Development of Functional Fermented Meat Products Using Agro-Food Byproducts
Abstract
1. Functional Fermented Food Products
2. Agro-Food Byproducts Used as Novel Ingredients in Production of Functional Foods
2.1. Antioxidant, Anti-Lipid and Anti-Microbial Applications
| Origin | Byproduct | Bioactive Compounds | Bioactive Properties/Application | References |
|---|---|---|---|---|
| Figs (Ficus carica L.) | Skin, seed, leaf of figs | Flavonoids (flavonoid glycosides and prenylated), coumarins, phenolic acids, terpenoids, alkaloids. | Antioxidant, antidiabetic, anticancer activity, neuroprotective effect, anti-inflammatory, anti-insecticidal activity, and antimicrobial activity | [53,54] |
| Blackcurrant (Ribes nigrum L.) | Pomace of blackcurrant | Vitamin C, carotenoids, and phytosterols, antocyanids (cyanidin and delphinidin derivatives), phenolic acids (protocatechuic, vanillic, ellagic, gallic, and syringic acid) | Antioxidant, anti-inflammatory activities, prevention of cardiovascular disease, educed systolic blood pressure | [55] |
| Blueberry (Vaccinium sp.) | Pomace of blueberry | Phenolic acids, flavonoids (anthocyanidins, tannins, anthocyanins, proanthocyanidins), vitamin C. | Antioxidant, anti-inflammation, neuro-protection, anti-metastatic, cardio-protective, antimicrobial, reno-protective, opthalmoprotective, anti-diabetic, hepato-protective, gastro-protective, anti-osteoporotic, anti-aging | [56] |
| Strawberry (ArAbutus unedo L.) | Pomace of strawberry | Phenolic acids (ellagic acid, cholorogenic acid); flavanols (catechin, procyanidin); anthocyanins (cyanidin-3-glucoside); fatty acids (α-linolenic, linoleic acid (ω-6)); vitamin E, vitamin C | Antiallergic, antibacterial, hepatoprotective, antithrombotic antiviral, urinary antiseptic, anti-inflammatory, anti-diarrheal, anti- hypertension, anti-diabetic, anticarcinogenic, neurodegenerative, and vasodilator effects | [57] |
| Pineapple (Ananas sativus L.) | Peel, core, pomace, and crown | Phenolic acids; vitamin C, vitamin D, vitamin E | Antioxidant, anti-inflammatory, antibacterial, antifungal, and anticancer activities | [58] |
| Lemon (Citrus limon L.) | Peel of lemon | Flavanones (eriodictyol, hesperidin, hesperetin, naringin); flavones (apigenin, diosmin); flavanols (quercetin, lymphocitrin); essential oils (limonene, β-pinene, γ-terpinene, α-pinene, myrcene, sabinene, geranial); ascorbic acid, organic acids, phenolic acids | Antimicrobial, anti-inflammatory, anticancer, antidiabetic, anti-obesogenic, anti-urolithic, and anti-cardiovascular disease effects | [59] |
| Pomegranate (Punica granatum L.) | Peel of pomegranate | Phenolic acids, flavonols (quercetin, kaempferol, (catechin), tannins (ellagic acid, 1,2,4-tri-O-galloyl-β-glucopyranose), anthocyanins | Antioxidant, antimicrobial, anti-inflammatory, antidiabetic, cytotoxic, anticancer activities | [60] |
| Apple (Malus domestica) | Peel, pomace, seed of apple | Phenolic acids, flavonoids, anthocyanins | Antioxidant | [61] |
| Tomato (Solanum lycopersicum) | Peel and seed of tomato | Carotenoids, phenolic acids and flavonoids, lycopene, dietary fiber | Antioxidant and radical scavenger | [62] |
| Grape (Vitis vinifera) | Pomace, seed, skin of grape | Anthocyanins, resveratrol, quercetin, kaempferol, catechins, phenolic acids and procyanidins | Antioxidant, anti-inflammatory, gut microbiota modulation, anti-obesity, cardioprotective, antidiabetic, hepatoprotective, anticancer, neuro-protective, antiproliferative anti-aging and antiaging activities | [63] |
| Eggs | Egg shells | Antioxidant peptides | Antioxidant activity, anti-inflammatory activity | [64,65] |
| Bovine | Buffalo horn | Peptides | Antioxidant | [66] |
| Bovine | Bovine hemoglobin hydrolysate | Peptides | Antibacterial, antihypertensive | [67] |
| Tomato | Tomato byproducts | Dietary fibers, proteins, carotenoids, tocopherols, polyphenols, lycopene | Anti-inflammatory, antiallergenic, antimicrobial, vasodilatory, antithrombotic, cardioprotective, antioxidant | [68,69] |
2.2. Thickeners, Gelling Agents, Texturizers and Colorants
| Product | Byproduct | Function | References |
|---|---|---|---|
| Fish | Fish bones, fish scales | Smooth and creamy texture, alternative of pork gelatin | [88,89] |
| Dairy | Whey protein and buttermilk | Texturizing agents | [90,91,92] |
| Onion | Onion hulls | [93] | |
| Spinach | Spinach stalks | [94] | |
| Tomato | Lycopene from tomato byproducts, carotenoids from tomato peels | Colorant | [95,96] |
| Grape | Wine pomace extract and flour | Colorant | [97] |
| Eggplant | Phenolic compounds from eggplant | Colorant | [98] |
| Potato | Phenolic compounds from potato peels | Colorant | [99] |
| Citrus fruits | Citrus peels | Colorant | [100,101,102] |
3. Extracts from Agro-Food Byproducts
4. Functional Fermented Meat Products
| Agro-Food Byproducts | Fermented Meat Products | Effect on the Properties of Fermented Meat Product | Reference |
|---|---|---|---|
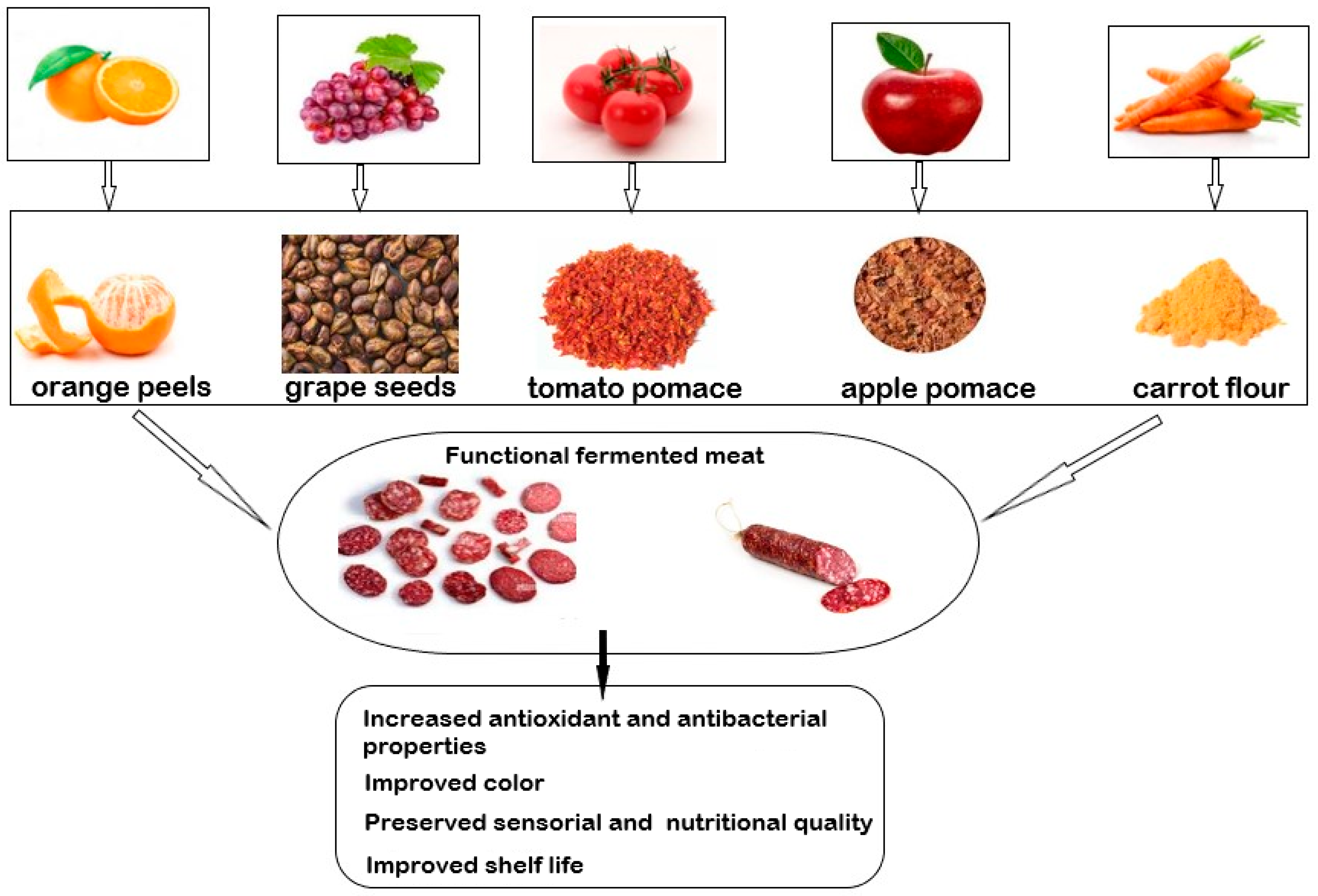
| Tomato pomace | Dry fermented sausage | Effective alternative to nitrates, leading to a reduction in the content of synthetic nitrates, high antioxidant and anti-lipid capacity, redness color, changed the fatty acid profile | [165] |
| Tomato pomace | Dry fermented sausage | The oxidative and microbiological stability, a higher heme iron content as well as higher carbonyl groups, saved the number of lactic acid bacteria | [166] |
| Tomato paste | Fermented sausages (mortadella) | Improved color and antioxidant activity, reduced lipid oxidation, and increased heme iron in dry fermented sausages, saved the texture | [167] |
| Tomato peels | Dry fermented sausage | High antioxidant capacity, sensory and textural properties very good, indicated a slight loss of lycopene after 21 days of ripening | [168] |
| Tomato powder | Beef sausage | Decreased pH, residual nitrite and moisture contents and increased protein, carbohydrate, ash, fiber and total calorie contents | [169] |
| Grape seed extract | Dry fermented sausage | Reduced the lipid oxidation, reduced the total volatile compounds from lipid oxidation. Sensorial analysis was good | [177] |
| Grape seed extract | Dry fermented sausage | High anti-lipid potential, low malondialdehyde values, high antimicrobial capacity, unchanged color | [178] |
| Grape seed flour | Fermented sausage “Basturma” | Improves the preservation against infectious pathogens and protein concentration | [179] |
| Grape seed extract | Beef sausage | Delayed lipid oxidation and prevented bacterial growth, kept the good quality and sensory characteristics | [180] |
| Grape pomace | Dry cured sausage (salchichón) | Decrease in TBARS value, increase in polyphenol content | [181] |
| Apple pomace | Turkey sausage | High total phenolic and fiber content, high antioxidant potential, strongly inhibited microorganism growth. | [188] |
| Apple pomace | Italian salami | Improved fiber and phenol content in salami and reduced the fat and calories | [189] |
| Apple pomace | Buffalo sausage | Increased water retention capacity, higher L color parameter, lower fat content, antimicrobial activity against S. aureus, P. aeruginosa, and L. Monocytogenes, emulsion stability | [190] |
| Orange fiber | Dry fermented sausage | Decrease in residual nitrite during fermentation, growth of Micrococcaceae (inhibits rancidity and stabilizes color) | [195] |
| Lemon fiber | Mortadella, a bologna-type sausage | Improved antimicrobial and antibacterial properties, improved the color, texture, and mineral composition, decreased residual nitrite levels. | [196] |
| Sugar beet (Beta vulgaris var. saccharifera L.) molasses | Sucuk | Substitute for sodium nitrate, complete replacement of sodium nitrate, improved the color | [213] |
| Cranberry skin powder | Dry fermented sausage | Reduced Salmonella enterica u Staphylococcus spp. cell count, preserved Lactobacillus spp. and Pediococcus spp. | [214] |
| Whey | Dry fermented sausage | Increased the nutritional value of non-nitrite organic fermented sausage, decreased pH and higher lactic acid bacteria content, improved the texture | [220] |
| Extracts of beer residue, chestnut leaves and peanut skin and micro-encapsulated fish oil | Spanish dry fermented sausage | Reduces the hexanal and total aldehyde content, antilipidemic effect of the mixture, partial replacement of pork back fat with microencapsulated fish oil | [221] |
