Magnetic Field-Assisted Electro-Fenton System Using Magnetite as a Sustainable Iron Source for Wastewater Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Magnetite Synthesis
2.2. Physicochemical Characterization of Synthesized Magnetite
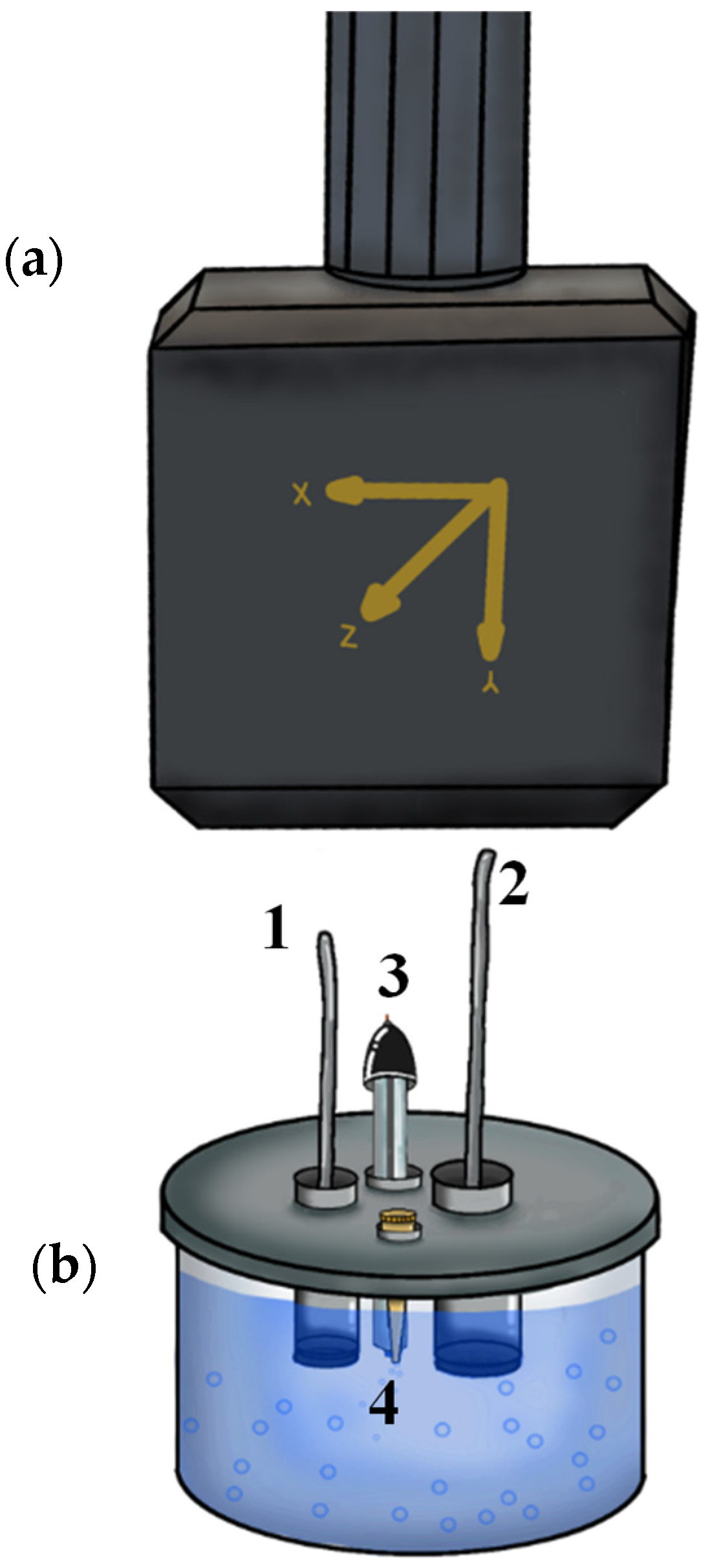
2.3. Performance of Synthesized Magnetite in an EF Process
3. Results
3.1. Physicochemical Characterization of Magnetite
3.2. Performance of Magnetite in an EF System
3.3. Study of Iron Ion Effects: Comparison Between Its Presence in Solution and Its Incorporation in Magnetite
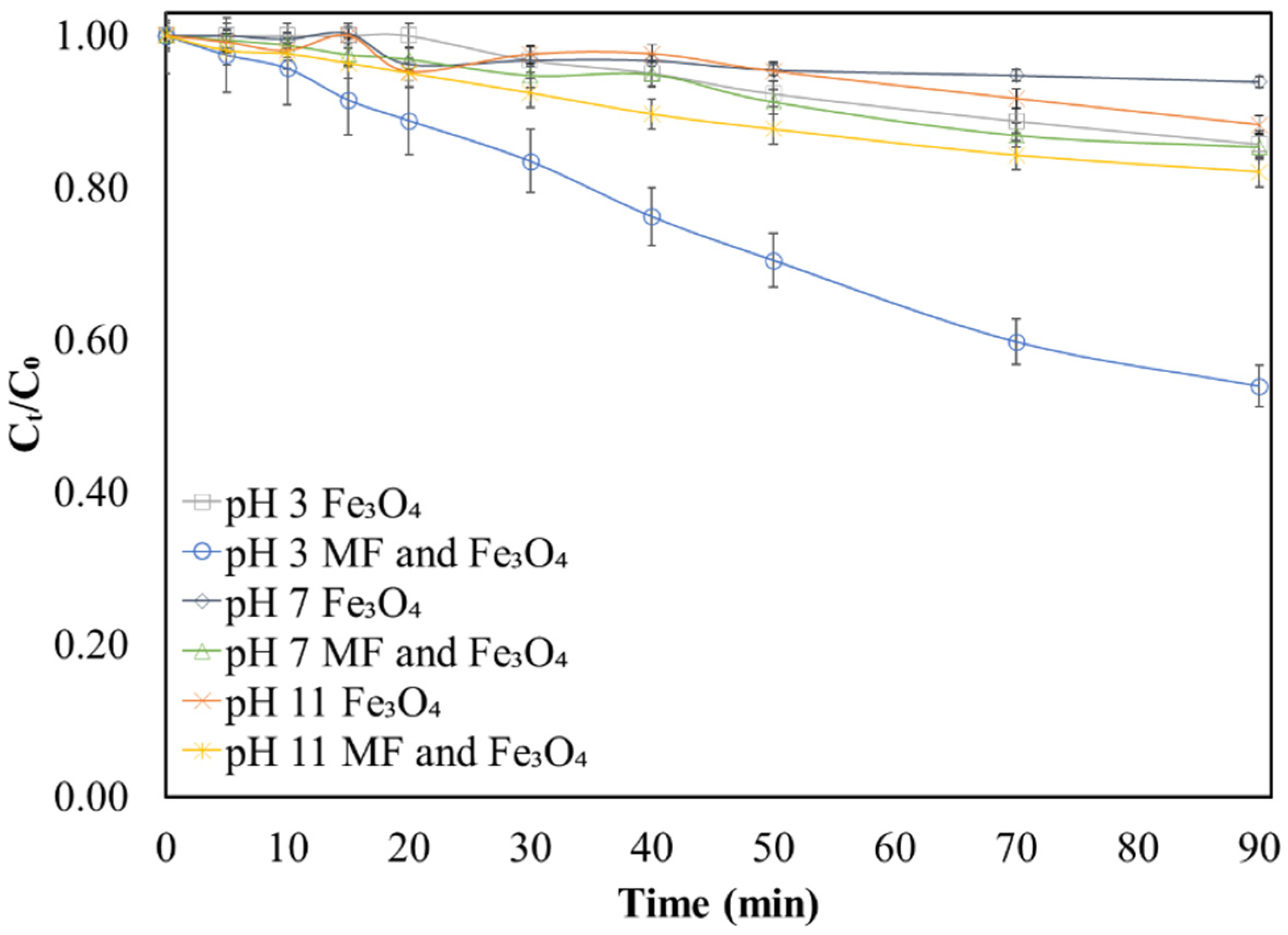
3.4. Influence of pH on Methylene Blue Decolorization Using FeSO4 and Fe3O4
3.5. Effect of Magnetic Stirring-Induced Magnetic Field on Decolorization
3.6. Effect of the Magnetite Ratio
3.7. Comparative Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CIQEC | Centro de Investigación en Química para la Economía Circular |
| EF | Electro-Fenton |
| Fe3O4 | Magnetite |
| FTIR | Fourier-transform infrared |
| SEM/EDS | Scanning Electron Microscopy and Energy-Dispersive X-ray Spectroscopy |
| XPS | X-ray Photoelectron Spectroscopy |
| EAOPs | Electrochemical advanced oxidation processes |
| •OH | Hydroxyl radicals |
| H2O2 | Hydrogen peroxide |
| ORR | Oxygen reduction reaction |
| MB | Methylene blue |
| FWHM | Full width at half maximum |
| B.E. | Binding energy |
| EO | Electrooxidation |
| σ | Standard deviation |
| S | Singlet |
| T | Triplet |
| •O2H | Hydroperoxyl radical |
| Fe | Iron |
| pHPZC | pH at the Point of Zero Charge |
References
- Choe, Y.J.; Kim, J.; Byun, J.Y.; Kim, S.H. An electro-Fenton system with magnetite coated stainless steel mesh as cathode. Catal. Today 2021, 359, 16–22. [Google Scholar] [CrossRef]
- Fernández, D.; Robles, I.; Rodríguez-Valadez, F.J.; Godínez, L.A. Novel arrangement for an electro-Fenton reactor that does not require addition of iron, acid and a final neutralization stage. Towards the development of a cost-effective technology for the treatment of wastewater. Chemosphere 2018, 199, 251–255. [Google Scholar] [CrossRef]
