Dimethyl Carbonate as a Mobile-Phase Modifier for Normal-Phase and Hydrophilic Interaction Liquid Chromatography
Abstract
1. Introduction
2. Materials and Methods
2.1. HILIC Instrumentation
2.2. HILIC Chemicals and Procedures
2.3. Normal-Phase LC Instrumentation
2.4. Normal-Phase LC Chemicals and Procedures
2.5. DMC Purity Analysis by GC
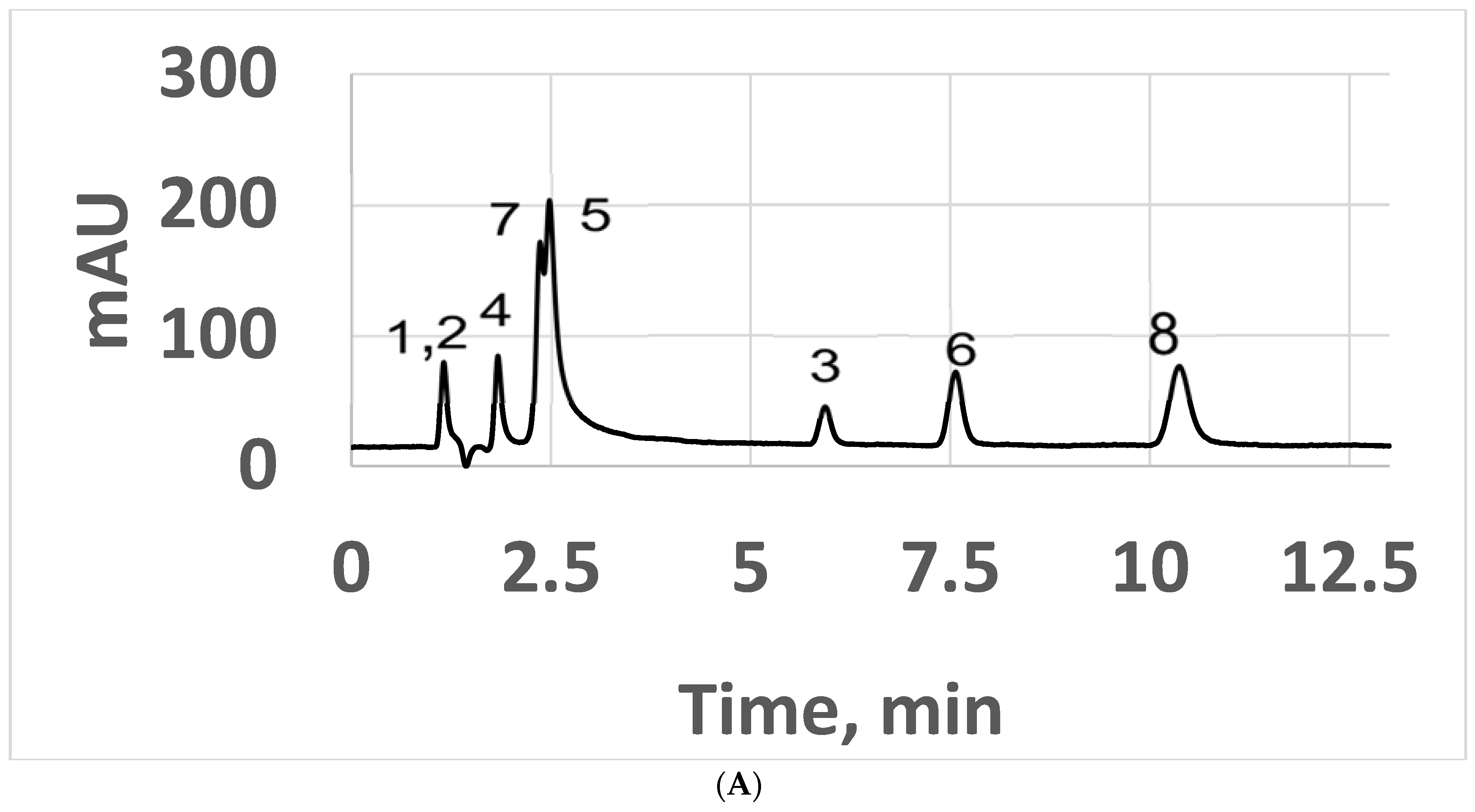
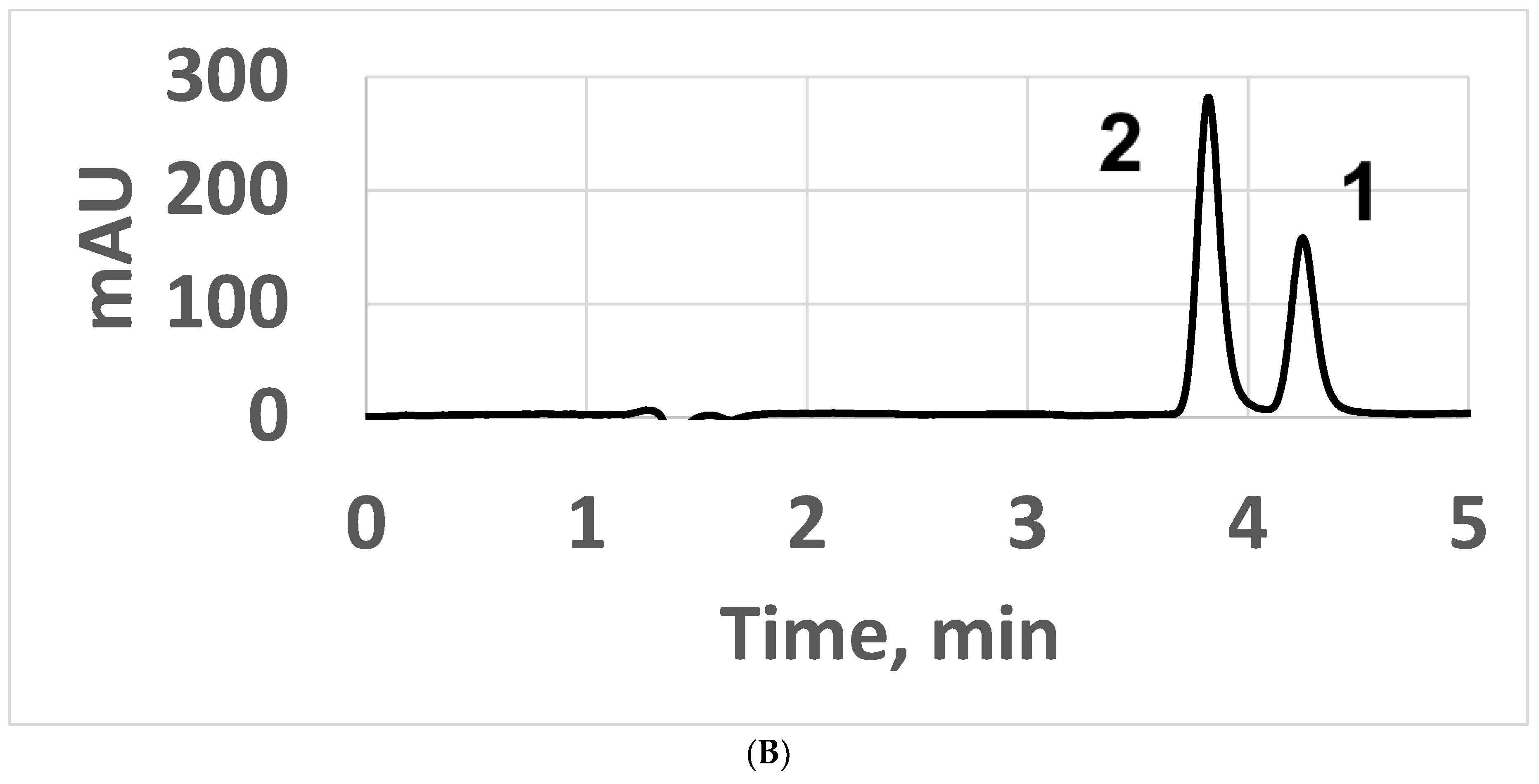
3. Results and Discussion
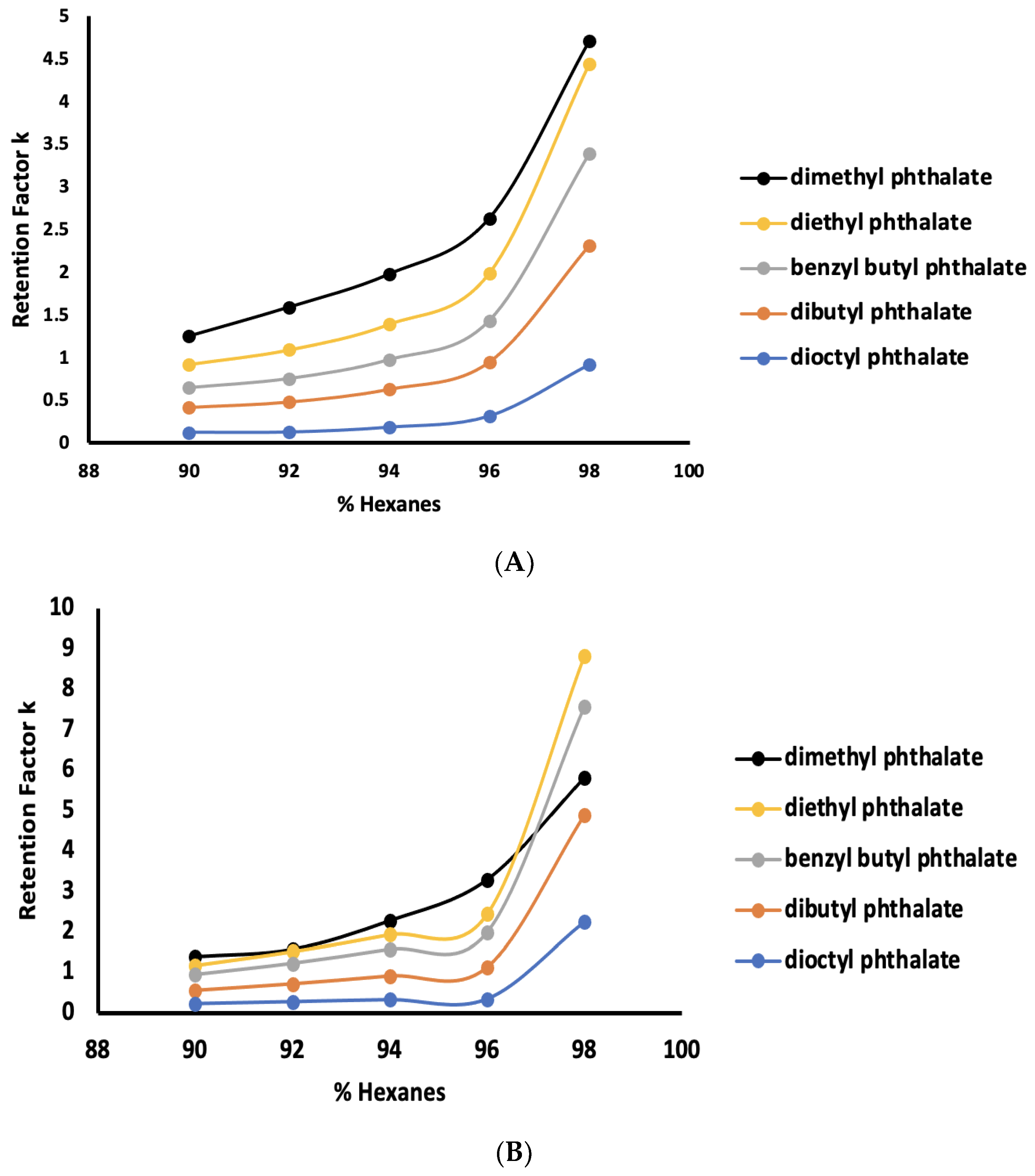
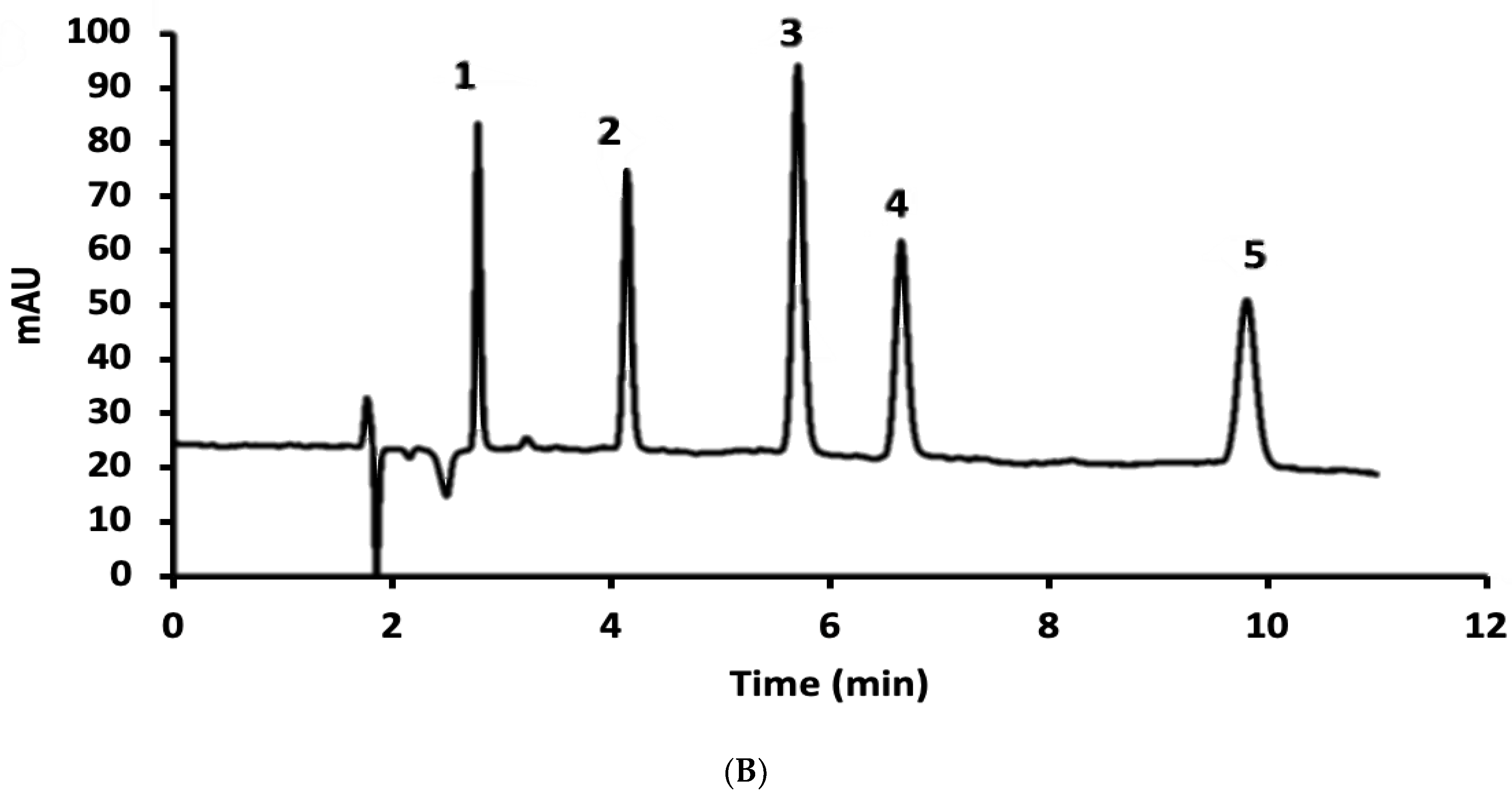
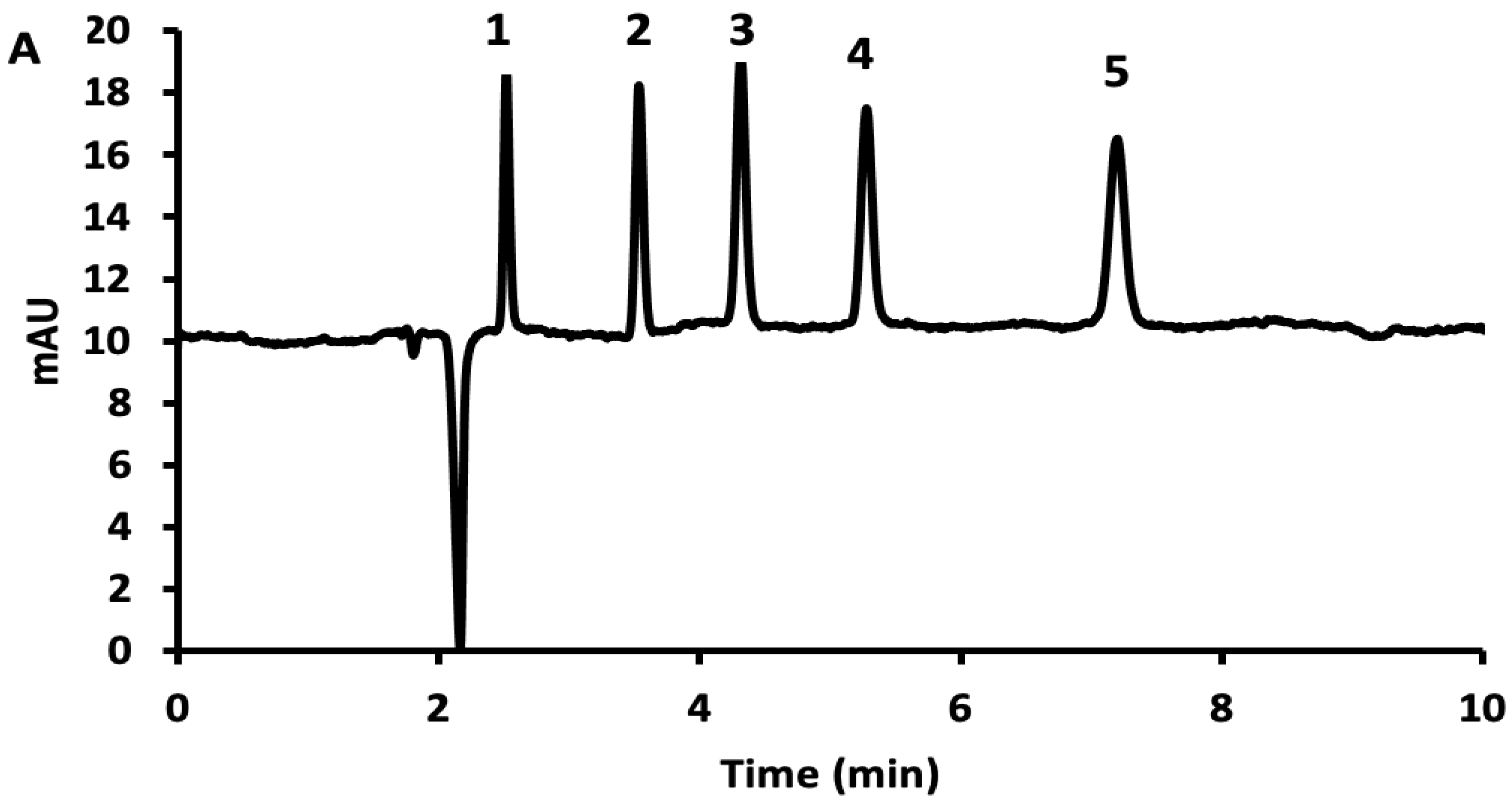
3.1. Normal-Phase LC
3.2. Hydrophilic Interaction Liquid Chromatography
3.2.1. Retention Factor Comparison between DMC and MeCN
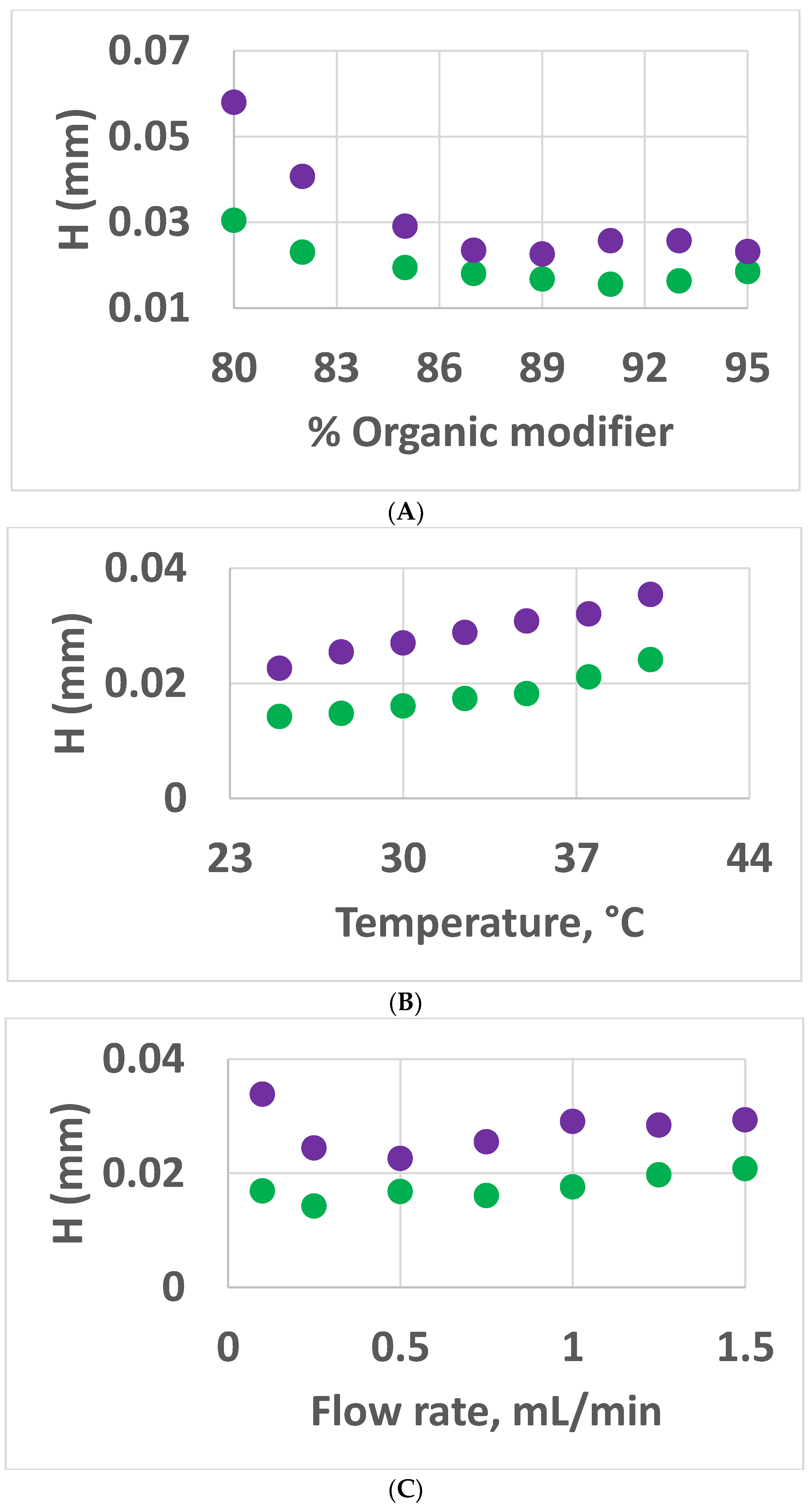
3.2.2. Van Deemter and Peak Asymmetry Comparison of DMC and MeCN
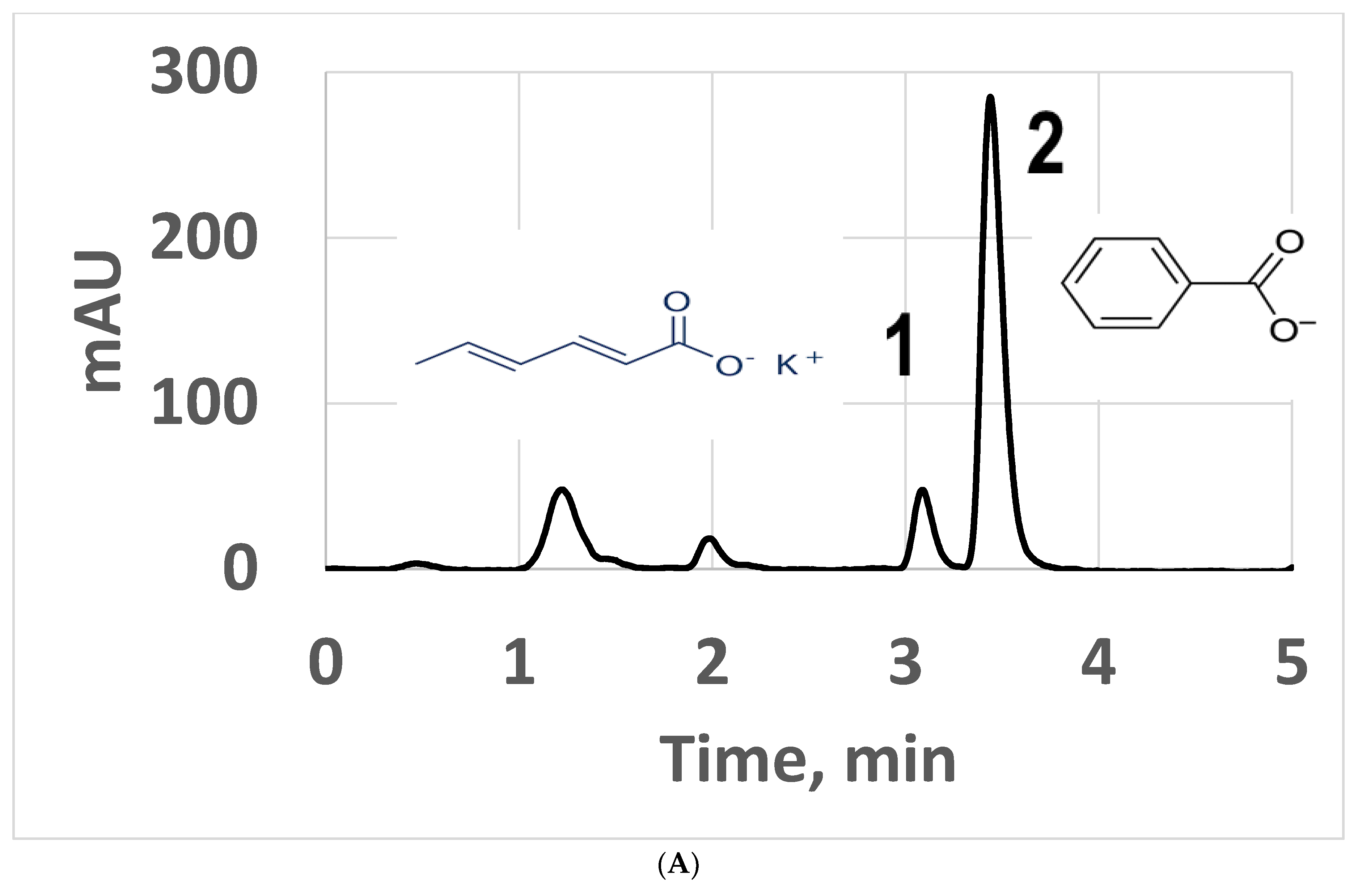
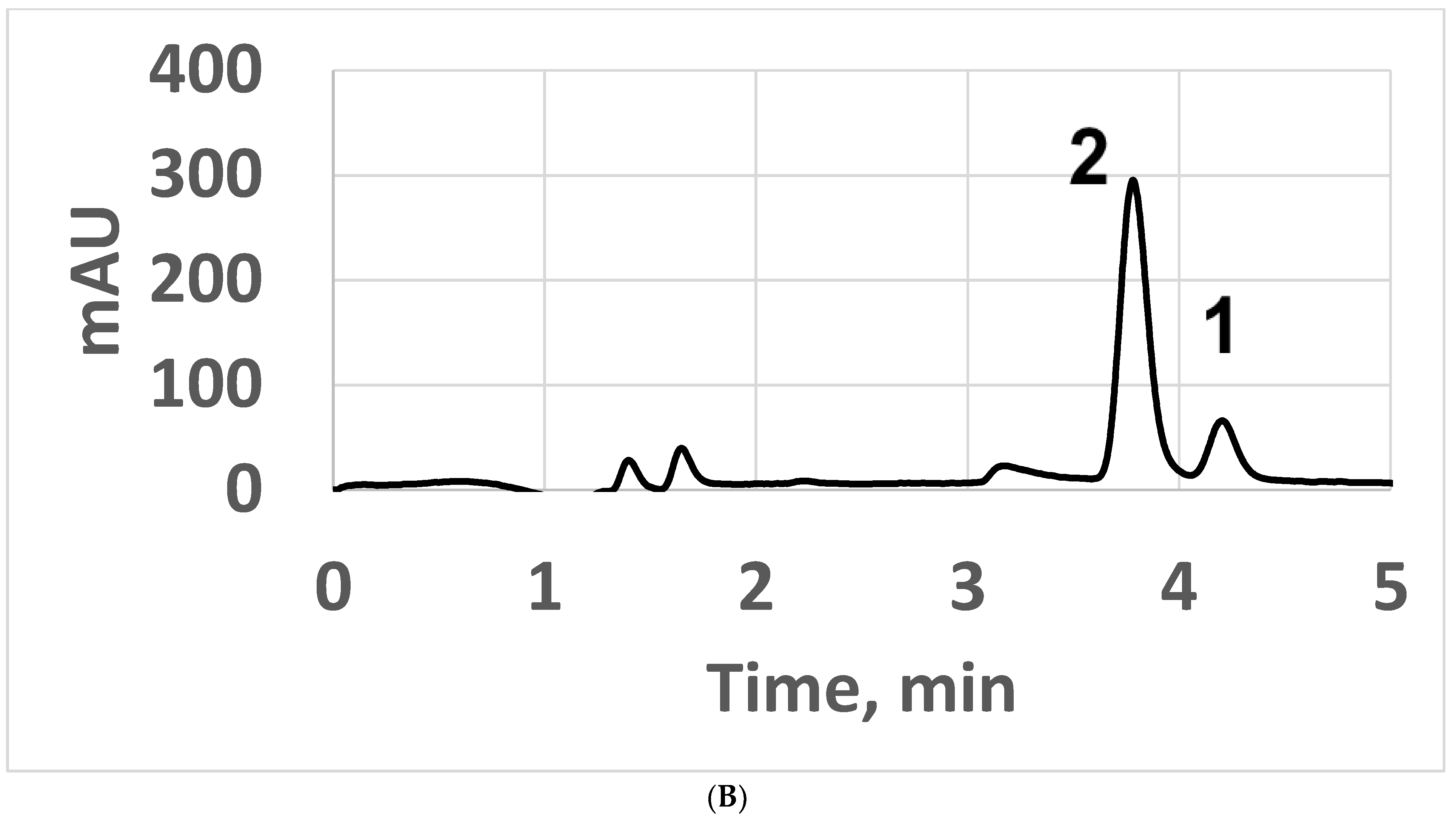
3.2.3. Applications Involving Positional Isomers and Drink Preservatives
3.2.4. Greenness Evaluation of DMC as a Mobile Phase for LC