| Pectin from fruit peel | Low-fat fermented sausage | Good texture profile of sausage, adding a small amount of pectin could replace the fat in sausage | [222] |
| Pectin and inulin | Frankfurter sausage | Fat could be replaced, moisture and ashes of sausages increased, shear force, hardness, fracturability, gumminess, and chewiness were slightly lower | [223] |
| Pistachio shells | Fermented sausage | Effective alternative to nitrates, leading to a reduction in the content of synthetic nitrates, high antioxidant and anti-lipid capacity | [224] |
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azino-bis-(3-etylbenzothiazoline-6-sulfonic acid |
| CUPRAC | Cupric reducing antioxidant capacity |
| DPPH | 1,1-diphenyl-2-picryhydrazyl |
| FMPs | Fermented meat products |
| FRAP | Ferric reducing antioxidant power |
| LAB | Lactic acid bacteria |
| ORAC | Oxygen radical absorbance capacity |
| PUFA | Polyunsaturated fatty acids |
| TRABS | Thiobarbituric acid reactive substances |
| TPC | Total phenolic content |
References
- Aziz, T.; Hussain, N.; Hameed, Z.; Lin, L. Elucidating the role of diet in maintaining gut health to reduce the risk of obesity, cardiovascular and other age-related inflammatory diseases: Recent challenges and future recommendations. Gut Microbes 2024, 16, 2297864. [Google Scholar] [CrossRef]
- Negi, R.; Jan, T.; Kaur, T.; Khan, S.S.; Yadav, N.; Rai, A.K.; Rustagi, S.; Shreaz, S.; Kour, D.; Ahmed, N.; et al. Bioactive compounds as plant-based functional foods for human health: Current scenario and future challenges. J. Appl. Biol. Biotechnol. 2025, 13, 1–23. [Google Scholar] [CrossRef]
- Subramanian, P.; Anandharamakrishnan, C. Introduction to functional foods and nutraceuticals. In Industrial Application of Functional Foods, Ingredients and Nutraceuticals; Subramanian, P., Anandharamakrishnan, C., Eds.; Academic Press: Cambridge, MA, USA, 2023; pp. 3–43. [Google Scholar]
- Arshad, M.S.; Khalid, W.; Ahmad, R.S.; Khan, M.K.; Ahmad, M.H.; Safdar, S.; Kousar, S.; Munir, H.; Shabbir, U.; Zafarullah, M.; et al. Functional foods and human health: An overview. In Functional Foods Phytochemicals and Health Promoting Potential; Arshad, M.S., Ahmad, M.H., Eds.; IntechOpen: London, UK, 2021; p. 3. [Google Scholar] [CrossRef]
- Granato, D.; Barba, F.J.; Kovačević, D.B.; Lorenzo, J.M.; Cruz, A.G.; Putnik, P. Functional foods: Product development, technological trends, efficacy testing, and safety. Annu. Rev. Food Sci. Technol. 2020, 11, 93–118. [Google Scholar] [CrossRef]
- Goksen, G.; Demir, D.; Dhama, K.; Kumar, M.; Shao, P.; Xie, F.; Echegaray, N.; Lorenzo, J.M. Mucilage polysaccharide as a plant secretion: Potential trends in food and biomedical applications. Int. J. Biol. Macromol. 2023, 230, 123146. [Google Scholar] [CrossRef]
- Indriyani, N.N.; Anshori, J.A.; Permadi, N.; Nurjanah, S.; Julaeha, E. Bioactive components and their activities from different parts of Citrus aurantifolia (Christm.) Swingle for food development. Foods 2023, 12, 2036. [Google Scholar] [CrossRef] [PubMed]
- Lazović, M.Č.; Jović, M.D.; Petrović, M.; Dimkić, I.Z.; Gašić, U.M.; Opsenica, D.M.M.; Ristivojević, P.M.; Trifković, J.Đ. Potential application of green extracts rich in phenolics for innovative functional foods: Natural deep eutectic solvents as media for isolation of biocompounds from berries. Food Funct. 2024, 15, 4122–4139. [Google Scholar] [CrossRef]
- Steinkraus, K.H. Fermentations in world food processing. Compr. Rev. Food Sci. Food Saf. 2002, 1, 23–32. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Merenstein, D.J.; Reid, G.; Gibson, G.R.; Rastall, R.A. Probiotics and prebiotics in intestinal health and disease: From biology to the clinic. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Tamang, J.P.; Watanabe, K.; Holzapfel, W.H. Diversity of microorganisms in global fermented foods and beverages. Front. Microbiol. 2016, 7, 377. [Google Scholar] [CrossRef]
- Toldrá, F. Biochemical changes during the fermentation of dry-cured meat products. Meat Sci. 2006, 77, 71–80. [Google Scholar] [CrossRef]
- Leroy, F.; Verluyten, J.; De Vuyst, L. Functional meat starter cultures for improved sausage fermentation. Int. J. Food Microbiol. 2006, 106, 270–285. [Google Scholar] [CrossRef]
- Tadesse, S.A.; Emire, S.A. Production and processing of antioxidant bioactive peptides: A driving force for the functional food market. Heliyon 2020, 6, e04765. [Google Scholar] [CrossRef] [PubMed]
- Stobiecka, M.; Król, J.; Brodziak, A. Antioxidant activity of milk and dairy products. Animals 2022, 12, 245. [Google Scholar] [CrossRef]
- Conte, P.; Pulina, S.; Del Caro, A.; Fadda, C.; Urgeghe, P.P.; De Bruno, A.; Difonzo, G.; Caponio, F.; Romeo, R.; Piga, A. Gluten-free breadsticks fortified with phenolic-rich extracts from olive leaves and olive mill wastewater. Foods 2021, 10, 923. [Google Scholar] [CrossRef]
- Zhu, Y.; Lao, F.; Pan, X.; Wu, J. Food protein-derived antioxidant peptides: Molecular mechanism, stability and bioavailability. Biomolecules 2022, 12, 1622. [Google Scholar] [CrossRef]
- Rogel-Castillo, C.; Latorre-Castañeda, M.; Muñoz-Muñoz, C.; Agurto-Muñoz, C. Seaweeds in food: Current trends. Plants 2023, 12, 2287. [Google Scholar] [CrossRef]
- Cuamatzin-García, L.; Rodríguez-Rugarcía, P.; El-Kassis, E.G.; Galicia, G.; Meza-Jiménez, M.D.L.; Baños-Lara, M.D.R.; Zaragoza-Maldonado, D.S.; Pérez-Armendáriz, B. Traditional fermented foods and beverages from around the world and their health benefits. Microorganisms 2022, 10, 1151. [Google Scholar] [CrossRef] [PubMed]
- Ubeyitogullari, A.; Ahmadzadeh, S.; Kandhola, G.; Kim, J. Polysaccharide-based porous biopolymers for enhanced bioaccessibility and bioavailability of bioactive food compounds: Challenges, advances, and opportunities. Compr. Rev. Food Sci. Food Saf. 2022, 21, 4610–4639. [Google Scholar] [CrossRef]
- Chourasia, R.; Phukon, L.C.; Abedin, M.M.; Padhi, S.; Singh, S.P.; Rai, A.K. Bioactive peptides in fermented foods and their application: A critical review. Syst. Microbiol. Biomanuf. 2023, 3, 88–109. [Google Scholar] [CrossRef]
- Laya, A.; Wangso, H.; Fernandes, I.; Djakba, R.; Oliveira, J.; Carvalho, E. Bioactive ingredients in traditional fermented food condiments: Emerging products for prevention and treatment of obesity and type 2 diabetes. J. Food Qual. 2023, 2023, 5236509. [Google Scholar] [CrossRef]
- Harahap, I.A.; Suliburska, J.; Karaca, A.C.; Capanoglu, E.; Esatbeyoglu, T. Fermented soy products: A review of bioactives for health from fermentation to functionality. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70080. [Google Scholar] [CrossRef] [PubMed]
- Galanakis, C.M.; Rizou, M.; Aldawoud, T.M.S.; Ucak, I.; Rowan, N.J. Innovations and technology disruptions in the food sector within the COVID-19 pandemic and post-lockdown era. Trends Food Sci. Technol. 2021, 110, 193–200. [Google Scholar] [CrossRef]
- Liu, Z.; de Souza, T.S.P.; Holland, B.; Dunshea, F.; Barrow, C.; Suleria, H.A.R. Valorization of food waste to produce value-added products based on its bioactive compounds. Processes 2023, 11, 840. [Google Scholar] [CrossRef]
- Ezeorba, T.P.C.; Okeke, E.S.; Mayel, M.H.; Nwuche, C.O.; Ezike, T.C. Recent advances in biotechnological valorization of agro-food wastes: Optimizing integrated approaches for sustainable biorefinery and circular bioeconomy. Bioresour. Technol. Rep. 2024, 26, 101823. [Google Scholar] [CrossRef]
- Xie, Y.; Kang, R.; Tang, D. The flavone baicalein and its use in gastrointestinal disease. In Dietary Interventions in Liver Disease; Elsevier: Amsterdam, The Netherlands, 2019; pp. 145–155. [Google Scholar] [CrossRef]
- Sut, S.; Dall’Acqua, S.; Zengin, G.; Senkardes, I.; Bulut, G.; Cvetanović, A.; Stupar, A.; Mandić, A.; Picot-Allain, C.; Dogan, A.; et al. Influence of different extraction techniques on the chemical profile and biological properties of Anthemis cotula L.: Multifunctional aspects for potential pharmaceutical applications. J. Pharm. Biomed. Anal. 2019, 173, 75–85. [Google Scholar] [CrossRef]
- Varghese, S.A.; Pulikkalparambil, H.; Promhuad, K.; Srisa, A.; Laorenza, Y.; Jarupan, L.; Nampitch, T.; Chonhenchob, V.; Harnkarnsujarit, N. Renovation of agro-waste for sustainable food packaging: A review. Polymers 2023, 15, 648. [Google Scholar] [CrossRef] [PubMed]
- Ngoc, L.T.N.; Moon, J.-Y.; Lee, Y.-C. Insights into bioactive peptides in cosmetics. Cosmetics 2023, 10, 111. [Google Scholar] [CrossRef]