- Brillas, E.; Sirés, I.; Oturan, M.A. Electro-fenton process and related electrochemical technologies based on fenton’s reaction chemistry. Chem. Rev. 2009, 109, 6570–6631. [Google Scholar] [CrossRef]
- Nidheesh, P.V.; Gandhimathi, R.; Velmathi, S.; Sanjini, N.S. Magnetite as a heterogeneous electro Fenton catalyst for the removal of Rhodamine B from aqueous solution. RSC Adv. 2014, 4, 5698–5708. [Google Scholar] [CrossRef]
- Hao, X.L.; Zou, L.Y.; Zhang, G.S.; Zhang, Y.B. Magnetic field assisted Fenton reactions for the enhanced degradation of methyl blue. Chin. Chem. Lett. 2009, 20, 99–101. [Google Scholar] [CrossRef]
- Dan, H.; Kong, Y.; Yue, Q.; Liu, J.; Xu, X.; Kong, W.; Gao, Y.; Gao, B. Magnetic field-enhanced radical intensity for accelerating norfloxacin degradation under FeCu/rGO photo-Fenton catalysis. Chem. Eng. J. 2021, 420, 127634. [Google Scholar] [CrossRef]
- Pirsaheb, M.; Moradi, S.; Shahlaei, M.; Wang, X.; Farhadian, N. Simultaneously implement of both weak magnetic field and aeration for ciprofloxacin removal by Fenton-like reaction. J. Environ. Manag. 2019, 246, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Ozbey Unal, B.; Bilici, Z.; Ugur, N.; Isik, Z.; Harputlu, E.; Dizge, N.; Ocakoglu, K. Adsorption and Fenton oxidation of azo dyes by magnetite nanoparticles deposited on a glass substrate. J. Water Process Eng. 2019, 32, 100897. [Google Scholar] [CrossRef]
- Herrera, A.; Rodríguez, H.; Torres-Lugo, M.; Rinaldi, C. Functionalization of Magnetite (Fe3O4) Nanoparticles for Cancer Treatment. NSTI-Nanotech 2006, 2, 75–78. [Google Scholar]
- Cai, G.; Zhu, X.; Li, K.; Qi, X.; Wei, Y.; Wang, H.; Hao, F. Self-enhanced and efficient removal of arsenic from waste acid using magnetite as an in situ iron donator. Water Res. 2019, 157, 269–280. [Google Scholar] [CrossRef]
- Vallejo-Espinosa, G.; Pérez-Larios, A.; Nava-Andrade, K. Influence of temperature and stirring on the synthesis of magnetite nanoparticles. Mater. Lett. 2024, 372, 136980. [Google Scholar] [CrossRef]
- Mbuyazi, T.B.; Ajibade, P.A. Photocatalytic degradation of eosin yellow and quinaldine red under visible light irradiation by magnetite nanoparticles. J. Mol. Struct. 2025, 1334, 141862. [Google Scholar] [CrossRef]