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Waterlot, C.; Ghinet, A.; Lipka, E. Core-shell particles: A way to greening liquid chromatography in environmental applications. Curr. Chromatogr. 2018, 5, 78–90. [Google Scholar] [CrossRef]
- Aly, A.A.; Gorecki, T. Green chromatography and related techniques. In Green Analytical Chemistry. Green Chemistry and Sustainable Technology; Plotka-Wasylka, J., Namiesnik, J., Eds.; Springer: Singapore, 2019. [Google Scholar]
- Dogan, A.; Eylem, C.; Nursabah, N.E.B. An overview of green chromatographic analysis from pharmaceutical preparations. Curr. Anal. Chem. 2021, 17, 1260–1277. [Google Scholar] [CrossRef]
- Zotou, A. An overview of recent advances in HPLC instrumentation. Cent. Eur. J. Chem. 2012, 10, 554–569. [Google Scholar] [CrossRef]
- Napolitano-Tabares, P.I.; Negrin-Santmaria, I.; Gutierrez-Serpa, A.; Pino, V. Recent efforts to increase greenness in chromatography. Curr. Opin. Green Sustain. Chem. 2021, 32, 100536. [Google Scholar] [CrossRef]
- Dembek, M.; Bocian, S. Pure water as a mobile phase in liquid chromatography techniques. Trends Anal. Chem. 2020, 123, 115793. [Google Scholar] [CrossRef]
