Mezcal as a Novel Source of Mixed Yeasts Inocula for Wine Fermentation
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains and Inoculum Growth Conditions
2.2. Setup of Minifermentation Conditions
2.3. Mixed Yeasts Populations Quantification
2.4. Biomass and CO2 Production Quantification
2.5. Sugar Consumption and Metabolite Quantification by HPLC
2.6. Volatile Compound Quantification by GC-MS
2.7. Statistical Analyses
3. Results
3.1. Sampling and Yeast Identification
3.2. Fermentation Performance of the Mezcal Yeast Strains in Semi-Synthetic Medium M3
3.3. Volatile Productions of the Mezcal Yeast Strains in Medium M3
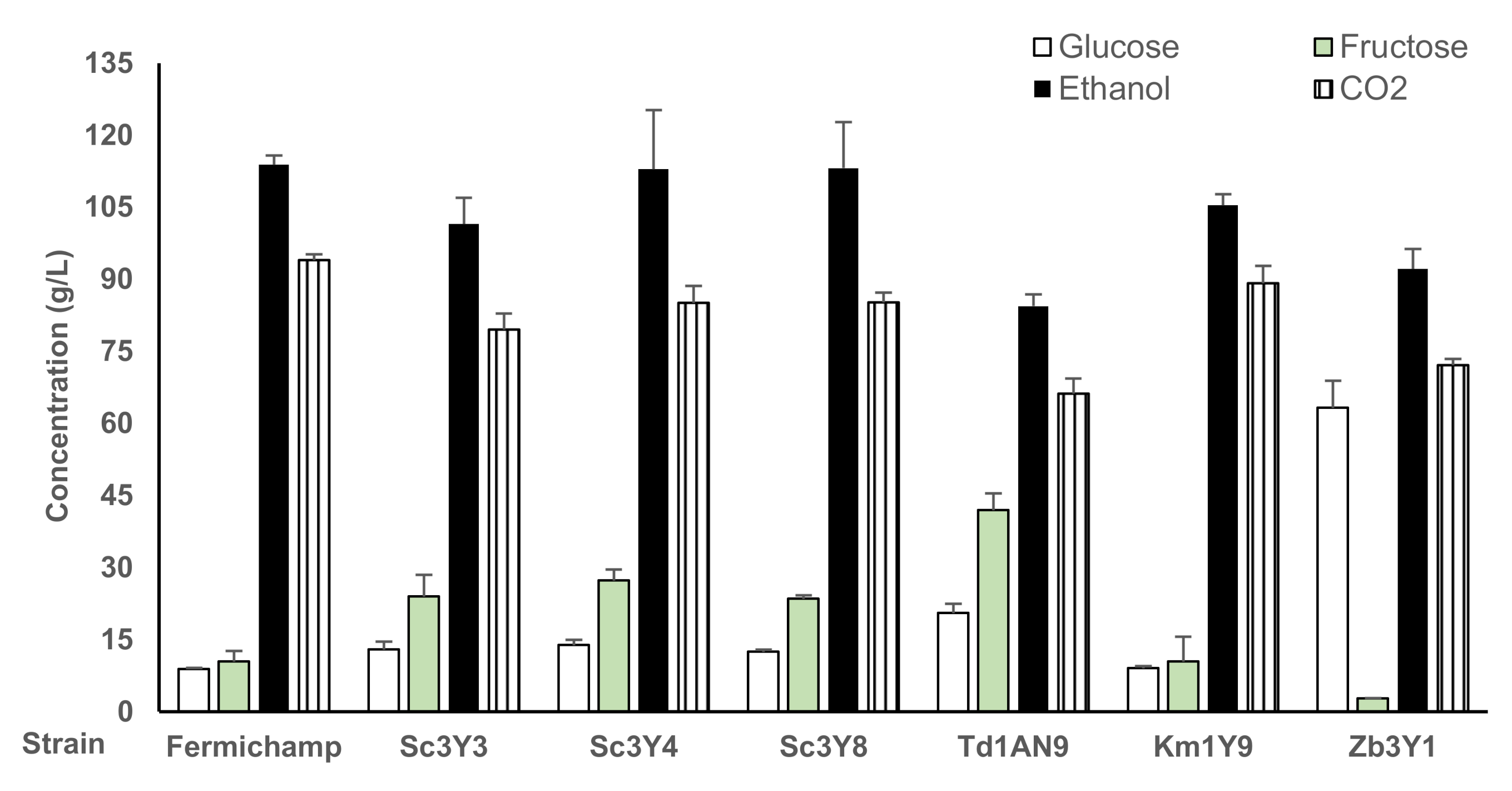
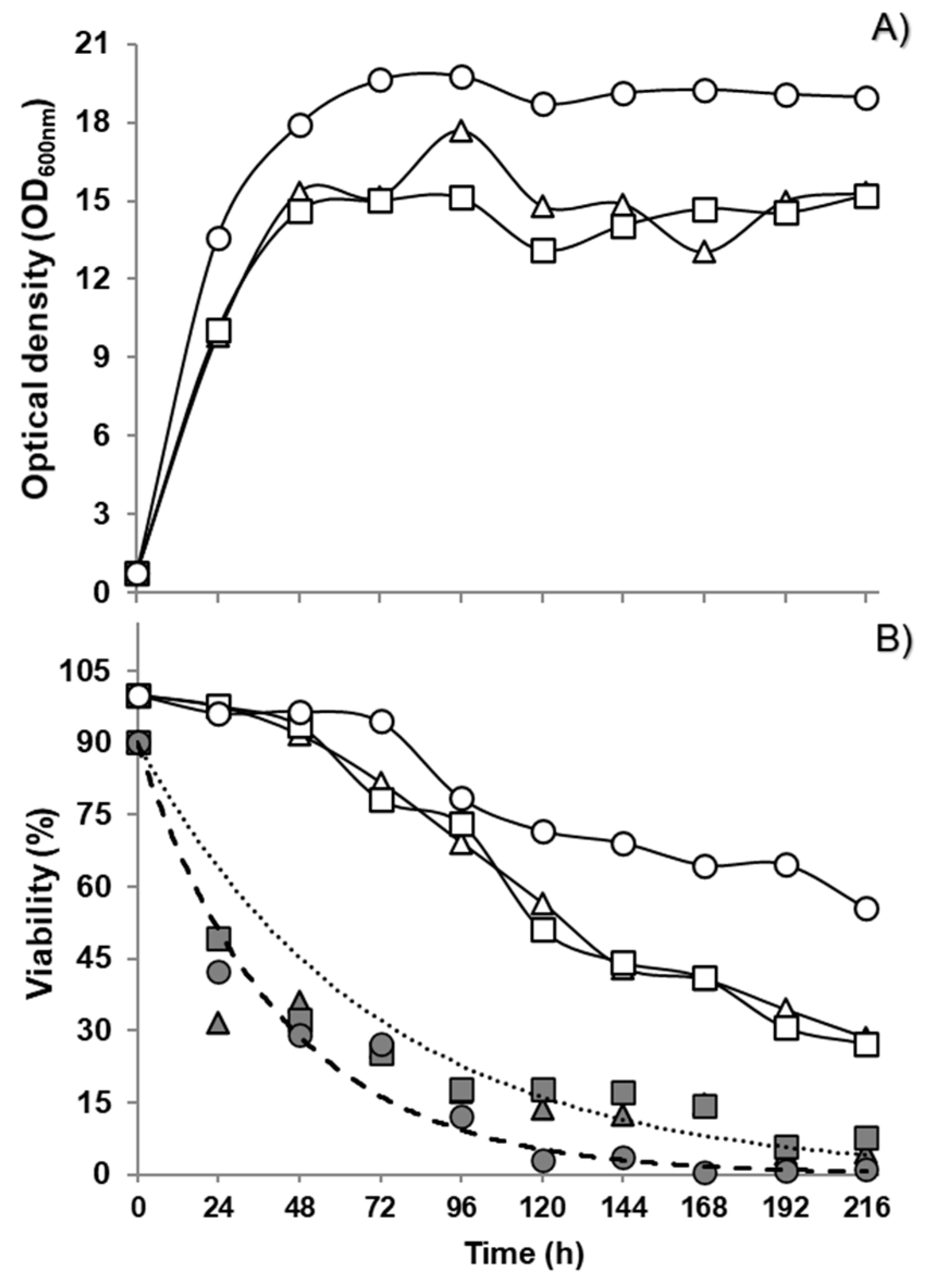
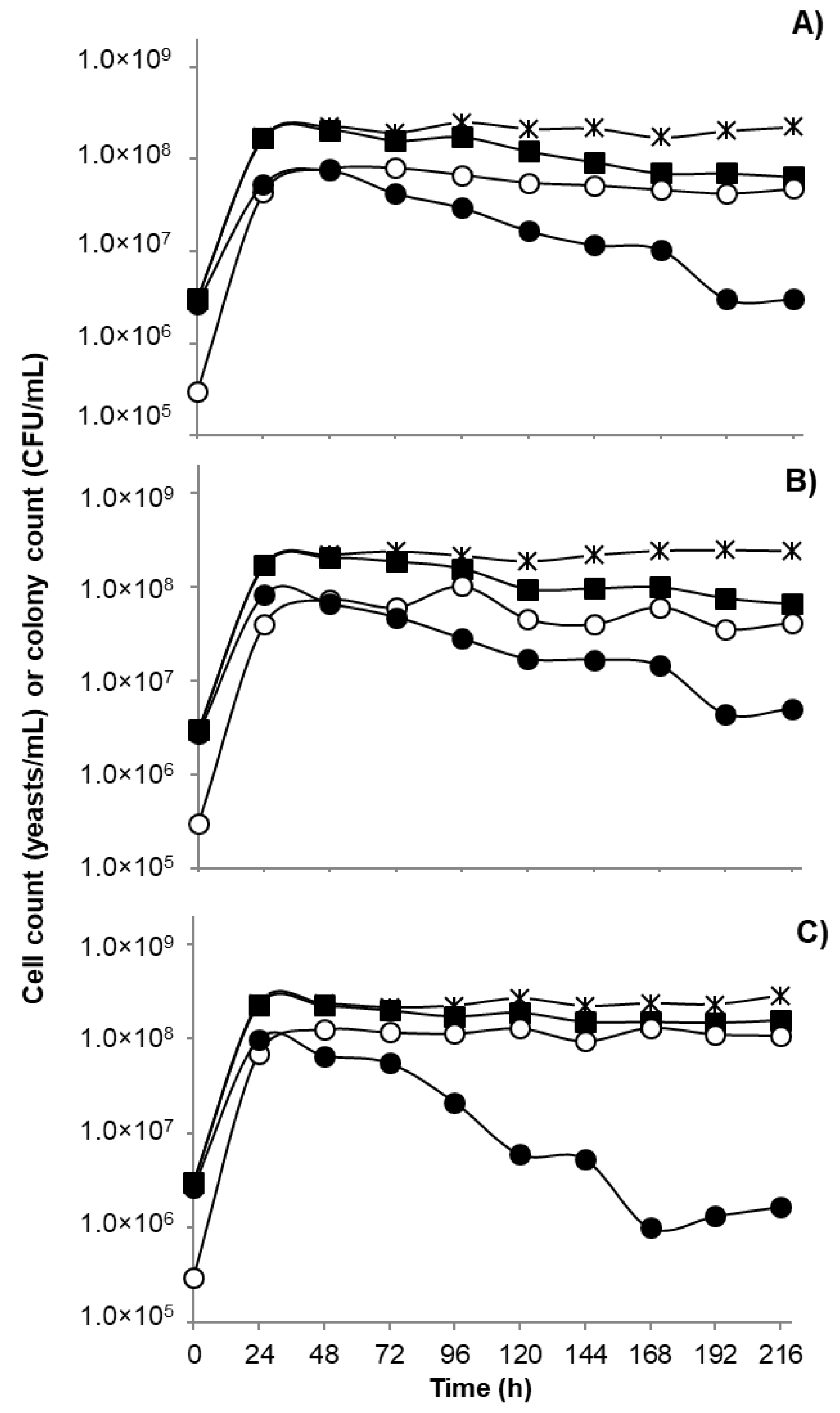
3.4. Fermentation Performance and Volatile Production of the Selected Yeasts, Individually and as Mixed Inoculum in Grape Juice Medium
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrajón, N.; Capece, A.; Arévalo-Villena, M.; Briones, A.; Romano, P. Co-inoculation of different Saccharomyces cerevisiae strains and influence on volatile composition of wines. Food Microbiol. 2011, 28, 1080–1086. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-López, F.N.; Querol, A.; Barrio, E. Application of a substrate inhibition model to estimate the effect of fructose concentration on the growth of diverse Saccharomyces cerevisiae strains. J. Ind. Microbiol. Biotechnol. 2009, 36, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Zuzuarregui, A.; del Olmo, M.L. Analyses of stress resistance under laboratory conditions constitute a suitable criterion for wine yeast selection. Antonie Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2004, 85, 271–280. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Greetham, D.; Wimalasena, T.T.; Phister, T.G.; Cabellos, J.M.; Arroyo, T. The phenotypic characterization of yeast strains to stresses inherent to wine fermentation in warm climates. J. Appl. Microbiol. 2016, 121, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Belda, I.; Ruiz, J.; Alastruey-Izquierdo, A.; Navascués, E.; Marquina, D.; Santos, A. Unraveling the enzymatic basis of wine “flavorome”: A phylo-functional study of wine related yeast species. Front. Microbiol. 2016, 7, 12. [Google Scholar] [CrossRef]
