Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis
Abstract
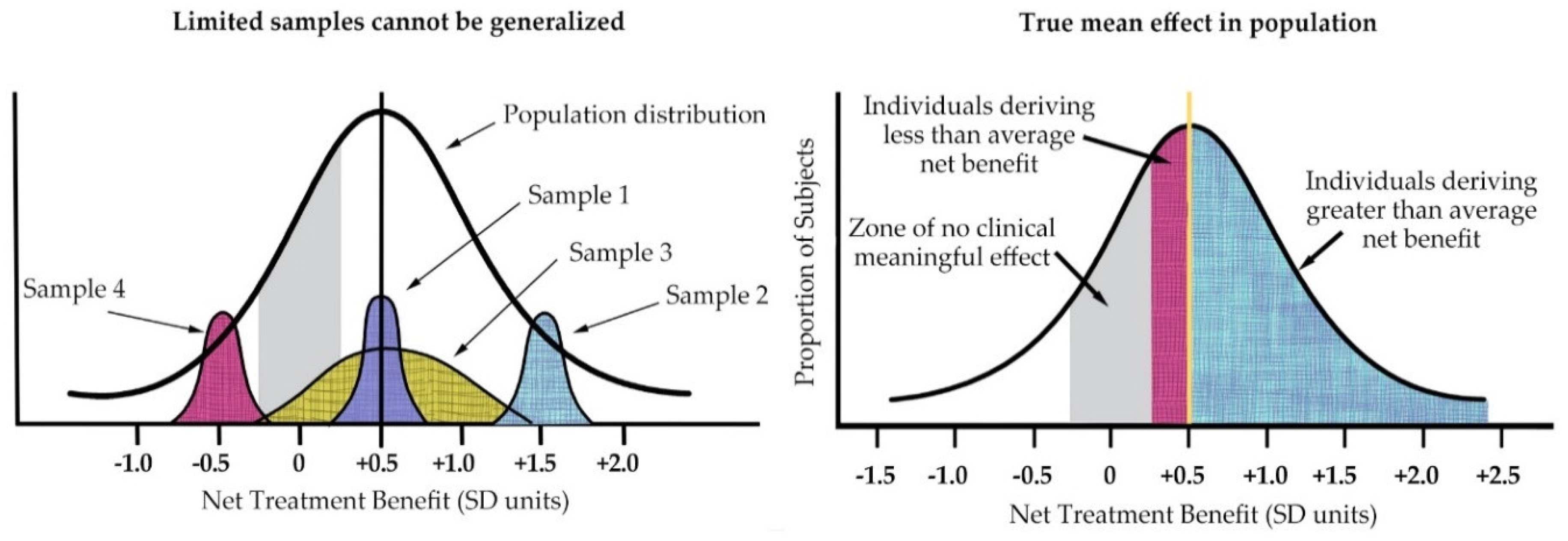
1. Introduction
2. Materials and Methods
2.1. Data Sources
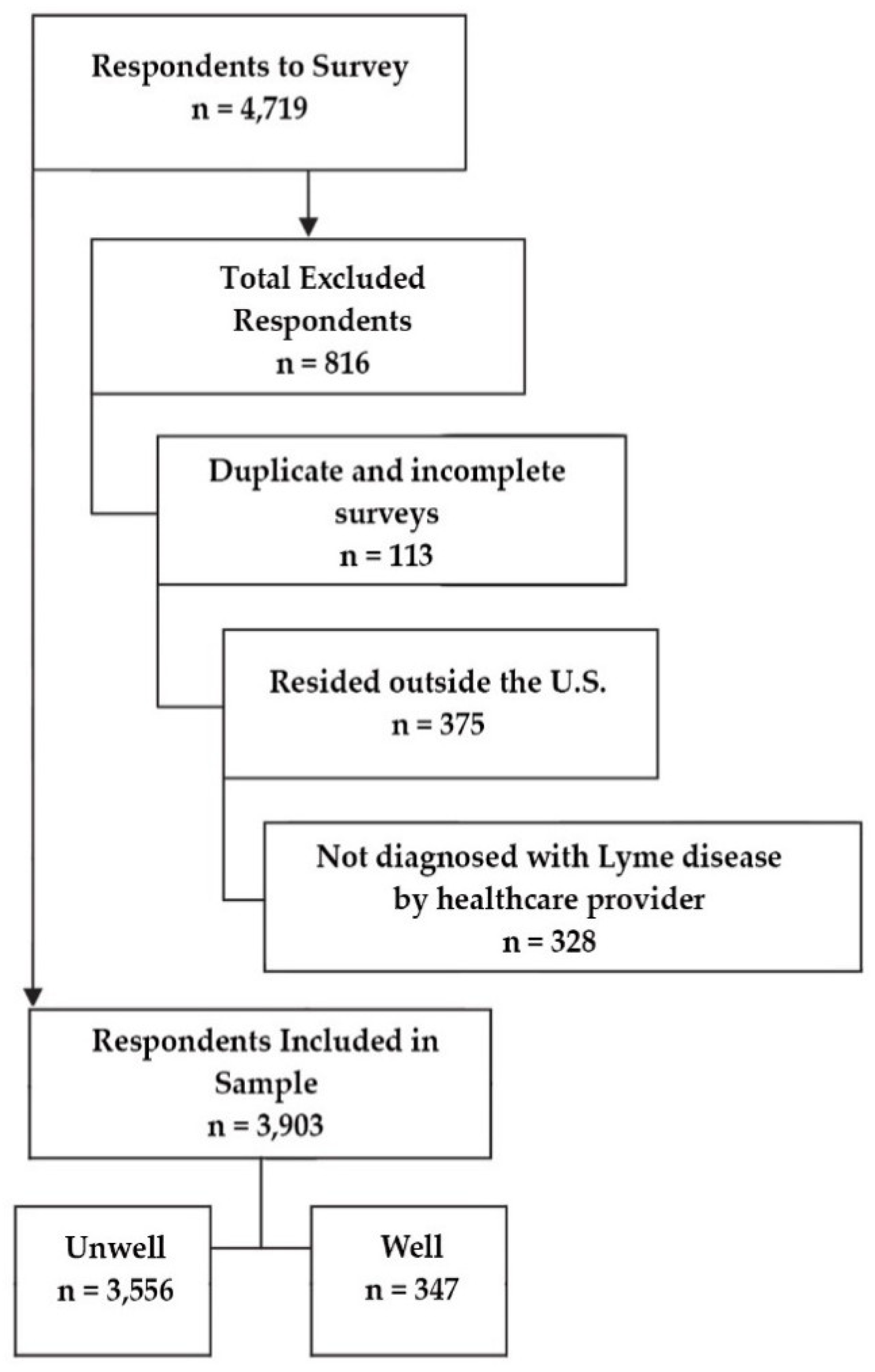
2.2. Study Participants
2.3. Global Rating of Change Scale
3. Results
3.1. General Observations
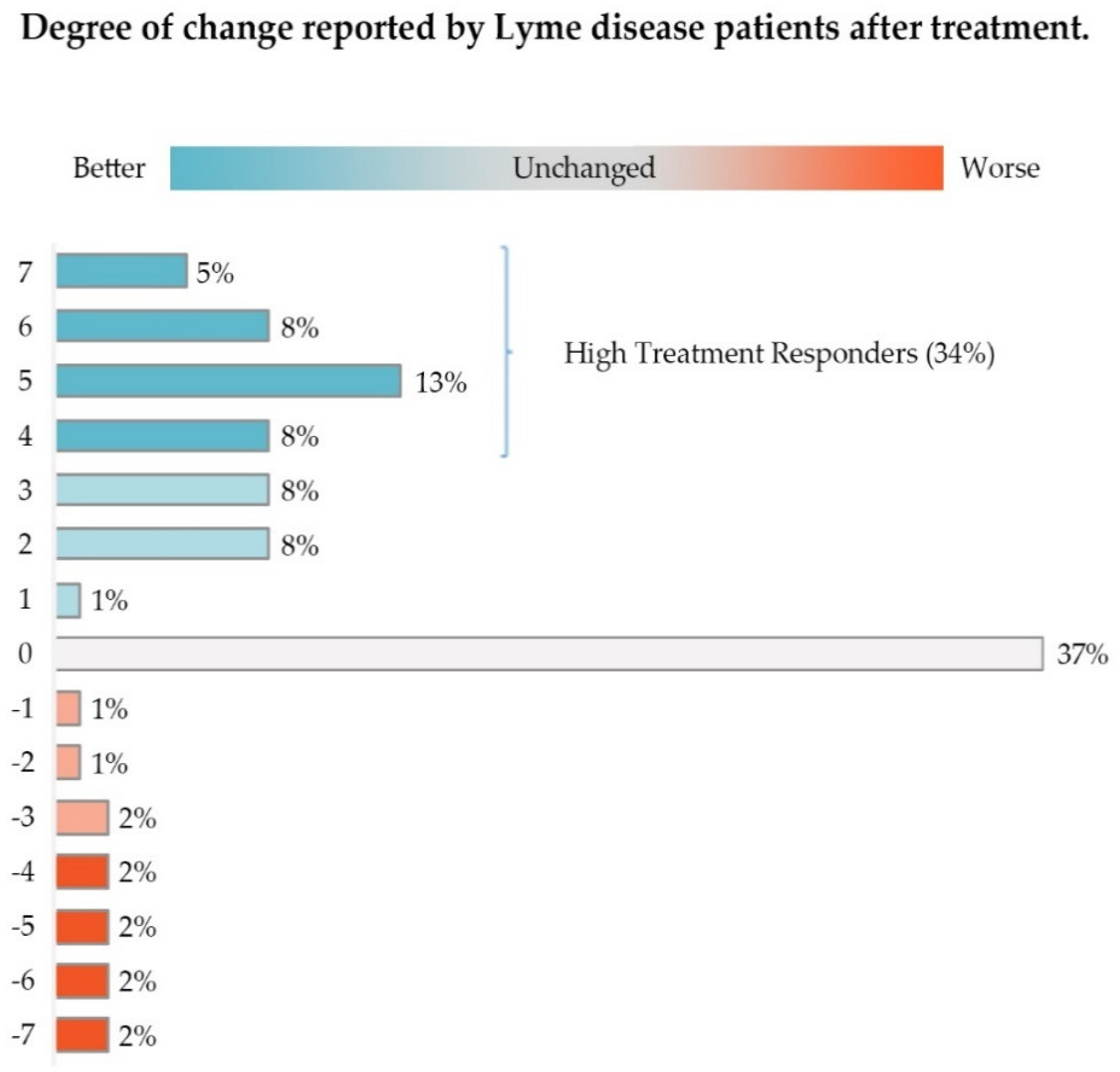
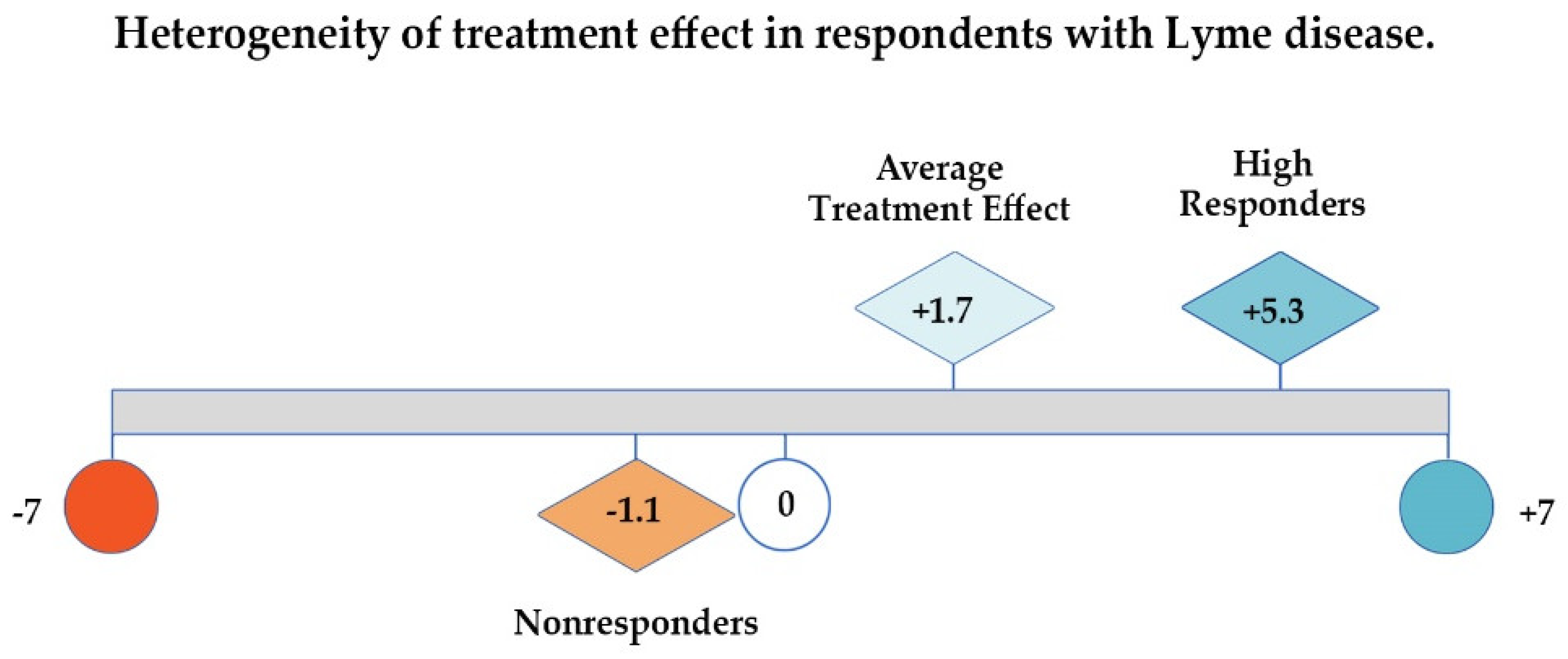
3.2. Global Rating of Change Scale
4. Discussion
4.1. The Value of Patient-Generated Health Data
“Health-related data—including health history, symptoms, biometric data, treatment history, lifestyle choices, and other information—created, recorded, gathered, or inferred by or from patients or their designees (i.e., care partners or those who assist them) to help address a health concern.”[44]
- improve recruitment,
- identify patient research cohorts,
- conduct natural history studies,
- integrate patient-reported and clinical data from multiple sources into single registry,
- stimulate new research on the causes, treatments, and outcomes of diseases,
- accelerate research, knowledge discovery, and scientific insights from patients with under-researched diseases, and
- enhance creative data mining within and across diseases [50].
- enroll diverse patient populations,
- evaluate care as it is actually provided in real-world practice,
- assess complex treatment patterns and treatment combinations, and
- offer the ability to evaluate patient outcomes when clinical trials are not practical or are difficult to conduct (for example, when long-term outcomes are important) [22].