- Cermeño, M.; Bascón, C.; Amigo-Benavent, M.; Felix, M.; FitzGerald, R.J. Identification of peptides from edible silkworm pupae (Bombyx mori) protein hydrolysates with antioxidant activity. J. Funct. Foods 2022, 92, 105052. [Google Scholar] [CrossRef]
- Jiang, J.; Xiong, Y.L. Natural antioxidants as food and feed additives to promote health benefits and quality of meat products: A review. Meat Sci. 2016, 120, 107–117. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.; Gómez, B.; Barba, F.J.; Mora, L.; Pérez-Santaescolástica, C.; Toldrá, F. Bioactive peptides as natural antioxidants in food products—A review. Trends Food Sci. Technol. 2018, 79, 136–147. [Google Scholar] [CrossRef]
- Carocho, M.; Morales, P.; Ferreira, I.C. Antioxidants: Reviewing the chemistry, food applications, legislation and role as preservatives. Trends Food Sci. Technol. 2018, 71, 107–120. [Google Scholar] [CrossRef]
- Roca-Saavedra, P.; Méndez-Vilabrille, V.; Miranda, J.M.; Nebot, C.; Cardelle-Cobas, A.; Franco, C.M.; Cepeda, A. Food additives, contaminants and other minor components: Effects on human gut microbiota—A review. J. Physiol. Biochem. 2017, 74, 69–83. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef]
- Benchabane, L.; Samghouli, A.; Abderrahim, L.A.; Allali, H. Chemical composition and biological activity of Thymus willdenowii essential oil and its application in yogurt. J. Food Meas. Charact. 2022, 16, 4165–4175. [Google Scholar]
- Fawzy, N.; Abo El-Nor, S.A.H.; El-Deeb, D.A.; El-Tanboly, E.S.; El Sayed, M.A. The role of thyme and clove essential oil nanoemulsions in quality improvement of soft cheese. Suez Canal Vet. Med. J. 2024, 29, 99–108. [Google Scholar] [CrossRef]
- El Gabali, T.M.; Jadain, O.A.; El Zubeir, I.E. Effect of addition of Syrian thyme (Thymus syriacus) on physiochemical and sensory quality of Sudanese Mudaffara cheese during storage. J. Food Sci. Technol. 2023, 60, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Lončar, B.; Pezo, L.; Iličić, M.; Kanurić, K.; Vukić, D.; Degenek, J.; Vukić, V. Modeling and Optimization of Herb-Fortified Fresh Kombucha Cheese: An Artificial Neural Network Approach for Enhancing Quality Characteristics. Foods 2024, 13, 548. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Zhou, W.; Yu, J.; Zhao, L.; Wang, K.; Hu, Z.; Liu, X. By-products of fruit and vegetables: Antioxidant properties of extractable and non-extractable phenolic compounds. Antioxidants 2023, 12, 418. [Google Scholar] [CrossRef]
- Faraoni, P.; Laschi, S. Bioactive compounds from agrifood by-products: Their use in medicine and biology. Int. J. Mol. Sci. 2024, 25, 5776. [Google Scholar] [CrossRef]
- Khursheed, S.; Akhtar, M.; Saeed, S.M.A.; Fazal, A.; Rehman, F.; Bashir, Z.; Khanam, U.; Hani, U.; Mustafa, H.M.; Haris, M.; et al. Valorization of pomegranate and banana by-products into value-added products: Influence on physicochemical, rheological, and consumer attributes. Indus. J. Biosci. Res. 2025, 3, 427–440. [Google Scholar] [CrossRef]
- Villamil-Galindo, E.; Van de Velde, F.; Piagentini, A.M. Strawberry agro-industrial by-products as a source of bioactive compounds: Effect of cultivar on the phenolic profile and the antioxidant capacity. Bioresour. Bioprocess. 2021, 8, 61. [Google Scholar] [CrossRef]
- Devatkal, S.K.; Narsaiah, K.; Borah, A. Effect of salt, kinnow and pomegranate fruit by-product extracts on colour and oxidative stability of raw chicken patties during refrigerated storage. J. Food Sci. Technol. 2011, 48, 472–477. [Google Scholar] [CrossRef]
- Nour, V.; Trandafir, I.; Cosmulescu, S. Antioxidant capacity, phenolic compounds and mineral content of blackcurrant (Ribes nigrum L.) leaves as influenced by harvesting date and extraction method. Ind. Crops Prod. 2014, 53, 133–139. [Google Scholar] [CrossRef]
- Lafarga, T.; Hayes, M. Bioactive peptides from meat muscle and by-products: Generation, functionality and application as functional ingredients. Meat Sci. 2014, 98, 227–239. [Google Scholar] [CrossRef]
- Toldrá, F.; Mora, L.; Reig, M. Current developments in meat by-products. In New Aspects of Meat Quality, 2nd ed.; Purslow, P., Ed.; Woodhead Publishing: Cambridge, UK, 2022; pp. 649–665. [Google Scholar]
- Sánchez, A.; Vázquez, A. Bioactive peptides: A review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Toldrá, F.; Mora, L.; Reig, M. New insights into meat by-product utilization. Meat Sci. 2016, 120, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Ryder, K.; Ha, M.; Bekhit, A.E.-D.; Carne, A. Characterization of novel fungal and bacterial protease preparations and evaluation of their ability to hydrolyse meat myofibrillar and connective tissue proteins. Food Chem. 2015, 172, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, Y.; Guo, Y.; Jiang, Y.; Wen, L.; Yang, B. New insights of fig (Ficus carica L.) as a potential functional food. Trends Food Sci. Technol. 2023, 140, 104146. [Google Scholar] [CrossRef]
- Rasool, I.F.u.; Aziz, A.; Khalid, W.; Koraqi, H.; Siddiqui, S.A.; Al-Farga, A.; Lai, W.-F.; Ali, A. Industrial application and health prospective of fig (Ficus carica) by-products. Molecules 2023, 28, 960. [Google Scholar] [CrossRef]
- Blejana, A.M.; Nour, V.; Păcularu-Burada, B.; Popescu, S.M. Wild bilberry, blackcurrant, and blackberry by-products as a source of nutritional and bioactive compounds. Int. J. Food Prop. 2023, 26, 1579–1595. [Google Scholar] [CrossRef]
- Pedišić, S.; Zorić, Z.; RepaJić, M.; Levaj, B.; Dobrincic, A.; Balbino, S.; Ćošić, Z.; Dragović-Uzelac, V.; Garofulić, I.E. Valorization of berry fruit by-products: Bioactive compounds, extraction, health benefits, encapsulation and food applications. Foods 2025, 14, 1354. [Google Scholar] [CrossRef]
- Pukalskienė, M.; Pukalskas, A.; Dienaitė, L.; Revinytė, S.; Pereira, C.V.; Matias, A.A.; Venskutonis, P.R. Recovery of bioactive compounds from strawberry (Fragaria × ananassa) pomace by conventional and pressurized liquid extraction and assessment of their bioactivity in human cell cultures. Foods 2021, 10, 1780. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.I.; Martins, C.F.; Amaral, R.A.; Brito, L.; Saraiva, J.A.; Vicente, A.A.; Moldão-Martins, M. Pineapple (Ananas comosus L.) by-products valorization: Novel bioingredients for functional foods. Molecules 2021, 26, 3216. [Google Scholar] [CrossRef]
- Wang, Z.; Fu, Q.; Hao, G.; Gu, Y.; Sun, T.; Gao, L.; Wang, B.; Wang, S.; Zheng, X.; Yang, Z.; et al. Physical aspects, phytochemical profiles, and nutritional properties of lemon (Citrus limon) slices under different drying technologies. Foods 2025, 14, 2586. [Google Scholar] [CrossRef]
- Noreen, S.; Hashmi, B.; Aja, P.M.; Atoki, A.V. Phytochemicals and pharmacology of pomegranate (Punica granatum L.): Nutraceutical benefits and industrial applications. Front. Nutr. 2025, 12, 1528897. [Google Scholar] [CrossRef]
- Aqilah, N.M.N.; Rovina, K.; Felicia, W.X.L.; Vonnie, J.M.A. A review on the potential bioactive components in fruits and vegetable wastes as value-added products in the food industry. Molecules 2023, 28, 2631. [Google Scholar] [CrossRef] [PubMed]
- Szabo, K.; Varvara, R.A.; Ciont, C.; Macri, A.M.; Vodnar, D.C. An updated overview on the revalorization of bioactive compounds derived from tomato production and processing by-products. J. Clean. Prod. 2025, 497, 145151. [Google Scholar] [CrossRef]
- Karastergiou, A.; Gancel, A.; Jourdes, M.; Teissedre, P. Transforming winemaking waste: Grape pomace as a sustainable source of bioactive compounds. OENO One 2025, 59, 2. [Google Scholar] [CrossRef]
- Zhao, Q.-C.; Zhao, J.-Y.; Ahn, D.U.; Jin, Y.-G.; Huang, X. Separation and identification of highly efficient antioxidant peptides from eggshell membrane. Antioxidants 2019, 8, 495. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, Z.; Ma, M.; Huang, X.; Guyonnet, V.; Xiong, H. Exploration of novel DPP-IV inhibitory peptides from discarded eggshell membrane: An integrated in silico and in vitro study. Food Biosci. 2024, 59, 104036. [Google Scholar] [CrossRef]
- Liu, R.; Wang, M.; Duan, J.-A. Antipyretic and antioxidant activities of the aqueous extract of Cornu bubali (water buffalo horn). Am. J. Chin. Med. 2010, 38, 293–306. [Google Scholar] [CrossRef]