- De la Torre-González, F.J.; Narváez-Zapata, J.A.; López-y-López, V.E.; Larralde-Corona, C.P. Ethanol tolerance is decreased by fructose in Saccharomyces and non-Saccharomyces yeasts. LWT Food Sci. Technol. 2016, 67. [Google Scholar] [CrossRef]
- Peter, J.; De Chiara, M.; Friedrich, A.; Yue, J.-X.; Pflieger, D.; Bergström, A.; Sigwalt, A.; Barre, B.; Freel, K.; Llored, A.; et al. Genome evolution across 1011 Saccharomyces cerevisiae isolates. Nature 2018, 556, 339–344. [Google Scholar] [CrossRef]
- Oliva Hernández, A.A.; Taillandier, P.; Reséndez Pérez, D.; Narváez Zapata, J.A.; Larralde Corona, C.P. The effect of hexose ratios on metabolite production in Saccharomyces cerevisiae strains obtained from the spontaneous fermentation of mezcal. Antonie Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2013, 103, 833–843. [Google Scholar] [CrossRef]
- Vergara-Álvarez, I.; Quiroz-Figueroa, F.; Tamayo-órdónñez, M.C.; Oliva-Hernández, A.A.; Larralde-Corona, C.P.; Narváez-Zapata, J.A. Flocculation and expression of FLO genes of a Saccharomyces cerevisiae mezcal strain with high stress tolerance. Food Technol. Biotechnol. 2019, 57, 544–553. [Google Scholar] [CrossRef]
- Yan, G.; Zhang, B.; Joseph, L.; Waterhouse, A.L. Effects of initial oxygenation on chemical and aromatic composition of wine in mixed starters of Hanseniaspora vineae and Saccharomyces cerevisiae. Food Microbiol. 2020, 90, 103460. [Google Scholar] [CrossRef]
- Camarasa, C.; Sanchez, I.; Brial, P.; Bigey, F.; Dequin, S. Phenotypic landscape of Saccharomyces cerevisiae during wine fermentation: Evidence for origin-dependent metabolic traits. PLoS ONE 2011, 6, e25147. [Google Scholar] [CrossRef]
