Carbon Dots–TiO2 Hybrid Nanomaterials with Enhanced Photochemical Properties and Photodynamic Therapy Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
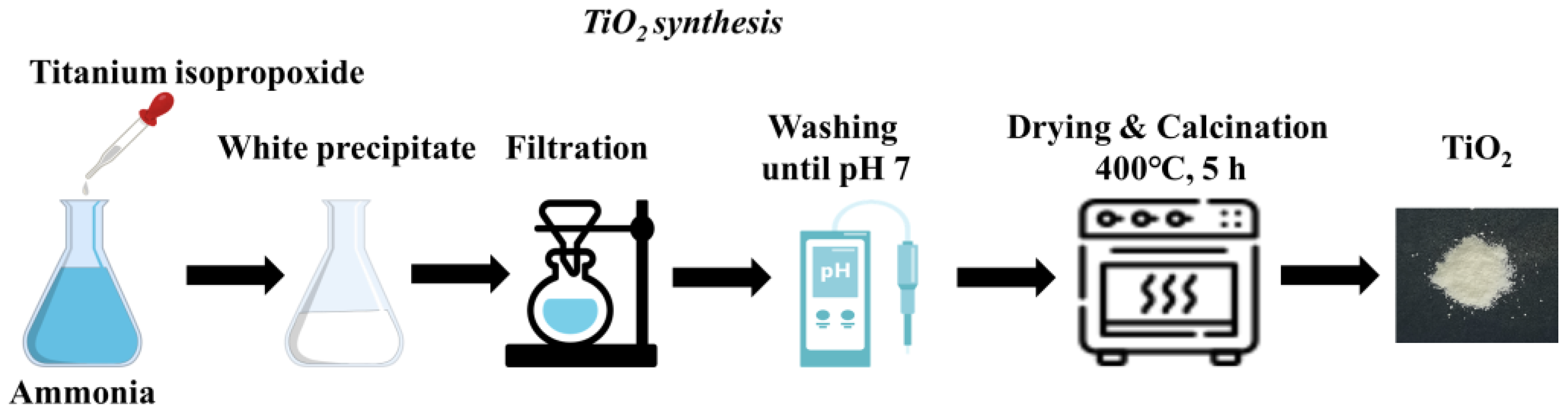
2.2. Synthesis of Titanium Dioxide (TiO2)
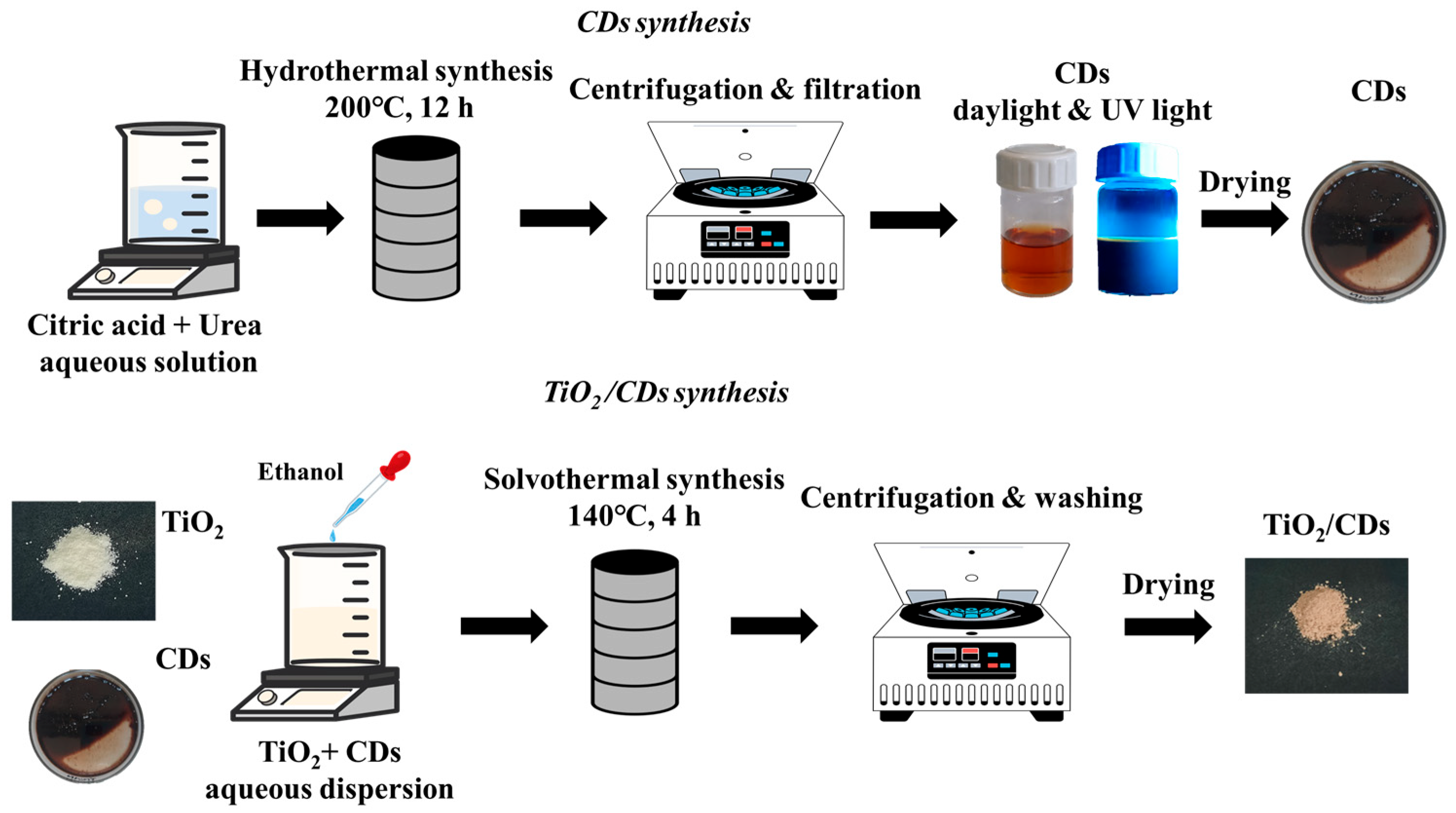
2.3. Synthesis of Carbon Dots (CDs) and TiO2/CDs Hybrid Materials
2.4. Characterization Methods
2.5. Irradiation Device
2.6. Reactive Oxygen Species (ROS) Studies
2.7. In Vitro Cell Studies
2.7.1. Cell Culture
2.7.2. Cell Viability Evaluation
2.7.3. Dark Toxicity Studies
2.7.4. Light Toxicity Studies
2.7.5. PDT Studies
2.7.6. Intracellular Localization
2.7.7. Statistical Analysis
3. Results and Discussion
3.1. Characterization of the Photosensitizers TiO2 and TiO2/CDs
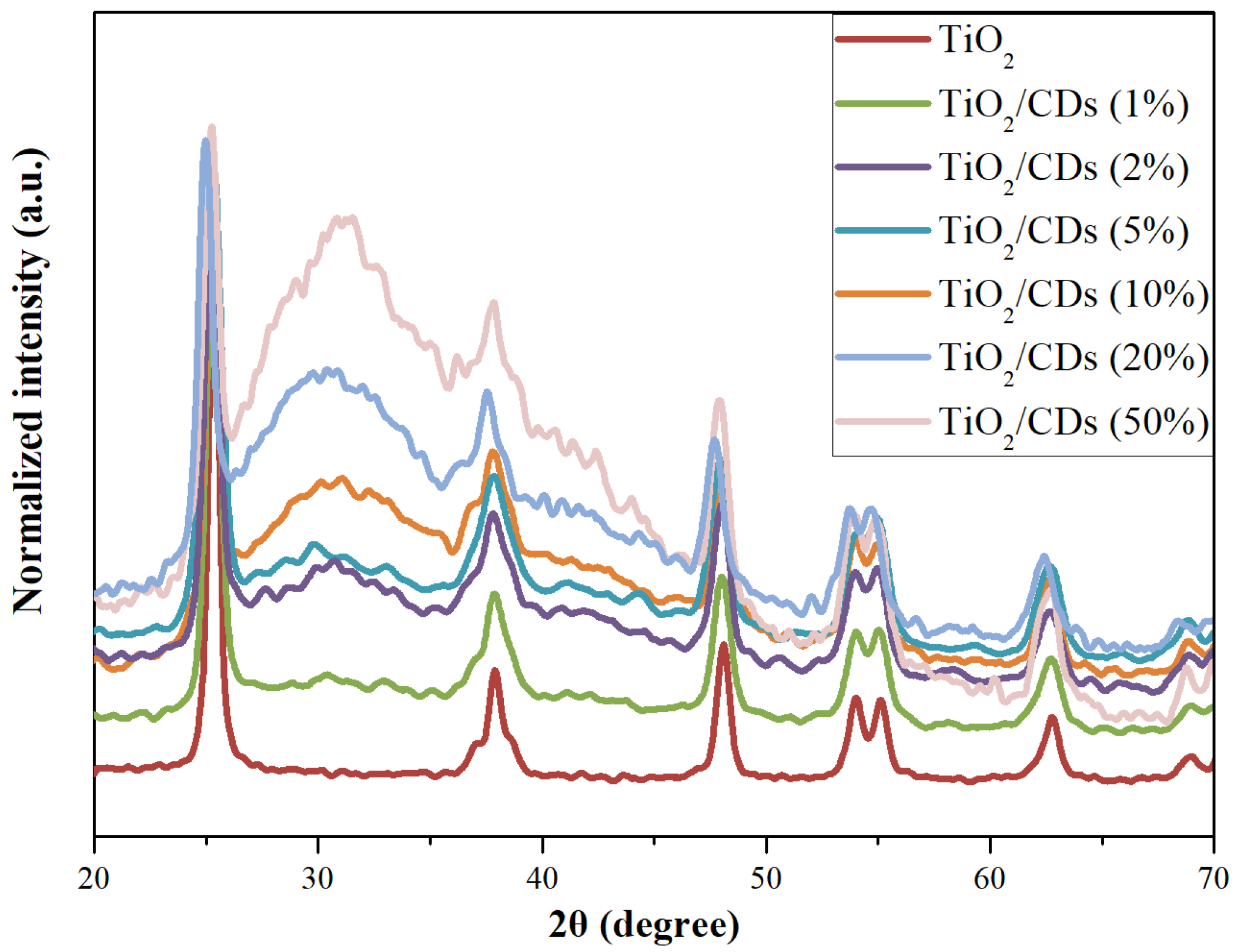
3.1.1. X-Ray Diffraction Analysis
3.1.2. Micro-Raman Spectroscopy
3.1.3. High-Resolution Transmission Electron Microscopy (HR-TEM) Analysis
3.1.4. Fourier Transform Infrared Spectroscopy (FT-IR Spectroscopy)
3.1.5. Zeta Potential
3.1.6. Thermogravimetric (TG) Analysis
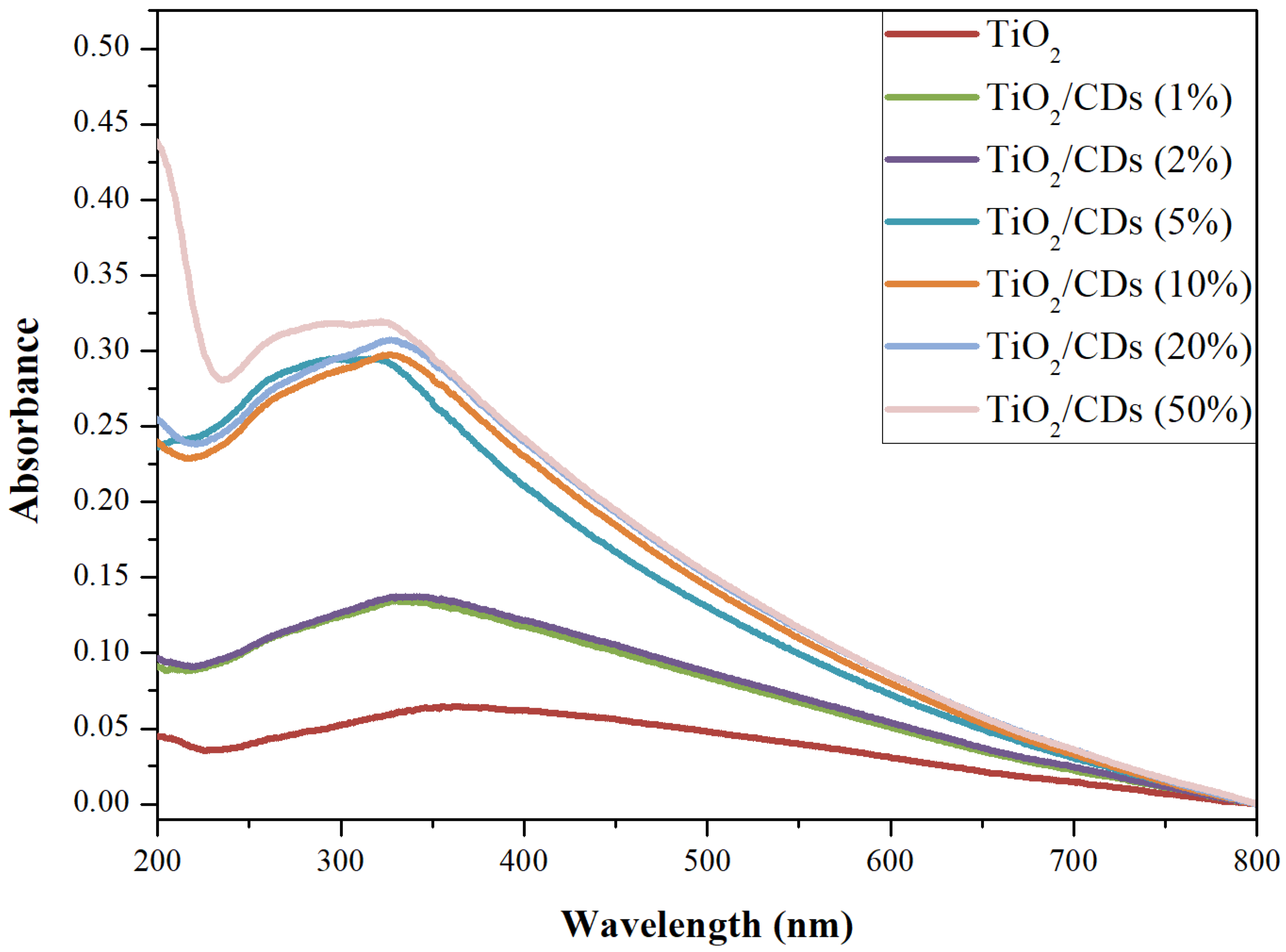
3.1.7. Ultraviolet-Visible (UV-Vis) and Photoluminescence (PL) Spectroscopy
3.2. Reactive Oxygen Species (ROS) Generation Studies
3.3. In Vitro Studies in Cancer Cells
3.3.1. Dark Cytotoxicity
3.3.2. Light Cytotoxicity
3.3.3. Photodynamic Therapy (PDT) Efficiency
3.3.4. Intracellular Localization
3.4. In Vitro Studies in Healthy Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Niculescu, A.-G.; Grumezescu, A.M. Photodynamic Therapy—An Up-to-Date Review. Appl. Sci. 2021, 11, 3626. [Google Scholar] [CrossRef]
- Karagianni, A.; Alexandratou, E.; Terrones, M.; Kordatos, K.V. Carbon-based nanomaterials in photodynamic therapy of cancer. Carbon 2025, 238, 119986. [Google Scholar] [CrossRef]
- Karagianni, A.; Tsierkezos, N.G.; Prato, M.; Terrones, M.; Kordatos, K.V. Application of carbon-based quantum dots in photodynamic therapy. Carbon 2023, 203, 273–310. [Google Scholar] [CrossRef]
- Chen, J.; Fan, T.; Xie, Z.; Zeng, Q.; Xue, P.; Zheng, T.; Chen, Y.; Luo, X.; Zhang, H. Advances in nanomaterials for photodynamic therapy applications: Status and challenges. Biomaterials 2020, 237, 119827. [Google Scholar] [CrossRef] [PubMed]
- Aebisher, D.; Rogóż, K.; Myśliwiec, A.; Dynarowicz, K.; Wiench, R.; Cieślar, G.; Kawczyk-Krupka, A.; Bartusik-Aebisher, D. The use of photodynamic therapy in medical practice. Front. Oncol. 2024, 14, 1373263. [Google Scholar] [CrossRef] [PubMed]
- Correia, J.H.; Rodrigues, J.A.; Pimenta, S.; Dong, T.; Yang, Z. Photodynamic Therapy Review: Principles, Photosensitizers, Applications, and Future Directions. Pharmaceutics 2021, 13, 1332. [Google Scholar] [CrossRef] [PubMed]
