Influence of Polyethylene Glycol on Leaf Anatomy, Stomatal Behavior, Water Loss, and Some Physiological Traits of Date Palm Plantlets Grown In Vitro and Ex Vitro
Abstract
1. Introduction
2. Results and Discussion
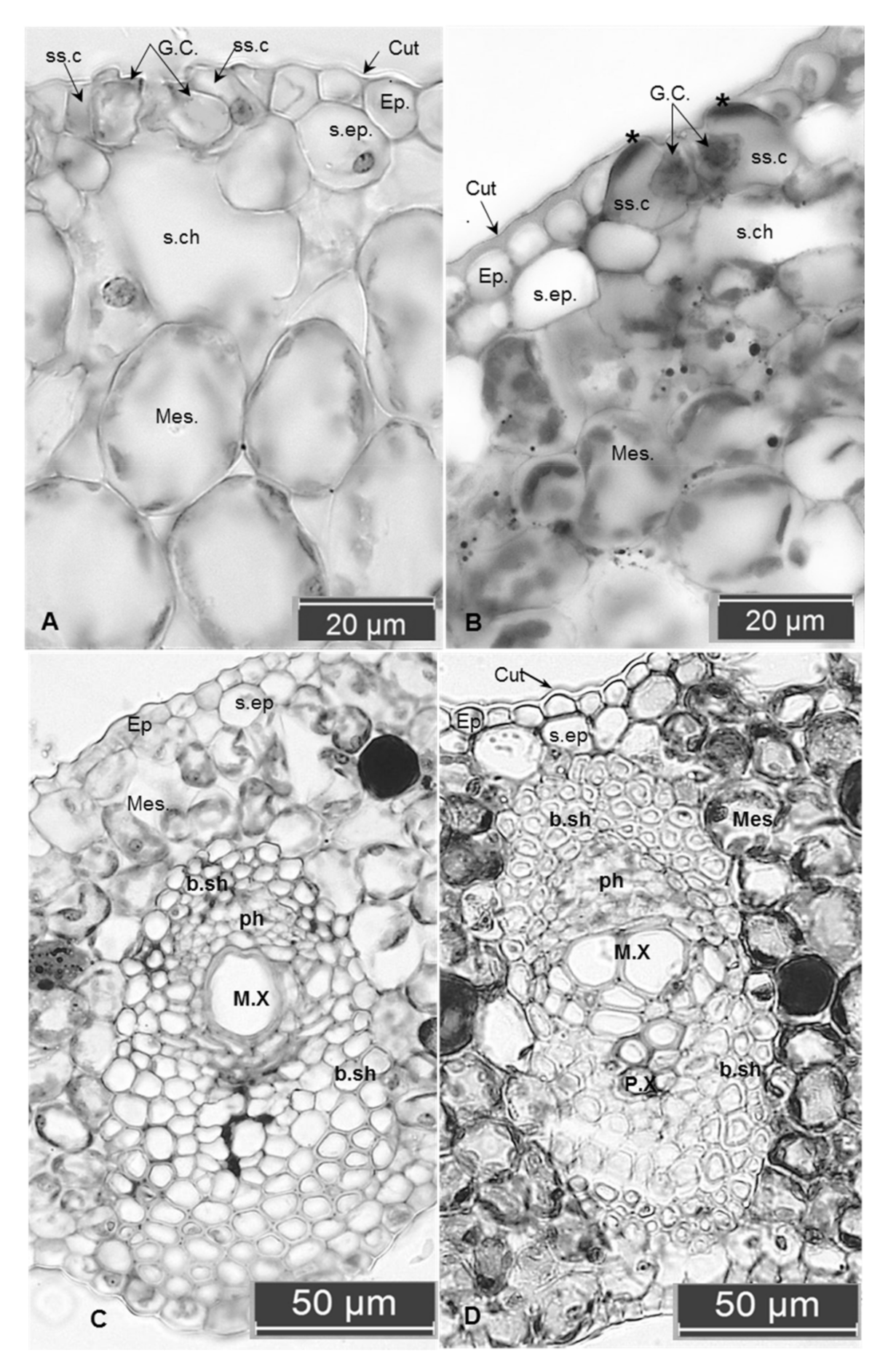
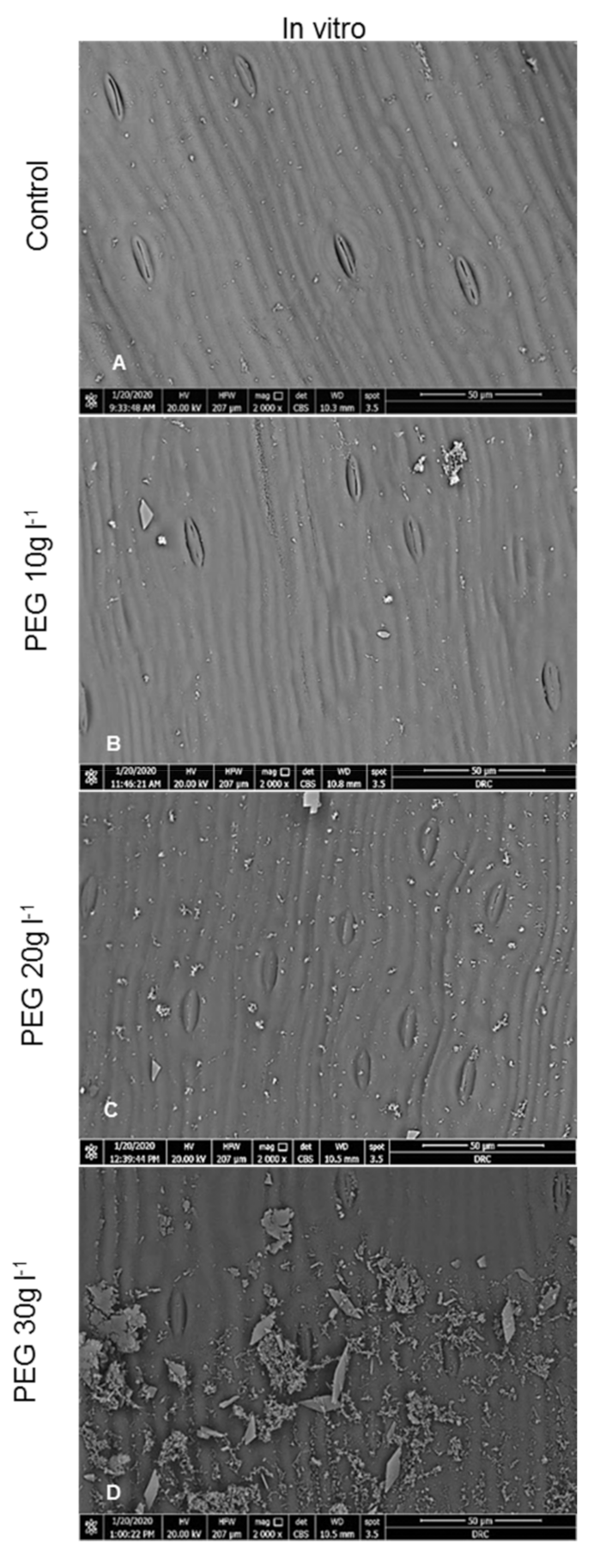
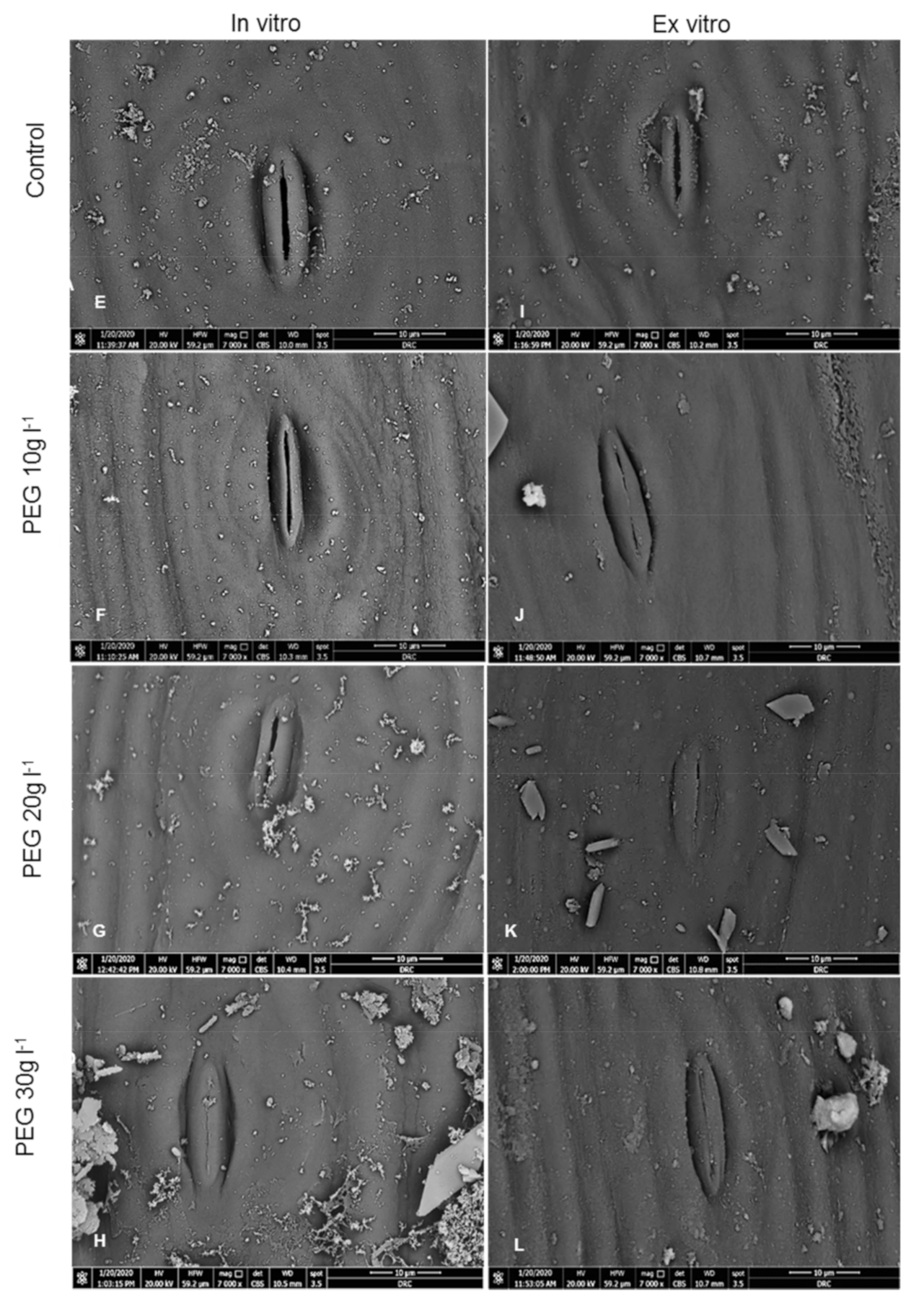
2.1. Anatomical Examinations
2.2. Water Loss %
2.3. Photosynthetic Pigments and Reducing Sugars
2.4. Survival Percentage (%)
3. Materials and Methods
3.1. Plant Material and Growth Conditions
3.2. Leaf Anatomy and Surface Ultrastructure (Scanning Electron Microscope, SEM)
3.3. Determination of the Water Loss %
3.4. Photosynthetic Pigments
3.5. Determination of Reducing Sugars
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Al-Khayri, J. Date palm Phoenix dactylifera L. micropropagation. In Protocols for Micropropagation of Woody Trees and Fruits; Jain, S., Haggman, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Abd El Bar, O.H.A.; El Dawayati, M.M. Histological changes on regeneration in vitro culture of date palm (Phoenix dactylifera) leaf explants. Aust. J. Crop Sci. 2014, 8, 848–855. [Google Scholar]
- Al-Khayri, J.M. In vitro germination of somatic embryos in date palm: Effect of auxin concentration and strength of MS salts. Curr. Sci. 2003, 84, 680–683. [Google Scholar]
- Quiroz-Figueroa, F.R.; Rojas-Herrera, R.; Galaz-Avalos, R.M.; Loyola-Vargas, V.M. Embryo production through somatic embryogenesis can be used to study cell differentiation in plants. Plant Cell Tissue Organ Cult 2006, 86, 285. [Google Scholar] [CrossRef]
- Al Kaabi, H.; Rhiss, A.; Hassan, M. Effect of auxins and cytokinins on the in vitro production of date palm bud generative tissues and on the number of differentiated buds. In Proceedings of the Second International Conference on Date Palms, Al Ain, UAE, 25–27 March 2001; pp. 47–86. [Google Scholar]
- Al-Khateeb, A. Role of cytokinin and auxin on the multiplication stage of date palm (Phoenix dactylifera L.) cv. Sukry. Biotechnology 2006, 5, 349–352. [Google Scholar] [CrossRef]
- Hazarika, B.; Parthasarathy, V.; Nagaraju, V. Influence of in vitro preconditioning of Citrus sp. microshoots with sucrose on their ex vitro establishment. Ind. J. Hortic. 2004, 61, 29–31. [Google Scholar]
- Pospóšilová, J.; Tichá, I.; Kadleček, P.; Haisel, D.; Plzáková, Š. Acclimatization of micropropagated plants to ex vitro conditions. Biol. Plant. 1999, 42, 481–497. [Google Scholar] [CrossRef]
- Pospisilova, J.; Synková, H.; Haisel, D.; Semoradova, S. Acclimation of plantlets to ex vitro conditions: Effects of air humidity, irradiance, CO2 concentration and abscisic acid (a Review). Acta Hortic. 2007, 748, 29. [Google Scholar] [CrossRef]
