Analysis of the Effects of Process Parameters on Start-Up Operation in Continuous Wet Granulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Manufacturing Methods
2.3. Characterization Methods
2.4. Assessment Methods
2.4.1. Torque Profile
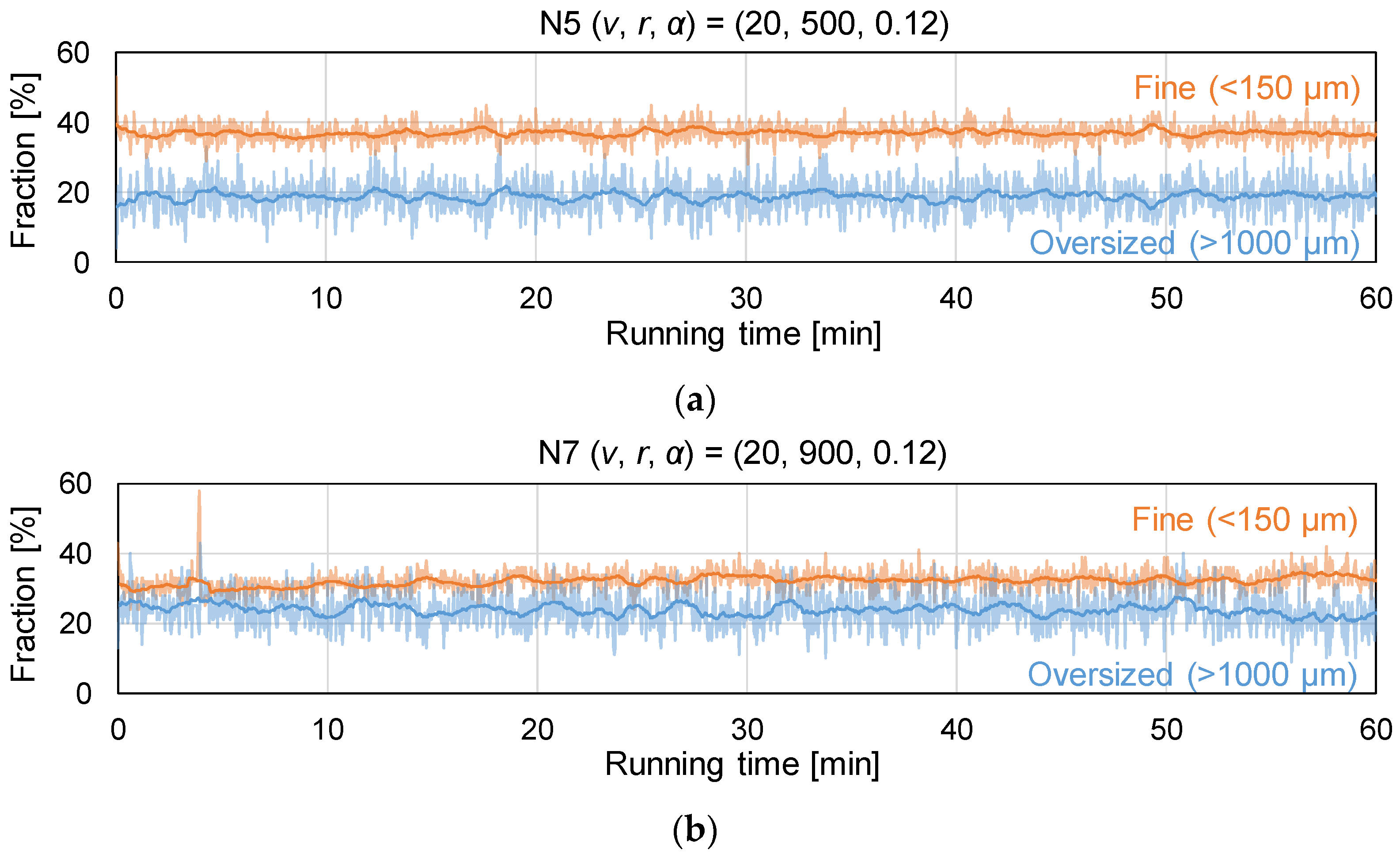
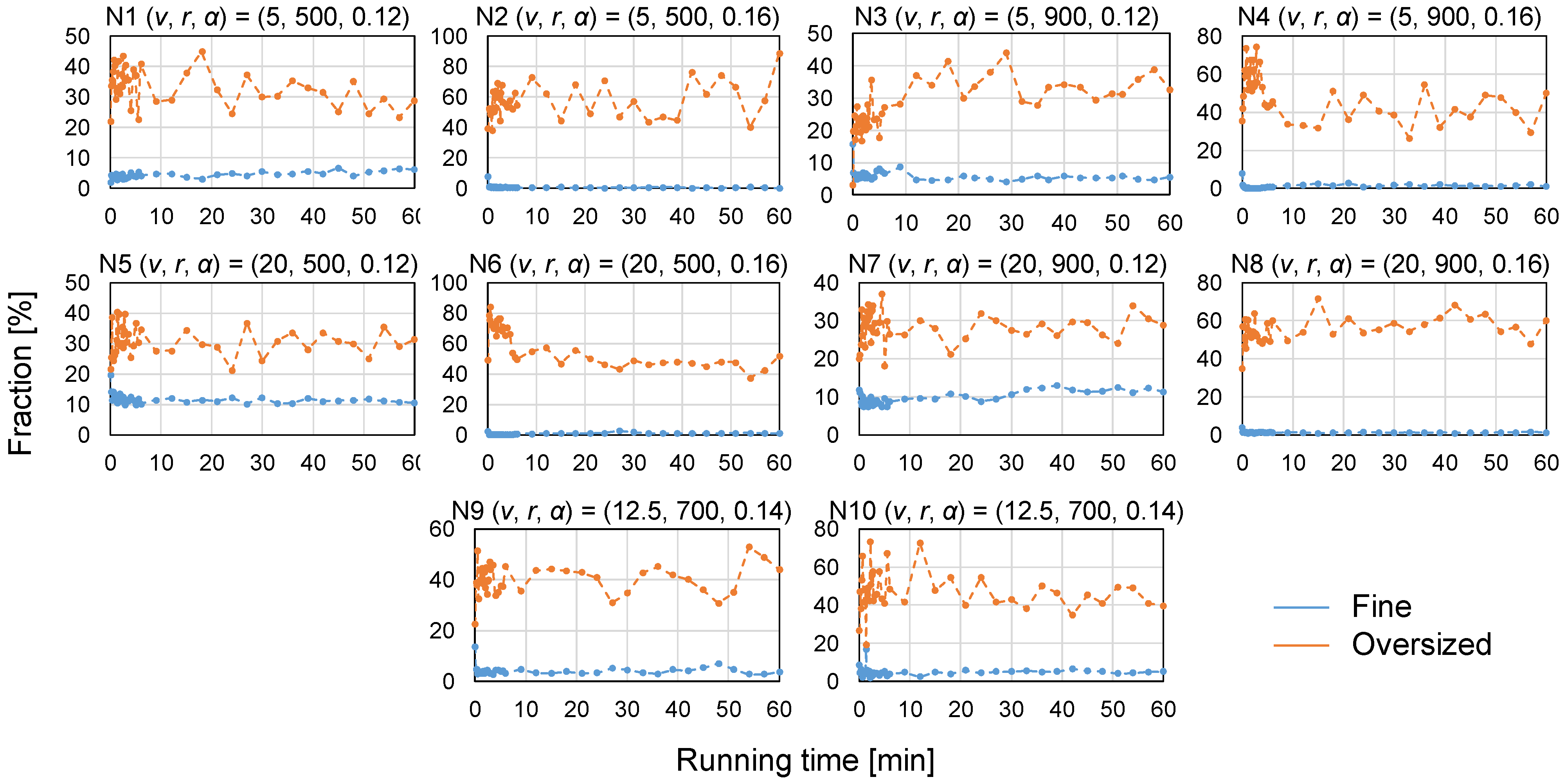
2.4.2. Granule Size Distribution
- Visual comparison.
