Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends
Abstract
1. Introduction
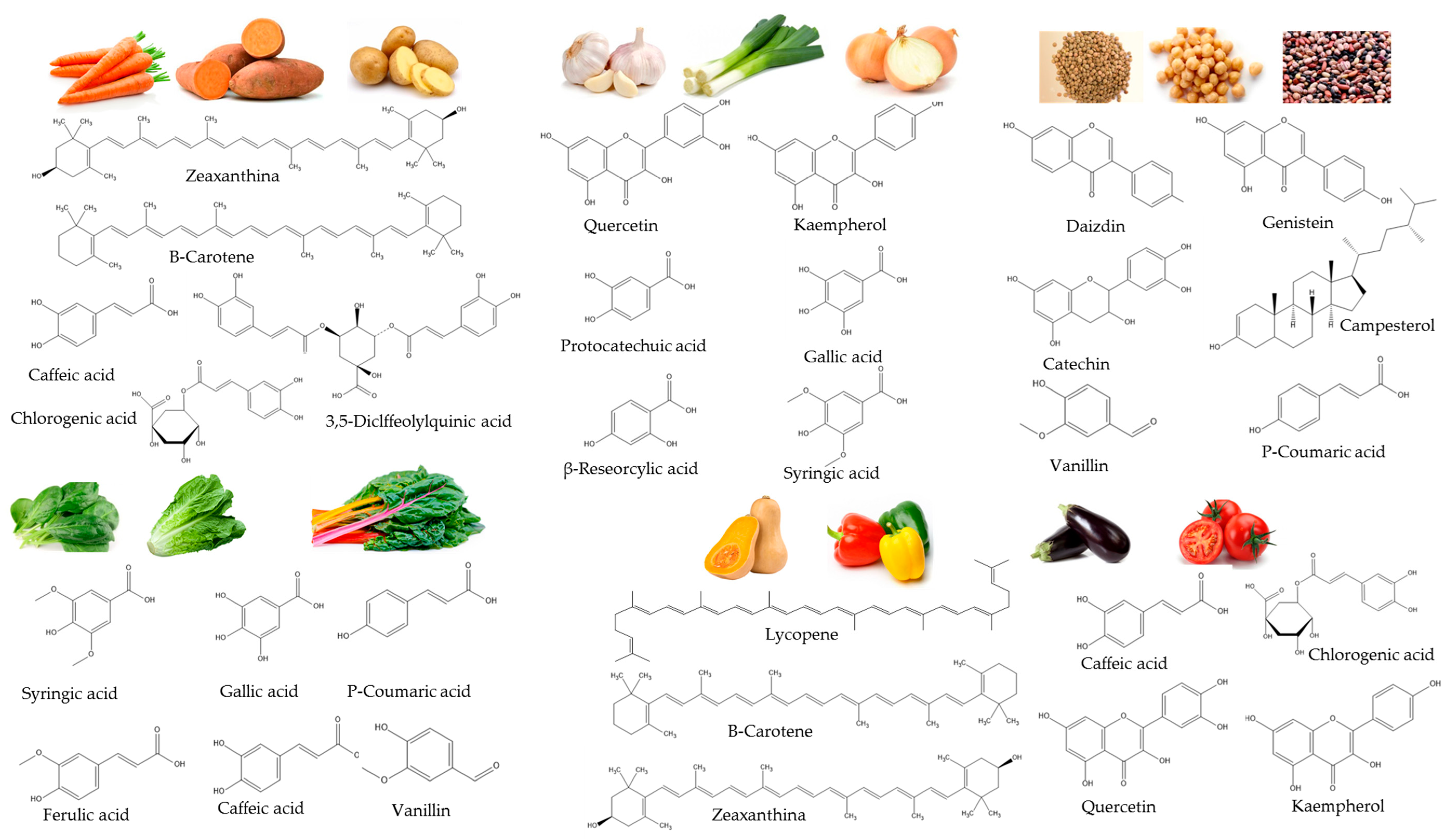
2. Main Raw Materials Used in Vegetable Soups and Cream Production
2.1. Fruit Vegetables
2.1.1. Pepper (Capsicum spp.)
2.1.2. Eggplant (Solanum melongena L.)
2.1.3. Tomato (Solanum lycopersicum)
2.1.4. Pumpkin (Cucurbita spp.)
2.2. Green Leafy Vegetables
2.2.1. Chards (Beta vulgaris var. cicla)
2.2.2. Spinach (Spinacia oleracea)
2.3. Tubers
2.3.1. Potato (Solanum tuberosum)
2.3.2. Sweet Potato (Ipomea batatas L.)
2.3.3. Carrots (Daucus carota)
2.4. Bulbs
2.4.1. Onions (Allium cepa L.)
2.4.2. Leek (Allium ampeloprasum var. porrum)
2.4.3. Garlic (Allium sativum)
2.5. Legumes
2.5.1. Lentil (Lens culinaris)
2.5.2. Bean (Phaseolus vulgaris L.)
2.5.3. Chickpea (Cicer aretianum L.)
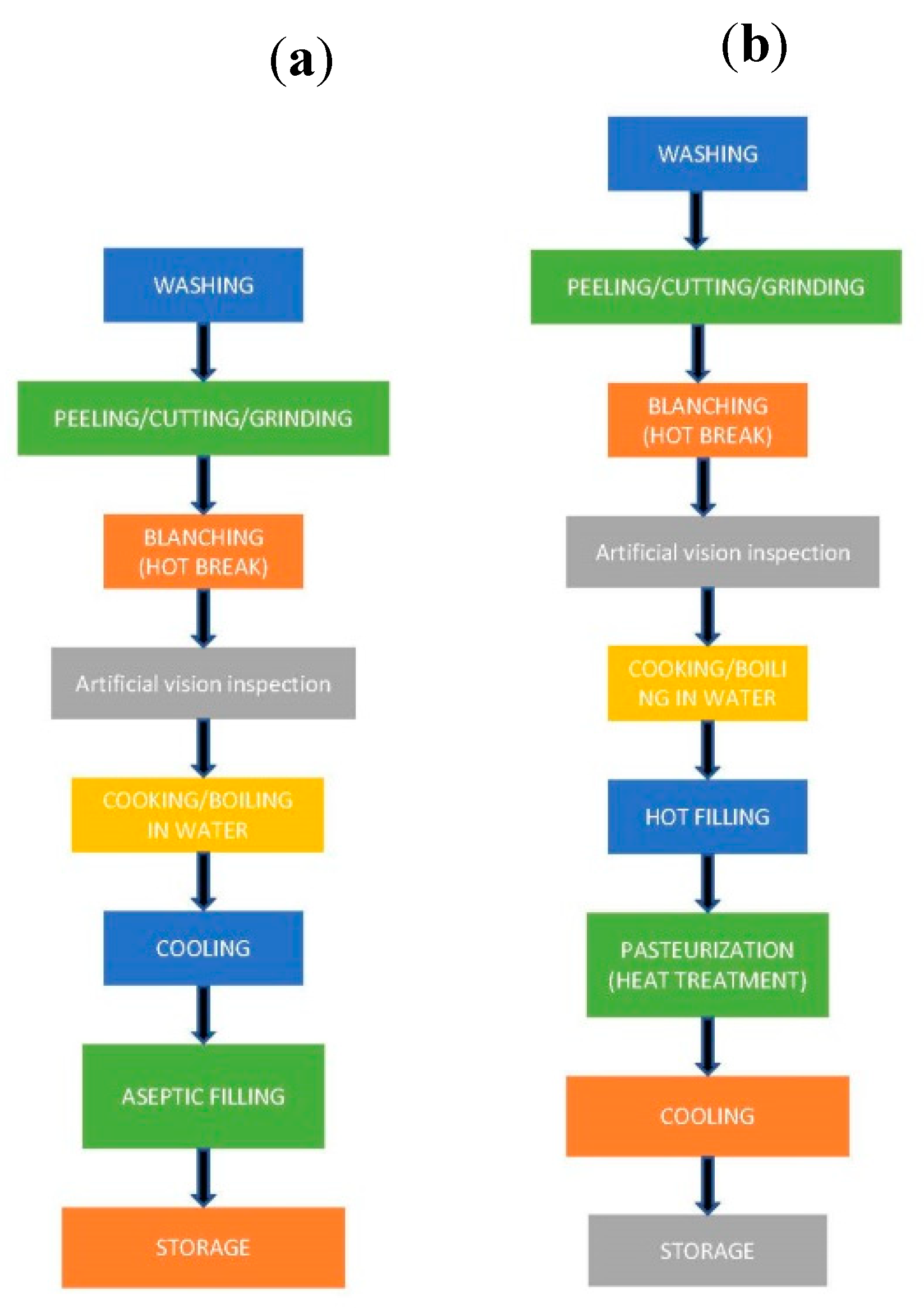
3. Processing Effects on the Main Components of Raw Materials
4. Health Benefits of the Intake of Vegetable Soups and Creams
4.1. Nutritional Properties
4.2. To Promote Body Hydration
4.3. Satiating Properties
5. New Trends in the Development of Vegetable Soups and Creams
5.1. Healthier Nutrition
- Substitution of animal protein by vegetable proteins: Overwhelming evidence shows that overconsumption of meat is bad not only for human health but also for environmental health and that moving towards a more plant-based diet is more sustainable [186]. This change is urgently needed to ensure One Health objectives (United Nations Sustainable Development Goals) for people and the planet are achieved [187]. The innovation opportunities in this sector include the substitution of thickened agents from animal origin (milk, cream, butter, meat, or fish broth, etc.) by others from vegetable origin such as certain legumes [151,152,188,189], ancient cereals [188,190], pseudocereals [191,192], seaweeds [193], and others.
- Salt reduction: In this aspect, it should not be forgotten that canned soup is one of the leading processed food categories containing high quantities of sodium. Although it has been suggested that intake of sodium rich foods in excess of immediate bodily needs is a preventative mechanism serving to embed sodium sources in memory and thus ward off hyponatremic challenge, nowadays much of the sodium that individuals and manufacturers place in foods is present because people “like” the taste of sodium rich food better than the taste of the same food without sodium [194,195]. WHO reported that high sodium consumption (>2 g/day, equivalent to 5 g salt/day) and insufficient potassium intake (less than 3.5 g/day) contribute to high blood pressure and increase the risk of heart disease and stroke. Most people consume too much salt—on average 9–12 g per day, or around twice the recommended maximum level of intake. In view of the severity of excess salt intake with foods the WHO Member States have agreed to reduce the global population’s intake of salt by a relative 30% by 2025. To achieve this goal, one of the proposed actions is that authorities have to work with the private sector to improve the availability and accessibility of low-salt products [196]. Several authors have studied the influence of salt taste threshold on acceptability and purchase intent of reformulated reduced sodium vegetable soups and the results highlighted the importance of both taste characteristics and the inclusion of the consumer in the reformulation process of reduced salt foods [197,198]. The study from Mitchell et al. [198] also showed that salt reductions of up to 48% can be achieved in commercial vegetable soup samples without affecting consumers liking for the meal.