- Franco-Duarte, R.; Umek, L.; Mendes, I.; Castro, C.C.; Fonseca, N.; Martins, R.; Silva-Ferreira, A.C.; Sampaio, P.; Pais, C.; Schuller, D. New integrative computational approaches unveil the Saccharomyces cerevsiae pheno-metabolomic fermentative profile and allow strain selection for winemaking. Food Chem. 2016, 211, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Andorrà, I.; Berradre, M.; Rozès, N.; Mas, A.; Guillamón, J.M.; Esteve-Zarzoso, B. Effect of pure and mixed cultures of the main wine yeast species on grape must fermentations. Eur. Food Res. Technol. 2010, 231, 215–224. [Google Scholar] [CrossRef]
- Pérez-Nevado, F.; Albergaria, H.; Hogg, T.; Girio, F. Cellular death of two non-Saccharomyces wine-related yeasts during mixed fermentations with Saccharomyces cerevisiae. Int. J. Food Microbiol. 2006, 108, 336–345. [Google Scholar] [CrossRef]
- Nissen, P.; Nielsen, D.; Arneborg, N. Viable Saccharomyces cerevisiae cells at high concentrations cause early growth arrest of non-Saccharomyces yeasts in mixed cultures by a cell - Cell contact-mediated mechanism. Yeast 2003, 20, 331–341. [Google Scholar] [CrossRef]
- Kosel, J.; Čadež, N.; Schuller, D.; Carreto, L.; Franco-Duarte, R.; Raspor, P. The influence of Dekkera bruxellensis on the transcriptome of Saccharomyces cerevisiae and on the aromatic profile of synthetic wine must. FEMS Yeast Res. 2017, fox018, 9184–9196. [Google Scholar] [CrossRef]
- Maturano, Y.P.; Assof, M.; Fabani, M.P.; Nally, M.C.; Jofré, V.; Rodríguez Assaf, L.A.; Toro, M.E.; Castellanos de Figueroa, L.I.; Vazquez, F. Enzymatic activities produced by mixed Saccharomyces and non-Saccharomyces cultures: Relationship with wine volatile composition. Antonie Leeuwenhoek Int. J. Gen. Mol. Microbiol. 2015, 108, 1239–1256. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, Z. Volatile compounds of young wines from cabernet sauvignon, cabernet gernischet and chardonnay varieties grown in the loess plateau region of China. Molecules 2010, 15, 9184–9196. [Google Scholar] [CrossRef]
- Romano, P.; Fiore, C.; Paraggio, M.; Caruso, M.; Capece, A. Function of yeast species and strains in wine flavour. Int. J. Food Microbiol. 2003, 86, 169–180. [Google Scholar] [CrossRef]
- Ando, H.; Kurata, A.; Kishimoto, N. Antimicrobial properties and mechanism of volatile isoamyl acetate, a main flavour component of Japanese sake (Ginjo-shu). J. Appl. Microbiol. 2015, 118, 873–880. [Google Scholar] [CrossRef]
