Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology
Abstract
1. Introduction
1.1. Advances in Plant Virus Diagnostics
1.2. Loop Mediated Isothermal Amplification (LAMP)
2. Principles of LAMP
2.1. General Considerations
2.1.1. Initial Step
2.1.2. Cycling Amplification and Elongation Step
2.1.3. LAMP Acceleration
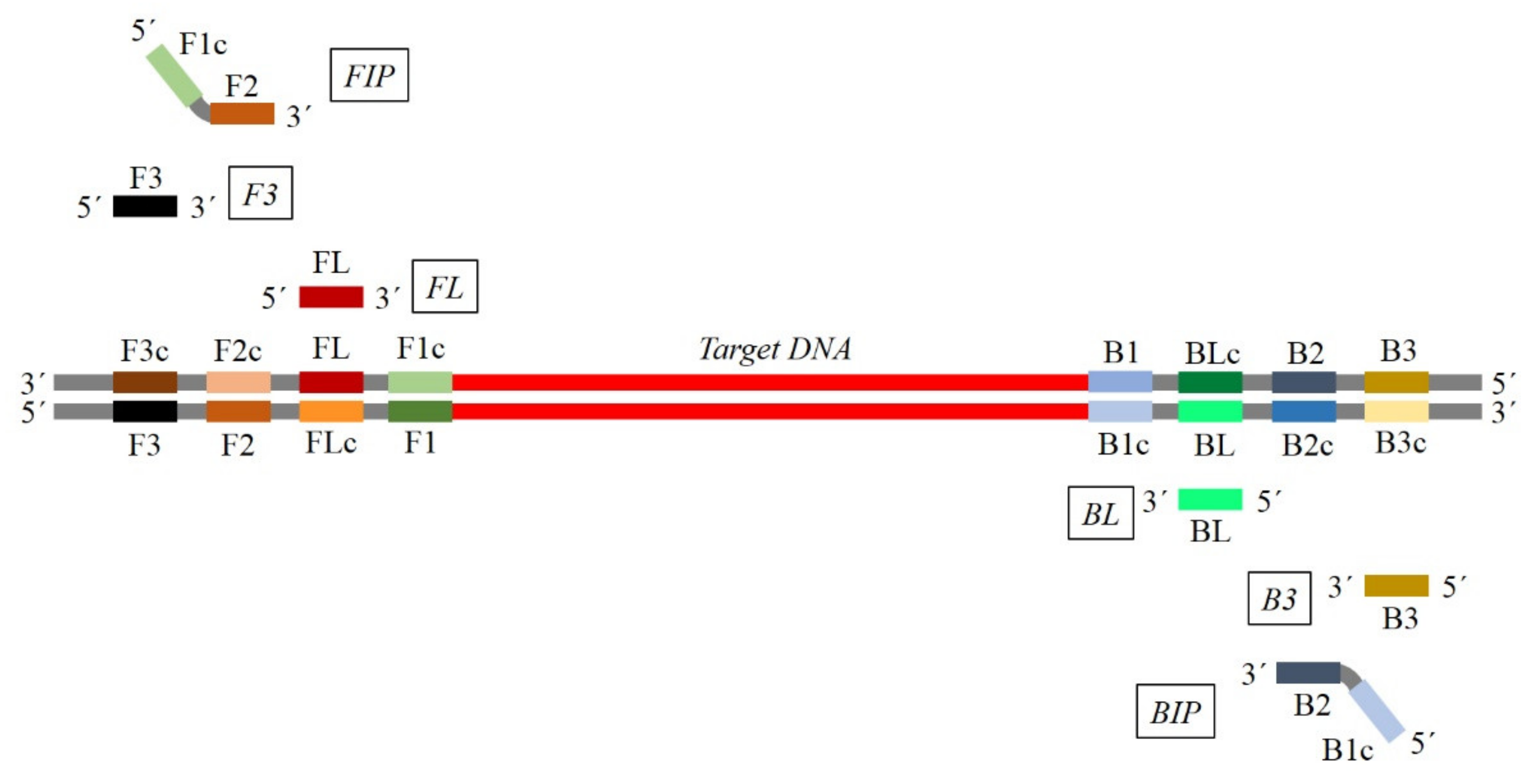
3. General and Specific Considerations for Primers Design
Key Factors for Primer Design
4. LAMP Primer Design Software
5. Sample Preparation Methods for Viruses and Viroids Detection
6. Reverse Transcription (RT)-LAMP
7. Visualization Methods of Amplification Products
7.1. End Point-Based Methods
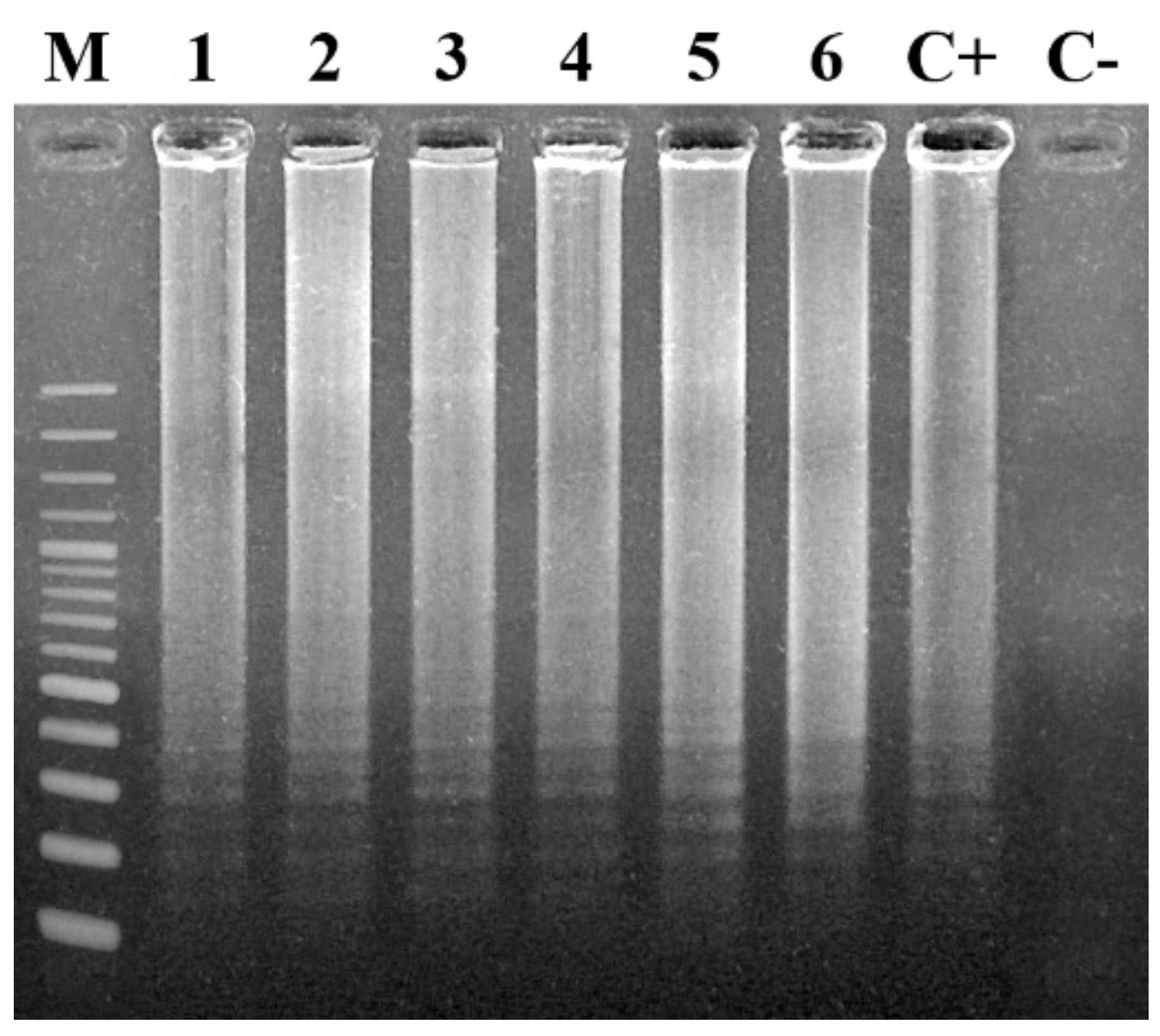
7.1.1. Agarose Gel Electrophoresis
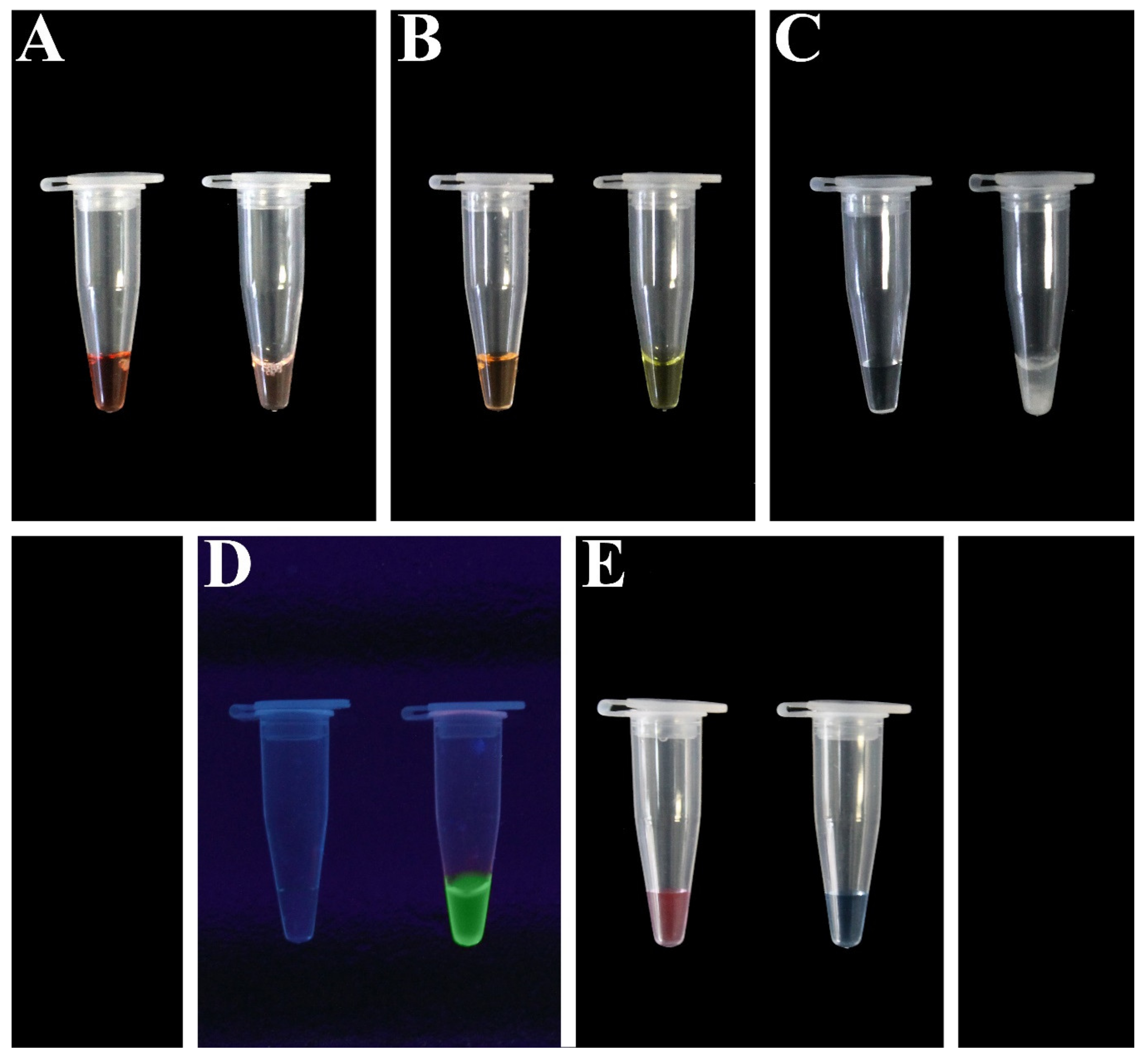
7.1.2. Fluorescent Dyes
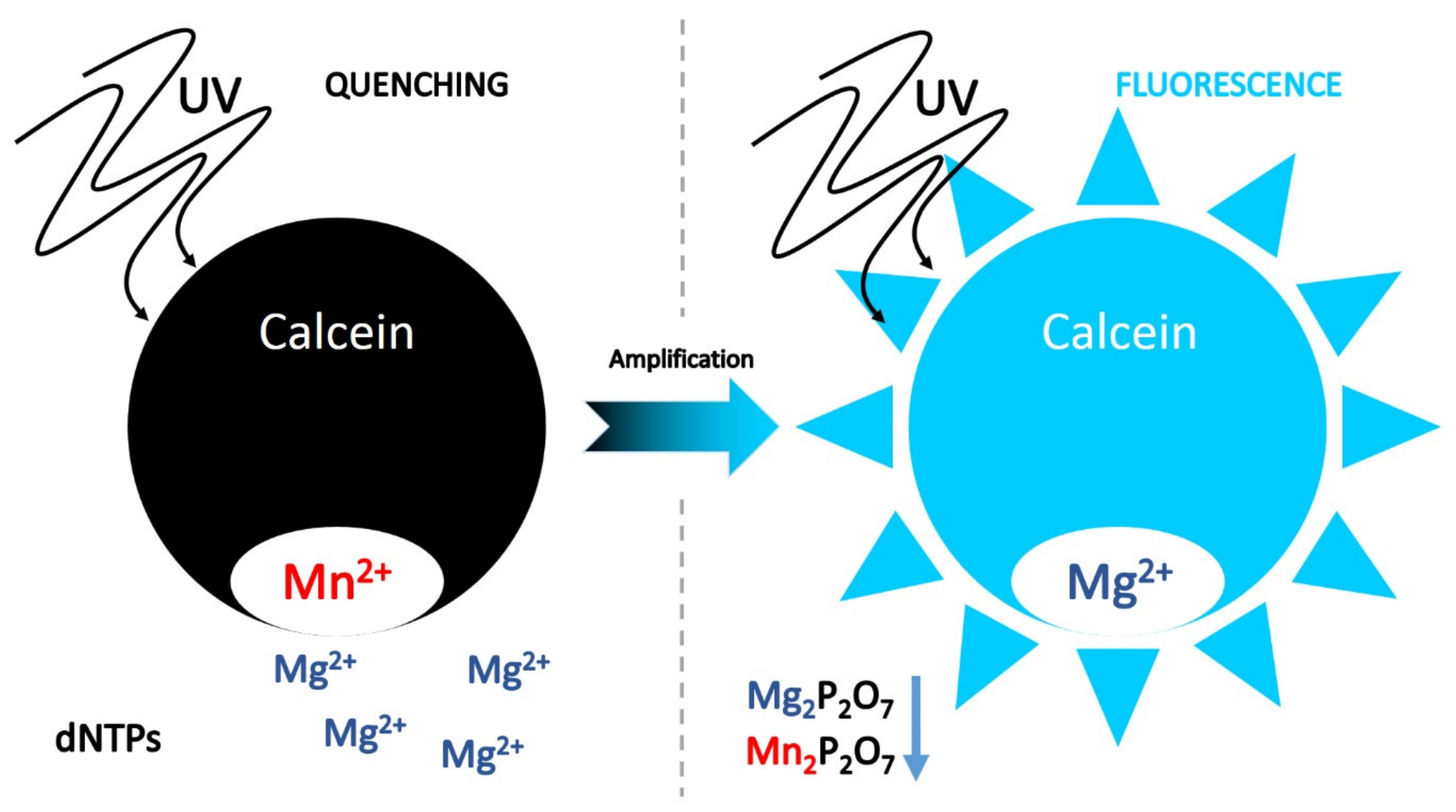
7.1.3. Ion Indicators
7.2. Real-Time-Based Methods
8. LAMP Application in Plant Virology
9. LAMP: Evolution and Recent Applications
9.1. Multiplex LAMP (mLAMP)
9.2. Micro LAMP (μLAMP)
9.3. Digital LAMP (dLAMP)
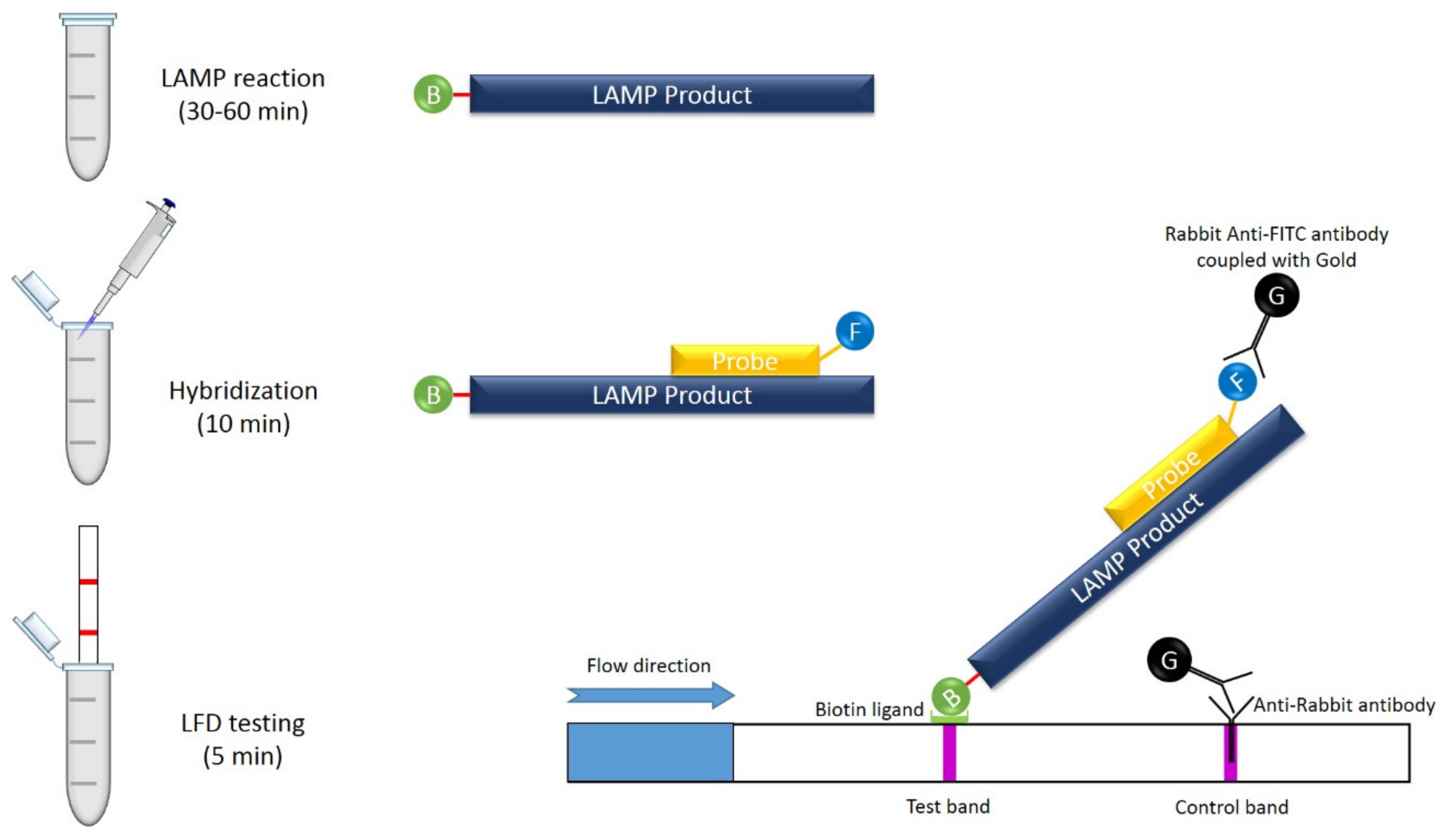
9.4. LAMP Combined with Lateral Flow Assay (LFA)
9.5. Immunocapture-RT-LAMP (IC-RT-LAMP)
9.6. Electric LAMP (eLAMP)
9.7. Lab-on-a-Disc (LoaD) LAMP Platform
9.8. Lyophilized LAMP
9.9. FRET-Based Assimilating Probe for Sequence-Specific Real-Time Monitoring of LAMP
10. Advantages and Drawbacks of LAMP Assay
- -
- Compared with other RNA/DNA amplification methods (i.e., PCR, RT-PCR, and RT-qPCR), LAMP shows a similar sensitivity and specificity [187].
- -
- A typical LAMP test is rapid, and it is completed in about one hour; if loop primers are used, it requires no more than 30 min [24].
- -
- LAMP works at a constant temperature, thanks to the high strand displacement activity of Bst polymerase [15].
- -
- -
- When combined with reverse transcription, amplification of RNA sequences can be carried out in a LAMP assay with high efficiency.
- -
- The robustness of the LAMP allows to also analyze unprocessed samples, which can be used as a template, as the activity of the Bst polymerase is not influenced by the presence of inhibiting substances. For example, in plant virus diagnosis, it could be possible to use direct crude plant extracts in order to avoid total RNA or DNA extraction, shortening the processing time, allowing the simultaneous analysis of multiple samples, and drastically reducing the total cost for single analysis [12].
- -
