Innovating the Concept and Practice of Two-Dimensional Gel Electrophoresis in the Analysis of Proteomes at the Proteoform Level
Abstract
1. Introduction
2. Definition of Spots and Pixels
3. Relationship between Proteoform, Protein, and Proteome
4. A 2DE Spot Contains Many Proteoforms Derived from Different Genes
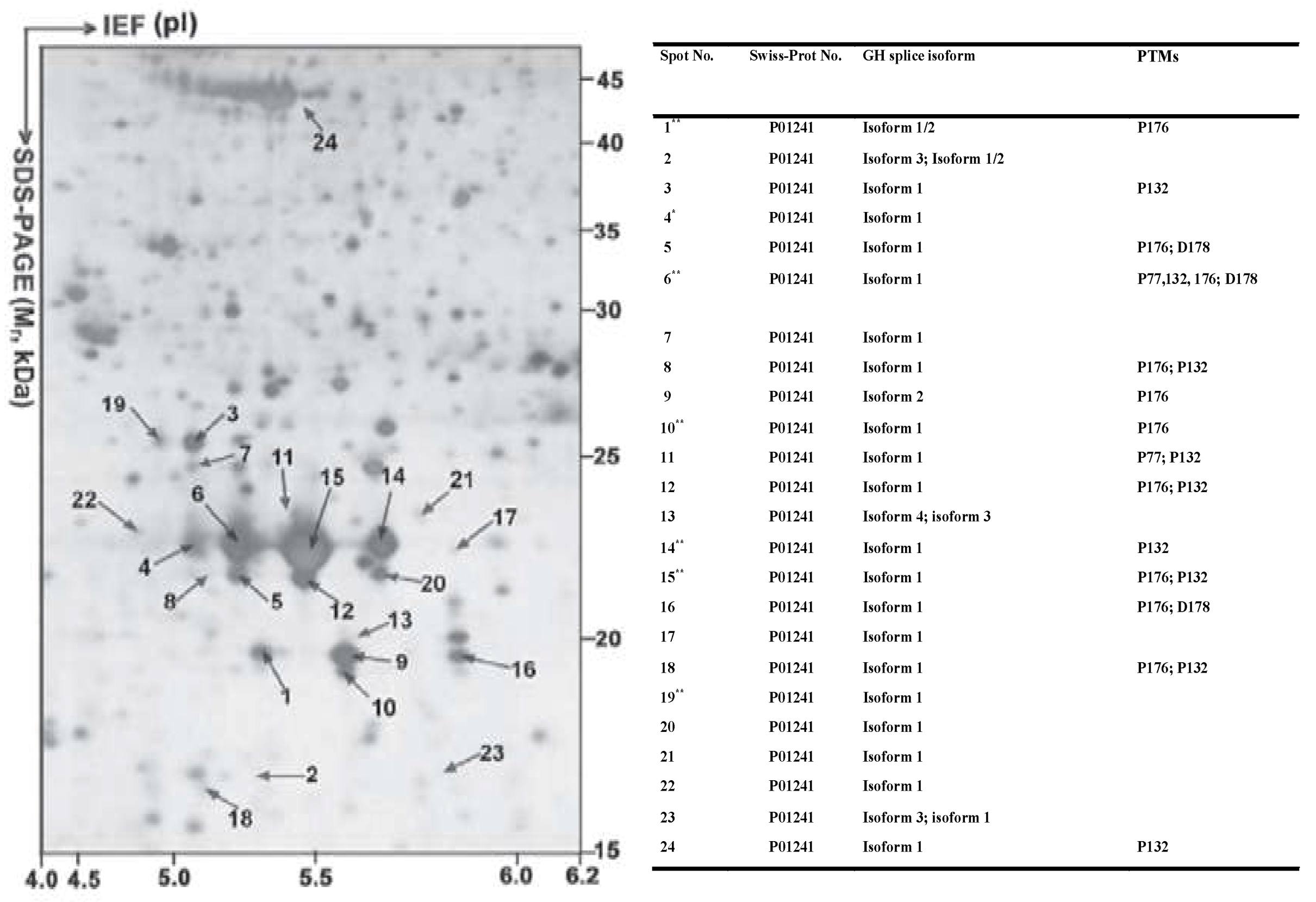
5. Proteoforms from One Gene are Distributed within Different 2DE Spots
6. Most Proteoforms in a 2DE Spot are of Low-Abundance
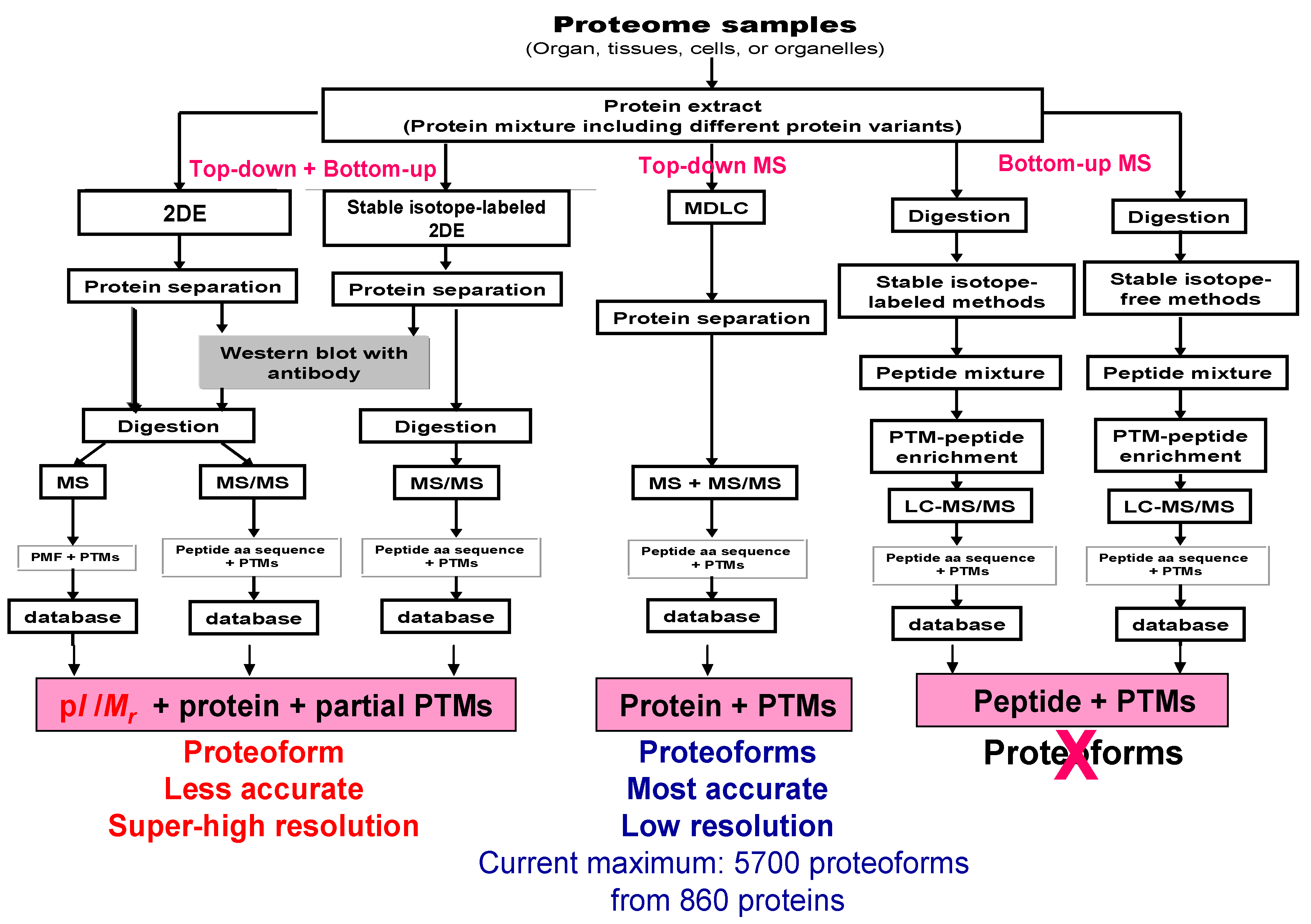
7. Stable Isotope-Labeled 2DE-LC-MS/MS for the Large-Scale Analysis of Proteoforms
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 2DE | two-dimensional gel electrophoresis |
| DDA | data-dependent acquisition |
| DIA | data-independent acquisition |
| hGH | human growth hormone |
| hPRL | human prolactin |
| IEF | isoelectric focusing |
| IPG | immobilized pH gradient gel |
| ICAT | isotope-coded affinity tags |
| iTRAQ | isobaric tags for relative and absolute quantification |
| IU | image unit |
| LC | liquid chromatography |
| Mr | relative molecular mass |
| MS | mass spectrometry |
| MS/MS | tandem mass spectrometry |
| OD | optical density |
| pI | isoelectric point |
| PTM | post-translational modification |
| SDS-PAGE | sodium dodecyl sulfate-polyacrylamide gel electrophoresis |
| SILAC | stable isotope labeling of amino acids in cell culture |
| SRM | selected reaction monitoring |
| SWATH | sequential window acquisition of all theoretical mass spectra |
| TMT | tandem mass tags |
References
- Smithies, O.; Poulik, M.D. Two-dimensional electrophoresis of serum proteins. Nature 1956, 177, 1033. [Google Scholar] [CrossRef] [PubMed]
- Raymond, S. Acrylamide gel electrophoresis. Ann. N. Y. Acad. Sci. 1964, 121, 350–365. [Google Scholar] [CrossRef] [PubMed]
- Kaltschmidt, E.; Wittmann, H.G. Ribosomal proteins. VII. Two-dimensional polyacrylamide gel electrophoresis for fingerprinting of ribosomal proteins. Anal. Biochem. 1970, 36, 401–412. [Google Scholar] [CrossRef]
- MacGillivray, A.J.; Rickwood, D. The heterogeneity of mouse-chromatin nonhistone proteins as evidenced by two-dimensional polyacrylamide-gel electrophoresis and ion-exchange chromatography. Eur. J. Biochem. 1974, 41, 181–190. [Google Scholar] [CrossRef]
- Klose, J. Protein mapping by combined isoelectric focusing and electrophoresis of mouse tissues. A novel approach to testing for induced point mutations in mammals. Humangenetik 1975, 26, 231–243. [Google Scholar]
- Scheele, G.A. Two-dimensional gel analysis of soluble proteins. Charaterization of guinea pig exocrine pancreatic proteins. J. Biol. Chem. 1975, 250, 5375–5385. [Google Scholar]
- Iborra, F.; Buhler, J.M. Protein subunit mapping. A sensitive high resolution method. Anal. Biochem. 1976, 74, 503–511. [Google Scholar] [CrossRef]
- O’Farrell, P.H. High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 1975, 250, 4007–4021. [Google Scholar]
- Zhan, X.; Desiderio, D.M. Heterogeneity analysis of the human pituitary proteome. Clin. Chem. 2003, 49, 1740–1751. [Google Scholar] [CrossRef]