- Sugar reduction: The sugar content in commercial vegetable soups can result surprising. For example, Gallager [199] reported amount of sugar up to 20 g per tube in commercial root and tomato vegetable soups. The WHO recommends people take no more than 24 g a day. Traditionally the addition of sugar to soups (mainly as isoglucose) has been made with the goal of making the soup tastier, reducing acid taste, or increasing creamy sensation, but it is not necessary if the balance of ingredients is correct [200]. In this sense, most of the new soup formulations are not added sugars.
- Fiber addition: Dietary fiber, which is not easily hydrolyzed by the human digestive system, reduces the risk of cardiovascular diseases, obesity, diabetes, and gastrointestinal disorders. It mainly consists of lignin and plant polysaccharides, and their physiological effect is determined by physical and chemical properties, such as the polysaccharide degree of polymerization and crosslinking, cell wall integrity, and particle size. Although vegetables soups are considered good sources of dietary fiber (depending on the main vegetable used in their formulation) [146], the deficit in the amount of dietary fiber in our current diet has led to an increase in the supply of fiber-enriched foods. Apart from the nutritional purpose, fiber can be used in vegetable soups for technological purposes as bulking or gelling agent or fat substitute [201]. Some of the ingredients used for this enrichment are quinoa grains (13–15% dietary fiber content, mainly insoluble dietary fiber) [202,203], legumes (7–10% dietary fiber content depending on the type) [189], and fiber extracts obtained from coproducts of vegetable-food processing [204,205,206]. These coproducts represent a major disposal problem for the industry and its transformation into value-added products, as fibers are, may contribute to diminish the problem and to recover valuable biomass and nutrients [204,205,206]. The use of fibers from new origins that are currently not fully exploited and the possibility of modifying the fibers by chemical, enzymatic and/or physical treatments will probably widen the fields of application for dietary fibers [207,208].
- Fat reduction: Consumer concerns about excess fat consumption of some types of lipids have led to the development by the food industry of a new category of foods to meet this demand. The development of reduced-fat foods with the same desirable attributes as the corresponding full-fat foods has created a distinct challenge to food manufacturers. In view to achieve this nutritional item, different vegetable proteins and starches have been applied as fat substitutes in soups. Several authors have reported that the water binding enhancement by whey proteins is important in reduced-fat and fat-free formulations to retain the increased water in the formula, and to replace fat and maintain texture and yield [209]. Whey proteins contribute to increased opacity and help reduced-fat soups and sauces retain a good visual appeal, with the creamy appearance of regular products. Whey proteins provide fat-like functions of lubricity and significantly improve the mouthfeel of low-fat soups and sauces. Emulsification properties of whey proteins aid in efficient dispersion of fat by forming interfacial membranes around oil globules that prevent creaming, coalescence, and oiling off [210,211]. In addition, whey proteins contribute to increased shelf-life in terms of product appearance and consumer acceptability. Interesting results have also been obtained using cross-linking tapioca starch (CLTS) as fat substitute in soups. Variations of swelling power, solubility, pasting, gelatinization, and rheological properties of the CLTS were found. Thermogravimetric analysis exhibited higher thermal stability for the CLTS granules compared to the native one. Among the samples, soup containing the 1.0%-CLTS exhibited the strongest gel characteristic and the greatest shear resistant properties. The 1.0%-CLTS improved the textural properties and sensory quality of soups [212]. Cox et al. [213] tried to characterize the changes in the drivers of liking when sodium and fat levels were varied in a model retorted soup system. They reported that formulation modifications that would decrease intensities of attributes that characterize lower fat and sodium soups, such as sour (taste and aftertaste), grainy (texture), and darkness (appearance), will aid in higher consumer acceptance of these soups.
5.2. High Added Value
5.3. More Natural (Clean Label, Organic Ingredients, Less Invasive Production Processes, Coproducts Valorization, etc.)
5.4. Shelf-Life Innovations
5.5. Products Aimed at Specific Population Groups
- 6.
- Adults can reduce their energy intake from the entire meal by consuming low-energy dense foods before the main course as a first course, increasing their satiety or even for diner due to its easy digestion which is positive for comforting and not unbalanced sleep. Regarding that, The Spanish Society of Communitarian Nutrition recommends the introduction of soups and creams at dinners [220].
- 7.
- Children’s vegetable consumption is generally far below the recommended standards, and effective strategies are required to support additional consumption [221]; attractive and nutritive soups and creams could increase their vegetable consumption.
- 8.
- Elderly people are encouraged to consume soups and creams four or more times a week because helps to achieve the essential fluid intake to meet daily requirements due to their high water content. In addition, their high vitamins and minerals content contributes to achieve others daily nutritional requirements, mainly in elderly [169].
- 9.
- Celiacs. Celiac disease is a major public health problem worldwide. Approximately 3 million people in Europe and another 3 million people in the United States are estimated to be affected by celiac disease [222]. Traditionally semi-prepared soups and creams are made of wheat (or another gluten-containing cereal) and texturizing agents to contribute to their creamy taste and texture because it has been reported that the absence of gluten leads to a low viscosity [223]. Several ingredients have been tested in order to make these food products suitable for celiacs, being quinoa flour, which showed the better results. Quinoa has a great ability to form emulsions, a high water holding capacity and high solubility. Its high amylopectin content contributes to its ability to reach the gelatinization temperature in less time than corn and in a similar time as wheat with lower retrogradation. This represents a good gel stability [224], which is difficult to achieve when wheat flour is used to produce semi-prepared soups and creams. Quinoa flour is an excellent ingredient for soups and creams not only for these technological properties but also for its nutritional value (50–60% carbohydrates, 7–10% dietetic fiber, 12–23% protein content with great amino acid balance, and vitamins [225].
- 10.
- Patients whose intake of solids is considerably reduced due to several pathological reasons. In this case, the formulation must be optimized in view of compensating the small food volumes with the highest nutrients and energy without misleading aspects such as an adequate texture that allows its correct deglutition [169].
Author Contributions
Funding
Conflicts of Interest
References
- Sheevani, T. Soups, Meaning and Classification. Your Article library.com. Available online: https://www.yourarticlelibrary.com/home-science/food-production/soup-meaning-and-classification-food-production/86444 (accessed on 6 September 2020).
- Bhg.com. Thickening Soups. Available online: https://www.bhg.com/recipes/how-to/cooking-techniques/thickening-soups/ (accessed on 9 June 2020).
- Smith, A.F. The Oxford Encyclopedia of Food and Drink in America, 2nd ed.; Oxford University Press: Oxford, UK, 2013; pp. 242–249. [Google Scholar]
- El Wakeel, M.A. Ultra-Structure and Functional Properties of Some Dry Mixes of Food. Master’s Thesis, Faculty of Agriculture, Ain Shams University, Cairo, Egypt, 2007. [Google Scholar]
- Osman, M.A.; El-Damaty, S.; Shaheen, A.; Ibrahim, M.M. Production of precooked dehydrated soups. Preparation of beef meat, vegetable soup and chemical, sensory and keeping quality evaluations. Egypt J. Food Sci. 1991, 19, 257–267. [Google Scholar]
- Rekha, M.N.; Yadav, A.R.; Dharmesh, S.; Chauhan, A.S.; Ramteke, R.S. Evaluation of antioxidant properties of dry soup mix extracts containing dill (Anethum sowa L.) leaf. Food Bioprocess. Technol. 2010, 3, 441–449. [Google Scholar] [CrossRef]
- Farzana, T.; Mohajan, S.; Saha, T.; Hossain, N.; Haque, Z. Formulation and nutritional evaluation of a healthy vegetable soup powder supplemented with soy flour, mushroom, and moringa leaf. Food Sci. Nutr. 2017, 5, 911–920. [Google Scholar] [CrossRef] [PubMed]
- De Cicco, A. The Fruit and Vegetable Sector in the EU—A Statistical Overview. 2019. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/The_fruit_and_vegetable_sector_in_the_EU_-_a_statistical_overview (accessed on 20 October 2020).
- MAPA (Ministerio de Agricultura, Pesca y Alimentación) Informe del Consumo Alimentario en España 2019; Ministerio de Agricultura, Pesca y Alimentación: Madrid, Spain, 2020.
- Roberts, W., Jr. Soup in the U.S. Mintel Group: London, England. June 2019. Available online: https://store.mintel.com/report/soup-us-june-2019 (accessed on 15 July 2020).
