1. Introduction
A sudden pressure drop in fluidic systems, such as centrifugal pumps, hydro turbines, and diesel engine injectors, leads to local pressures lower than the saturation vapor pressure of the working fluid, which triggers the phase change phenomenon known as cavitation. The destructive effects on the turbomachinery as a result of cavitating flows, as well as methods for prevention of cavitation, have attracted much attention. Numerical and experimental methods have aided the researchers for these purposes. For instance, Deng et al. [
1] developed a numerical model to characterize cavitation in centrifugal pump impellers in transporting water and diesel. They could include the effects of viscosity and surface tension in their proposed model. In another study, Escaler et al. [
2] studied cavitating flows in hydro turbines and its negative instability effect on the performance due to the draft tube swirl. They also presented different types of cavitation, namely leading edge cavitation, traveling bubble cavitation, draft tube swirl, inter-blade vortex cavitation, and Von Karman vortex cavitation. The above-mentioned studies proved the destructive potential of hydrodynamic cavitation, which could be utilized in different applications as well.
The advances in small scale bubble generation and the confirmation of the massive energy release upon bubble collapse caused a paradigm shift. Many researchers have already started to exploit the energy released upon the collapse of cavitation bubbles in different applications such as energy harvesting [
3,
4], water treatment [
5,
6], biological applications such as urinary stone treatment [
7], food industry [
8], and heat transfer [
9,
10].
According to the thermodynamic phase diagram [
11], phase change phenomena happen as a result of temperature increase (boiling) or pressure drop (cavitation) in fluidic systems. Sudden pressure drop happens as the fluid experiences a sudden decrease in the cross-sectional area of the channel due to the existence of a small flow restrictive element such as orifice [
12] or venturi [
13], which could lead to hydrodynamic cavitation, while the propagation of acoustic waves in the bulk of the fluid forming low-pressure nodes [
14] could trigger acoustic cavitation.
In hydrodynamic cavitation, different parameters such as thermophysical properties of the working fluid, geometrical dimensions, and surface roughness alter the flow behavior in fluidic systems. The generation of cavitating flows in microfluidic devices has been receiving increasing attention. As a result of research efforts in this emerging field, there are already parametric studies in the literature. For instance, Ghorbani et al. [
15] studied the effect of various lateral wall roughness element geometries in micro orifices fabricated on silicon. They could show that the equilateral triangle roughness elements increased the intensity of cavitating flows and assisted the development of twin cavities along the channel. In this study, they concluded that small roughness elements and shorter total length raised the intensity. In another study, Carpenter et al. [
16] studied the effect of different geometrical cavitation generators on the emulsification of oil in water. They fabricated seven different geometries, including both orifice plate and venturi pipe, and could prove the direct dependence of geometry on the size of generated droplets. The generated nanoemulsion of mustard oil in water was significantly stable after their experiments under thermal stress and centrifugal condition. They could also demonstrate that hydrodynamic cavitation was 11 times more energy-efficient compared to ultrasonication. The thermophysical properties of the working fluid also play a crucial role. Hence, many studies concentrated on these parameters [
17,
18]. Since the generation of hydrodynamic cavitating flows in microfluidic devices is rather cheap, and this type of reactors are easy to scale up, they have received considerable attention in the literature. The stationary sections of these reactors and their easy fabrication process flows increase the popularity and efficiency of these reactors.
All the above-mentioned studies would have a larger impact if they could be implemented in industrial or clinical applications. For instance, Gevari et al. [
3] utilized intensified cavitating flows by means of Perfluoropentane (PFC5) droplet addition to the working fluid to harvest more energy from the heat generation upon bubble collapse via micro thermoelectric generators. Dular et al. [
6] used the energy of collapsing bubbles for water and wastewater treatment. They could demonstrate the effectiveness of cavitating flows on removal and deactivation of microorganisms available in water sources. They could successfully remove various organisms such as bacteria, viruses, pharmaceutical pollutions, and algae from the water. Various studies on water treatment application with hydrodynamic cavitation have manifested the importance of finding a cheap and easy method to remove microorganisms from water, such as bacteria [
19,
20].
Salmonella enterica subsp. enterica, serovar typhimurium (S. typhimurium) is a Gram-negative bacterium, which abundantly exists in the intestines of humans and mammals.