- The result visualization can be performed with naked-eye or real-time methods, through the use of SYBR Green I, EtBr, HNB, or Calcein [171]. There is no need for post-amplification processing, reducing the time and the high risk of contamination.
- -
- The detection limit of LAMP assay is comparable to an end point PCR, while this assay is higher compared with other isothermal amplification techniques such as NASBA, 3SR, and SDA, all of which having a limit of detection of less than 10 copies [15].
Author Contributions
Funding
Conflicts of Interest
References
- Clark, M.F.; Adams, A.N. Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol. 1977, 34, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Boonham, N.; Kreuze, J.; Winter, S.; Van der Vlugt, R.; Bergervoet, J.; Tomlinson, J.; Mumford, R. Methods in virus diagnostics: From ELISA to next generation sequencing. Virus Res. 2014, 186, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Mullis, K.; Faloona, F.; Scharf, S.; Saiki, R.K.; Horn, G.T.; Erlich, H. Specific enzymatic amplification of DNA in vitro: The polymerase chain reaction. Cold Spring Harb. Symp. Quant. Biol. 1986, 51, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Davino, S.; Calari, A.; Davino, M.; Tessitori, M.; Bertaccini, A.; Bellardi, M.G. Virescence of tenweeks stock associated to phytoplasma infection in Sicily. Bull. Insectol. 2007, 60, 279–280. [Google Scholar]
- Davino, S.; Panno, S.; Rangel, E.A.; Davino, M.; Bellardi, M.G.; Rubio, L. Population genetics of Cucumber mosaic virus infecting medicinal, aromatic and ornamental plants from northen Italy. Arch. Virol. 2012, 157, 739–745. [Google Scholar] [CrossRef]
- Panno, S.; Davino, S.; Rubio, L.; Rangel, E.; Davino, M.; García-Hernández, J.; Olmos, A. Simultaneous detection of the seven main tomato-infecting RNA viruses by two multiplex reverse transcription polymerase chain reactions. J. Virol. Methods 2012, 186, 152–156. [Google Scholar] [CrossRef][Green Version]
- Panno, S.; Ferriol, I.; Rangel, E.A.; Olmos, A.; Han, C.G.; Martinelli, F.; Rubio, L.; Davino, S. Detection and identification of Fabavirus species by one-step RT-PCR and multiplex RT-PCR. J. Virol. Methods 2014, 197, 77–82. [Google Scholar] [CrossRef]
- Vunsh, R.O.N.; Rosner, A.; Stein, A. The use of the polymerase chain reaction (PCR) for the detection of bean yellow mosaic virus in gladiolus. Ann. Appl. Biol. 1990, 117, 561–569. [Google Scholar] [CrossRef]
- PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 3 February 2020).