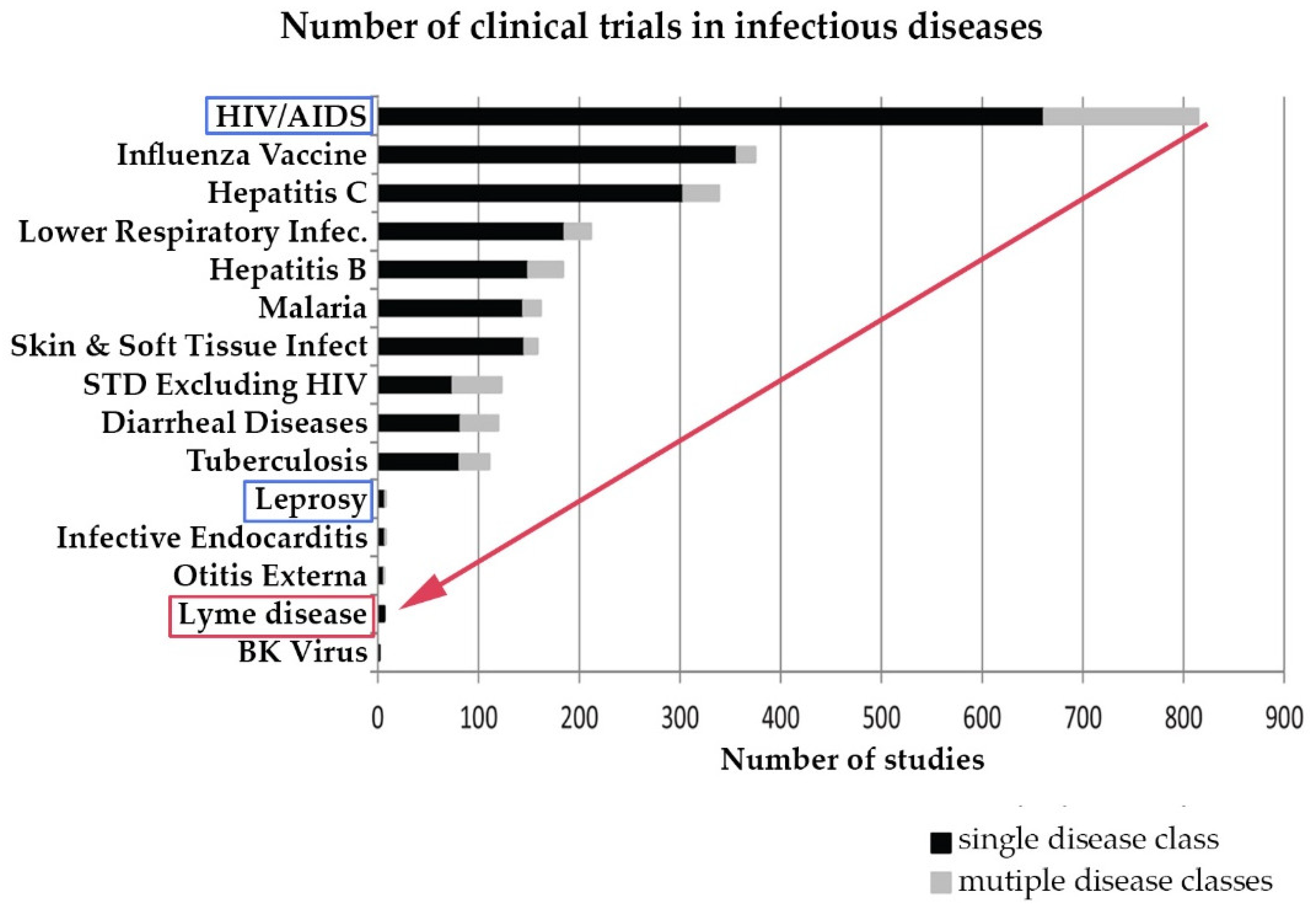
4.2. The Need to Accelerate Research in Treatment of Lyme Disease
4.3. Samples and Outcomes in Lyme Disease Studies Reflect a Heterogeneous Patient Population
4.4. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. MyLymeData Patient Registry Survey Overview
| Medical History | Patient Characteristics |
|
|
| Treatment Risks and Benefits | Changes over Time |
|
|
Appendix B. Potential Clinical Research Criteria for Pragmatic Big Data Trials
- Required
- ○
- Physician diagnosed
- ○
- Clinical symptoms consistent with Lyme disease
- ○
- Late stage Lyme disease (untreated for ≥6 months after symptom onset) or chronic Lyme disease (persistent symptoms for ≥6 months after treatment with <30 days of antibiotics)
- Strongly supportive
- ○
- Positive serological testing
- ○
- Fulfills CDC surveillance Western blot criteria
- ○
- Fulfills Ma/Engstrom criteria
- ○
- Seropositive for other tick-borne conditions
- Supportive
- ○
- History of EM rash
- ○
- Known or possible tick bite
- ○
- Risk of exposure
Appendix C. Data Sources and Limitations
| Data Source | Strengths | Weaknesses |
| Claims Data | Ubiquitous Large Standardized | Insurance covered care only Designed for billing purposes No “results” Nonspecific Not timely |
| Academic case data | Well characterized samples Rigorous methodology Lab measures Assessments (Results) | EHR siloed Physician time constraints Practice constraints limit treatments Convenience sample |
| Community clinical data | Real-world clinical care N-of-1 practices Lab measures | EHR siloed or paper/nonexistent Physician time constraints Convenience sample |
| Patient registries | Real-world clinical care Standardized Timely Labor burden on patient Cross-silo | Emerging innovation Patient generated Self-selected sample Accuracy/Recall |
References
- Centers for Disease Control and Prevention. CDC Provides Estimate of Americans Diagnosed with Lyme Disease Each Year. 2013. Available online: http://www.cdc.gov/media/releases/2013/p0819-lyme-disease.html (accessed on 30 September 2018).