- Adje, E.Y.; Balti, R.; Kouach, M.; Dhulster, P.; Guillochon, D.; Nedjar-Arroume, N. Obtaining antimicrobial peptides by controlled peptic hydrolysis of bovine hemoglobin. Int. J. Biol. Macromol. 2011, 49, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Ciurlia, L.; Bleve, M.; Rescio, L. Supercritical carbon dioxide co-extraction of tomatoes (Lycopersicum esculentum L.) and hazelnuts (Corylus avellana L.): A new procedure for obtaining a source of natural lycopene. J. Supercrit. Fluids 2009, 49, 338–344. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Sanchez-Zapata, E.; Sayas-Barberá, E.; Sendra, E.; Pérez-Álvarez, J.A.; Fernández-López, J. Tomato and tomato by-products: Human health benefits of lycopene and its application to meat products—A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1032–1049. [Google Scholar] [CrossRef]
- Sung, K.; Khan, S.A.; Nawaz, M.S.; Cerniglia, C.E.; Tamplin, M.L.; Phillips, R.W.; Kelley, L.C. Lysozyme as a barrier to growth of Bacillus anthracis strain Sterne in liquid egg white, milk and beef. Food Microbiol. 2011, 28, 1231–1234. [Google Scholar] [CrossRef]
- Barbiroli, A.; Bonomi, F.; Capretti, G.; Iametti, S.; Manzoni, M.; Piergiovanni, L.; Rollini, M. Antimicrobial activity of lysozyme and lactoferrin incorporated in cellulose-based food packaging. Food Control 2012, 26, 387–392. [Google Scholar] [CrossRef]
- Novais, C.; Molina, A.K.; Abreu, R.M.V.; Santo-Buelga, C.; Ferreira, I.C.F.R.; Pereira, C.; Barros, L. Natural food colorants and preservatives: A review, a demand, and a challenge. J. Agric. Food Chem. 2022, 70, 2789–2805. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; González-Aguilar, G.A. Use of additives to preserve the quality of fresh-cut fruits and vegetables. In Advances in Fresh-Cut Fruits and Vegetables Processing; CRC Press: Boca Raton, FL, USA, 2010; pp. 233–256. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Antonio, A.L.; Oliveira, M.B.P.P.; Ferreira, I.C.F.R. A comparative study between natural and synthetic antioxidants: Evaluation of their performance after incorporation into biscuits. Food Chem. 2017, 216, 342–346. [Google Scholar] [CrossRef] [PubMed]
- McCusker, M.M.; Durrani, K.; Payette, M.J.; Suchecki, J. An eye on nutrition: The role of vitamins, essential fatty acids, and antioxidants in age-related macular degeneration, dry eye syndrome, and cataract. Clin. Dermatol. 2016, 34, 276–285. [Google Scholar] [CrossRef]
- Roldán, E.; Sánchez-Moreno, C.; de Ancos, B.; Cano, M.P. Characterization of onion (Allium cepa L.) by-products as food ingredients with antioxidant and antibrowning properties. Food Chem. 2008, 108, 907–916. [Google Scholar] [CrossRef]
- Larrosa, M.; Llorach, R.; Espín, J.C.; Tomás-Barberán, F.A. Increase of antioxidant activity of tomato juice upon functionalisation with vegetable by-product extracts. LWT–Food Sci. Technol. 2002, 35, 532–542. [Google Scholar] [CrossRef]
- Chandel, V.; Biswas, D.; Roy, S.; Vaidya, D.; Verma, A.; Gupta, A. Current advancements in pectin: Extraction, properties and multifunctional applications. Foods 2022, 11, 2683. [Google Scholar] [CrossRef]
- Karim, A.A.; Bhat, R. Fish gelatin: Properties, challenges, and prospects as an alternative to mammalian gelatins. Food Hydrocoll. 2009, 23, 563–576. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Giménez, B.; López-Caballero, M.E.; Montero, M.P. Functional and bioactive properties of collagen and gelatin from alternative sources: A review. Food Hydrocoll. 2011, 25, 1813–1827. [Google Scholar] [CrossRef]
- Smithers, G.W. Whey-ing up the options—Yesterday, today and tomorrow. Int. Dairy J. 2015, 48, 2–14. [Google Scholar] [CrossRef]
- Gbogouri, G.; Linder, M.; Fanni, J.; Parmentier, M. Influence of hydrolysis degree on the functional properties of salmon by-products hydrolysates. J. Food Sci. 2004, 69, C615–C622. [Google Scholar] [CrossRef]
- Sathivel, S.; Bechtel, P.J.; Babbitt, J.; Prinyawiwatkul, W.; Negulescu, I.I.; Reppond, K.D. Properties of protein powders from arrowtooth flounder (Atheresthes stomias) and herring (Clupea harengus) by-products. J. Agric. Food Chem. 2004, 52, 5040–5046. [Google Scholar] [CrossRef]
- Sathivel, S.; Bechtel, P.J.; Babbitt, J.; Smiley, S.; Crapo, C.; Reppond, K.D.; Prinyawiwatkul, W. Biochemical and functional properties of herring (Clupea harengus) by-product hydrolysates. J. Food Sci. 2003, 68, 2196–2200. [Google Scholar] [CrossRef]
- Vieira, E.F.; Gomes, L.R.; Grosso, C.; Delerue-Matos, C. Recent advances on seaweed-derived pigments for food application and current legal framework. Foods 2025, 14, 3265. [Google Scholar] [CrossRef]
- Cazón, P.; Silva, A.S. Natural pigments from food wastes: New approaches for the extraction and encapsulation. Curr. Opin. Green Sustain. Chem. 2024, 47, 100929. [Google Scholar] [CrossRef]
- Ofori, J.A.; Hsieh, Y.-H.P. Issues related to the use of blood in food and animal feed. Crit. Rev. Food Sci. Nutr. 2014, 54, 687–697. [Google Scholar] [CrossRef]
- Boronat, Ò.; Sintes, P.; Celis, F.; Díez, M.; Ortiz, J.; Aguiló-Aguayo, I.; Martin-Gómez, H. Development of added-value culinary ingredients from fish waste: Fish bones and fish scales. Int. J. Gastron. Food Sci. 2023, 31, 100657. [Google Scholar] [CrossRef]
- Abdel-Moemin, A.R. Healthy cookies from cooked fish bones. Food Biosci. 2015, 12, 114–121. [Google Scholar] [CrossRef]
- Akalın, A.; Unal, G.; Dinkci, N.; Hayaloglu, A.A. Microstructural, textural, and sensory characteristics of probiotic yogurts fortified with sodium calcium caseinate or whey protein concentrate. J. Dairy Sci. 2012, 95, 3617–3628. [Google Scholar] [CrossRef]
- Zhang, T.; McCarthy, J.; Wang, G.; Liu, Y.; Guo, M. Physiochemical properties, microstructure, and probiotic survivability of nonfat goats’ milk yogurt using heat-treated whey protein concentrate as fat replacer. J. Food Sci. 2015, 80, M788–M794. [Google Scholar] [CrossRef] [PubMed]
- Saffon, M.; Richard, V.; Jiménez-Flores, R.; Gauthier, S.F.; Britten, M.; Pouliot, Y. Behavior of heat-denatured whey: Buttermilk protein aggregates during the yogurt-making process and their influence on set-type yogurt properties. Foods 2013, 2, 444–459. [Google Scholar] [CrossRef]
- Iriondo-DeHond, M.; Miguel, E.; del Castillo, M.D. Food by-products as sustainable ingredients for innovative and healthy dairy foods. Nutrients 2018, 10, 1358. [Google Scholar] [CrossRef]
- Saraç, M.G.; Dogan, M. Incorporation of dietary fiber concentrates from fruit and vegetable wastes in butter: Effects on physicochemical, textural, and sensory properties. Eur. Food Res. Technol. 2016, 242, 1331–1342. [Google Scholar] [CrossRef]
- Kaur, D.; Wani, A.A.; Singh, D.P.; Sogi, D.S. Shelf-life enhancement of butter, ice cream, and mayonnaise by addition of lycopene. Int. J. Food Prop. 2011, 14, 1217–1231. [Google Scholar] [CrossRef]
- Rizk, E.M.; El-Kady, A.T.; El-Bialy, A.R. Characterization of carotenoids (lyco-red) extracted from tomato peels and their use as natural colorants and antioxidants in ice cream. Ann. Agric. Sci. 2014, 59, 53–61. [Google Scholar] [CrossRef]
- Tseng, A.; Zhao, Y. Wine grape pomace as antioxidant dietary fiber for enhancing nutritional value and improving storability of yogurt and salad dressing. Food Chem. 2013, 138, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Anthocyanins, color and antioxidant properties of eggplant (Solanum melongena L.) and violet pepper (Capsicum annuum L.) peel extracts. Z. Naturforschung C 2006, 61, 527–535. [Google Scholar] [CrossRef]
- De Ancos, B.; Colina-Coca, C.; González-Peña, D.; Sánchez-Moreno, C. Bioactive compounds from vegetable and fruit by-products. In Biotechnology of Bioactive Compounds: Sources and Applications; Wiley: Hoboken, NJ, USA, 2015; pp. 1–36. [Google Scholar] [CrossRef]
- Ajila, C.; Leelavathi, K.; Rao, U.P. Improvement of dietary fiber content and antioxidant properties in soft dough biscuits with the incorporation of mango peel powder. J. Cereal Sci. 2008, 48, 319–326. [Google Scholar] [CrossRef]
- Tung, Y.-C.; Chang, W.-T.; Li, S.; Wu, J.-C.; Badmeav, V.; Ho, C.-T.; Pan, M.-H. Citrus peel extracts attenuate obesity and modulate gut microbiota in mice with high-fat diet-induced obesity. Food Funct. 2018, 9, 3363–3373. [Google Scholar] [CrossRef]
- Ghasemi, K.; Ghasemi, Y.; Ebrahimzadeh, M.A. Antioxidant activity, phenol and flavonoid contents of 13 citrus species peels and tissues. Pak. J. Pharm. Sci. 2009, 22, 277–281. [Google Scholar] [PubMed]
- Food and Agriculture Organization of the United Nations. What Is Organic Agriculture? Food and Agriculture Organization of the United Nations: Rome, Italy, 2020; Available online: http://www.fao.org/3/ad818s/ad818s03.htm (accessed on 17 June 2020).
