Quality of Life and Disease Impact of Atopic Dermatitis and Psoriasis on Children and Their Families
Abstract
1. Introduction
2. Measurement of QoL
3. Disease Impact of AD
3.1. AD Impact Relative to Other Diseases
3.2. Psychosocial/Mental Comorbidity
3.3. Impact of Comorbidities on QoL
4. Disease Impact of Psoriasis
4.1. Psoriasis Impact Relative to Other Diseases
4.2. Psychosocial/Mental Comorbidity
4.3. Impact of Comorbidities on QoL
5. Strategies to Improve QoL
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Eichenfield, L.F.; Tom, W.L.; Chamlin, S.L.; Feldman, S.R.; Hanifin, J.M.; Simpson, E.L.; Berger, T.G.; Bergman, J.N.; Cohen, D.E.; Cooper, K.D.; et al. Guidelines of care for the management of atopic dermatitis: Section 1. Diagnosis and assessment of atopic dermatitis. J. Am. Acad. Dermatol. 2014, 70, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.; Stewart, A.; von Mutius, E.; Cookson, W.; Anderson, H.R.; International Study of Asthma. Is eczema really on the increase worldwide? J. Allergy Clin. Immunol. 2008, 121, 947–954.e915. [Google Scholar] [CrossRef] [PubMed]
- Shaw, T.E.; Currie, G.P.; Koudelka, C.W.; Simpson, E.L. Eczema prevalence in the United States: Data from the 2003 National Survey of Children’s Health. J. Investig. Dermatol. 2011, 131, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Simpson, E.L. Associations of childhood eczema severity: A US population based study. Dermat. Contact Atopic Occup. Drug 2014, 25, 107. [Google Scholar] [CrossRef] [PubMed]
- Kuo, I.-H.; Yoshida, T.; De Benedetto, A.; Beck, L.A. The cutaneous innate immune response in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2013, 131, 266–278. [Google Scholar] [CrossRef] [PubMed]
- Koblenzer, C.S. Itching and the atopic skin. J. Allergy Clin. Immunol. 1999, 104, S109–S113. [Google Scholar] [CrossRef]
- Tollefson, M.M.; Crowson, C.S.; McEvoy, M.T.; Kremers, H.M. Incidence of psoriasis in children: A population-based study. J. Am. Acad. Dermatol. 2010, 62, 979–987. [Google Scholar] [CrossRef]
- Michalek, I.; Loring, B.; John, S. A systematic review of worldwide epidemiology of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 205–212. [Google Scholar] [CrossRef]
- Farber, E.M.; Nall, L. The natural history of psoriasis in 5600 patients. Dermatology 1974, 148, 1–18. [Google Scholar] [CrossRef]
- Guttman-Yassky, E.; Nograles, K.E.; Krueger, J.G. Contrasting pathogenesis of atopic dermatitis and psoriasis—Part II: Immune cell subsets and therapeutic concepts. J. Allergy Clin. Immunol. 2011, 127, 1420–1432. [Google Scholar] [CrossRef]
- Prignano, F.; Ricceri, F.; Pescitelli, L.; Lotti, T. Itch in psoriasis: Epidemiology, clinical aspects and treatment options. Clin. Cosmet. Investig. Dermatol. CCID 2009, 2, 9. [Google Scholar] [CrossRef] [PubMed]
- Szepietowski, J.; Reich, A. Pruritus in psoriasis: An update. Eur. J. Pain 2016, 20, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Lawson, V.; Lewis-Jones, M.S.; Finlay, A.Y.; Reid, P.; Owens, R.G. The family impact of childhood atopic dermatitis: The Dermatitis Family Impact Questionnaire. Br. J. Dermatol. 1998, 138, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Beattie, P.; Lewis-Jones, M. A comparative study of impairment of quality of life in children with skin disease and children with other chronic childhood diseases. Br. J. Dermatol. 2006, 155, 145–151. [Google Scholar] [CrossRef]