- Aucott, J.N.; Rebman, A.W.; Crowder, L.A.; Kortte, K.B. Post-treatment Lyme disease syndrome symptomatology and the impact on life functioning: Is there something here? Qual. Life Res. 2013, 22, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, A.G.; Herman, R.J.; Rebman, A.; Moon, K.A.; Aucott, J.; Heaney, C.; Schwartz, B.S. Obstacles to diagnosis and treatment of Lyme disease in the USA: A qualitative study. BMJ Open 2018, 8, e021367. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Aylward, A.; Stricker, R.B. Healthcare access and burden of care for patients with Lyme disease: A large united states survey. Health Policy 2011, 102, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Wilcox, S.; Mankoff, J.; Stricker, R.B. Severity of chronic Lyme disease compared to other chronic conditions: A quality of life survey. PeerJ 2014, 2, e322. [Google Scholar] [CrossRef] [PubMed]
- Asch, E.S.; Bujak, D.I.; Weiss, M.; Peterson, M.G.; Weinstein, A. Lyme disease: An infectious and postinfectious syndrome. J. Rheumatol. 1994, 21, 454–461. [Google Scholar] [PubMed]
- Shadick, N.A.; Phillips, C.B.; Logigian, E.L.; Steere, A.C.; Kaplan, R.F.; Berardi, V.P.; Duray, P.H.; Larson, M.G.; Wright, E.A.; Ginsburg, K.S.; et al. The long-term clinical outcomes of Lyme disease. A population-based retrospective cohort study. Ann. Intern. Med. 1994, 121, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Shadick, N.A.; Phillips, C.B.; Sangha, O.; Logigian, E.L.; Kaplan, R.F.; Wright, E.A.; Fossel, A.H.; Fossel, K.; Berardi, V.; Lew, R.A.; et al. Musculoskeletal and neurologic outcomes in patients with previously treated Lyme disease. Ann. Intern. Med. 1999, 131, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Treib, J.; Fernandez, A.; Haass, A.; Grauer, M.T.; Holzer, G.; Woessner, R. Clinical and serologic follow-up in patients with neuroborreliosis. Neurology 1998, 51, 1489–1491. [Google Scholar] [CrossRef] [PubMed]
- Goswami, N.D.; Pfeiffer, C.D.; Horton, J.R.; Chiswell, K.; Tasneem, A.; Tsalik, E.L. The state of infectious diseases clinical trials: A systematic review of clinicaltrials.Gov. PLoS ONE 2013, 8, e77086. [Google Scholar] [CrossRef] [PubMed]
- Report of the Pathogenesis, Transmission, and Treatment Subcommittee to the Tick-Borne Disease Working Group. 4 May 2018. Available online: Https://www.Hhs.Gov/ash/advisory-committees/tickbornedisease/reports/pathogenesis-transmission-2018-5-9/index.Html (accessed on 30 September 2018).
- Zhang, X.; Meltzer, M.I.; Pena, C.A.; Hopkins, A.B.; Wroth, L.; Fix, A.D. Economic impact of Lyme disease. Emerg. Infect. Dis. 2006, 12, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Hook, S.A.; Nelson, C.A.; Mead, P.S. U.S. Public’s experience with ticks and tick-borne diseases: Results from national healthstyles surveys. Ticks Tick Borne Dis. 2015, 6, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Mead, P. Statement by Paul Mead, M.D., M.P.H., Medical Epidemiologist, Division of Vector-Borne Infectious Diseases, National Center for Infectious Diseases, Center for Disease Control and Prevention, U.S. Department of Health and Human Services at Hearing: Cdc’s Lyme Disease Prevention and Control Activities before the Connecticut Department of Public Health and the Connecticut Attorney General’s Office On 29 January 2004. Available online: http://www.hhs.gov/asl/testify/t040129.html (accessed on 30 September 2018).
- Wormser, G.P.; Dattwyler, R.J.; Shapiro, E.D.; Halperin, J.J.; Steere, A.C.; Klempner, M.S.; Krause, P.J.; Bakken, J.S.; Strle, F.; Stanek, G.; et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: Clinical practice guidelines by the infectious diseases society of america. Clin. Infect. Dis. 2006, 43, 1089–1134. [Google Scholar] [CrossRef] [PubMed]
- Fallon, B.A.; Keilp, J.G.; Corbera, K.M.; Petkova, E.; Britton, C.B.; Dwyer, E.; Slavov, I.; Cheng, J.; Dobkin, J.; Nelson, D.R.; et al. A randomized, placebo-controlled trial of repeated iv antibiotic therapy for Lyme encephalopathy. Neurology 2008, 70, 992–1003. [Google Scholar] [CrossRef] [PubMed]
- Klempner, M.; Hu, L.; Evans, J.; Schmid, C.; Johnson, G.; Trevino, R.; Norton, D.; Levy, L.; Wall, D.; McCall, J.; et al. Two controlled trials of antibiotic treatment in patients with persistent symptoms and a history of Lyme disease. N. Engl. J. Med. 2001, 345, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Krupp, L.B.; Hyman, L.G.; Grimson, R.; Coyle, P.K.; Melville, P.; Ahnn, S.; Dattwyler, R.; Chandler, B. Study and treatment of post Lyme disease (Stop-LD): A randomized double masked clinical trial. Neurology 2003, 60, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Wormser, G.P. Clinical practice. Early Lyme disease. N. Engl. J. Med. 2006, 354, 2794–2801. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, R.I.; Hayes-Conroy, A.; Caricchio, R.; Singer, B.H. From evidence based medicine to medicine based evidence. Am. J. Med. 2017, 130, 1246–1250. [Google Scholar] [CrossRef] [PubMed]
- RAND Health. 36-Item Short Form Survey (SF-36). Available online: https://www.rand.org/health/surveys_tools/mos/36-item-short-form/terms.html (accessed on 30 September 2018).