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef]
- Li, M.; Fan, Y.; Zhang, X.; Hou, W.; Tang, Z. Fruit and vegetable intake and risk of type 2 diabetes mellitus: Meta-analysis of prospective cohort studies. BMJ Open 2014, 4, e005497. [Google Scholar] [CrossRef]
- Jaiwal, A.K. Nutritional, Composition and Antioxidant Properties of Fruits and Vegetables; Academic Press: London, UK, 2020. [Google Scholar]
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–516. [Google Scholar] [CrossRef]
- Angelino, D.; Godos, J.; Ghelfi, F.; Tieri, M.; Titta, L.; Lafranconi, A.; Marventano, S.; Alonzo, E.; Gambera, A.; Sciacca, S.; et al. Fruit and vegetable consumption and health outcomes: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2019, 70, 652–667. [Google Scholar] [CrossRef]
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef]
- Lock, K.; Pomerleau, J.; Causer, L.; Altmann, D.R.; McKee, M. Global and regional burden of disease attributable to selected major risk factors. Bull. World Health Organ. 2005, 83, 100–108. [Google Scholar]
- Zhan, J.; Liu, Y.J.; Cai, L.B.; Xu, F.R.; Xie, T.; He, Q.Q. Fruit and vegetable consumption and risk of cardiovascular disease: A meta-analysis of prospective cohort studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 1650–1663. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G.; Kalle-Uhlmann, T.; Arregui, M.; Buijsse, B.; Boeing, H. Fruit and vegetable consumption and changes in anthropometric variables in adult populations: A systematic review and meta-analysis of prospective cohort studies. PLoS ONE 2015, 10, e0140846. [Google Scholar] [CrossRef] [PubMed]
- Holst, B.; Williamson, G. Nutrients and phytochemicals: From bioavailability to bioefficacy beyond antioxidants. Curr. Opin. Biotechnol. 2008, 19, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Bogusz, S.; Marco, P.H.; Valderrama, P.; Damasceno, F.C.; Aranda, M.S.; Zini, C.A.; Caramao, E.B.; Tavares Melo, A.M.; Filho, J.T.; Godoy, H.T. Analysys of volatile compounds in Capsicum spp. by headspace solid-phase microextraction and GC-TOFMS. J. Anal. Methods Chem. 2015, 22, 521–529. [Google Scholar]
- Mitic, V.D.; Cvetkovic, J.S.; Stankov-Jovanovic, V.P.; Dimitrijevic, M.V.; Stojanovic, G.S. Characterization of pepper genotypes from Serbia as a function of maturity by antioxidant activity with chemometric analysis. Anal. Lett. 2016, 49, 2234–2245. [Google Scholar] [CrossRef]
- de Sá Mendes, N.; Branco de Andrade Gonçalves, É.C. The role of bioactive components found in peppers. Trends Food Sci. Technol. 2020, 99, 229–243. [Google Scholar] [CrossRef]
- Rodrigues, C.A.; Nicácio, A.E.; Jardim, I.C.S.F.; Vsentainer, J.V.; Maldanera, L. Determination of phenolic compounds in red sweet pepper (Capsicum annuum L.) using a modified QuEChERS method and UHPLC-MS/MS analysis and its relation to antioxidant activity. J. Braz. Chem. Soc. 2019, 30, 1229–1240. [Google Scholar] [CrossRef]
- Carvalho, A.V.; de Andrade Mattietto, R.; de Oliveira Rios, A. Bioactive compounds and antioxidant activity of pepper (Capsicum spp.) genotypes. J. Food Sci. Technol. 2015, 52, 7457–7464. [Google Scholar] [CrossRef]
- Baenas, N.; Belović, M.; Ilic, N.; Moreno, D.A.; García-Viguera, C. Industrial use of pepper (Capsicum annum L.) derived products: Technological benefits and biological advantages. Food Chem. 2019, 274, 872–885. [Google Scholar] [CrossRef]
- Gürbüz, N.; Uluişik, S.; Frary, A.; Frary, A.; Doğanlar, S. Health benefits and bioactive compounds of eggplant. Food Chem. 2018, 268, 602–610. [Google Scholar] [CrossRef]
- Salerno, L.; Modica, M.N.; Pittalà, V.; Romeo, G.; Siracusa, M.A.; Di Giacomo, C.; Acquaviva, R. Antioxidant activity and phenolic content of microwave-assisted Solanum melongena extracts. Sci. World J. 2014, 719486. [Google Scholar] [CrossRef]
- Todaro, A.; Cimino, F.; Rapisarda, P.; Catalano, A.E.; Barbagallo, R.N.; Spagna, G. Recovery of anthocyanins from eggplant peel. Food Chem. 2009, 114, 434–439. [Google Scholar] [CrossRef]
- Ferarsa, S.; Zhang, W.; Moulai-Mostefa, N.; Ding, L.; Jaffrin, M.Y.; Grimi, N. Recovery of anthocyanins and other phenolic compounds from purple eggplant peels and pulps using ultrasonic-assisted extraction. Food Bioprod. Process. 2018, 109, 19–28. [Google Scholar] [CrossRef]
- García-Salas, P.; Gómez-Caravaca, A.M.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Identification and quantification of phenolic compounds in diverse cultivars of eggplant grown in different seasons by high-performance liquid chromatography coupled to diode array detector and electrospray-quadrupole-time of flight-mass spectrometry. Food Res. Int. 2015, 57, 114–122. [Google Scholar] [CrossRef]
- Sadilova, E.; Stintzing, F.C.; Carle, R. Anthocyanins, colour and antioxidant properties of eggplant (Solanum melongena L.) and violet pepper (Capsicum annuum L.) peel extracts. Z. Nat. C 2006, 61, 527–535. [Google Scholar] [CrossRef]
- Alarcón-Flores, M.I.; Romero-González, R.; Vidal, J.L.M.; Frenich, A.G. Multiclass determination of phytochemicals in vegetables and fruits by ultra-high-performance liquid chromatography coupled to tandem mass spectrometry. Food Chem. 2013, 141, 1120–1129. [Google Scholar] [CrossRef]
- Koley, T.K.; Tiwari, S.K.; Sarkar, A.; Nishad, J.; Goswami, A.; Singh, B. Antioxidant potential of indian eggplant: Comparison among white, purple and green genotypes using chemometrics. J. Agric. 2019, 8, 9–20. [Google Scholar] [CrossRef]
- Frond, A.D.; Luhas, C.I.; Stirbu, I.; Leopold, L.; Socaci, S.; Andreea, S.; Ayvaz, H.; Andreea, S.; Mihai, S.; Diaconeasa, Z.; et al. Phytochemical characterization of five edible purple-reddish vegetables: Anthocyanins, flavonoids, and phenolic acid derivatives. Molecules 2019, 24, 1536. [Google Scholar] [CrossRef]
- WPTC (World Producer Tomato Council). World Production Estimate of Tomatoes for Processing. 2014. Available online: http://www.wptc.to (accessed on 20 October 2020).
- Viuda-Martos, M.; Sánchez-Zapata, E.; Sayas-Barberá, E.; Sendra, E.; Pérez-Alvarez, J.A.; Fernández-López, J. Tomato and Tomato byproducts. Human health benefits of lycopene and its application to meat products: A review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1032–1049. [Google Scholar] [CrossRef]
- Frusciante, L.; Carli, P.; Ercolano, M.R.; Pernice, R.; Di Matteo, A.; Fogliano, V.; Pellegrini, N. Antioxidant nutritional quality of tomato. Mol. Nutr. Food Res. 2007, 51, 609–617. [Google Scholar] [CrossRef]
- Vallverdu-Queralta, A.; Arranz, S.; Medina-Remón, A.; Casals, I.; Lamuela-Raventós, R.M. Changes in phenolic content of tomato products during storage. J. Agric. Food Chem. 2011, 59, 9358–9365. [Google Scholar] [CrossRef]
- Giovanelli, G.; Lavelli, V.; Peri, C.; Nobili, S. Variation in antioxidant components of tomato during vine and post-harvest ripening. J. Sci. Food Agric. 1999, 79, 1583–1588. [Google Scholar] [CrossRef]
- Gómez-Romero, M.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Metabolite profiling and quantification of phenolic compounds in methanol extracts of tomato fruit. Phytochemistry 2010, 71, 1848–1864. [Google Scholar] [CrossRef] [PubMed]
- Sargent, S.A.; Maynard, D.N. Cucurbits. In Crop Post-Harvest: Science and Technology; Rees, D., Farrell, G., Orchard, J., Eds.; Wiley Publishing Online: Oxford, UK, 2012. [Google Scholar]
- Kulczyński, B.; Gramza-Michałowska, A. The profile of carotenoids and other bioactive molecules in various pumpkin fruits (Cucurbita maxima Duchesne) cultivars. Molecules 2019, 24, 3212. [Google Scholar] [CrossRef] [PubMed]
- Kulczyński, B.; Gramza-Michałowska, A. The profile of secondary metabolites and other bioactive compounds in Cucurbita pepo L. and Cucurbita moschata pumpkin cultivars. Molecules 2019, 24, 2945. [Google Scholar] [CrossRef] [PubMed]
- Priori, D.; Valduga, E.; Villela, J.C.B.; Mistura, C.C.; Vizzotto, M.; Valgas, R.A.; Barbieri, R.L. Characterization of bioactive compounds, antioxidant activity and minerals in landraces of pumpkin (Cucurbita moschata) cultivated in Southern Brazil. Food Sci. Technol. 2017, 37, 33–40. [Google Scholar] [CrossRef]
- Bemfeito, C.M.; Carneiro, J.d.S.; Carvalho, E.E.N.; Coli, P.C.; Pereira, R.C.; Vilas, E.V.d.B. Nutritional and functional potential of pumpkin (Cucurbita moschata) pulp and pequi peel flours. J. Food Sci. Technol. 2020, 57, 3920–3925. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Singh, V.; Shukla, S.; Rai, A.R. Phenolic content and antioxidant capacity of selected cucurbit fruits extracted with different solvents. J. Nutr. Food Sci. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Mzoughi, Z.; Chahdoura, H.; Chakroun, Y.; Cámara, M.; Fernández-Ruiz, V.; Morales, P.; Majdoub, H. Wild edible Swiss chard leaves (Beta vulgaris L. var. cicla): Nutritional, phytochemical composition and biological activities. Food Res. Int. 2019, 119, 612–621. [Google Scholar] [CrossRef]
- Pyo, Y.H.; Lee, T.C.; Logendra, L.; Rosen, R.T. Antioxidant activity and phenolic compounds of Swiss chard (Beta vulgaris subspecies cycla) extracts. Food Chem. 2004, 85, 19–26. [Google Scholar] [CrossRef]