- Zhan, X.; Desiderio, D.M.; Wang, X.; Zhan, X.; Guo, T.; Li, M.; Peng, F.; Chen, X.; Yang, H.; Zhang, P.; et al. Identification of the proteomic variations of invasive relative to non-invasive non-functional pituitary adenomas. Electrophoresis 2014, 35, 2184–2194. [Google Scholar]
- Wasinger, V.C.; Cordwell, S.J.; Cerpa-Poljak, A.; Yan, J.X.; Gooley, A.A.; Wilkins, M.R.; Duncan, M.W.; Harris, R.; Williams, K.L.; Humphery-Smith, I. Progress with gene-product mapping of the Mollicutes: Mycoplasma genitalium. Electrophoresis 1995, 16, 1090–1094. [Google Scholar] [CrossRef] [PubMed]
- Klose, J.; Kobalz, U. Two-dimensional electrophoresis of proteins: An updated protocol and implications for a functional analysis of the genome. Electrophoresis 1995, 16, 1034–1059. [Google Scholar] [CrossRef] [PubMed]
- Zimny-Arndt, U.; Schmid, M.; Ackermann, R.; Jungblut, P.R. Classical proteomics: Two-dimensional electrophoresis/MALDI mass spectrometry. Methods Mol. Biol. 2009, 492, 65–91. [Google Scholar] [PubMed]
- Görg, A.; Obermaier, C.; Boguth, G.; Harder, A.; Scheibe, B. The current state of two-dimensional electrophoresis with immobilized pH gradients. Electrophoresis 2000, 21, 1037–1053. [Google Scholar] [CrossRef]
- Zhan, X.; Desiderio, D.M. Differences in the spatial and quantitative reproducibility between two second-dimensional gel electrophoresis systems. Electrophoresis 2003, 24, 1834–1846. [Google Scholar] [CrossRef]
- Zhan, X.; Desiderio, D.M. Spot volume vs. amount of protein loaded onto a gel: A detailed, statistical comparison of two gel electrophoresis systems. Electrophoresis 2003, 24, 1818–1833. [Google Scholar] [CrossRef]
- D’Silva, A.M.; Hyett, J.A.; Coorssen, J.R. A routine ‘top-down’ approach to analysis of the human serum proteome. Proteomes 2017, 5, 13. [Google Scholar] [CrossRef]
- D’Silva, A.M.; Hyett, J.A.; Coorssen, J.R. Proteomic analysis of first trimester maternal serum to identify candidate biomarkers potentially predictive of spontaneous preterm birth. J. Proteom. 2018, 178, 31–42. [Google Scholar] [CrossRef]
- Butt, R.H.; Coorssen, J.R. Postfractionation for enhanced proteomic analyses: Routine electrophoretic methods increase the resolution of standard 2D-PAGE. J. Proteome Res. 2005, 4, 982–991. [Google Scholar] [CrossRef]
- Churchward, M.A.; Butt, R.H.; Lang, J.C.; Hsu, K.K.; Coorssen, J.R. Enhanced detergent extraction for analysis of membrane proteomes by two-dimensional gel electrophoresis. Proteome Sci. 2005, 3, 5. [Google Scholar] [CrossRef]
- Butt, R.H.; Coorssen, J.R. Pre-extraction sample handling by automated frozen disruption significantly improves subsequent proteomic analyses. J. Proteome Res. 2006, 5, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.P.; Prasad, K.A.; Padula, M.P.; Coorssen, J.R. Deep imaging: How much of the proteome does current top-down technology already resolve? PLoS ONE 2014, 9, e86058. [Google Scholar] [CrossRef] [PubMed]
- Noaman, N.; Abbineni, P.S.; Withers, M.; Coorssen, J.R. Coomassie staining provides routine (sub)femtomole in-gel detection of intact proteoforms: Expanding opportunities for genuine top-down proteomics. Electrophoresis 2017, 38, 3086–3099. [Google Scholar] [CrossRef] [PubMed]