- Hazarika, B. Morpho-physiological disorders in in vitro culture of plants. Sci. Hortic. 2006, 108, 105–120. [Google Scholar] [CrossRef]
- Chandra, S.; Bandopadhyay, R.; Kumar, V.; Chandra, R. Acclimatization of tissue cultured plantlets: From laboratory to land. Biotech. Lett. 2010, 32, 1199–1205. [Google Scholar] [CrossRef]
- Fabbri, A.; Sutter, E.; Dunston, S.K. Anatomical changes in persistent leaves of tissuecultured strawberry plants after removal from culture. Sci. Hortic. 1986, 28, 331–337. [Google Scholar] [CrossRef]
- Asayesh, Z.M.; Vahdati, K.; Aliniaeifard, S.; Askari, N. Enhancement of ex vitro acclimation of walnut plantlets through modification of stomatal characteristics in vitro. Sci. Hortic. 2017, 220, 114–121. [Google Scholar] [CrossRef]
- Vahdati, K.; Asayesh, Z.M.; Aliniaeifard, S.; Leslie, C. Improvement of ex vitro desiccation through elevation of CO2 concentration in the atmosphere of culture vessels during in vitro growth. HortScience 2017, 52, 1006–1012. [Google Scholar] [CrossRef]
- Awad, M.A. Promotive effects of a 5-aminolevulinic acid-based fertilizer on growth of tissue culture-derived date palm plants (Phoenix dactylifera L.) during acclimatization. Sci. Hortic. 2008, 118, 48–52. [Google Scholar] [CrossRef]
- Awad, M.A.; Soaud, A.; El-Konaissi, S. Effect of exogenous application of anti-stress substances and elemental sulfur on growth and stress tolerance of tissue culture derived plantlets of date palm (Phoenix dactylifera L.) cv ‘Khalas’ during acclimatization. J. Appl. Hortic. 2006, 8, 129–134. [Google Scholar] [CrossRef]
- Zaid, A.; de Wet, P.F. Date palm propagation. In Date Palm Cultivation; Zaid, A., Arias-Jimenez, E., Eds.; FAO Plant Production and Protection Paper, No.156; FAO: Rome, Italy, 1999. [Google Scholar]
- Pospíšilová, J.; Wilhelmová, N.A.; Synková, H.; Čatský, J.; Krebs, D.; Tichá, I.; Hanáčková, B.; Snopek, J. Acclimation of tobacco plantlets to ex vitro conditions as affected by application of abscisic acid. J. Exp. Bot. 1998, 49, 863–869. [Google Scholar] [CrossRef]
- Sutter, E.G. Morphological, physical and chemical characteristics of epicuticular wax on ornamental plants regenerated in vitro. Ann. Bot. 1985, 55, 321–329. [Google Scholar] [CrossRef]
- Zaid, A.; Hughes, H. In vitro acclimatization of date palm (Phoenix dactylifera L.) plantlets: A quantitative comparison of epicuticular leaf wax as a function of polyethylene glycol treatment. Plant Cell Rep. 1995, 15, 111–114. [Google Scholar] [CrossRef]
