Bismuth Oxychloride@Graphene Oxide/Polyimide Composite Nanofiltration Membranes with Excellent Self-Cleaning Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of the Composite Membrane
2.3. Characterizations
2.4. Separation and Photocatalytic Performance Examination of BiOCl@GO/PI Membrane
2.5. Photocatalytic Mechanism and Cycle Stability Study of BiOCl@GO/PI Membrane
3. Results and Discussion
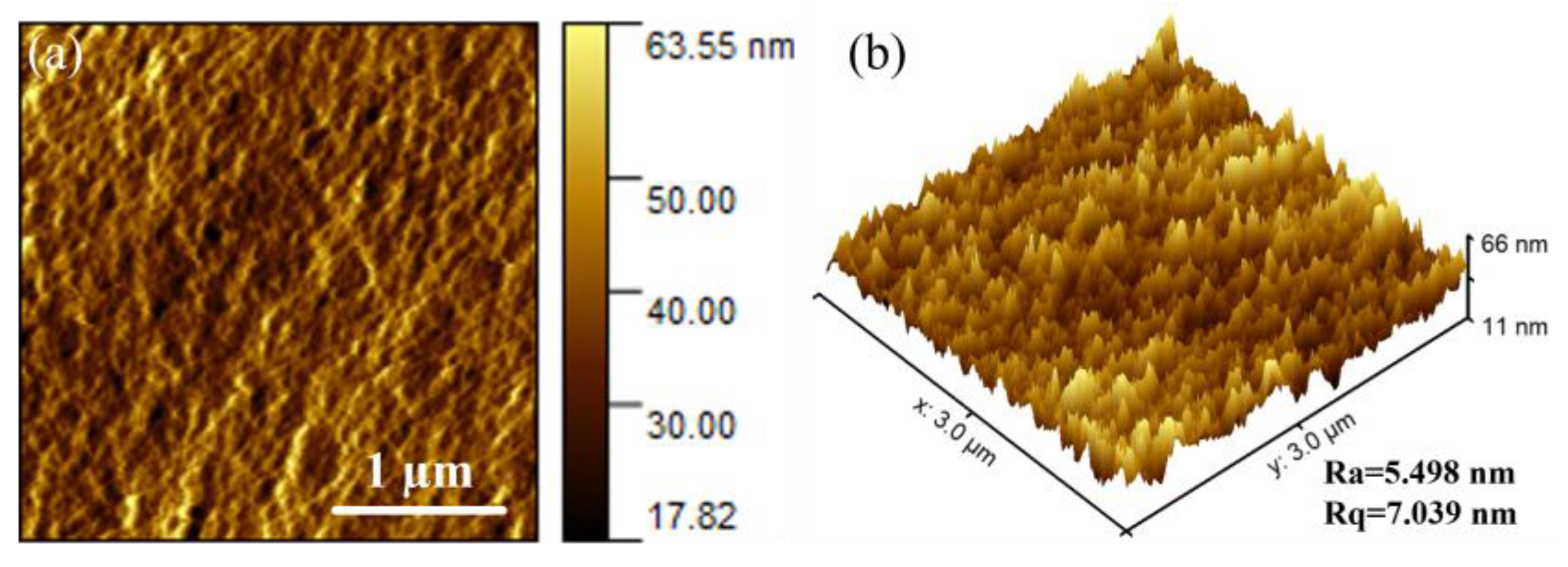
3.1. Membrane Characterization
3.2. Separation Performance of the BiOCl@GO/PI Membrane
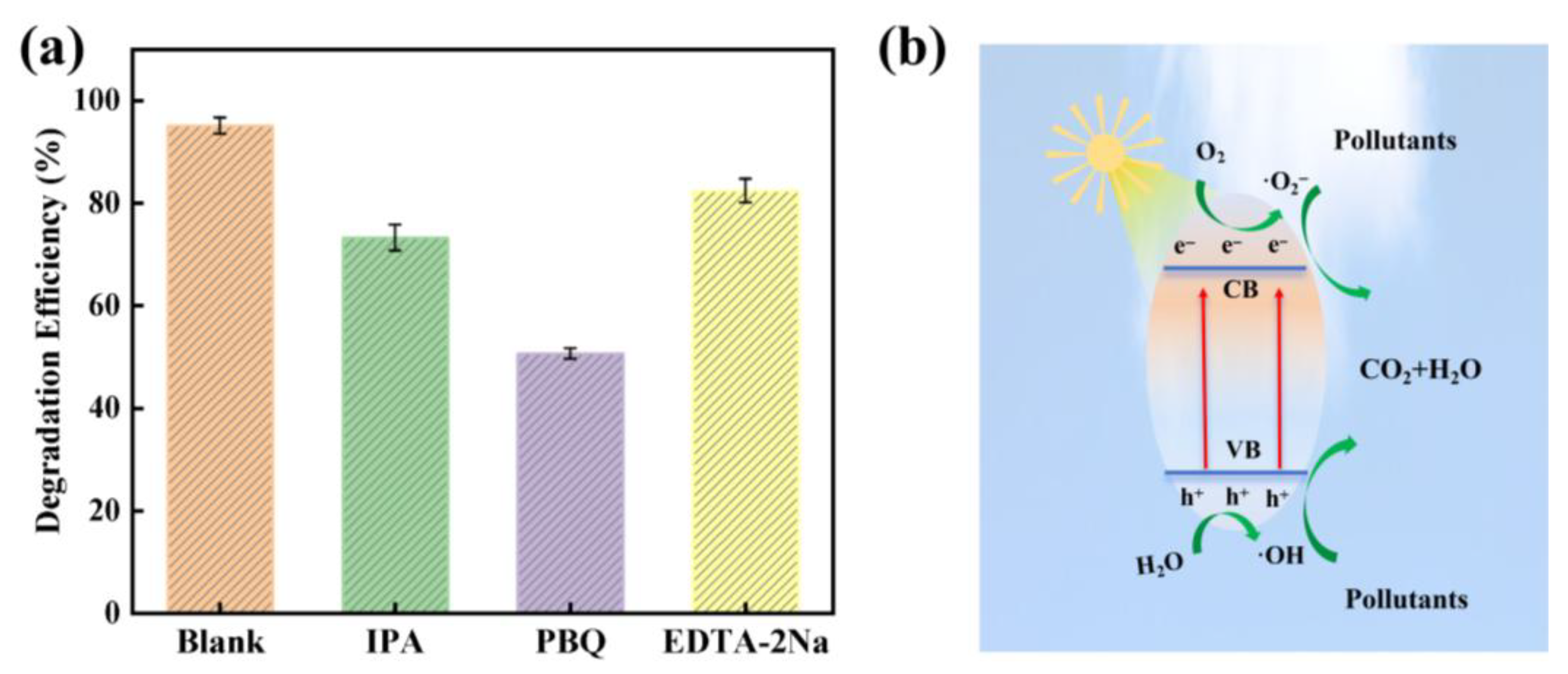
3.3. Self-Cleaning Performance of the BiOCl@GO/PI Membrane
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, C.; Sun, H.; Wang, N.; An, Q.-F. Robust ZIF-8 and Its Derivative Composite Membrane for Antibiotic Desalination with High Performance. Sep. Purif. Technol. 2023, 307, 122857. [Google Scholar] [CrossRef]
- Yang, Z.; Lin, Q.; Zeng, G.; Zhao, S.; Yan, G.; Ang, M.B.M.Y.; Chiao, Y.-H.; Pu, S. Ternary Hetero-Structured BiOBr/Bi2MoO6@MXene Composite Membrane: Construction and Enhanced Removal of Antibiotics and Dyes from Water. J. Membr. Sci. 2023, 669, 121329. [Google Scholar] [CrossRef]
- Basu, S.; Balakrishnan, M. Polyamide Thin Film Composite Membranes Containing ZIF-8 for the Separation of Pharmaceutical Compounds from Aqueous Streams. Sep. Purif. Technol. 2017, 179, 118–125. [Google Scholar] [CrossRef]
- Li, Y.; Han, M.; Chen, J.; Tan, X.; Xie, W.; Lou, H.; Chen, Z.; Gu, H. In-Situ Grown Magnetic Nanoparticles on Graphene Oxide for Removal of Hexavalent Chromium from Wastewater. Colloids Surf. A Physicochem. Eng. Asp. 2025, 712, 136430. [Google Scholar] [CrossRef]
- Li, Y.; Chen, J.; Tan, X.; Lou, H.; Gu, H. Aniline-p-Phenylenediamine Copolymer for Removal of Hexavalent Chromium from Wastewater. Separations 2024, 11, 327. [Google Scholar] [CrossRef]
- Feng, F.; Dong, G.; Wang, J.; Zhu, Z.; Liu, X.; Han, R. Bioinspired PDA/PVDF Composite Membrane with Excellent Antibiotic Desalination Properties. Emerg. Mater. Res. 2025, 14, 381–388. [Google Scholar] [CrossRef]
- Zeng, J.; Xie, W.; Guo, Y.; Zhao, T.; Zhou, H.; Wang, Q.; Li, H.; Guo, Z.; Xu, B.B.; Gu, H. Magnetic Field Facilitated Electrocatalytic Degradation of Tetracycline in Wastewater by Magnetic Porous Carbonized Phthalonitrile Resin. Appl. Catal. B Environ. 2024, 340, 123225. [Google Scholar] [CrossRef]
