Regulation of Symbiotic Nitrogen Fixation in Legume Root Nodules
Abstract
1. Introduction
2. Overview of the Control of the Legumes´ Symbiotic Nitrogen Fixation
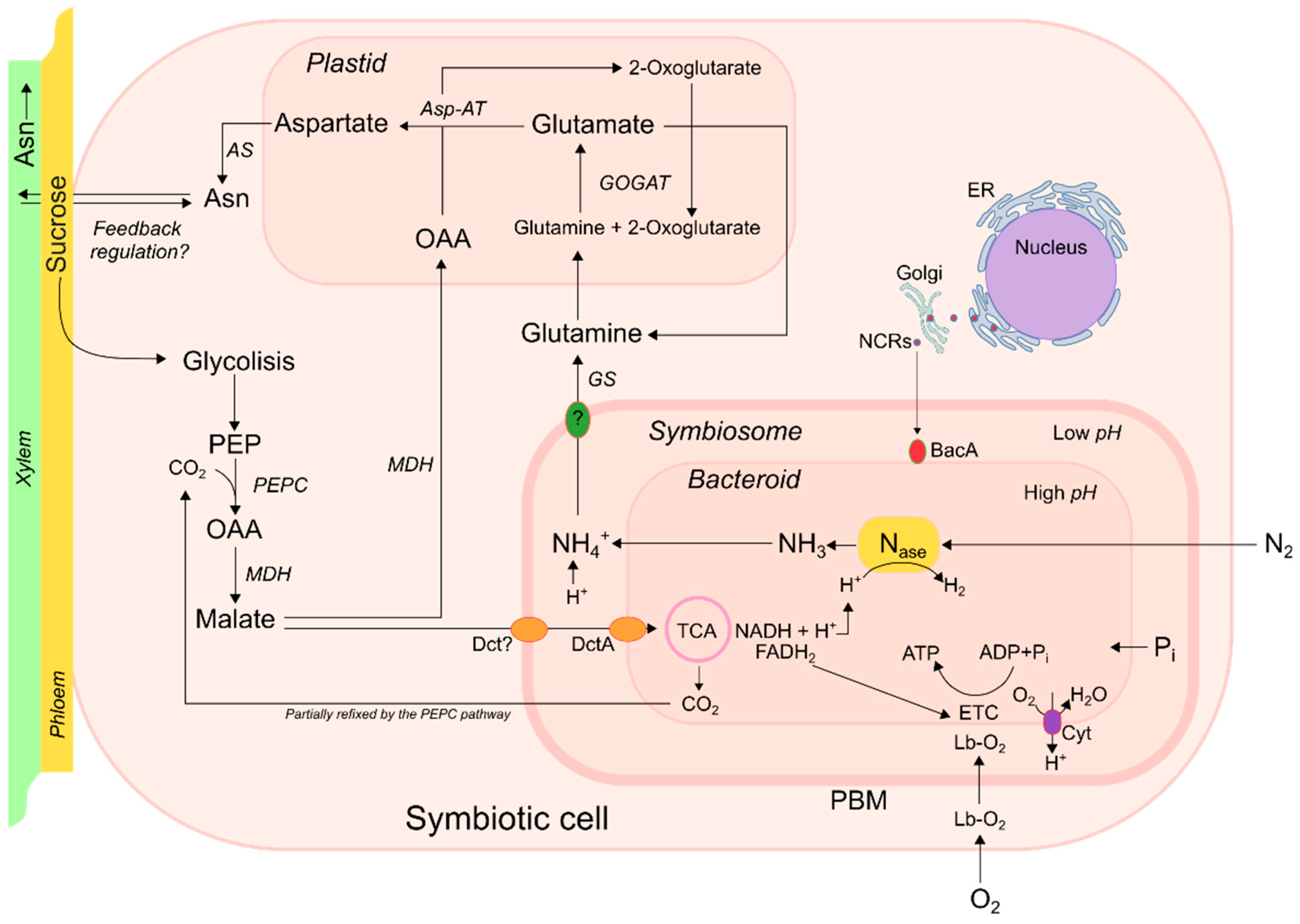
3. Nitrogen-Metabolism
4. Carbon-Metabolism
5. Nodule Oxygen Supply
6. Oxidative Stress
7. Molecular Control of N2 Fixation
8. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Jacob, C.; Carrasco, B.; Schwember, A.R. Advances in breeding and biotechnology of legume crops. Plant Cell Tissue Organ Cult. PCTOC 2016, 127, 561–584. [Google Scholar] [CrossRef]
- FAO FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 26 September 2018).
- Peix, A.; Ramírez-Bahena, M.H.; Velázquez, E.; Bedmar, E.J. Bacterial associations with legumes. Crit. Rev. Plant Sci. 2015, 34, 17–42. [Google Scholar] [CrossRef]
- White, J.; Prell, J.; James, E.K.; Poole, P. Nutrient sharing between symbionts. Plant Physiol. 2007, 144, 604–614. [Google Scholar] [CrossRef]
- Herridge, D.F.; Peoples, M.B.; Boddey, R.M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 2008, 311, 1–18. [Google Scholar] [CrossRef]
- Adams, M.A.; Buchmann, N.; Sprent, J.I.; Bucley, T.N.; Turnbull, T.L. Crops, nitrogen, water: Are legumes friend, foe, or misunderstood ally? Trends Plant Sci. 2018, 23, 539–550. [Google Scholar] [CrossRef]
- Poole, P.; Ramachandran, V.; Terpolilli, J. Rhizobia: From saprophytes to endosymbionts. Nat. Rev. Microbiol. 2018, 16, 291–303. [Google Scholar] [CrossRef]
- Buhian, W.P.; Bensmihen, S. Mini-review: Nod factor regulation of phytohormone signaling and homeostasis during rhizobia-legume symbiosis. Front. Plant Sci. 2018, 9, 1247. [Google Scholar] [CrossRef]
- Miller, A.J.; Fan, X.; Shen, Q.; Smith, S.J. Amino acids and nitrate as signals for the regulation of nitrogen acquisition. J. Exp. Bot. 2008, 59, 111–119. [Google Scholar] [CrossRef]
- Vance, C.P. Carbon and nitrogen metabolism in legume nodules. In Nitrogen-Fixing Leguminous Symbioses; Dilworth, M.J., James, E.K., Sprent, J.I., Newton, W.E., Eds.; Nitrogen Fixation: Origins, Applications, and Research Progress; Springer: Dordrecht, The Netherlands, 2008; pp. 293–320. ISBN 978-1-4020-3545-6. [Google Scholar]
- Minchin, F.R.; James, E.K.; Becana, M. Oxygen diffusion, production of reactive oxygen and nitrogen species, and antioxidants in legume nodules. In Nitrogen-Fixing Leguminous Symbioses; Dilworth, M.J., James, E.K., Sprent, J.I., Newton, W.E., Eds.; Nitrogen Fixation: Origins, Applications, and Research Progress; Springer: Dordrecht, The Netherlands, 2008; pp. 321–362. ISBN 978-1-4020-3545-6. [Google Scholar]
- Becana, M.; Matamoros, M.A.; Udvardi, M.; Dalton, D.A. Recent insights into antioxidant defenses of legume root nodules. New Phytol. 2010, 188, 960–976. [Google Scholar] [CrossRef]
- Valentine, A.J.; Benedito, V.A.; Kang, Y. Legume nitrogen fixation and soil abiotic stress: From physiology to genomics and beyond. In Annual Plant Reviews; Wiley-Blackwell: Hoboken, NJ, USA, 2011; pp. 207–248. ISBN 978-1-4443-2860-8. [Google Scholar]
- Ferguson, B.J.; Gresshoff, P.M. Physiological implications of legume nodules associated with soil acidity. In Legume Nitrogen Fixation in a Changing Environment; Springer: Cham, Switzerland, 2015; pp. 113–125. ISBN 978-3-319-06211-2. [Google Scholar]
- González, E.M.; Larrainzar, E.; Marino, D.; Wienkoop, S.; Gil-Quintana, E.; Arrese-Igor, C. Physiological responses of N2-fixing legumes to water limitation. In Legume Nitrogen Fixation in a Changing Environment; Springer: Cham, Switzerland, 2015; pp. 5–33. ISBN 978-3-319-06211-2. [Google Scholar]
- Berrada, H.; Fikri-Benbrahim, K. Taxonomy of the Rhizobia: Current perspectives. Br. Microbiol. Res. J. 2014, 4, 616–639. [Google Scholar] [CrossRef]
- Sprent, J.I. Nodulation in Legumes; Royal Botanic Gardens: Kew, UK, 2001; ISBN 1-84246-013-7.
- Sprent, J.I. Legume Nodulation: A Global Perspective; John Wiley & Sons: Hoboken, NJ, USA, 2009; ISBN 1-4443-1639-7. [Google Scholar]
- Schulze, J. How are nitrogen fixation rates regulated in legumes? J. Plant Nutr. Soil Sci. 2004, 167, 125–137. [Google Scholar] [CrossRef]
- Sulieman, S.; Tran, L.-S.P. Asparagine: An amide of particular distinction in the regulation of symbiotic nitrogen fixation of legumes. Crit. Rev. Biotechnol. 2013, 33, 309–327. [Google Scholar] [CrossRef]
- del Pozo, A.; Garnier, E.; Aronson, J. Contrasted nitrogen utilization in annual C3 grass and legume crops: Physiological explorations and ecological considerations. Acta Oecol. 2000, 21, 79–89. [Google Scholar] [CrossRef]
- Adams, M.A.; Turnbull, T.L.; Sprent, J.I.; Buchmann, N. Legumes are different: Leaf nitrogen, photosynthesis, and water use efficiency. Proc. Natl. Acad. Sci. USA 2016, 113, 4098–4103. [Google Scholar] [CrossRef]