- El-Maghrabey, M.H.; Sivasankar, V.; El-Shaheny, R.N. (Eds.) Green Chemical Analysis and Sample Preparations; Springer Nature: London, UK, 2022. [Google Scholar]
- van der Wal, S.J. Low viscosity organic modifiers in reversed-phase HPLC. Chromatographia 1985, 20, 274–278. [Google Scholar] [CrossRef]
- Sangle, M.; Shirsath, A.; Gondkar, S.; Kulkarni, A. Fundamentals and recent progress on green high performance liquid chromatography. Int. J. Res. Anal. Rev. 2022, 9, 69. [Google Scholar]
- Gilomen, K.; Stauffer, H.P.; Meyer, V.R. Detoxification of acetonitrile—water wastes from liquid chromatography. Chromatographia 1995, 41, 488–491. [Google Scholar]
- Yabr´e, M.; Ferey, L.; Som´e, I.T.; Gaudin, K. Greening reversed-phase liquid chromatography methods using alternative solvents for pharmaceutical analysis. Molecules 2018, 23, 1065. [Google Scholar] [CrossRef]
- Micale, F.; Albu, F.; Lorgulescu, E.-E.; Medvedovici, A.; Tache, F. Ethyl lactate as a greener alternative to acetonitrile in RPLC: A realistic approach. J. Chrom. Sci. 2015, 53, 1701–1707. [Google Scholar] [CrossRef]