- Tollefson, M.M.; Finnie, D.M.; Schoch, J.J.; Eton, D.T. Impact of childhood psoriasis on parents of affected children. J. Am. Acad. Dermatol. 2017, 76, 286–289.e285. [Google Scholar] [CrossRef]
- Lifschitz, C. The impact of atopic dermatitis on quality of life. Ann. Nutr. Metab. 2015, 66, 34–40. [Google Scholar] [CrossRef]
- Do Amaral, C.S.F.; March, M.d.F.B.P.; Sant‘Anna, C.C. Instruments that measure the quality of life in atopic dermatitis among children and adolescents. Clin. Investig. 2013, 3, 935–942. [Google Scholar] [CrossRef]
- Augustin, M.; Langenbruch, A.K.; Gutknecht, M.; Radtke, M.A.; Blome, C. Quality of life measures for dermatology: Definition, evaluation, and interpretation. Curr. Dermatol. Rep. 2012, 1, 148–159. [Google Scholar] [CrossRef]
- Holme, S.; Man, I.; Sharpe, J.; Dykes, P.; Lewis-Jones, M.; Finlay, A. The Children’s Dermatology Life Quality Index: Validation of the cartoon version. Br. J. Dermatol. 2003, 148, 285–290. [Google Scholar] [CrossRef]
- Neri, E.; Agostini, F.; Gremigni, P.; Gobbi, F.; Casu, G.; Chamlin, S.L.; Monti, F. Italian validation of the Childhood Atopic Dermatitis Impact Scale: A contribution to its clinical application. J. Investig. Dermatol. 2012, 132, 2534–2543. [Google Scholar] [CrossRef]
- McKenna, S.P.; Doward, L.C. Quality of life of children with atopic dermatitis and their families. Curr. Opin. Allergy Clin. Immunol. 2008, 8, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Halioua, B.; Beumont, M.G.; Lunel, F. Quality of life in dermatology. Int. J. Dermatol. 2000, 39, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Chernyshov, P.; Tomas-Aragones, L.; Manolache, L.; Marron, S.; Salek, M.; Poot, F.; Oranje, A.; Finlay, A.Y.; EADV Quality of Life Task Force. Quality of life measurement in atopic dermatitis. Position paper of the European Academy of Dermatology and Venereology (EADV) Task Force on quality of life. J. Eur. Acad. Derm. Venereol. 2017, 31, 576–593. [Google Scholar] [CrossRef] [PubMed]
- Lewis-Jones, M.; Finlay, A. The Children’s Dermatology Life Quality Index (CDLQI): Initial validation and practical use. Br. J. Dermatol. 1995, 132, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Lewis-Jones, M.; Finlay, A.; Dykes, P. The infants’ dermatitis quality of life index. Br. J. Dermatol. 2001, 144, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Heinl, D.; Prinsen, C.; Sach, T.; Drucker, A.; Ofenloch, R.; Flohr, C.; Apfelbacher, C. Measurement properties of quality-of-life measurement instruments for infants, children and adolescents with eczema: A systematic review. Br. J. Dermatol. 2017, 176, 878–889. [Google Scholar] [CrossRef] [PubMed]
- Rehal, B.; Armstrong, A. Health outcome measures in atopic dermatitis: A systematic review of trends in disease severity and quality-of-life instruments 1985–2010. PLoS ONE 2011, 6, e17520. [Google Scholar] [CrossRef]
- Schmitt, J.; Apfelbacher, C.; Spuls, P.I.; Thomas, K.S.; Simpson, E.L.; Furue, M.; Chalmers, J.; Williams, H.C. The Harmonizing Outcome Measures for Eczema (HOME) roadmap: A methodological framework to develop core sets of outcome measurements in dermatology. J. Investig. Dermatology 2015, 135, 24–30. [Google Scholar] [CrossRef]
- HOME VII Meeting. HOME—Harmonising Outcome Measures for Eczema. Available online: www.homeforeczema.org (accessed on 10 April 2019).