- de Oliveira Guidolin, T.; Possolli, N.M.; Polla, M.B.; Wermuth, T.B.; de Oliveira, T.F.; Eller, S.; Montedo, O.R.K.; Arcaro, S.; Cechinel, M.A.P. Photocatalytic pathway on the degradation of methylene blue from aqueous solutions using magnetite nanoparticles. J. Clean. Prod. 2021, 318, 128556. [Google Scholar] [CrossRef]
- Shahid, M.K.; Kim, Y.; Choi, Y.G. Magnetite synthesis using iron oxide waste and its application for phosphate adsorption with column and batch reactors. Chem. Eng. Res. Des. 2019, 148, 169–179. [Google Scholar] [CrossRef]
- Kuila, S.K.; Chatterjee, R.; Ghosh, D. Reduction kinetics and characterization study of synthetic magnetite micro fines. In Minerals, Metals and Materials Series; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 615–631. [Google Scholar]
- Kim, K.-C.; Kim, E.-K.; Lee, J.-O.; Kim, Y.-S. Characterization of Magnetic Nanoparticles Synthesized by Sonomechanical Method. In Proceedings of the IEEE Nanotechnology Materials and Devices Conference, Gyeongju, Republic of Korea, 22–25 October 2006; pp. 600–601. [Google Scholar] [CrossRef]
- Schwaminger, S.P.; Bauer, D.; Fraga-García, P.; Wagnerb, F.E.; Berensmeier, S. Oxidation of magnetite nanoparticles: Impact on surface and crystal properties. CrystEngComm 2017, 19, 246–255. [Google Scholar] [CrossRef]
- More, S.; Raut, S.; Premkumar, S.; Bhopale, S.; Bhoraskar, S.; Morea, M.; Mathe, V. Structural and morphological tuning of iron oxide polymorphs by ECR plasma-assisted thermal oxidation. RSC Adv. 2020, 10, 32088–32101. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Guo, H.; Chai, C.; Li, N.; Lin, X.; Xu, W. Anodized graphite felt as an efficient cathode for in-situ hydrogen peroxide production and Electro-Fenton degradation of rhodamine B. Chemosphere 2022, 286, 131936. [Google Scholar] [CrossRef]
- Banda-Alemán, J.A.; Camacho-Callejas, M.; Salazar-López, M.L.; Robles, I.; Acosta-Santoyo, G.; García-Espinoza, J.D.; Rodríguez-Valadez, F.J.; Manriquez, J.; Godínez, L.A. Alternating polarization of Fe modified carbon fiber materials. towards the development of single-electrode electro-Fenton batch reactors. Electrochim. Acta 2023, 450, 142282. [Google Scholar] [CrossRef]