- Gontijo, L.N.; da Silva, C.O.; Stort, L.G.; Duarte, R.M.T.; Betanho, C.; Tassi, E.M.M. Nutritional composition of vegetables grown in organic and conventional cultivation systems in Uberlândia, MG. Afr. J. Agric. Res. 2017, 12, 1848–1851. [Google Scholar] [CrossRef]
- Popa, M.E.; Mitelut, A.C.; Popa, E.E.; Stan, A.; Popa, V.I. Organic Foods Contribution to Nutritional Quality and Value. Trends Food Sci. Technol. 2019, 84, 15–18. [Google Scholar] [CrossRef]
- Reeve, J.R.; Hoagland, L.A.; Villalba, J.J.; Carr, P.M.; Atucha, A.; Cambardella, C.; Davis, D.R.; Delate, K. Organic Farming Soil. Health, and Food Quality: Considering Possible Links. Adv. Agron. 2016, 137, 319–368. [Google Scholar] [CrossRef]
- Manyaku, A.; Witbooi, H.; Laubscher, C.P. The significance of organic horticulture in mitigating climate change and promoting the production of healthier fruits and vegetables. Appl. Sci. 2024, 14, 4966. [Google Scholar] [CrossRef]
- Lairon, D. Nutritional quality and safety of organic food: A review. Agron. Sustain. Dev. 2010, 30, 33–41. [Google Scholar] [CrossRef]
- Rabiee, M.; Kaviani, B.; Sedaghathoor, S.; Eslami, A. Nutritional and qualitative comparison of temperate fruits from conventional and organic orchards. Sci. Rep. 2025, 15, 6835. [Google Scholar] [CrossRef]
- Suja, G.; Byju, G.; Jyothi, A.N.; Veena, S.S.; Sreekumar, J. Yield, quality and soil health under organic vs. conventional farming in taro. Sci. Hortic. 2017, 218, 334–343. [Google Scholar] [CrossRef]
- Gąstoł, M.; Domagała-Świątkiewicz, I.; Krosniak, M. Organic versus conventional—A comparative study on quality and nutritional value of fruit and vegetable juices. Biol. Agric. Hortic. 2011, 27, 310–319. [Google Scholar] [CrossRef]
- Kazimierczak, R.; Srednicka-Tober, D.; Hallmann, E.; Kopczynska, K.; Zarzynska, K. The impact of organic vs. conventional agricultural practices on selected quality features of eight potato cultivars. Agronomy 2019, 9, 799. [Google Scholar] [CrossRef]
- Lombardo, S.; Pandino, G.; Mauromicale, G. The influence of growing environment on the antioxidant and mineral content of early crop potato. J. Food Compos. Anal. 2013, 32, 28–35. [Google Scholar] [CrossRef]
- Vrček, I.V.; Čepo, D.V.; Rašić, D.; Peraica, M.; Žuntar, I.; Bojić, M.; Mendaš, G.; Medić-Šarić, M. A comparison of the nutritional value and food safety of organically and conventionally produced wheat flours. Food Chem. 2014, 143, 522–529. [Google Scholar] [CrossRef]
- Carillo, P.; Cacace, D.; Pascale, S.D.; Rapacciuolo, M.; Fuggi, A. Organic vs. traditional potato powder. Food Chem. 2012, 133, 1264–1273. [Google Scholar] [CrossRef]
- Verma, R.; Saini, J.P.; Dhaliwal, Y.S.; Bhat, F.M. Comparative study of organic and inorganic legumes for their chemical composition, nutritional and amino acid profiling—An Analytical Study. J. Hum. Nutr. Food Sci. 2023, 11, 1173. [Google Scholar] [CrossRef]
- Maggio, A.; Carillo, P.; Bulmetti, G.S.; Fuggi, A.; Barbieri, G.; Pascale, S.D. Potato yield and metabolic profiling under conventional and organic farming. Eur. J. Agron. 2008, 28, 343–350. [Google Scholar] [CrossRef]
- Hunter, D.; Foster, M.; McArthur, J.O.; Ojha, R.; Petocz, P.; Samman, S. Evaluation of the micronutrient composition of plant foods produced by organic and conventional agricultural methods. Crit. Rev. Food Sci. Nutr. 2011, 51, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Guo, L.; Jiang, G.; Song, Y.; Muminov, M.A. Advances of organic products over conventional productions with respect to nutritional quality and food security. Acta Ecol. Sin. 2018, 38, 53–60. [Google Scholar] [CrossRef]
- Warman, P.R.; Havard, K.A. Yield, vitamin and mineral contents of organically and conventionally grown potatoes and sweet corn. Agric. Ecosyst. Environ. 1998, 68, 207–216. [Google Scholar] [CrossRef]
- Araújo, D.F.d.S.; da Silva, A.M.R.B.; Lima, L.L.d.A.; Vasconcelos, M.A.d.S.; Andrade, S.A.C.; Sarubbo, L.A. The concentration of minerals and physicochemical contaminants in conventional and organic vegetables. Food Control 2014, 44, 242–248. [Google Scholar] [CrossRef]
- Sheng, J.P.; Liu, C.; Shen, L. Analysis of nutrients and minerals in organic and traditional cherry tomato by ICP-OES method. Spectrosc. Spectr. Anal. 2009, 29, 2244–2246. [Google Scholar]
- Reddy, A.A.; Vimala, Y.; Goudar, G.; Mergu, N.; Rao, J.S. Nutritional compilation of commonly consumed organic and conventional fruits and vegetables from India. Food Humanit. 2023, 1, 1652–1658. [Google Scholar] [CrossRef]
- Worthington, V. Nutritional quality of organic versus conventional fruits, vegetables, and grains. J. Altern. Complement. Med. 2001, 7, 161–173. [Google Scholar] [CrossRef]
- de González, M.T.N.; Osburn, W.N.; Hardin, M.D.; Longnecker, M.; Garg, H.K.; Bryan, N.S.; Keeton, J.T. A survey of nitrate and nitrite concentrations in conventional and organic-labeled raw vegetables at retail. J. Food Sci. 2015, 80, C942–C949. [Google Scholar] [CrossRef]
- Chausali, N.; Saxena, J. Conventional versus organic farming: Nutrient status. In Advances in Organic Farming; Woodhead Publishing: Cambridge, UK, 2021; pp. 241–254. [Google Scholar] [CrossRef]
- Mohamed, N. Determination of nitrate content in organic and conventionally grown vegetable leaves in Sri Lanka using spectrophotometry. J. Nutr. Health Sci. 2017, 1, 16. [Google Scholar]
- Xu, M.J.; He, W.L. Effects of organic, special and conventional farming systems on vegetable quality. Acta Agric. Jiangxi 2009, 21, 68–70. [Google Scholar]
- Calderon, R.; García-Hernández, J.; Palma, P.; Leyva-Morales, J.B.; Godoy, M.; Zambrano-Soria, M.; Bastidas-Bastidas, P.J.; Valenzuela, G. Heavy metals and metalloids in organic and conventional vegetables from Chile and Mexico: Implications for human health. J. Food Compos. Anal. 2023, 123, 105527. [Google Scholar] [CrossRef]
- Brazinskiene, V.; Asakaviciute, R.; Miezeliene, A.; Alencikiene, G.; Ivanauskas, L.; Jakstas, V.; Viskelis, P.; Razukas, A. Effect of farming systems on yield, quality parameters and sensory properties of conventionally and organically grown potato (Solanum tuberosum L.) tubers. Food Chem. 2014, 145, 903–909. [Google Scholar] [CrossRef]
- Czech, A.; Szmigielski, M.; Sembratowicz, I. Nutritional value and antioxidant capacity of organic and conventional vegetables of the genus. Sci. Rep. 2022, 12, 18713. [Google Scholar] [CrossRef]
- Cruz-Carrión, Á.; Ruiz de Azua, M.J.; Muguerza, B.; Mulero, M.; Bravo, F.I.; Arola-Arnal, A.; Suarez, M. Organic vs. Non-Organic Plant-Based Foods—A Comparative Study on Phenolic Content and Antioxidant Capacity. Plants 2023, 12, 183. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, C.; Wilches-Pérez, D.; Hallman, E.; Kazimierczak, R.; Rembiałkowska, E. Organic versus conventional beetroot: Bioactive compounds and antioxidant properties. LWT 2019, 116, 108552. [Google Scholar] [CrossRef]
- Faller, A.L.K.; Fialho, E. Polyphenol content and antioxidant capacity in organic and conventional plant foods. J. Food Compos. Anal. 2010, 23, 561–568. [Google Scholar] [CrossRef]
- Carbonaro, M.; Mattera, M.; Nicoli, S.; Bergamo, P.; Cappelloni, M. Modulation of antioxidant compounds in organic vs. conventional fruit (Peach, Prunus persica L., and Pear, Pyrus communis L.). J. Agric. Food Chem. 2002, 50, 5458–5462. [Google Scholar] [CrossRef]
- Barański, M.; Rempelos, L.; Iversen, P.O.; Leifert, C. Effects of organic food consumption on human health; The jury is still out! Food Nutr. Res. 2017, 61, 1287333. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.D.; Li, J.; Xiong, R.-G.; Saimaiti, A.; Huang, S.-Y.; Wu, S.-X.; Yang, Z.-J.; Shang, A.; Zhao, C.-N.; Gan, R.-Y.; et al. Bioactive compounds, health benefits and food applications of grape. Foods 2022, 11, 2755. [Google Scholar] [CrossRef]