- Jungblut, P.; Klose, J. Composition and genetic variability of Heparin-Sepharose CL-6B protein fractions obtained from the solubilized proteins of mouse organs. Biochem. Genet. 1986, 24, 925–939. [Google Scholar] [CrossRef]
- Santos, F.M.; Albuquerque, T.; Gaspar, L.M.; Dias, J.M.L.; Castro, E.; Sousa, J.P.; Paradela, A.; Tomaz, C.T.; Passarinha, L.A. Refinement of two-dimensional electrophoresis for vitreous proteome profiling using an artificial neural network. Anal. Bioanal. Chem. 2019, 411, 5115–5126. [Google Scholar] [CrossRef] [PubMed]
- Posch, A. Sample preparation guidelines for two-dimensional electrophoresis. Arch. Physiol. Biochem. 2014, 120, 192–197. [Google Scholar] [CrossRef]
- Kočevar, N.; Grazio, S.F.; Komel, R. Two-dimensional gel electrophoresis of gastric tissue in an alkaline pH range. Proteomics. 2014, 14, 311–321. [Google Scholar] [CrossRef]
- Moche, M.; Albrecht, D.; Maaß, S.; Hecker, M.; Westermeier, R.; Büttner, K. The new horizon in 2D electrophoresis: New technology to increase resolution and sensitivity. Electrophoresis 2013, 34, 1510–1518. [Google Scholar] [CrossRef]
- Zabel, C.; Klose, J. High-resolution large-gel 2DE. Methods Mol. Biol. 2009, 519, 311–338. [Google Scholar]
- Afjehi-Sadat, L.; Lubec, G. Identification of enzymes and activity from two-dimensional gel electrophoresis. Nat. Protoc. 2007, 2, 2318–2324. [Google Scholar] [CrossRef]
- Khoudoli, G.A.; Porter, I.M.; Blow, J.J.; Swedlow, J.R. Optimisation of the two-dimensional gel electrophoresis protocol using the Taguchi approach. Proteome Sci. 2004, 2, 6. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kwiatkowski, M.; Wurlitzer, M.; Krutilin, A.; Kiani, P.; Nimer, R.; Omidi, M.; Mannaa, A.; Bussmann, T.; Bartkowiak, K.; Kruber, S.; et al. Homogenization of tissues via picosecond-infrared laser (PIRL) ablation: Giving a closer view on the in-vivo composition of protein species as compared to mechanical homogenization. J. Proteom. 2016, 134, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Li, J.; Guo, T.; Yang, H.; Li, M.; Sang, S.; Li, X.; Desiderio, D.M.; Zhan, X. Nitroproteins in human astrocytomas discovered by gel electrophoresis and tandem mass spectrometry. J. Am. Soc. Mass Spectrum. 2015, 26, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.; Yang, H.; Peng, F.; Li, J.; Mu, Y.; Long, Y.; Cheng, T.; Huang, Y.; Li, Z.; Lu, M.; et al. How many proteins can be identified in a 2-DE gel spot within an analysis of a complex human cancer tissue proteome? Electrophoresis 2018, 39, 965–980. [Google Scholar] [CrossRef]
- Zhan, X.; Li, N.; Zhan, X.; Qian, S. Revival of 2DE-LC/MS in proteomics and its potential for large-scale study of human proteoforms. Med One 2018, 3, e180008. [Google Scholar]
- Schmidt, F.; Donahoe, S.; Hagens, K.; Mattow, J.; Schaible, U.E.; Kaufmann, S.H.E.; Aebersold, R.; Jungblut, P.R. Complementary analysis of the Mycobacterium tuberculosis proteome by two-dimensional electrophoresis and isotope coded affinity tag technology. Mol. Cell. Proteom. 2004, 3, 24–42. [Google Scholar] [CrossRef]