- Anucha, C.B.; Altin, I.; Bacaksiz, E.; Stathopoulos, V.N. Titanium dioxide (TiO2)-based photocatalyst materials activity enhancement for contaminants of emerging concern (CECs) degradation: In the light of modification strategies. Chem. Eng. J. Adv. 2022, 10, 100262. [Google Scholar] [CrossRef]
- Mojgan, R.; Ehsan, S.; Mostafa, Z. High photoluminescence and afterglow emission of nitrogen-doped graphene quantum dots/TiO2 nanocomposite for use as a photodynamic therapy photosensitizer. Appl. Phys. A 2024, 130, 144. [Google Scholar] [CrossRef]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B Environ. 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Kang, X.; Liu, S.; Dai, Z.; He, Y.; Song, X.; Tan, Z. Titanium Dioxide: From Engineering to Applications. Catalysts 2019, 9, 191. [Google Scholar] [CrossRef]
- Sargazi, S.; Simge, E.; Gelen, S.S.; Rahdar, A.; Bilal, M.; Arshad, R.; Ajalli, N.; Khan, M.F.A.; Pandey, S. Application of titanium dioxide nanoparticles in photothermal and photodynamic therapy of cancer: An updated and comprehensive review. J. Drug Deliv. Sci. Technol. 2022, 75, 103605. [Google Scholar] [CrossRef]
- Siwińska-Stefańska, K.; Jesionowski, T.; Siwińska-Stefańska, K.; Jesionowski, T. Advanced Hybrid Materials Based on Titanium Dioxide for Environmental and Electrochemical Applications. In Titanium Dioxide; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Khmiri, Y.; Attia, A.; Jallouli, N.; Chabanon, E.; Charcosset, C.; Mahouche-Chergui, S.; Dammak, L.; Algieri, C.; Chakraborty, S.; Ben Amar, R. Synthesis of a cost-effective ZnO/zeolite photocatalyst for paracetamol removal. Emergent Mater. 2025, 8, 7715–7731. [Google Scholar] [CrossRef]
- Attia, A.; Elboughdiri, N.; Ghernaout, D.; Carbonnier, B.; Amar, R.B.; Mahouche-Chergui, S. Enhancing the generation and stabilization of ZnO nanoparticles on modified clay with polyethylenimine to improve the photodegradation of dyes in textile wastewater. J. Water Process Eng. 2025, 73, 107711. [Google Scholar] [CrossRef]
- Reddy, K.R.; Hassan, M.; Gomes, V.G. Hybrid nanostructures based on titanium dioxide for enhanced photocatalysis. Appl. Catal. A General. 2015, 489, 1–16. [Google Scholar] [CrossRef]
- Falara, P.P.; Zourou, A.; Kordatos, K.V. Recent advances in Carbon Dots/2-D hybrid materials. Carbon 2022, 195, 219–245. [Google Scholar] [CrossRef]
- Sargin, I.; Yanalak, G.; Arslan, G.; Patir, I.H. Green synthesized carbon quantum dots as TiO2 sensitizers for photocatalytic hydrogen evolution. Int. J. Hydrogen Energy 2019, 44, 21781–21789. [Google Scholar] [CrossRef]