S. typhimurium causes toxic syndromes through the intake of contaminated water or food products such as fruit juice, milk, yogurt, cheese, and salad [
21,
22]. The origin of the toxication is lipopolysaccharides (LPS) located on bacterial membranes. LPSs are composed of Lipid A and oligosaccharide antigen units (O-antigens and Core-antigens), which constitute almost 75% of the bacterial membrane surface. O-antigens are positioned at the edge of the LPS units, and they are the principal target of antibodies produced by immune system cells. LPSs contribute to the conformational change of the bacterial surface and can make the bacteria unrecognizable by the immune system cells, and eventually, the biological system becomes infected by the bacteria. The existence of bacteria in main water or food sources might cause severe health problems if not treated effectively and continuously. Developing countries suffer from a lack of clean water sources [
23] due to the increasing growth of the population and the process of urbanization. As old pipelines running to open channels of drain water are highly susceptible to contamination, water quickly becomes contaminated at gathering parts in most of the cases [
24]. Some processes involved in water treatment are physical and chemical processes, including filtration, slow sand filter (SSF), and cavitation.
Regarding the cavitation method, numerous new-fangled systems were considered. To set an example, Panda et al. [
25] provided a liquid whistle hydrodynamic cavitation reactor (LWHCR) for the treatment of dicofol, known as a hazardous pollutant due to its troublemaking endocrine features. In the abovementioned study, major parameters, including the initial concentration of the pollutant, pH, and temperature, were studied. Following this, Parthasarathy et al. [
26] explored LWHCR as a system for producing submicron emulsions using the hydrodynamic-assisted emulsification process. Initial pressure and distance between the blade and orifice plate were the parameters examined in this study. In another study, Tang et al. [
27] improved high-pressure LWHCR for the generation of highly stable water-in-oil-in-water emulsions for the controlled release of ferrous fumarate extensively used for the treatment of a common type of anemia, namely, iron deficiency.
In this study, hydrodynamic cavitation is achieved as a result of a sudden decrease in the cross-sectional area of the fluid path, which gives rise to a sudden increase in the velocity of the working fluid. Three microfluidic devices are fabricated using conventional microfabrication methods on silicon and are bonded to the glass. The microfluidic devices withstand high-pressures (up to 8.30 MPa). First, the effect of the thermophysical properties of the working fluid (using deionized water and phosphate-buffered saline (PBS)) is studied in the microfluidic devices, and the flow behavior is compared for the case of water and PBS. Cavitation number, Reynolds number, cavitation flow rate, and tensile strength of the working fluid are parameters to analyze the flow behavior. The presence of bacteria, i.e., S. typhimurium acts as a solid interface in the working fluid to enhance heterogeneous bubble nucleation. In the last section of this study, a suspension of this bacteria in PBS is prepared and is used as the working fluid. Cavitating flows are visualized in the presence of the bacteria. Finally, the deactivation performance of the device is assessed on one of the microfluidic devices.
As a result, this study focuses on hydrodynamic cavitation from two perspectives: first, the effects of the thermophysical properties of working fluids on cavitating flows, and second, the effect of energy release upon bubble collapse on the activity of bacteria. The high deactivation efficiency of the proposed device is also presented and discussed.
2. Materials and Methods
The microfluidic devices used in the experiments housed micro orifices etched in silicon and covered with glass. The designed and assembled experimental setup in this study introduced the working fluid to the system at an inlet pressure, and the high-speed camera recorded flow patterns during the experiments. Pressure sensors were also installed to measure the pressures at three locations (inlet, nozzle entrance, and extension). The system performance with different working fluids, namely, water, PBS, and suspension of PBS/bacteria, were obtained and compared.
The effect of the presence of S. typhimurium bacteria on cavitating flow patterns and deactivation effect of collapsing bubbles (on the Salmonella typhimurium bacteria suspended in sterile phosphate-buffered saline (PBS)) were assessed in this study. The scanning electron microscopy (SEM) method was also implemented to monitor the physical morphology of the bacteria.