- Zhu, Z.; Feng, F.; Li, J.; Wang, G.; Dong, G.; Han, R. Selective Solvent Resistant Polyimide Composite Membranes with Efficient Separation Performance in Antibiotics. Results Eng. 2025, 29, 108653. [Google Scholar] [CrossRef]
- Lim, S.K.; Goh, K.; Bae, T.-H.; Wang, R. Polymer-Based Membranes for Solvent-Resistant Nanofiltration: A Review. Chin. J. Chem. Eng. 2017, 25, 1653–1675. [Google Scholar] [CrossRef]
- Dong, G.; Feng, F.; Su, S.; Zhu, Z.; Liu, X.; Han, R. Facile Preparation of Quaternary Ammonium Graft-Modified PVDF Nanofiltration Membrane with Excellent Antibiotic Desalination and Antibacterial Performance. J. Water Process Eng. 2026, 81, 109284. [Google Scholar] [CrossRef]
- Cheng, K.; Zhang, N.; Yang, N.; Hou, S.; Ma, J.; Zhang, L.; Sun, Y.; Jiang, B. Rapid and Robust Modification of PVDF Ultrafiltration Membranes with Enhanced Permselectivity, Antifouling and Antibacterial Performance. Sep. Purif. Technol. 2021, 262, 118316. [Google Scholar] [CrossRef]
- Han, R.; Feng, F.; Zhu, Z.; Dong, G.; Liu, X.; Guo, M.; Yang, X.; Qin, J.; Wang, H.; Dai, Y.; et al. Engineering Multifunctional MDPCs@MOFs via Selective Thermal Etching Integrates Low-Resistance Transport with Strong Adsorption for Enhanced CO2/N2 Separation. J. Colloid Interface Sci. 2025, 708, 139760. [Google Scholar] [CrossRef]
- Khan, A.; Li, W.; Ma, X.; Dong, M.; Geng, L.; Khan, S.S.; Li, Y.; Khan, S.; Yasin, G. Large-Scale with Uniformly Dispersed Silver Nanoparticles Decorated Poly(Acrylonitrile) (PAN) Membrane Preparation for Efficient Photocatalytic Reactions and Antimicrobial Activity. Opt. Mater. 2024, 149, 115063. [Google Scholar] [CrossRef]
- Yu, S.; Wang, Y.; Sun, F.; Wang, R.; Zhou, Y. Novel Mpg-C3N4/TiO2 Nanocomposite Photocatalytic Membrane Reactor for Sulfamethoxazole Photodegradation. Chem. Eng. J. 2018, 337, 183–192. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, H.; Fan, X.; Lv, F.; Chen, S.; Quan, X. Fabrication of TiO2 Nanofiber Membranes by a Simple Dip-Coating Technique for Water Treatment. Surf. Coat. Technol. 2016, 298, 45–52. [Google Scholar] [CrossRef]
- Chen, C.; Fei, L.; Wang, B.; Xu, J.; Li, B.; Shen, L.; Lin, H. MOF-Based Photocatalytic Membrane for Water Purification: A Review. Small 2024, 20, 2305066. [Google Scholar] [CrossRef]