- Yan, Y.; Kuang, W.; Shi, L.; Ye, X.; Yang, Y.; Xie, X.; Shi, Q.; Tan, S. Carbon quantum dot-decorated TiO2 for fast and sustainable antibacterial properties under visible-light. J. Alloys Compd. 2019, 777, 234–243. [Google Scholar] [CrossRef]
- Madrid, A.; Martín-Pardillos, A.; Bonet-Aleta, J.; Sancho-Albero, M.; Martinez, G.; Calzada-Funes, J.; Martin-Duque, P.; Santamaria, J.; Hueso, J.L. Nitrogen-doped carbon nanodots deposited on titania nanoparticles: Unconventional near-infrared active photocatalysts for cancer therapy. Catal. Today 2023, 419, 114154. [Google Scholar] [CrossRef]
- Ramachandran, P.; Lee, C.Y.; Doong, R.-A.; Oon, C.E.; Thanh, N.T.K.; Lee, H.L. A titanium dioxide/nitrogen-doped graphene quantum dot nanocomposite to mitigate cytotoxicity: Synthesis, characterisation, and cell viability evaluation. RSC Adv. 2020, 10, 21795–21805. [Google Scholar] [CrossRef]
- Ramachandran, P.; Khor, B.-K.; Lee, C.Y.; Doong, R.-A.; Oon, C.E.; Thanh, N.T.K.; Lee, H.L. N-Doped Graphene Quantum Dots/Titanium Dioxide Nanocomposites: A Study of ROS-Forming Mechanisms, Cytotoxicity and Photodynamic Therapy. Biomedicines 2022, 10, 421. [Google Scholar] [CrossRef] [PubMed]
- Jabbar, M.S.; Mahmood, O.A.; Jameel, Z.N.; Jamal, N.J. Synthesis and Characterization of TiO2-CQDs Nanocomposites and Testing of Their Anticancer Potential. Int. J. Nanoelectron. Mater. (IJNeaM) 2024, 17, 627–633. [Google Scholar] [CrossRef]
- Liu, J.; Chen, K.-Y.; Wang, J.; Du, M.; Gao, Z.-Y.; Song, C.-X. Preparation and Photocatalytic Properties of N-Doped Graphene/TiO2 Composites. J. Chem. 2020, 2020, 2928189. [Google Scholar] [CrossRef]
- Shen, K.; Xue, X.; Wang, X.; Hu, X.; Tian, H.; Zheng, W. One-step synthesis of band-tunable N, S co-doped commercial TiO2/graphene quantum dots composites with enhanced photocatalytic activity. RSC Adv. 2017, 7, 23319–23327. [Google Scholar] [CrossRef]
- Mi, Y.; Wang, N.; Fang, X.; Cao, J.; Tao, M.; Cao, Z. Interfacial polymerization nanofiltration membrane with visible light photocatalytic self-cleaning performance by incorporation of CQD/TiO2. Sep. Purif. Technol. 2021, 277, 119500. [Google Scholar] [CrossRef]
- Mintz, K.J.; Bartoli, M.; Rovere, M.; Zhou, Y.; Hettiarachchi, S.D.; Paudyal, S.; Chen, J.; Domena, J.B.; Liyanage, P.Y.; Sampson, R.; et al. A deep investigation into the structure of carbon dots. Carbon 2021, 173, 433–447. [Google Scholar] [CrossRef]
- Shafique, M.; Mahr, M.S.; Yaseen, M.; Bhatti, H.N. CQD/TiO2 nanocomposite photocatalyst for efficient visible light-driven purification of wastewater containing methyl orange dye. Mater. Chem. Phys. 2022, 278, 125583. [Google Scholar] [CrossRef]
- Gao, Y.; Pu, X.; Zhang, D.; Ding, G.; Shao, X.; Ma, J. Combustion synthesis of graphene oxide–TiO2 hybrid materials for photodegradation of methyl orange. Carbon 2012, 50, 4093–4101. [Google Scholar] [CrossRef]