- Finlay, A.Y.; Khan, G. Dermatology Life Quality Index (DLQI)—A simple practical measure for routine clinical use. Clin. Exp. Dermatol. 1994, 19, 210–216. [Google Scholar] [CrossRef]
- Basra, M.; Sue-Ho, R.; Finlay, A. The Family Dermatology Life Quality Index: Measuring the secondary impact of skin disease. Br. J. Dermatol. 2007, 156, 528–538. [Google Scholar] [CrossRef]
- Smidt, A.C.; Lai, J.-S.; Cella, D.; Patel, S.; Mancini, A.J.; Chamlin, S.L. Development and validation of Skindex-Teen, a quality-of-life instrument for adolescents with skin disease. Arch. Dermatol. 2010, 146, 865–869. [Google Scholar] [CrossRef] [PubMed]
- Drake, L.; Prendergast, M.; Maher, R.; Breneman, D.; Korman, N.; Satoib, Y.; Beusterien, K.M.; Lawrence, I. The impact of tacrolimus ointment on health-related quality of life of adult and pediatric patients with atopic dermatitis. J. Am. Acad. Dermatol. 2001, 44, S65–S72. [Google Scholar] [CrossRef] [PubMed]
- Chamlin, S.L.; Lai, J.-S.; Cella, D.; Frieden, I.J.; Williams, M.L.; Mancini, A.J.; Chren, M.-M. Childhood Atopic Dermatitis Impact Scale: Reliability, discriminative and concurrent validity, and responsiveness. Arch. Dermatol. 2007, 143, 768–772. [Google Scholar] [CrossRef] [PubMed]
- McKenna, S.P.; Doward, L.C.; Meads, D.M.; Tennant, A.; Lawton, G.; Grueger, J. Quality of life in infants and children with atopic dermatitis: Addressing issues of differential item functioning across countries in multinational clinical trials. Health Qual. Life Outcomes 2007, 5, 45. [Google Scholar] [CrossRef]
- Baars, R.M.; Atherton, C.I.; Koopman, H.M.; Bullinger, M.; Power, M. The European DISABKIDS project: Development of seven condition-specific modules to measure health related quality of life in children and adolescents. Health Qual. Life Outcomes 2005, 3, 70. [Google Scholar] [CrossRef]
- McKenna, S.P.; Whalley, D.; Dewar, A.L.; Erdman, R.A.; Kohlmann, T.; Niero, M.; Baró, E.; Cook, S.A.; Crickx, B.; Frech, F. International development of the parents’ index of quality of life in atopic dermatitis (PIQoL-AD). Qual. Life Res. 2005, 14, 231–241. [Google Scholar] [CrossRef]
- Kondo-Endo, K.; Ohashi, Y.; Nakagawa, H.; Katsunuma, T.; Ohya, Y.; Kamibeppu, K.; Masuko, I. Development and validation of a questionnaire measuring quality of life in primary caregivers of children with atopic dermatitis (QPCAD). Br. J. Dermatol. 2009, 161, 617–625. [Google Scholar] [CrossRef]
- Von Rüden, U.; Staab, D.; Kehrt, R.; Wahn, U. Entwicklung und Validierung eines krankheitsspezifischen Fragebogens zur Erfassung der Lebensqualität von Eltern neurodermitiskranker Kinder. Z. Gesundh. J. Public Health 1999, 7, 335. [Google Scholar] [CrossRef]
- Oostveen, A.M.; De Jong, E.M.; Evers, A.W.; Donders, A.; De Van Kerkhof, P.; Seyger, M. Reliability, responsiveness and validity of Scalpdex in children with scalp psoriasis: The Dutch study. Acta Derm. Venereol. 2014, 94, 198–202. [Google Scholar] [CrossRef]
- Su, J.C.; Kemp, A.S.; Varigos, G.A.; Nolan, T.M. Atopic eczema: Its impact on the family and financial cost. Arch. Dis. Child. 1997, 76, 159–162. [Google Scholar] [CrossRef]
- Elliott, B.E.; Luker, K. The experiences of mothers caring for a child with severe atopic eczema. J. Clin. Nurs. 1997, 6, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Camfferman, D.; Kennedy, J.D.; Gold, M.; Martin, A.J.; Lushington, K. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med. Rev. 2010, 14, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Stores, G.; Burrows, A.; Crawford, C. Physiological sleep disturbance in children with atopic dermatitis: A case control study. Pediatric Dermatol. 1998, 15, 264–268. [Google Scholar] [CrossRef] [PubMed]
