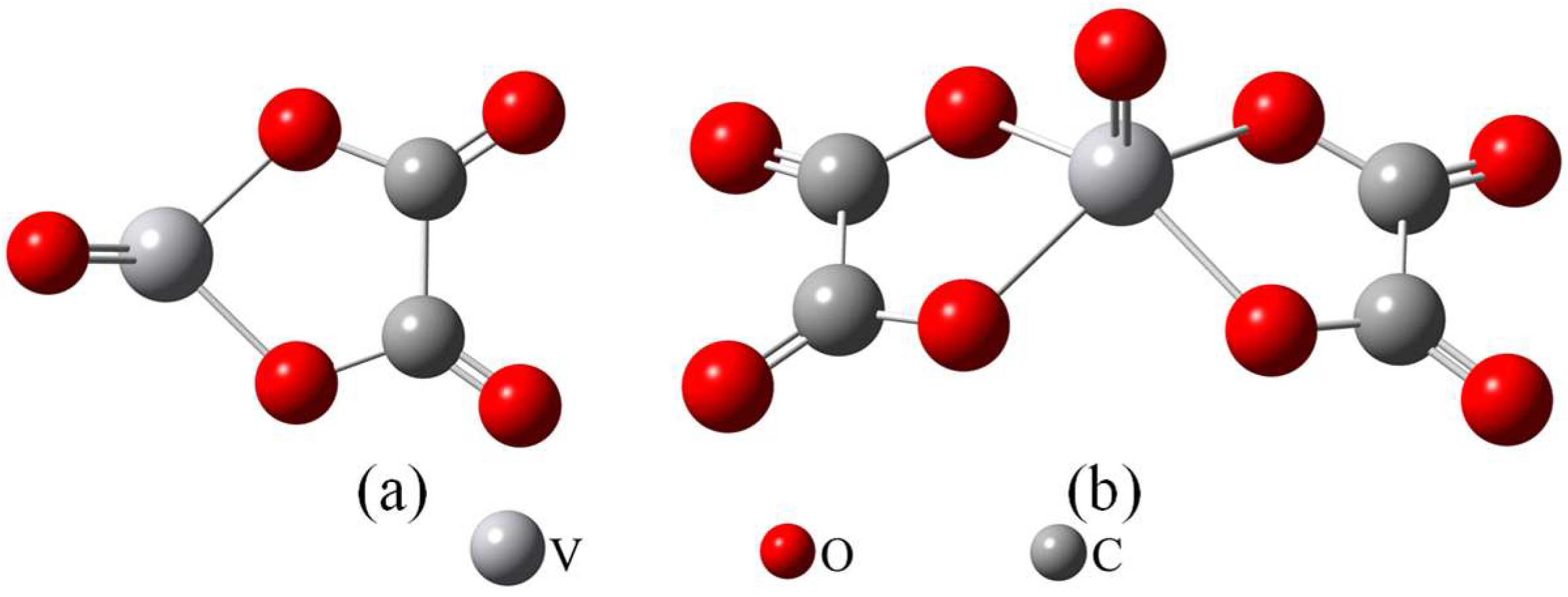
19]. The oxalic acid leachate derived from vanadium-bearing shale contains multiple high-valent metal ions, including vanadium, iron, and aluminum. These ions exist in the oxalic acid system as stable anionic complexes, such as [VO(C
2O
4)
2]
2−, [Fe(C
2O
4)
2]
−, and [Al(C
2O
4)
2]
− [
20,
21,
22]. Differences in the stability and coordination behavior of these complexes provide a basis for their selective separation via solvent extraction. Research indicates that the selectivity in vanadium extraction depends critically on the coordination interaction between the functional groups of the extractant and the vanadium complexes, as well as on the competitive partitioning behavior of various metal oxalate complexes between the aqueous and organic phases [
23,
24]. In our previous study [
25], using Aliquat 336 as the extractant under optimized conditions, selective separation of vanadium and aluminum was achieved, with a vanadium extraction efficiency of 99.06% and an aluminum co-extraction rate of only 7.95%. Through analytical techniques such as FTIR and ESI-MS, combined with thermodynamic modeling, it was determined that vanadium and aluminum predominantly exist as [VO(C
2O
4)
2]
2− and [Al(C
2O
4)
2]
−, respectively, in the oxalic acid leachate of vanadium-bearing shale. This confirmed that the extraction mechanism for both metals follows an anion exchange pathway, wherein [VO(C
2O
4)
2]
2− forms an ion-association complex with the quaternary ammonium cation (R
4N
+) and transfers into the organic phase. However, since both [VO(C
2O
4)
2]
2− and [Al(C
2O
4)
2]
− are anionic species, the molecular-level origin of the selective extraction of vanadium over aluminum by the quaternary ammonium-based extractant Aliquat 336 remains unclear and has not been systematically investigated.