- Asayesh, Z.M.; Vahdati, K.; Aliniaeifard, S.; Askari, N. Investigation of physiological components involved in low water conservation capacity of in vitro walnut plants. Sci. Hortic. 2017, 224, 1–7. [Google Scholar] [CrossRef]
- Dias, M.; Pinto, G.; Santos, C. Acclimatization of micropropagated plantlets induces an antioxidative burst: A case study with Ulmus minor Mill. Photosynth 2011, 49, 259–266. [Google Scholar] [CrossRef]
- Pospíšilová, J.; Synková, H.; Haisel, D.; Baťková, P. Effect of abscisic acid on photosynthetic parameters during ex vitro transfer of micropropagated tobacco plantlets. Bio. Plant. 2009, 53, 11–20. [Google Scholar] [CrossRef]
- Dami, I.; Hughes, H. Leaf anatomy and water loss of in vitro PEG-treated ‘Valiant’ grape. Plant Cell Tissue Organ Cult. 1995, 42, 179–184. [Google Scholar] [CrossRef]
- Zaid, A.; Hughes, H. Water loss and polyethylene glycol-mediated acclimatization of in vitro-grown seedlings of 5 cultivars of date palm (Phoenix dactylifera L.) plantlets. Plant Cell Rep. 1995, 14, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Short, K.; Warburton, J.; Roberts, A. In vitro hardening of cultured cauliflower and chrysanthemum plantlets to humidity. Acta Hortic. 1987, 212, 329–334. [Google Scholar] [CrossRef]
- El Dawayati, M.M.; Bar, O.H.A.E.; Zaid, Z.E.; El Din, A.F.Z. In vitro morpho-histological studies of newly developed embryos from abnormal malformed embryos of date palm cv. Gundila under desiccation effect of polyethelyne glycol treatments. Ann. Agric. Sci. 2012, 57, 117–128. [Google Scholar] [CrossRef]
- Ali-Ahmad, M.; Hughes, H.G.; Safadi, F. Studies on stomatal function, epicuticular wax and stem-root transition region of polyethylene glycol-treated and nontreated in vitro grape plantlets. Vitr. Cell. Dev. Biol. Plant 1998, 34, 1–7. [Google Scholar] [CrossRef]
- Aliniaeifard, S.; Asayesh, Z.M.; Driver, J.; Vahdati, K. Stomatal features and desiccation responses of Persian walnut leaf as caused by in vitro stimuli aimed at stomatal closure. Trees 2020, 34, 1219–1232. [Google Scholar] [CrossRef]
- Hazarika, B.; Nagaraju, V.; Parthasarathy, V.; Bhowmik, G. Biochemical basis of acclimatization of micropropagated plantlets–A review. Agric. Rev. 2001, 22, 65–67. [Google Scholar]
- Taiz, L.; Zeiger, E. Plant Physiology: Stress Physiology; Sinauer Associates: Sunderland, MA, USA, 2006. [Google Scholar]
- Evert, R.F. Esau’s Plant Anatomy: Meristems, Cells, and Tissues of the Plant Body: Their Structure, Function, and Development, 3rd ed.; John Wiley & Sons, Inc., Publication: Hoboken, NJ, USA, 2006; pp. 222–223. [Google Scholar] [CrossRef]