- Maucieri, C.; Nicoletto, C.; Zanin, G.; Xiccato, G.; Borin, M.; Sambo, P. Composition and quality traits of vegetables grown in a low-tech aquaponic system at different fish stocking densities. J. Sci. Food Agric. 2020, 100, 4310–4318. [Google Scholar] [CrossRef]
- Gennari, L.; Felletti, M.; Blasa, M.; Angelino, D.; Celeghini, C.; Corallini, A.; Ninfali, P. Total extract of Beta vulgaris var. cicla seeds versus its purified phenolic components: Antioxidant activities and antiproliferative effects against colon cancer cells. Phytochem. Anal. 2011, 22, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Sacan, O.; Yanardag, R. Antioxidant and antiacetylcholinesterase activities of chard (Beta vulgaris L. var. cicla). Food Chem. Toxicol. 2010, 48, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Aehle, E.; Grandic, S.R.-L.; Ralainirina, R.; Baltora-Rosset, S.; Mesnard, F.; Prouillet, C.; Mazière, J.C.; Fliniaux, M.A. Development and evaluation of an enriched natural antioxidant preparation obtained from aqueous spinach (Spinacia oleracea) extracts by an adsorption procedure. Food Chem. 2004, 86, 579–585. [Google Scholar] [CrossRef]
- Roberts, J.L.; Moreau, R. Functional properties of spinach (Spinacia oleracea L.) phytochemicals and bioactives. Food Funct. 2016, 7, 3337–3353. [Google Scholar] [CrossRef]
- Chu, Y.H.; Sun, J.; Wu, X.; Liu, R.H. Antioxidant and antiproliferative activities of common vegetables. J. Sci. Food Agric. 2002, 50, 6910–6916. [Google Scholar] [CrossRef]
- Yosefi, Z.; Tabaraki, R.; Gharneh, H.A.A.; Mehrabi, A.A. Variation in antioxidant activity, total phenolics, and nitrate in spinach. Int. J. Veg. Sci. 2010, 16, 233–242. [Google Scholar] [CrossRef]
- Ko, S.H.; Park, J.H.; Kim, S.Y.; Lee, S.W.; Chun, S.S.; Park, E. Antioxidant effects of spinach (Spinacia oleracea L.) supplementation in hyperlipidemic Rats. Prev. Nutr. Food Sci. 2014, 19, 19–26. [Google Scholar] [CrossRef]
- Sánchez Maldonado, A.; Mudge, E.; Gänzle, M.; Schieber, A. Extraction and fractionation of phenolic acids and glycoalkaloids from potato peels using acidified water/ethanol-based solvents. Food Res. Int. 2014, 65, 27–34. [Google Scholar] [CrossRef]
- Habeebullah, S.F.K.; Grejsen, H.D.; Jacobsen, C. Potato peel extract as a natural antioxidant in chilled storage of minced horse mackerel (Trachurus trachurus): Effect on lipid and protein oxidation. Food Chem. 2012, 131, 843–851. [Google Scholar]
- Blessington, T.; Nzaramba, M.; Scheuring, D.; Hale, A.; Reddivari, L.; Miller, J. Cooking methods and storage treatments of potato: Effects on carotenoids, antioxidant activity, and phenolics. Am. J. Potato Res. 2010, 87, 479–491. [Google Scholar] [CrossRef]
- Navarre, D.; Pillai, S.; Shakya, R.; Holden, M. HPLC Profiling of phenolics in diverse potato genotypes. Food Chem. 2011, 127, 34–41. [Google Scholar] [CrossRef]
- Oertel, A.; Matros, A.; Hartmann, A.; Arapitsas, P.; Dehmer, K.J.; Martens, S.; Mock, H.P. Metabolite profiling of red and blue potatoes revealed cultivar and tissue specific patterns for anthocyanins and other polyphenols. Plants 2017, 246, 281–297. [Google Scholar] [CrossRef] [PubMed]
- André, C.M.; Oufir, M.; Guignard, C.; Hoffmann, L.; Hausman, J.F.; Evers, D.; Larondelle, Y. Antioxidant profiling of native Andean potato tubers (Solanum tuberosum L.) reveals cultivars with high levels of beta-carotene, alpha-tocopherol, chlorogenic acid, and petanin. J. Agric. Food Chem. 2007, 55, 10839–10849. [Google Scholar] [CrossRef] [PubMed]
- Bonierbale, M.; Grüneberg, W.; Amoros, W.; Burgos, G.; Salas, E.; Porras, E.; zum Felde, T. Total and individual carotenoid profiles in Solanum phureja cultivated potatoes: II. Development and application of near-infrared reflectance spectroscopy (NIRS) calibrations for germplasm characterization. J. Food Comp. Anal. 2009, 22, 509–516. [Google Scholar] [CrossRef]
- Hamouz, K.; Lachman, J.; Pazderů, K.; Tomášek, J.; Hejtmánková, K.; Pivec, V. Differences in anthocyanin content and antioxidant activity of potato tubers with different flesh colour. Plant Soil Environ. 2011, 57, 478–485. [Google Scholar] [CrossRef]
- Sasaki, K.; Han, J.; Shimozono, H.; Villareal, M.O.; Isoda, H. Caffeoylquinic acid-rich purple sweet potato extract, with or without anthocyanin, imparts neuroprotection and contributes to the improvement of spatial learning and memory of SAMP8 mouse. J. Agric. Food Chem. 2013, 61, 5037–5045. [Google Scholar] [CrossRef] [PubMed]
- Grace, M.H.; Yousef, G.G.; Gustafson, S.J.; Truong, V.D.; Yencho, G.C.; Lila, M.A. Phytochemical changes in phenolics, anthocyanins, ascorbic acid, and carotenoids associated with sweet potato storage and impacts on bioactive properties. Food Chem. 2014, 145, 717–724. [Google Scholar] [CrossRef]
- Lim, S.; Xu, J.; Kim, J.; Chen, T.Y.; Su, X.; Standard, J.; Carey, E.; Griffin, J.; Herndon, B.; Katz, B.; et al. Role of anthocyanin-enriched purple-fleshed sweet potato p40 in colorectal cancer prevention. Mol. Nutr. Food Res. 2013, 57, 1908–1917. [Google Scholar] [CrossRef]
- Esatbeyoglu, T.; Rodríguez-Werner, M.; Schlösser, A.; Winterhalter, P.; Rimbach, G. Fractionation, enzyme inhibitory and cellular antioxidant activity of bioactives from purple sweet potato (Ipomoea batatas). Food Chem. 2017, 221, 447–456. [Google Scholar] [CrossRef]
- Ishiguro, K.; Yoshinaga, M.; Kai, Y.; Maoka, T.; Yoshimoto, M. Composition, content and antioxidative activity of the carotenoids in yellow-fleshed sweet potato (Ipomoea batatas L.). Breed Sci. 2010, 60, 324–329. [Google Scholar] [CrossRef]
- Teow, C.C.; Truong, V.D.; McFeeters, R.F.; Thompson, R.L.; Pecota, K.V.; Yencho, C.G. Antioxidant activities, phenolic and β-carotene contents of sweet potato genotypes with varying flesh colours. Food Chem. 2007, 103, 829–838. [Google Scholar] [CrossRef]
- Ji, H.; Zhang, H.; Li, H.; Li, Y. Analysis on the nutrition composition and antioxidant activity of different types of sweet potato cultivars. Food Nutr. Sci. 2015, 6, 161–167. [Google Scholar] [CrossRef]
- Clementz, A.; Torresi, P.A.; Molli, J.S.; Cardell, D.; Mammarella, E.; Yori, J.C. Novel method for valorization of by-products from carrot discards. LW—Food Sci. Technol. 2019, 100, 374–380. [Google Scholar] [CrossRef]
- Nicolle, C.; Simon, G.; Rock, E.; Amouroux, P.; Rémésy, C. Genetic variability influences carotenoid, vitamin, phenolic, and mineral content in white, yellow, purple, orange, and dark-orange carrot cultivars. J. Am. Soc. Hortic. Sci. 2004, 129, 523–529. [Google Scholar] [CrossRef]
- Dias, J.S. Major classes of phytonutraceuticals in vegetables and health benefits: A review. J. Nutr. Therapeut. 2012, 1, 31–62. [Google Scholar]
- Narayan, M.S.; Venkataraman, L.V. Characterisation of anthocyanins derived from carrot (Daucus carota) cell culture. Food Chem. 2000, 70, 361–363. [Google Scholar] [CrossRef]
- Montilla, E.C.; Arzaba, M.R.; Hillebrand, S.; Winterhalter, P. Anthocyanin composition of black carrot (Daucus carota ssp. sativus var. atrorubens Alef.) cultivars Antonina, Beta Sweet, Deep Purple, and Purple Haze. J. Agric. Food Chem. 2011, 59, 3385–3390. [Google Scholar] [CrossRef]
- Zhang, D.; Hamauzu, Y. Phenolic compounds and their antioxidant properties in different tissues of carrots (Daucus carota L.). J. Food Agric. Environ. 2004, 2, 95–100. [Google Scholar]
- Dong, R.; Yu, Q.; Liao, W.; Liu, S.; He, Z.; Hu, X.; Chen, Y.; Xie, J.; Nie, S.; Xie, M. Composition of bound polyphenols from carrot dietary fiber and its in vivo and in vitro antioxidant activity. Food Chem. 2021, 339, 127879. [Google Scholar] [CrossRef]
- Algarra, M.; Fernandes, A.; Mateus, N.; de Freitas, V.; Esteves da Silva, J.C.G.; Casado, J. Athocyanin profile and antioxidant capacity of black carrots (Daucus carota L. ssp. sativus var. atrorubens Alef.) from Cuevas Bajas, Spain. J. Food Comp. Anal. 2014, 33, 71–76. [Google Scholar] [CrossRef]
- Lanzotti, V. The analysis of onion and garlic. J. Chromatograph. A 2006, 1112, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Metrani, R.; Singh, J.; Acharya, P.; Acharya, P.; Jayaprakasha, G.K. Comparative Metabolomics Profiling of polyphenols, nutrients and antioxidant activities of two red onion (Allium cepa L.) cultivars. Plants 2020, 9, 1077. [Google Scholar] [CrossRef] [PubMed]
- Lombard, K.A.; Geoffriau, E.; Peffley, E. Flavonoid quantification in onion by spectrophotometric and high-performance liquid chromatography analysis. Hort. Sci. 2002, 37, 682–685. [Google Scholar] [CrossRef]
- Panche, A.; Diwan, A.; Chandra, S. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Mojzer, B.E.; Hrnčič, M.K.; Škerget, M.; Knez, Z.; Bren, U. Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef] [PubMed]
- Akdeniz, B.; Sumnu, G.; Sahin, S. Microencapsulation of phenolic compounds extracted from onion (Allium cepa) skin. J. Food Process. Preserv. 2018, 42, e13648. [Google Scholar] [CrossRef]
- Bystricka, J.; Musilova, J.; Vollmannova, A.; Timoracka, M.; Kavalcova, P. Bioactive components of onion (Allium cepa L.)—A review. Acta Aliment. 2013, 42, 11–22. [Google Scholar] [CrossRef]