2.1. Microfluidic Devices Geometry
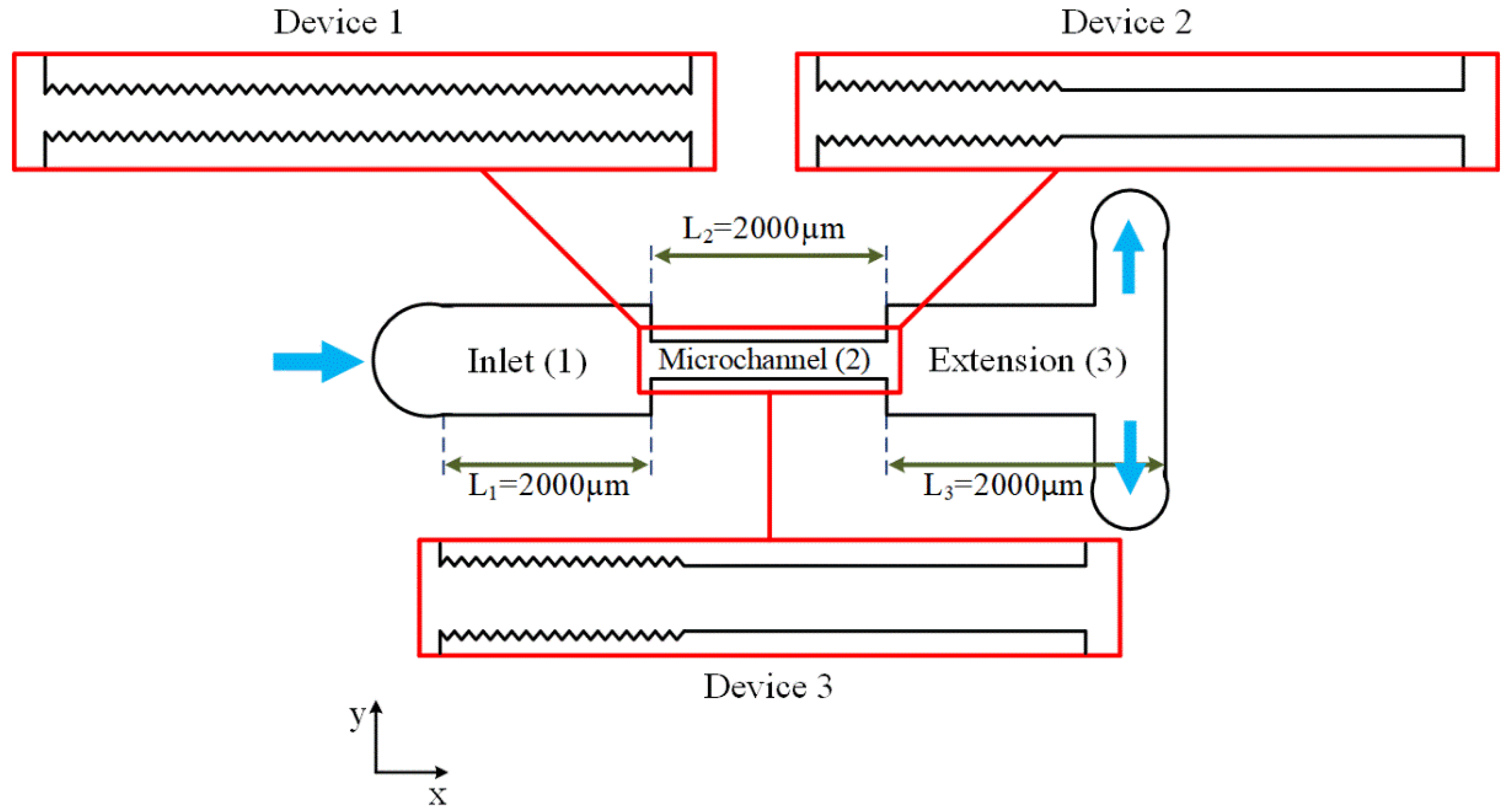
Three microfluidic devices with different geometries were employed in the experiments to study cavitating flows. As shown in
Figure 1, the microfluidic devices consisted of three main sections, namely, inlet, microchannel, and extension. The length of all sections was 2000 µm, while the width of the inlet and extension were 900 µm. Three narrow channels connected to the inlet, microchannel, and extension sections of the microfluidic devices acted as pressure ports to measure the static pressure of the working fluid at these points. According to our previous studies, engineered lateral roughness elements (equilateral triangles) were included on the microchannel walls to intensify cavitating flows in the microfluidic devices [
15]. The geometry of the roughness elements was studied, and it was shown that smaller wall roughness elements, in terms of height, and shorter elements, in terms of total length, resulted in both earlier inception of cavitating flows in the microchannel section and easier formation of twin cavities along the microchannel [
15]. The small roughness elements enhanced the heterogeneous nucleation of bubbles in the microfluidic devices [
28]. It was also demonstrated that the narrower microchannel section leads to more intense cavitating flows and lower energy requirement to reach the developed cavitating flow regime [
15].