- Jungblut, P.; Thiede, B.; Zimny-Arndt, U.; Müller, E.C.; Scheler, C.; Wittmann-Liebold, B.; Otto, A. Resolution power of two-dimensional electrophoresis and identification of proteins from gels. Electrophoresis 1996, 17, 839–847. [Google Scholar] [CrossRef]
- Jungblut, P.R.; Holzhütter, H.G.; Apweiler, R.; Schlüter, H. The speciation of the proteome. Chem. Cent. J. 2008, 2, 16. [Google Scholar] [CrossRef]
- Schlüter, H.; Apweiler, R.; Holzhütter, H.G.; Jungblut, P.R. Finding one’s way in proteomics: A protein species nomenclature. Chem. Cent. J. 2009, 3, 11. [Google Scholar] [CrossRef]
- Smith, L.M.; Kelleher, N.L. Consortium for Top Down Proteomics. Proteoform: A single term describing protein complexity. Nat. Methods 2013, 10, 186–187. [Google Scholar] [CrossRef]
- Coorssen, J.R.; Yergey, A.L. Proteomics is analytical chemistry: Fitness-for-purpose in the application of top-down and bottom-up analyses. Proteomes 2015, 3, 440–453. [Google Scholar] [CrossRef] [PubMed]
- Naryzhny, S.N.; Lisitsa, A.V.; Zgoda, V.G.; Ponomarenko, E.A.; Archakov, A.I. 2DE-based approach for estimation of number of protein species in a cell. Electrophoresis 2014, 35, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Ponomarenko, E.A.; Poverennaya, E.V.; Ilgisonis, E.V.; Pyatnitskiy, M.A.; Kopylov, A.T.; Zgoda, V.G.; Lisitsa, A.V.; Archakov, A.I. The size of the human proteome: The width and depth. Int. J. Anal. Chem. 2016, 2016, 7436849. [Google Scholar] [CrossRef] [PubMed]
- Aebersold, R.; Agar, J.N.; Amster, I.J.; Baker, M.S.; Bertozzi, C.R.; Boja, E.S.; Costello, C.E.; Cravatt, B.F.; Fenselau, C.; Garcia, B.A.; et al. How many human proteoforms are there? Nat. Chem. Biol. 2018, 14, 206–214. [Google Scholar] [CrossRef]
- Kelleher, N.L. A cell-based approach to the human proteome project. J. Am. Soc. Mass Spectrom. 2012, 23, 1617–1624. [Google Scholar] [CrossRef]
- Campostrini, N.; Areces, L.B.; Rappsilber, J.; Pietrogrande, M.C.; Dondi, F.; Pastorino, F.; Ponzoni, M.; Righetti, P.G. Spot overlapping in two-dimensional maps: A serious problem ignored for much too long. Proteomics 2005, 5, 2385–2395. [Google Scholar] [CrossRef]
- Thiede, B.; Koehler, C.J.; Strozynski, M.; Treumann, A.; Stein, R.; Zimny-Arndt, U.; Schmid, M.; Jungblut, P.R. High resolution quantitative proteomics of HeLa cells protein species using stable isotope labeling with amino acids in cell culture(SILAC), two-dimensional gel electrophoresis(2DE) and nano-liquid chromatograpohy coupled to an LTQ-OrbitrapMass spectrometer. Mol. Cell. Proteom. 2013, 12, 529–538. [Google Scholar]
- Lim, H.; Eng, J.; Yates, J.R., 3rd; Tollaksen, S.L.; Giometti, C.S.; Holden, J.F.; Adams, M.W.; Reich, C.I.; Olsen, G.J.; Hays, L.G. Identification of 2D-gel proteins: A comparison of MALDI/TOF peptide mass mapping to mu LC-ESI tandem mass spectrometry. J. Am. Soc. Mass Spectrom. 2003, 14, 957–970. [Google Scholar] [CrossRef]
- Naryzhny, S.N.; Maynskova, M.A.; Zgoda, V.G.; Ronzhina, N.L.; Kleyst, O.A.; Vakhrushev, I.V.; Archakov, A.I. Virtual-experimental 2DE approach in chromosome-centric human proteome project. J. Proteome Res. 2016, 15, 525–530. [Google Scholar] [CrossRef]