- Tenth-percentile, median, and ninth-percentile diameter.
- Fractions of fines and oversized granules, or granulation yield.
- Fitting by probability distributions, e.g., Gaussian distribution.
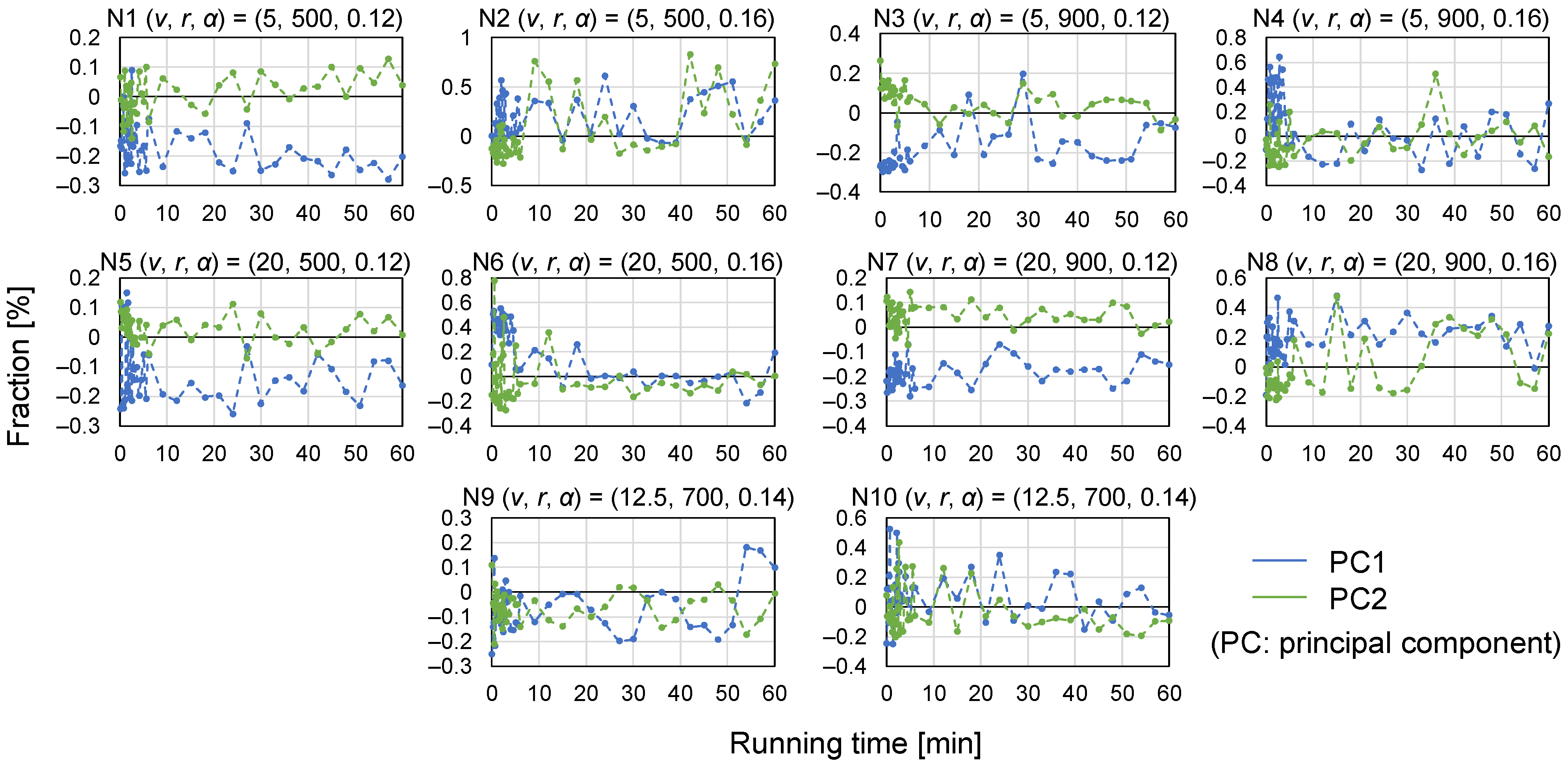
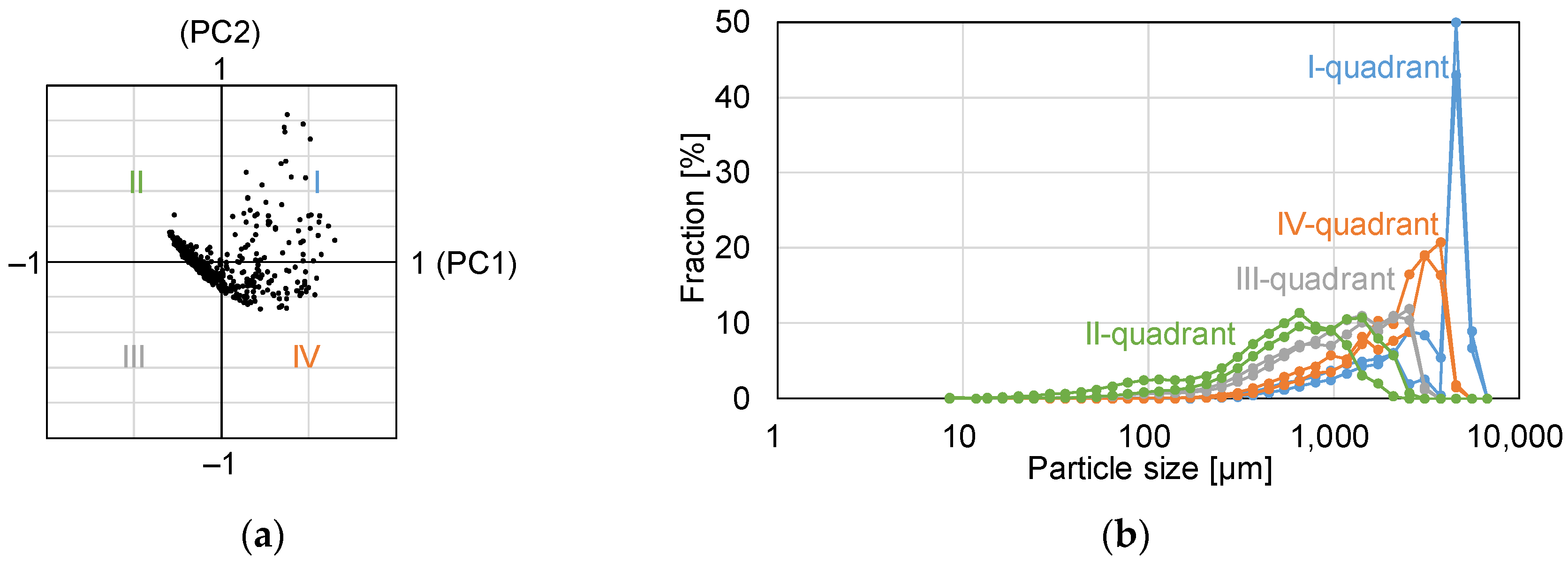
- Multivariate analysis.