- Gliklich, R.D.N.; Leavy, M. (Eds.) Registries for Evaluating Patient Outcomes: A User’s Guide, 3rd ed.; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2014.
- Centers for Disease Control and Prevention. Hospital Utilization (in Non-Federal Short-Stay Hospitals). Available online: http://www.cdc.gov/nchs/fastats/hospital.htm (accessed on 30 September 2018).
- Centers for Disease Control and Prevention. Measuring Healthy Days; CDC Atlanta: Atlanta, GA, USA, 2000.
- Centers for Disease Control and Prevention. Behavioral Risk Factor Surveillance Survey Data. Available online: http://apps.nccd.cdc.gov/s_broker/WEATSQL.exe/weat/freq_analysis.hsql?survey_year=2010 (accessed on 30 September 2018).
- Centers for Disease Control and Prevention. National Ambulatory Medical Care Survey: 2010 Summary Tables. Available online: http://www.cdc.gov/nchs/data/ahcd/namcs_summary/2010_namcs_web_tables.pdf (accessed on 30 September 2018).
- Centers for Disease Control and Prevention. National Ambulatory Medical Care Survey: 2010 Outpatient Department Summary Tables. Available online: http://www.cdc.gov/nchs/data/ahcd/nhamcs_outpatient/2010_opd_web_tables.pdf (accessed on 30 September 2018).
- Centers for Disease Control and Prevention. National Ambulatory Medical Care Survey: 2010 Emergency Department Summary Tables. Available online: http://www.cdc.gov/nchs/data/ahcd/nhamcs_emergency/2010_ed_web_tables.pdf (accessed on 30 September 2018).
- Agency for Healthcare Research and Quality. Medical Expenditure Panel Survey. Available online: http://meps.ahrq.gov/mepsweb/ (accessed on 19 March 2014).
- Junghard, O.; Halling, K. Responsiveness of measures of heartburn improvement in non-erosive reflux disease. Health Qual. Life Outcomes 2007, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Kamper, S.J.; Maher, C.G.; Mackay, G. Global rating of change scales: A review of strengths and weaknesses and considerations for design. J. Man. Manip. Ther. 2009, 17, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.C.; Hart, D.L.; Stratford, P.W.; Mioduski, J.E. Baseline dependency of minimal clinically important improvement. Phys. Ther. 2011, 91, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Gliklich, R.D.N.; Leavy, M.; Christian, J.B. (Eds.) 21st Century Patient Registries. In Ebook Addendum to Registries for Evaluating Patient Outcomes: A User’s Guide, 3rd ed.; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2018. [Google Scholar]
- Stricker, R.B.; Fessler, M. Chronic lyme disease: A working case definition. Am. J. Infect. Dis. 2018, 14, 1–44. [Google Scholar] [CrossRef]
- Wang, Y.C.; Sindhu, B.S.; Kapellusch, J.; Yen, S.C.; Lehman, L. Global rating of change: Perspectives of patients with lumbar impairments and of their physical therapists. Physiother. Theory Pract. 2018, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jaeschke, R.; Singer, J.; Guyatt, G. Measurement of health status: Ascertaining the minimal clinically important difference. Control. Clin. Trials 1989, 10, 407–415. [Google Scholar] [CrossRef]
- Dahabreh, I.J.; Hayward, R.; Kent, D.M. Using group data to treat individuals: Understanding heterogeneous treatment effects in the age of precision medicine and patient-centred evidence. Int. J. Epidemiol. 2016, 45, 2184–2193. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.; Hannon, J.; Hegedus, E.J.; Kavchak, A.E. Clinimetrics corner: A closer look at the minimal clinically important difference (MCID). J. Man. Manip. Ther. 2012, 20, 160–166. [Google Scholar] [CrossRef] [PubMed]
- National Center for Health Statistics. Health, United States, 2016: With Chartbook on Long-Term Trends in Health; National Center for Health Statistics: Hyattsville, MD, USA, 2017.
- Chomel, B.B.; Kasten, R.W. Bartonellosis, an increasingly recognized zoonosis. J. Appl. Microbiol. 2010, 109, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Lantos, P.M.; Maggi, R.G.; Ferguson, B.; Varkey, J.; Park, L.P.; Breitschwerdt, E.B.; Woods, C.W. Detection of bartonella species in the blood of veterinarians and veterinary technicians: A newly recognized occupational hazard? Vector Borne Zoonotic Dis. 2014, 14, 563–570. [Google Scholar] [CrossRef] [PubMed]
- Workman, T.A. Agency for Healthcare Research and Quality. Engaging Patients in Information Sharing and Data Collection: The Role of Patient-Powered Registries and Research Networks. 2013. Available online: https://www.ncbi.nlm.nih.gov/books/NBK164514/ (accessed on 30 September 2018).
- Mader, L.B.; Harris, T.; Kläger, S.; Wilkinson, I.B.; Hiemstra, T.F. Inverting the patient involvement paradigm: Defining patient led research. Res. Involv. Engagem. 2018, 4, 21. [Google Scholar] [CrossRef] [PubMed]
- Howie, L.; Hirsch, B.; Locklear, T.; Abernethy, A.P. Assessing the value of patient-generated data to comparative effectiveness research. Health Aff. 2014, 33, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. National Amyotrophic Lateral Sclerosis (ALS) Registry. Available online: https://www.cdc.gov/als/ALSJoinALSRegistry.html (accessed on 30 September 2018).