- Zhang, Q.; Bao, N.; Wang, X.; Hu, X.; Miao, X.; Chaker, M.; Ma, D. Advanced Fabrication of Chemically Bonded Graphene/TiO2 Continuous Fibers with Enhanced Broadband Photocatalytic Properties and Involved Mechanisms Exploration. Sci. Rep. 2016, 6, 38066. [Google Scholar] [CrossRef]
- Falara, P.P.; Ibrahim, I.; Zourou, A.; Sygellou, L.; Sanchez, D.E.; Romanos, G.E.; Givalou, L.; Antoniadou, M.; Arfanis, M.K.; Han, C.; et al. Bi-functional photocatalytic heterostructures combining titania thin films with carbon quantum dots (C-QDs/TiO2) for effective elimination of water pollutants. Environ. Sci. Pollut. Res. 2023, 30, 124976–124991. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, W.; Zhang, Y.; Liu, S. An Efficient Two-Step Technique for Nitrogen-Doped Titanium Dioxide Synthesizing: Visible-Light-Induced Photodecomposition of Methylene Blue. J. Phys. Chem. C 2007, 111, 1010–1014. [Google Scholar] [CrossRef]
- Raseel Rahman, M.K.; Riscob, B.; Bhatt, R.; Bhaumik, I.; Ganesamoorthy, S.; Vijayan, N.; Bhagavannarayana, G.; Kumar Kumal, A.; Nair, L. Investigations on Crystalline Perfection, Raman Spectra and Optical Characteristics of Transition Metal (Ru) Co-Doped Mg:LiNbO3 Single Crystals. ACS Omega 2021, 6, 10807–10815. [Google Scholar] [CrossRef]
- Inoue, F.; Ando, R.A.; Corio, P. Raman evidence of the interaction between multiwalled carbon nanotubes and nanostructured TiO2. J. Raman Spectrosc. 2011, 42, 1379–1383. [Google Scholar] [CrossRef]
- Chanda, A.; Rout, K.; Vasundhara, M.; Joshi, S.R.; Singh, J. Structural and magnetic study of undoped and cobalt doped TiO2 nanoparticles. RSC Adv. 2018, 8, 10939–10947. [Google Scholar] [CrossRef]
- Choudhury, B.; Choudhury, A. Local structure modification and phase transformation of TiO2 nanoparticles initiated by oxygen defects, grain size, and annealing temperature. Int. Nano Lett. 2013, 3, 55. [Google Scholar] [CrossRef]
- Hirata, T.; Kitajima, M.; Nakamura, K.G.; Asari, E. Infrared and Raman spectra of solid solutions Ti1−xZrxO2 (x ≤ 0.1). J. Phys. Chem. Solids 1994, 55, 349–355. [Google Scholar] [CrossRef]
- Bersani, D.; Lottici, P.P.; Ding, X.-Z. Phonon confinement effects in the Raman scattering by TiO2 nanocrystals. Appl. Phys. Lett. 1998, 72, 73–75. [Google Scholar] [CrossRef]
- Stepanidenko, E.A.; Skurlov, I.D.; Khavlyuk, P.D.; Onishchuk, D.A.; Koroleva, A.V.; Zhizhin, E.V.; Arefina, I.A.; Kurdyukov, D.A.; Eurov, D.A.; Golubev, V.G.; et al. Carbon Dots with an Emission in the Near Infrared Produced from Organic Dyes in Porous Silica Microsphere Templates. Nanomaterials 2022, 12, 543. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.C.; Robertson, J. Resonant Raman spectroscopy of disordered, amorphous, and diamondlike carbon. Phys. Rev. B 2001, 64, 075414. [Google Scholar] [CrossRef]