- Yang, J.; Meyers, K.J.; Van der Heide, J.; Liu, R.H. Varietal differences in phenolic content and antioxidant and antiproliferative activities of onions. J. Agric. Food Chem. 2004, 52, 6787–6793. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Park, S.W. Total phenolics, antioxidant and xanthine oxidase inhibitory activity of three coloured onions (Allium cepa L.). Front Life Sci. 2013, 7, 224–228. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, C.; Feng, B.; Chen, B.; Jin, L.; Shen, Y. UPLC-ESI-MS/MS based identification and antioxidant, antibacterial, cytotoxic activities of aqueous extracts from storey onion (Allium cepa L. var. proliferum Regel). Food Res. Int. 2020, 130, 108969. [Google Scholar] [CrossRef]
- Zill-E-Huma, H.; Vian, M.A.; Fabiano-Tixier, A.-S.; Elmaataoui, M.; Dangles, O.; Chemat, F. A remarkable influence of microwave extraction: Enhancement of antioxidant activity of extracted onion varieties. Food Chem. 2011, 127, 1472–1480. [Google Scholar] [CrossRef]
- Kovarovič, J.; Bystrická, J.; Fehér, A.; Lenková, M. Evaluation and comparison of bioactive substances in selected species of the genus Allium. Potravinarstvo 2017, 11, 702–708. [Google Scholar] [CrossRef][Green Version]
- Termine, E.; Lairon, D.; Taupier-Letage, B.; Gautier, S.; Lafont, R.; Lafont, H. Yield and content in nitrates, minerals and ascorbic acid of leeks and turnips grown under mineral or organic nitrogen fertilization. Plant Foods Hum. Nutr. 1987, 37, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Radovanović, B.; Mladenović, J.; Radovanović, A.; Pavlović, R.; Nikolić, V. Phenolic composition, antioxidant, antimicrobial and cytotoxic activites of Allium porrum L. (Serbia) extracts. J. Food Nutr. Res. 2015, 3, 564–569. [Google Scholar]
- Donna, L.D.; Mazzotti, F.; Taverna, D.; Napoli, A.; Sindona, G. Structural characterisation of malonyl flavonols in leek (Allium porrum L.) using high-performance liquid chromatography and mass spectrometry. Phytochem. Anal. 2014, 25, 207–212. [Google Scholar] [CrossRef]
- Bernaert, N.; De Loose, M.; Van Droogenbroeck, B. Antioxidant capacity of different leek types and the comparison with its related species. Acta Hort. 2012, 969, 25–232. [Google Scholar] [CrossRef]
- Bernaert, N.; Wouters, D.; De Vuyst, L.; De Paepe, D.; De Clercq, H.; Van Bockstaele, E.; De Loose, M.; Van Droogenbroeck, B. Antioxidant changes of leek (Allium ampeloprasum var. porrum) during spontaneous fermentation of the white shaft and green leaves. J. Sci. Food Agric. 2013, 93, 2146–2153. [Google Scholar] [CrossRef]
- Kavalcová, P.; Bystrická, J.; Tomáš, J.; Karovičová, J.; Kuchtová, V. Evaluation and comparison of the content of total polyphenols and antioxidant activity in onion, garlic and leek. Potravinarstvo 2014, 8, 272–276. [Google Scholar] [CrossRef]
- Rouf, R.; Uddin, S.J.; Sarker, D.K.; Islam, M.T.; Ali, E.S.; Shilpi, J.A.; Sarker, S.D. Antiviral potential of garlic (Allium sativum) and its organosulfur compounds: A systematic update of pre-clinical and clinical data. Trends Food Sci. Technol. 2020, 104, 219–234. [Google Scholar] [CrossRef]
- Chekki, R.Z.; Snoussi, A.; Hamrouni, I.; Bouzouita, N. Chemical composition, antibacterial and antioxidant activities of Tunisian garlic (Allium sativum) essential oil and ethanol extract. Med. J. Chem. 2014, 3, 947–956. [Google Scholar]
- Fei, M.L.I.; Tong, L.I.; Wei, L.I.; De Yang, L. Changes in antioxidant capacity, levels of soluble sugar, total polyphenol, organosulfur compound and constituents in garlic clove during storage. Ind. Crops Prod. 2015, 69, 137–142. [Google Scholar] [CrossRef]
- Liu, J.; Ji, F.; Chen, F.M.; Guo, W.; Yang, M.L.; Huang, S.X.; Zhang, F.; Liu, Y.S. Determination of garlic phenolic compounds using supercritical fluid extraction coupled to supercritical fluid chromatography/tandem mass spectrometry. J. Pharm. Biomed. Anal. 2018, 159, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Gorinstein, S.; Park, Y.S.; Heo, B.G.; Namiesnik, J.; Leontowicz, H.; Leontowicz, M.; Kang, S.G. A comparative study of phenolic compounds and antioxidant and antiproliferative activities in frequently consumed raw vegetables. Eur. Food Res. Technol. 2009, 228, 903–911. [Google Scholar] [CrossRef]
- Bozin, B.; Mimica-Dukic, N.; Samojlik, I.; Goran, A.; Igic, R. Phenolics as antioxidants in garlic (Allium sativum L., Alliaceae). Food Chem. 2008, 111, 925–929. [Google Scholar] [CrossRef]
- Nuutila, A.M.; Puupponen-Pimiä, R.; Aarni, M.; Oksman-Caldentey, K.M. Comparison of antioxidant activities of onion and garlic extracts by inhibition of lipid peroxidation and radical scavenging activity. Food Chem. 2003, 81, 485–493. [Google Scholar] [CrossRef]
- Shang, A.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Tang, G.Y.; Corke, H.; Li, H.B. Bioactive compounds and biological functions of garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ragaee, S.; Marcone, M.F.; Abdel-Aal, E.M. Composition of phenolic acids and antioxidant properties of selected pulses cooked with different heating conditions. Foods 2020, 9, 908. [Google Scholar] [CrossRef]
- Roy, F.; Boye, J.I.; Simpson, B.K. Bioactive proteins and peptides in pulse crops: Pea, chickpea and lentil. Food Res. Int. 2010, 43, 432–442. [Google Scholar] [CrossRef]
- Iqbal, A.; Khalil, I.A.; Ateeq, N.; Khan, M.S. Nutritional quality of important food legumes. Food Chem. 2006, 97, 331–335. [Google Scholar] [CrossRef]
- Benmeziane-Derradji, F.; Djermoune-Arkoub, L.; Ayat, N.E.H. Impact of roasting on the physicochemical, functional properties, antioxidant content and microstructure changes of Algerian lentil (Lens culinaris) flour. J. Food Meas. Charact. 2020, 14, 2840–2853. [Google Scholar] [CrossRef]
- Xu, B.; Chang, S.K. Phenolic substance characterization and chemical and cell-based antioxidant activities of 11 lentils grown in the Northern United States. J. Agric. Food Chem. 2010, 58, 1509–1517. [Google Scholar] [CrossRef]
- Amarowicz, R.; Estrella, I.; Hernández, T.; Duenas, M.; Troszynska, A.; Kosinska, A.; Pegg, R.B. Antioxidant activity of a red lentil extract and its fractions. Int. J. Mol. Sci. 2009, 10, 5513–5527. [Google Scholar] [CrossRef] [PubMed]
- Amarowicz, R.; Estrella, I.; Hernandez, T.; Robredo, S.; Troszynska, A.; Kosinska, A.; Pegg, R.B. Free radical-scavenging capacity, antioxidant activity, and phenolic composition of green lentil (Lens culinaris). Food Chem. 2010, 121, 705–711. [Google Scholar] [CrossRef]
- Thompson, L.U.; Boucher, B.A.; Liu, Z.; Cotterchio, M.; Kreiger, N. Phytoestrogen content of foods consumed in Canada, including isoflavones, lignans, and coumestan. Nutr. Cancer 2006, 54, 184–201. [Google Scholar] [CrossRef] [PubMed]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R.; O’Brien, N.M. Phytosterol, squalene, tocopherol content and fatty acid profile of selected seeds, grains, and legumes. Plant Foods Hum. Nutr. 2007, 62, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Alshikh, N.; Costa de Camargo, A.; Shahidi, F. Phenolics of selected lentil cultivars: Antioxidant activities and inhibition of low-density lipoprotein and DNA damage. J. Funct. Foods 2015, 18, 1022–1038. [Google Scholar] [CrossRef]
- Guzmán-Maldonado, S.H.; Acosta-Gallegos, J.A.; Álvarez, M.; García, D.; Loarca, P. Calidad alimentaria y potencial nutracéutico del frijol (Phaseolus vulgaris L.). Agric. Tec. Mex. Agric. Técnica México 2002, 28, 159–173. [Google Scholar]
- Dinelli, G.; Bonetti, A.; Minelli, M.; Marotti, I.; Catizone, P.; Mazzanti, A. Content of flavonols in Italian bean (Phaseolus vulgaris L.) ecotypes. Food Chem. 2006, 99, 105–114. [Google Scholar] [CrossRef]
- Lopéz, A.; El-Naggar, T.; Duenas, M.; Ortega, T.; Estrella, I.; Hernández, T.; Carretero, E.M. Effect of cooking and germination on phenolic composition and biological properties of dark beans (Phaseolus vulgaris L.). Food Chem. 2013, 138, 547–555. [Google Scholar] [CrossRef]
- Chen, P.X.; Dupuis, J.H.; Marcone, M.F.; Pauls, P.K.; Liu, R.; Liu, Q.; Tsao, R. Physicochemical properties and in vitro digestibility of cooked regular and nondarkening cranberry beans (Phaseolus vulgaris L.) and their effects on bioaccessibility, phenolic composition, and antioxidant activity. J. Agric. Food Chem. 2015, 63, 10448–10458. [Google Scholar] [CrossRef]
- Nikmaram, N.; Dar, B.N.; Roohinejad, S.; Koubaa, M.; Barba, F.J.; Greiner, R.; Johnson, S.K. Recent advances in γ-aminobutyric acid (GABA) properties in pulses: An overview. J. Sci. Food Agric. 2017, 97, 2681–2689. [Google Scholar] [CrossRef]
- Boye, J.; Zare, F.; Pletch, A. Pulse proteins: Processing, characterization, functional properties and applications in food and feed. Food Res. Int. 2010, 43, 414–431. [Google Scholar] [CrossRef]
- Jukanti, A.K.; Gaur, P.M.; Gowda, C.L.L.; Chibbar, R.N. Nutritional quality and health benefits of chickpea (Cicer arietinum L.): A review. Br. J. Nutr. 2012, 108, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Champ, M.J.M. Non-nutrient bioactive substances of pulses. Br. J. Nutr. 2002, 88 (Suppl. 3), S307–S319. [Google Scholar] [CrossRef] [PubMed]