- PatientsLikeMe. Available online: https://www.patientslikeme.com/ (accessed on 30 September 2018).
- Stanford University and Volunteers. Cancerbase. Available online: https://cancerbase.org/ (accessed on 30 September 2018).
- The Duchenne Registry. Available online: https://www.duchenneregistry.org/ (accessed on 30 September 2018).
- Fleurence, R.L.; Beal, A.C.; Sheridan, S.E.; Johnson, L.B.; Selby, J.V. Patient-powered research networks aim to improve patient care and health research. Health Aff. 2014, 33, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Groft, S. Patient Registries As a Prelude to Clinical Trials and Post-Approval Studies; Pediatric Devices for Rare Diseases Food and Drug Administration: White Oak, MD, USA, 8 January 2014.
- Chawla, N.V.; Davis, D.A. Bringing big data to personalized healthcare: A patient-centered framework. J. Gen. Intern. Med. 2013, 28 (Suppl. 3), S660–S665. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Hiv Surveillance Report. 2016; Volume 28. Available online: http://www.cdc.gov/hiv/library/reports/hiv-surveillance.html (accessed on 30 September 2018).
- Centers for Disease Control and Prevention. Lyme and Other Tickborne Diseases. Available online: https://www.cdc.gov/media/dpk/diseases-and-conditions/lyme-disease/index.html (accessed on 30 September 2018).
- National Institute of Allergy and Infectious Diseases. Hiv Infections Drop 18 Percent in Six Years. 14 February 2017. Available online: https://www.hiv.gov/blog/new-hiv-infections-drop-18-percent-in-six-yearsNew (accessed on 30 September 2018).
- Health Resources and Services Administration. National Hansen’s Disease (Leprosy) Program Caring and Curing Since 1894. Available online: https://www.hrsa.gov/hansens-disease/index.html (accessed on 30 September 2018).
- Biau, D.J.; Kernéis, S.; Porcher, R. Statistics in brief: The importance of sample size in the planning and interpretation of medical research. Clin. Orthop. Relat. Res. 2008, 466, 2282–2288. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.H.; Mills, E.J.; Elbourne, D. In the era of systematic reviews, does the size of an individual trial still matter. PLoS Med. 2008, 5, e4. [Google Scholar] [CrossRef] [PubMed]
- Delong, A.K.; Blossom, B.; Maloney, E.; Phillips, S.E. Antibiotic retreatment of Lyme disease in patients with persistent symptoms: A biostatistical review of randomized, placebo-controlled, clinical trials. Contemp. Clin. Trials 2012, 33, 1132–1142. [Google Scholar] [CrossRef] [PubMed]
- Kravitz, R.L.; Duan, N.; Braslow, J. Evidence-based medicine, heterogeneity of treatment effects, and the trouble with averages. Milbank Q. 2004, 82, 661–687. [Google Scholar] [CrossRef] [PubMed]
- Aucott, J.N.; Crowder, L.A.; Kortte, K.B. Development of a foundation for a case definition of post-treatment Lyme disease syndrome. Int. J. Infect. Dis. 2013, 17, e443–e449. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Mervine, P.; Potter, M. New patient-powered research tool can be used to answer important questions about Lyme disease. In Proceedings of the Poster Presentation at the LDA/Columbia Conference, St. Paul, MN, USA, 16 October 2016. [Google Scholar]
- Aucott, J.; Morrison, C.; Munoz, B.; Rowe, P.C.; Schwarzwalder, A.; West, S.K. Diagnostic challenges of early Lyme disease: Lessons from a community case series. BMC Infect. Dis. 2009, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Aucott, J.N.; Seifter, A. Misdiagnosis of early Lyme disease as the summer flu. Orthop. Rev. 2011, 3, e14. [Google Scholar] [CrossRef] [PubMed]
- Stricker, R.B.; Johnson, L. Gender bias in chronic Lyme disease. J. Womens Health 2009, 18, 1717–1718. [Google Scholar] [CrossRef] [PubMed]
- Schwarzwalder, A.; Schneider, M.F.; Lydecker, A.; Aucott, J.N. Sex differences in the clinical and serologic presentation of early Lyme disease: Results from a retrospective review. Gend. Med. 2010, 7, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Adrion, E.R.; Aucott, J.; Lemke, K.W.; Weiner, J.P. Health care costs, utilization and patterns of care following Lyme disease. PLoS ONE 2015, 10, e0116767. [Google Scholar] [CrossRef] [PubMed]
- Kent, D.M.; Hayward, R.A. Limitations of applying summary results of clinical trials to individual patients: The need for risk stratification. JAMA 2007, 298, 1209–1212. [Google Scholar] [CrossRef] [PubMed]
- Connor, S. Glaxo Chief: Our Drugs Do Not Work on Most Patients. The Independent, 8 December 2003. [Google Scholar]