- Ji, C.; Han, Q.; Zhou, Y.; Wu, J.; Shi, W.; Gao, L.; Leblanc, R.M.; Peng, Z. Phenylenediamine-derived near infrared carbon dots: The kilogram-scale preparation, formation process, photoluminescence tuning mechanism and application as red phosphors. Carbon 2022, 192, 198–208. [Google Scholar] [CrossRef]
- Luo, Y.; Cui, C.; Zhang, X.; Jiang, Y.; Xiang, Z.; Ji, C.; Peng, Z. Carbon Dots-Based Fluorescence Assay for the Facile and Reliable Detection of Ag+ in Natural Water and Serum Samples. Molecules 2023, 28, 1566. [Google Scholar] [CrossRef] [PubMed]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Fawaz, W.; Hasian, J.; Alghoraibi, I. Synthesis and physicochemical characterization of carbon quantum dots produced from folic acid. Sci. Rep. 2023, 13, 18641. [Google Scholar] [CrossRef]
- Prasannan, A.; Imae, T. One-Pot Synthesis of Fluorescent Carbon Dots from Orange Waste Peels. Ind. Eng. Chem. Res. 2013, 52, 15673–15678. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, W.; Lin, H.; Yao, C.; He, Y.; Ran, X.; Guo, L.; Li, T. Facet-dependent photocatalytic and photoelectric properties of CQDs/TiO2 composites under visible irradiation. J. Alloys Compd. 2022, 920, 165896. [Google Scholar] [CrossRef]
- Choi, D.; Ham, S.; Jang, D.-J. Visible-light photocatalytic reduction of Cr(VI) via carbon quantum dots-decorated TiO2 nanocomposites. J. Environ. Chem. Eng. 2018, 6, 1–8. [Google Scholar] [CrossRef]
- Saud, P.S.; Pant, B.; Alam, A.-M.; Ghouri, Z.K.; Park, M.; Kim, H.-Y. Carbon quantum dots anchored TiO2 nanofibers: Effective photocatalyst for waste water treatment. Ceram. Int. 2015, 41, 11953–11959. [Google Scholar] [CrossRef]
- Ren, P.; Fu, X.; Zhang, Y. Carbon Quantum Dots-TiO2 Nanocomposites with Enhanced Catalytic Activities for Selective Liquid Phase Oxidation of Alcohols. Catal. Lett. 2017, 147, 1679–1685. [Google Scholar] [CrossRef]
- Hazarika, D.; Karak, N. Unprecedented Influence of Carbon Dot@TiO2 Nanohybrid on Multifaceted Attributes of Waterborne Hyperbranched Polyester Nanocomposite. ACS Omega 2018, 3, 1757–1769. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Ganesan, S.; Manikandan, V.; Sivaselvam, S.; AlSalhi, M.S.; Asemi, N.N.; Angayarkanni, J.; Rajendiran, N.; Lo, H.-M. Facile synthesis of carbon/titanium oxide quantum dots from lignocellulose-rich mandarin orange peel extract via microwave irradiation: Synthesis, characterization and bio-imaging application. Int. J. Biol. Macromol. 2023, 241, 124546. [Google Scholar] [CrossRef] [PubMed]