- Peoples, M.B.; Craswell, E.T. Biological nitrogen fixation: Investments, expectations and actual contributions to agriculture. Plant Soil 1992, 141, 13–39. [Google Scholar] [CrossRef]
- Adams, M.A.; Buckley, T.N.; Salter, W.T.; Buchmann, N.; Blessing, C.H.; Turnbull, T.L. Contrasting responses of crop legumes and cereals to nitrogen availability. New Phytol. 2018, 217, 1475–1483. [Google Scholar] [CrossRef]
- Streeter, J. Inhibition of legume nodule formation and N2 fixation by nitrate. Crit. Rev. Plant Sci. 1988, 7, 1–23. [Google Scholar] [CrossRef]
- Cabeza, R.A.; Koester, B.; Liese, R.; Lingner, A.; Baumgarten, V.; Dirks, J.; Salinas-Riester, G.; Pommerenke, C.; Dittert, K.; Schulze, J. An RNA sequencing transcriptome analysis reveals novel insights into molecular aspects of the nitrate impact on the nodule activity of Medicago truncatula. Plant Physiol. 2014, 164, 400–411. [Google Scholar] [CrossRef]
- Schubert, S. Nitrogen assimilation by legumes-processes and ecological limitations. Fertil. Res. 1995, 42, 99–107. [Google Scholar] [CrossRef]
- Liese, R.; Schulze, J.; Cabeza, R.A. Nitrate application or P deficiency induce a decline in Medicago truncatula N2-fixation by similar changes in the nodule transcriptome. Sci. Rep. 2017, 7, 46264. [Google Scholar] [CrossRef]
- Soussana, J.-F.; Tallec, T. Can we understand and predict the regulation of biological N2 fixation in grassland ecosystems? Nutr. Cycl. Agroecosyst. 2010, 88, 197–213. [Google Scholar] [CrossRef]
- Hartwig, U.A. The regulation of symbiotic N2 fixation: A conceptual model of N feedback from the ecosystem to the gene expression level. Perspect. Plant Ecol. Evol. Syst. 1998, 1, 92–120. [Google Scholar] [CrossRef]
- Jeudy, C.; Ruffel, S.; Freixes, S.; Tillard, P.; Santoni, A.L.; Morel, S.; Journet, E.-P.; Duc, G.; Gojon, A.; Lepetit, M.; et al. Adaptation of Medicago truncatula to nitrogen limitation is modulated via local and systemic nodule developmental responses. New Phytol. 2010, 185, 817–828. [Google Scholar] [CrossRef]
- Fischinger, S.A.; Hristozkova, M.; Mainassara, Z.-A.; Schulze, J. Elevated CO2 concentration around alfalfa nodules increases N2 fixation. J. Exp. Bot. 2010, 61, 121–130. [Google Scholar] [CrossRef]
- Fischinger, S.A.; Drevon, J.-J.; Claassen, N.; Schulze, J. Nitrogen from senescing lower leaves of common bean is re-translocated to nodules and might be involved in a N-feedback regulation of nitrogen fixation. J. Plant Physiol. 2006, 163, 987–995. [Google Scholar] [CrossRef]
- Cabeza, R.A.; Liese, R.; Fischinger, S.A.; Sulieman, S.; Avenhaus, U.; Lingner, A.; Hein, H.; Koester, B.; Baumgarten, V.; Dittert, K.; et al. Long-term non-invasive and continuous measurements of legume nodule activity. Plant J. 2015, 81, 637–648. [Google Scholar] [CrossRef]
- Czyzewicz, N.; Yue, K.; Beeckman, T.; Smet, I.D. Message in a bottle: Small signalling peptide outputs during growth and development. J. Exp. Bot. 2013, 64, 5281–5296. [Google Scholar] [CrossRef]
- Djordjevic, M.A.; Mohd-Radzman, N.A.; Imin, N. Small-peptide signals that control root nodule number, development, and symbiosis. J. Exp. Bot. 2015, 66, 5171–5181. [Google Scholar] [CrossRef]
- Sulieman, S.; Schulze, J. Phloem-derived γ-aminobutyric acid (GABA) is involved in upregulating nodule N2 fixation efficiency in the model legume Medicago truncatula. Plant Cell Environ. 2010, 33, 2162–2172. [Google Scholar] [CrossRef]
- Sulieman, S. Does GABA increase the efficiency of symbiotic N2 fixation in legumes? Plant Signal. Behav. 2011, 6, 32–36. [Google Scholar] [CrossRef]
- Sulieman, S.; Fischinger, S.A.; Gresshoff, P.M.; Schulze, J. Asparagine as a major factor in the N-feedback regulation of N2 fixation in Medicago truncatula. Physiol. Plant. 2010, 140, 21–31. [Google Scholar] [CrossRef]
- Forde, B.G.; Lea, P.J. Glutamate in plants: Metabolism, regulation, and signalling. J. Exp. Bot. 2007, 58, 2339–2358. [Google Scholar] [CrossRef]
- Neo, H.H.; Layzell, D.B. Phloem glutamine and the regulation of O2 diffusion in legume nodules. Plant Physiol. 1997, 113, 259–267. [Google Scholar] [CrossRef]
- Seabra, A.R.; Pereira, P.A.; Becker, J.D.; Carvalho, H.G. Inhibition of glutamine synthetase by phosphinothricin leads to transcriptome reprograming in root nodules of Medicago truncatula. Mol. Plant. Microbe Interact. 2012, 25, 976–992. [Google Scholar] [CrossRef]
- Matamoros, M.A.; Kim, A.; Peñuelas, M.; Ihling, C.; Griesser, E.; Hoffmann, R.; Fedorova, M.; Frolov, A.; Becana, M. Protein carbonylation and glycation in legume nodules. Plant Physiol. 2018, 177, 1510–1528. [Google Scholar] [CrossRef]