In the first part of this study, the performance of the microfluidic devices (with water and PBS as the working fluids) were studied separately, and the suitable device for the second section was chosen based on the experimental results.
In the second part of this study, the effects of bacteria presence in PBS on the flow patterns, as well as the effects of cavitation bubble collapse on bacteria deactivation, were investigated. Since the bacteria agglomerated at the inlet of the microchannel and clogged the device, the geometry of the microfluidic device was different in this part. In addition, in order to have more intense cavitating flows, the width of the microchannel section should be increased. On the other hand, as stated above, the shorter total length of the roughness elements leads to a lower energy requirement of the system, which increases the working efficiency of the whole device. As a result, the microfluidic devices used in this section were 152 and 400 µm in width and 50 µm in depth, resulting in hydraulic diameters of 75.24 and 88.8 µm, respectively. The height of the roughness elements was 1.5 and 3 µm for the second and third devices, respectively, covering one-third of the microchannel.
Figure 1 and
Table 1 include the dimensions and configurations of all the three devices used in this study.
2.2. Microfluidic Device Fabrication
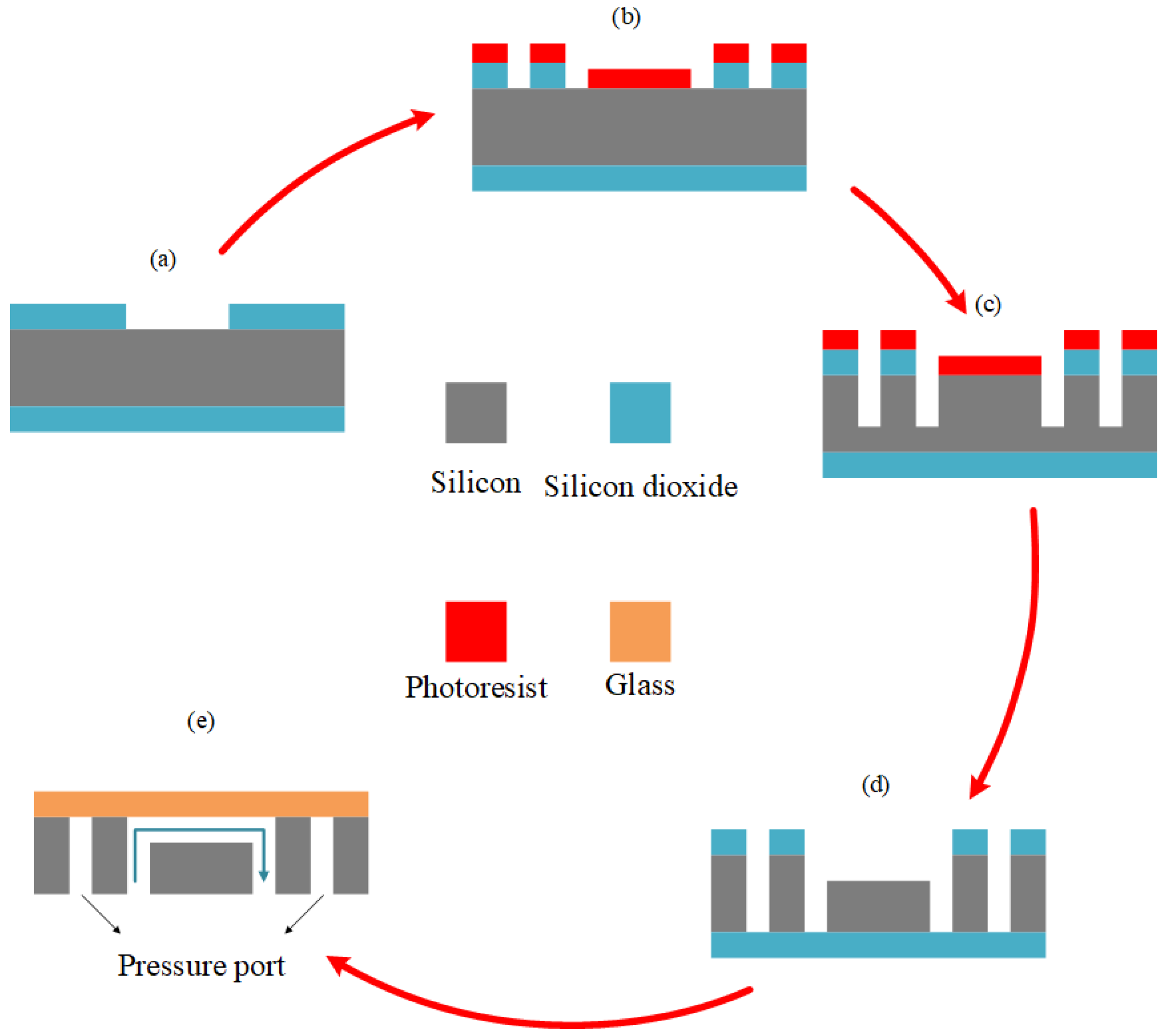
Since the experiments were performed at high pressures, the microfluidic devices were made of silicon and were bonded to glass to withstand high pressures. For this purpose, a 500 nm thick layer of SiO
2 was thermally grown and deposited on both sides of a <100> silicon wafer. The pattern of the microfluidic device was transferred on the surface of the substrate using MLA 150 maskless aligner photolithography machine with high resolution on a 2-µm thick layer of photoresist (AZ-ECI) as a positive photoresist. The employed software for the designs was Layout Editor. The exposure energy for photolithography was 320 mJ/cm
2. The SiO
2 layer was dry-etched, and the residual photoresist was stripped off the surface (
Figure 2a). The second photolithography was performed using a second mask to open inlet, outlets, and pressure ports of the microfluidic device. A second dry etching of SiO
2 (
Figure 2b) followed by 320 µm deep reactive ion etching (D-RIE) of silicon leads to
Figure 2c. Thereafter, the photoresist on top was removed, and another 50-µm deep dry etching of silicon was performed (
Figure 2d). The initially grown layers of SiO
2 were removed by the wet etching process, and the geometry of the whole device, including the inlet, outlets, and pressure ports openings, was achieved. An anodic bonding process was also employed to bond Borofloat 33 glass to the silicon substrate after cleaning (