- Joshi, P.; Joshi, N.; Purohit, S. Stomatal characteristics during micropropagation of Wrightia tomentosa. Biol. Plan. 2006, 50, 275–278. [Google Scholar] [CrossRef]
- Wardle, K.; Dobbs, E.B.; Short, K.C. In vitro acclimatization of aseptically cultured plantets to humidity. J. Am. Soc. Hortic. Sci. 1983, 108, 386–389. [Google Scholar]
- Brainerd, K.; Fuchigami, L. Stomatal functioning of in vitro and greenhouse apple leaves in darkness, mannitol, ABA, and CO2. J. Exp. Bot. 1982, 33, 388–392. [Google Scholar] [CrossRef]
- Sutter, E. Stomatal and cuticular water loss from apple, cherry, and sweetgum plants after removal from in vitro culture. J. Am. Soci. Hortic. Sci. 1988, 113, 234–238. [Google Scholar]
- Abd Elbar, O.H.; Farag, R.E.; Shehata, S.A. Effect of putrescine application on some growth, biochemical and anatomical characteristics of Thymus vulgaris L. under drought stress. Ann. Agric. Sci. 2019, 64, 129–137. [Google Scholar] [CrossRef]
- Guenni, O.; Douglas, M.; Baruch, Z. Responses to drought of five Brachiaria species. I. Biomass production, leaf growth, root distribution, water use and forage quality. Plant Soil 2002, 243, 229–241. [Google Scholar] [CrossRef]
- Kim, T.H.; Bohmer, M.; Hu, H.H.; Nishimura, N.; Schroeder, J.I. Guard Cell Signal Transduction Network: Advances in Understanding Abscisic Acid, CO2, and Ca2+ Signaling. Annu. Rev. Plant Biol. 2010, 61, 561–591. [Google Scholar] [CrossRef] [PubMed]
- Arve, L.; Torre, S.; Olsen, J.; Tanino, K. Stomatal responses to drought stress and air humidity. In Abiotic Stress in Plants-Mechanisms and Adaptations; Intech Open: London, UK, 2011. [Google Scholar]
- Riederer, M.; Schneider, G. The effect of the environment on the permeability and composition of Citrus leaf cuticles. Planta 1990, 1990. 180, 154–165. [Google Scholar] [CrossRef]
- Kosma, D.K.; Jenks, M.A. Eco-physiological and molecular-genetic determinants of plant cuticle function in drought and salt stress tolerance. In Advances in Molecular Breeding toward Drought and Salt Tolerant Crops; Jenks, M.A., Hasegawa, P.M., Jain, S.M., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 91–120. [Google Scholar]
- Bi, H.; Kovalchuk, N.; Langridge, P.; Tricker, P.J.; Lopato, S.; Borisjuk, N. The impact of drought on wheat leaf cuticle properties. BMC Plant Biol. 2017, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Safadi, F.; Hughes, H. Comparison of the diffusive resistance of Polyethylene glycol treated and non-treated tissue culture Tobacco plantlets. HortScience 1990, 25, 1105b–1105. [Google Scholar] [CrossRef]