Quantum chemistry, as a high-precision computational tool in materials science, has been widely applied in recent years to chemical systems such as organic molecules, organometallic complexes, and intricate multicomponent media [
26]. With regard to the molecular structure and properties of extractants, quantum chemical calculations and geometry optimizations enable the elucidation of electronic structural features and relative stabilities of metal oxalate complexes at the molecular level [
27,
28], thereby providing a theoretical foundation for understanding the selective separation mechanisms of vanadium and aluminum in quaternary ammonium-based extraction systems. In this study, density functional theory (DFT) calculations are employed. Building upon previous experimental findings, interfacial extraction models involving [VO(C
2O
4)
2]
2− and [Al(C
2O
4)
2]
− with quaternary ammonium cations (R
4N
+) are constructed to investigate the mechanism underlying the selective extraction of vanadium over aluminum from oxalic acid leachate of vanadium-bearing shale. This work thus contributes to refining the theoretical framework for purification and enrichment processes within oxalic acid-based hydrometallurgical systems.
2. Experimental Methods and Reagents
2.1. Composition Analysis of Oxalic Acid Leachate of Vanadium-Bearing Shale
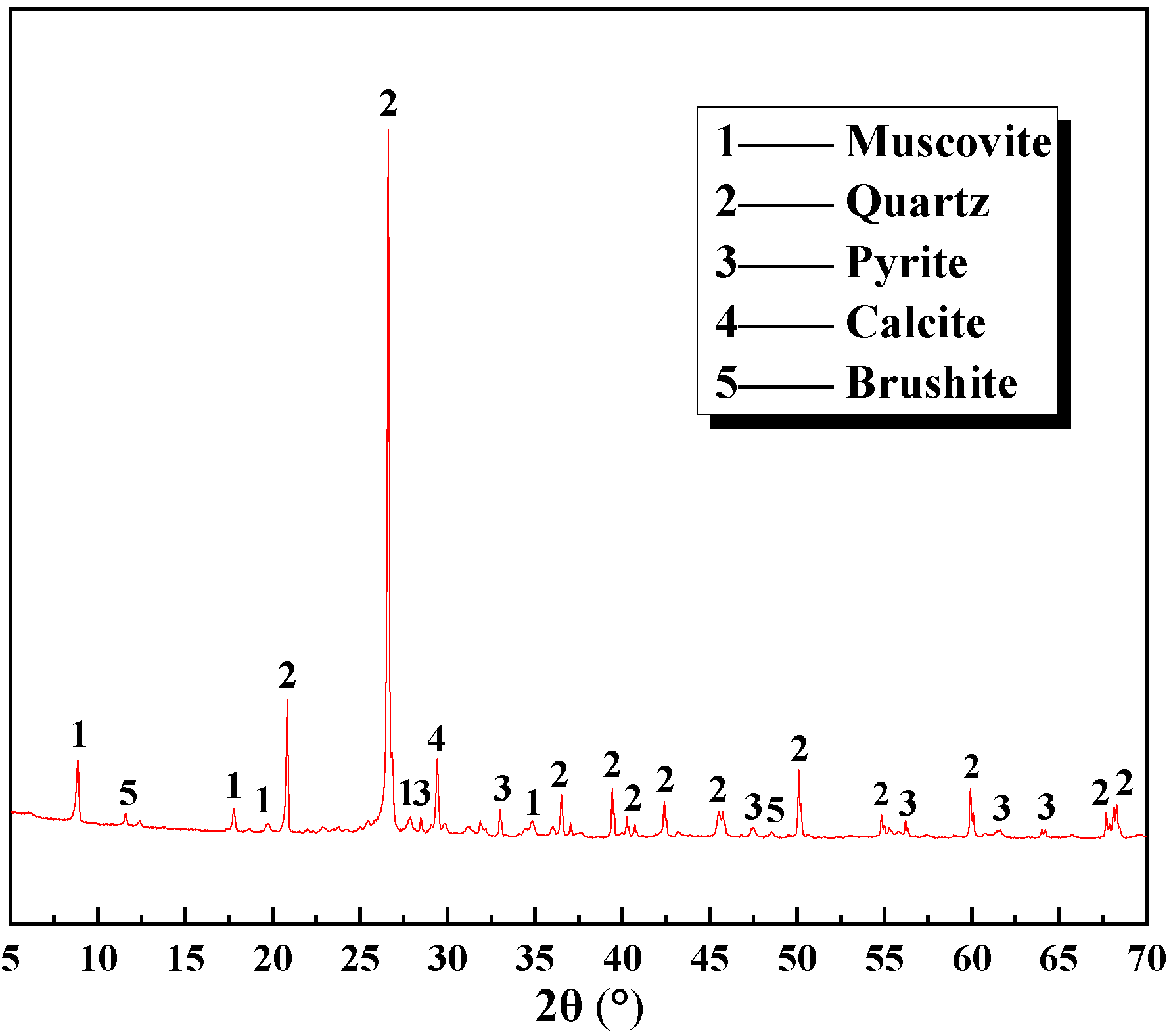
The vanadium-bearing shale was sourced from Hubei. Its phase composition is shown in
Figure 1 [
29], multi-element chemical analysis is shown in
Table 1.
Based on the previously reported methodology [
25], the preparation of oxalic acid leachate from vanadium-bearing shale was carried out as follows: the raw shale sample was roasted at 850 °C for 1 h, followed by leaching at 95 °C for 4 h using 60% oxalic acid and 5% CaF
2, with the L/S = 1:1. After leaching, iron ions were selectively removed from the solution to complete the leachate [
9,
10]. The initial pH of the oxalic acid leachate used in this study was 0.65. The chemical composition of the oxalic acid leachate of shale was analyzed, and the results are presented in
Table 2.
The results presented in
Table 2 indicated that the vanadium concentration in this pickle liquor was relatively low, at only 1.57 g/L. However, the solution contains a variety of impurity ions at high concentrations. Notably, the aluminum concentration reaches as high as 14.23 g/L. Therefore, the pickle liquor constitutes a complex system characterized by low vanadium content, high acidity, and the presence of multiple interfering species, rendering the purification and enrichment of vanadium from this solution particularly challenging.