Figure 2e).
2.3. The Experimental Setup of the Fluidic System
The working fluids (water, PBS, PBS/bacteria suspension) were kept in a stainless steel fluid container (Swagelok, Erbusco, Italy), which was connected to a high-pressure pure nitrogen tank (Linde Gas, Gabze, Kocaeli, Turkey) from the top, which drove the fluid into the system via proper fittings and stainless steel piping (Swagelok, Erbusco BS, Italy). A needle fine control valve (Swagelok, Erbusco BS, Italy) was installed to control the flow and inlet pressure during the experiments. The microfluidic device was installed and sandwiched on a homemade aluminum package, which facilitated flow visualization. The homemade package consisted of one inlet, which was connected to the fluid container and one outlet, where the fluid exiting the microfluidic device could leave the system. Three pressure sensors (Omega, Manchester, UK, with accuracy value of ±0.25% and the range of up to 3000 psi) were installed to measure P
1, P
2, and P
3 (
Figure 3). Micro O-rings and tight connections were used to avoid any leakage in the system. A double-shutter CMOS high-speed camera (Phantom v310 with a resolution of 1280 × 800 pixels and a pixel size of 0.02 mm, along with a macro camera lens (type K2 DistaMax) with the focal length of 50 mm and
f-number of 1.2) was used to record the flow patterns during the experiments. The package was installed at 20 cm distance from the camera so that it would be in the focal area. A point halogen light source was used to provide the required background light for better visualization of the flow patterns. The volume flow rate of the system (as measured for each data point and the velocity of the working fluid inside the microfluidic device by dividing a reference volume of the exiting fluid by the elapsed time for this volume to leave the system) was utilized for Reynolds and cavitation numbers calculation.
Figure 3 shows the schematic of the experimental setup, which was constructed in similar lines with our previous studies (with minor changes such as the removal of the filter) [
18].
The PBS/bacteria suspension was collected in sterile biological sample containers upon exiting the microfluidic device and was restored for the next cycle of cavitating flow. Sterile serological pipettes were used to reload the sample into the fluid container in the last part of the experiments (deactivation effects of cavitating flows). The experiments were performed in one shot, and the excess bacteria were kept at 4 °C until they were loaded into the system.
The uncertainties in experimental parameters are given in
Table 2. They were provided by the manufacturer’s specification sheets or were obtained using the propagation of the uncertainty method presented in the study of Kline and McClintock [
29].