- Leyva-Ruiz, D.; Treviño-Reséndez, J.; Godínez, L.A.; Robles, I.; Acosta-Santoyo, G.; García-Espinoza, J.D. Electrochemical degradation of carbamazepine in water by a flow-through and pilot-scale reactor with carbon felt electrodes: Parametric study under realistic operational conditions. J. Environ. Chem. Eng. 2024, 12, 113232. [Google Scholar] [CrossRef]
- Rodríguez-Chueca, J.; Mediano, A.; Pueyo, N.; García-Suescun, I.; Mosteo, R.; Ormad, M.P. Degradation of chloroform by Fenton-like treatment induced by electromagnetic fields: A case of study. Chem. Eng. Sci. 2016, 156, 89–96. [Google Scholar] [CrossRef]
- Peña, M.R.; Díaz, C.E.B. Procesos de Oxidación Avanzada en el Tratamiento de Agua; Universidad Autónoma del Estado de México, Instituto Literario núm: Toluca, Estado de México, Mexico, 2020; ISBN 978-607-633-188-0. (PDF). [Google Scholar]
- Do, T.M.; Byun, J.Y.; Kim, S.H. An electro-Fenton system using magnetite coated metallic foams as cathode for dye degradation. Catal. Today 2017, 295, 48–55. [Google Scholar] [CrossRef]
- Kuntail, J.; Kumar, U.; Sinha, I. Insight into photo-Fenton reaction mechanism on a magnetite-GO nanocomposite: Computational and experimental investigations. Mol. Catal. 2022, 528, 112491. [Google Scholar] [CrossRef]
- Pujol, A.A.; León, I.; Cárdenas, J.; Sepúlveda-Guzmán, S.; Manríquez, J.; Sirés, I.; Bustos, E. Degradation of phenols by heterogeneous electro-Fenton with a Fe3O4-chitosan composite and a boron-doped diamond anode. Electrochim. Acta 2020, 337, 135784. [Google Scholar] [CrossRef]
- Teixeira, A.P.C.; Tristão, J.C.; Araujo, M.H.; Oliveira, L.C.A.; Moura, F.C.C.; Ardisson, J.D.; Amorim, C.C.; Lago, R.M. Iron: A Versatile Element to Produce Materials for Environmental Applications. J. Braz. Chem. Soc. 2012, 23, 1579–1593. [Google Scholar] [CrossRef]
- Xiong, X.; Sun, Y.; Sun, B.; Song, W.; Sun, J.; Gao, N.; Qiaoa, J.; Guan, X. Enhancement of the advanced Fenton process by weak magnetic field for the degradation of 4-nitrophenol. RSC Adv. 2015, 5, 13357–13365. [Google Scholar] [CrossRef]