- King, C.A.; Purcell, L.C. Inhibition of N2 fixation in soybean is associated with elevated ureides and amino acids. Plant Physiol. 2005, 137, 1389–1396. [Google Scholar] [CrossRef]
- Ladrera, R.; Marino, D.; Larrainzar, E.; González, E.M.; Arrese-Igor, C. Reduced carbon availability to bacteroids and elevated ureides in nodules, but not in shoots, are involved in the nitrogen fixation response to early drought in soybean. Plant Physiol. 2007, 145, 539–546. [Google Scholar] [CrossRef]
- Bustos-Sanmamed, P.; Tovar-Méndez, A.; Crespi, M.; Sato, S.; Tabata, S.; Becana, M. Regulation of nonsymbiotic and truncated hemoglobin genes of Lotus japonicus in plant organs and in response to nitric oxide and hormones. New Phytol. 2011, 189, 765–776. [Google Scholar] [CrossRef]
- Larrainzar, E.; Wienkoop, S.; Scherling, C.; Kempa, S.; Ladrera, R.; Arrese-Igor, C.; Weckwerth, W.; González, E.M. Carbon metabolism and bacteroid functioning are involved in the regulation of nitrogen fixation in Medicago truncatula under drought and recovery. Mol. Plant. Microbe Interact. 2009, 22, 1565–1576. [Google Scholar] [CrossRef]
- Carter, A.M.; Tegeder, M. Increasing nitrogen fixation and seed development in soybean requires complex adjustments of nodule nitrogen metabolism and partitioning processes. Curr. Biol. 2016, 26, 2044–2051. [Google Scholar] [CrossRef]
- Garneau, M.G.; Tan, Q.; Tegeder, M. Function of pea amino acid permease AAP6 in nodule nitrogen metabolism and export, and plant nutrition. J. Exp. Bot. 2018, 69, 5205–5219. [Google Scholar] [CrossRef]
- Unkovich, M.J.; Baldock, J.; Peoples, M.B. Prospects and problems of simple linear models for estimating symbiotic N2 fixation by crop and pasture legumes. Plant Soil 2010, 329, 75–89. [Google Scholar] [CrossRef]
- Espinoza, S.; Ovalle, C.; Zagal, E.; Matus, I.; Tay, J.; Peoples, M.B.; del Pozo, A. Contribution of legumes to wheat productivity in Mediterranean environments of central Chile. Field Crops Res. 2012, 133, 150–159. [Google Scholar] [CrossRef]
- Anglade, J.; Billen, G.; Garnier, J. Relationships for estimating N2 fixation in legumes: Incidence for N balance of legume-based cropping systems in Europe. Ecosphere 2015, 6, 1–24. [Google Scholar] [CrossRef]
- Kaschuk, G.; Kuyper, T.W.; Leffelaar, P.A.; Hungria, M.; Giller, K.E. Are the rates of photosynthesis stimulated by the carbon sink strength of rhizobial and arbuscular mycorrhizal symbioses? Soil Biol. Biochem. 2009, 41, 1233–1244. [Google Scholar] [CrossRef]
- Schulze, J.; Adgo, E.; Merbach, W. Carbon costs associated with N2 fixation in Vicia faba L and Pisum sativum L over a 14-day period. Plant Biol. 1999, 1, 625–631. [Google Scholar] [CrossRef]
- Arrese-Igor, C.; González, E.; Marino, D.; Ladrera, R.; Larrainzar, E.; Gil Quintana, E. Physiological responses of legume nodules to drought. Plant Stress 2011, 5, 24–31. [Google Scholar]
- Vance, C.P.; Heichel, G.H. Carbon in N2 fixation: Limitation or exquisite adaptation. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1991, 42, 373–390. [Google Scholar] [CrossRef]
- Redondo, F.J.; de la Peña, T.C.; Morcillo, C.N.; Lucas, M.M.; Pueyo, J.J. Overexpression of flavodoxin in bacteroids induces changes in antioxidant metabolism leading to delayed senescence and starch accumulation in alfalfa root nodules. Plant Physiol. 2009, 149, 1166–1178. [Google Scholar] [CrossRef]
- Vidal, R.; Gerbaud, A.; Vidal, D.; Drevon, J.J. A short-term decrease in nitrogenase activity (C2H2 reduction) is induced by exposure of soybean shoots to their CO2 compensation point. Plant Physiol. 1995, 108, 1455–1460. [Google Scholar] [CrossRef][Green Version]
- Rogers, A.; Ainsworth, E.A.; Leakey, A.D.B. Will elevated carbon dioxide concentration amplify the benefits of nitrogen fixation in lgumes? Plant Physiol. 2009, 151, 1009–1016. [Google Scholar] [CrossRef]
- Lam, S.K.; Chen, D.; Norton, R.; Armstrong, R.; Mosier, A.R. Nitrogen dynamics in grain crop and legume pasture systems under elevated atmospheric carbon dioxide concentration: A meta-analysis. Glob. Chang. Biol. 2012, 18, 2853–2859. [Google Scholar] [CrossRef]
- Sanz-Sáez, Á.; Heath, K.D.; Burke, P.V.; Ainsworth, E.A. Inoculation with an enhanced N2-fixing Bradyrhizobium japonicum strain (USDA110) does not alter soybean (Glycine max Merr.) response to elevated [CO2]. Plant Cell Environ. 2015, 38, 2589–2602. [Google Scholar] [CrossRef]
- Li, Y.; Yu, Z.; Liu, X.; Mathesius, U.; Wang, G.; Tang, C.; Wu, J.; Liu, J.; Zhang, S.; Jin, J. Elevated CO2 increases nitrogen fixation at the reproductive phase contributing to various yield responses of soybean cultivars. Front. Plant Sci. 2017, 8, 1546. [Google Scholar] [CrossRef]
- Udvardi, M.; Poole, P. Transport and metabolism in legume-rhizobia symbioses. Annu. Rev. Plant Biol. 2013, 64, 781–805. [Google Scholar] [CrossRef]