2.4. Bacteria Culturing
Bacteria cultures were prepared from −80 °C glycerol stocks and used freshly for each assay. Salmonella enterica subsp. enterica serovar typhimurium (ATCC®14028™) was cultured in Luria–Bertani broth (LB) medium (10 g tryptone, 5 g yeast extract, 5 g NaCl, Sigma, Munich, Germany), until the logarithmic growth phase (around 108 CFU/mL) was reached. Bacteria were incubated at 37 °C for 16 h in a shaker at 220 rpm. Growth medium was centrifuged at 5000 rpm for 5 min, the supernatant was removed, and the pellet was washed once with PBS and was dispersed in sterile PBS buffer before cavitation experiments. Agar plates were prepared from LB medium supplemented with agar (Sigma, Munich, Germany) and incubated under similar conditions without shaking.
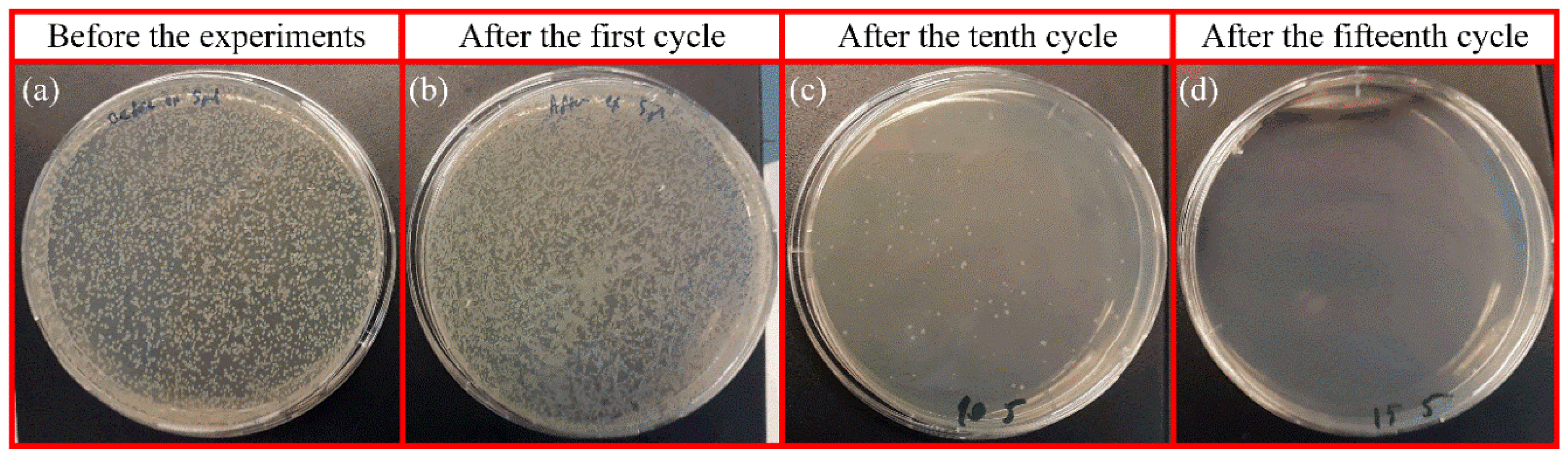
The bacteria suspension was diluted by 1000 mL PBS to obtain a concentration of 105 CFU/mL before cavitation assay. Then, 100 µl bacteria suspension was kept as a positive control to spread on agar plates. After the cavitation process, collected samples and control samples were diluted to 1:10 with sterile PBS. The diluted bacterial suspensions were spread over the LB agar medium by using sterile glass beads. The plates were placed in an incubator at 37 °C overnight, under dark condition. After incubation, the plates were observed for evaluation of the antibacterial effect.