2.2. Reagents Used in the Experiments
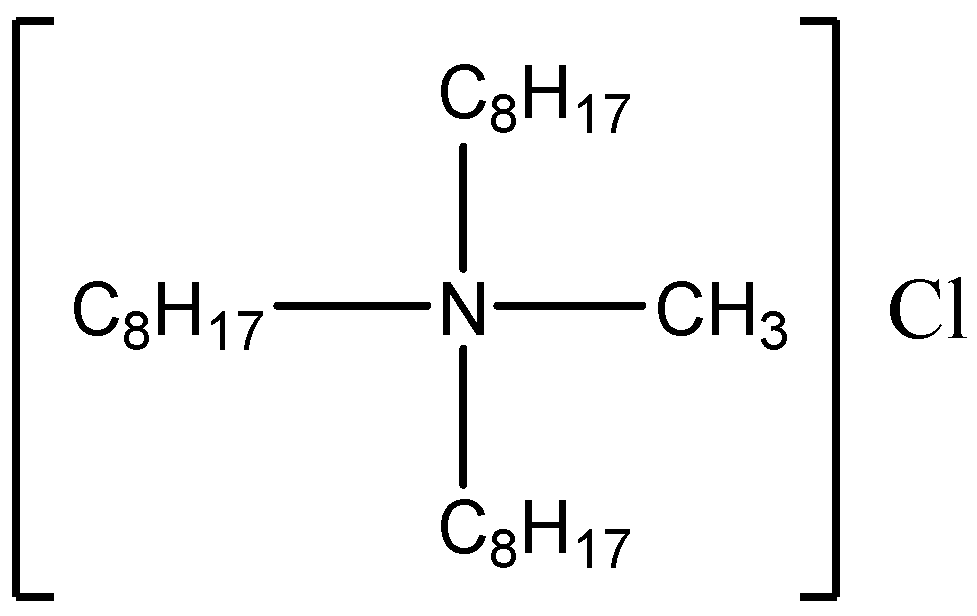
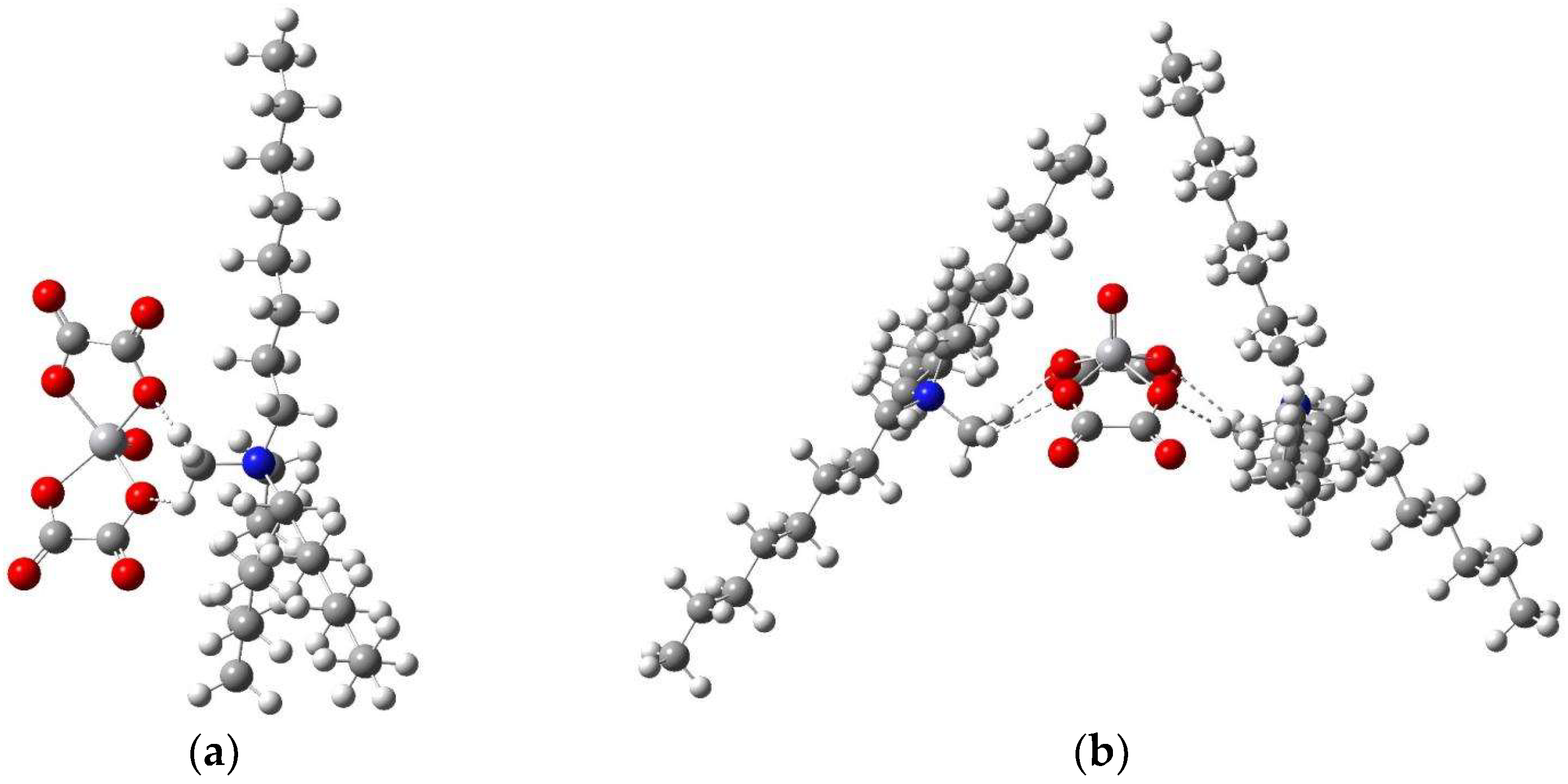
The Aliquat 336 extractant was provided by Zhengzhou Qinshi Technology Co., Ltd. (Zhengzhou, China), and its structure is shown in
Figure 2. The oxalic acid and Tri-butyl phosphate (TBP) were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). All reagents were of analytical grade.