2.4.3. Cost Analysis
3. Results and Discussion
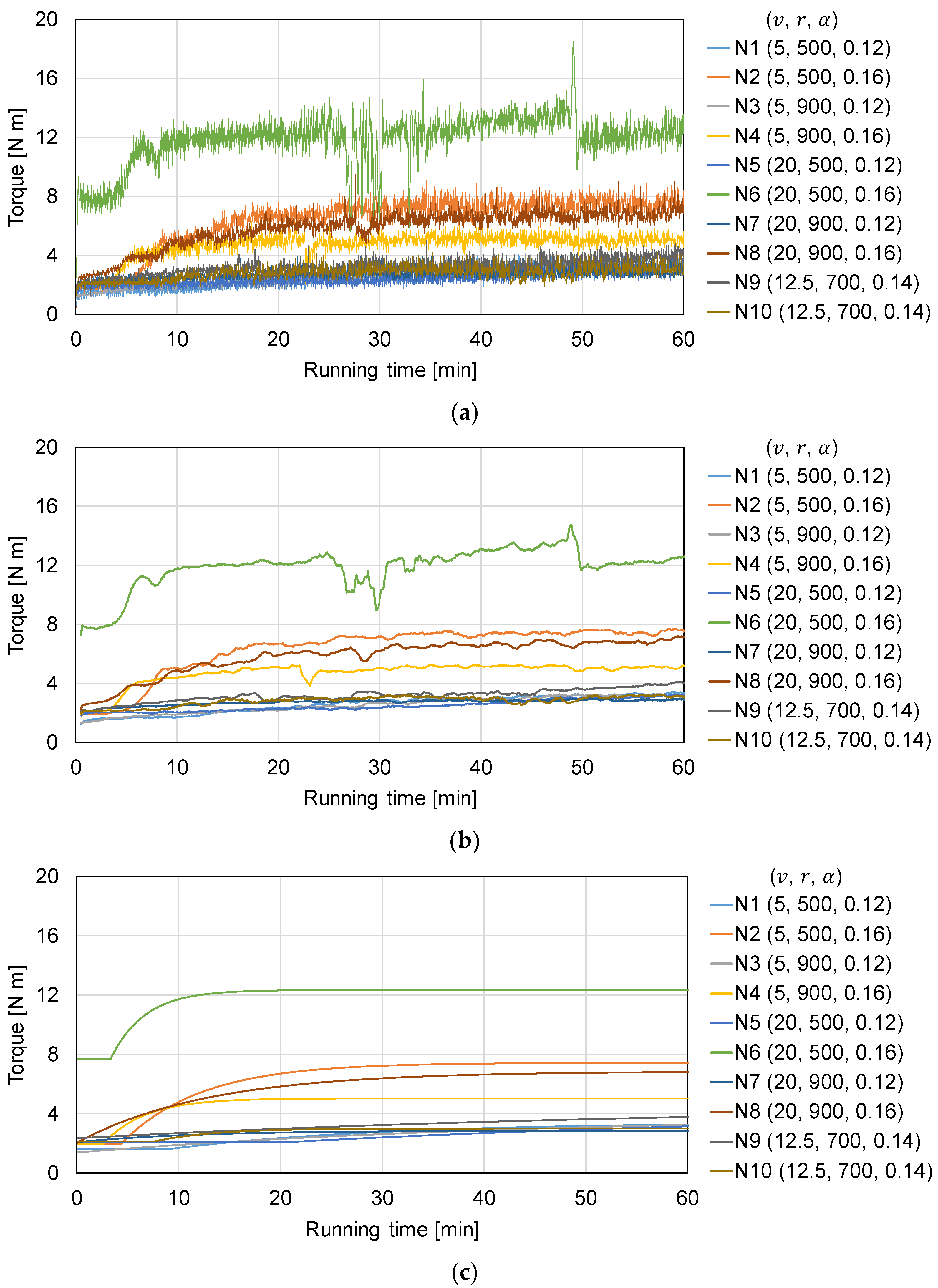
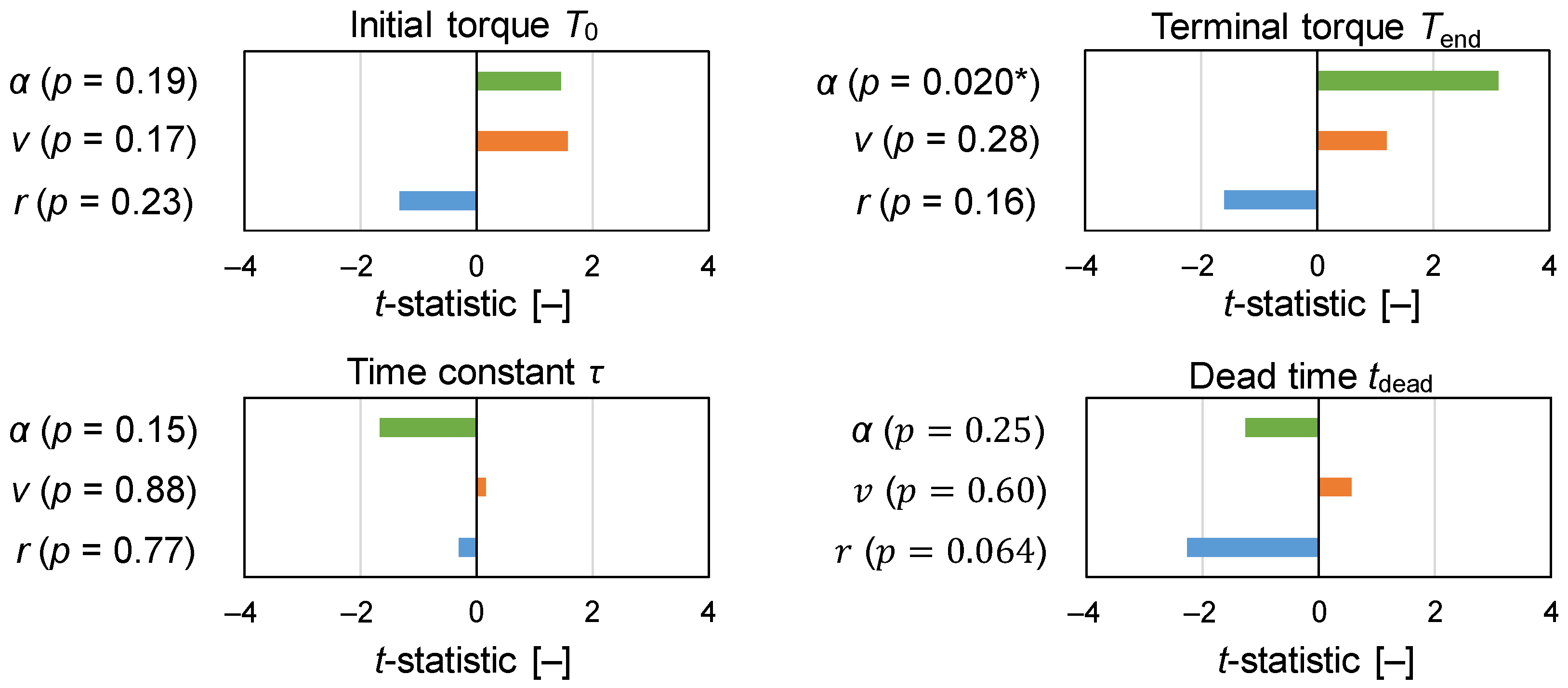
3.1. Assessment of Torque Profile
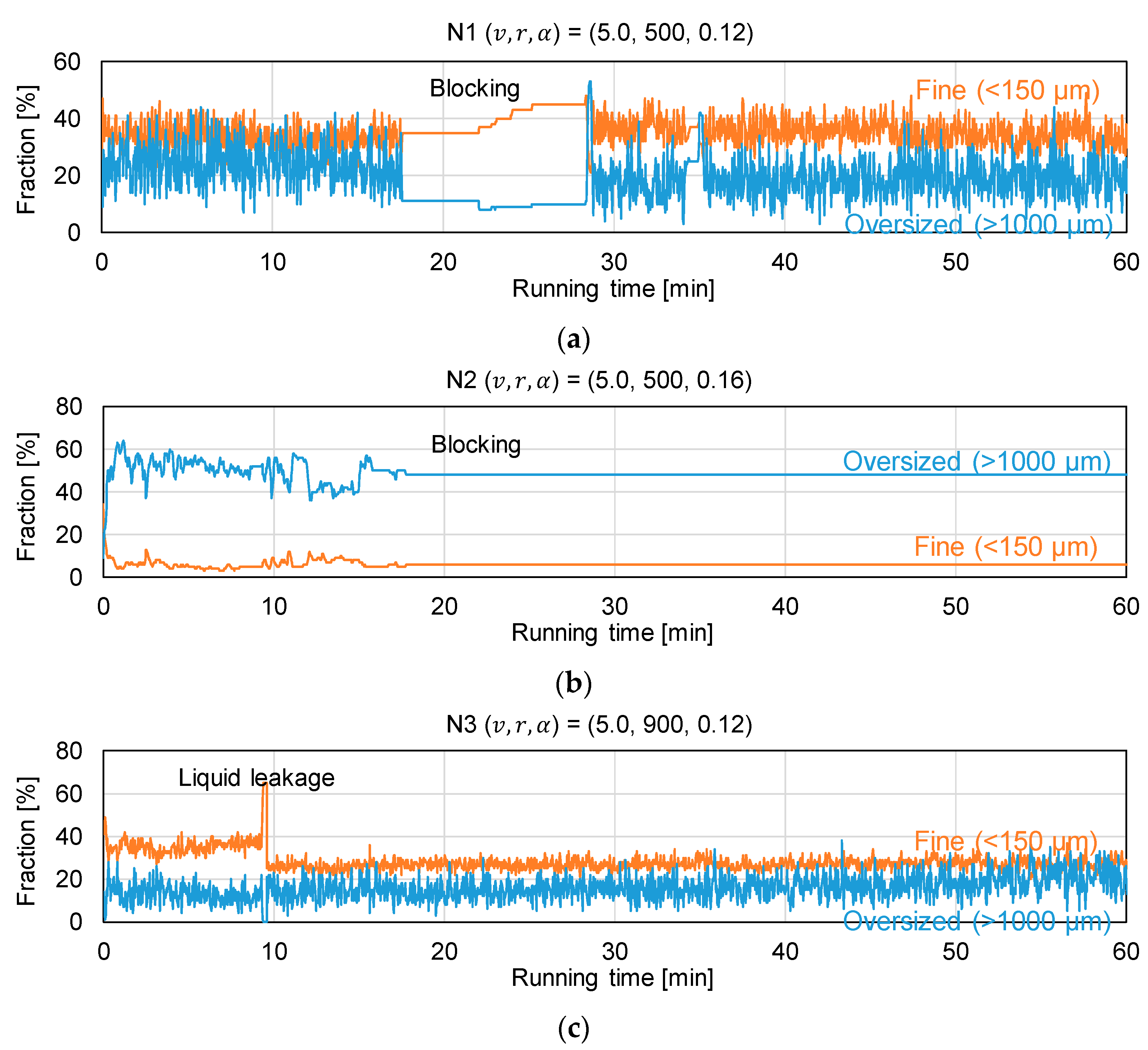
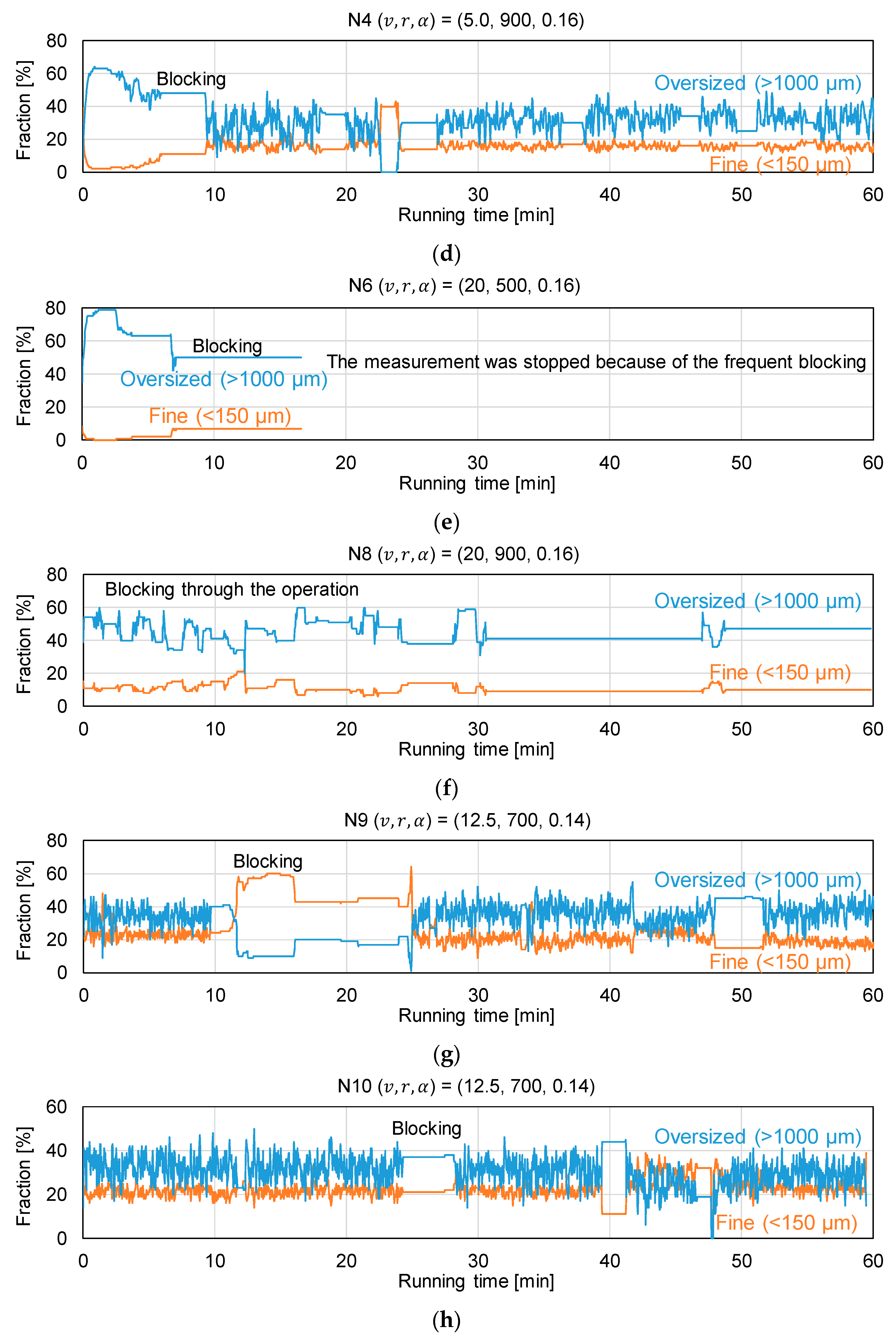
3.2. Assessment of Granule Size Distribution
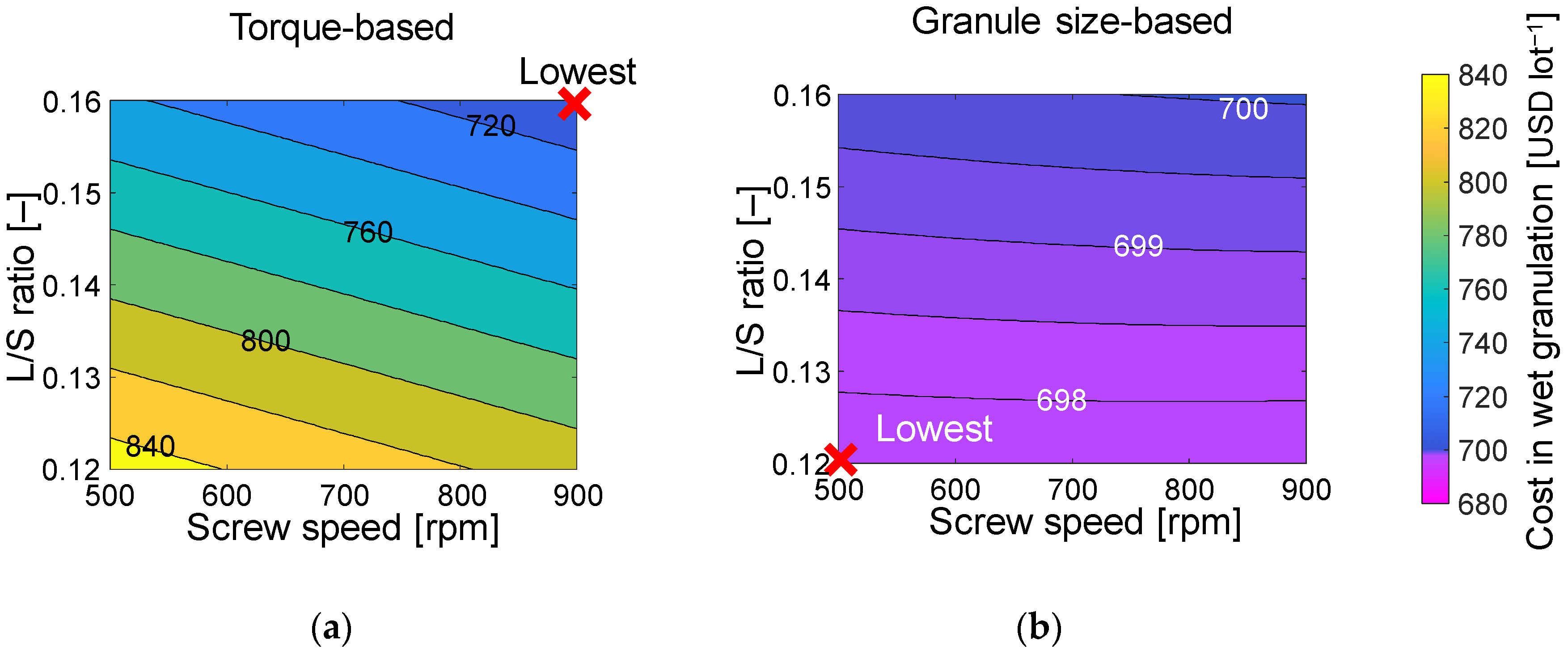
3.3. Economic Assessment with Different Start-Up Criteria
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| API | active pharmaceutical ingredient |
| DoE | design of experiments |
| HPMC | hydroxypropylmethylcellulose |
| L/D | length-to-diameter |
| L/S | liquid-to-solid |
| PAT | process analytical technology |
| PC | principal component |
| PCA | principal component analysis |
| RBF | radial basis function |
| RMSE | root mean square error |
| Nomenclature | |
| Variable | Description |
| cest | estimated coefficient for each parameter |
| Celec | electricity price [USD KWh−1] |
| Cenergy | energy cost [USD lot−1] |
| Cmaterial | raw material cost [USD lot–1] |
| CRM | unit price of raw materials [USD kg−1] |
| CWG | cost of wet granulation [USD lot−1] |
| mlot | lot size [kg lot−1] |
| r | screw speed [rpm] |
| SE | standard error of the coefficient cest |
| t | running time [min] |
| T | torque [N m] |
| tdead | dead time [min] |
| Tend | terminal torque [N m] |
| tstart | time needed for a start-up operation [h lot−1] |
| tstat | t-statistic [–] |
| T0 | initial torque [N m] |
| v | manufacturing rate [kg h−1] |
| α | liquid-to-solid ratio [–] |
| γ | hyperparameter in RBFs |
| σ | hyperparameter in RBFs |
| τ | time constant [min] |
| φ | RBFs |
Appendix A
References
- Lee, S.L.; O’Connor, T.F.; Yang, X.; Cruz, C.N.; Chatterjee, S.; Madurawe, R.D.; Moore, C.M.V.; Yu, L.X.; Woodcock, J. Modernizing pharmaceutical manufacturing: From batch to continuous production. J. Pharm. Innov. 2015, 10, 191–199. [Google Scholar] [CrossRef] [Green Version]
- Teżyk, M.; Milanowski, B.; Ernst, A.; Lulek, J. Recent progress in continuous and semi-continuous processing of solid oral dosage forms: A review. Drug Dev. Ind. Pharm. 2016, 42, 1195–1214. [Google Scholar] [CrossRef]
- Domokos, A.; Nagy, B.; Szilágyi, B.; Marosi, G.; Nagy, Z.K. Integrated Continuous Pharmaceutical Technologies—A Review. Org. Process Res. Dev. 2021, 25, 721–739. [Google Scholar] [CrossRef]
- Ierapetritou, M.; Muzzio, F.; Reklaitis, G. Perspectives on the continuous manufacturing of powder-based pharmaceutical processes. AIChE J. 2016, 62, 1846–1862. [Google Scholar] [CrossRef]
- Vanhoorne, V.; Vervaet, C. Recent progress in continuous manufacturing of oral solid dosage forms. Int. J. Pharm. 2020, 579, 119194. [Google Scholar] [CrossRef]