- Chamlin, S.L.; Mattson, C.L.; Frieden, I.J.; Williams, M.L.; Mancini, A.J.; Cella, D.; Chren, M.-M. The price of pruritus: Sleep disturbance and cosleeping in atopic dermatitis. Arch. Pediatrics Adolesc. Med. 2005, 159, 745–750. [Google Scholar] [CrossRef]
- Daud, L.; Garralda, M.; David, T. Psychosocial adjustment in preschool children with atopic eczema. Arch. Dis. Child. 1993, 69, 670–676. [Google Scholar] [CrossRef]
- Mitchell, A.E.; Fraser, J.A.; Morawska, A.; Ramsbotham, J.; Yates, P. Parenting and childhood atopic dermatitis: A cross-sectional study of relationships between parenting behaviour, skin care management, and disease severity in young children. Int. J. Nurs. Stud. 2016, 64, 72–85. [Google Scholar] [CrossRef]
- Absolon, C.; Cottrell, D.; Eldridge, S.; Glover, M. Psychological disturbance in atopic eczema: The extent of the problem in school-aged children. Br. J. Dermatol. 1997, 137, 241–245. [Google Scholar] [CrossRef]
- Bullinger, M.; Schmidt, S.; Petersen, C.; DISABKIDS group. Assessing quality of life of children with chronic health conditions and disabilities: A European approach. Int. J. Rehabil. Res. 2002, 25, 197–206. [Google Scholar] [CrossRef]
- Holm, E.A.; Wulf, H.C.; Stegmann, H.; Jemec, G.B. Life quality assessment among patients with atopic eczema. Br. J. Dermatol. 2006, 154, 719–725. [Google Scholar] [CrossRef]
- Mozaffari, H.; Pourpak, Z.; Pourseyed, S.; Farhoodi, A.; Aghamohammadi, A.; Movahadi, M.; Gharaghozloo, M.; Moin, M. Quality of life in atopic dermatitis patients. J. Microbiol. Immunol. Infect. Wei Mian Yu Gan Ran Za Zhi 2007, 40, 260–264. [Google Scholar]
- Chamlin, S.L.; Frieden, I.J.; Williams, M.L.; Chren, M.-M. Effects of atopic dermatitis on young American children and their families. Pediatrics 2004, 114, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Lapidus, C.; Kerr, P. Social impact of atopic dermatitis. Med. Health 2001, 84, 294–295. [Google Scholar]
- Slattery, M.J.; Essex, M.J.; Paletz, E.M.; Vanness, E.R.; Infante, M.; Rogers, G.M.; Gern, J.E. Depression, anxiety, and dermatologic quality of life in adolescents with atopic dermatitis. J. Allergy Clin. Immunol. 2011, 128, 668. [Google Scholar] [CrossRef] [PubMed]
- Yaghmaie, P.; Koudelka, C.W.; Simpson, E.L. Mental health comorbidity in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2013, 131, 428–433. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.; Apfelbacher, C.; Chen, C.-M.; Romanos, M.; Sausenthaler, S.; Koletzko, S.; Bauer, C.-P.; Hoffmann, U.; Krämer, U.; Berdel, D. Infant-onset eczema in relation to mental health problems at age 10 years: Results from a prospective birth cohort study (German Infant Nutrition Intervention plus). J. Allergy Clin. Immunol. 2010, 125, 404–410. [Google Scholar] [CrossRef]
- Lee, S.; Shin, A. Association of atopic dermatitis with depressive symptoms and suicidal behaviors among adolescents in Korea: The 2013 Korean Youth Risk Behavior Survey. BMC Psychiatry 2017, 17, 3. [Google Scholar] [CrossRef] [PubMed]
- Noh, H.-M.; Cho, J.J.; Park, Y.S.; Kim, J.-H. The relationship between suicidal behaviors and atopic dermatitis in Korean adolescents. J. Health Psychol. 2016, 21, 2183–2194. [Google Scholar] [CrossRef]