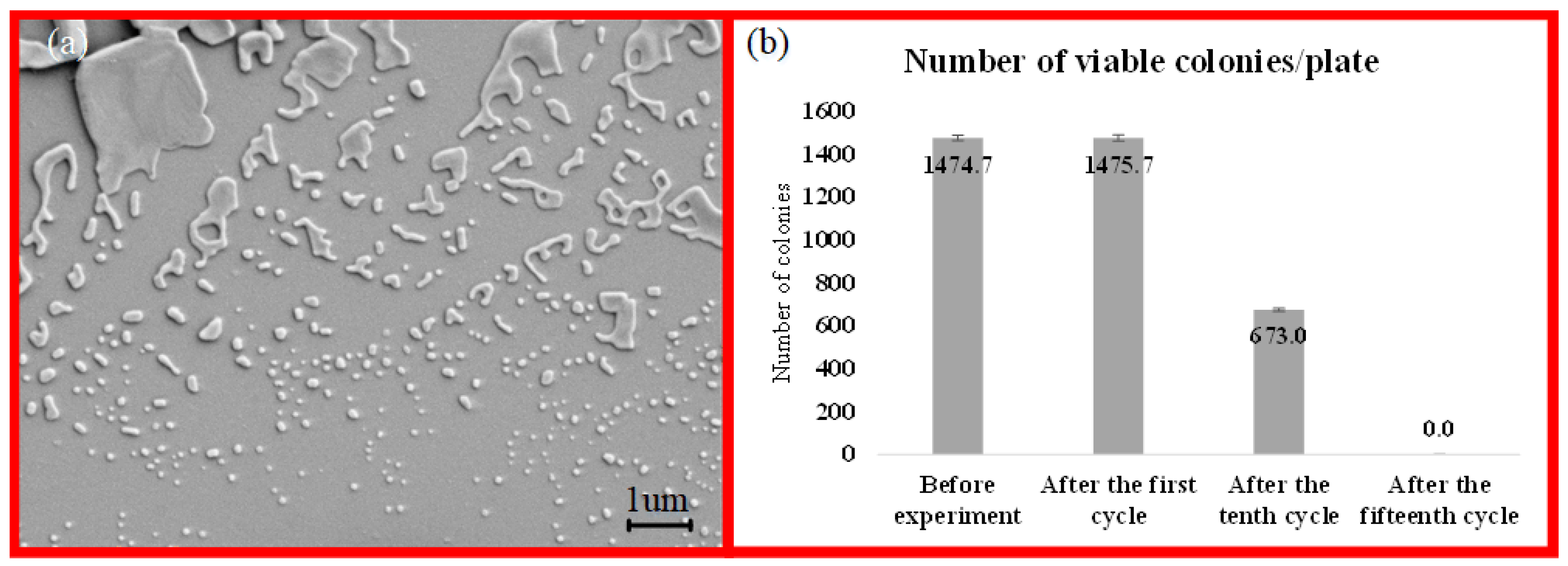
The number of viable bacterial colonies was calculated using the ImageJ software, v.152.a. All Petri dish images were combined in one PNG file and adjusted for brightness/contrast. Every plate image was then converted into an 8-bit image, and threshold corrections were applied. Finally, the particle analysis tool was applied to all dishes under identical analysis conditions to obtain the number of colonies per plate.
2.5. SEM Sample Preparation
Further understanding of bacteria morphology, including size and shape, was investigated by scanning electron microscopy (SEM, Leo Supra 35VP, Oberkochen, Germany) using an acceleration voltage of 3 kV. In order to provide a sample for SEM analysis, 10 µl of the sample was drop-casted on a silicon wafer and was left to dry at room temperature for 3 h. The sample was coated by gold-palladium alloy to avoid any surface charge of the bacteria and to make the surface homogeneous for image processing.
3. Theory
In order to characterize hydrodynamic cavitation, the main two parameters are employed: cavitation number and flowrate. Cavitation number is usually used to represent the intensity of cavitating flows in a fluidic system. The cavitation number is expressed as [
28]
where
P is the inlet pressure,
P1 in this study,
Pvap is the saturation vapor pressure of the working fluid,
is the density of the working fluid, and V is the (velocity in the orifice, maximum velocity) of the fluid along the microchannel.
P1 is measured using the pressure sensor in the experimental setup, and
Pvap and
ρ are read from the available look-up tables for each working fluid. The volumetric flow rate of the system is measured during the experiments at each inlet pressure, while the value of the velocity is calculated at the beginning of the microchannel (orifice), where the fluid has the maximum velocity.
The inlet pressure is gradually increased during the experiments so that the transition between flow patterns could be observed and accurate images could be recorded by the high-speed camera. According to Equation (1), the cavitation number decreases with an increase in the inlet pressure, and the inception occurs either in the microchannel (orifice) or in the extension of the microfluidic devices. The decreasing trend in the cavitation number continues, and the twin cavities emerge and move along the microchannel until the supercavitation condition becomes visible. At this point, the intensity of the cavitating flow reaches its maximum in the device. Beyond this point, the flow rate becomes saturated, and the velocity cannot be increased anymore with the inlet pressure. This leads to an increase in the cavitation number, which marks the choked flow condition. Thus, inception, developed cavitating flow, and supercavitation are observed during the experiments.