- Kryvoruchko, I.S.; Sinharoy, S.; Torres-Jerez, I.; Sosso, D.; Pislariu, C.I.; Guan, D.; Murray, J.; Benedito, V.A.; Frommer, W.B.; Udvardi, M.K. MtSWEET11, a nodule-specific sucrose transporter of Medicago truncatula. Plant Physiol. 2016, 171, 554–565. [Google Scholar] [CrossRef]
- Udvardi, M.K.; Price, G.D.; Gresshoff, P.M.; Day, D.A. A dicarboxylate transporter on the peribacteroid membrane of soybean nodules. FEBS Lett. 1988, 231, 36–40. [Google Scholar] [CrossRef]
- Takanashi, K.; Sasaki, T.; Kan, T.; Saida, Y.; Sugiyama, A.; Yamamoto, Y.; Yazaki, K. A Dicarboxylate Transporter, LjALMT4, Mainly Expressed in Nodules of Lotus japonicus. Mol. Plant. Microbe Interact. 2016, 29, 584–592. [Google Scholar] [CrossRef]
- Oldroyd, G.E.D.; Murray, J.D.; Poole, P.S.; Downie, J.A. The rules of engagement in the legume-rhizobial symbiosis. Annu. Rev. Genet. 2011, 45, 119–144. [Google Scholar] [CrossRef]
- Liu, A.; Contador, C.A.; Fan, K.; Lam, H.-M. Interaction and Regulation of Carbon, Nitrogen, and Phosphorus Metabolisms in Root Nodules of Legumes. Front. Plant Sci. 2018, 9, 1860. [Google Scholar] [CrossRef]
- Whitehead, L.F.; Day, D.A.; Tyerman, S.D. Divalent cation gating of an ammonium permeable channel in the symbiotic membrane from soybean nodules. Plant J. 1998, 16, 313–324. [Google Scholar] [CrossRef]
- Fischinger, S.K.A. Die Bedeutung der CO2-Fixierung von Leguminosenknöllchen für ihre Aktivität und Effizienz. Ph.D. Thesis, University of Goettingen, Goettingen, Germany, 2009. [Google Scholar]
- Aleman, L.; Ortega, J.L.; Martinez-Grimes, M.; Seger, M.; Holguin, F.O.; Uribe, D.J.; Garcia-Ibilcieta, D.; Sengupta-Gopalan, C. Nodule-enhanced expression of a sucrose phosphate synthase gene member (MsSPSA) has a role in carbon and nitrogen metabolism in the nodules of alfalfa (Medicago sativa L.). Planta 2010, 231, 233–244. [Google Scholar] [CrossRef][Green Version]
- Baier, M.C.; Barsch, A.; Küster, H.; Hohnjec, N. Antisense repression of the Medicago truncatula nodule-enhanced sucrose synthase leads to a handicapped nitrogen fixation mirrored by specific alterations in the symbiotic transcriptome and metabolome. Plant Physiol. 2007, 145, 1600–1618. [Google Scholar] [CrossRef][Green Version]
- Gebril, S.; Seger, M.; Villanueva, F.M.; Ortega, J.L.; Bagga, S.; Sengupta-Gopalan, C. Transgenic alfalfa (Medicago sativa) with increased sucrose phosphate synthase activity shows enhanced growth when grown under N2-fixing conditions. Planta 2015, 242, 1009–1024. [Google Scholar] [CrossRef]
- Warembourg, F.R.; Roumet, C. Why and how to estimate the cost of symbiotic N2 fixation? A progressive approach based on the use of 14C and 15N isotopes. In Ecology of Arable Land—Perspectives and Challenges; Developments in Plant and Soil Sciences; Springer: Dordrecht, The Netherlands, 1989; pp. 31–41. ISBN 978-94-010-6950-2. [Google Scholar]
- Atkins, C.; Smith, P.; Mann, A.; Thumfort, P. Localization of carbonic anhydrase in legume nodules. Plant Cell Environ. 2001, 24, 317–326. [Google Scholar] [CrossRef]
- Gálvez, S.; Hirsch, A.M.; Wycoff, K.L.; Hunt, S.; Layzell, D.B.; Kondorosi, A.; Crespi, M. Oxygen regulation of a nodule-located carbonic anhydrase in alfalfa. Plant Physiol. 2000, 124, 1059–1068. [Google Scholar] [CrossRef]
- Miller, S.S.; Boylan, K.L.M.; Vance, C.P. Alfalfa root nodule carbon dioxide fixation: III. Immunological studies of nodule phosphoenolpyruvate carboxylase. Plant Physiol. 1987, 84, 501–508. [Google Scholar] [CrossRef]
- Schulze, J.; Kupietz, A.; Merbach, W. N2 fixation in different varieties of Vicia faba L. and Pisum sativum L. during pod-filling. Agribiol. Res. 1998, 51, 261–269. [Google Scholar]
- Egli, M.A.; Griffith, S.M.; Miller, S.S.; Anderson, M.P.; Vance, C.P. Nitrogen assimilating enzyme activities and enzyme protein during development and senescence of effective and plant gene-controlled ineffective alfalfa nodules. Plant Physiol. 1989, 91, 898–904. [Google Scholar] [CrossRef]
- Miller, S.S.; Driscoll, B.T.; Gregerson, R.G.; Gantt, J.S.; Vance, C.P. Alfalfa malate dehydrogenase (MDH): Molecular cloning and characterization of five different forms reveals a unique nodule-enhanced MDH. Plant J. 1998, 15, 173–184. [Google Scholar] [CrossRef]
- Fedorova, M.; Tikhonovich, I.A.; Vance, C.P. Expression of C-assimilating enzymes in pea (Pisum sativum L.) root nodules. In situ localization in effective nodules. Plant Cell Environ. 1999, 22, 1249–1262. [Google Scholar] [CrossRef]
- Schulze, J.; Tesfaye, M.; Litjens, R.H.M.G.; Bucciarelli, B.; Trepp, G.; Miller, S.; Samac, D.; Allan, D.; Vance, C.P. Malate plays a central role in plant nutrition. Plant Soil 2002, 247, 133–139. [Google Scholar] [CrossRef]