- Patel, K.R.; Immaneni, S.; Singam, V.; Rastogi, S.; Silverberg, J.I. Association between atopic dermatitis, depression, and suicidal ideation: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2019, 80, 402–410. [Google Scholar] [CrossRef]
- Van Oers, H.A.; Haverman, L.; Limperg, P.F.; van Dijk-Lokkart, E.; Maurice-Stam, H.; Grootenhuis, M. Anxiety and depression in mothers and fathers of a chronically ill child. Matern. Child Health J. 2014, 18, 1993–2002. [Google Scholar] [CrossRef]
- Ho, R.C.; Giam, Y.; Ng, T.; Mak, A.; Goh, D.; Zhang, M.W.; Cheak, A.; Van Bever, H.P. The influence of childhood atopic dermatitis on health of mothers, and its impact on Asian families. Pediatric Allergy Immunol. 2010, 21, 501–507. [Google Scholar] [CrossRef]
- Charman, C.; Morris, A.; Williams, H. Topical corticosteroid phobia in patients with atopic eczema. Br. J. Dermatol. 2000, 142, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Silverberg, J.I.; Gelfand, J.M.; Margolis, D.J.; Boguniewicz, M.; Fonacier, L.; Grayson, M.H.; Simpson, E.L.; Ong, P.Y.; Fuxench, Z.C.C. Patient burden and quality of life in atopic dermatitis in US adults: A population-based cross-sectional study. Ann. Allergy Asthma Immunol. 2018, 121, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.L. Comorbidity in atopic dermatitis. Curr. Dermatol. Rep. 2012, 1, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Hammer-Helmich, L.; Linneberg, A.; Obel, C.; Thomsen, S.F.; Møllehave, L.T.; Glümer, C. Mental health associations with eczema, asthma and hay fever in children: A cross-sectional survey. BMJ Open 2016, 6, e012637. [Google Scholar] [CrossRef] [PubMed]
- Palit, A.; Handa, S.; Bhalla, A.K.; Kumar, B. A mixed longitudinal study of physical growth in children with atopic dermatitis. Indian J. Dermatol. Venereol. Leprol. 2007, 73, 171. [Google Scholar] [PubMed]
- Agostoni, C.; Grandi, F.; Scaglioni, S.; Giannì, M.L.; Torcoletti, M.; Radaelli, G.; Fiocchi, A.; Riva, E. Growth pattern of breastfed and nonbreastfed infants with atopic dermatitis in the first year of life. Pediatrics 2000, 106, e73. [Google Scholar] [CrossRef] [PubMed]
- Ellison, J.; Patel, L.; Kecojevic, T.; Foster, P.; David, T.; Clayton, P. Pattern of growth and adiposity from infancy to adulthood in atopic dermatitis. Br. J. Dermatol. 2006, 155, 532–538. [Google Scholar] [CrossRef]
- Mitchell, A.E. Bidirectional relationships between psychological health and dermatological conditions in children. Psychol. Res. Behav. Manag. 2018, 11, 289. [Google Scholar] [CrossRef]
- Manzoni, A.P.D.d.S.; Pereira, R.L.; Townsend, R.Z.; Weber, M.B.; Nagatomi, A.R.d.S.; Cestari, T.F. Assessment of the quality of life of pediatric patients with the major chronic childhood skin diseases. An. Bras. Dermatol. 2012, 87, 361–368. [Google Scholar] [CrossRef]
- Varni, J.W.; Globe, D.R.; Gandra, S.R.; Harrison, D.J.; Hooper, M.; Baumgartner, S. Health-related quality of life of pediatric patients with moderate to severe plaque psoriasis: Comparisons to four common chronic diseases. Eur. J. Pediatrics 2012, 171, 485–492. [Google Scholar] [CrossRef]