2.3. Extraction Experiments
Organic phase preparation: The extractant (Aliquat 336), phase modifier (TBP), and diluent (sulfonated kerosene) were poured into a beaker in specified proportions, stirred evenly to obtain the organic phase.
Extraction experiment: A magnetic stir bar was placed in a beaker, which was then positioned on a magnetic stirrer. The pickle liquor and organic phase were added to the beaker at the specified volume ratio and mixed under controlled stirring for the designated extraction time. Following the reaction, the biphasic mixture was transferred to a separatory funnel to allow complete phase separation, yielding the loaded organic phase and the raffinate. Assuming negligible volume changes during the extraction process, the extraction efficiency (
E1), distribution ratios (
D), separation factor of ions a and b after extraction (
βa,b) were calculated according to Equations (1)–(5), respectively.
In the formula: C0, C1, and C2 are the ion concentrations in the feed solution, raffinate, and loaded organic phase, respectively (g/L), V1 and V2 are the volumes of the feed solution and loaded organic phase, respectively (L), DV and DAl were distribution ratios of vanadium and aluminum, respectively.
2.4. Determination Method
The grades and concentrations of vanadium were accurately determined using the ammonium ferrous sulfate titration method [
30], a well-established technique for redox-based quantification. Oxalate concentration was quantified via potassium permanganate titration [
31], ensuring reliable measurement under acidic conditions. The grades and concentrations of other elements in both vanadium-bearing shale and the corresponding leachate were systematically analyzed by inductively coupled plasma optical emission spectrometry (ICP-OES, IRIS Intrepid II, Thermo Electron Corporation, Waltham, MA, USA), providing high sensitivity and multi-element detection capability. Phase compositions were comprehensively characterized by X-ray diffraction (XRD, D8 Advance, Bruker, Berlin, Germany), enabling definitive identification of crystalline phases. Together, this suite of analytical methods ensures a robust and accurate assessment of both elemental distribution and mineralogical characteristics.
2.5. Quantum Chemical Calculations
This section employed Gaussian 16W software c01 to perform structural optimization of the proposed extracted complexes in the vanadium–aluminum oxalic acid system. Frequency calculations were conducted on the optimized geometries to obtain thermodynamic parameters. The reaction Gibbs free energy (∆G) for each extraction process was calculated, enabling a comparative analysis of the binding affinities between the extractant and the vanadium or aluminum complexes.
In quantum chemical calculations using Gaussian 16W, the initial step involved constructing the molecular structural models, followed by analysis of the computational results. Structural optimization was subsequently carried out within the framework of Density Functional Theory (DFT) using the B3LYP/6-31G(d) basis set for the extractant, the vanadium and aluminum oxalate complexes, and their corresponding extracted complexes. Thereafter, frequency calculations were performed on all optimized structures under the same theoretical level to confirm the nature of stationary points (minima) and to derive thermodynamic corrections. The Gibbs free energy changes (∆G) for both extraction and reverse extraction (stripping) reactions were computed and compared, allowing for an assessment of the relative binding energies associated with the extraction and recovery of vanadium and aluminum.