- Chambers, P.J.; Borneman, A.R.; Varela, C.; Cordente, A.G.; Bellon, J.R.; Tran, T.M.T.; Henschke, P.A.; Curtin, C.D. Ongoing domestication of wine yeast: Past, present and future. Aust. J. Grape Wine Res. 2015, 21, 642–650. [Google Scholar] [CrossRef]

| Species | Strain ID | GenBank Accession Number | Fermentation Stage of Isolation * |
|---|---|---|---|
| Fermichamp | - | ||
| Sc3Y2 | JQ824877 | Final | |
| Sc3Y3 | JQ824872 | Final | |
| Sc3Y4 | JQ824875 | Final | |
| Sc3Y5 | JQ824869 | Final | |
| Sc3Y8 | JQ824874 | Final | |
| Saccharomyces cerevisiae | Scmosca3 | KT945088 | fruit fly on the vat surface |
| Sc3D6 | JQ824876 | Final | |
| Sc3D5 | KT945085 | Final | |
| Sc3D4 | KT945086 | Final | |
| Sc3D2 | JQ824871 | Final | |
| Sc4Y3 | KT945087 | Trapiche | |
| Km4D3 | KT945094 | Trapiche | |
| Kluyveromyces marxianus | Km1D5 | KT945093 | Trapiche |
| Km1Y9 | KT945092 | Early | |
| Td1AN9 | KT945090 | Early | |
| Torulaspora delbrueckii | Td1AN2 | KT945089 | Early |
| Td1AN1 | KT945091 | Early | |
| Pichia kluyveri | Pk4D6 | KT945083 | Trapiche |
| Yamadazyma mexicana | Pm1AN3 | KT945081 | Early |
| Meyerozyma guilliermondii | Pg1Y12 | KT945082 | Early |
| Clavispora lusitaniae | Cl4Y4 | KT945080 | Trapiche |
| Candida parapsilosis | Cp1Y7 | KT945079 | Early |
| Rhodotorula mucilaginosa | RmP12 | KT945095 | Early |
| Zygosaccharomyces bailii | Zb3Y1 | KT945084 | Final |
| Strain | Glucose | Fructose | Ethanol | CO2 | Glycerol | Acetic Acid | Dry Weight |
|---|---|---|---|---|---|---|---|
| (g/L) | |||||||
| Fermichamp | 0.0 ± 0.0 | 4.7 ± 1.7 | 67.2 ± 1.4 | 97.7 ± 5.7 | 2.6 ± 0.1 | 0.5 ± 0.1 | 6.0 ± 0.3 |
| Sc3Y2 | 29.9 ± 3.9 | 54.8 ± 3.1 | 38.0 ± 0.2 | 49.8 ± 2.0 | 3.3 ± 0.2 | 0.0 ± 0.0 | 7.0 ± 1.7 |
| Sc3Y3 | 9.9 ± 1.4 | 21.7 ± 2.3 | 58.8 ± 13.5 | 75.8 ± 19.2 | 7.3 ± 1.0 | 0.8 ± 0.1 | 4.9 ± 0.5 |
| Sc3Y4 | 3.9 ± 1.4 | 13.5 ± 2.3 | 61.4 ± 4.7 | 83.0 ± 11.4 | 2. 7 ± 0.1 | 0.5 ± 0.03 | 3.9 ± 0.4 |
| Sc3Y5 | 9.9 ± 6.7 | 26.9 ± 9.7 | 57.7 ± 7.3 | 72.2 ± 11.6 | 6.7 ± 0.1 | 0.5 ± 0.3 | 4.5 ± 0.3 |
| Sc3Y8 | 9.9 ± 1.5 | 22.1 ± 1.3 | 53.3 ± 5.8 | 84.5 ± 4.36 | 2.6 ± 0.2 | 0.6 ± 0.2 | 3.8 ± 0.2 |