- Le Roux, M.; Phiri, E.; Khan, W.; Şakiroğlu, M.; Valentine, A.; Khan, S. Expression of novel cytosolic malate dehydrogenases (cMDH) in Lupinus angustifolius nodules during phosphorus starvation. J. Plant Physiol. 2014, 171, 1609–1618. [Google Scholar] [CrossRef]
- Layzell, D.B. Oxygen regulation and adenylates in legume nodules. In Nitrogen Fixation: From Molecules to Crop Productivity; Current Plant Science and Biotechnology in Agriculture; Springer: Dordrecht, The Netherlands, 2000; pp. 367–368. ISBN 978-0-7923-6233-3. [Google Scholar]
- Marchal, K.; Vanderleyden, J. The “oxygen paradox” of dinitrogen-fixing bacteria. Biol. Fertil. Soils 2000, 30, 363–373. [Google Scholar] [CrossRef]
- Hunt, S.; King, B.J.; Layzell, D.B. Effects of gradual increases in O2 concentration on nodule activity in soybean. Plant Physiol. 1989, 91, 315–321. [Google Scholar] [CrossRef]
- Witty, J.F.; Minchin, F.R.; Skot, L.; Sheehy, J.E. Nitrogen fixation and oxygen in legume root nodules. Oxf. Surv. Plant Mol. Cell Biol. 1986, 3, 275–314. [Google Scholar]
- Bergersen, F.J. Regulation of nitrogen fixation in infected cells of leguminous root nodules in relation to O2 supply. Plant Soil 1997, 191, 189–203. [Google Scholar] [CrossRef]
- Vance, C.P.; Miller, S.S.; Driscoll, B.T.; Robinson, D.L.; Trepp, G.; Gantt, J.S.; Samas, D.A. Nodule carbon metabolism: Organic acids for N2 fixation. In Biological Nitrogen Fixation for the 21st Century; Current Plant Science and Biotechnology in Agriculture; Springer: Dordrecht, The Netherlands, 1998; pp. 443–448. ISBN 978-94-010-6169-8. [Google Scholar]
- Denison, R.F.; Kinraide, T.B. Oxygen-induced membrane depolarizations in legume root nodules (possible evidence for an osmoelectrical mechanism controlling nodule gas permeability). Plant Physiol. 1995, 108, 235–240. [Google Scholar] [CrossRef]
- Avenhaus, U.; Cabeza, R.A.; Liese, R.; Lingner, A.; Dittert, K.; Salinas-Riester, G.; Pommerenke, C.; Schulze, J. Short-term molecular acclimation processes of legume nodules to increased external oxygen concentration. Front. Plant Sci. 2016, 6, 1133. [Google Scholar] [CrossRef]
- Tjepkema, J.D.; Yocum, C.S. Measurement of oxygen partial pressure within soybean nodules by oxygen microelectrodes. Planta 1974, 119, 351–360. [Google Scholar] [CrossRef]
- Witty, J.F. Microelectrode measurements of hydrogen concentrations and gradients in legume nodules. J. Exp. Bot. 1991, 42, 765–771. [Google Scholar] [CrossRef]
- Witty, J.F.; Minchin, F.R. Hydrogen measurements provide direct evidence for a variable physical barrier to gas diffusion in legume nodules. J. Exp. Bot. 1998, 49, 1015–1020. [Google Scholar] [CrossRef]
- Lazali, M.; Drevon, J.J. The nodule conductance to O2 diffusion increases with phytase activity in N2-fixing Phaseolus vulgaris L. Plant Physiol. Biochem. 2014, 80, 53–59. [Google Scholar] [CrossRef]
- Kleinert, A.; le Roux, M.; Kang, Y.; Valentine, A.J. Oxygen and the regulation of N2 fixation in legume nodules under P scarcity. In Legume Nitrogen Fixation in Soils with Low Phosphorus Availability; Springer: Cham, Switzerland, 2017; pp. 97–109. ISBN 978-3-319-55728-1. [Google Scholar]
- Ott, T.; van Dongen, J.T.; Gu¨nther, C.; Krusell, L.; Desbrosses, G.; Vigeolas, H.; Bock, V.; Czechowski, T.; Geigenberger, P.; Udvardi, M.K. Symbiotic leghemoglobins are crucial for nitrogen fixation in legume root nodules but not for general plant growth and development. Curr. Biol. 2005, 15, 531–535. [Google Scholar] [CrossRef]
- Appleby, C.A. Leghemoglobin and rhizobium respiration. Annu. Rev. Plant Physiol. 1984, 35, 443–478. [Google Scholar] [CrossRef]
- Uchiumi, T.; Shimoda, Y.; Tsuruta, T.; Mukoyoshi, Y.; Suzuki, A.; Senoo, K.; Sato, S.; Kato, T.; Tabata, S.; Higashi, S.; et al. Expression of symbiotic and nonsymbiotic globin genes responding to microsymbionts on Lotus japonicus. Plant Cell Physiol. 2002, 43, 1351–1358. [Google Scholar] [CrossRef]
- Wisniewski, J.-P.; Rathbun, E.A.; Knox, J.P.; Brewin, N.J. Involvement of diamine oxidase and peroxidase in insolubilization of the extracellular matrix: Implications for pea nodule initiation by Rhizobium leguminosarum. Mol. Plant. Microbe Interact. 2000, 13, 413–420. [Google Scholar] [CrossRef]
- Rubio, M.C.; James, E.K.; Clemente, M.R.; Bucciarelli, B.; Fedorova, M.; Vance, C.P.; Becana, M. Localization of superoxide dismutases and hydrogen peroxide in legume root nodules. Mol. Plant. Microbe Interact. 2004, 17, 1294–1305. [Google Scholar] [CrossRef]
- Puppo, A.; Pauly, N.; Boscari, A.; Mandon, K.; Brouquisse, R. Hydrogen peroxide and nitric oxide: Key regulators of the legume-rhizobium and mycorrhizal symbioses. Antioxid. Redox Signal. 2012, 18, 2202–2219. [Google Scholar] [CrossRef]