- Nallar, M.; Tenaglia, N.; Morose, G.; Wong, H.-W. Safer solvent blends for food, dye, and environmental analyses using reversed-phase high performance liquid chromatography. Chromatographia 2021, 21, 769–780. [Google Scholar] [CrossRef]
- Habib, A.; Mabrouk, M.M.; Fekry, M.; Mansour, F.R. Glycerol as a new mobile phase modifier for green liquid chromatographic determination of ascorbic acid and glutathione in pharmaceutical tablets. J. Pharm. Biomed. Anal. 2014, 95, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.F.; Danielson, N.D. Ultra-high performance micellar liquid chromatography comparing Tween 20 and Tween 40 for the determination of hydroxycinnamic acids. Separations 2022, 9, 61. [Google Scholar] [CrossRef]
- Ali, A.F.; Danielson, N.D. Micellar and sub-micellar liquid chromatography of terephthalic acid contaminants using a C18 column coated with Tween 20. Anal. Chim. Acta 2020, 1105, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Angel, M.J.; Carda-Broch, S.; Garcia-Alvarez-Coque, M.C. Ionic liquids as mobile phase additives and immobilized on stationary phases in liquid chromatography. In Ionic Liquids in Analytical Chemistry; Ruiz-Angel, M.J., Carda-Broch, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 203–234. [Google Scholar]