- Zaky, A.A.; Akram, M.U.; Rybak, K.; Witrowa-Rajchert, D.; Nowacka, M. Bioactive compounds from plants and by-products: Novel extraction methods, applications, and limitations. AIMS Mol. Sci. 2024, 11, 150–188. [Google Scholar] [CrossRef]
- Majida, I.; Khana, S.; Aladelb, A.; Dara, A.H.; Adnan, M.; Khand, M.I.; Awadelkareem, A.M.; Ashraf, S.A. Recent insights into green extraction techniques as efficient methods for the extraction of bioactive components and essential oils from foods. J. Food 2023, 21, 101–114. [Google Scholar] [CrossRef]
- Herzyk, F.; Piłakowska-Pietras, D.; Korzeniowska, M. Supercritical extraction techniques for obtaining biologically active substances from a variety of plant by-products. Foods 2024, 13, 1713. [Google Scholar] [CrossRef]
- Ranade, A.; Malav, O.P. Fermented meat products—A review. Int. J. Adv. Biochem. Res. 2025, 9, 1003–1011. [Google Scholar] [CrossRef]
- Flores, M.; Piornos, J.A. Fermented meat sausages and the challenge of their plant-based alternatives: A comparative review on aroma-related aspects. Meat Sci. 2021, 182, 108636. [Google Scholar] [CrossRef]
- Sirini, N.; Munekata, P.E.S.; Lorenzo, J.M.; Stegmayer, M.Á.; Pateiro, M.; Pérez-Álvarez, J.Á.; Sepúlveda, N.; Sosa-Morales, M.E.; Teixeira, A.; Fernández-López, J.; et al. Development of Healthier and Functional Dry Fermented Sausages: Present and Future. Foods 2022, 11, 1128. [Google Scholar] [CrossRef] [PubMed]
- Karwowska, M.; Munekata, P.E.S.; Lorenzo, J.M.; Tomasevic, I. Functional and Clean Label Dry Fermented Meat Products: Phytochemicals, Bioactive Peptides, and Conjugated Linoleic Acid. Appl. Sci. 2022, 12, 5559. [Google Scholar] [CrossRef]
- Toldrá, F. Biochemistry of Fermented Meat. In Food Biochemistry and Food Processing, 2nd ed.; Simpson, B.K., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 331–343. [Google Scholar] [CrossRef]
- Skowron, K.; Budzyńska, A.; Grudlewska-Buda, K.; Wiktorczyk-Kapischke, N.; Andrzejewska, M.; Wałecka-Zacharska, E.; Gospodarek-Komkowska, E. Two Faces of Fermented Foods—The Benefits and Threats of Its Consumption. Front. Microbiol. 2022, 13, 845166. [Google Scholar] [CrossRef]
- Šojić, B.V.; Petrović, L.S.; Mandić, A.I.; Sedej, I.J.; Džinić, N.R.; Tomović, V.M.; Jokanović, M.R.; Tasić, T.A.; Škaljac, S.B.; Ikonić, P.M. Lipid oxidative changes in traditional dry fermented sausage Petrovská klobása during storage. Hem. Ind. 2014, 68, 27–34. [Google Scholar] [CrossRef]
- Coton, M.; Deniel, F.; Mounier, J.; Joubrel, R.; Robieu, E.; Pawtowski, A.; Jeuge, S.; Taminiau, B.; Daube, G.; Coton, E.; et al. Microbial ecology of French dry fermented sausages and mycotoxin risk evaluation during storage. Front. Microbiol. 2021, 12, 737140. [Google Scholar] [CrossRef]
- Ji, X.; Liu, J.; Li, J.; Xiaoxia, F. The hidden diet: Synthetic antioxidants in packaged food and their impact on human exposure and health. Environ. Int. 2024, 186, 108613. [Google Scholar] [CrossRef]
- European Union. European Commission Regulation (EU) 2018/1481 of 4 October 2018 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council and the Annex to Commission Regulation (EU) No 231/2012 as regards octyl gallate (E 311) and dodecyl gallate (E 312). Off. J. Eur. Union 2018, L251, 13–18. [Google Scholar]
- Zhang, Y.; Zhang, Y.; Jia, J.; Peng, H.; Qian, Q.; Pan, Z.; Liu, D. Nitrite and nitrate in meat processing: Functions and alternatives. Curr. Res. Nutr. Food Sci. 2023, 6, 100470. [Google Scholar] [CrossRef]
- European Union; European Commission. Commission Regulation (EU) No 1129/2011 of 11 November 2011 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council by establishing a Union list of food additives. Off. J. Eur. Union 2011, 54, 205–211. [Google Scholar]
- Boruah, B.; Ray, S. Current progress in the valorization of food industrial by-products for the development of functional food products. Food Sci. Appl. Biotech. 2024, 7, 289–317. [Google Scholar] [CrossRef]
- Prakash, A.; Baskaran, R. Acerola, an untapped functional superfruit: A review on latest frontiers. J. Food Sci. Technol. 2018, 55, 3373–3384. [Google Scholar] [CrossRef]
- Monteiro, S.A.; Barbosa, M.M.; da Silva, F.F.M.; Bezerra, R.F.; da Silva Maia, K. Preparation, phytochemical and bromatological evaluation of flour obtained from the acerola (Malpighia punicifolia) agroindustrial residue with potential use as fiber source. LWT 2020, 134, 110142. [Google Scholar] [CrossRef]
- da Silva, N.C.; Dourado, P.L.A.; Tosi, M.M. A review about acerola (Malpighia emarginata DC) by-products as a promising raw material for the generation of green products. Braz. J. Food Technol. 2023, 26, e2023039. [Google Scholar] [CrossRef]
- Rezende, Y.R.R.S.; Nogueira, J.P.; Narain, N. Microencapsulation of extracts of bioactive compounds obtained from acerola (Malpighia emarginata DC) pulp and residue by spray and freeze drying: Chemical, morphological and chemometric characterization. Food Chem. 2018, 254, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidants: A comprehensive review. Arch. Toxicol. 2025, 99, 1893–1997. [Google Scholar] [CrossRef]
- Shao, H.-B.; Chu, L.-Y.; Lu, Z.-H.; Kang, C.-M. Primary antioxidant free radical scavenging and redox signaling pathways in higher plant cells. Int. J. Biol. Sci. 2007, 7, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Choe, E.; Min, D.B. Mechanisms of antioxidants in the oxidation of foods. Compr. Rev. Food Sci. Food Saf. 2009, 8, 345–358. [Google Scholar] [CrossRef]
- Gao, X.; Xiao, Y.; Li, W.; Xu, L.; Yuan, J. Synergistic effects of antioxidant blends: A comparative study on oxidative stability of lipids in feed matrices. Antioxidants 2025, 14, 981. [Google Scholar] [CrossRef] [PubMed]
- Bugatti, V.; Brachi, P.; Viscusi, G.; Gorrasi, G. Valorization of tomato processing residues through the production of active bio-composites for packaging. Appl. Front. Mater. 2019, 6, 34. [Google Scholar] [CrossRef]
- Szabo, K.; Cătoi, A.F.; Vodnar, D.C. Bioactive compounds extracted from tomato processing by-products as a source of valuable nutrients. Plant Foods Hum. Nutr. 2018, 73, 268–277. [Google Scholar] [CrossRef]
- Eslami, E.; Carpentieri, S.; Pataro, G.; Ferrari, G. A comprehensive overview of tomato processing by-product valorization by conventional methods versus emerging technologies. Foods 2023, 12, 166. [Google Scholar] [CrossRef]
- Skwarek, P.; Karwowska, M. Fatty acids profile and antioxidant properties of raw fermented sausages with the addition of tomato pomace. Biomolecules 2022, 12, 1695. [Google Scholar] [CrossRef]
- Skwarek, P.; Karwowska, M. The Effect of Tomato Pomace on the Oxidative and Microbiological Stability of Raw Fermented Sausages with Reduced Addition of Nitrites. Int. J. Food Sci. 2025, 2025, 6146090. [Google Scholar] [CrossRef]
- Doménech-Asensi, G.; García-Alonso, J.; Martínez, E.; Santaella, M.; Martín-Pozuelo, G.; Bravo, S.; Periago, M.J. Effect of the Addition of Tomato Paste on the Nutritional and Sensory Properties of Mortadela. Meat Sci. 2013, 93, 213–219. [Google Scholar] [CrossRef]
- Calvo, M.M.; García, M.L.; Selgas, M.D. Dry Fermented Sausages Enriched with Lycopene from Tomato Peel. Meat Sci. 2008, 80, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Ghafouri-Oskuei, H.; Javadi, A.; Asl, M.R.S.; Azadmard-Damirchi, S.; Armin, M. Quality Properties of Sausage Incorporated with Flaxseed and Tomato Powders. Meat Sci. 2020, 161, 107957. [Google Scholar] [CrossRef] [PubMed]
- Lopes, J.d.C.; Madureira, J.; Margaca, F.M.A.; Cabo Verde, S. Grape Pomace: A Review of Its Bioactive Phenolic Compounds, Health Benefits, and Applications. Molecules 2025, 30, 362. [Google Scholar] [CrossRef]