- Wetmur, J.G. DNA probes: Applications of the principles of nucleic acid hybridization. Crit. Rev. Biochem. Mol. Biol. 1991, 26, 227–259. [Google Scholar] [CrossRef]
- Mumford, R.A.; Walsh, K.; Barker, I.; Boonham, N. Detection of Potato mop top virus and Tobacco rattle virus using a multiplex real-time fluorescent reverse-transcription polymerase chain reaction assay. Phytopathology 2000, 90, 448–453. [Google Scholar] [CrossRef]
- Panno, S.; Ruiz-Ruiz, S.; Caruso, A.G.; Alfaro-Fernandez, A.; San Ambrosio, M.I.F.; Davino, S. Real-time reverse transcription polymerase chain reaction development for rapid detection of Tomato brown rugose fruit virus and comparison with other techniques. PeerJ 2019, 7, e7928. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, J.; Pingoud, A.; Hahn, M. Real-time PCR-based method for the estimation of genome sizes. Nucleic Acids Res. 2003, 31, e56. [Google Scholar] [CrossRef] [PubMed]
- Ponchel, F.; Toomes, C.; Bransfield, K.; Leong, F.T.; Douglas, S.H.; Field, S.L.; Robinson, P.A. Real-time PCR based on SYBR-Green I fluorescence: An alternative to the TaqMan assay for a relative quantification of gene rearrangements, gene amplifications and micro gene deletions. BMC Biotechnol. 2003, 3, 18. [Google Scholar] [CrossRef] [PubMed]
- Notomi, T.; Okayama, H.; Masubuchi, H.; Yonekawa, T.; Watanabe, K.; Amino, N.; Hase, T. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res. 2000, 28, e63. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Teng, D.; Guan, Q.; Tian, F.; Wang, J. Detection of Roundup Ready soybean by loop-mediated isothermal amplification combined with a lateral-flow dipstick. Food Control 2013, 29, 213–220. [Google Scholar] [CrossRef]
- Li, J.J.; Xiong, C.; Liu, Y.; Liang, J.S.; Zhou, X.W. Loop-Mediated Isothermal Amplification (LAMP): Emergence as an Alternative Technology for Herbal Medicine Identification. Front. Plant Sci. 2016, 7, 1956. [Google Scholar] [CrossRef]
- Mayboroda, O.; Katakis, I.; O’Sullivan, C.K. Multiplexed isothermal nucleic acid amplification. Anal Biochem. 2018, 545, 20–30. [Google Scholar] [CrossRef]
- Srividya, A.; Maiti, B.; Chakraborty, A.; Chakraborty, G. Loop Mediated Isothermal Amplification: A Promising Tool for Screening Genetic Mutations. Mol. Diagn. 2019, 23, 723–733. [Google Scholar] [CrossRef]
- Ye, J.; Xu, M.; Tian, X.; Cai, S.; Zeng, S. Research advances in the detection of miRNA. J. Pharm. Anal. 2019, 90, 217–226. [Google Scholar] [CrossRef]
- Martinelli, F.; Scalenghe, R.; Davino, S.; Panno, S.; Scuderi, G.; Ruisi, P.; Villa, P.; Stroppiana, D.; Boschetti, M.; Goulart, L.R.; et al. Advanced methods of plant disease detection. A review. Agron. Sustain. Dev. 2014, 35, 1–25. [Google Scholar] [CrossRef]
- Mori, Y.; Notomi, T. Loop-mediated isothermal amplification (LAMP): A rapid, accurate, and cost-effective diagnostic method for infectious diseases. J. Infect. Chemother. 2009, 15, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Tomita, N.; Mori, Y.; Kanda, H.; Notomi, T. Loop-mediated isothermal amplification (LAMP) of gene sequences and simple visual detection of products. Nat. Protoc. 2008, 3, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Nagamine, K.; Hase, T.; Notomi, T. Accelerated reaction by loop-mediated isothermal amplification using loop primers. Mol. Cell. Probes 2002, 16, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Chander, Y.; Koelbl, J.; Puckett, J.; Moser, M.J.; Klingele, A.J.; Liles, M.R.; Carrias, A.; Mead, D.A.; Schoenfeld, T.W. A novel thermostable polymerase for RNA and DNA loop-mediated isothermal amplification (LAMP). Front. Microbiol. 2014, 5, 395. [Google Scholar] [CrossRef] [PubMed]
- Hadersdorfer, J.; Neumüller, M.; Treutter, D.; Fischer, T.C. Fast and reliable detection of Plum pox virus in woody host plants using the Blue LAMP protocol. Ann. Appl. Biol. 2011, 159, 456–466. [Google Scholar] [CrossRef]
- Walsh, H.A.; Pietersen, G. Rapid detection of Grapevine leafroll-associated virus type 3 using a reverse transcription loop-mediated amplification method. J. Virol. Methods 2013, 194, 308–316. [Google Scholar] [CrossRef]
- Zhang, J.; Borth, W.; Lin, B.; Melzer, M.; Shen, H.; Pu, X.; Sun, D.; Nelson, S.; Hu, J. Multiplex detection of three banana viruses by reverse transcription loop-mediated isothermal amplification (RT-LAMP). Trop. Plant Pathol. 2018, 43, 543–551. [Google Scholar] [CrossRef]
- Mori, Y.; Hirano, T.; Notomi, T. Sequence specific visual detection of LAMP reactions by addition of cationic polymers. BMC Biotechnol. 2006, 6, 3. [Google Scholar] [CrossRef]
- Varga, A.; James, D. Use of reverse transcription loop-mediated isothermal amplification for the detection of Plum pox virus. J. Virol. Methods 2006, 138, 184–190. [Google Scholar] [CrossRef]
- Almasi, M.A. Establishment and application of a reverse transcription loop-mediated isothermal amplification assay for detection of Grapevine fanleaf virus. Mol. Biol. 2016, 4, 55935592. [Google Scholar] [CrossRef]
- Budziszewska, M.; Wieczorek, P.; Obrępalska-Stęplowska, A. One-step reverse transcription loop-mediated isothermal amplification (RT-LAMP) for detection of tomato torrado virus. Arch. Virol. 2016, 161, 1359–1364. [Google Scholar] [CrossRef] [PubMed]
- Francois, P.; Tangomo, M.; Hibbs, J.; Bonetti, E.J.; Boehme, C.C.; Notomi, T.; Perkins, M.D.; Schrenzel, J. Robustness of a loop-mediated isothermal amplification reaction for diagnostic applications. FEMS Immunol. Med. Microbiol. 2011, 62, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Si Ammour, M.; Bilodeau, G.J.; Tremblay, D.M.; Van der Heyden, H.; Yaseen, T.; Varvaro, L.; Carisse, O. Development of Real-Time Isothermal Amplification Assays for On-Site Detection of Phytophthora infestans in Potato Leaves. Plant Dis. 2017, 101, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Elvira-González, L.; Puchades, A.V.; Carpino, C.; Alfaro-Fernandez, A.; Font-San-Ambrosio, M.I.; Rubio, L.; Galipienso, L. Fast detection of Southern tomato virus by one-step transcription loop-mediated isothermal amplification (RT-LAMP). J. Virol. Methods. 2017, 241, 11–14. [Google Scholar] [CrossRef]
- Kogovšek, P.; Mehle, N.; Pugelj, A.; Jakomin, T.; Schroers, H.J.; Ravnikar, M.; Dermastia, M. Rapid loop-mediated isothermal amplification assays for grapevine yellows phytoplasmas on crude leaf-vein homogenate has the same performance as qPCR. Eur. J. Plant Pathol. 2017, 148, 75–84. [Google Scholar] [CrossRef]
- Romero Romero, J.L.; Carver, G.D.; Arce Johnson, P.; Perry, K.L.; Thompson, J.R. A rapid, sensitive and inexpensive method for detection of grapevine red blotch virus without tissue extraction using loop-mediated isothermal amplification. Arch. Virol. 2019, 164, 1453–1457. [Google Scholar] [CrossRef]
- Selvaraj, V.; Maheshwari, Y.; Hajeri, S.; Yokomi, R. A rapid detection tool for VT isolates of Citrus tristeza virus by immunocapture-reverse transcriptase loop-mediated isothermal amplification assay. PLoS ONE 2019, 14, e0222170. [Google Scholar] [CrossRef]
- Wilisiani, F.; Tomiyama, A.; Katoh, H.; Hartono, S.; Neriya, Y.; Nishigawa, H.; Natsuaki, T. Development of a LAMP assay with a portable device for real-time detection of begomoviruses under field conditions. J. Virol. Methods 2019, 265, 71–76. [Google Scholar] [CrossRef]
- Kaneko, H.; Kawana, T.; Fukushima, E.; Suzutani, T. Tolerance of loop-mediated isothermal amplification to a culture medium and biological substances. J. Biochem. Biophys. Methods 2007, 70, 499–501. [Google Scholar] [CrossRef]
- Abd-Elsalam, K.A. Bioinformatic tools and guidelines for PCR primer design. Afr. J. Biotechnol. 2003, 2, 91–95. [Google Scholar]
- PrimerExplorer. Available online: https://primerexplorer.jp/e/ (accessed on 3 February 2020).