Cavitation flowrate is the critical flow rate, at which the inception of the cavitating flow is detected in the microfluidic device. Cavitation flowrate is expressed as [
30]
where
W and
w are the width of the inlet and microchannel, respectively.
Cd is the discharge coefficient of the device, which was explained and discussed in detail in our previous study [
8]. Equation (2) displays the effect of the thermophysical properties of the working fluid on the cavitation flowrate. The saturation vapor pressure directly affects this value, while the density has an adverse relationship with cavitation flowrate, which underlines the importance of the working fluid type on flow behavior.
In addition, the tensile strength of the working fluid, which is an indicator of its resistance against forming cavities in the bulk of fluid, could also be used to explain the flow patterns. The tensile strength of the working fluid is given as Equation (3) [
11]
where Δ
PC is the pressure difference between the bubble and the surroundings,
S is the surface tension, and
RC is the critical radius of the bubbles before collapsing. The lower tensile strength of the working fluid leads to earlier inception of the cavitating flow. Equation (3) emphasizes the importance of surface tension of the working fluid on the nucleation of bubbles encountering a low-pressure region.
Besides the mentioned parameters to characterize cavitating flows and Reynolds number, Equation (4) is an important dimensionless number in any fluidic system to characterize the flow pattern inside the channels [
31]
where
and µ are the density and dynamic viscosity of the working fluid, respectively. The corresponding values are listed in
Table 3 for water and PBS. On the other hand, V is the velocity of the fluid flow, and D
h is the hydraulic diameter of the channel. D
h for rectangular microchannels with side values of
a and
b is calculated as [
31]
Due to the small size and slow fluid flow in most of the microfluidic systems, the Reynolds number is very small, and the fluid shows a laminar behavior in the microscale channels. However, the Reynolds number is high enough to lead to turbulent flow behavior as a result of the high velocity of the fluid inside the system. The Reynolds number in these experiments is calculated at the beginning of the microchannel, where the velocity is at its maximum value. A Reynolds number of above 4000 shows a turbulent flow behavior in fluidic systems [
31].
5. Conclusions
In the first part of this study, water and PBS were tested as the working fluids in the fabricated microfluidic devices, and the differences in the flow behavior were discussed with the perspective of thermophysical differences. The results confirm the importance of surface tension and density of the working fluid for the nucleation of cavitating bubbles. The inception of hydrodynamic cavitation at the inlet happened at 2.41 MPa for the case of PBS, while it occurred at 3.10 MPa for the case of water. The tendency of PBS to form bubbly flow rather than bubble cloud was also recognized. The intensity of cavitating flows in the extension of the microfluidic devices was also another difference between the working fluids in this study. PBS led to a more intense cavitation flow regime in the extension compared to the case of water.
The second part of this study is dedicated to the effect of the presence of S. typhimurium in the working fluid on the flow patterns. The irregular shape of the bacteria in the working fluid acted as a solid interface, which promoted the inception of cavitation by increasing the heterogeneous nucleation sites. The energy released upon the collapse of the cavitating bubbles had an impact on the viability of the bacteria in the working fluid. The viability test on the suspended bacteria in the fluid exhibited a significant decrease in the colony number of bacteria after experiments. A deactivation efficiency of 100% was attained in the designed microfluidic devices.
A change in the design of the microfluidic devices, such as putting more micro-orifices on one chip, could further augment the output of the system. On the other hand, in some applications such as energy harvesting, intensifying the cavitating flow in microfluidic devices is of great importance. Reaching developed cavitating flows at lower inlet pressures could increase the efficiency of energy harvesting systems. Hence, using bacteria (non-toxic species) in the working fluid with this purpose could be a solution to raise the efficiency of energy harvesting systems, which are based on the “hydrodynamic cavitation on chip” concept. The major advantages of the proposed system are the low cost of fabrication and high bacteria deactivation efficiency.
The proposed concept in the study could be well utilized for energy harvesting as well as for water treatment. Since bacteria are found abundant in nature, and their culture is not a very costly procedure, they could be considered as strong candidates in energy harvesting systems to enhance performance. The species of the bacteria could be changed in this regard to ensure a safe and more environmentally-friendly platform. In addition, the design of such microfluidic devices could be further optimized.