- Bhutani, M.; Gaur, S.S.; Shams, R.; Dash, K.K.; Shaikh, A.M.; Bela, K. Valorization of Grape By-Products: Insights into Sustainable Industrial and Nutraceutical Applications. Future Food 2025, 12, 100710. [Google Scholar] [CrossRef]
- Caponio, G.R.; Minervini, F.; Tamma, G.; Gambacorta, G.; De Angelis, M. Promising Application of Grape Pomace and Its Agri-Food Valorization: Source of Bioactive Molecules with Beneficial Effects. Sustainability 2023, 15, 9075. [Google Scholar] [CrossRef]
- Silva, V.; Igrejas, G.; Falco, V.; Santos, T.; Torres, C.; Oliveira, A.; Pereira, E.J.; Amaral, S.A.; Poeta, P. Chemical Composition, Antioxidant and Antimicrobial Activity of Phenolic Compounds Extracted from Wine Industry By-Products. Food Control 2018, 92, 516–522. [Google Scholar] [CrossRef]
- Di Stefano, V.; Buzzanca, C.; Melilli, M.G.; Indelicato, S.; Mauro, M.; Vazzana, M.; Arizza, V.; Lucarini, M.; Durazzo, A.; Bongiorno, D. Polyphenol Characterization and Antioxidant Activity of Grape Seeds and Skins from Sicily: A Preliminary Study. Sustainability 2022, 14, 6702. [Google Scholar] [CrossRef]
- Jebur, A.B.; El-Sayed, R.A.; El-Demerdash, F.M. Grape Seeds: Nutritional Value, Health Advantages, and Industrial Applications. Food Med. Homol. 2026, 3, 9420100. [Google Scholar] [CrossRef]
- Songsermsakul, P.; Pornphairin, E.; Porasuphatana, S. Comparison of antioxidant activity of grape seed extract and fruits containing high β-carotene, vitamin C, and E. Int. J. Food Prop. 2013, 16, 643–648. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; González-Rodríguez, R.M.; Sánchez, M.; Amado, I.R.; Franco, D. Effects of Natural (Grape Seed and Chestnut Extract) and Synthetic Antioxidants (Buthylated Hydroxytoluene, BHT) on the Physical, Chemical, Microbiological and Sensory Characteristics of Dry Cured Sausage “Chorizo”. Food Res. Int. 2013, 54, 611–620. [Google Scholar] [CrossRef]
- Ivanov, Y.; Godjevargova, T.; Atanasova, M.; Nakov, G. The Effect of Grape Seed Extract on Lipid Oxidation, Color Change, and Microbial Growth in a Beef–Pork Sausage Model System. Molecules 2025, 30, 1739. [Google Scholar] [CrossRef] [PubMed]
- Fadhil, Y.S. Effect of Grape Seed Extract on the Quality of Local Meat Product (Basturma) during Storage. Food Sci. Technol. 2023, 43, e4423. [Google Scholar] [CrossRef]
- El-Zainy, A.; Morsy, A.; Sedki, A. Polyphenols Extracted from Grape Seeds and Its Effects as Antioxidant and Antimicrobial on Beef Sausage. J. Food Dairy Sci. 2016, 7, 19–25. [Google Scholar] [CrossRef]
- D’Arrigo, M.; Petrón, M.J.; Delgado-Adámez, J.; García-Parra, J.J.; Martín-Mateos, M.J.; Ramírez-Bernabé, M.R. Dry-Cured Sausages “Salchichón” Manufactured with a Valorized Ingredient from Red Grape Pomace (Var. Tempranillo). Foods 2024, 13, 3133. [Google Scholar] [CrossRef] [PubMed]
- Skinner, R.C.; Gigliotti, J.C.; Ku, K.-M.; Tou, J.C. A Comprehensive Analysis of the Composition, Health Benefits, and Safety of Apple Pomace. Nutr. Rev. 2018, 76, 893–909. [Google Scholar] [CrossRef]
- Gonelimali, F.D.; Szabo-Notin, B.; Szalokidorko, L.; Ribarski, A.; Mate, M. Evaluation of Polyphenol Extraction from Apple Pomace. AGROFOR Int. J. 2021, 6, 103–110. [Google Scholar] [CrossRef]
- Gonelimali, F.D.; Szabo-Notin, B.; Mate, M. Optimal Drying Conditions for Valorization of Industrial Apple Pomace: Potential Source of Food Bioactive Compounds. Prog. Agric. Eng. Sci. 2021, 17, 69–75. [Google Scholar] [CrossRef]
- Jung, J.; Cavender, G.; Zhao, Y. Impingement Drying for Preparing Dried Apple Pomace Flour and Its Fortification in Bakery and Meat Products. J. Food Sci. Technol. 2015, 52, 5568–5578. [Google Scholar] [CrossRef] [PubMed]
- Pascoalino, L.A.; Finimundy, T.C.; Pires, T.C.; Pereira, C.; Barros, L.; Ferreira, I.C.; Oliveira, M.B.P.; Barreira, J.C.; Reis, F.S. Investigating Bioactive Compounds in Apple Pomace: Potential to Develop High Added-Value Products from an Industrial Surplus. Food Biosci. 2025, 63, 105664. [Google Scholar] [CrossRef]
- Ciurlă, L.; Enache, I.-M.; Buterchi, I.; Mihalache, G.; Lipsa, F.D.; Patras, A. A New Approach to Recover Bioactive Compounds from Apple Pomace: Healthy Jelly Candies. Foods 2025, 14, 39. [Google Scholar] [CrossRef]
- Koishybayeva, A.; Korzeniowska, M. Utilization and Effect of Apple Pomace Powder on Quality Characteristics of Turkey Sausages. Foods 2024, 13, 2807. [Google Scholar] [CrossRef]
- Grispoldi, L.; Ianni, F.; Blasi, F.; Pollini, L.; Crotti, S.; Cruciani, D.; Cenci-Goga, B.T.; Cossignani, L. Apple Pomace as Valuable Food Ingredient for Enhancing Nutritional and Antioxidant Properties of Italian Salami. Antioxidants 2022, 11, 1221. [Google Scholar] [CrossRef]
- Younis, K.; Ahmad, S. Waste Utilization of Apple Pomace as a Source of Functional Ingredient in Buffalo Meat Sausage. Cogent Food Agric. 2015, 1, 1119397. [Google Scholar] [CrossRef]
- Rashwan, A.K.; Bai, H.; Osman, A.I.; Eltohamy, K.M.; Chen, Z.; Younis, H.A.; Al-Fatesh, A.; Rooney, D.W.; Yap, P.S. Recycling Food and Agriculture By-Products to Mitigate Climate Change: A Review. Environ. Chem. Lett. 2023, 21, 3351–3375. [Google Scholar] [CrossRef]
- Vilas-Boas, A.A.; Magalhães, D.; Campos, D.A.; Porretta, S.; Dellapina, G.; Poli, G.; Istanbullu, Y.; Demir, S.; San Martín, Á.M.; García-Gómez, P.; et al. Innovative Processing Technologies to Develop a New Segment of Functional Citrus-Based Beverages: Current and Future Trends. Foods 2022, 11, 3859. [Google Scholar] [CrossRef]
- Rao, N.; Sharma, M.; Sharma, A. Development of Product Rich in Dietary Fiber and Antioxidant Prepared from Lemon Peel. Glob. J. Biol. Agric. Health Sci. 2016, 5, 120–123. [Google Scholar]
- Yang, Y.-Y.; Ma, S.; Wang, X.-X.; Zheng, X.-L. Modification and Application of Dietary Fiber in Foods. J. Chem. 2017, 2017, 9340427. [Google Scholar] [CrossRef]
- Fernández-López, J.; Sendra, E.; Sayas-Barberá, E.; Navarro, C.; Pérez-Alvarez, J.A. Physico-Chemical and Microbiological Profiles of “Salchichón” (Spanish Dry-Fermented Sausage) Enriched with Orange Fiber. Meat Sci. 2008, 80, 410–417. [Google Scholar] [CrossRef]
- Magalhães, D.; Rodrigues, C.V.; Botella-Martinez, C.; Muñoz-Tebar, N.; Pérez-Álvarez, J.A.; Viuda-Martos, M.; Teixeira, P.; Pintado, M. Lemon Dietary Fibre-Based Powder as a Promising Ingredient for the Food Industry: Enhancing Mortadella Nutritional Quality. Foods 2025, 14, 1693. [Google Scholar] [CrossRef]
- Delgado-Pando, G.; Ekonomou, S.I.; Stratakos, A.C.; Pintado, T. Clean Label Alternatives in Meat Products. Foods 2021, 10, 1615. [Google Scholar] [CrossRef]
- Kim, M.; Bae, S.M.; Yoo, Y.; Park, J.; Jeong, Y.J. Clean-Label Strategies for the Replacement of Nitrite, Ascorbate, and Phosphate in Meat Products: A Review. Foods 2025, 14, 2442. [Google Scholar] [CrossRef]
- Jeong, J.Y.; Bae, S.M.; Yoon, J.; Jeong, D.H.; Gwak, S.H. Effect of Using Vegetable Powders as Nitrite/Nitrate Sources on the Physicochemical Characteristics of Cooked Pork Products. Food Sci. Anim. Resour. 2020, 40, 831–843. [Google Scholar] [CrossRef]
- Pennisi, L.; Verrocchi, E.; Paludi, D.; Vergara, A. Effects of Vegetable Powders as Nitrite Alternative in Italian Dry Fermented Sausage. Ital. J. Food Saf. 2020, 9, 8422. [Google Scholar] [CrossRef]
- Alahakoon, A.U.; Jayasena, D.D.; Ramachandra, S.; Jo, C. Alternatives to Nitrite in Processed Meat: Up to Date. Trends Food Sci. Technol. 2015, 45, 37–49. [Google Scholar] [CrossRef]
- Alessi, A.C.S.B.; Brito, N.L.H.; Droval Arcain, A.A.; Reitz Cardoso, F.A. Evaluation of Celery Extract as a Natural Alternative to Sodium Nitrite in Fresh Chicken Sausages during Refrigerated Storage. J. Sci. Food Agric. 2025, 106, 2251–2258. [Google Scholar] [CrossRef]
- Bhusal, A.; Muriana, P.M. Isolation and Characterization of Nitrate Reducing Bacteria for Conversion of Vegetable-Derived Nitrate to ‘Natural Nitrite’. Appl. Microbiol. 2021, 1, 11–23. [Google Scholar] [CrossRef]