- Torres, C.; Vitalis, E.A.; Baker, B.R.; Gardner, S.N.; Torres, M.W.; Dzenitis, J.M. LAVA: An Open-Source approach to designing LAMP (Loop-Mediated Isothermal Amplification) DNA signatures. BMC Bioinform. 2011, 12, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.L.; Yen, C.H.; Tu, C.F. Immunocapture loop-mediated isothermal amplification assays for the detection of canine parvovirus. J. Virol. Methods 2017, 249, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Bertolini, E.; Moreno, A.; Capote, N.; Olmos, A.; De Luis, A.; Vidal, E.; Pérez-Panadés, J.; Cambra, M. Quantitative detection of Citrus tristeza virus in plant tissues and single aphids by real-time RT-PCR. Eur. J. Plant Pathol. 2008, 120, 177–188. [Google Scholar] [CrossRef]
- Osman, F.; Rowhani, A. Application of a spotting sample preparation technique for the detection of pathogens in woody plants by RT-PCR and real-time PCR (TaqMan). J. Virol. Methods 2006, 133, 130–136. [Google Scholar] [CrossRef]
- Fu, S.; Qu, G.; Guo, S.; Ma, L.; Zhang, N.; Zhang, S.; Zhang, S.; Gao, S.; Shen, Z. Applications of loop-mediated isothermal DNA amplification. Appl. Biochem. Biotechnol. 2011, 163, 845–850. [Google Scholar] [CrossRef]
- Nie, X. Reverse transcription loop-mediated isothermal amplification of DNA for detection of Potato virus Y. Plant Dis. 2005, 89, 605–610. [Google Scholar] [CrossRef]
- Galvez, L.C.; Barbosa, C.F.C.; Koh, R.B.L.; Aquino, V.M. Loop-mediated isothermal amplification (LAMP) assays for the detection of abaca bunchy top virus and banana bunchy top virus in abaca. Crop Prot. 2020, 131, 105101. [Google Scholar] [CrossRef]
- Peng, D.; Xie, J.; Qiang, W.; Ling, K.S.; Guo, L.; Fan, Z.; Zhou, T. One-step reverse transcription loop-mediated isothermal amplification assay for detection of Apple chlorotic leaf spot virus. J. Virol. Methods 2017, 248, 154–158. [Google Scholar] [CrossRef]
- Lu, Y.; Yao, B.; Wang, G.; Hong, N. The detection of ACLSV and ASPV in pear plants by RT-LAMP assays. J. Virol. Methods 2018, 252, 80–85. [Google Scholar] [CrossRef]
- Lee, S.H.; Ahn, G.; Kim, M.S.; Jeong, O.C.; Lee, J.H.; Kwon, H.G.; Kim, Y.H.; Ahn, J.Y. Poly-adenine-Coupled LAMP Barcoding to Detect Apple Scar Skin Viroid. ACS Comb. Sci. 2018, 20, 472–481. [Google Scholar] [CrossRef]
- Lixin, G. Detection of Arabis mosaic virus by the reverse transcription loop-mediated isothermal amplification method. Plant Prot. 2015, 39, 91–95. [Google Scholar]
- Zhang, J.; Borth, W.B.; Lin, B.; Dey, K.K.; Melzer, M.J.; Shen, H.; Pu, X.; Sun, D.; Hu, J.S. Deep sequencing of banana bract mosaic virus from flowering ginger (Alpinia purpurata) and development of an immunocapture RT-LAMP detection assay. Arch. Virol. 2016, 161, 1783–1795. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zhang, J.; Xia, Z.; Li, Y.; Huang, J.; Fan, Z. Rapid and sensitive detection of Banana bunchy top virus by loop-mediated isothermal amplification. J. Virol. Methods 2012, 185, 254–258. [Google Scholar] [CrossRef]
- Peng, J.; Fan, Z.; Huang, J. Rapid detection of banana streak virus by loop-mediated isothermal amplification assay in South China. J. Phytopathol. 2012, 160, 248–250. [Google Scholar] [CrossRef]
- Zarzyńska-Nowak, A.; Hasiów-Jaroszewska, B.; Jeżewska, M. Molecular analysis of barley stripe mosaic virus isolates differing in their biological properties and the development of reverse transcription loop-mediated isothermal amplification assays for their detection. Arch. Virol. 2018, 163, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Liu, Y.; Wang, X.F. Reverse transcription loop-mediated isothermal amplification of DNA for detection of Barley yellow dwarf viruses in China. J. Virol. Methods 2010, 169, 211–221. [Google Scholar] [CrossRef]
- Lee, S.; Kim, H.; Lee, J.Y.; Rho, J.Y. Development of rapid and highly sensitive detection of Bean common mosaic necrosis virus in leguminous crops using loop-mediated isothermal amplification assay. J. Virol. Methods 2017, 249, 117–120. [Google Scholar] [CrossRef]
- Wei, Q.W.; Yu, C.; Zhang, S.Y.; Yang, C.Y.; Miriam, K.; Zhang, W.N.; Dou, D.L.; Tao, X.R. One-step detection of Bean pod mottle virus in soybean seeds by the reverse-transcription loop-mediated isothermal amplification. Virol. J. 2012, 9, 187. [Google Scholar] [CrossRef]
- Kil, E.; Cho, S.; Byun, H.; Kim, J.; Hwang, H.; Auh, C.; Heo, N.; Shin, Y.; Lee, S. Differential identification of three species of Curtovirus using loop-mediated isothermal amplification. Acta Virol. 2014, 58, 160–166. [Google Scholar] [CrossRef]
- Almasi, M.A.; Almasi, G. Development and evaluation of a reverse transcription loop-mediated isothermal amplification assay for detection of beet necrotic yellow vein virus. Arch. Virol. 2017, 162, 495–500. [Google Scholar] [CrossRef]
- Tomlinson, J.A.; Ostoja-Starzewska, S.; Adams, I.P.; Miano, D.W.; Abidrabo, P.; Kinyua, Z.; Alicai, T.; Dickinson, M.J.; Peters, D.; Boonham, N.; et al. Loop-mediated isothermal amplification for rapid detection of the causal agents of cassava brown streak disease. J. Virol. Methods 2013, 191, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Roy, S.; Sharma, S.K.; Dutta, S.K.; Chandra, S.; Ngachan, S.V. Reverse transcription loop-mediated isothermal amplification (RT-LAMP) assay for rapid diagnosis of chilli veinal mottle virus. Arch. Virol. 2016, 161, 1957–1961. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, S.; Tamura, M.; Maejima, H.; Takahashi, R.; Kuwayama, S.; Tsuji, T.; Yoshida, T.; Itoh, K.; Hashizume, H.; Nakajima, Y.; et al. Differential detection of Wheat yellow mosaic virus, Japanese soil-borne wheat mosaic virus and Chinese wheat mosaic virus by reverse transcription loop-mediated isothermal amplification reaction. J. Virol. Methods 2013, 189, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Jung, Y.; Kil, E.J.; Kim, J.; Thi Tran, D.; Choi, S.K.; Yoon, J.Y.; Cho, W.K.; Lee, S. Loop-mediated isothermal amplification for the rapid detection of Chrysanthemum chlorotic mottle viroid (CChMVd). J. Virol. Methods 2013, 193, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Fukuta, S.; Matsumoto, Y.; Hasegawa, T.; Kojima, H.; Hotta, M.; Miyake, N. Development of reverse transcription loop-mediated isothermal amplification assay as a simple detection method of Chrysanthemum stem necrosis virus in chrysanthemum and tomato. J. Virol. Methods 2016, 236, 29–34. [Google Scholar] [CrossRef]
- Liu, X.L.; Zhao, X.T.; Muhammad, I.; Ge, B.B.; Hong, B. Multiplex reverse transcription loop-mediated isothermal amplification for the simultaneous detection of CVB and CSVd in chrysanthemum. J. Virol. Methods 2014, 210, 26–31. [Google Scholar] [CrossRef]
- Liu, H.; Wu, W.; Tan, J.; Li, Y.; Mi, W.; Jiang, L.; Wu, Y. Development and evaluation of a one-step reverse transcription loop-mediated isothermal amplification for detection of Citrus leaf blotch virus. J. Virol. Methods 2019, 270, 150–152. [Google Scholar] [CrossRef]
- Ghosh, D.K.; Warghane, A.; Biswas, K.K. Rapid and sensitive detection of Citrus tristeza virus using reverse transcription Loop-mediated Isothermal Amplification (RT-LAMP) assay. Methods Mol. Biol. 2019, 2015, 143–150. [Google Scholar]
- Warghane, A.; Misra, P.; Bhose, S.; Biswas, K.K.; Sharma, A.K.; Reddy, M.K.; Ghosh, D.K. Development of a simple and rapid reverse transcription-loop mediated isothermal amplification (RT-LAMP) assay for sensitive detection of Citrus tristeza virus. J. Virol. Methods 2017, 250, 6–10. [Google Scholar] [CrossRef]
- Anthony Johnson, A.M.; Dasgupta, I.; Sai Gopal, D.V. Development of loop-mediated isothermal amplification and SYBR green real-time PCR methods for the detection of Citrus yellow mosaic badnavirus in citrus species. J. Virol. Methods 2014, 203, 9–14. [Google Scholar] [CrossRef]
- Thanarajoo, S.S.; Kong, L.L.; Kadir, J.; Lau, W.H.; Vadamalai, G. Detection of Coconut cadang-cadang viroid (CCCVd) in oil palm by reverse transcription loop-mediated isothermal amplification (RT-LAMP). J. Virol. Methods 2014, 202, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Bhuvitarkorn, S.; Klinkong, S.; Reanwarakorn, K. Enhancing Columnea latent viroid detection using reverse transcription loop-mediated isothermal amplification (RT-LAMP). Int. J. Agric. Technol. 2019, 15, 215–228. [Google Scholar]
- Li, J.Y.; Wei, Q.W.; Liu, Y.; Tan, X.Q.; Zhang, W.N.; Wu, J.Y.; Charimbu, M.K.; Hu, B.S.; Cheng, Z.B.; Yu, C.; et al. One-step reverse transcription loop-mediated isothermal amplification for the rapid detection of cucumber green mottle mosaic virus. J. Virol. Methods 2013, 193, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Bhat, A.I.; Siljo, A.; Deeshma, K.P. Rapid detection of Piper yellow mottle virus and Cucumber mosaic virus infecting black pepper (Piper nigrum) by loop mediated isothermal amplification (LAMP). J. Virol. Methods 2013, 193, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Shi, M.; Xia, Z.; Huang, J.; Fan, Z. Detection of cucumber mosaic virus isolates from banana by one-step reverse transcription loop-mediated isothermal amplification. Arch. Virol. 2012, 157, 2213–2217. [Google Scholar] [CrossRef]
- Fan, X.; Du, Y.; Cai, Y.; Zhang, Y.; Zhao, X.; Liang, J.; Xu, Y. Rapid and sensitive detection of cucumber mosaic virus by reverse transcription loop-mediated isothermal amplification. Acta Biochim. Biophys. Sin. 2019, 51, 223–226. [Google Scholar] [CrossRef]
- Wang, Z.; Gu, Q.; Sun, H.; Li, H.; Sun, B.; Liang, X.; Yuan, Y.; Liu, R.; Shi, Y. One-step reverse transcription loop mediated isothermal amplification assay for sensitive and rapid detection of Cucurbit chlorotic yellows virus. J. Virol. Methods 2014, 195, 63–66. [Google Scholar] [CrossRef]
- Waliullah, S.; Ling, K.-S.; Cieniewicz, E.J.; Oliver, J.E.; Ji, P.; Ali, M.E. Development of Loop-Mediated Isothermal Amplification Assay for Rapid Detection of Cucurbit Leaf Crumple Virus. Int. J. Mol. Sci. 2020, 21, 1756. [Google Scholar] [CrossRef]
- Lee, M.S.; Yang, M.J.; Hseu, Y.C.; Lai, G.H.; Chang, W.T.; Hsu, Y.H.; Lin, M.K. One-step reverse transcription loop-mediated isothermal amplification assay for rapid detection of Cymbidium mosaic virus. J. Virol. Methods 2011, 173, 43–48. [Google Scholar] [CrossRef]