- Kumar, A.; Gernaey, K.V.; De Beer, T.; Nopens, I. Model-based analysis of high shear wet granulation from batch to continuous processes in pharmaceutical production—A critical review. Eur. J. Pharm. Biopharm. 2013, 85, 814–832. [Google Scholar] [CrossRef] [Green Version]
- Portier, C.; Vervaet, C.; Vanhoorne, V. Continuous Twin Screw Granulation: A Review of Recent Progress and Opportunities in Formulation and Equipment Design. Pharmaceutics 2021, 13, 668. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Hwang, K.M.; Cho, C.H.; Nguyen, T.T.; Seok, S.H.; Hwang, K.M.; Kim, J.Y.; Park, C.W.; Rhee, Y.S.; Park, E.S. Application of continuous twin screw granulation for the metformin hydrochloride extended release formulation. Int. J. Pharm. 2017, 529, 410–422. [Google Scholar] [CrossRef]
- Kumar, A.; Dhondt, J.; Vercruysse, J.; De Leersnyder, F.; Vanhoorne, V.; Vervaet, C.; Remon, J.P.; Gernaey, K.V.; De Beer, T.; Nopens, I. Development of a process map: A step towards a regime map for steady-state high shear wet twin screw granulation. Powder Technol. 2016, 300, 73–82. [Google Scholar] [CrossRef]
- Liu, H.; Galbraith, S.C.; Ricart, B.; Stanton, C.; Smith-Goettler, B.; Verdi, L.; O’Connor, T.; Lee, S.; Yoon, S. Optimization of critical quality attributes in continuous twin-screw wet granulation via design space validated with pilot scale experimental data. Int. J. Pharm. 2017, 525, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Ryckaert, A.; Stauffer, F.; Funke, A.; Djuric, D.; Vanhoorne, V.; Vervaet, C.; De Beer, T. Evaluation of torque as an in-process control for granule size during twin-screw wet granulation. Int. J. Pharm. 2021, 602, 120642. [Google Scholar] [CrossRef]
- Pawar, P.; Clancy, D.; Gorringe, L.; Barlow, S.; Hesketh, A.; Elkes, R. Development and Scale-Up of Diversion Strategy for Twin Screw Granulation in Continuous Manufacturing. J. Pharm. Sci. 2020, 109, 3439–3450. [Google Scholar] [CrossRef]
- Kumar, A.; Alakarjula, M.; Vanhoorne, V.; Toiviainen, M.; De Leersnyder, F.; Vercruysse, J.; Juuti, M.; Ketolainen, J.; Vervaet, C.; Remon, J.P.; et al. Linking granulation performance with residence time and granulation liquid distributions in twin-screw granulation: An experimental investigation. Eur. J. Pharm. Sci. 2016, 90, 25–37. [Google Scholar] [CrossRef]
- Meng, W.; Kotamarthy, L.; Panikar, S.; Sen, M.; Pradhan, S.; Marc, M.; Litster, J.D.; Muzzio, F.J.; Ramachandran, R. Statistical analysis and comparison of a continuous high shear granulator with a twin screw granulator: Effect of process parameters on critical granule attributes and granulation mechanisms. Int. J. Pharm. 2016, 513, 357–375. [Google Scholar] [CrossRef]