- Nguyen, N.T.T.; Nguyen, B.X.; Habibi, N.; Dabirimirhosseinloo, M.; Oliveira, L.d.A.; Terada, N.; Sanada, A.; Kamata, A.; Koshio, K. Effect of Organic and Synthetic Fertilizers on Nitrate, Nitrite, and Vitamin C Levels in Leafy Vegetables and Herbs. Plants 2025, 14, 917. [Google Scholar] [CrossRef]
- Ibrahim, M.N.; Jaafar, H.Z.E.; Karimi, E.; Ghasemzadeh, A. Impact of Organic and Inorganic Fertilizers Application on the Phytochemical and Antioxidant Activity of Kacip Fatimah (Labisia pumila Benth). Molecules 2013, 18, 10973–10988. [Google Scholar] [CrossRef]
- Alkazzaz, S.R.M.A.; Abdelrahman, H.A.; Ahmed, A.M.; Dora, E.-D.H.I.; Helal, I.M.; Ahmed, N.I.H. Effect of Replacing Sodium Nitrite with Celery on Sensory and Chemical Quality of Popular Dry Sausage. J. Adv. Vet. Res. 2022, 12, 404–408. [Google Scholar]
- Jin, S.-K.; Choi, J.S.; Yang, H.-S.; Park, T.-S.; Yim, D.-G. Natural Curing Agents as Nitrite Alternatives and Their Effects on the Physicochemical, Microbiological Properties and Sensory Evaluation of Sausages during Storage. Meat Sci. 2018, 146, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Posthuma, J.A.; Rasmussen, F.D.; Sullivan, G.A. Effects of Nitrite Source, Reducing Compounds, and Holding Time on Cured Color Development in a Cured Meat Model System. LWT 2018, 95, 47–50. [Google Scholar] [CrossRef]
- Katmer, B.; Kaya, M. The Effect of Swiss Chard Powder as a Curing Agent on Volatile Compound Profile and Other Qualitative Properties of Heat-Treated Sucuk. Foods 2025, 14, 3785. [Google Scholar] [CrossRef]
- Sheng, S.; Silva, E.M.; Ricke, S.C.; Claus, J.R. Characterization of Volatilized Compounds in Conventional and Organic Vegetable-Source Alternative Meat-Curing Ingredients. Molecules 2025, 30, 835. [Google Scholar] [CrossRef]
- Cau, S.; Tilocca, M.G.; Spanu, C.; Soro, B.; Tedde, T.; Salza, S.; Melillo, R.; Piras, G.; Virgilio, S.; Vodret, B.; et al. Detection of Celery (Apium graveolens) Allergen in Foods of Animal and Plant Origin by Droplet Digital PCR Assay. Food Control 2021, 130, 108407. [Google Scholar] [CrossRef]
- Sheng, S.; Silva, E.M.; Ricke, S.C.; Claus, J.R. Organic and Conventional Alternative Curing Ingredients Effects on Quality and Sensory Attributes of Deli-Style Turkey. Poult. Sci. 2025, 104, 105370. [Google Scholar] [CrossRef]
- Dilek, N.M.; Karakaya, M. Natural Alternative Curing Agent in Fermented Sucuk Production: Sugar Beet (Beta vulgaris var. saccharifera L.) Molasses. Selcuk J. Agric. Food Sci. 2022, 36, 380–386. [Google Scholar] [CrossRef]
- Lau, A.T.Y.; Arvaj, L.; Strange, P.; Goodwin, M.; Barbut, S.; Balamurugan, S. Effect of Cranberry Pomace on the Physicochemical Properties and Inactivation of Salmonella during the Manufacture of Dry Fermented Sausages. Curr. Res. Food Sci. 2021, 4, 636–645. [Google Scholar] [CrossRef]
- Zhou, J.; Li, D.; Zhang, X.; Liu, C.; Chen, Y. Valorization of Protein-Rich Waste and Its Application. Sci. Total Environ. 2023, 901, 166141. [Google Scholar] [CrossRef]
- Karwowska, M.; Kononiuk, A. Addition of Acid Whey Improves Organic Dry-Fermented Sausage without Nitrite Production and Its Nutritional Value. Int. J. Food Sci. Technol. 2018, 53, 246–253. [Google Scholar] [CrossRef]
- Selani, M.M.; Herrero, A.M.; Ruiz-Capillas, C. Plant Antioxidants in Dry Fermented Meat Products with a Healthier Lipid Profile. Foods 2022, 11, 3558. [Google Scholar] [CrossRef]
- Stadnik, J.; Kęska, P.; Gazda, P.; Siłka, L.; Kołożyn-Krajewska, D. Influence of LAB Fermentation on the Color Stability and Oxidative Changes in Dry-Cured Meat. Appl. Sci. 2022, 12, 11736. [Google Scholar] [CrossRef]
- Faustino, M.; Veiga, M.; Sousa, P.; Costa, E.M.; Silva, S.; Pintado, M. Agro-Food Byproducts as a New Source of Natural Food Additives. Molecules 2019, 24, 1056. [Google Scholar] [CrossRef]
- Valdés, A.; Burgos, N.; Jiménez, A.; Garrigós, M.C. Natural Pectin Polysaccharides as Edible Coatings. Coatings 2015, 5, 865–886. [Google Scholar] [CrossRef]
- Ahmad, S.S.; Khalid, M.; Younis, K. Interaction Study of Dietary Fibers (Pectin and Cellulose) with Meat Proteins Using Bioinformatics Analysis: An In-Silico Study. LWT 2020, 119, 108889. [Google Scholar] [CrossRef]
- Huynh, D.T.; Tran, U.P.N. Low-Fat Vietnamese Sausage (Lap Xuong) Formulated with Passion-Fruit Peel Pectin as a Sustainable Fat Replacer. Chem. Eng. Trans. 2015, 122, 319–324. [Google Scholar] [CrossRef]
- Méndez-Zamora, G.; García-Macías, J.A.; Santellano-Estrada, E.; Chávez-Martínez, A.; Durán-Meléndez, L.A.; Silva-Vázquez, R.; Quintero-Ramos, A. Fat Reduction in the Formulation of Frankfurter Sausages Using Inulin and Pectin. Food Sci. Technol. Camp. 2015, 35, 25–31. [Google Scholar] [CrossRef]
- Lashgari, S.S.; Noorolahi, Z.; Sahari, M.A.; Gavlighi, H.A. Improvement of Oxidative Stability and Textural Properties of Fermented Sausage via Addition of Pistachio Hull Extract. Food Sci. Nutr. 2020, 8, 2920–2928. [Google Scholar] [CrossRef] [PubMed]
- Fiorentini, M.; Kinchla, A.J.; Nolden, A.A. Role of Sensory Evaluation in Consumer Acceptance of Plant-Based Meat Analogs and Meat Extenders: A Scoping Review. Foods 2020, 9, 1334. [Google Scholar] [CrossRef] [PubMed]
- Bouked, F. Chickpea (Cicer arietinum L.) Protein as a Prospective Plant-Based Ingredient: A Review. Food Sci. Technol. 2021, 56, 5435–5444. [Google Scholar] [CrossRef]
- Marques, C.; Correia, E.; Dinis, L.-T.; Vilela, A. An Overview of Sensory Characterization Techniques: From Classical Descriptive Analysis to the Emergence of Novel Profiling Methods. Foods 2022, 11, 255. [Google Scholar] [CrossRef]
- Ray, S. Sensory Properties of Foods and Their Measurement Methods. In Techniques to Measure Food Safety and Quality; Khan, M.S., Rahman, M.S., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2021; pp. 345–381. [Google Scholar] [CrossRef]
- Drake, M.A.; Delahunty, C.M. Chapter 20—Sensory Character of Cheese and Its Evaluation. In Cheese: Chemistry, Physics and Microbiology, 4th ed.; Academic Press: London, UK, 2017; pp. 517–545. [Google Scholar] [CrossRef]
- Pereira, A.; Lee, H.C.; Lammert, R., Jr.; Wolberg, C., Jr.; Ma, D.; Immoos, C.; Casassa, F.; Kang, I. Effects of Red-Wine Grape Pomace on the Quality and Sensory Attributes of Beef Hamburger Patty. Food Sci. Technol. 2022, 57, 1814–1823. [Google Scholar] [CrossRef]
- Parrini, S.; Sirtori, F.; Acciaioli, A.; Becciolini, V.; Crovetti, A.; Franci, O.; Romani, A.; Scardigli, A.; Bozzi, R. Effect of Replacement of Synthetic vs. Natural Curing Agents on Quality Characteristics of Cinta Senese Frankfurter-Type Sausage. Animals 2020, 10, 14. [Google Scholar] [CrossRef]
- Šojić, B.; Pavlić, B.; Tomović, V.; Kočić-Tanackov, S.; Đurović, S.; Zeković, Z.; Belović, M.; Torbica, A.; Jokanović, M.; Urumović, N.; et al. Tomato Pomace Extract and Organic Peppermint Essential Oil as Effective Sodium Nitrite Replacement in Cooked Pork Sausages. Food Chem. 2020, 330, 127202. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ivanov, Y.; Atanasova, M.; Godjevargova, T. Development of Functional Fermented Meat Products Using Agro-Food Byproducts. Processes 2026, 14, 602. https://doi.org/10.3390/pr14040602
Ivanov Y, Atanasova M, Godjevargova T. Development of Functional Fermented Meat Products Using Agro-Food Byproducts. Processes. 2026; 14(4):602. https://doi.org/10.3390/pr14040602
Chicago/Turabian StyleIvanov, Yavor, Milka Atanasova, and Tzonka Godjevargova. 2026. "Development of Functional Fermented Meat Products Using Agro-Food Byproducts" Processes 14, no. 4: 602. https://doi.org/10.3390/pr14040602
APA StyleIvanov, Y., Atanasova, M., & Godjevargova, T. (2026). Development of Functional Fermented Meat Products Using Agro-Food Byproducts. Processes, 14(4), 602. https://doi.org/10.3390/pr14040602