- Ishikawa, K.; Maejima, K.; Netsu, O.; Fukuoka, M.; Nijo, T.; Hashimoto, M.; Takata, D.; Yamaji, Y.; Namba, S. Rapid detection of fig mosaic virus using reverse transcription loop-mediated isothermal amplification. J. Gen. Plant Pathol. 2015, 81, 382–389. [Google Scholar] [CrossRef]
- Almasi, M.A.; Almasi, G. Colorimetric immunocapture loop mediated isothermal amplification assay for detection of Impatiens necrotic spot virus (INSV) by GineFinder™ dye. Eur. J. Plant Pathol. 2018, 150, 533–538. [Google Scholar] [CrossRef]
- Fukuta, S.; Iida, T.; Mizukami, Y.; Ishida, A.; Ueda, J.; Kanbe, M.; Ishimoto, Y. Detection of Japanese yam mosaic virus by RT-LAMP. Arch. Virol. 2003, 148, 1713–1720. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xie, Z.; Fletcher, J.D.; Wang, Y.; Wang, R.; Guo, Z.; He, Y. Rapid and Sensitive Detection of Lettuce Necrotic Yellows Virus and Cucumber Mosaic Virus Infecting Lettuce (Lactuca sativa L.) by Reverse Transcription Loop-Mediated Isothermal Amplification. Plant Pathol. J. 2020, 36, 76. [Google Scholar] [PubMed]
- Zhao, B.; Yang, D.; Zhang, Y.; Xu, Y.; Zhao, X.; Liang, J.; Fan, X.; Du, Y.; Zhu, Z.; Shi, B.; et al. Rapid visual detection of lily mottle virus using a loop-mediated isothermal amplification method. Arch. Virol. 2018, 63, 545–548. [Google Scholar] [CrossRef]
- He, X.; Xue, F.; Xu, S.; Wang, W. Rapid and sensitive detection of Lily symptomless virus by reverse transcription loop-mediated isothermal amplification. J. Virol. Methods 2016, 238, 38–41. [Google Scholar] [CrossRef]
- Zhao, X.; Du, Y.; Zhang, Y.; Liang, J.; Cai, Y.; Xu, Y.; Yang, D. Effective detection of Lily Symptomless Virus using the reverse transcription loop–mediated isothermal amplification method. Australas. Plant Pathol. 2019, 48, 373–374. [Google Scholar] [CrossRef]
- Tahzima, R.; Foucart, Y.; Peusens, G.; Beliën, T.; Massart, S.; De Jonghe, K. New sensitive and fast detection of Little cherry virus 1 using loop-mediated isothermal amplification (RT-LAMP). J. Virol. Methods 2019, 265, 91–98. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, F.; Yu, Y.; Qiu, Z. The Application of Reverse Transcription-loopmediated Isothermal Amplification for the Rapid Detection of Maize Chlorotic Dwarf Virus. Agric. Sci. Technol. 2017, 18, 2450–2453. [Google Scholar]
- Chen, L.; Jiao, Z.; Liu, D.; Liu, X.; Xia, Z.; Deng, C.; Zhou, T.; Fan, Z. One-step reverse transcription loop-mediated isothermal amplification for the detection of Maize chlorotic mottle virus in maize. J. Virol. Methods 2017, 240, 49–53. [Google Scholar] [CrossRef]
- Qiao, N.; Dai, H.; Liu, J.; Zhu, X.; Li, J.; Zhang, D.; Liu, Y. Detection of melon necrotic spot virus by one-step reverse transcription loop-mediated isothermal amplification assay. PLoS ONE 2020, 15, e0230023. [Google Scholar] [CrossRef]
- Zeng, R.; Xu, L.H.; Gao, S.G.; Ni, X.H.; Chen, C.L.; Chen, J.C.; Dai, F.M. One-step reverse transcription loop-mediated isothermal amplification assay for rapid detection of melon yellow spot virus. Eur. J. Plant Pathol. 2016, 145, 119–124. [Google Scholar] [CrossRef]
- Meena, P.N.; Kharbikar, L.L.; Rana, R.S.; Satpathy, S.; Shanware, A.; Sivalingam, P.N.; Nandanwar, S. Detection of Mesta yellow vein mosaic virus (MeYVMV) in field samples by a loop-mediated isothermal amplification reaction. J. Virol. Methods 2019, 263, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, X.; Li, W.; Zhang, J.; Xiao, Z.; Zhou, Z.; Liu, T.; Li, Y.; Wang, F.; Zhang, S.; et al. Rapid detection of milk vetch dwarf virus by loop-mediated isothermal amplification. J. Virol. Methods 2018, 261, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Almasi, M.A. Development of a colorimetric reverse transcription loop-mediated isothermal amplification assay for the detection of Mirafiori lettuce big-vein virus. Arch. Virol. 2017, 162, 2775–2780. [Google Scholar] [CrossRef] [PubMed]
- Tiberini, A.; Tomlinson, J.; Micali, G.; Fontana, A.; Albanese, G.; Tomassoli, L. Development of a reverse transcription-loop mediated isothermal amplification (LAMP) assay for the rapid detection of onion yellow dwarf virus. J. Virol. Methods 2019, 271, 113680. [Google Scholar] [CrossRef]
- Shen, W.; Tuo, D.; Yan, P.; Li, X.; Zhou, P. Detection of Papaya leaf distortion mosaic virus by reverse-transcription loop-mediated isothermal amplification. J. Virol. Methods 2014, 195, 174–179. [Google Scholar] [CrossRef]
- Shen, W.; Tuo, D.; Yan, P.; Yang, Y.; Li, X.; Zhou, P. Reverse transcription loop-mediated isothermal amplification assay for rapid detection of Papaya ringspot virus. J. Virol. Methods 2014, 204, 93–100. [Google Scholar] [CrossRef]
- Boubourakas, I.N.; Fukuta, S.; Kyriakopoulou, P.E. Sensitive and rapid detection of peach latent mosaic viroid by the reverse transcription loop-mediated isothermal amplification. J. Virol. Methods 2009, 160, 63–68. [Google Scholar] [CrossRef]
- Hasiów-Jaroszewska, B.; Borodynko, N. Detection of Pepino mosaic virus isolates from tomato by one-step reverse transcription loop-mediated isothermal amplification. Arch. Virol 2013, 158, 2153–2156. [Google Scholar] [CrossRef]
- Ling, K.S.; Li, R.; Bledsoe, M. Pepino mosaic virus genotype shift in North America and development of a loop-mediated isothermal amplification for rapid genotype identification. Virol. J. 2013, 10, 117. [Google Scholar] [CrossRef]
- Tangkanchanapas, P.; Höfte, M.; De Jonghe, K. Reverse transcription loop-mediated isothermal amplification (RT-LAMP) designed for fast and sensitive on-site detection of Pepper chat fruit viroid (PCFVd). J. Virol. Methods 2018, 259, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Zhang, D.; Zheng, L.; Peng, J.; Li, F.; Zhang, S.; Liu, Y. Development of a reverse transcription loop-mediated isothermal amplification assay for the rapid detection of Pepper mottle virus. Can. J. Plant Pathol. 2016, 38, 506–510. [Google Scholar] [CrossRef]
- Komatsu, K.; Maejima, K.; Fujita, N.; Netsu, O.; Tomomitsu, T.; Arie, T.; Teraoka, T.; Namba, S. A detection method based on reverse transcription loop-mediated isothermal amplification for a genetically heterogeneous plantago asiatica mosaic virus. J. Gen. Plant Pathol. 2015, 81, 297–303. [Google Scholar] [CrossRef]
- Almasi, M.A.; Manesh, M.E.; Jafary, H.; Dehabadi, S.M.H. Visual detection of Potato leafroll virus by loop-mediated isothermal amplification of DNA with the Gene Finder TM dye. J. Virol. Methods 2013, 192, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Raigond, B.; Verma, A.; Jandrajupalli, S.; Kochhar, T.; Sharma, S.; Chakrabarti, S.K. Squash Print Reverse Transcription Loop-Mediated Isothermal Amplification Assay for Detection of Potato leafroll virus in Single Aphid and in Potato. Potato Res. 2020, 63, 1–14. [Google Scholar] [CrossRef]
- Tsutsumi, N.; Yanagisawa, H.; Fujiwara, Y.; Ohara, T. Detection of Potato Spindle Tuber Viroid by Reverse Transcription Loop-mediated Isothermal Amplification. Res. Bull. Pl. Prot. Jpn. 2010, 46, 61–67. [Google Scholar]
- Jeong, J.; Cho, S.Y.; Lee, W.H.; Lee, K.J.; Ju, H.J. Development of a Rapid Detection Method for Potato virus X by Reverse Transcription Loop-Mediated Isothermal Amplification. Plant Pathol. J. 2015, 31, 219–225. [Google Scholar] [CrossRef]
- Raigond, B.; Verma, A.; Roach, S.; Kochhar, T.; Arjunan, J.; Kumar, R.; Chakrabarti, S.K. One-step reverse transcription loop-mediated isothermal amplification: A simple, sensitive and rapid assay for detection of potato virus X in potato leaves and tubers. Indian Phytopathol. 2019, 72, 321–328. [Google Scholar] [CrossRef]
- Treder, K.; Chołuj, J.; Zacharzewska, B.; Babujee, L.; Mielczarek, M.; Burzyński, A.; Rakotondrafara, A.M. Optimization of a magnetic capture RT-LAMP assay for fast and real-time detection of potato virus Y and differentiation of N and O serotypes. Arch. Virol. 2018, 163, 447–458. [Google Scholar] [CrossRef]
- Hasiów-Jaroszewska, B.; Stachecka, J.; Minicka, J.; Sowiński, M.; Borodynko, N. Variability of Potato virus Y in Tomato Crops in Poland and Development of a Reverse-Transcription Loop-Mediated Isothermal Amplification Method for Virus Detection. Phytopathology 2015, 105, 1270–1276. [Google Scholar] [CrossRef]
- Zong, X.; Wang, W.; Wei, H.; Wang, J.; Chen, X.; Xu, L.; Zhu, D.; Tan, Y.; Liu, Q. Rapid detection of Prunus necrotic ringspot virus using magnetic nanoparticle-assisted reverse transcription loop-mediated isothermal amplification. J. Virol. Methods 2014, 208, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Sasaya, T. Detection methods for rice viruses by a reverse-transcription loop-mediated isothermal amplification (RT-LAMP). Methods Mol. Biol. 2015, 1236, 49–59. [Google Scholar] [PubMed]
- Du, L.; Shi, W.; Li, X.; Lan, Y.; Sun, F.; Fan, Y.; Zhou, T.; Zhou, Y. A reverse-transcription loop-mediated isothermal amplification (RT-LAMP) assay for detecting the pathogen of maize rough dwarf disease in China. Australas. Plant Pathol. 2019, 48, 485–489. [Google Scholar] [CrossRef]
- Le, D.T.; Netsu, O.; Uehara-Ichiki, T.; Shimizu, T.; Choi, I.R.; Omura, T.; Sasaya, T. Molecular detection of nine rice viruses by a reverse-transcription loop-mediated isothermal amplification assay. J. Virol. Methods 2010, 170, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Lai, D.; Zhang, Y.; Huang, Q.; Yin, G.; Pennerman, K.K.; Liu, Z.; Guo, A. Reverse transcription loop-mediated isothermal amplification to rapidly detect Rice ragged stunt virus. Saudi J. Biol. Sci. 2018, 25, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Keizerweerd, A.T.; Chandra, A.; Grisham, M.P. Development of a reverse transcription loop-mediated isothermal amplification (RT-LAMP) assay for the detection of Sugarcane mosaic virus and Sorghum mosaic virus in sugarcane. J. Virol. Methods 2015, 212, 23–29. [Google Scholar] [CrossRef]
- Zhou, T.; Du, L.; Fan, Y.; Zhou, Y. Reverse transcription loop-mediated isothermal amplification of RNA for sensitive and rapid detection of southern rice black-streaked dwarf virus. J. Virol. Methods 2012, 180, 91–95. [Google Scholar] [CrossRef]