- Elmaghrabi, A.M.; Rogers, H.J.; Francis, D.; Ochatt, S.J. PEG induces high expression of the cell cycle checkpoint gene WEE1 in embryogenic callus of Medicago truncatula: Potential link between cell cycle checkpoint regulation and osmotic stress. Front. Plant Sci. 2017, 8, 1479. [Google Scholar] [CrossRef]
- Abd El-Maboud, M.M.A.; Abd Elbar, O.H. Adaptive responses of Limoniastrum monopetalum (L.) Boiss. growing naturally at different habitats. Plant Physiol. Rep. 2020, 25, 325–344. [Google Scholar] [CrossRef]
- Abd Elbar, O.H. Development of the successive cambia in Sesuvium verrucosum Raf (Aizoaceae). Ann. Agric. Sci. 2015, 60, 203–208. [Google Scholar] [CrossRef][Green Version]
- Cha-um, S.; Takabe, T.; Kirdmanee, C. Physio-biochemical responses of oil palm (Elaeis guineensis Jacq.) seedlings to mannitol-and polyethylene glycol-induced iso-osmotic stresses. Plant Prod. Sci. 2012, 15, 65–72. [Google Scholar] [CrossRef][Green Version]
- Zhang, J.; Jia, W.; Yang, J.; Ismail, A.M. Role of ABA in integrating plant responses to drought and salt stresses. Field Crop. Res. 2006, 97, 111–119. [Google Scholar] [CrossRef]
- Kong, L.; Yeung, E.C. Effects of silver nitrate and polyethylene glycol on white spruce (Picea glauca) somatic embryo development: Enhancing cotyledonary embryo formation and endogenous ABA content. Physiol. Plant. 1995, 93, 298–304. [Google Scholar] [CrossRef]
- Darko, E.; Végh, B.; Khalil, R.; Marček, T.; Szalai, G.; Pál, M.; Janda, T. Metabolic responses of wheat seedlings to osmotic stress induced by various osmolytes under iso-osmotic conditions. PLoS ONE 2019, 14, e0226151. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Duan, X.; Wang, Y.; Zhu, K.; Zhang, J.; Wang, R.; Hu, H.; Qi, F.; Pan, J.; Yan, Y.; et al. Methane protects against polyethylene glycol-induced osmotic stress in maize by improving sugar and ascorbic acid metabolism. Sci. Rep. 2017, 7, srep46185. [Google Scholar] [CrossRef] [PubMed]
- Stasolla, C.; van Zyl, L.; Egertsdotter, U.; Craig, D.; Liu, W.; Sederoff, R.R. The effects of polyethylene glycol on gene expression of developing white spruce somatic embryos. Plant Physiol. 2003, 131, 49–60. [Google Scholar] [CrossRef]
- Handa, S.; Bressan, R.A.; Handa, A.K.; Carpita, N.C.; Hasegawa, P.M. Solutes contributing to osmotic adjustment in cultured plant cells adapted to water stress. Plant Physiol. 1983, 73, 834–843. [Google Scholar] [CrossRef]
- Zanella, M.; Borghi, G.L.; Pirone, C.; Thalmann, M.; Pazmino, D.; Costa, A.; Santelia, D.; Trost, P.; Sparla, F. β-amylase 1 (BAM1) degrades transitory starch to sustain proline biosynthesis during drought stress. J. Exp. Bot. 2016, 67, 1819–1826. [Google Scholar] [CrossRef]