- Quintero-Soto, M.F.; Saracho-Peña, A.G.; Chavez-Ontiveros, J. Phenolic profiles and their contribution to the antioxidant activity of selected chickpea genotypes from Mexico and ICRISAT collections. Plant Foods Hum. Nutr. 2018, 73, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, Y.; Dueñas, M.; Estrella, I.; Hernández, M.T.; Benitez, V.; Esteban, R.M.; Martín-Cabrejas, M.A. Phenolic profile and antioxidant capacity of chickpeas (Cicer arietinum L.) as affected by a dehydration process. Plant Foods Hum. Nutr. 2011, 66, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Rashmi, H.B.; Negi, P.S. Phenolic acids from vegetables: A review on processing stability and health benefits. Food Res. Int. 2020, 136, 109298. [Google Scholar] [CrossRef]
- Pellegrini, N.; Chiavaro, E.; Gardana, C.; Mazzeo, T.; Contino, D.; Gallo, M.; Riso, P.; Fogliano, V.; Porrini, M. Effect of different cooking methods on color, phytochemical concentration, and antioxidant capacity of raw and frozen brassica vegetables. J. Agric. Food Chem. 2010, 58, 4310–4321. [Google Scholar] [CrossRef]
- Jeż, M.; Błaszczak, W.; Zielińska, D.; Wiczkowski, W.; Białobrzewski, I. Carotenoids and lipophilic antioxidant capacities of tomato purées as affected by high hydrostatic pressure processing. Int. J. Food Sci. Technol. 2020, 55, 65–73. [Google Scholar] [CrossRef]
- Zaro, M.J.; Vicente, A.R.; Ortiz, C.M.; Chaves, A.R.; Concellón, A. Eggplant. In Handbook of Vegetable Preservation and Processing; Huy, Y.H., Evranuz, Ö., Eds.; Taylor & Francys: Abingdon, VA, USA, 2015; Chapter 21; pp. 479–595. [Google Scholar]
- Barbagallo, R.; Chisari, M.; Caputa, G. Effects of calcium citrate and ascorbate as inhibitors of browning and softening in minimally processed ‘Birgah’ eggplants. Postharvest Biol. Technol. 2012, 73, 107–114. [Google Scholar] [CrossRef]
- Kaur, R.; Kaur, K. Effect of processing on color, rheology and bioactive compounds of different sweet pepper purees. Plant Foods Hum. Nutr. 2020, 75, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M.T.K.; Tribst, A.A.L.; Pedro, E.D.; Augusto, P.E.D. High pressure homogenization in fruit and vegetable juice and puree. Processing: Effects on quality, stability and phytochemical profile. Innov. Food Process. Technol. 2021, 3, 1–7. [Google Scholar]
- Sergio, L.; Boari, F.; Pieralice, M.; Linsalata, V.; Cantore, V.; Di Venere, D. Bioactive phenolics and antioxidant capacity of some wild edible greens as affected by different cooking treatments. Foods 2020, 9, 1320. [Google Scholar] [CrossRef] [PubMed]
- Miglio, C.; Chiavaro, E.; Visconti, A.; Fogliano, V.; Pellegrini, N. Effects of different cooking methods on nutritional and physicochemical characteristics of selected vegetables. J. Agric. Food Chem. 2008, 56, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Arias-Rico, J.; Macías-León, F.J.; Alanís-García, E.; Cruz-Cansino, N.; Jaramillo-Morales, O.A.; Barrera-Gálvez, R.; Ramírez-Moreno, E. Study of edible plants: Effects of boiling on nutritional, antioxidant, and physicochemical properties. Foods 2020, 9, 599. [Google Scholar] [CrossRef] [PubMed]
- Provesi, J.G.; Dias, C.O.; Amante, E.R. Changes in carotenoids during processing and storage of pumpkin puree. Food Chem. 2011, 128, 195–202. [Google Scholar] [CrossRef]
- Sungpuag, P.; Tangchitpianvit, S.; Chittchang, U.; Wasantwisut, E. Retinol and beta carotene content of indigenous raw and homeprepared foods in Northeast Thailand. Food Chem. 1999, 64, 163–167. [Google Scholar] [CrossRef]
- Song, L.; Thornalley, P.J. Effect of storage, processing and cooking on glucosinolate content of Brassica vegetables. Food Chem. Toxic. 2007, 45, 216–224. [Google Scholar] [CrossRef]
- Galgano, F.; Favati, F.; Caruso, M.; Pietrafesa, A.; Natella, S. The influence of processing and preservation on the retention of health-promoting compounds in broccoli. J. Food Sci. 2007, 72, S130–S135. [Google Scholar] [CrossRef]
- Volden, J.; Borge, G.I.A.; Bengtsson, G.B.; Hansen, M.; Thygesen, I.E.; Wicklund, T. Effect of thermal treatment on glucosinolates and antioxidant-related parameters in red cabbage (Brassica oleracea L. ssp. capitata f. rubra). Food Chem. 2008, 109, 595–605. [Google Scholar] [CrossRef]
- Martínez, S.; Armesto, J.; Gómez-Limia, L.; Carballo, J. Impact of processing and storage on the nutritional and sensory properties and bioactive components of Brassica spp. A review. Food Chem. 2020, 313, 126065. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.A.; Rehab, F.M.A. Bioactive compounds and antioxidant activity of fresh and processed white cauliflower. Biomed. Res. Int. 2013, 7, 367819. [Google Scholar]
- Alam, M.K.; Sams, S.; Rana, Z.; Akhtaruzzamana, M.; Islam, S.N. Minerals, vitamin C, and effect of thermal processing on carotenoids composition in nine varieties orange-fleshed sweet potato (Ipomoea batatas L.). J. Food Compos. Anal. 2020, 92, 103582. [Google Scholar] [CrossRef]
- Dutta, D.; Dutta, A.; Raychaudhuri, U.; Chakraborty, R. Rheological characteristics and thermal degradation kinetics of β -carotene in pumpkin puree. J. Food Eng. 2006, 76, 538–546. [Google Scholar] [CrossRef]
- Gaur, S.; Shivhare, U.S.; Sarkar, B.C.; Ahmed, J. Thermal chlorophyll degradation kinetics of mint leaves puree. Int. J. Food Prop. 2007, 10, 853–865. [Google Scholar] [CrossRef]
- Arevalo, I.; Guzmán-Maldonado, S.H.; Sanchez, S.M.M.; Acosta-Gallegos, J.A. Steaming and toasting reduce the nutrimental quality, total phenols and antioxidant capacity of fresh kabuli chickpea (Cicer arietinum L.). Plant Foods Hum. Nutr. 2020, 75, 628–634. [Google Scholar] [CrossRef]
- Liu, K.; Zheng, J.; Wang, X.; Chen, F. Effects of household cooking processes on mineral, vitamin B, and phytic acid contents and mineral bioaccessibility in rice. Food Chem. 2019, 280, 59–64. [Google Scholar] [CrossRef]
- Grundya, M.M.L.; Momanyib, D.K.; Hollanda, C.; Kawakac, F.; Malinda-Salimd, S.T.; Boydd, B.J.; Bajkae, B.; Mulet-Caberof, A.I.; Bishop, J.; Owinob, W.O. Effects of grain source and processing methods on the nutritional profile and digestibility of grain amaranth. J. Funct. Foods 2020, 72, 104065. [Google Scholar] [CrossRef]
- Wu, W.C.; Chen, C.Y. Weight loss induced by whole grain-rich diet is through a gut microbiota-independent mechanism. World J. Diabetes 2020, 11, 6969707. [Google Scholar] [CrossRef]
- Abdel-Haleem, A.M.; Omran, A.A. Preparation of dried vegetarian soup supplemented with some legumes. Food Nutr. Sci. 2014, 5, 2274–2285. [Google Scholar] [CrossRef]
- van Buren, L.V.; Grün, C.H.; Basendowski, S.; Spraul, M.; Newson, R.; Eilander, A. Nutritional quality of dry vegetable soups. Nutrients 2019, 11, 1270. [Google Scholar] [CrossRef] [PubMed]
- CDCP (Center for Disease Control and Prevention). CDCP Guide to Strategies to Increase the Consumption of Fruits and Vegetables; U.S. Department of Health and Human Services: Atlanta, GA, USA, 2011.
- EFSA (European Food Science Agency). Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- Manhivi, V.E.; Sultanbawa, Y.; Sivajumar, D. Enhancement of the phytonutrient content of a gluten-free soup using a composite of vegetables. Int. J. Food Prop. 2020, 23, 1051–1065. [Google Scholar] [CrossRef]
- Paterson, E.; Gordon, M.H.; Niwat, C.; George, T.W.; Parr, L.; Waroonphan, S.; Lovegrove, J.A. Supplementation with fruit and vegetable soups and beverages increases plasma carotenoid concentrations but does not alter markers of oxidative stress or cardiovascular risk factors. J. Nutr. 2006, 136, 2849–2855. [Google Scholar] [CrossRef] [PubMed]
- Dolan, L.C.; Matulka, R.A.; Burdock, G.A. Naturally occurring food toxins. Toxins 2010, 9, 2289–2332. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Koh, W.Y.; Paek, W.K.; Lim, J. The sources of chemical contaminants in food and their health implications. Front. Pharmacol. 2017, 8, 830. [Google Scholar] [CrossRef]
- Adornetto, A.; Pagliara, V.; Di Renzo, G.; Arcone, R. Polychlorinated biphenyls impair dibutyryl cAMP-induced astrocytic differentiation in rat C6 glial cell line. FEBS Open Bio 2013, 3, 459–466. [Google Scholar] [CrossRef]
- Tuck, C.J.; Biesikierski, J.R.; Schmid-Grendelmeier, P.; Pohl, D. Food intolerances. Nutrients 2019, 11, 1684. [Google Scholar] [CrossRef]
- Abdel-Moemin, A.R. Consumer satisfaction and nutrient profile of reformulated dry soups. Agricultura 2014, 11, 27–34. [Google Scholar]
- Trevijano. Comercial Soups. 2020. Available online: https://www.trevijano.com/producto/sopa-juliana/ (accessed on 1 September 2020).
- BADALI. Base de Datos de Alimentos. 2020. Available online: https://badali.umh.es (accessed on 4 November 2020).
- EFSA (European Food Safety Authority). Dietary Reference Values for Nutrients Summary Report. EFSA Supporting Publ. 2017, 14, e15121. [Google Scholar]
- WHO (World Health Organization). Global Strategy on Diet, Physical Activity and Health. 2004. Available online: https://apps.who.int/iris/handle/10665/43035 (accessed on 10 October 2020).