- Hwang, K.M.; Cho, C.H.; Yoo, S.D.; Cha, K.I.; Park, E.S. Continuous twin screw granulation: Impact of the starting material properties and various process parameters. Powder Technol. 2019, 356, 847–857. [Google Scholar] [CrossRef]
- Vercruysse, J.; Córdoba Díaz, D.; Peeters, E.; Fonteyne, M.; Delaet, U.; Van Assche, I.; De Beer, T.; Remon, J.P.; Vervaet, C. Continuous twin screw granulation: Influence of process variables on granule and tablet quality. Eur. J. Pharm. Biopharm. 2012, 82, 205–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vanhoorne, V.; Bekaert, B.; Peeters, E.; De Beer, T.; Remon, J.P.; Vervaet, C. Improved tabletability after a polymorphic transition of delta-mannitol during twin screw granulation. Int. J. Pharm. 2016, 506, 13–24. [Google Scholar] [CrossRef] [Green Version]
- Vanhoorne, V.; Vanbillemont, B.; Vercruysse, J.; De Leersnyder, F.; Gomes, P.; De Beer, T.; Remon, J.P.; Vervaet, C. Development of a controlled release formulation by continuous twin screw granulation: Influence of process and formulation parameters. Int. J. Pharm. 2016, 505, 61–68. [Google Scholar] [CrossRef]
- Liu, H.; Ricart, B.; Stanton, C.; Smith-Goettler, B.; Verdi, L.; O’Connor, T.; Lee, S.; Yoon, S. Design space determination and process optimization in at-scale continuous twin screw wet granulation. Comput. Chem. Eng. 2019, 125, 271–286. [Google Scholar] [CrossRef]
- Meng, W.; Oka, S.; Liu, X.; Omer, T.; Ramachandran, R.; Muzzio, F.J. Effects of process and design parameters on granule size distribution in a continuous high shear granulation process. J. Pharm. Innov. 2017, 12, 283–295. [Google Scholar] [CrossRef]
- Matsunami, K.; Nagato, T.; Hasegawa, K.; Sugiyama, H. Determining key parameters of continuous wet granulation for tablet quality and productivity: A case in ethenzamide. Int. J. Pharm. 2020, 579, 119160. [Google Scholar] [CrossRef]
- Meng, W.; Román-Ospino, A.D.; Panikar, S.S.; O’Callaghan, C.; Gilliam, S.J.; Ramachandran, R.; Muzzio, F.J. Advanced process design and understanding of continuous twin-screw granulation via implementation of in-line process analytical technologies. Adv. Powder Technol. 2019, 30, 879–894. [Google Scholar] [CrossRef]
- Meng, W.; Rao, K.S.; Snee, R.D.; Ramachandran, R.; Muzzio, F.J. A comprehensive analysis and optimization of continuous twin-screw granulation processes via sequential experimentation strategy. Int. J. Pharm. 2019, 556, 349–362. [Google Scholar] [CrossRef]
- Nasr, M.M.; Krumme, M.; Matsuda, Y.; Trout, B.L.; Badman, C.; Mascia, S.; Cooney, C.L.; Jensen, K.D.; Florence, A.; Johnston, C.; et al. Regulatory perspectives on continuous pharmaceutical manufacturing: Moving from theory to practice. J. Pharm. Sci. 2017, 106, 3199–3206. [Google Scholar] [CrossRef]