- Naryzhny, S.N.; Maynskova, M.A.; Zgoda, V.G.; Ronzhina, N.L.; Novikova, S.E.; Belyakova, N.V.; Kleyst, O.A.; Legina, O.K.; Pantina, R.A. Proteomic profiling of highgrade glioblastoma using virtual-experimental 2DE. J. Proteom. Bioinform. 2016, 9, 158–165. [Google Scholar] [CrossRef]
- Zhan, X.; Desiderio, D.M. A reference map of a human pituitary adenoma proteome. Proteomics 2003, 3, 699–713. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhan, X.; Li, M.; Li, G.; Zhang, P.; Xiao, Z.; Shao, M.; Peng, F.; Hu, R.; Chen, Z. Mitochondrial proteomics of nasopharyngeal carcinoma metastasis. BMC Med. Genom. 2012, 5, 62. [Google Scholar] [CrossRef] [PubMed]
- Arentz, G.; Weiland, F.; Oehler, M.K.; Hoffmann, P. State of the art of 2D DIGE. Proteom. Clin. Appl. 2015, 9, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, F.; Centeno, D.; Rofidal, V.; Tauzin, M.; Martin, O.; Sonamerer, N.; Rossignol, M. Different impact of staining procedures using visible stains and fluorescent dyes for large-scale investigation of proteomes by MALDI-TOF mass spectrometry. J. Proteome Res. 2006, 5, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Gauci, V.J.; Padula, M.P.; Coorssen, J.R. Coomassie blue staining for high sensitivity gel-based proteomics. J. Proteom. 2013, 90, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Liew, Y.K.; Neela, V.; Hamat, R.A.; Nordin, S.A.; Chong, P.P. Modified silver staining in 2DE improves protein detection even at extremely low sample concentration. Electrophoresis 2013, 34, 397–400. [Google Scholar] [CrossRef]
- Gauci, V.J.; Wright, E.P.; Coorssen, J.R. Quantitative proteomics: Assessing the spectrum of in-gel protein detection methods. J. Chem. Biol. 2011, 4, 3–29. [Google Scholar] [CrossRef]
- Swindell, W.R.; Remmer, H.A.; Sarkar, M.K.; Xing, X.; Barnes, D.H.; Wolterink, L.; Voorhees, J.J.; Nair, R.P.; Johnston, A.; Elder, J.T.; et al. Proteogenomic analysis of psoriasis reveals discordant and concordant changes in mRNA and protein abundance. Genome Med. 2015, 7, 86. [Google Scholar] [CrossRef]
- Mehdi, A.M.; Patrick, R.; Bailey, T.L.; Bod’en, M. Predicting the dynamics of protein abundance. Mol. Cell. Proteom. 2014, 13, 1330–1340. [Google Scholar] [CrossRef]
- Bienvenut, W.V.; Sanchez, J.C.; Karmime, A.; Rouge, V.; Rose, K.; Binz, P.A.; Hochstrasser, D.F. Toward a clinical molecular scanner for proteome research: Parallel protein chemical processing before and during western blot. Anal. Chem. 1999, 71, 4800–4807. [Google Scholar] [CrossRef]
- Naryzhny, S. Inventory of proteoforms as a current challenge of proteomics: Some technical aspects. J. Proteom. 2019. [Google Scholar] [CrossRef] [PubMed]
- Naryzhny, S.N. Towards the Full Realization of 2DE Power. Proteomes 2016, 4, 33. [Google Scholar] [CrossRef] [PubMed]
- Lander, E.S.; Linton, L.M.; Birren, B.; Nusbaum, C.; Zody, M.C.; Baldwin, J.; Devon, K.; Dewar, K.; Doyle, M.; FitzHugh, W.; et al. International Human Genome Sequencing Consortium. Initial sequencing and analysis of the human genome. Nature 2001, 409, 860–921. [Google Scholar] [PubMed]