- Randa, H.; Lomholt, J.; Skov, L.; Zachariae, R. Health-related quality of life in adolescents with psoriasis: An interview-based study. Br. J. Dermatol. 2018, 178, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- De Jager, M.E.; De Jong, E.M.; Evers, A.W.; Van De Kerkhof, P.C.; Seyger, M.M. The burden of childhood psoriasis. Pediatric Dermatol. 2011, 28, 736–737. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, S.P.; Gross, J. A comparative study of pediatric onset psoriasis with adult onset psoriasis. Pediatric Dermatol. 2000, 17, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.A.; Simpson, F.C.; Gupta, A.K. Psoriasis and sleep disorders: A systematic review. Sleep Med. Rev. 2016, 29, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Seyhan, M.; Coşkun, B.K.; Sağlam, H.; Özcan, H.; Karincaoğlu, Y. Psoriasis in childhood and adolescence: Evaluation of demographic and clinical features. Pediatrics Int. 2006, 48, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Kimball, A.B.; Wu, E.Q.; Guérin, A.; Andrew, P.Y.; Tsaneva, M.; Gupta, S.R.; Bao, Y.; Mulani, P.M. Risks of developing psychiatric disorders in pediatric patients with psoriasis. J. Am. Acad. Dermatol. 2012, 67, 651–657.e652. [Google Scholar] [CrossRef] [PubMed]
- Bilgic, A.; Bilgic, Ö.; Akış, H.K.; Eskioğlu, F.; Kılıç, E.Z. Psychiatric symptoms and health-related quality of life in children and adolescents with psoriasis. Pediatric Dermatol. 2010, 27, 614–617. [Google Scholar] [CrossRef]
- Golics, C.; Basra, M.; Finlay, A.; Salek, M. Adolescents with skin disease have specific quality of life issues. Dermatology 2009, 218, 357–366. [Google Scholar] [CrossRef]
- Remröd, C.; Sjöström, K.; Svensson, Å. Psychological differences between early-and late-onset psoriasis: A study of personality traits, anxiety and depression in psoriasis. Br. J. Dermatol. 2013, 169, 344–350. [Google Scholar] [CrossRef]
- Basavaraj, K.H.; Navya, M.A.; Rashmi, R. Stress and quality of life in psoriasis: An update. Int. J. Dermatol. 2011, 50, 783–792. [Google Scholar] [CrossRef]
- Nyfors, A.; Lemholt, K. Psoriasis in children: A short review and a survey of 245 cases. Br. J. Dermatol. 1975, 92, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Gånemo, A.; Wahlgren, C.F.; Svensson, Å. Quality of life and clinical features in Swedish children with psoriasis. Pediatric Dermatol. 2011, 28, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Randa, H.; Todberg, T.; Skov, L.; Larsen, L.S.; Zachariae, R. Health-related quality of life in children and adolescents with psoriasis: A systematic review and meta-analysis. Acta Derm. Venereol. 2017, 97, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.E.; Cohen, J.M.; Ho, R.S. Psoriasis and suicidality: A review of the literature. Dermatol. Ther. 2019, 32, e12771. [Google Scholar] [CrossRef] [PubMed]
- Tadros, A.; Vergou, T.; Stratigos, A.; Tzavara, C.; Hletsos, M.; Katsambas, A.; Antoniou, C. Psoriasis: Is it the tip of the iceberg for the quality of life of patients and their families? J. Eur. Acad. Derm. Venereol. 2011, 25, 1282–1287. [Google Scholar] [CrossRef] [PubMed]
- Augustin, M.; Glaeske, G.; Radtke, M.; Christophers, E.; Reich, K.; Schäfer, I. Epidemiology and comorbidity of psoriasis in children. Br. J. Dermatol. 2010, 162, 633–636. [Google Scholar] [CrossRef]
- Matusiewicz, D.; Koerber, A.; Schadendorf, D.; Wasem, J.; Neumann, A. Childhood psoriasis—An analysis of German health insurance data. Pediatric Dermatol. 2014, 31, 8–13. [Google Scholar] [CrossRef]
- Mercy, K.M.; Paller, A.S. The relationship between obesity and psoriasis in the pediatric population: Implications and future directions. Cutis 2013, 92, 107. [Google Scholar]
- Paller, A.S.; Mercy, K.; Kwasny, M.J.; Choon, S.E.; Cordoro, K.M.; Girolomoni, G.; Menter, A.; Tom, W.L.; Mahoney, A.M.; Oostveen, A.M. Association of pediatric psoriasis severity with excess and central adiposity: An international cross-sectional study. JAMA Dermatol. 2013, 149, 166–176. [Google Scholar] [CrossRef]