| Scmosca3 | 0.0 ± 0.0 | 2.7 ± 0.5 | 67.9 ± 0.4 | 94.0 ± 0.46 | 6.9 ± 0.2 | 0.3 ± 0.1 | 5.7 ± 0.2 |
| Sc3D6 | 0.3 ± 0.5 | 6.6 ± 2.7 | 68.4 ± 7.1 | 90.7 ± 1.42 | 8.2 ± 0.5 | 0.5 ± 0.03 | 5.9 ± 0.2 |
| Sc3D5 | 1.1 ± 1.6 | 9.0 ± 10.0 | 65.1 ± 4.9 | 90.0 ± 8.0 | 6.3 ± 0.2 | 0.3 ± 0.1 | 5.7 ± 0.1 |
| Sc3D4 | 0.4 ± 0.6 | 6.5 ± 5.6 | 66.0 ± 2.8 | 89.4 ± 7.3 | 6.9 ± 0.3 | 0.5 ± 0.03 | 5.4 ± 0.2 |
| Sc3D2 | 1.2 ± 0.3 | 12.5 ± 1.7 | 65.4 ± 1.2 | 88.3 ± 2.0 | 8.3 ± 0.1 | 0.9 ± 0.01 | 5.6 ± 0.1 |
| Sc4Y3 | 0.0 ± 0.0 | 3.6 ± 2.9 | 67.2 ± 0.2 | 92.9 ± 0.6 | 7.4 ± 0.1 | 0.3 ± 0.01 | 5.7 ± 0.1 |
| Km4D3 | 30.2 ± 24.2 | 51.6 ± 17.8 | 34.5 ± 17.2 | 53.6 ± 24.2 | 5.3 ± 2.39 | 0.5 ± 0.5 | 4.8 ± 0.2 |
| Km1D5 | 33.2 ± 0.5 | 57.2 ± 3.9 | 29.8 ± 2.1 | 46.0 ± 2.5 | 5.8 ± 0.67 | 0.2 ± 0.2 | 6.6 ± 0.6 |
| Km1Y9 | 12.2 ± 12.9 | 34.8 ± 16.6 | 45.3 ± 17.2 | 66.5 ± 19.4 | 6.2 ± 1.90 | 0.5 ± 0.3 | 5.1 ± 0.6 |
| Td1AN9 | 7.1 ± 5.2 | 27.8 ±10.3 | 50.6 ± 7.6 | 77.9 ± 6.5 | 6.6 ± 0.7 | 0.9 ± 0.2 | 4.5 ± 0.5 |
| Td1AN2 | 59.1 ± 3.0 | 67.9 ± 4. 7 | 20.0 ± 3.1 | 27.4 ± 4.4 | 3.8 ± 0.4 | 0.0 ± 0.0 | 6. 9 ± 0.8 |
| Td1AN1 | 35.2 ± 3.9 | 58.3 ± 4.5 | 31.2 ± 0.2 | 37.4 ± 2.6 | 2.9 ± 0.03 | 0.0 ± 0.0 | 8.2 ± 0.4 |
| Pk4D6 | 37.0 ± 4.8 | 60.8 ± 7.7 | 25.2 ± 2.9 | 34.7 ± 0.5 | 5.4 ± 0.7 | 0.2 ± 0.03 | 6.15 ± 0.5 |
| Pm1AN3 | 25.9 ± 4.9 | 51.4 ± 3.6 | 39.7 ± 3.7 | 49.6 ± 6.5 | 2.8 ± 0.2 | 0.0 ± 0.0 | 5.86 ± 0.7 |
| Pg1Y12 | 83.0 ± 0.4 | 83.0 ± 0.4 | 6.3 ± 0.2 | 6.7 ± 1.3 | 0.4 ± 0.04 | 0.0 ± 0.0 | 7.4 ± 1.0 |
| Cl4Y4 | 24.8 ± 12.4 | 48.6 ± 10.8 | 40.0 ± 7.0 | 49.3 ± 11.7 | 3.03 ± 0.5 | 0.0 ± 0.0 | 6.2 ± 1.0 |
| Cp1Y7 | 45.8 ± 0.2 | 64.0 ± 0.3 | 19 ± 0.01 | 28.6 ± 5.1 | 5.7 ± 0.2 | 0.2 ± 0.0 | 7.8 ±1.4 |
| RmP12 | 70.8 ± 19.6 | 76.2 ± 7.6 | 11.6 ± 13.9 | 13.3 ± 9.9 | 1.4 ± 1.9 | 0.5 ± 0.03 | 5.0 ± 1.3 |
| Zb3Y1 | 46.2 ± 17.7 | 0.64 ± 0.3 | 56.3 ± 2.0 | 66.6 ± 10.0 | 8.4 ± 0.1 | 0.3 ± 0.02 | 6.4 ± 0.6 |
| Strain | Isoamyl Alcohol | Isoamyl Acetate | Phenyl Ethyl Acetate | Ethyl Decanoate | Ethyl Octanoate | Ethyl Hexanoate | Ethyl Butyrate |
|---|---|---|---|---|---|---|---|
| (mg/L) | (µg/L) | ||||||