- Arthikala, M.K.; Sánchez-López, R.; Nava, N.; Santana, O.; Cárdenas, L.; Quinto, C. RbohB, a Phaseolus vulgaris NADPH oxidase gene, enhances symbiosome number, bacteroid size, and nitrogen fixation in nodules and impairs mycorrhizal colonization. New Phytol. 2014, 202, 886–900. [Google Scholar] [CrossRef]
- Marino, D.; González, E.M.; Arrese-Igor, C. Drought effects on carbon and nitrogen metabolism of pea nodules can be mimicked by paraquat: Evidence for the occurrence of two regulation pathways under oxidative stresses. J. Exp. Bot. 2006, 57, 665–673. [Google Scholar] [CrossRef]
- Damiani, I.; Pauly, N.; Puppo, A.; Brouquisse, R.; Boscari, A. Reactive oxygen species and nitric oxide control early steps of the legume—Rhizobium symbiotic interaction. Front. Plant Sci. 2016, 7, 454. [Google Scholar] [CrossRef]
- Hichri, I.; Boscari, A.; Castella, C.; Rovere, M.; Puppo, A.; Brouquisse, R. Nitric oxide: A multifaceted regulator of the nitrogen-fixing symbiosis. J. Exp. Bot. 2015, 66, 2877–2887. [Google Scholar] [CrossRef]
- Xue, Y.; Liu, Z.; Gao, X.; Jin, C.; Wen, L.; Yao, X.; Ren, J. GPS-SNO: Computational prediction of protein S-nitrosylation sites with a modified GPS algorithm. PLoS ONE 2010, 5, e11290. [Google Scholar] [CrossRef]
- Melo, P.M.; Silva, L.S.; Ribeiro, I.; Seabra, A.R.; Carvalho, H.G. Glutamine synthetase is a molecular target of nitric oxide in root nodules of Medicago truncatula and is regulated by tyrosine nitration. Plant Physiol. 2011, 157, 1505–1517. [Google Scholar] [CrossRef]
- Oger, E.; Marino, D.; Guigonis, J.-M.; Pauly, N.; Puppo, A. Sulfenylated proteins in the Medicago truncatula–Sinorhizobium meliloti symbiosis. J. Proteomics 2012, 75, 4102–4113. [Google Scholar] [CrossRef]
- Ferguson, B.J.; Indrasumunar, A.; Hayashi, S.; Lin, M.-H.; Lin, Y.-H.; Reid, D.E.; Gresshoff, P.M. Molecular analysis of legume nodule development and autoregulation. J. Integr. Plant Biol. 2010, 52, 61–76. [Google Scholar] [CrossRef]
- Reid, D.E.; Ferguson, B.J.; Hayashi, S.; Lin, Y.-H.; Gresshoff, P.M. Molecular mechanisms controlling legume autoregulation of nodulation. Ann. Bot. 2011, 108, 789–795. [Google Scholar] [CrossRef]
- Ferguson, B.J.; Mens, C.; Hastwell, A.H.; Zhang, M.; Su, H.; Jones, C.H.; Chu, X.; Gresshoff, P.M. Legume nodulation: The host controls the party. Plant Cell Environ. 2019, 42, 41–51. [Google Scholar] [CrossRef]
- Hastwell, A.H.; Corcilius, L.; Williams, J.T.; Gresshoff, P.M.; Payne, R.J.; Ferguson, B.J. Triarabinosylation is required for nodulation-suppressive CLE peptides to systemically inhibit nodulation in Pisum sativum. Plant Cell Environ. 2019, 42, 188–197. [Google Scholar] [CrossRef]
- Tsikou, D.; Yan, Z.; Holt, D.B.; Abel, N.B.; Reid, D.E.; Madsen, L.H.; Bhasin, H.; Sexauer, M.; Stougaard, J.; Markmann, K. Systemic control of legume susceptibility to rhizobial infection by a mobile microRNA. Science 2018, 362, 233–236. [Google Scholar] [CrossRef]
- Takahara, M.; Magori, S.; Soyano, T.; Okamoto, S.; Yoshida, C.; Yano, K.; Sato, S.; Tabata, S.; Yamaguchi, K.; Shigenobu, S.; et al. TOO MUCH LOVE, a novel kelch repeat-containing F-box protein, functions in the long-distance regulation of the legume-rhizobium symbiosis. Plant Cell Physiol. 2013, 54, 433–447. [Google Scholar] [CrossRef]
- Nishida, H.; Handa, Y.; Tanaka, S.; Suzaki, T.; Kawaguchi, M. Expression of the CLE-RS3 gene suppresses root nodulation in Lotus japonicus. J. Plant Res. 2016, 129, 909–919. [Google Scholar] [CrossRef]
- Reid, D.E.; Ferguson, B.J.; Gresshoff, P.M. Inoculation- and nitrate-induced CLE peptides of soybean control NARK-dependent nodule formation. Mol. Plant. Microbe Interact. 2011, 24, 606–618. [Google Scholar] [CrossRef]
- Okamoto, S.; Ohnishi, E.; Sato, S.; Takahashi, H.; Nakazono, M.; Tabata, S.; Kawaguchi, M. Nod factor/nitrate-induced CLE genes that drive HAR1-mediated systemic regulation of nodulation. Plant Cell Physiol. 2009, 50, 67–77. [Google Scholar] [CrossRef]
- Nishida, H.; Tanaka, S.; Handa, Y.; Ito, M.; Sakamoto, Y.; Matsunaga, S.; Betsuyaku, S.; Miura, K.; Soyano, T.; Kawaguchi, M.; et al. A NIN-LIKE PROTEIN mediates nitrate-induced control of root nodule symbiosis in Lotus japonicus. Nat. Commun. 2018, 9, 499. [Google Scholar] [CrossRef]
- Venkateshwaran, M.; Ané, J.-M. Legumes and nitrogen fixation: Physiological, molecular, evolutionary perspectives, and applications. In The Molecular and Physiological Basis of Nutrient Use Efficiency in Crops; Wiley-Blackwell: Hoboken, NJ, USA, 2011; pp. 457–489. ISBN 978-0-470-96070-7. [Google Scholar]