- Sasikala, V.; Karthik, P.; Ravichandran, S.; Prakash, N.; Rajesh, J.; Mukkannan, A. Facile Synthesis of BiOCl Incorporated Polymer Membrane for Effective Photocatalytic Dye Degradation. J. Polym. Environ. 2024, 32, 1937–1951. [Google Scholar] [CrossRef]
- Duan, H.; Zhuang, Z.; Yang, J.; Zhang, S.; Wang, L. Confined Gas Transport in Low-Dimensional Materials. Int. J. Smart Nano Mater. 2024, 15, 127–164. [Google Scholar] [CrossRef]
- Salemi, F.; Karimzadeh, F.; Abbasi, M.-H.; Moradi, F.; Pham, D.H.; Kim, J. Effective Parameters on Polydimethylsiloxane/Graphene Composite-Based Triboelectric Nanogenerator Performance. Int. J. Smart Nano Mater. 2024, 15, 365–386. [Google Scholar] [CrossRef]
- Ruiz-Torres, C.A.; Kang, J.; Kang, S.; Kim, J.P.; Kim, C.; Kang, K.M.; Cho, K.M.; Nam, Y.T.; Chang, Y.-Y.; Byon, C.; et al. Oxidation-Controlled Nanoporous Graphene Laminate Membranes via Intercalation Chemistry for Desalination. J. Membr. Sci. 2025, 735, 124515. [Google Scholar] [CrossRef]
- Ma, L.; Shi, X.; Zhang, G.; Wen, X.; Liu, C.; Ma, X.; Jiang, M.; Li, J.; Zhang, L. A Stable Bimetallic MOF (Ti, Zr)-Graphene Oxide Composite Membrane with Synergistic Anti-Fouling Strategy of Photocatalysis and Hydration Layer for Efficient Separation of Oily Emulsions. Sep. Purif. Technol. 2026, 382, 136016. [Google Scholar] [CrossRef]
- Joseph, A.; Yelekar, G.R.; Vijayanandan, A. Efficiency of Titanium Dioxide-Reduced Graphene Oxide in Carbamazepine Removal: A Comparative Study of Adsorption, Photocatalysis, and Ultrafiltration Techniques. Appl. Catal. O Open 2024, 193, 206967. [Google Scholar] [CrossRef]
- Runlin, H.; Chaoyue, W.; Congcong, B.; Hanli, W. Facile Preparation of High Performance GO/Mn3O4/PVDF Composite Membranes with Intercalation of Manganese Oxide Nanowires. RSC Adv. 2023, 13, 19002–19010. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Shi, G.; Shen, J.; Peng, B.; Zhang, B.; Wang, Y.; Bian, F.; Wang, J.; Li, D.; Qian, Z.; et al. Ion Sieving in Graphene Oxide Membranes via Cationic Control of Interlayer Spacing. Nature 2017, 550, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, L.; Chen, R.; Liu, Q.; Liu, J.; Yu, J.; Liu, P.; Duan, J.; Wang, J. Surface Morphology Properties and Antifouling Activity of Bi2WO6/Boron-Grafted Polyurethane Composite Coatings Realized via Multiple Synergy. J. Colloid Interface Sci. 2022, 626, 815–823. [Google Scholar] [CrossRef]