- Vercruysse, J.; Delaet, U.; Van Assche, I.; Cappuyns, P.; Arata, F.; Caporicci, G.; De Beer, T.; Remon, J.P.; Vervaet, C. Stability and repeatability of a continuous twin screw granulation and drying system. Eur. J. Pharm. Biopharm. 2013, 85, 1031–1038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pauli, V.; Kleinebudde, P.; Krumme, M. Predictive model-based process start-up in pharmaceutical continuous granulation and drying. Pharmaceutics 2020, 12, 67. [Google Scholar] [CrossRef] [Green Version]
- Matsunami, K.; Nagato, T.; Hasegawa, K.; Sugiyama, H. A large-scale experimental comparison of batch and continuous technologies in pharmaceutical tablet manufacturing using ethenzamide. Int. J. Pharm. 2019, 559, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Matsunami, K.; Sternal, F.; Yaginuma, K.; Tanabe, S.; Nakagawa, H.; Sugiyama, H. Superstructure-based process synthesis and economic assessment under uncertainty for solid drug product manufacturing. BMC Chem. Eng. 2020, 2, 6. [Google Scholar] [CrossRef] [Green Version]
- Matsunami, K.; Miyano, T.; Arai, H.; Nakagawa, H.; Hirao, M.; Sugiyama, H. Decision Support Method for the Choice between Batch and Continuous Technologies in Solid Drug Product Manufacturing. Ind. Eng. Chem. Res. 2018, 57, 9798–9809. [Google Scholar] [CrossRef]
- Metta, N.; Ghijs, M.; Schäfer, E.; Kumar, A.; Cappuyns, P.; Van Assche, I.; Singh, R.; Ramachandran, R.; De Beer, T.; Ierapetritou, M.; et al. Dynamic flowsheet model development and sensitivity analysis of a continuous pharmaceutical tablet manufacturing process using the wet granulation route. Processes 2019, 7, 234. [Google Scholar] [CrossRef] [Green Version]
- Fonteyne, M.; Wickström, H.; Peeters, E.; Vercruysse, J.; Ehlers, H.; Peters, B.-H.; Remon, J.P.; Vervaet, C.; Ketolainen, J.; Sandler, N.; et al. Influence of raw material properties upon critical quality attributes of continuously produced granules and tablets. Eur. J. Pharm. Biopharm. 2014, 87, 252–263. [Google Scholar] [CrossRef]
- Verstraeten, M.; Van Hauwermeiren, D.; Lee, K.; Turnbull, N.; Wilsdon, D.; am Ende, M.; Doshi, P.; Vervaet, C.; Brouckaert, D.; Mortier, S.T.F.C.; et al. In-depth experimental analysis of pharmaceutical twin-screw wet granulation in view of detailed process understanding. Int. J. Pharm. 2017, 529, 678–693. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Thompson, M.R.; O’Donnell, K.P. Examining drug hydrophobicity in continuous wet granulation within a twin screw extruder. Int. J. Pharm. 2015, 496, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Ge Wang, L.; Morrissey, J.P.; Barrasso, D.; Slade, D.; Clifford, S.; Reynolds, G.; Ooi, J.Y.; Litster, J.D. Model driven design for twin screw granulation using mechanistic-based population balance model. Int. J. Pharm. 2021, 120939. [Google Scholar] [CrossRef]