- Roest Crollius, H.; Jaillon, O.; Bernot, A.; Dasilva, C.; Bouneau, L.; Fischer, C.; Fizames, C.; Wincker, P.; Brottier, P.; Quétier, F.; et al. Estimate of human gene number provided by genome-wide analysis using Tetraodon nigroviridis DNA sequence. Nat. Genet. 2000, 25, 235–238. [Google Scholar] [CrossRef] [PubMed]
- Ewing, B.; Green, P. Analysis of expressed sequence tags indicates 35,000 human genes. Nat. Genet. 2000, 25, 232–234. [Google Scholar] [CrossRef] [PubMed]
- Abascal, F.; Juan, D.; Jungreis, I.; Kellis, M.; Martinez, L.; Rigau, M.; Rodriguez, J.M.; Vazquez, J.; Tress, M.L. Loose ends: Almost one in five human genes still have unresolved coding status. Nucleic Acids Res. 2018, 46, 7070–7084. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Seilhamer, J. A comparison of selected mRNA and protein abundances in human liver. Electrophoresis 1997, 18, 533–537. [Google Scholar] [CrossRef]
- Gygi, S.P.; Rochon, Y.; Franza, B.R.; Aebersold, R. Correlation between protein and mRNA abundance in yeast. Mol. Cell. Biol. 1999, 19, 1720–1730. [Google Scholar] [CrossRef]
- Zhan, X.; Long, Y.; Zhan, X.; Mu, Y. Consideration of statistical vs. biological significances for omics data-based pathway network analysis. Med One 2017, 1, e170002. [Google Scholar]
- Zhan, X.; Long, Y.; Lu, M. Exploration of variations in proteome and metabolome for predictive diagnostics and personalized treatment algorithms: Innovative approach and examples for potential clinical application. J. Proteom. 2018, 188, 30–40. [Google Scholar] [CrossRef]
- Zhan, X.; Giorgianni, F.; Desiderio, D.M. Proteomics analysis of growth hormone isoforms in the human pituitary. Proteomics 2005, 5, 1228–1241. [Google Scholar] [CrossRef] [PubMed]
- Qian, S.; Yang, Y.; Li, N.; Cheng, T.; Wang, X.; Liu, J.; Li, X.; Desiderio, D.M.; Zhan, X. Prolactin variants in human pituitaries and pituitary adenomas identified with two-dimensional gel electrophoresis and mass spectrometry. Front. Endocrinol. 2018, 9, 468. [Google Scholar] [CrossRef] [PubMed]
- Scheler, C.; Müller, E.C.; Stahl, J.; Müller-Werdan, U.; Salnikow, J.; Jungblut, P.R. Identification and characterization of heat shock protein 27 protein species in human myocardial two-dimensional electrophoresis patterns. Electrophoresis 1997, 18, 2823–2831. [Google Scholar] [CrossRef] [PubMed]
- Klose, J.; Nock, C.; Herrmann, M.; Stühler, K.; Marcus, K.; Blüggel, M.; Krause, E.; Schalkwyk, L.C.; Rastan, S.; Brown, S.D.; et al. Genetic analysis of the mouse brain proteome. Nat. Genet. 2002, 30, 385–393. [Google Scholar] [CrossRef]
- Szabo, Z.; Szomor, J.S.; Foeldi, I.; Janaky, T. Mass spectrometry-based label free quantification of gel separated proteins. J. Proteom. 2012, 75, 5544–5553. [Google Scholar] [CrossRef]
- Schaffer, L.V.; Millikin, R.J.; Miller, R.M.; Anderson, L.C.; Fellers, R.T.; Ge, Y.; Kelleher, N.L.; LeDuc, R.D.; Liu, X.; Payne, S.H.; et al. Identification and quantification of proteoforms by mass spectrometry. Proteomics 2019, 19, e1800361. [Google Scholar] [CrossRef]
- Li, H.; Wongkongkathep, P.; Van Orden, S.L.; Ogorzalek Loo, R.R.; Loo, J.A. Revealing ligand binding sites and quantifying subunit variants of noncovalent protein complexes in a single native top-down FTICR MS experiment. J. Am. Soc. Mass Spectrom. 2014, 25, 2060–2068. [Google Scholar] [CrossRef]