- Swerlick, R.A.; Zhang, C.; Patel, A.; Chren, M.M.; Chen, S. The Skindex-mini: A streamlined QOL measurement tool suitable for routine use in clinic. J. Am. Acad. Dermatol. 2018, in press. [Google Scholar] [CrossRef]
- Oostveen, A.M.; Spillekom-van Koulil, S.; Otero, M.E.; Klompmaker, W.; Evers, A.W.; Seyger, M.M. Development and design of a multidisciplinary training program for outpatient children and adolescents with psoriasis and their parents. J. Dermatol. Treat. 2013, 24, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.; Cunningham, K.; Perlmutter, J.; Gottlieb, A. Systematic review of health-related quality of life in adolescents with psoriasis. Dermatology 2016, 232, 541–549. [Google Scholar] [CrossRef] [PubMed]
- LeBovidge, J.S.; Elverson, W.; Timmons, K.G.; Hawryluk, E.B.; Rea, C.; Lee, M.; Schneider, L.C. Multidisciplinary interventions in the management of atopic dermatitis. J. Allergy Clin. Immunol. 2016, 138, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Ricci, G.; Bendandi, B.; Aiazzi, R.; Patrizi, A.; Masi, M. Three years of Italian experience of an educational program for parents of young children affected by atopic dermatitis: Improving knowledge produces lower anxiety levels in parents of children with atopic dermatitis. Pediatric Dermatol. 2009, 26, 1–5. [Google Scholar] [CrossRef]
- Barbarot, S.; Stalder, J. Therapeutic patient education in atopic eczema. Br. J. Dermatol. 2014, 170, 44–48. [Google Scholar] [CrossRef]
- Premerlani, M.; Ludewig, Y.; Schnopp, C.; Ring, J. Eczema school: Practical approaches in an efficient module of tertiary prevention programs. In Handbook of Atopic Eczema; Springer: Berlin/Heidelberg, Germany, 2006; pp. 576–581. [Google Scholar]
- Williams, H.C. Educational programmes for young people with eczema. Br. Med J. Publ. Group 2006, 332, 923–924. [Google Scholar] [CrossRef]
- Wollenberg, A.; Barbarot, S.; Bieber, T.; Christen-Zaech, S.; Deleuran, M.; Fink-Wagner, A.; Gieler, U.; Girolomoni, G.; Lau, S.; Muraro, A. Consensus-based European guidelines for treatment of atopic eczema (atopic dermatitis) in adults and children: Part II. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 850–878. [Google Scholar] [CrossRef]
- Takaoka, R.; Aoki, V.; Campos, T. Support groups for patients with atopic dermatitis: A nine-year experience. Dermatol. Psychosom. 2000, 1, 33–38. [Google Scholar]
- Fox, F.E.; Rumsey, N.; Morris, M. “Ur skin is the thing that everyone sees and you cant change it!”: Exploring the appearance-related concerns of young people with psoriasis. Dev. Neurorehabilit. 2007, 10, 133–141. [Google Scholar] [CrossRef]
| AD | Psoriasis | |
|---|---|---|
| Lesion configuration | Erythematous scaly, crusted papules, lichenification, excoriation | Sharply demarcated, erythematous scaly plaques |
| Scale | Fine | Silvery-white, thick |
| Oozing/crust | Common | Rare |
| Distribution | Flexor surfaces (neck, antecubital and popliteal fossae), face, chest, wrist, back of hands and feet | Extensor surfaces of elbows and knees, scalp, sacral region, chest, face, abdomen |
| Symptoms | Pruritus | Stinging, burning |
| Photographic example | 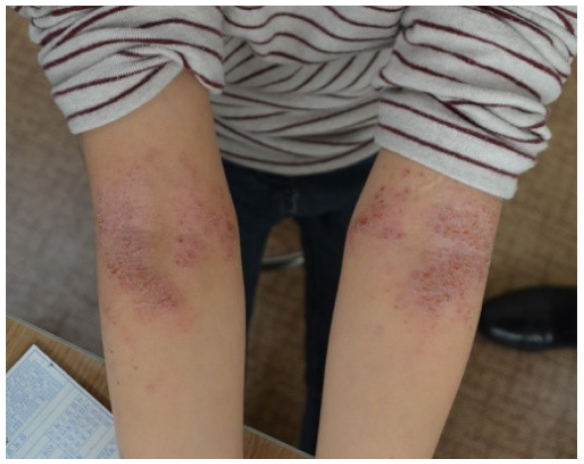 | 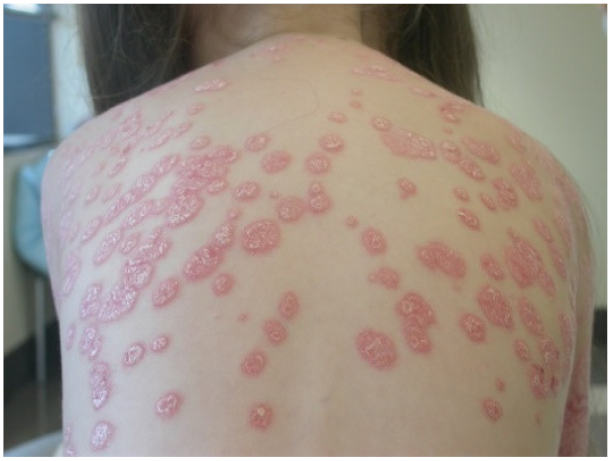 |
| Dermatology-Specific Tools | Disease-Specific Tools | |
|---|---|---|
| AD | Psoriasis | |