- Feng, F.; Zhu, Z.; Li, J.; Su, S.; Dong, G.; Wang, G.; Liu, X.; Han, R. In-Situ Grown Electrospun PI/BiOCl Nanofiber Membranes for Efficient Photocatalytic Degradation of Antibiotics. J. Photochem. Photobiol. A Chem. 2026, 474, 117014. [Google Scholar] [CrossRef]
- Liao, H.; Liu, C.; Zhong, J.; Li, J. Fabrication of BiOCl with Adjustable Oxygen Vacancies and Greatly Elevated Photocatalytic Activity by Using Bamboo Fiber Surface Embellishment. Colloids Surf. A Physicochem. Eng. Asp. 2022, 634, 127892. [Google Scholar] [CrossRef]
- Le, T.M.H.; Chuchak, R.; Sairiam, S. Empowering TiO2–Coated PVDF Membranes Stability with Polyaniline and Polydopamine for Synergistic Separation and Photocatalytic Enhancement in Dye Wastewater Purification. Sci. Rep. 2024, 14, 15969. [Google Scholar] [CrossRef]
- Han, R.; Wu, K.; Xu, L. Facile Preparation of Loose P84 Copolyimide/GO Composite Membrane with Excellent Selectivity and Solvent Resistance. Polymers 2022, 14, 1353. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Liao, J.; Xue, Y.; Lin, Q.; Yang, Z.; Yan, G.; Zeng, G.; Sengupta, A. Ultrahigh-Flux and Self-Cleaning Composite Membrane Based on BiOCl-PPy Modified MXene Nanosheets for Contaminants Removal from Wastewater. J. Membr. Sci. 2022, 644, 120188. [Google Scholar] [CrossRef]
- Han, R.; Feng, F.; Zhu, Z.; Su, S.; Wang, J.; Shi, C.; Dong, G.; Gu, H. Innovative Bi-Functional Cellulose Acetate/BiOCl Photocatalytic Membrane with Excellent Antibiotics Degradation and Photoresponsive Antimicrobial Performance. Colloids Surf. A Physicochem. Eng. Asp. 2026, 730, 138959. [Google Scholar] [CrossRef]
- Kral, M.; Dendisova, M.; Svoboda, J.; Cernescu, A.; Svecova, M.; Johnson, C.M.; Pop-Georgievski, O.; Matejka, P. Nano-FTIR Spectroscopy of Surface Confluent Polydopamine Films—What Is the Role of Deposition Time and Substrate Material? Colloids Surf. B Biointerfaces 2024, 235, 113769. [Google Scholar] [CrossRef]
- Liu, H.; Yang, C.; Huang, J.; Chen, J.; Zhong, J.; Li, J. Ionic Liquid-Assisted Hydrothermal Preparation of BiOI/BiOCl Heterojunctions with Enhanced Separation Efficiency of Photo-Generated Charge Pairs and Photocatalytic Performance. Inorg. Chem. Commun. 2020, 113, 107806. [Google Scholar] [CrossRef]