- Kent, D.M.; Nelson, J.; Dahabreh, I.J.; Rothwell, P.M.; Altman, D.G.; Hayward, R.A. Risk and treatment effect heterogeneity: Re-analysis of individual participant data from 32 large clinical trials. Int. J. Epidemiol. 2016, 45, 2075–2088. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, N.; Franklin, P.; Haynes, K. Direct-to-patient registry and other patient-centric designs. In 21st Century Patient Registries: Registries for Evaluating Patient Outcomes: A User’s Guide, 3rd ed.; Gliklich, R.E., Dreyer, N.A., Leavy, M.B., Eds.; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2018. [Google Scholar]
- Cohen, D.J.; Keller, S.R.; Hayes, G.R.; Dorr, D.A.; Ash, J.S.; Sittig, D.F. Integrating patient-generated health data into clinical care settings or clinical decision-making: Lessons learned from project healthdesign. JMIR Hum. Factors 2016, 3, e26. [Google Scholar] [CrossRef] [PubMed]
- Wood, W.A.; Bennett, A.V.; Basch, E. Emerging uses of patient generated health data in clinical research. Mol. Oncol. 2015, 9, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Bayliss, M.; Rendas-Baum, R.; White, M.K.; Maruish, M.; Bjorner, J.; Tunis, S.L. Health-related quality of life (HRQL) for individuals with self-reported chronic physical and/or mental health conditions: Panel survey of an adult sample in the united states. Health Qual. Life Outcomes 2012, 10, 154. [Google Scholar] [CrossRef] [PubMed]
- Bytzer, P. Assessment of reflux symptom severity: Methodological options and their attributes. Gut 2004, 53 (Suppl. 4), iv28–iv34. [Google Scholar] [CrossRef] [PubMed]
| Variable | Count (% of Working Sample) |
|---|---|
| Gender | |
| Female | 3250 (83%) |
| Mean age | 49 |
| Education a | |
| High school or less | 340 (9%) |
| Some college or associate degree | 1265 (34%) |
| Bachelor degree | 1139 (31%) |
| Graduate school degree | 945 (25%) |
| Family income b | |
| <$25k | 485 (14%) |
| $25–50k | 542 (15%) |
| $50–75k | 547 (16%) |
| $75–100k | 408 (12%) |
| >$100k | 1025 (29%) |
| Geography c | |
| East | 1274 (33%) |
| Midwest | 571 (15%) |
| South | 1021 (26%) |
| West | 1004 (26%) |
| Variable | Count (% of Working Sample) |
|---|---|
| Current stage of illness | |
| Chronic LD a | 61% |
| Late untreated LD b | 18% |
| Early Lyme disease c | 6% |
| Don’t know/Other | 15% |
| Stage when diagnosed | |
| Late untreated LD b | 70% |
| Early Lyme disease c | 22% |
| Don’t know/Other | 8% |
| Key diagnostic factors | |
| Clinician diagnosed (entry criteria for registry) d | 100% |
| Recollection of tick bite | 41% |
| Recollection of EM rash e | 34% |
| With supportive lab tests | 78% |
| 1 or more coinfection | 60% |
| Self-reported health status as fair or poor | 65% |
| Disabled (with or without disability benefits) | 32% |
| Better/Worse/Unchanged | Degree of Change | Likert Score | n (% of Total) | Assigned Group |
|---|---|---|---|---|
| Better | Hardly better at all | 1 | 43 (1.22) | Low Responders |
| A little better | 2 | 269 (7.61) | Low Responders | |
| Somewhat better | 3 | 298 (8.43) | Low Responders | |
| Total | 17.26% | Low Responders | ||
| Moderately better | 4 | 295 (8.34) | High Responders | |
| A good deal better | 5 | 450 (12.73) | High Responders | |
| A great deal better | 6 | 289 (8.17) | High Responders | |
| A very great deal better | 7 | 191 (5.40) | High Responders | |
| Total | 34.64% | High Responders | ||
| Total Better | 51.9% | |||
| Unchanged a | 0 | 1293 (36.57) | Nonresponders | |
| Worse | A very great deal worse | −7 | 64 (1.81) | Nonresponders |
| A great deal worse | −6 | 64 (1.81) | Nonresponders | |
| A good deal worse | −5 | 85 (2.40) | Nonresponders | |
| Moderately worse | −4 | 71 (2.01) | Nonresponders | |
| Somewhat worse | −3 | 66 (1.87) | Nonresponders | |
| A little worse | −2 | 35 (0.99) | Nonresponders | |
| Hardly worse at all | −1 | 23 (0.65) | Nonresponders | |
| Total Worse | 11.54% | Nonresponders | ||
| Total | 100% |
| Study Type | Trial | Screened | Enrolled | Yield | Time to Recruit |
|---|---|---|---|---|---|
| RCT * | Klempner (2001) | 1996 | 129 | 7% | 3.3 years |
| RCT | Krupp (2003) | 512 | 56 | 11% | 2.5 years |
| RCT | Fallon (2008) | 3368 | 37 | 1% | 4 years |
| Big Data | Johnson (2014) | 5357 | 3090 | 58% | 6 months |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, L.; Shapiro, M.; Mankoff, J. Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis. Healthcare 2018, 6, 124. https://doi.org/10.3390/healthcare6040124
Johnson L, Shapiro M, Mankoff J. Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis. Healthcare. 2018; 6(4):124. https://doi.org/10.3390/healthcare6040124
Chicago/Turabian StyleJohnson, Lorraine, Mira Shapiro, and Jennifer Mankoff. 2018. "Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis" Healthcare 6, no. 4: 124. https://doi.org/10.3390/healthcare6040124
APA StyleJohnson, L., Shapiro, M., & Mankoff, J. (2018). Removing the Mask of Average Treatment Effects in Chronic Lyme Disease Research Using Big Data and Subgroup Analysis. Healthcare, 6(4), 124. https://doi.org/10.3390/healthcare6040124