- Petrak, D. Simultaneous measurement of particle size and particle velocity by the spatial filtering technique. Part. Part. Syst. Charact. 2002, 19, 391–400. [Google Scholar] [CrossRef]
- Silva, A.F.T.; Burggraeve, A.; Denon, Q.; Van Der Meeren, P.; Sandler, N.; Van Den Kerkhof, T.; Hellings, M.; Vervaet, C.; Remon, J.P.; Lopes, J.A.; et al. Particle sizing measurements in pharmaceutical applications: Comparison of in-process methods versus off-line methods. Eur. J. Pharm. Biopharm. 2013, 85, 1006–1018. [Google Scholar] [CrossRef]
- Van Hauwermeiren, D.; Stock, M.; De Beer, T.; Nopens, I. Predicting pharmaceutical particle size distributions using kernel mean embedding. Pharmaceutics 2020, 12, 271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Hauwermeiren, D. On the Simulation of Particle Size Distributions in Continuous Pharmaceutical Wet Granulation. Ph.D. Thesis, Universiteit Gent, Ghent, Belgium, 2020. [Google Scholar]
- Arthur, G.; Herbrich, R.; Smola, A.; Bousquet, O.; Bernhard, S. Kernel methods for measuring independence. J. Mach. Learn. Res. 2005, 6, 2075–2129. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar] [CrossRef]
- Leuenberger, H. New trends in the production of pharmaceutical granules: The classical batch concept and the problem of scale-up. Eur. J. Pharm. Biopharm. 2001, 52, 279–288. [Google Scholar] [CrossRef]
- Matsui, Y.; Ando, Y.; Murakami, T.; Watano, S. Prediction of pharmaceutical product properties by compression test of wet granulated masses. J. Soc. Powder Technol. Japan 2019, 56, 328–338. [Google Scholar] [CrossRef]
- Iveson, S.M.; Litster, J.D.; Hapgood, K.; Ennis, B.J. Nucleation, growth and breakage phenomena in agitated wet granulation processes: A review. Powder Technol. 2001, 117, 3–39. [Google Scholar] [CrossRef]
- Gentzler, M.; Michaels, J.N.; Tardos, G.I. Quantification of segregation potential for polydisperse, cohesive, multi-component powders and prediction of tablet die-filling performance—A methodology for practical testing, re-formulation and process design. Powder Technol. 2015, 285, 96–102. [Google Scholar] [CrossRef]
- Liss, E.D.; Conway, S.L.; Zega, J.A.; Glasser, B.J. Segregation of powders during gravity flow through vertical pipes. Pharm. Technol. 2004, 28, 78–96. [Google Scholar]
- Kotamarthy, L.; Ramachandran, R. Mechanistic understanding of the effects of process and design parameters on the mixing dynamics in continuous twin-screw granulation. Powder Technol. 2021, 390, 73–85. [Google Scholar] [CrossRef]