| - Dermatology Life Quality Index (DLQI) [30] - Children’s Dermatology Life Quality Index (CDLQI) [24] - Infants’ Dermatitis Quality of Life Index (IDQoL) [25] - Family Dermatology Life Quality Index (FDLQI) [31] - Skindex-Teen [32] - Toddler Quality of Life Survey [33] | - Dermatitis Family Index (DFI) [13] - Childhood Atopic Dermatitis Impact Scale (CADIS) [34] - Childhood Impact of Atopic Dermatitis (CIAD) [35] - DISABKIDS Atopic Dermatitis Module (DISABKIDS-ADM) [36] - Parents’ Index of Quality of Life in Atopic Dermatitis (PIQoL-AD) [37] - Quality of Life in Primary Caregivers of children with Atopic Dermatitis (QPCAD) [38] - The Quality of Life in Parents of Children with Atopic Dermatitis [39] | - Children’s Scalpdex in Psoriasis [40] |
| AD | Psoriasis | |
|---|---|---|
| Overall QoL impairment | - Greatest negative impact on HRQoL among chronic skin disorders including urticaria, alopecia, acne, and localized eczema [14] | - Greater negative impact than those seen in children with epilepsy, enuresis, diabetes, and vitiligo [14,70] - Comparable impairment to those seen in children with other chronic diseases such as arthritis or asthma [15,71] |
| Itch and sleep disturbance | - Frequent scratching due to itching [42] - Decreased sleep efficiency, trouble getting to sleep, reduced total sleep time, difficulty waking up, daytime drowsiness, and irritability [43,44] | - Itching frequently [72,73,74] - Disturbed sleep due to pruritus and pain [75] |
| Psychosocial/mental comorbidities | - Behavioral and maladjustment problems, sibling rivalry, abnormal psychological development, childhood low self-esteem, and lack of socialization skills [41], excessive dependency, clinginess, fearfulness [52], and damaged social functioning [53] - Increased risk of ADHD, depression, anxiety, conduct disorder, and autism [55] | - Embarrassment, shame, behavior avoidance, teasing, bullying, stigmatization, disrupted body image, decrease in self-confidence, and social isolation [77,78,79] - Increased risk of anxiety and depression [77] |
| Comorbidities | - Comorbid asthma, hay fever, food allergies, cutaneous infections, and possibly cardio-metabolic comorbidities [63,64] - Shorter stature, delayed growth, and early childhood obesity [66,67,68] | - Increased risk of concomitant obesity, hypertension, heart disease, diabetes, and autoimmune disorders [87,88,89,90] |
| Impact for caregivers and family | - Greater impact than having a child with type 1 diabetes [41] - Sleep deprivation [45], anxiety, depression [60], exhaustion, frustration, helplessness, feelings of guilt, and instability of spousal and other familial relationships [41,53] | - Adverse impact on emotional well-being including stress, sadness, frustration, and depressed mood [15] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Na, C.H.; Chung, J.; Simpson, E.L. Quality of Life and Disease Impact of Atopic Dermatitis and Psoriasis on Children and Their Families. Children 2019, 6, 133. https://doi.org/10.3390/children6120133
Na CH, Chung J, Simpson EL. Quality of Life and Disease Impact of Atopic Dermatitis and Psoriasis on Children and Their Families. Children. 2019; 6(12):133. https://doi.org/10.3390/children6120133
Chicago/Turabian StyleNa, Chan Ho, Janice Chung, and Eric L. Simpson. 2019. "Quality of Life and Disease Impact of Atopic Dermatitis and Psoriasis on Children and Their Families" Children 6, no. 12: 133. https://doi.org/10.3390/children6120133
APA StyleNa, C. H., Chung, J., & Simpson, E. L. (2019). Quality of Life and Disease Impact of Atopic Dermatitis and Psoriasis on Children and Their Families. Children, 6(12), 133. https://doi.org/10.3390/children6120133