- Nia, M.H.; Rezaei-tavirani, M.; Nikoofar, A.R.; Masoumi, H.; Nasr, R.; Hasanzadeh, H.; Jadidi, M.; Shadnush, M. Stabilizing and dispersing methods of TiO2 nanoparticles in biological studies. Arch. Adv. Biosci. 2015, 6, 96–105. [Google Scholar] [CrossRef]
- Mahmood, A.; Shi, G.; Wang, Z.; Rao, Z.; Xiao, W.; Xie, X.; Sun, J. Carbon quantum dots-TiO2 nanocomposite as an efficient photocatalyst for the photodegradation of aromatic ring-containing mixed VOCs: An experimental and DFT studies of adsorption and electronic structure of the interface. J. Hazard. Mater. 2021, 401, 123402. [Google Scholar] [CrossRef]
- Vasconcelos, H.C.; Meirelles, M.; Özmenteş, R.; Korkut, A. Vacuum Ultraviolet Spectroscopic Analysis of Structural Phases in TiO2 Sol–Gel Thin Films. Coatings 2025, 15, 19. [Google Scholar] [CrossRef]
- Yu, H.; Zhao, Y.; Zhou, C.; Shang, L.; Peng, Y.; Cao, Y.; Wu, L.-Z.; Tung, C.-H.; Zhang, T. Carbon quantum dots/TiO2 composites for efficient photocatalytic hydrogen evolution. J. Mater. Chem. A 2014, 2, 3344–3351. [Google Scholar] [CrossRef]
- Song, F.; Sun, H.; Ma, H.; Gao, H. Porous TiO2/Carbon Dot Nanoflowers with Enhanced Surface Areas for Improving Photocatalytic Activity. Nanomaterials 2022, 12, 2536. [Google Scholar] [CrossRef] [PubMed]
- Usenko, E.; Glamazda, A.; Svidzerska, A.; Valeev, V.; Laguta, A.; Petrushenko, S.; Karachevtsev, V. DNA: TiO2 nanoparticle nanoassemblies: Effect of temperature and nanoparticle concentration on aggregation. J. Nanoparticle Res. 2023, 25, 113. [Google Scholar] [CrossRef]
- He, Z.; Que, W. Surface scattering and reflecting: The effect on light absorption or photocatalytic activity of TiO2 scattering microspheres. Phys. Chem. Chem. Phys. 2013, 15, 16768–16773. [Google Scholar] [CrossRef] [PubMed]
- Li, X.Z.; Li, F.B. Study of Au/Au3+-TiO2 Photocatalysts toward Visible Photooxidation for Water and Wastewater Treatment. Environ. Sci. Technol. 2001, 35, 2381–2387. [Google Scholar] [CrossRef]
- Sun, M.; Qu, S.; Ji, W.; Jing, P.; Li, D.; Qin, L.; Cao, J.; Zhang, H.; Zhao, J.; Shen, D. Towards efficient photoinduced charge separation in carbon nanodots and TiO2 composites in the visible region. Phys. Chem. Chem. Phys. 2015, 17, 7966–7971. [Google Scholar] [CrossRef]
- Falara, P.P.; Chatzikonstantinou, N.; Zourou, A.; Tsipas, P.; Sakellis, E.; Alexandratou, E.; Nasikas, N.K.; Kordatos, K.V.; Antoniadou, M. Optimizing Carbon Dot—TiO2 Nanohybrids for Enhanced Photocatalytic Hydrogen Evolution. Materials 2025, 18, 1023. [Google Scholar] [CrossRef]
- Deng, Y.; Chen, M.; Chen, G.; Zou, W.; Zhao, Y.; Zhang, H.; Zhao, Q. Visible–Ultraviolet Upconversion Carbon Quantum Dots for Enhancement of the Photocatalytic Activity of Titanium Dioxide. ACS Omega 2021, 6, 4247–4254. [Google Scholar] [CrossRef]