- Sutton, A.T.; Fraige, K.; Leme, G.M.; da Silva Bolzani, V.; Hilder, E.F.; Cavalheiro, A.J.; Arrua, R.D.; Funari, C.S. Natural deep eutectic solvents as the major mobile phase components in high-performance liquid chromatography—Searching for alternatives to organic solvents. Anal. Bioanal. Chem. 2018, 419, 3705–3713. [Google Scholar] [CrossRef]
- Varsha, N.; Suvarna, B.; Pratibha, V.; Soni, M.; Ashok, B. Replacement of acetonitrile by mixtures of propylene carbonate and methanol as organic modifier in mobile phases for RPLC separation mechanism: Application to the assay of alprazolam and sertraline in combined pharmaceutical formulations. J. Liq. Chrom. Rel. Technol. 2012, 35, 2643–2654. [Google Scholar] [CrossRef]
- Varsha, N.; Pratibha, V.; Soni, M.; Ashok, B.; Suvarna, B. Estimation of paracetamol and lornoxicam by isocratic, gradient, and elevated temperature HPLC using propylene carbonate. J. Liq. Chrom. Rel. Technol. 2014, 37, 1094–1103. [Google Scholar] [CrossRef]
- Dogan, A.; Basci, N.E. Green bioanalytical and pharmaceutical analysis of voriconazole and tadalafil by HPLC. Curr. Pharm. Anal. 2017, 13, 495–504. [Google Scholar] [CrossRef]
- Cheragi, M.; Albu, F.; Udrescu, S.; Raducanu, N.; Medvedovici, A. Greener bioanalytical approach for LC/MS-MS assay of enalapril and enalaprilat in human plasma with total replacement of acetonitrile throughout all analytical stages. J. Chromatogr. B. 2013, 927, 124–132. [Google Scholar] [CrossRef]
- Aly, A.A.; Gorecki, T.; Omar, M.A. Green approaches to comprehensive two-dimensional liquid chromatography (LC × LC). J. Chromatogr. Open 2022, 2, 100046. [Google Scholar] [CrossRef]
- Aly, A.A.; Gorecki, T. Green comprehensive two-dimensional liquid chromatography (LC × LC) for the analysis of phenolic compounds in grape juices and wine. Anal. Bioanal. Chem. 2022. [Google Scholar] [CrossRef]
- Tache, F.; Udrescu, S.; Albu, F.; Micăle, F.; Medvedovici, A. Greening pharmaceutical applications of liquid chromatography through using propylene carbonate-ethanol mixtures instead of acetonitrile as organic modifier in the mobile phases. J. Pharm. Biomed. Anal. 