- Anderson, L.C.; DeHart, C.J.; Kaiser, N.K.; Fellers, R.T.; Smith, D.F.; Greer, J.B.; LeDuc, R.D.; Blakney, G.T.; Thomas, P.M.; Kelleher, N.L.; et al. Identification and characterization of human proteoforms by top-down LC-21 Tesla FT-ICR mass spectrometry. J. Proteome Res. 2017, 16, 1087–1096. [Google Scholar] [CrossRef]
- Fornelli, L.; Durbin, K.R.; Fellers, R.T.; Early, B.P.; Greer, J.B.; LeDuc, R.D.; Compton, P.D.; Kelleher, N.L. Advancing top-down analysis of the human proteome using a benchtop quadrupole-Orbitrap mass spectrometer. J. Proteome Res. 2017, 16, 609–618. [Google Scholar] [CrossRef]
- Fornelli, L.; Srzentić, K.; Huguet, R.; Mullen, C.; Sharma, S.; Zabrouskov, V.; Fellers, R.T.; Durbin, K.R.; Compton, P.D.; Kelleher, N.L. Accurate sequence analysis of a monoclonal antibody by top-down and middle-down Orbitrap mass spectrometry applying multiple ion activation techniques. Anal. Chem. 2018, 90, 8421–8429. [Google Scholar] [CrossRef]
- McCool, E.N.; Lubeckyj, R.A.; Shen, A.; Chen, D.; Kou, Q.; Liu, X.; Sun, L. Deep top-down proteomics using capillary zone electrophoresis-tandem mass spectrometry: Identification of 5700 proteoforms from the Escherichia coli proteome. Anal. Chem. 2018, 90, 5529–5533. [Google Scholar] [CrossRef] [PubMed]
- McCool, E.N.; Lubeckyj, R.; Shen, X.; Kou, Q.; Liu, X.; Sun, L. Large-scale top-down proteomics using capillary zone electrophoresis tandem mass spectrometry. J. Vis. Exp. 2018, 140, e58644. [Google Scholar] [CrossRef] [PubMed]
- Wiese, S.; Reidegeld, K.A.; Meyer, H.E.; Warscheid, B. Protein labeling by iTRAQ: A new tool for quantitative mass spectrometry in proteome research. Proteomics 2007, 7, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Prudova, A.; auf dem Keller, U.; Butler, G.S.; Overall, C.M. Multiplex N-terminome analysis of MMP-2 and MMP-9 substrate degradomes by iTRAQ-TAILS quantitative proteomics. Mol. Cell. Proteom. 2010, 9, 894–911. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, J.; Timms, J.F. Quantitative profiling of serum samples using TMT protein labelling, fractionation and LC-MS/MS. Methods 2011, 54, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.W.; Tholey, A. Tandem mass tag protein labeling for top-down identification and quantification. Anal. Chem. 2012, 84, 161–170. [Google Scholar] [CrossRef]
- Gygi, S.P.; Rist, B.; Gerber, S.A.; Turecek, F.; Gelb, M.H.; Aebersold, R. Quantitative analysis of complex protein mixtures using isotope-coded affinity tags. Nat. Biotechnol. 1999, 17, 994–999. [Google Scholar] [CrossRef]
- Blein-Nicolas, M.; Zivy, M. Thousand and one ways to quantify and compare protein abundances in label-free bottom-up proteomics. Biochim. Biophys. Acta Proteins Proteom. 2015, 1864, 883–895. [Google Scholar] [CrossRef]
- Lange, V.; Picotti, P.; Domon, B.; Aebersold, R. Selected reaction monitoring for quantitative proteomics: A tutorial. Mol. Syst. Biol. 2008, 4, 222. [Google Scholar] [CrossRef]
- Aebersold, R.; Bensimon, A.; Collins, B.C.; Ludwig, C.; Sabido, E. Applications and developments in targeted proteomics: From SRM to DIA/SWATH. Proteomics 2016, 16, 2065–2067. [Google Scholar] [CrossRef]