- Ramalingam, A.; Kudapa, H.; Pazhamala, L.T.; Weckwerth, W.; Varshney, R.K. Proteomics and metabolomics: Two emerging areas for legume improvement. Front. Plant Sci. 2015, 6, 1116. [Google Scholar] [CrossRef]
- Omrane, S.; Ferrarini, A.; D’Apuzzo, E.; Rogato, A.; Delledonne, M.; Chiurazzi, M. Symbiotic competence in Lotus japonicus is affected by plant nitrogen status: Transcriptomic identification of genes affected by a new signalling pathway. New Phytol. 2009, 183, 380–394. [Google Scholar] [CrossRef]
- Cabeza, R.A.; Liese, R.; Lingner, A.; von Stieglitz, I.; Neumann, J.; Salinas-Riester, G.; Pommerenke, C.; Dittert, K.; Schulze, J. RNA-seq transcriptome profiling reveals that Medicago truncatula nodules acclimate N2 fixation before emerging P deficiency reaches the nodules. J. Exp. Bot. 2014, 65, 6035–6048. [Google Scholar] [CrossRef]
- Ruffel, S.; Freixes, S.; Balzergue, S.; Tillard, P.; Jeudy, C.; Martin-Magniette, M.L.; van der Merwe, M.J.; Kakar, K.; Gouzy, J.; Fernie, A.R.; et al. Systemic signaling of the plant nitrogen status triggers specific transcriptome responses depending on the nitrogen source in Medicago truncatula. Plant Physiol. 2008, 146, 2020–2035. [Google Scholar] [CrossRef]
- Van de Velde, W.; Zehirov, G.; Szatmari, A.; Debreczeny, M.; Ishihara, H.; Kevei, Z.; Farkas, A.; Mikulass, K.; Nagy, A.; Tiricz, H.; et al. Plant peptides govern terminal differentiation of bacteria in symbiosis. Science 2010, 327, 1122–1126. [Google Scholar] [CrossRef]
- Haag, A.F.; Arnold, M.F.F.; Myka, K.K.; Kerscher, B.; Dall’Angelo, S.; Zanda, M.; Mergaert, P.; Ferguson, G.P. Molecular insights into bacteroid development during Rhizobium–legume symbiosis. FEMS Microbiol. Rev. 2013, 37, 364–383. [Google Scholar] [CrossRef]
- Graham, P.H.; Vance, C.P. Legumes: Importance and constraints to greater use. Plant Physiol. 2003, 131, 872–877. [Google Scholar] [CrossRef]
- Israel, D.W. Investigation of the role of phosphorus in symbiotic dinitrogen fixation. Plant Physiol. 1987, 84, 835–840. [Google Scholar] [CrossRef]
- Robson, A.; O’hara, G.; Abbott, L. Involvement of phosphorus in nitrogen fixation by subterranean clover (Trifolium subterraneum L.). Funct. Plant Biol. 1981, 8, 427–436. [Google Scholar] [CrossRef]
- Schulze, J.; Temple, G.; Temple, S.J.; Beschow, H.; Vance, C.P. Nitrogen fixation by white lupin under phosphorus deficiency. Ann. Bot. 2006, 98, 731–740. [Google Scholar] [CrossRef]
- Israel, D.W. Symbiotic dinitrogen fixation and host-plant growth during development of and recovery from phosphorus deficiency. Physiol. Plant. 1993, 88, 294–300. [Google Scholar] [CrossRef]
- Hernández, G.; Valdés-López, O.; Ramírez, M.; Goffard, N.; Weiller, G.; Aparicio-Fabre, R.; Fuentes, S.I.; Erban, A.; Kopka, J.; Udvardi, M.K.; et al. Global changes in the transcript and metabolic profiles during symbiotic nitrogen fixation in phosphorus-stressed common bean plants. Plant Physiol. 2009, 151, 1221–1238. [Google Scholar] [CrossRef]
- Ramírez, M.; Flores-Pacheco, G.; Reyes, J.L.; Luz Alvarez, A.; Drevon, J.J.; Girard, L.; Hernández, G. Two common bean genotypes with contrasting response to phosphorus deficiency show variations in the microRNA 399-mediated PvPHO2 regulation within the PvPHR1 signaling pathway. Int. J. Mol. Sci. 2013, 14, 8328–8344. [Google Scholar] [CrossRef]
- Vance, C.P. Quantitative trait loci, epigenetics, sugars, and microRNAs: Quaternaries in phosphate acquisition and use. Plant Physiol. 2010, 154, 582–588. [Google Scholar] [CrossRef]
- Esfahani, M.N.; Kusano, M.; Nguyen, K.H.; Watanabe, Y.; Ha, C.V.; Saito, K.; Sulieman, S.; Herrera-Estrella, L.; Tran, L.-S.P. Adaptation of the symbiotic Mesorhizobium—Chickpea relationship to phosphate deficiency relies on reprogramming of whole-plant metabolism. Proc. Natl. Acad. Sci. USA 2016, 113, E4610–E4619. [Google Scholar] [CrossRef]
- Stevens, G.G.; Pérez-Fernández, M.A.; Morcillo, R.J.L.; Kleinert, A.; Hills, P.; Brand, D.J.; Steenkamp, E.T.; Valentine, A.J. Roots and Nodules Response Differently to P Starvation in the Mediterranean-Type Legume Virgilia divaricata. Front. Plant Sci. 2019, 10, 73. [Google Scholar] [CrossRef]
- Nizampatnam, N.R.; Schreier, S.J.; Damodaran, S.; Adhikari, S.; Subramanian, S. microRNA160 dictates stage-specific auxin and cytokinin sensitivities and directs soybean nodule development. Plant J. 2015, 84, 140–153. [Google Scholar] [CrossRef]
- Yan, Z.; Hossain, M.S.; Wang, J.; Valdés-López, O.; Liang, Y.; Libault, M.; Qiu, L.; Stacey, G. miR172 regulates soybean nodulation. Mol. Plant. Microbe Interact. 2013, 26, 1371–1377. [Google Scholar] [CrossRef]
- Chen, X. microRNA biogenesis and function in plants. FEBS Lett. 2005, 579, 5923–5931. [Google Scholar] [CrossRef]