2013, 75, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Pyo, S.H.; Park, J.H.; Chang, T.S.; Hatti-Kaul, R. Dimethyl carbonate as a green chemical. Curr. Opin. Green Sustain. Chem. 2017, 5, 61–66. [Google Scholar] [CrossRef]
- Lajin, B.; Goessler, W. Introducing dimethyl carbonate as a new eluent in HPLC-ICP-MS: Stronger elution with less carbon. J. Anal. At. Spectrom. 2021, 36, 1272–1279. [Google Scholar] [CrossRef]
- Buljan, A.; Roje, M. Application of green chiral chromatography in enantioseparation of newly syntehsized racemic marinoepoxides. Mar. Drugs 2022, 20, 530. [Google Scholar] [CrossRef] [PubMed]
- Jurin, M.; Kontrec, D.; Drazic, T.; Roje, M. Enantioseparation of syn- and anti-4,5-disubstituted hydantoins by HPLC and SFC on immobilized polysaccharides-based chiral stationary phases. Separations 2022, 9, 157. [Google Scholar] [CrossRef]
- Propylene Carbonate Technical Data. Available online: https://www.lyondellbasell.com/globalassets/documents/chemicals-technical-literature/lyondellbasell-chemicals-technicalliterature-propylene-carbonate-2417.pdf (accessed on 11 January 2023).