- Gilham, B.; Hall, R.; Woods, J.L. Vegetables and legumes in new Australasian food launches: How are they being used and are they a healthy choice? Nutr. J. 2018, 17, 104. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, F.M.; Visentainer, J.V.; de Souza, N.E. Proximate composition and quantification of fatty acids in major brazilian brands of margarine and vegetable creams marketed in brazil. Braz. J. Food Nutr. 2011, 22, 181–189. [Google Scholar]
- Benelam, B.; Wyness, L. Hydration and health: A review. Nutr. Bull. 2010, 35, 3–25. [Google Scholar] [CrossRef]
- Nieto, G.; Ros, G. Key nutritional aspects for the design of soups, creams and sauces. Technifood 2020, 130, 78–81. [Google Scholar]
- EFSA (European Food Science Agency). Panel on Dietetic Products, Nutrition, and Allergies (NDA). Opinion on Dietary reference values for water. EFSA J. 2010, 8, 1459. [Google Scholar]
- Chambers, L.; McCrickerd, K.; Yeomans, M.R. Optimising foods for satiety. Trends Food Sci. Technol. 2015, 41, 149–160. [Google Scholar] [CrossRef]
- Vincente, A.R.; Manganaris, G.A.; Ortiz, C.M.; Sozzi, G.O.; Crisosto, C.H. Nutritional Quality of Fruits and Vegetables. In Postharvest Handling; Academic Press: Oxford, UK, 2014; pp. 69–122. [Google Scholar]
- Bertrais, S.; Galan, P.; Renault, N.; Zarebska, M.; Preziosi, P.; Hercberg, S. Consumption of soup and nutritional intake in French adults: Consequences for nutritional status. J. Hum. Nutr. Diet. 2001, 14, 121–128. [Google Scholar] [CrossRef]
- Moreira, P.; Padrão, P. Educational, economic and dietary determinants of obesity in Portuguese adults: A cross-sectional study. Eat. Behav. 2006, 7, 220–228. [Google Scholar] [CrossRef]
- Kuroda, M.; Ohta, M.; Okufuji, T.; Takigami, C.; Eguchi, M.; Hayabuchi, H.; Ikeda, M. Frequency of soup intake is inversely associated with body mass index, waist circumference, and waist-to-hip ratio, but not with other metabolic risk factors in Japanese men. J. Acad. Nutr. Diet. 2011, 111, 137–142. [Google Scholar] [CrossRef]
- Zhu, Y.; Hollis, J.H. Soup consumption is associated with a reduced risk of overweight and obesity but not metabolic syndrome in US adults: NHANES 2003–2006. PLoS ONE 2013, 8, e75630. [Google Scholar] [CrossRef]
- Flood, J.E.; Rolls, B.J. Soup preloads in a variety of forms reduce meal energy intake. Appetite 2007, 49, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Buttriss, J.L. Food reformulation: The challenges to the food industry. Proc. Nutr. Soc. 2013, 72, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Diez, J.; Bilal, U.; Franco, M. Unique features of the Mediterranean food environment: Implications for the prevention of chronic diseases Rh: Mediterranean food environments. Eur. J. Clin. Nutr. 2019, 72, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Bilal, U.; Jones-Smith, J.; Diez, J.; Lawrence, R.S.; Celentano, D.D.; Franco, M. Neighborhood social and economic change and retail food environment change in Madrid (Spain): The heart healthy hoods study. Health Place 2018, 51, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Scarmozzino, F.; Visioli, F. Covid-19 and the subsequent lockdown modified dietary habits of almost half the population in an Italian sample. Foods 2020, 9, 675. [Google Scholar] [CrossRef] [PubMed]
- Gressier, M.; Sassi, F.; Frost, G. Healthy foods and healthy diets. How government policies can steer food reformulation. Nutrients 2020, 12, 1992. [Google Scholar] [CrossRef]
- Monteiro, C.; Cannon, G. The Food System: Ultra-processed products. 2014 Position paper. Product reformulation will not improve public health. Public Health Nutr. 2014, 5, 140–168. [Google Scholar]
- Mintel. Global Food & Drink Trends: How Did We Do? Available online: www.mintel.com/blog/food-market-news/2017-global-food-drink-trends-how-did-we-do-12 (accessed on 12 September 2020).
- WHO (World Health Organization). Regional Office for the Eastern Mediterranean. Healthy diet. 2019. Available online: https://apps.who.int/iris/handle/10665/325828 (accessed on 15 September 2020).
- Rust, N.; Ridding, L.; Ward, C.; Clark, B.; Kehoee, L.; Dora, M.; Whittingham, M.J.; McGowani, P.; Chaudhary, A.; Reynolds, C.J.; et al. How to transition to reduced-meat diets that benefit people and the planet. Sci. Total Environ. 2020, 718, 137208. [Google Scholar] [CrossRef]
- Hawkes, C.; Popkin, B.M. Can the sustainable development goals reduce the burden of nutrition-related non-communicable diseases without truly addressing major food system reforms? BMC Med. 2015, 13, 143–149. [Google Scholar] [CrossRef]
- Di Monaco, R.; Cavella, S.; Torrieri, E.; Masi, P. Consumer acceptability of vegetable soups. J. Sens. Stud. 2007, 22, 81–98. [Google Scholar] [CrossRef]
- Maphosa, Y.; Jideani, V.A. The Role of Legumes in Human Nutrition. In Functional Food: Improve Health through Adequate Food; Chavarrri-Hueda, M., Ed.; IntechOpen: Zagreb, Croatia, 2017; Chapter 6; pp. 103–122. [Google Scholar]
- McKevith, B. Nutritional aspects of cereals. Nutr. Bull. 2004, 29, 11–142. [Google Scholar] [CrossRef]
- Pérez-Rea, D.; Antezana-Gómez, R. The Funcionality of Pseudocereal Starches. In Starch in Food, 2nd ed.; Sjöö, M., Nilsson, L., Eds.; Elsevier: Sawston, UK, 2018; pp. 509–542. [Google Scholar]
- Jacobsen, S.E.; Mujica, A.; Ortiz, R. The global potential for quinoa and other Andean crops. Food Rev. Int. 2003, 19, 139–148. [Google Scholar] [CrossRef]
- Jayasinghe, P.S.; Pahalawattaarachch, V.; Ranaweera, K.K.D.S. Formulation of nutritionally superior and low-cost seaweed based soup mix powder. J. Food Process. Technol. 2016, 7, 1000571. [Google Scholar]
- Bourne, L.T.; Langenhoven, M.L.; Steyn, K.; Jooste, P.L.; Laubscher, J.A.; Van Der Vyver, E. Nutrient intake in the urban African population of the Cape Peninsula, South Africa. The brisk study. Cent. Afr. J. Med. 1993, 3, 238–246. [Google Scholar]
- Malherbe, M.; Walsh, C.M.; Van Der Merwe, C.A. Consumer acceptability and salt perception of food with a reduced sodium content. J. Fam. Ecol. Consum. Sci. 2003, 31, 12–20. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Salt Reduction. 29 April 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/salt-reduction (accessed on 18 August 2020).
- Liem, D.G.; Miremadi, F.; Zandstra, E.H.; Keast, R.S.J. Health labelling can influence taste perception and use of table salt for reduced-sodium products. Public Health Nutr. 2012, 12, 2340–2347. [Google Scholar] [CrossRef]
- Mitchell, M.; Brunton, N.P.; Wilkinson, M.G. The influence of salt taste threshold on acceptability and purchase intent of reformulated reduced sodium vegetable soups. Food Qual. Prefer. 2013, 28, 356–360. [Google Scholar] [CrossRef]
- Gallager, J. Seven Teaspoons of Sugar Added to Healthy Soups. Irish Sunday Mirror. 19 March 2017. Available online: https://www.pressreader.com/similar/281831463549429 (accessed on 15 September 2020).
- Pérez-Alvarez, J.A.; Botella-Martínez, C.; Navarro-Rodríguez de Vera, C.; Sayas-Barberá, M.E.; Viuda-Martos, M.; Sánchez-Zapata, M.; Fernández-López, J. Research & Development during COVID-19 pandemic: Elaboration of healthy soups and creams. Technifood 2020, 130, 85–87. [Google Scholar]
- Guillon, F.; Champ, M. Structural and physical properties of dietary fibres, and consequences of processing on human physiology. Food Res. Int. 2000, 33, 233–245. [Google Scholar] [CrossRef]
- Pellegrini, M.; Lucas-González, R.; Ricci, A.; Fontecha, J.; Fernández-López, J.; Pérez-Alvarez, J.A.; Viuda-Martos, M. Chemical, fatty acid, polyphenolic profile, techno-functional and antioxidant properties of flours obtained from quinoa (Chenopodium quinoa willd) seeds. Ind. Crops Prod. 2018, 11, 38–46. [Google Scholar] [CrossRef]
- Repo-Carrasco-Valencia, R.A.M.; Serna, L.A. Quinoa (Chenopodium quinoa, Willd.) as a source of dietary fiber and other functional components. Food Sci. Technol. 2011, 31, 225–230. [Google Scholar] [CrossRef]
- López-Marcos, M.; Bailina, C.; Viuda-Martos, M.; Pérez-Alvarez, J.A.; Fernández-López, J. Properties of dietary fibers from agroindustrial coproducts as source for fiber-enriched foods. Food Bioprocess Technol. 2015, 8, 2400–2408. [Google Scholar] [CrossRef]
- Sánchez-Zapata, E.; Fuentes-Zaragoza, E.; Fernández-López, J.; Sendra, E.; Sayas, E.; Navarro, C.; Pérez-Alvarez, J.A. Preparation of dietary fiber poder from tiger nut (Cyperus esculentus) milk (“horchata”) byproducts and its physicochemical properties. J. Agric. Food Chem. 2009, 57, 7719–7725. [Google Scholar] [CrossRef] [PubMed]
- García-Amezquita, L.E.; Tejada-Ortigoza, V.; Serna-Saldivar, S.O.; Welti-Chanes, J. dietary fiber concentrates from fruit and vegetable by-products: Processing, modification, and application as functional ingredients. Food Bioprocess Technol. 2018, 11, 1439–1463. [Google Scholar] [CrossRef]
- Laufenberg, G.; Kunz, B.; Nystroem, M. Transformation ofvegetable waste into value added products: (A) the upgrading concept, (B) practical implementations. Biores. Technol. 2003, 87, 167–198. [Google Scholar] [CrossRef]
- Escalada Pla, M.F.; Ponce, N.M.; Stortz, C.A.; Gerschenson, L.N.; Rojas, A.M. Composition and functional properties of enriched fiber products obtained from pumpkin (Cucurbita moschata Duchesne ex Poiret). LWT-Food Sci. Technol. 2007, 40, 1176–1185. [Google Scholar] [CrossRef]
- Jhonson, B.R. Whey Protein Concentrates in Low Fat Applications; U.S. Dairy Export Council: Arlington, TX, USA, 2020. [Google Scholar]
- McClements, D.J.; Demetriades, K. An integrated approach to the development of reduced-fat food emulsions. Crit. Rev. Food Sci. Nutr. 1998, 38, 511–536. [Google Scholar] [CrossRef]
- McClements, D.J.; Keogh, M.K. Physical properties of cold-setting gels formed from heat-denatured whey protein isolate. J. Food Sci. Agric. 1995, 69, 7–14. [Google Scholar] [CrossRef]
- Wongsagonsup, R.; Pujchakarn, T.; Jitrakbumrung, S.; Chaiwat, W.; Fuongfuchat, A.; Varavinit, S.; Dangtipe, S.; Suphantharika, M. Effect of cross-linking on physicochemical properties of tapioca starch and its application in soup product. Carboh. Polym. 2014, 101, 656–665. [Google Scholar] [CrossRef]
- Cox, G.O.; Lee, Y.; Lee, S.Y. Drivers of liling in a model retorted creamy tomato soup system with varying levels of sodium, fat, and herbs. J. Food Sci. 2019, 84, 2610–2618. [Google Scholar] [CrossRef]
- Pastor, S. Tendencias en el Desarrollo de Nuevos Productos; AINIA-Technological Institute for the Food Industry: Valencia, Spain, 2020.