- Ke, J.; Li, X.; Zhao, Q.; Liu, B.; Liu, S.; Wang, S. Upconversion carbon quantum dots as visible light responsive component for efficient enhancement of photocatalytic performance. J. Colloid. Interface Sci. 2017, 496, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Khojiev, S.; Hojiyeva, G.; Tursunkulov, O.; Chen, D.; Mohamed, H.S.H.; Kholikov, A.; Akbarov, K.; Liu, J.; Li, Y. Red Emissive Upconversion Carbon Quantum Dots Modifying TiO2 for Enhanced Photocatalytic Performance. ACS Appl. Nano Mater. 2023, 6, 14669–14679. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, H.; Liu, J.; Liu, Y.; Kang, Z. Carbon quantum dots serving as spectral converters through broadband upconversion of near-infrared photons for photoelectrochemical hydrogen generation. J. Mater. Chem. A 2013, 1, 11529–11533. [Google Scholar] [CrossRef]
- Pan, J.; Sheng, Y.; Zhang, J.; Wei, J.; Huang, P.; Zhang, X.; Feng, B. Preparation of carbon quantum dots/TiO2 nanotubes composites and their visible light catalytic applications. J. Mater. Chem. A 2014, 2, 18082–18086. [Google Scholar] [CrossRef]
- Zourou, A.; Ntziouni, A.; Karagianni, A.; Alizadeh, N.; Argirusis, N.; Antoniadou, M.; Sourkouni, G.; Kordatos, K.V.; Argirusis, C. Recent Advances in Carbon Dots-Based Photocatalysts for Water Treatment Applications. Inorganics 2025, 13, 286. [Google Scholar] [CrossRef]
- Rasheed, T.; Anwar, M.T.; Ferry, D.B.; Ali, A.; Imran, M. Zero-dimensional luminescent carbon dots as fascinating analytical tools for the treatment of pharmaceutical based contaminants in aqueous media. Environ. Sci. Water Res. Technol. 2023, 10, 12–28. [Google Scholar] [CrossRef]
- Ma, R.; Xu, Z.; Zhang, C.; Li, H.; Chen, J.; Fan, J.; Shi, Q. Highly efficient reduction of Cr(VI) from industries sewage using novel biomass-driven carbon dots modified TiO2 under sunlight. Chem. Eng. J. 2024, 500, 157480. [Google Scholar] [CrossRef]
- Liu, W.; Li, Y.; Liu, F.; Jiang, W.; Zhang, D.; Liang, J. Visible-light-driven photocatalytic degradation of diclofenac by carbon quantum dots modified porous g-C3N4: Mechanisms, degradation pathway and DFT calculation. Water Res. 2019, 151, 8–19. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Li, X. Carbon quantum dots modified 3D flower-like BiOCl nanostructures with enhanced visible light photocatalytic degradation of rhodamine B. Discov. Mater. 2019, 5, 30. [Google Scholar] [CrossRef]
- Jourshabani, M.; Long, N.V.D.; Asrami, M.R.; Pho, Q.H.; Lee, B.-K.; Hessel, V. Nitrogen-doped carbon quantum dot as electron acceptor anchored on graphitic carbon nitride nanosheet for improving rhodamine B degradation. Mater. Sci. Eng. B 2024, 305, 117417. [Google Scholar] [CrossRef]