- Wang, W.-M.; Li, X.; Du, X.; Wu, Q.-Y. A feasible approach for azo dye degradation using natural magnetite in heterogeneous Fenton oxidation. Water Cycle 2022, 3, 100–105. [Google Scholar] [CrossRef]
- Hernández-Rodríguez, E.A.; Castillo-Suárez, L.A.; Teutli-Sequeira, E.A.; Martínez-Miranda, V.; Vázquez-Mejía, G. Electro-oxidation and solar electro-oxidation of commercial carbamazepine: Effect of the support electrolyte. Sep. Sci. Technol. 2022, 57, 465–483. [Google Scholar] [CrossRef]
- Rodríguez-Chueca, J.; Mediano, A.; Ormad, M.P.; Mosteo, R.; Ovelleiro, J.L. Disinfection of wastewater effluents with the Fenton-like process induced by electromagnetic fields. Water Res. 2014, 60, 250–258. [Google Scholar] [CrossRef]
- Shi, Z.; Zhang, R.; Zhang, J. Role of weak magnetic field for enhanced oxidation of orange G by magnetic Fenton. Environ. Sci. Pollut. Res. 2021, 28, 59834–59843. [Google Scholar] [CrossRef]
- Novikov, V.V.; Yablokova, E.V.; Fesenko, E.E. The effect of weak magnetic fields on the production of reactive oxygen species in neutrophils. Biophysics 2016, 61, 959–962. [Google Scholar] [CrossRef]
- Giwa, A.R.A.; Bello, I.A.; Olabintan, A.B.; Belloa, O.S.; Saleh, T.A. Kinetic and thermodynamic studies of fenton oxidative decolorization of methylene blue. Heliyon 2020, 6, e04454. [Google Scholar] [CrossRef] [PubMed]
- Garzón-Cucaita, V.; Carriazo, J.G. Óxidos de hierro como catalizadores de procesos tipo Fenton con potencial aplicación en tecnologías de remoción de contaminantes. TecnoLógicas 2022, 25, e2393. [Google Scholar] [CrossRef]
- Muzenda, C.; Arotiba, O.A. Improved Magnetite Nanoparticle Immobilization on a Carbon Felt Cathode in the Heterogeneous Electro-Fenton Degradation of Aspirin in Wastewater. ACS Omega 2022, 7, 19261–19269. [Google Scholar] [CrossRef] [PubMed]






| Element | B.E (eV) | FWHM (eV) | Atomic Composition (%) |
|---|---|---|---|
| Cl2p | 198.1 | 0.93 | 0.33 |
| O1s | 529.8 | 1.31 | 61.11 |
| Fe2p | 710.5 | 4.00 | 38.51 |
| Na1s | 1073.5 | 0.14 | 0.05 |
| Fe Source | pH | Fetot Initial (mg/L) | Fetot Final (mg/L) | Decolorization (%) | σ |
|---|---|---|---|---|---|