- Wilson, S.M.; Burton, R.A.; Doblin, M.S.; Stone, B.A.; Newbigin, E.J.; Fincher, G.B.; Bacic, A. Temporal and spatial appearance of wall polysaccharides during cellularization of barley (Hordeum vulgare) endosperm. Planta 2006, 224, 655. [Google Scholar] [CrossRef]
- Hazarika, B. Acclimatization of tissue-cultured plants. Curr. Sci. 2003, 85, 1704–1712. [Google Scholar]
- Shim, S.-W.; Hahn, E.-J.; Paek, K.-Y. In vitro and ex vitro growth of grapevine rootstock5BB’as influenced by number of air exchanges and the presence or absence of sucrose in culture media. Plant Cell Tissue Organ. Cult. 2003, 75, 57–62. [Google Scholar] [CrossRef]
- Aliniaeifard, S.; Matamoros, P.M.; van Meeteren, U. Stomatal malfunctioning under low VPD conditions: Induced by alterations in stomatal morphology and leaf anatomy or in the ABA signaling? Physiol. Plant. 2014, 152, 688–699. [Google Scholar] [CrossRef]
- Ziv, M.; Schwartz, A.; Fleminger, D. Malfunctioning stomata in vitreous leaves of carnation (Dianthus caryophyllus) plants propagated in vitro; implications for hardening. Plant Sci. 1987, 52, 127–134. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Abdelbar, O.H. Histological analysis of the developmental stages of direct somatic embryogenesis induced from in vitro leaf explants of date palm. In Date Palm Biotechnology Protocols; Springer: Totowa, NJ, USA, 2017; Volume I, pp. 145–162. [Google Scholar] [CrossRef]
- Harley, M.; Ferguson, I. The role of the SEM in pollen morphology and plant systematics. Scanning Electron Microscopy in Taxonomy and Functional Morphology; Oxford University Press: Oxford, UK, 1990; pp. 45–68. [Google Scholar]
- Moran, R. Formulae for determination of chlorophyllous pigments extracted with N, N-dimethylformamide. Plant Physiol. 1982, 69, 1376–1381. [Google Scholar] [CrossRef] [PubMed]
- Wellburn, A.R. The spectral determination of chlorophylls a and b, as well as total carotenoids, using various solvents with spectrophotometers of different resolution. J. Plant Physiol. 1994, 144, 307–313. [Google Scholar] [CrossRef]
- Marsden, W.L.; Gray, P.P.; Nippard, G.J.; Quinlan, M.R. Evaluation of the DNS method for analysing lignocellulosic hydrolysates. J. Chem. Technol. Biotechnol. 1982, 32, 1016–1022. [Google Scholar] [CrossRef]
- Snedecor, G.W.; Cochran, W.G. Statistical Methods, 7th ed.; Iowa State University Press: Ames, IA, USA, 1980; pp. 80–86. [Google Scholar]
- Gomez, K.A.; Gomez, A.A. Statistical Procedures for Agricultural Research; John Wiley & Sons: Hoboken, NJ, USA, 1984. [Google Scholar] [CrossRef]