- Petukhov, D.I.; Weston, J.; Valeev, R.G.; Johnson, D.J. Graphene Oxide Surface Modification of Reverse Osmosis (RO) Membrane via Langmuir–Blodgett Technique: Balancing Performance and Antifouling Properties. Membranes 2024, 14, 172. [Google Scholar] [CrossRef] [PubMed]
- Petukhov, D.I.; Chumakov, A.P.; Johnson, D.J. Effect of Competition between Swelling and Dye Adsorption on the Performance and Selectivity of Graphene Oxide Membranes. Nanoscale 2025, 17, 25572–25588. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Du, J.; Shi, Y.; Fang, X.; Ma, X.; Liu, D.; Cao, D.; Li, J.; Chen, R.; Wu, C.; et al. Highly Active 3D Composites for a Flow-Through Photocatalytic Membrane Reactor toward Water Micropollutant Removal. ACS Mater. Lett. 2024, 6, 427–437. [Google Scholar] [CrossRef]










| Name | GO (mg) | DA·HCl (mg) | BiCl3 (mg) |
|---|---|---|---|
| M0 | 0 | 0 | 0 |
| M1 | 20 | 10 | 2.5 |
| M2 | 20 | 20 | 2.5 |
| M3 | 20 | 20 | 20 |
| Element | Atomic % |
|---|---|
| Bi 4f | 10.53 |
| C 1s | 45.97 |
| Cl 2p | 13.91 |
| N 1s | 5.84 |
| O 1s | 23.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Han, R.; Feng, F.; Zhu, Z.; Li, J.; Kou, Y.; Yan, C.; Gu, H. Bismuth Oxychloride@Graphene Oxide/Polyimide Composite Nanofiltration Membranes with Excellent Self-Cleaning Performance. Separations 2026, 13, 37. https://doi.org/10.3390/separations13010037
Han R, Feng F, Zhu Z, Li J, Kou Y, Yan C, Gu H. Bismuth Oxychloride@Graphene Oxide/Polyimide Composite Nanofiltration Membranes with Excellent Self-Cleaning Performance. Separations. 2026; 13(1):37. https://doi.org/10.3390/separations13010037
Chicago/Turabian StyleHan, Runlin, Faxiang Feng, Zanming Zhu, Jiale Li, Yiting Kou, Chaowei Yan, and Hongbo Gu. 2026. "Bismuth Oxychloride@Graphene Oxide/Polyimide Composite Nanofiltration Membranes with Excellent Self-Cleaning Performance" Separations 13, no. 1: 37. https://doi.org/10.3390/separations13010037
APA StyleHan, R., Feng, F., Zhu, Z., Li, J., Kou, Y., Yan, C., & Gu, H. (2026). Bismuth Oxychloride@Graphene Oxide/Polyimide Composite Nanofiltration Membranes with Excellent Self-Cleaning Performance. Separations, 13(1), 37. https://doi.org/10.3390/separations13010037