- Alpert, J. Hydrophilic interaction chromatography for the separation of peptides, nucleic acids and other polar compounds. J. Chrom. 1990, 499, 177–196. [Google Scholar] [CrossRef]
- Grumbach, E.S.; Wagrowski-Diehl, D.M.; Mazzeo, J.R.; Alden, B.; Iraneta, P.C. Hydrophilic interaction chromatography using silica columns for the retention of polar analytes and enhanced ESI-MS sensitivity. LC GC N. Am. 2004, 22, 1010–1023. [Google Scholar]
- Buszewski, B.L.; Noga, S. Hydrophilic interaction liquid chromatography (HILIC)-a powerful separation technique. Anal. Bioanal. Chem. 2012, 402, 231–247. [Google Scholar] [CrossRef]
- Foley, J.P.; Dorsey, J.G. Equations for Calculation of Chromatographic Figures of Merit for Ideal and Skewed Peaks. Anal. Chem. 1983, 35, 730–737. [Google Scholar] [CrossRef]
- Ma, J.; Zhang, W.; Lu, S.; Yu, R. Determination of purity and components in dimethyl carbonate by gas chromatography. Anal. Lett. 2004, 37, 2557–2563. [Google Scholar] [CrossRef]
- Hao, Z.; Xiao, B.; Weng, N. Impact of column temperature and mobile phase components on selectivity of hydrophilic interaction liquid chromatography (HILIC). J. Sep. Sci. 2008, 31, 1449–1464. [Google Scholar] [CrossRef] [PubMed]
- Burgos-Gil, R.; Peris-Garcia, E.; Ruiz-Angel, M.J.; Baeza-Baeza, J.J.; Garcia-Alvarez-Coque, M.C. Protocol to compare column performance applied to hydrophilic interaction liquid chromatography. Microchem. J. 2019, 149, 103973. [Google Scholar] [CrossRef]
- Chen, T.; Zhu, L.; Lu, H.; Song, G.; Li, Y.; Zhou, H.; Li, P.; Zhu, W.; Xu, H.; Shao, L. Preparation and application of covalently bonded polysaccharide modified stationary phase for per aqueous liquid chromatography. Anal. Chim. Acta 2017, 964, 195–202. [Google Scholar] [CrossRef]
- Kannaiah, K.P.; Sugumaran, A.; Chanduluru, H.K.; Rathinam, S. Environmental impact of greenness assessment tools in liquid chromatography—A review. Microchem. J. 2021, 170, 106685. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Wojnowski, W.; Tobiszewski, M. AGREE Analytical GREEnness Metric Approach and Software. Anal. Chem. 2020, 92, 10076–10082. [Google Scholar] [CrossRef]
- Chen, K.; Lynen, F.; De Beer, M.; Hitzel, L.; Ferguson, P.; Hanna-Brown, M.; Sandra, P. Selectivity optimization in green chromatography by gradient stationary phase optimized selectivity liquid chromatography, J. Chromatogr. A. 2010, 1217, 7222–7230. [Google Scholar] [CrossRef] [PubMed]
- Treadway, J.W.; Philibert, G.S.; Olesik, S.V. Enhanced fluidity liquid chromatography for hydrophilic interaction separation of nucleosides. J. Chromatogr. A. 2011, 1218, 5897–5902. [Google Scholar] [CrossRef] [PubMed]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boes, P.D.; Elleman, S.R.; Danielson, N.D. Dimethyl Carbonate as a Mobile-Phase Modifier for Normal-Phase and Hydrophilic Interaction Liquid Chromatography. Separations 2023, 10, 70. https://doi.org/10.3390/separations10020070
Boes PD, Elleman SR, Danielson ND. Dimethyl Carbonate as a Mobile-Phase Modifier for Normal-Phase and Hydrophilic Interaction Liquid Chromatography. Separations. 2023; 10(2):70. https://doi.org/10.3390/separations10020070
Chicago/Turabian StyleBoes, Philip D., Sophie R. Elleman, and Neil D. Danielson. 2023. "Dimethyl Carbonate as a Mobile-Phase Modifier for Normal-Phase and Hydrophilic Interaction Liquid Chromatography" Separations 10, no. 2: 70. https://doi.org/10.3390/separations10020070
APA StyleBoes, P. D., Elleman, S. R., & Danielson, N. D. (2023). Dimethyl Carbonate as a Mobile-Phase Modifier for Normal-Phase and Hydrophilic Interaction Liquid Chromatography. Separations, 10(2), 70. https://doi.org/10.3390/separations10020070