| PEG g L−1 | Adaxial Surface | Abaxial Surface | ||||||
|---|---|---|---|---|---|---|---|---|
| Density | Opened | Closed | Aperture Length | Density | Opened | Closed | Aperture Length | |
| 0.0 | 140.6 | 133.74 a | 6.82 d | 13.67 a | 159.5 | 150.9 a | 8.54 c | 14.67 a |
| 10 | 140.6 | 101.2 b | 39.40 c | 10.74 ab | 154.4 | 114.9 b | 39.42 b | 11.32 b |
| 20 | 138.9 | 75.46 c | 63.42 b | 9.54 bc | 145.8 | 99.46 c | 46.26 b | 10.45 b |
| 30 | 149.4 | 41.10 d | 108.1 a | 7.36 c | 151.4 | 51.42 d | 89.18 a | 7.77 c |
| LSD0.05 | NS | 15.29 | 20.93 | 3.30 | NS | 14.78 | 16.46 | 1.305 |
| PEG g L−1 | Adaxial Surface | Abaxial Surface | ||||||
|---|---|---|---|---|---|---|---|---|
| Density | Opened | Closed | Aperture Length | Density | Opened | Closed | Aperture Length | |
| 0.0 | 145.8 | 109.7 a | 36.04 c | 11.44 a | 187.0 a | 145.8 a | 41.16 b | 12.32 a |
| 10 | 147.7 | 86.02 b | 61.66 b | 7.87 b | 158.0 b | 101.6 b | 56.44 b | 8.71 b |
| 20 | 154.3 | 41.12 c | 115.0 a | 6.15 c | 168.1 ab | 54.86 c | 113.2 a | 6.99 c |
| 30 | 154.1 | 30.58 c | 123.5 a | 3.14 d | 157.34 b | 33.46 d | 124.0 a | 3.43 d |
| LSD0.05 | NS | 14.06 | 20.15 | 1.59 | 19.54 | 17.75 | 21.28 | 1.181 |
| Time (A) | PEG g L−1 (B) | Mean (A) | |||
|---|---|---|---|---|---|
| 0.0 | 10 | 20 | 30 | ||
| In vitro | |||||
| 30 min | 24.19 q | 22.94 q | 22.91 q | 20.43 r | 20.62 H |
| 60 min | 36.30 n | 33.35 o | 33.38 o | 30.33 p | 33.27 G |
| 90 min | 42.24 jk | 38.76 m | 36.61 n | 34.92 no | 38.13 F |
| 120 min | 44.52 i | 43.93 ij | 39.64 lm | 38.74 m | 41.71 E |
| 150 min | 50.54 de | 46.85 h | 42.57 jk | 41.30 kl | 45.32 D |
| 180 min | 54.46 c | 51.67 d | 46.55 h | 44.64 i | 49.33 C |
| 210 min | 56.67 b | 53.83 c | 48.68 fg | 47.81 gh | 51.75 B |
| 240 min | 60.38 a | 55.31 bc | 50.28 Ef | 49.22 efg | 53.80 A |
| Mean (B) | 46.13 A | 43.33 B | 40.03 C | 38.42 D | |
| Ex vitro | |||||
| 30 min | 22.32 n | 21.19 no | 20.43 o | 18.35 p | 20.6 H |
| 60 min | 33.13 k | 31.11 l | 30.34 l | 24.49 m | 29.77 G |
| 90 min | 36.63 hi | 34.50 jk | 33.35 k | 30.85 l | 33.83 F |
| 120 min | 39.50 g | 37.30 h | 35.30 ij | 33.27 k | 36.35 E |
| 150 min | 42.93 ef | 40.28 g | 37.78 h | 36.37 hi | 39.34 D |
| 180 min | 48.18 cd | 44.54 e | 40.81 g | 40.36 g | 43.47 C |
| 210 min | 51.57 b | 46.93 d | 42.82 f | 42.80 f | 46.03 B |
| 240 min | 55.66 a | 49.33 c | 46.60 d | 43.97 ef | 48.89 A |
| Mean (B) | 41.24 A | 38.16 B | 35.93 C | 33.81 D | |
| LSD0.05 for | In vitro | Ex vitro | |||
| A | 0.4913 | 0.5907 | |||
| B | 0.6949 | 0.8354 | |||
| AB | 1.702 | 1.671 | |||