- Kuan, C.P.; Wu, M.T.; Lu, Y.L.; Huang, H.C. Rapid detection of squash leaf curl virus by loop-mediated isothermal amplification. J. Virol. Methods 2010, 169, 61–65. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, Y.; Wu, Y.; Hao, X. Rapid Detection of Watermelon Viruses by Reverse Transcription Loop-Mediated Isothermal Amplification. J. Phytopathol. 2016, 164, 330–336. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; Choi, J.; Kim, S.K.; Jang, W. Development of Simple and Rapid Diagonosis Method for Strawberry Latent Ringspot Virus in Plants Using Loop-Mediated Isothermal Amplification Assay. J. Plant Pathol. Microbiol. 2016, 7, 377. [Google Scholar] [CrossRef]
- Anandakumar, L.; Bagyalakshmi, K.; Muthuramalingam, T.R.; Nithya, K.; Parameswari, B.; Viswanathan, R. Reverse transcription loop-mediated isothermal amplification based rapid detection of Sugarcane mosaic virus and Sugarcane streak mosaic virus associated with mosaic disease of sugarcane. Indian Phytopathol. 2020, 1–10. [Google Scholar] [CrossRef]
- Amata, R.L.; Fernandez, E.; Filloux, D.; Martin, D.P.; Rott, P.; Roumagnac, P. Prevalence of Sugarcane yellow leaf virus in sugarcane-producing regions in Kenya revealed by reverse-transcription loop-mediated isothermal amplification method. Plant Dis. 2016, 100, 260–268. [Google Scholar] [CrossRef]
- Zhao, L.; Cheng, J.; Hao, X.; Tian, X.; Wu, Y. Rapid detection of tobacco viruses by reverse transcription loop-mediated isothermal amplification. Arch. Virol. 2012, 157, 2291–2298. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, Z.; Qian, Y.; Mu, J.; Shen, L.; Wang, F.; Yang, J. Rapid detection of tobacco mosaic virus using the reverse transcription loop-mediated isothermal amplification method. Arch. Virol. 2010, 155, 1681–1685. [Google Scholar] [CrossRef] [PubMed]
- Gawande, S.P.; Raghavendra, K.P.; Monga, D.; Nagrale, D.T.; Kranthi, S. Rapid detection of Tobacco streak virus (TSV) in cotton (Gossypium hirsutum) based on Reverse Transcription Loop Mediated Isothermal Amplification (RT-LAMP). J. Virol. Methods 2019, 270, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Huang, C.; Wu, Z.; Zhang, X.; Wang, Y. Detection of Tomato aspermy virus infecting chrysanthemums by LAMP. Sci. Agric. Sin. 2010, 43, 1288–1294. [Google Scholar]
- Hasiów-Jaroszewska, B.; Budzyńska, D.; Borodynko, N.; Pospieszny, H. Rapid detection of genetically diverse tomato black ring virus isolates using reverse transcription loop-mediated isothermal amplification. Arch. Virol. 2015, 160, 3075–3078. [Google Scholar] [CrossRef]
- Sarkes, A.; Fu, H.; Feindel, D.; Harding, M.W.; Feng, J. Development and evaluation of a loop-mediated isothermal amplification (LAMP) assay for the detection of Tomato brown rugose fruit virus (ToBRFV). bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhao, L.M.; Li, G.; Gao, Y.; Zhu, Y.R.; Liu, J.; Zhu, X.P. Reverse transcription loop-mediated isothermal amplification assay for detecting tomato chlorosis virus. J. Virol. Methods 2015, 213, 93–97. [Google Scholar] [CrossRef]
- Karwitha, M.; Feng, Z.K.; Shen, Y.; Essendi, W.; Zhang, W.N.; Li, J.Y.; Tao, X.R. Rapid detection of Tomato chlorosis virus from infected plant and whitefly by one-step reverse transcription loop-mediated isothermal amplification. J. Phytopathol. 2016, 164, 255–263. [Google Scholar] [CrossRef]
- Sui, X.; Zhang, S.; Wu, Z.; Ling, K.S. Reverse transcription loop-mediated isothermal amplification for species-specific detection of tomato chlorotic spot orthotospovirus. J. Virol. Methods 2018, 253, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Arutselvan, R.; Reddy, M.K.; Makeshkumar, T. Rapid detection of tomato leaf curl Bengaluru virus through loop mediated isothermal amplification assay. Virus Dis. 2017, 28, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Naganur, P.; Premchand, U.; Shankarappa, K.S.; Mesta, R.K.; Manjunatha, C.; Patil, C.V. Development of a Loop-Mediated Isothermal Amplification Assay for Detection of Tomato Leaf Curl New Delhi Virus in Ridge Gourd [Luffa acutangula (L.) Roxb.]. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 2282–2295. [Google Scholar] [CrossRef]
- Jeevalatha, A.; Kaundal, P.; Kumar, R.; Raigond, B.; Kumar, R.; Sharma, S.; Chakrabarti, S.K. Optimized loop-mediated isothermal amplification assay for Tomato leaf curl New Delhi virus-[potato] detection in potato leaves and tubers. Eur. J. Plant Pathol. 2018, 150, 565–573. [Google Scholar] [CrossRef]
- Li, R.; Ling, K.S. Development of reverse transcription loop-mediated isothermal amplification assay for rapid detection of an emerging potyvirus: Tomato necrotic stunt virus. J. Virol. Methods 2014, 200, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, S.; Ohishi, K.; Yoshida, K.; Mizukami, Y.; Ishida, A.; Kanbe, M. Development of immunocapture reverse transcription loop-mediated isothermal amplification for the detection of tomato spotted wilt virus from chrysanthemum. J. Virol. Methods 2004, 121, 49–55. [Google Scholar] [CrossRef]
- Fukuta, S.; Kato, S.; Yoshida, K.; Mizukami, Y.; Ishida, A.; Ueda, J.; Kanbe, M.; Ishimoto, Y. Detection of Tomato yellow leaf curl virus by loop-mediated isothermal amplification reaction. J. Virol. Methods 2003, 112, 35–40. [Google Scholar] [CrossRef]
- Zhao, L.; Hao, X.; Liu, H.; Wang, Q.; Wu, Y. Rapid Detection of Turnip mosaic virus by Reverse Transcription Loop-Mediated Isothermal Amplification. J. Phytopathol. 2014, 162, 693–696. [Google Scholar] [CrossRef]
- Congdon, B.S.; Kehoe, M.A.; Filardo, F.F.; Coutts, B.A. In-field capable loop-mediated isothermal amplification detection of Turnip yellows virus in plants and its principal aphid vector Myzus persicae. J. Virol. Methods 2019, 265, 15–21. [Google Scholar] [CrossRef]
- Lee, S.; Kim, J.H.; Choi, J.Y.; Jang, W.C. Loop-mediated isothermal amplification assay to rapidly detect wheat streak mosaic virus in quarantined plants. Plant Pathol. J. 2015, 31, 438. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Liu, X.J.; Li, D.W.; Yu, J.L.; Han, C.G. Rapid detection of wheat yellow mosaic virus by reverse transcription loop-mediated isothermal amplification. Virol. J. 2011, 8, 550. [Google Scholar] [CrossRef] [PubMed]
- Nkere, C.K.; Oyekanmi, J.O.; Silva, G.; Bömer, M.; Atiri, G.I.; Onyeka, J.; Maroya, N.G.; Seal, S.E.; Kumar, P.L. Chromogenic detection of yam mosaic virus by closed-tube reverse transcription loop-mediated isothermal amplification (CT-RT-LAMP). Arch. Virol. 2018, 163, 1057–1061. [Google Scholar] [CrossRef]
- Kuan, C.P.; Deng, T.C.; Huang, H.C.; Chi, H.H.; Lu, Y.L. Use of Reverse Transcription Loop-Mediated Isothermal Amplification for the Detection of Zucchini yellow mosaic virus. J. Phytopathol. 2014, 162, 238–244. [Google Scholar] [CrossRef]
- Mori, Y.; Nagamine, K.; Tomita, N.; Notomi, T. Detection of loop-mediated isothermal amplification reaction by turbidity derived from magnesium pyrophosphate formation. Biochem. Biophys. Res. Commun. 2001, 289, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, T.; Sonobe, T.; Hayashi, K. Loop-mediated isothermal amplification for direct detection of Mycobacterium tuberculosis complex, M. avium, and M. intracellulare in sputum samples. J. Clin. Microbiol. 2003, 41, 2616–2622. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Kitao, M.; Tomita, N.; Notomi, T. Real-time turbidimetry of LAMP reaction for quantifying template DNA. J. Biochem. Biophys. Methods 2004, 59, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Parida, M.; Horioke, K.; Ishida, H.; Dash, P.K.; Saxena, P.; Jana, A.M.; Islam, M.A.; Inoue, S.; Hosaka, N.; Morita, K. Rapid detection and differentiation of dengue virus serotypes by a real-time reverse transcription-loop-mediated isothermal amplification assay. J. Clin. Microbiol. 2005, 43, 2895–2903. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Kokeguchi, S.; Fujimoto, C.; Tanimoto, I.; Yoshizumi, W.; Nishimura, F.; Takashiba, S. Detection of periodontal pathogen Porphyromonas gingivalis by loop-mediated isothermal amplification method. FEMS Immunol. Med. Microbiol. 2005, 43, 233–239. [Google Scholar] [CrossRef]
- Boehme, C.C.; Nabeta, P.; Henostroza, G.; Raqib, R.; Rahim, Z.; Gerhardt, M.; Sanga, E.; Hoelscher, M.; Notomi, T.; Hase, T.; et al. Operational feasibility of using loop-mediated isothermal amplification for diagnosis of pulmonary tuberculosis in microscopy centers of developing countries. J. Clin. Microbiol. 2007, 45, 1936–1940. [Google Scholar] [CrossRef]
- Parida, M.; Posadas, G.; Inoue, S.; Hasebe, F.; Morita, K. Real-time reverse transcription loop-mediated isothermal amplification for rapid detection of West Nile virus. J. Clin. Microbiol. 2004, 42, 257–263. [Google Scholar] [CrossRef]
- Monis, P.T.; Giglio, S.; Saint, C.P. Comparison of SYTO9 and SYBR Green I for real-time polymerase chain reaction and investigation of the effect of dye concentration on amplification and DNA melting curve analysis. Anal. Biochem. 2005, 340, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Parida, M.; Sannarangaiah, S.; Dash, P.K.; Rao, P.V.L.; Morita, K. Loop mediated isothermal amplification (LAMP): A new generation of innovative gene amplification technique; perspectives in clinical diagnosis of infectious diseases. Rev. Med. Virol. 2008, 18, 407–421. [Google Scholar] [CrossRef] [PubMed]
- Curtis, K.A.; Rudolph, D.L.; Owen, S.M. Rapid detection of HIV-1 by reverse-transcription, loop-mediated isothermal amplification (RT-LAMP). J. Virol. Methods 2008, 151, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Dukes, J.P.; King, D.P.; Alexandersen, S. Novel reverse transcription loop-mediated isothermal amplification for rapid detection of foot-and-mouth disease virus. Arch. Virol. 2006, 151, 1093–1106. [Google Scholar] [CrossRef]
- Hill, J.; Beriwal, S.; Chandra, I.; Paul, V.K.; Kapil, A.; Singh, T.; Goyal, A.; Jahnukainen, T.; Johnson, J.R.; Tarr, P.I.; et al. Loop-mediated isothermal amplification assay for rapid detection of common strains of Escherichia coli. J. Clin. Microbiol. 2008, 46, 2800–2804. [Google Scholar] [CrossRef]
- Zoheir, K.M.; Allam, A.A. A rapid method for sexing the bovine embryo. Anim. Reprod. Sci. 2010, 119, 92–96. [Google Scholar] [CrossRef]