- Autelitano, F.; Loyaux, D.; Roudières, S.; Déon, C.; Guette, F.; Fabre, P.; Ping, Q.; Wang, S.; Auvergne, R.; Badarinarayana, V.; et al. Identification of novel tumor-associated cell surface sialoglycoproteins in human glioblastoma tumors using quantitative proteomics. PLoS ONE 2014, 9, e110316. [Google Scholar] [CrossRef] [PubMed]
- Schelletter, L.; Albaum, S.; Walter, S.; Noll, T.; Hoffrogge, R. Clonal variations in CHO IGF signaling investigated by SILAC-based phosphoproteomics and LFQ-MS. Appl. Microbiol. Biotechnol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.M.; Kelleher, N.L. Proteoforms as the next proteomics currency. Science 2018, 359, 1106–1107. [Google Scholar] [CrossRef] [PubMed]
- Kusch, K.; Uecker, M.; Liepold, T.; Möbius, W.; Hoffmann, C.; Neumann, H.; Werner, H.B.; Jahn, O. Partial immunoblotting of 2D-Gels: A novel method to identify post-translationally modified proteins exemplified for the myelin acetylome. Proteomes 2017, 5, 3. [Google Scholar] [CrossRef]


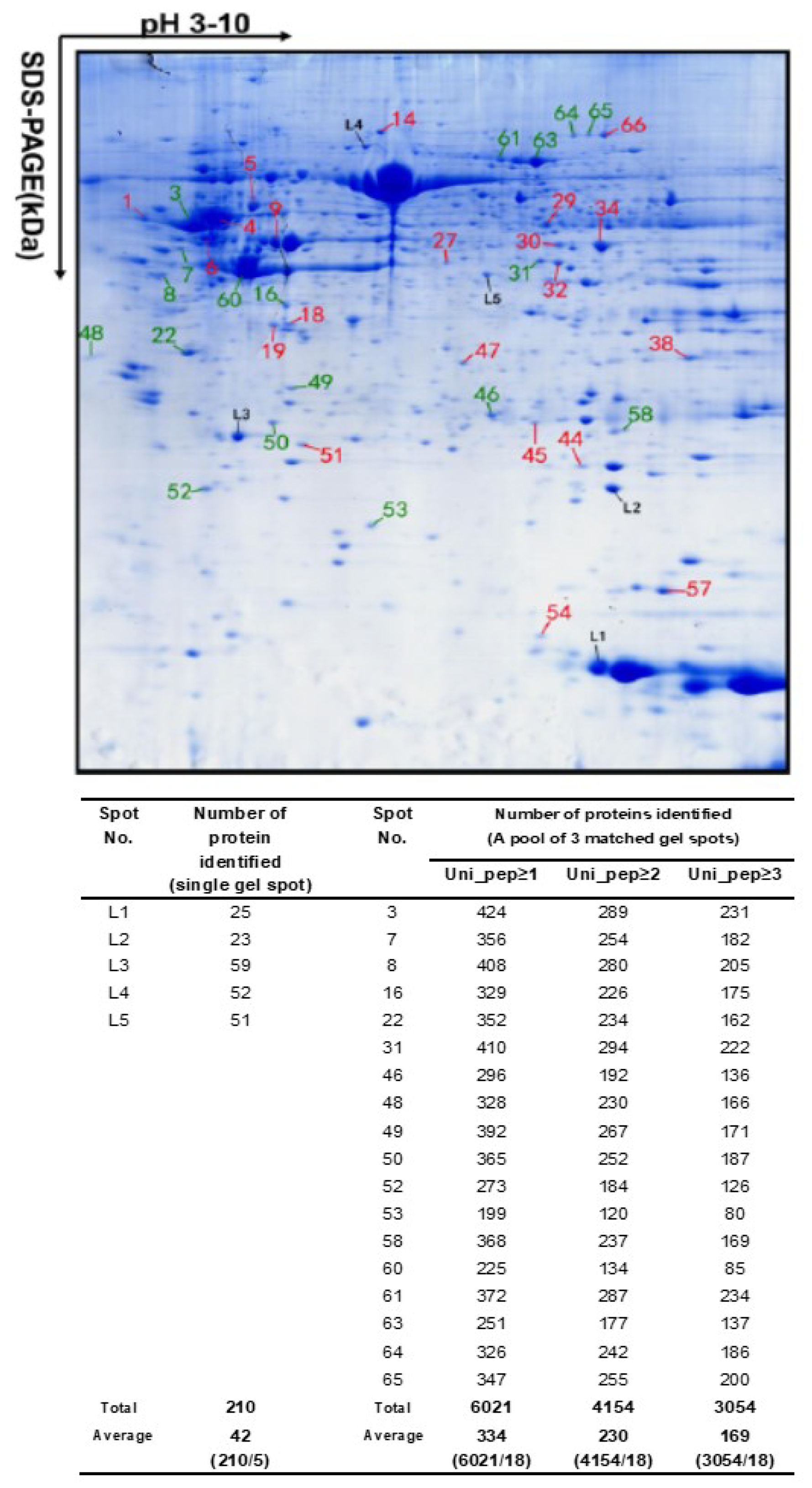
| Spot No. | Total (n) | The Number of Proteins by emPAI Range | ||||
|---|---|---|---|---|---|---|
| >100 (n) | 100–10 (n) | 10–1 (n) | 1–0.1 (n) | 0.1–0.01 (n) | ||
| 3 | 289 | 7 | 3 | 28 | 105 | 146 |
| 7 | 254 | 2 | 6 | 19 | 105 | 122 |
| 8 | 280 | 2 | 2 | 22 | 130 | 124 |
| 16 | 226 | - | 8 | 32 | 89 | 97 |
| 22 | 234 | 1 | 3 | 27 | 122 | 81 |
| 31 | 294 | - | 4 | 36 | 125 | 129 |
| 46 | 192 | 1 | 4 | 19 | 106 | 62 |
| 48 | 230 | - | 1 | 29 | 132 | 68 |
| 49 | 267 | 3 | 2 | 29 | 139 | 94 |
| 50 | 252 | - | 1 | 40 | 126 | 85 |
| 52 | 184 | 1 | - | 33 | 99 | 51 |
| 53 | 120 | - | - | 12 | 61 | 47 |
| 58 | 237 | 2 | 4 | 31 | 129 | 71 |
| 60 | 134 | 2 | 4 | 6 | 60 | 62 |
| 61 | 287 | 1 | 6 | 28 | 116 | 136 |
| 63 | 177 | 1 | 2 | 24 | 53 | 97 |
| 64 | 242 | - | 1 | 18 | 93 | 130 |
| 65 | 255 | - | 1 | 19 | 85 | 150 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhan, X.; Li, B.; Zhan, X.; Schlüter, H.; Jungblut, P.R.; Coorssen, J.R. Innovating the Concept and Practice of Two-Dimensional Gel Electrophoresis in the Analysis of Proteomes at the Proteoform Level. Proteomes 2019, 7, 36. https://doi.org/10.3390/proteomes7040036
Zhan X, Li B, Zhan X, Schlüter H, Jungblut PR, Coorssen JR. Innovating the Concept and Practice of Two-Dimensional Gel Electrophoresis in the Analysis of Proteomes at the Proteoform Level. Proteomes. 2019; 7(4):36. https://doi.org/10.3390/proteomes7040036
Chicago/Turabian StyleZhan, Xianquan, Biao Li, Xiaohan Zhan, Hartmut Schlüter, Peter R. Jungblut, and Jens R. Coorssen. 2019. "Innovating the Concept and Practice of Two-Dimensional Gel Electrophoresis in the Analysis of Proteomes at the Proteoform Level" Proteomes 7, no. 4: 36. https://doi.org/10.3390/proteomes7040036
APA StyleZhan, X., Li, B., Zhan, X., Schlüter, H., Jungblut, P. R., & Coorssen, J. R. (2019). Innovating the Concept and Practice of Two-Dimensional Gel Electrophoresis in the Analysis of Proteomes at the Proteoform Level. Proteomes, 7(4), 36. https://doi.org/10.3390/proteomes7040036