| Fermichamp | 74.0 ± 4.2 | 43.0 ± 2.8 | 76.5 ± 23.3 | 13.0 ± 5.0 | 49.5 ± 17.7 | 59.0 ± 8.5 | ND |
| Sc3Y2 | 47.5 ± 4.9 | D | 12.5 ± 4.95 | D | D | ND | ND |
| Sc3Y3 | 62.5 ± 24.7 | 25.5 ± 19.1 | 28.0 ± 1.4 | 7.5 ± 2.1 | 31.0 ± 1.4 | 53.5± 14.9 | ND |
| Sc3Y4 | 66.0 ±1.4 | 36.0 ± 2.8 | 49.0 ± 4.2 | 21.0 ± 1.7 | 58.0 ± 0.01 | 73.5 ± 6.4 | ND |
| Sc3Y5 | 52.5 ± 12.0 | 25.0 ± 14.1 | 123 ± 5.0 | 8.5 ± 0.7 | 15 ± 7.1 | 48.5± 16.3 | ND |
| Sc3Y8 | 49.0 ± 4.2 | D | 16.0 ± 1.4 | D | D | ND | ND |
| Scmosca3 | 59.5 ± 21.9 | 28.0 ± 0.0 | 187 ± 40.0 | 7.5 ± 5.0 | 77.0 ± 8.0 | 76.0 ± 9.0 | ND/D |
| Sc3D6 | 92.0 ± 7.1 | 42.0 ± 1.4 | 32.0 ± 12.7 | 9.0 ± 2.8 | 35.5 ± 12.0 | 69.0 ± 7.1 | 215 ± 8 |
| Sc3D5 | 78.0 ± 8.5 | 27.0 ± 1.4 | 35.5 ± 12.0 | 12.0 ± 4.2 | 83.0 ± 14.1 | 82.0 ± 8.5 | 205 ± 8 |
| Sc3D4 | 112 ± 0.7 | 46.5 ± 2.1 | 42.5 ± 3.5 | 15.5 ± 5.0 | 74.5 ± 19.1 | 81.0 ± 8.5 | 0.0 |
| Sc3D2 | 67.5 ± 9.2 | 32.0 ± 2.8 | 47.0 ± 9.9 | 10.0 ± 0.01 | 48.0 ± 1.4 | 55.0 ± 4.2 | 0.0 |
| Sc4Y3 | 74.0 ± 1.4 | 30.5 ± 0.7 | 25.5 ± 16.3 | 13.5 ± 6.4 | 71.0 ± 18.4 | 85.5 ± 0.7 | 230 ± 8 |
| Km4D3 | 60.5 ± 27.6 | 33 ± 0.0 | 1693 ± 574 | D | 2.0 ± 0.01 | ND | ND |
| Km1D5 | 15.0 ± 1.4 | 1774 ± 588 | 2422 ± 186 | D | 5.5 ± 0.7 | ND | ND |
| Km1Y9 | 79.5 ± 23.3 | 23.5 ± 19.0 | 2772 ± 743 | D | 2.0 ± 0.01 | ND | ND |
| Td1AN9 | 75.5 ± 9.2 | 13.5 ± 0.7 | 2594 ± 395 | D | D | ND | ND |
| Td1AN2 | 44.0 ± 21.2 | ND | 100 ± 13 | D | 3.0 ± 0.01 | ND | ND |
| Td1AN1 | 31.0 ± 4.2 | ND | 5.0 ± 1.0 | D | D | ND | ND |
| Pk4D6 | 11.0 ± 0.0 | 414 ± 136 | 4754 ± 821 | D | 2.0 ± 0.01 | ND | ND |
| Pm1AN3 | 41.0 ± 5.7 | ND | 19.5 ± 3.5 | D | D | ND | ND |
| Pg1Y12 | 15.5 ± 0.7 | ND | 1.0 ± 0.01 | D | D | ND | ND |
| Cp1Y7 | ND | 353 ± 64 | 5211 ± 452 | D | 2.0 ± 0.01 | ND | ND |
| Cl4Y4 | 45.5 ± 5.0 | ND | 13.0 ± 6.0 | D | D | ND | ND |
| RmP12 | 21.0 ± 0.0 | ND | ± 9.8 | D | 3.0 ± 0.01 | ND | ND |
| Zb3Y1 | 61.0 ± 2.8 | 12.0 ± 4.2 | 143 ± 36.1 | D | D | ND | ND |
| Strain | Dry Weight | Acetic Acid | Glycerol | Fermentation Power (FP) | Fermentation Purity | YEtOH/S |
|---|---|---|---|---|---|---|
| g/L | % v/v | gacet.ac/FP | ||||