- Zoheir, K.M.A.; Allam, A.A. A rapid improved method for sexing embryo of water buffalo. Theriogenology 2011, 76, 83–87. [Google Scholar] [CrossRef]
- Goto, M.; Honda, E.; Ogura, A.; Nomoto, A.; Hanaki, K.I. Colorimetric detection of loop-mediated isothermal amplification reaction by using hydroxy naphthol blue. Biotechniques 2009, 46, 167–172. [Google Scholar] [CrossRef]
- Lee, S.Y.; Huang, J.G.; Chuang, T.L.; Sheu, J.C.; Chuang, Y.K.; Holl, M.; Meldrum, D.R.; Leeb, C.; Lin, C.W. Compact optical diagnostic device for isothermal nucleic acids amplification. Sens. Actuators B Chem. 2008, 133, 493–501. [Google Scholar] [CrossRef]
- Nagamine, K.; Watanabe, K.; Ohtsuka, K.; Hase, T.; Notomi, T. Loop-mediated isothermal amplification reaction using a nondenatured template. Clin. Chem. 2001, 47, 1742–1743. [Google Scholar] [CrossRef] [PubMed]
- Peyrefitte, C.N.; Boubis, L.; Coudrier, D.; Bouloy, M.; Grandadam, M.; Tolou, H.J.; Plumet, S. Real-time reverse-transcription loop-mediated isothermal amplification for rapid detection of Rift Valley fever virus. J. Clin. Microbiol. 2008, 46, 3653–3659. [Google Scholar] [CrossRef]
- Thiessen, L.D.; Neill, T.M.; Mahaffee, W.F. Development of a quantitative loop-mediated isothermal amplification assay for the field detection of Erysiphe necator. PeerJ 2018, 6, e4639. [Google Scholar] [CrossRef] [PubMed]
- Puchades, A.V.; Carpino, C.; Alfaro-Fernandez, A.; Font-San-Ambrosio, M.I.; Davino, S.; Guerri, J.; Rubio, L.; Galipienso, L. Detection of Southern tomato virus by molecular hybridisation. Ann. Appl. Biol. 2017, 171, 172–178. [Google Scholar] [CrossRef]
- Mugasa, C.M.; Laurent, T.; Schoone, G.J.; Kager, P.A.; Lubega, G.W.; Schallig, H.D. Nucleic acid sequence-based amplification with oligochromatography for detection of Trypanosoma brucei in clinical samples. J. Clin. Microbiol. 2009, 47, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.T.; Little, M.C.; Nadeau, J.G.; Shank, D.D. Isothermal in vitro amplification of DNA by a restriction enzyme/DNA polymerase system. Proc. Natl. Acad. Sci. USA 1992, 89, 392–396. [Google Scholar] [CrossRef]
- Lizardi, P.M.; Huang, X.; Zhu, Z.; Bray-Ward, P.; Thomas, D.C.; Ward, D.C. Mutation detection and single-molecule counting using isothermal rolling-circle amplification. Nat. Genet. 1998, 19, 225. [Google Scholar] [CrossRef]
- Gill, P.; Ghaemi, A. Nucleic acid isothermal amplification technologies—A review. Nucleosides Nucleotides Nucleic Acids 2008, 27, 224–243. [Google Scholar] [CrossRef]
- Mori, Y.; Kanda, H.; Notomi, T. Loop-mediated isothermal amplification (LAMP): Recent progress in research and development. J. Infect. Chemother. 2013, 19, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Njiru, Z.K. Loop-mediated isothermal amplification technology: Towards point of care diagnostics. PLoS Negl. Trop. Dis. 2012, 6, e1572. [Google Scholar] [CrossRef]
- LaBarre, P.; Hawkins, K.R.; Gerlach, J.; Wilmoth, J.; Beddoe, A.; Singleton, J.; Boyle, D.; Weigl, B. A simple, inexpensive device for nucleic acid amplification without electricity—Toward instrument-free molecular diagnostics in low-resource settings. PLoS ONE 2011, 6, e19738. [Google Scholar] [CrossRef]
- Iseki, H.; Alhassan, A.; Ohta, N.; Thekisoe, O.M.; Yokoyama, N.; Inoue, N.; Nambota, A.; Yasuda, J.; Igarashi, I. Development of a multiplex loop-mediated isothermal amplification (mLAMP) method for the simultaneous detection of bovine Babesia parasites. J. Microbiol. Methods 2007, 71, 281–287. [Google Scholar] [CrossRef]
- Tanner, N.A.; Zhang, Y.; Evans, T.C., Jr. Simultaneous multiple target detection in real-time loop-mediated isothermal amplification. Biotechniques 2012, 53, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Liu, Y.; Kong, J.; Jiang, X. Loop-mediated isothermal amplification integrated on microfluidic chips for point-of-care quantitative detection of pathogens. Anal. Chem. 2010, 82, 3002–3006. [Google Scholar] [CrossRef]
- Gansen, A.; Herrick, A.M.; Dimov, I.K.; Lee, L.P.; Chiu, D.T. Digital LAMP in a sample self-digitization (SD) chip. Lab Chip 2012, 12, 2247–2254. [Google Scholar] [CrossRef]
- Lin, X.; Huang, X.; Urmann, K.; Xie, X.; Hoffmann, M.R. Digital Loop-Mediated Isothermal Amplification on a Commercial Membrane. ACS Sens. 2019, 4, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Soliman, H.; El-Matbouli, M. Loop mediated isothermal amplification combined with nucleic acid lateral flow strip for diagnosis of cyprinid herpes virus-3. Mol. Cell. Probes 2010, 24, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Rigano, L.A.; Malamud, F.; Orce, I.G.; Filippone, M.P.; Marano, M.R.; Do Amaral, A.M.; Castagnaro, A.P.; Vojnov, A.A. Rapid and sensitive detection of Candidatus Liberibacter asiaticus by loop mediated isothermal amplification combined with a lateral flow dipstick. BMC Microbiol. 2014, 14, 86. [Google Scholar] [CrossRef]
- Salinas, N.R.; Little, D.P. Electric LAMP: Virtual loop-mediated isothermal AMPlification. ISRN Bioinform. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Kinahan, D.J.; Julius, L.A.; Schoen, C.; Dreo, T.; Ducrée, J. Automated DNA purification and multiplexed lamp assay preparation on a centrifugal microfluidic “Lab-on-a-Disc” platform. In Proceedings of the 2018 IEEE Micro Electro Mechanical Systems (MEMS), Belfast, UK, 21–25 January 2018; pp. 1134–1137. [Google Scholar]
- Masilamani, S.M.; Sasitharan, D.; Manohar, P.W. Associated technologies ensures complete loop mediated isothermal amplification platform for pathogen diagnosis. Afr. J. Biotechnol. 2013, 12. [Google Scholar] [CrossRef]
- Abdullahi, U.F.; Naim, R.; Taib, W.R.W.; Saleh, A.; Muazu, A.; Aliyu, S.; Baig, A.A. Loop-mediated isothermal amplification (LAMP), an innovation in gene amplification: Bridging the gap in molecular diagnostics; a review. Indian J. Sci. Technol. 2015, 8, 1. [Google Scholar] [CrossRef]
- Davino, S.; Panno, S.; Arrigo, M.; La Rocca, M.; Caruso, A.G.; Bosco, G.L. Planthology: An application system for plant diseases management. Chem. Eng. Trans. 2017, 58, 619–624. [Google Scholar]
- Kubota, R.; Alvarez, A.M.; Su, W.W.; Jenkins, D.M. FRET-based assimilating probe for sequence specific real-time monitoring of loop-mediated isothermal amplification (LAMP). Biol. Eng. Trans. 2011, 4, 81–100. [Google Scholar] [CrossRef]
- Yi, J.Z.; Zhang, W.D.; Zhang, D.Y. Molecular Zipper: A fluorescent probe for realtime isothermal DNA amplification. Nucleic Acids Res. 2006, 34, e81. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.G.M.; Bhaskar, K.R.H.; Salam, M.A.; Akther, T.; Pluschke, G.; Mondal, D. Diagnostic accuracy of loop-mediated isothermal amplification (LAMP) for detection of Leishmania DNA in buffy coat from visceral leishmaniasis patients. Parasites Vectors 2012, 5, 280. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Karthik, K.; Chakraborty, S.; Tiwari, R.; Kapoor, S.; Kumar, A.; Thomas, P. Loop-mediated isothermal amplification of DNA (LAMP): A new diagnostic tool lights the world of diagnosis of animal and human pathogens: A review. Pak. J. Biol. Sci. 2014, 17, 151–166. [Google Scholar] [CrossRef] [PubMed]
- Bai, Z.; Shi, L.; Hu, C.; Chen, X.; Qi, J.; Ba, X.; Peng, Q.; Chen, Y.; Chen, H.; Guo, A. Development of a loop-mediated isothermal amplification assay for sensitive and rapid detection of Mycoplasma bovis. Afr. J. Biotechnol. 2011, 10, 12333–12338. [Google Scholar]
- He, L.; Xu, H.S. Development of a multiplex loop-mediated isothermal amplification (mLAMP) method for the simultaneous detection of white spot syndrome virus and infectious hypodermal and hematopoietic necrosis virus in penaeid shrimp. Aquaculture 2011, 311, 94–99. [Google Scholar] [CrossRef]
- Chen, X.; Wang, X.; Jin, N.; Zhou, Y.; Huang, S.; Miao, Q.; Zhu, Q.; Xu, J. Endpoint visual detection of three genetically modified rice events by loop-mediated isothermal amplification. Int. J. Mol. Sci. 2012, 13, 14421–14433. [Google Scholar] [CrossRef]
- Lee, D.; La Mura, M.; Allnutt, T.R.; Powell, W. Detection of genetically modified organisms (GMOs) using isothermal amplification of target DNA sequences. BMC Biotechnol. 2009, 9, 7. [Google Scholar] [CrossRef]
- Hirayama, H.; Kageyama, S.; Moriyasu, S.; Sawai, K.; Onoe, S.; Takahashi, Y.; Katagiri, S.; Toen, K.; Watanabe, K.; Notomi, T.; et al. Rapid sexing of bovine preimplantation embryos using loop-mediated isothermal amplification. Theriogenology 2004, 62, 887–896. [Google Scholar] [CrossRef]
- Horibe, D.; Ochiai, T.; Shimada, H.; Tomonaga, T.; Nomura, F.; Gun, M.; Tanizawa, T.; Hayashi, H. Rapid detection of metastasis of gastric cancer using reverse transcription loop-mediated isothermal amplification. Int. J. Cancer 2007, 120, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Davino, S.; Panno, S.; Iacono, G.; Sabatino, L.; D’Anna, F.; Iapichino, G.; Olmos, A.; Scuderi, G.; Rubio, L.; Tomassoli, L.; et al. Genetic variation and evolutionary analysis of Pepino mosaic virus in Sicily: Insights into the dispersion and epidemiology. Plant Pathol. 2017, 66, 368–375. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Davino, S. The nucleotide sequence of a recombinant tomato yellow leaf curl virus strain frequently detected in Sicily isolated from tomato plants carrying the Ty-1 resistance gene. Arch. Virol. 2018, 163, 795–797. [Google Scholar] [CrossRef] [PubMed]
- Panno, S.; Caruso, A.G.; Troiano, E.; Luigi, M.; Manglli, A.; Vatrano, T.; Iacono, G.; Marchione, S.; Bertin, S.; Tomassoli, L.; et al. Emergence of tomato leaf curl New Delhi virus in Italy: Estimation of incidence and genetic diversity. Plant Pathol. 2019, 68, 601–608. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Davino, S. First report of tomato brown rugose fruit virus on tomato crops in Italy. Plant Dis. 2019, 103, 1443. [Google Scholar] [CrossRef]

| Species | Acronym | Genome | Family | Genus | Reference |
|---|---|---|---|---|---|
| Abaca bunchy top virus | ABTV | ssDNA(+) | Nanoviridae | Babuvirus | [49] |
| Apple chlorotic leaf spot virus | ACLSV | ssRNA(+) | Betaflexiviridae | Trichovirus | [50,51] |