| a Fe3O4 | 3 | 0.90 | 0.93 | 14.22 | 0.038 |
| 7 | 0.82 | 0.77 | 6.07 | 0.019 | |
| 11 | 0.85 | 0.81 | 11.69 | 0.036 | |
| b FeSO4 | 3 | 2.09 | 2.09 | 46.18 | 0.107 |
| 7 | 0.91 | 1.13 | 16.48 | 0.033 | |
| 11 | 0.90 | 0.93 | 9.57 | 0.028 |
| Catalyst/Setup | Process | Magnetic Field (G) | Pollutant (mg/L) | Efficiency (%)/Reaction Time | Ref. |
|---|---|---|---|---|---|
| Fe0 nanoparticles | Photo-Fenton | 300 | Ciprofloxacin (20) | 90%/60 min | [7] |
| Fe−Cu/rGO | Photo-Fenton | 250 | Norfloxacin (20) | 88%/60 min | [6] |
| Fe3O4 on Steel mesh | Electro-Fenton | - | Methylene Blue (10) | 100%/80–150 min | [1] |
| Fe3O4/Carbon Felt | Electro-Fenton | - | Aspirin (20) | 100%/140 min | [36] |
| Synthesized Fe3O4 | Electro-Fenton | 150 | Methylene Blue (20) | 49.2%/90 min | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hernández-Rodríguez, E.A.; García-Espinoza, J.D.; Treviño-Resendez, J.; Razo-Negrete, M.; Acosta-Santoyo, G.; Godínez, L.A.; Robles, I. Magnetic Field-Assisted Electro-Fenton System Using Magnetite as a Sustainable Iron Source for Wastewater Treatment. Processes 2026, 14, 264. https://doi.org/10.3390/pr14020264
Hernández-Rodríguez EA, García-Espinoza JD, Treviño-Resendez J, Razo-Negrete M, Acosta-Santoyo G, Godínez LA, Robles I. Magnetic Field-Assisted Electro-Fenton System Using Magnetite as a Sustainable Iron Source for Wastewater Treatment. Processes. 2026; 14(2):264. https://doi.org/10.3390/pr14020264
Chicago/Turabian StyleHernández-Rodríguez, Evelyn A., Josué D. García-Espinoza, José Treviño-Resendez, Mónica Razo-Negrete, Gustavo Acosta-Santoyo, Luis A. Godínez, and Irma Robles. 2026. "Magnetic Field-Assisted Electro-Fenton System Using Magnetite as a Sustainable Iron Source for Wastewater Treatment" Processes 14, no. 2: 264. https://doi.org/10.3390/pr14020264
APA StyleHernández-Rodríguez, E. A., García-Espinoza, J. D., Treviño-Resendez, J., Razo-Negrete, M., Acosta-Santoyo, G., Godínez, L. A., & Robles, I. (2026). Magnetic Field-Assisted Electro-Fenton System Using Magnetite as a Sustainable Iron Source for Wastewater Treatment. Processes, 14(2), 264. https://doi.org/10.3390/pr14020264