- Ouranidis, A.; Gkampelis, N.; Vardaka, E.; Karagianni, A.; Tsiptsios, D.; Nikolakakis, I.; Kachrimanis, K. Overcoming the solubility barrier of ibuprofen by the rational process design of a nanocrystal formulation. Pharmaceutics 2020, 12, 969. [Google Scholar] [CrossRef] [PubMed]
| Component | Substance | Composition [% (w/w)] |
|---|---|---|
| API | Theophylline | 5.0 |
| Filler | α-lactose monohydrate | 65.0 |
| Filler | Microcrystalline cellulose | 15.0 |
| Binder | Hydroxypropylmethylcellulose | 15.0 |
| Experiment | Manufacturing Rate v [kg h–1] | Screw Speed r [rpm] | L/S Ratio α [–] |
|---|---|---|---|
| N1 | 5.0 | 500 | 0.12 |
| N2 | 5.0 | 500 | 0.16 |
| N3 | 5.0 | 900 | 0.12 |
| N4 | 5.0 | 900 | 0.16 |
| N5 | 20.0 | 500 | 0.12 |
| N6 | 20.0 | 500 | 0.16 |
| N7 | 20.0 | 900 | 0.12 |
| N8 | 20.0 | 900 | 0.16 |
| N9 | 12.5 | 700 | 0.14 |
| N10 | 12.5 | 700 | 0.14 |
| Experiment | T0 [N m] | Tend [N m] | τ [min] | tdead [min] | RMSE |
|---|---|---|---|---|---|
| N1 | 1.61 | 3.43 | 20.7 | 8.84 | 0.0775 |
| N2 | 1.95 | 7.44 | 7.80 | 4.30 | 0.1765 |
| N3 | 1.40 | 3.91 | 44.1 | 2.89 × 10−14 | 0.0793 |
| N4 | 1.99 | 5.04 | 4.08 | 2.62 | 0.1776 |
| N5 | 2.10 | 4.30 | 60.0 | 20.8 | 0.0900 |
| N6 | 7.69 | 12.3 | 3.34 | 3.31 | 0.7117 |
| N7 | 2.09 | 2.86 | 11.0 | 1.05 × 10−15 | 0.0407 |
| N8 | 2.04 | 6.86 | 12.8 | 7.47 × 10−15 | 0.1823 |
| N9 | 2.36 | 4.62 | 60.0 | 1.75 × 10−13 | 0.1643 |
| N10 | 2.15 | 3.01 | 5.70 | 7.68 | 0.1312 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsunami, K.; Ryckaert, A.; Peeters, M.; Badr, S.; Sugiyama, H.; Nopens, I.; De Beer, T. Analysis of the Effects of Process Parameters on Start-Up Operation in Continuous Wet Granulation. Processes 2021, 9, 1502. https://doi.org/10.3390/pr9091502
Matsunami K, Ryckaert A, Peeters M, Badr S, Sugiyama H, Nopens I, De Beer T. Analysis of the Effects of Process Parameters on Start-Up Operation in Continuous Wet Granulation. Processes. 2021; 9(9):1502. https://doi.org/10.3390/pr9091502
Chicago/Turabian StyleMatsunami, Kensaku, Alexander Ryckaert, Michiel Peeters, Sara Badr, Hirokazu Sugiyama, Ingmar Nopens, and Thomas De Beer. 2021. "Analysis of the Effects of Process Parameters on Start-Up Operation in Continuous Wet Granulation" Processes 9, no. 9: 1502. https://doi.org/10.3390/pr9091502
APA StyleMatsunami, K., Ryckaert, A., Peeters, M., Badr, S., Sugiyama, H., Nopens, I., & De Beer, T. (2021). Analysis of the Effects of Process Parameters on Start-Up Operation in Continuous Wet Granulation. Processes, 9(9), 1502. https://doi.org/10.3390/pr9091502