| PEG g L−1 | Chla µg mL−1 | Chlb µg mL−1 | Chla,b µg ml−1 | Carotenoids µg mL−1 | Reducing Sugars mg g−1 DW |
|---|---|---|---|---|---|
| In vitro | |||||
| 0.0 | 24.225 b | 7.665 bc | 31.891 c | 6.610 b | 63.623 d |
| 10 | 26.232 b | 9.395 b | 35.627 b | 7.751 a | 132.10 c |
| 20 | 33.795 a | 11.267 a | 45.062 a | 8.591 a | 195.30 b |
| 30 | 20.989 c | 6.354 c | 27.343 d | 5.740 b | 260.059 a |
| LSD0.05 | 1.728 | 1.730 | 2.881 | 1.007 | 2.234 |
| Ex vitro | |||||
| 0.0 | 27.283 c | 10.82 a | 38.099 c | 7.79 a | 24.962 d |
| 10 | 30.993 b | 11.12 a | 42.113 b | 8.019 c | 55.157 c |
| 20 | 34.278 a | 11.60 a | 45.895 a | 8.454 a | 142.359 b |
| 30 | 24.514 d | 8.898 b | 33.412 d | 6.030 b | 304.604 a |
| LSD0.05 | 1.621 | 1.243 | 2.117 | 1.514 | 2.823 |
| Treatments | Survival Percentage (%) |
|---|---|
| PEG 0.0 g L−1 | 63% ab |
| PEG 10 g L−1 | 72% ab |
| PEG 20 g L−1 | 90% a |
| PEG 30 g L−1 | 45% b |
| LSD0.05 | 27.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Din, A.F.M.Z.E.; Ibrahim, M.F.M.; Farag, R.; El-Gawad, H.G.A.; El-Banhawy, A.; Alaraidh, I.A.; Rashad, Y.M.; Lashin, I.; El-Yazied, A.A.; Elkelish, A.; et al. Influence of Polyethylene Glycol on Leaf Anatomy, Stomatal Behavior, Water Loss, and Some Physiological Traits of Date Palm Plantlets Grown In Vitro and Ex Vitro. Plants 2020, 9, 1440. https://doi.org/10.3390/plants9111440
Din AFMZE, Ibrahim MFM, Farag R, El-Gawad HGA, El-Banhawy A, Alaraidh IA, Rashad YM, Lashin I, El-Yazied AA, Elkelish A, et al. Influence of Polyethylene Glycol on Leaf Anatomy, Stomatal Behavior, Water Loss, and Some Physiological Traits of Date Palm Plantlets Grown In Vitro and Ex Vitro. Plants. 2020; 9(11):1440. https://doi.org/10.3390/plants9111440
Chicago/Turabian StyleDin, Amal F. M. Zein El, Mohamed F. M. Ibrahim, Reham Farag, Hany G. Abd El-Gawad, Ahmed El-Banhawy, Ibrahim A. Alaraidh, Younes M. Rashad, Islam Lashin, Ahmed Abou El-Yazied, Amr Elkelish, and et al. 2020. "Influence of Polyethylene Glycol on Leaf Anatomy, Stomatal Behavior, Water Loss, and Some Physiological Traits of Date Palm Plantlets Grown In Vitro and Ex Vitro" Plants 9, no. 11: 1440. https://doi.org/10.3390/plants9111440
APA StyleDin, A. F. M. Z. E., Ibrahim, M. F. M., Farag, R., El-Gawad, H. G. A., El-Banhawy, A., Alaraidh, I. A., Rashad, Y. M., Lashin, I., El-Yazied, A. A., Elkelish, A., & Elbar, O. H. A. (2020). Influence of Polyethylene Glycol on Leaf Anatomy, Stomatal Behavior, Water Loss, and Some Physiological Traits of Date Palm Plantlets Grown In Vitro and Ex Vitro. Plants, 9(11), 1440. https://doi.org/10.3390/plants9111440