| Apple scar skin viroid | ASSVd | circRNA | Pospiviroidae | Apscaviroid | [52] |
| Apple stem pitting virus | ASPV | ssRNA(+) | Betaflexiviridae | Foveavirus | [51] |
| Arabis mosaic virus | ArMV | ssRNA(+) | Secoviridae | Nepovirus | [53] |
| Banana bract mosaic virus | BBMV | ssRNA(+) | Potyviridae | Potyvirus | [54] |
| Banana bunchy top virus | BBTV | ssDNA(+) | Nanoviridae | Babuvirus | [49,55] |
| Banana streak virus | BSV | dsDNA-RT | Caulimoviridae | Badnavirus | [56] |
| Barley stripe mosaic virus | BSMV | ssRNA(+) | Virgaviridae | Hordeivirus | [57] |
| Barley yellow dwarf virus | BYDV | ssRNA(+) | Luteoviridae | Luteovirus | [58] |
| Bean common mosaic necrosis virus | BCMNV | ssRNA(+) | Potyviridae | Potyvirus | [59] |
| Bean pod mottle virus | BPMV | ssRNA(+) | Secoviridae | Comovirus | [60] |
| Beet curly top virus | BCTV | Circ-ssDNA(+/−) | Geminiviridae | Curtovirus | [61] |
| Beet mild curly top virus | BMCTP | Circ-ssDNA(+/−) | Geminiviridae | Curtovirus | [61] |
| Beet necrotic yellow vein virus | BNYVV | ssRNA(+) | Benyviridae | Benyvirus | [62] |
| Beet severe curly top virus | BSVTV | Circ-ssDNA(+/−) | Geminiviridae | Curtovirus | [61] |
| Cassava brown streak virus | CBSV | ssRNA(+) | Potyviridae | Ipomovirus | [63] |
| Chilli veinal mottle virus | ChiVMV | ssRNA(+) | Potyviridae | Potyvirus | [64] |
| Chinese wheat mosaic virus | CWMV | ssRNA(+) | Virgaviridae | Furovirus | [65] |
| Chrysanthemum chlorotic mottle viroid | CCMVd | circRNA | Avsunviroidae | Pelamoviroid | [66] |
| Chrysanthemum stem necrosis virus | CSNV | ssRNA(+/−) | Tospoviridae | Orthotospovirus | [67] |
| Chrysanthemum stunt viroid | CSVd | CircRNA | Pospiviroidae | Pospiviroid | [68] |
| Chrysanthemum virus B | CVB | ssRNA(+) | Betaflexiviridae | Carlavirus | [68] |
| Citrus leaf blotch virus | CLBV | ssRNA(+) | Betaflexiviridae | Citrivirus | [69] |
| Citrus tristeza virus | CTV | ssRNA(+) | Closteroviridae | Closterosvirus | [38,70,71] |
| Citrus yellow mosaic virus | CYMV | dsDNA-RT | Caulimoviridae | Badnavirus | [72] |
| Coconut cadang-cadang viroid | CCCVd | CircRNA | Pospiviroidae | Cocadviroid | [73] |
| Columnea latent viroid | CLVd | CircRNA | Pospiviroidae | Pospiviroid | [74] |
| Cucumber green mottle mosaic virus | CGMV | ssRNA(+) | Virgaviridae | Tobamovirus | [75] |
| Cucumber mosaic virus | CMV | ssRNA(+) | Bromoviridae | Cucumovirus | [76,77,78] |
| Cucurbit chlorotic yellows virus | CCYV | ssRNA(+) | Closteroviridae | Crinivirus | [79] |
| Cucurbit leaf crumple virus | CuLCrV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [80] |
| Cymbidium mosaic virus | CymMV | ssRNA(+) | Alphaflexiviridae | Potexvirus | [81] |
| Fig mosaic virus | FMV | ssRNA(−) | Fimoviridae | Emaravirus | [82] |
| Grapevine leafroll-associated virus 3 | GLRaV-3 | ssRNA(+) | Closteroviridae | Ampelovirus | [27] |
| Grapevine red blotch virus | GRBD | ssDNA(+/−) | Geminiviridae | Grablovirus | [37] |
| Impatiens necrotic spot virus | INSV | ssRNA | Bunyaviridae | Tospovirus | [83] |
| Japanese soil-borne wheat mosaic virus | JSBWMV | ssRNA(+) | Virgaviridae | Furovirus | [65] |
| Japanese yam mosaic virus | JYMV | ssRNA(+) | Potyviridae | Potyvirus | [84] |
| Lettuce necrotic yellows virus | LNYV | ssRNA(−) | Rhabdoviridae | Cytorhabdovirus | [85] |
| Lily mottle virus | LMoV | ssRNA(+) | Potyviridae | Potyvirus | [86] |
| Lily symptomless virus | LSLV | ssRNA(+) | Betaflexiviridae | Carlavirus | [87,88] |
| Little cherry virus 1 | LChV-1 | ssRNA(+) | Closteroviridae | Velarivirus | [89] |
| Maize chlorotic dwarf virus | MCDV | ssRNA(+) | Sequiviridae | Waikavirus | [90] |
| Maize chlorotic mottle virus | MCMV | ssRNA(+) | Tombusviridae | Machlomovirus | [91] |
| Melon necrotic spot virus | MNSV | ssRNA(+) | Tombusviridae | Carmovirus | [92] |
| Melon yellow spot virus | MYSV | ssRNA(+/−) | Bunyaviridae | Tospovirus | [93] |
| Mesta yellow vein mosaic virus | MeYVMV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [94] |
| Milk vetch dwarf virus | MDV | ssDNA(+) | Nanoviridae | Nanovirus | [95] |
| Mirafiori lettuce big-vein virus | MiLBVV | ssRNA(−) | Ophioviridae | Ophiovirus | [96] |
| Onion yellow dwarf virus | OYSV | ssRNA(+) | Potyviridae | Potyvirus | [97] |
| Papaya leaf distortion mosaic virus | PLDMV | ssRNA(+) | Potyviridae | Potyvirus | [98] |
| Papaya ringspot virus | PRSV | ssRNA(+) | Potyviridae | Potyvirus | [99] |
| Peach latent mosaic viroid | PLMVd | CircRNA | Avsunviroidae | Pelamoviroid | [100] |
| Pepino mosaic virus | PepMV | ssRNA(+) | Alphaflexiviridae | Potexvirus | [101,102] |
| Pepper chat fruit viroid | PCFVd | CircRNA | Pospiviroidae | Pospiviroid | [103] |
| Pepper mottle virus | PepMoV | ssRNA(+) | Potyviridae | Potyvirus | [104] |
| Pepper yellow leaf curl Indonesia virus | PepYLCIDV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [39] |
| Piper yellow mottle virus | PYMoV | dsDNA | Caulimoviridae | Badnavirus | [76] |
| Plantago asiatica mosaic virus | PlAMV | ssRNA(+) | Alphaflexiviridae | Potexvirus | [105] |
| Plum pox virus | PPV | ssRNA(+) | Potyviridae | Potyvirus | [30] |
| Potato leafroll virus | PLRV | ssRNA(+) | Luteoviridae | Polerovirus | [106,107] |
| Potato spindle tuber viroid | PSTVd | Circ-ssRNA | Pospiviroidae | Pospiviroid | [108] |
| Potato virus X | PVX | ssRNA(+) | Alphaflexiviridae | Potexvirus | [109,110] |
| Potato virus Y | PVY | ssRNA(+) | Potyviridae | Potyvirus | [111,112] |
| Prunus necrotic ringspot virus | PNRSV | ssRNA(+) | Bromoviridae | Ilarvirus | [113] |
| Rice black-streaked dwarf virus | RBSDV | dsRNA | Reoviridae | Fijivirus | [114,115] |
| Rice dwarf virus | RDV | dsRNA | Reoviridae | Phytoreovirus | [114,116] |
| Rice gall dwarf virus | RGDV | dsRNA | Reoviridae | Phytoreovirus | [114,116] |
| Rice grassy stunt virus | RGSV | ssRNA(−) | Phenuiviridae | Tenuivirus | [114,116] |
| Rice ragged stunt virus | RRSV | dsRNA | Reoviridae | Oryzavirus | [114,115,116,117] |
| Rice stripe virus | RSV | ssRNA(−) | Phenuiviridae | Tenuivirus | [114,116] |
| Rice transitory yellowing virus | RTYV | ssRNA(−) | Rhabdoviridae | Nucleorhabdovirus | [114,116] |
| Rice tungro bacilliform virus | RTBV | dsDNA-RT | Caulimoviridae | Tungrovirus | [114,116] |
| Rice tungro spherical virus | RTSV | ssRNA(+) | Secoviridae | Waikavirus | [114,116] |
| Sorghum mosaic virus | SrMV | ssRNA(+) | Potyviridae | Potyvirus | [118] |
| Southern rice black-streaked dwarf virus | SRBSDV | dsRNA | Reoviridae | Fijivirus | [119] |
| Southern tomato virus | STV | dsRNA | Amalgaviridae | Amalgavirus | [35] |
| Squash leaf curl virus | SLCV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [120] |
| Squash mosaic virus | SqMV | ssRNA(+) | Comoviridae | Comovirus | [121] |
| Strawberry latent ringspot virus | SLRSV | ssRNA(+) | Secoviridae | Unassigned | [122] |
| Sugarcane mosaic virus | SCMV | ssRNA(+) | Potyviridae | Potyvirus | [118,123] |
| Sugarcane streak mosaic virus | SCSMV | ssRNA(+) | Potyviridae | Poacevirus | [123] |
| Sugarcane yellow leaf virus | SCYLV | ssRNA(+) | Luteoviridae | Polerovirus | [124] |
| Tobacco etch virus | TEV | ssRNA(+) | Potyviridae | Potyvirus | [125] |
| Tobacco mosaic virus | TMV | ssRNA(+) | Virgaviridae | Tobamovirus | [125,126] |
| Tobacco streak virus | TSV | ssRNA(+) | Bromoviridae | Ilarvirus | [127] |
| Tobacco vein banding mosaic virus | TVBMV | ssRNA(+) | Potyviridae | Potyvirus | [125] |
| Tomato aspermy virus | TAV | ssRNA(+) | Bromoviridae | Cucumovirus | [128] |
| Tomato black ring virus | TBRV | ssRNA(+) | Secoviridae | Nepovirus | [129] |
| Tomato brown rugose fruit virus | ToBRFV | ssRNA(+) | Virgaviridae | Tobamovirus | [130] |
| Tomato chlorosis virus | ToCV | ssRNA(+) | Closteroviridae | Crinivirus | [131,132] |
| Tomato chlorotic spot virus | TCSV | ssRNA(+/−) | Tospoviridae | Orthotospovirus | [133] |
| Tomato leaf curl Bengaluru virus | ToLCBaV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [134] |
| Tomato leaf curl New Delhi virus | ToLCNDV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [39,135,136] |
| Tomato necrotic stunt virus | ToNSV | ssRNA(+) | Potyviridae | Potyvirus | [137] |
| Tomato spotted wilt virus | TSWV | ssRNA(+/−) | Tospoviridae | Orthotospovirus | [138] |
| Tomato torrado virus | ToTV | ssRNA(+) | Secoviridae | Torradovirus | [32] |
| Tomato yellow leaf curl Kanchanaburi virus | TYLCKaV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [39] |
| Tomato yellow leaf curl virus | TYLCV | Circ-ssDNA(+/−) | Geminiviridae | Begomovirus | [139] |
| Turnip mosaic virus | TuMV | ssRNA(+) | Potyviridae | Potyvirus | [140] |
| Turnip yellows virus | TuYV | ssRNA(+) | Luteoviridae | Polerovirus | [141] |
| Ugandan cassava brown streak virus | UCBSV | ssRNA(+) | Potyviridae | Ipomovirus | [63] |
| Watermelon mosaic virus | WMV | ssRNA(+) | Potyviridae | Potyvirus | [121] |
| Wheat streak mosaic virus | WSMV | ssRNA(+) | Potyviridae | Tritimovirus | [142] |
| Wheat yellow mosaic virus | WYMV | ssRNA(+) | Potyviridae | Bymovirus | [65,143] |
| Yam mosaic virus | YMV | ssRNA(+) | Potyviridae | Potyvirus | [144] |
| Zucchini yellow mosaic virus | ZYMV | ssRNA(+) | Potyviridae | Potyvirus | [121,145] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panno, S.; Matić, S.; Tiberini, A.; Caruso, A.G.; Bella, P.; Torta, L.; Stassi, R.; Davino, S. Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology. Plants 2020, 9, 461. https://doi.org/10.3390/plants9040461
Panno S, Matić S, Tiberini A, Caruso AG, Bella P, Torta L, Stassi R, Davino S. Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology. Plants. 2020; 9(4):461. https://doi.org/10.3390/plants9040461
Chicago/Turabian StylePanno, Stefano, Slavica Matić, Antonio Tiberini, Andrea Giovanni Caruso, Patrizia Bella, Livio Torta, Raffaele Stassi, and Salvatore Davino. 2020. "Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology" Plants 9, no. 4: 461. https://doi.org/10.3390/plants9040461
APA StylePanno, S., Matić, S., Tiberini, A., Caruso, A. G., Bella, P., Torta, L., Stassi, R., & Davino, S. (2020). Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology. Plants, 9(4), 461. https://doi.org/10.3390/plants9040461