| Fermichamp | 9.0 ± 0.3 | 0.2 ± 0.0 | 9.1 ± 0.2 | 14.4 | 0.014 | 0.48 |
| Sc3Y3 | 6.2 ± 0.7 | 0.7 ± 0.2 | 10.2 ± 1.1 | 12.9 | 0.054 | 0.46 |
| Sc3Y4 | 6.0 ± 0.2 | 0.8 ± 0.1 | 10.0 ± 0. 9 | 14.3 | 0.056 | 0.53 |
| Sc3Y8 | 6.9 ± 0.4 | 0.8 ± 0.1 | 10.0 ± 0.8 | 14.3 | 0.056 | 0.51 |
| Td1AN9 | 4.7 ± 0.2 | 0.5 ± 0.0 | 8.7 ± 0.4 | 10.7 | 0.047 | 0.44 |
| Km1Y9 | 3.6 ± 0.3 | 0.7 ± 0.0 | 9.9 ± 0.0 | 13.4 | 0.052 | 0.45 |
| Zb3Y1 | 6.3 ± 0.3 | 0.3 ± 0.0 | 7.8 ± 0.5 | 11.7 | 0.026 | 0.49 |
| Individual or Mixed Inocula Sc/non-Sc (1:9) | Glucose | Fructose | Ethanol | CO2 | Glycerol | Ac. Acid | Dry Weight |
|---|---|---|---|---|---|---|---|
| (g/L) | |||||||
| Fermichamp | 6.3 ± 0.09 | 5.6 ± 1.2 | 68.0 ± 1.9 | 99.4 ± 3.0 | 8.0 ± 0.2 | 0.5 ± 0.1 | 11.4 ± 0.2 |
| Fcham/Td1AN9 | 3.4 ± 2.6 | 7.5 ± 2.7 | 82.2 ± 1.1 | 92.9 ± 1.6 | 7.9 ± 0.3 | 0.3 ± 0.1 | 8.6 ± 0.4 |
| Fcham/Km1Y9 | 3.5 ± 2.7 | 6.8 ± 1.7 | 84.5 ± 2.3 | 94.5 ± 3.9 | 8.1 ± 0.2 | 0.4 ± 0.1 | 8.4 ± 0.3 |
| Fcham/Zb3Y1 | 3.8 ± 3.0 | 7.8 ± 4.1 | 82.1 ± 1.0 | 91.8 ± 3.0 | 4.8 ± 1.0 | 1.2 ± 0.5 | 8.3 ± 0.6 |
| Sc3Y3 | 7.1 ± 0.5 | 14.0 ± 2.0 | 66.0 ± 1.2 | 98.6 ± 2.5 | 8.0 ± 0.2 | 0.5 ± 0.0 | 7.6 ± 0.4 |
| Sc3Y3/Td1AN9 | 9.2 ± 1.7 | 24.9 ± 4.7 | 75.7 ± 1.7 | 86.4 ± 2.5 | 8.4 ± 0.4 | 1.2 ± 0.2 | 5.9 ± 0.2 |
| Sc3Y3/Km1Y9 | 11.4 ± 3.4 | 31.9 ± 7.1 | 73.4 ± 6.5 | 77.1 ± 2.0 | 7.6 ± 1.1 | 1.2 ± 0.4 | 5.4 ± 0.2 |
| Sc3Y3/Zb3Y1 | 14.4 ± 6.0 | 17.9 ± 6.1 | 79.1 ± 3.9 | 84.4 ± 5.9 | 6.3 ± 2.0 | 1.2 ± 0.6 | 6.5 ± 0.6 |
| Sc3Y4 | 8.1 ± 1.8 | 16.4 ± 5.9 | 67.0 ± 3.3 | 94.7 ± 5.4 | 8.4 ± 0.3 | 0.6 ± 0.1 | 7.5 ± 0.7 |
| Sc3Y4/Td1AN9 | 11.3 ±2.3 | 30.0 ± 4.6 | 73.3 ± 1.7 | 78.9 ± 3.8 | 7.5 ± 0.1 | 0.5 ± 0.2 | 5.6 ± 0.3 |
| Sc3Y4/Km1Y9 | 9.2 ± 3.5 | 25.0 ± 8.9 | 76.4 ± 3.9 | 81.3 ± 4.1 | 7.1 ± 0.4 | 0.8 ± 0.3 | 5.1 ± 0.3 |
| Sc3Y4/Zb3Y1 | 20.8 ± 5.0 | 23.6 ± 5.1 | 71.8 ± 4.0 | 78.4 ± 5.8 | 5.0 ± 1.2 | 1.3 ± 0.8 | 5.2 ± 0.3 |
| Sc3Y8 | 9.5 ± 2.8 | 19.5 ± 7.4 | 65.8 ± 2.7 | 1010± 6.6 | 7.3 ± 1.1 | 0.76 ± 0.3 | 7.0 ± 0.6 |
| Sc3Y8/Td1AN9 | 15.4 ± 8.2 | 35.8 ± 14.3 | 70.0 ± 7.8 | 76.4 ± 11.1 | 7.8 ± 0.3 | 0.9 ± 0.4 | 5.6 ± 0.3 |