- Jones-Rhoades, M.W.; Bartel, D.P.; Bartel, B. MicroRNAs and their regulatory roles in plants. Annu. Rev. Plant Biol. 2006, 57, 19–53. [Google Scholar] [CrossRef]
- Alves-Junior, L.; Niemeier, S.; Hauenschild, A.; Rehmsmeier, M.; Merkle, T. Comprehensive prediction of novel microRNA targets in Arabidopsis thaliana. Nucleic Acids Res. 2009, 37, 4010–4021. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Q.; Zhang, J.; Wu, L.; Qi, Y.; Zhou, J.M. Identification of microRNAs involved in pathogen-associated molecular pattern-triggered plant innate immunity. Plant Physiol. 2010, 152, 2222–2231. [Google Scholar] [CrossRef]
- Covarrubias, A.A.; Reyes, J.L. Post-transcriptional gene regulation of salinity and drought responses by plant microRNAs. Plant Cell Environ. 2010, 33, 481–489. [Google Scholar] [CrossRef]
- Dong, Z.; Shi, L.; Wang, Y.; Chen, L.; Cai, Z.; Wang, Y.; Jin, J.; Li, X. Identification and dynamic regulation of microRNAs involved in salt stress responses in functional soybean nodules by high-throughput sequencing. Int. J. Mol. Sci. 2013, 14, 2717–2738. [Google Scholar] [CrossRef]
- Chiou, T.-J.; Aung, K.; Lin, S.-I.; Wu, C.-C.; Chiang, S.-F.; Su, C. Regulation of phosphate homeostasis by microRNA in Arabidopsis. Plant Cell Online 2006, 18, 412–421. [Google Scholar] [CrossRef]
- Abdel-Ghany, S.E.; Pilon, M. MicroRNA-mediated systemic down-regulation of copper protein expression in response to low copper availability in Arabidopsis. J. Biol. Chem. 2008, 283, 15932–15945. [Google Scholar] [CrossRef]
- Kawashima, C.G.; Yoshimoto, N.; Maruyama-Nakashita, A.; Tsuchiya, Y.N.; Saito, K.; Takahashi, H.; Dalmay, T. Sulphur starvation induces the expression of microRNA-395 and one of its target genes but in different cell types. Plant J. 2009, 57, 313–321. [Google Scholar] [CrossRef]
- Chiou, T.-J.; Lin, S.-I. Signaling network in sensing phosphate availability in plants. Annu. Rev. Plant Biol. 2011, 62, 185–206. [Google Scholar] [CrossRef]
- Gifford, M.L.; Dean, A.; Gutierrez, R.A.; Coruzzi, G.M.; Birnbaum, K.D. Cell-specific nitrogen responses mediate developmental plasticity. Proc. Natl. Acad. Sci. USA 2008, 105, 803–808. [Google Scholar] [CrossRef]
- Vidal, E.A.; Araus, V.; Lu, C.; Parry, G.; Green, P.J.; Coruzzi, G.M.; Gutiérrez, R.A. Nitrate-responsive miR393/AFB3 regulatory module controls root system architecture in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2010. [Google Scholar] [CrossRef]
- Combier, J.-P.; Frugier, F.; de Billy, F.; Boualem, A.; El-Yahyaoui, F.; Moreau, S.; Vernié, T.; Ott, T.; Gamas, P.; Crespi, M.; et al. MtHAP2-1 is a key transcriptional regulator of symbiotic nodule development regulated by microRNA169 in Medicago truncatula. Genes Dev. 2006, 20, 3084–3088. [Google Scholar] [CrossRef]
- Boualem, A.; Laporte, P.; Jovanovic, M.; Laffont, C.; Plet, J.; Combier, J.-P.; Niebel, A.; Crespi, M.; Frugier, F. MicroRNA166 controls root and nodule development in Medicago truncatula. Plant J. 2008, 54, 876–887. [Google Scholar] [CrossRef]
- Bustos-Sanmamed, P.; Mao, G.; Deng, Y.; Elouet, M.; Khan, G.A.; Bazin, J.; Turner, M.; Subramanian, S.; Yu, O.; Crespi, M.; et al. Overexpression of miR160 affects root growth and nitrogen-fixing nodule number in Medicago truncatula. Funct. Plant Biol. 2013, 40, 1208–1220. [Google Scholar] [CrossRef]
- D’haeseleer, K.; Den Herder, G.; Laffont, C.; Plet, J.; Mortier, V.; Lelandais-Brière, C.; De Bodt, S.; De Keyser, A.; Crespi, M.; Holsters, M.; et al. Transcriptional and post-transcriptional regulation of a NAC1 transcription factor in Medicago truncatula roots. New Phytol. 2011, 191, 647–661. [Google Scholar] [CrossRef]
- Wang, Y.; Li, K.; Chen, L.; Zou, Y.; Liu, H.; Tian, Y.; Li, D.; Wang, R.; Zhao, F.; Ferguson, B.J.; et al. MicroRNA167-directed regulation of the auxin response factors GmARF8a and GmARF8b is required for soybean nodulation and lateral root development. Plant Physiol. 2015, 168, 984–999. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwember, A.R.; Schulze, J.; del Pozo, A.; Cabeza, R.A. Regulation of Symbiotic Nitrogen Fixation in Legume Root Nodules. Plants 2019, 8, 333. https://doi.org/10.3390/plants8090333
Schwember AR, Schulze J, del Pozo A, Cabeza RA. Regulation of Symbiotic Nitrogen Fixation in Legume Root Nodules. Plants. 2019; 8(9):333. https://doi.org/10.3390/plants8090333
Chicago/Turabian StyleSchwember, Andrés R., Joachim Schulze, Alejandro del Pozo, and Ricardo A. Cabeza. 2019. "Regulation of Symbiotic Nitrogen Fixation in Legume Root Nodules" Plants 8, no. 9: 333. https://doi.org/10.3390/plants8090333
APA StyleSchwember, A. R., Schulze, J., del Pozo, A., & Cabeza, R. A. (2019). Regulation of Symbiotic Nitrogen Fixation in Legume Root Nodules. Plants, 8(9), 333. https://doi.org/10.3390/plants8090333