- Wang, F.; Wang, Y.; Feng, Y.; Zeng, Y.; Xie, Z.; Zhang, Q.; Su, Y.; Chen, P.; Liu, Y.; Yao, K.; et al. Novel ternary photocatalyst of single atom-dispersed silver and carbon quantum dots co-loaded with ultrathin g-C3N4 for broad spectrum photocatalytic degradation of naproxen. Appl. Catal. B Environ. 2018, 221, 510–520. [Google Scholar] [CrossRef]
- Wang, F.; Chen, P.; Feng, Y.; Xie, Z.; Liu, Y.; Su, Y.; Zhang, Q.; Wang, Y.; Yao, K.; Lv, W.; et al. Facile synthesis of N-doped carbon dots/g-C3N4 photocatalyst with enhanced visible-light photocatalytic activity for the degradation of indomethacin. Appl. Catal. B Environ. 2017, 207, 103–113. [Google Scholar] [CrossRef]
- Awang, H.; Peppel, T.; Strunk, J. Photocatalytic Degradation of Diclofenac by Nitrogen-Doped Carbon Quantum Dot-Graphitic Carbon Nitride (CNQD). Catalysts 2023, 13, 735. [Google Scholar] [CrossRef]














| Nanomaterial | FWHM (cm−1) |
|---|---|
| TiO2 | 13.29 ± 0.09 |
| TiO2/CDs (1%) | 13.40 ± 0.20 |
| TiO2/CDs (2%) | 13.27 ± 0.07 |
| TiO2/CDs (5%) | 12.45 ±0.08 |
| TiO2/CDs (10%) | 12.51 ± 0.07 |
| Nanomaterial | Zeta Potential (mV) |
|---|---|
| TiO2 | 1.33 |
| TiO2/CDs (1%) | 5.18 |
| TiO2/CDs (2%) | 13.4 |
| TiO2/CDs (5%) | 17.2 |
| TiO2/CDs (10%) | 18.7 |
| TiO2/CDs (20%) | −9.37 |
| TiO2/CDs (50%) | −16.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Karagianni, A.; Zourou, A.; Ntziouni, A.; Qu, C.; Terrones, M.; Argirusis, C.; Alexandratou, E.; Kordatos, K.V. Carbon Dots–TiO2 Hybrid Nanomaterials with Enhanced Photochemical Properties and Photodynamic Therapy Activity. Processes 2026, 14, 1048. https://doi.org/10.3390/pr14071048
Karagianni A, Zourou A, Ntziouni A, Qu C, Terrones M, Argirusis C, Alexandratou E, Kordatos KV. Carbon Dots–TiO2 Hybrid Nanomaterials with Enhanced Photochemical Properties and Photodynamic Therapy Activity. Processes. 2026; 14(7):1048. https://doi.org/10.3390/pr14071048
Chicago/Turabian StyleKaragianni, Alexandra, Adamantia Zourou, Afroditi Ntziouni, Conghang Qu, Mauricio Terrones, Christos Argirusis, Eleni Alexandratou, and Konstantinos V. Kordatos. 2026. "Carbon Dots–TiO2 Hybrid Nanomaterials with Enhanced Photochemical Properties and Photodynamic Therapy Activity" Processes 14, no. 7: 1048. https://doi.org/10.3390/pr14071048
APA StyleKaragianni, A., Zourou, A., Ntziouni, A., Qu, C., Terrones, M., Argirusis, C., Alexandratou, E., & Kordatos, K. V. (2026). Carbon Dots–TiO2 Hybrid Nanomaterials with Enhanced Photochemical Properties and Photodynamic Therapy Activity. Processes, 14(7), 1048. https://doi.org/10.3390/pr14071048