| Sc3Y8/Km1Y9 | 9.8 ± 2.3 | 27.7 ± 6.0 | 75.9 ± 1.0 | 80.9 ± 5.6 | 7.4 ± 0.3 | 1.0 ± 0.3 | 5.5 ± 0.4 |
| Sc3Y8/Zb3Y1 | 17.3 ± 5.2 | 23.0 ± 4.8 | 81.6 ± 7.0 | 79.6 ± 5.5 | 7.4 ± 1.0 | 0.7 ± 0.4 | 6.2 ± 0.6 |
| Strain | Isoamyl Alcohol | Isoamyl Acetate | Phenyl Ethyl Acetate | Ethyl Decanoate | Ethyl Octanoate | Ethyl Hexanoate |
|---|---|---|---|---|---|---|
| (mg/L) | (µg/L) | |||||
| Fermichamp | 174 ± 5.0 | 111 ± 9.2 | 133 ± 14.1 | 133 ± 2.1 | 61 ± 4.9 | 40 ± 2.1 |
| Sc3Y3 | 77 ± 4.5 | D | 70 ± 16.5 | 140 ± 5.3 | 64 ± 4.2 | 38 ± 3.5 |
| Sc3Y4 | 78 ± 0.6 | D | 63 ± 5.7 | 103 ± 9.3 | 63 ± 2.1 | 33 ± 2.3 |
| Sc3Y8 | 82 ± 4.0 | D | 82 ± 20.5 | 115 ± 27.6 | 63 ± 2.7 | 45 ± 10.4 |
| Km1Y9 | 131 ± 6.0 | 30 ± 11.2 | 5069 ± 291.2 | 39 ± 3.6 | 38 ± 1.0 | D |
| Td1AN9 | 143 ± 90 | 45 ± 14.2 | 3349 ± 166.8 | 32 ± 0.6 | D | 16 ± 1.5 |
| Zb3Y1 | 213 ± 200 | D | 139 ± 9.1 | 32 ± 2.1 | 34 ± 0.0 | 15 ± 0.0 |
| Fcham/Td1AN9 | 141 ± 18.8 | 55 ± 9.9 | 543 ± 92.4 | 105 ± 16.5 | 41 ± 8.3 | 23 ± 3.5 |
| Sc3Y4/Td1AN9 | 93 ± 3.2 | D | 475 ± 34.9 | 84 ± 11.5 | 37 ± 8.5 | 33 ± 7.0 |
| Sc3Y8/Td1AN9 | 85 ± 9.1 | D | 544 ± 8.0 | 102 ± 16.5 | 33 ± 4.0 | 26 ± 2.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De la Torre-González, F.J.; Narváez-Zapata, J.A.; Taillandier, P.; Larralde-Corona, C.P. Mezcal as a Novel Source of Mixed Yeasts Inocula for Wine Fermentation. Processes 2020, 8, 1296. https://doi.org/10.3390/pr8101296
De la Torre-González FJ, Narváez-Zapata JA, Taillandier P, Larralde-Corona CP. Mezcal as a Novel Source of Mixed Yeasts Inocula for Wine Fermentation. Processes. 2020; 8(10):1296. https://doi.org/10.3390/pr8101296
Chicago/Turabian StyleDe la Torre-González, Francisco Javier, José Alberto Narváez-Zapata, Patricia Taillandier, and Claudia Patricia Larralde-Corona. 2020. "Mezcal as a Novel Source of Mixed Yeasts Inocula for Wine Fermentation" Processes 8, no. 10: 1296. https://doi.org/10.3390/pr8101296
APA StyleDe la Torre-González, F. J., Narváez-Zapata, J. A., Taillandier, P., & Larralde-Corona, C. P. (2020). Mezcal as a Novel Source of Mixed Yeasts Inocula for Wine Fermentation. Processes, 8(10), 1296. https://doi.org/10.3390/pr8101296