- Cornelia, M.; Christianti, A. Utilization of modified starch from avocado (Persea Americana Mill.) seed in cream soup production. IOP Conf. Ser. Earth Environ. Sci. 2018, 102, 012074. [Google Scholar] [CrossRef]
- Singh, Y.; Prasad, K. Sorption isotherms modeling approach of rice-based instant soup mix stored under controlled temperature and humidity. Cogent Food Agric. 2015, 1, 1103683. [Google Scholar] [CrossRef]
- Wang, R.; Mujumdar, A.S.; Sun, J.C. Microwave freeze-drying characteristics and sensory quality of instant vegetable soup. Dry. Technol. 2009, 27, 962–968. [Google Scholar] [CrossRef]
- Williams, C. The Healthiest Frozen Foods in the Supermarket: Soup. Cookinglight.com. 2016. Available online: https://www.cookinglight.com/eating-smart/smart-choices/healthiest-frozen-soups (accessed on 12 November 2020).
- Wartrous, M. Packaging Innovations Seals Treehouse Soup Deal. FoodBusiness News 4/22/2014. Available online: https://www.foodbusinessnews.net/articles/4103-packaging-innovation-seals-treehouse-soup-deal (accessed on 13 July 2020).
- SENC (Spanish Society of Communitarian Nutrition). Guías alimentarias para la población Española; La nueva pirámide de la alimentación saludable. Nutr. Hosp. 2016, 33, 1–48. [Google Scholar]
- Spill, M.K.; Birch, L.L.; Roe, L.S.; Rolls, B.J. Hiding vegetables to reduce energy density: An effective strategy to increase children’s vegetable intake and reduce energy intake. Am. J. Clin. Nutr. 2011, 94, 735–741. [Google Scholar] [CrossRef]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global prevalence of celiac disease: Systematic review and meta-analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836. [Google Scholar] [CrossRef]
- Sánchez, H.D.; González, R.J.; Osella, C.A.; Torres, R.L.; de la Torre, M.A. Elaboración de pan sin gluten con harinas de arroz extrudidas. Cienc. Tecnol. Aliment. 2008, 6, 109–116. [Google Scholar] [CrossRef]
- James, L.E.A. Quinoa (Chenopodium quinoa Willd.): Composition, chemistry, nutritional, and functional properties. Adv. Food Nutr. Res. 2009, 58, 1–31. [Google Scholar]
- Bolívar-Monsalve, J.; Ceballos-González, C.; Ramírez-Toro, C.; Bolívar, G.A. Reduction in saponin content and production of gluten-free cream soup base using quinoa fermented with Lactobacillus plantarum. J. Food Process. Preserv. 2018, 42, e13495. [Google Scholar] [CrossRef]
| Type of Soup 1 | Energy | Proteins | Carbohydrates | Fiber | Fats | Sodium | Reference | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Kcal) | g | %DRI 2 | g | %DRI | g | %DRI | g | %DRI | mg | %DRI | ||
| Tomato S. (C) | 82 | 1.9 | 4.8 | 16.3 | 32.6 | nd | - | 1.0 | 3.7 | nd | - | [151] |
| Tomato S. (C) | 128 | 1.5 | 3.8 | 16.5 | 33 | traces | - | 6.3 | 23.3 | 1800 | 90 | [151] |
| Tomato S. (C) | 110 | - | - | - | - | 2.9 | 11.6 | - | - | 856 | 42.8 | [146] |
| Tomato S. (R) | 83 | 2.3 | 5.8 | 18 | 36 | 1 | 4 | 2 | 7.4 | 500 | 25 | [151] |
| Onion S. (C) | 58 | - | - | - | - | 5.2 | 20.8 | - | - | 843 | 42.2 | [146] |
| Pumpkin S. (C) | 98 | - | - | - | - | 2.8 | 11.2 | - | - | 891 | 44.6 | [146] |
| Lentil S. (C) | 155 | - | - | - | - | 11 | 44 | - | - | 825 | 41.3 | [146] |
| M. Veget. S. (C) | 90 | - | - | - | - | 4.3 | 17.2 | - | - | 713 | 35.7 | [146] |
| M. Veget. S. (C) | 44 | 1.0 | 2.5 | 8.2 | 16.4 | nd | - | 0.8 | 3.0 | 840 | 42 | [151] |
| M. Veget. S. (C) | 110 | 0.9 | 2.3 | 8.8 | 17.6 | 4.6 | 18.4 | 8 | 29.7 | 1686 | 84.3 | [151] |
| M. Veget. S. (C) | 41 | 0.6 | 1.5 | 8.1 | 16.2 | 2.4 | 9.6 | 0 | 0 | 90 | 4.5 | [152] |
| M. Veget. S. (C) | 41 | 1.1 | 2.8 | 9.4 | 18.8 | 1.1 | 4.4 | 0.9 | 3.3 | 355 | 17.8 | [149] |
| M. Veget. S. (R) | 175 | 3.9 | 9.8 | 21 | 42 | 9.2 | 36.8 | 8.3 | 30.7 | 325 | 17.8 | [151] |
| M. Veget. S. (R) | 42 | 1.0 | 2.5 | 6.5 | 13 | 2.4 | 9.6 | 0.8 | 3.0 | 168 | 8.4 | [149] |
| Mushroom S. (C) | 59 | 2.3 | 5.8 | 9.9 | 19.8 | nd | - | 1.1 | 4.1 | 720 | 36 | [151] |
| Mushroom S. (C) | 82 | 1.8 | 4.5 | 9.7 | 19.4 | 0.5 | 2 | 4 | 15.8 | 875 | 43.8 | [151] |
| Mushroom S. (R) | 71 | 2.0 | 5.0 | 13 | 26 | 0.8 | 3.2 | 1.3 | 4.8 | 400 | 20 | [151] |
| Veget. C. (C) | 97.5 | 2.5 | 6.3 | 12.75 | 25.5 | 3 | 12 | 3.75 | 13.9 | 500 | 25 | [153] |
| Vichyssoise (C) | 145 | 1.5 | 3.8 | 12.75 | 25.5 | 0.5 | 2 | 9.25 | 34.3 | 480 | 24 | [153] |
| Pumpkin C. (C) | 92.5 | 2.0 | 5.0 | 10 | 20 | 2.25 | 9 | 5 | 18.5 | 760 | 38 | [153] |
| Asparagus C. (C) | 67.5 | 1.75 | 4.4 | 9.25 | 18.5 | 1.25 | 5 | 2.25 | 8.3 | 690 | 34.5 | [153] |
| Veget. C. (C) | 62.5 | 1.75 | 4.4 | 11 | 22 | 3 | 12 | 0 | 0 | 750 | 37.5 | [153] |
| Med.Veget. S. (C) | 115 | 2.25 | 5.7 | 9.75 | 19.5 | 3 | 12 | 6.5 | 24.1 | 650 | 32.5 | [153] |
| Mushroom C. (C) | 52.5 | 1.5 | 3.8 | 9.75 | 19.5 | 0 | 1.25 | 4.6 | 830 | 41.5 | [153] | |
| M. Veget. S. (C) | 92.8 | 1.8 | 4.5 | 19.86 | 39.7 | 0.41 | 1.6 | 0.70 | 2.3 | - | - | [145] |
| M. Veget. S. + lentil (R) | 93.8 | 3.74 | 9.4 | 17.99 | 35.98 | 0.40 | 1.6 | 0.78 | 2.9 | - | - | [145] |
| M. Veget. S. + green peal (R) | 93.6 | 3.72 | 9.4 | 17.82 | 35.6 | 1.05 | 4.2 | 0.83 | 3.1 | - | - | [145] |
| M. Veget. S. + chickpea (R) | 96.2 | 3.38 | 8.5 | 17.82 | 35.6 | 0.45 | 1.8 | 1.28 | 4.7 | . | - | [145] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-López, J.; Botella-Martínez, C.; Navarro-Rodríguez de Vera, C.; Sayas-Barberá, M.E.; Viuda-Martos, M.; Sánchez-Zapata, E.; Pérez-Álvarez, J.A. Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends. Plants 2020, 9, 1769. https://doi.org/10.3390/plants9121769
Fernández-López J, Botella-Martínez C, Navarro-Rodríguez de Vera C, Sayas-Barberá ME, Viuda-Martos M, Sánchez-Zapata E, Pérez-Álvarez JA. Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends. Plants. 2020; 9(12):1769. https://doi.org/10.3390/plants9121769
Chicago/Turabian StyleFernández-López, Juana, Carmen Botella-Martínez, Casilda Navarro-Rodríguez de Vera, María Estrella Sayas-Barberá, Manuel Viuda-Martos, Elena Sánchez-Zapata, and José Angel Pérez-Álvarez. 2020. "Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends" Plants 9, no. 12: 1769. https://doi.org/10.3390/plants9121769
APA StyleFernández-López, J., Botella-Martínez, C., Navarro-Rodríguez de Vera, C., Sayas-Barberá, M. E., Viuda-Martos, M., Sánchez-Zapata, E., & Pérez-Álvarez, J. A. (2020). Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends. Plants, 9(12), 1769. https://doi.org/10.3390/plants9121769







