Bacillus Spp.: Efficient Biotic Strategy to Control Postharvest Diseases of Fruits and Vegetables
Abstract
1. Introduction
2. Bacillus spp. Capacity to Alleviate Postharvest Losses of Fruits and Vegetables
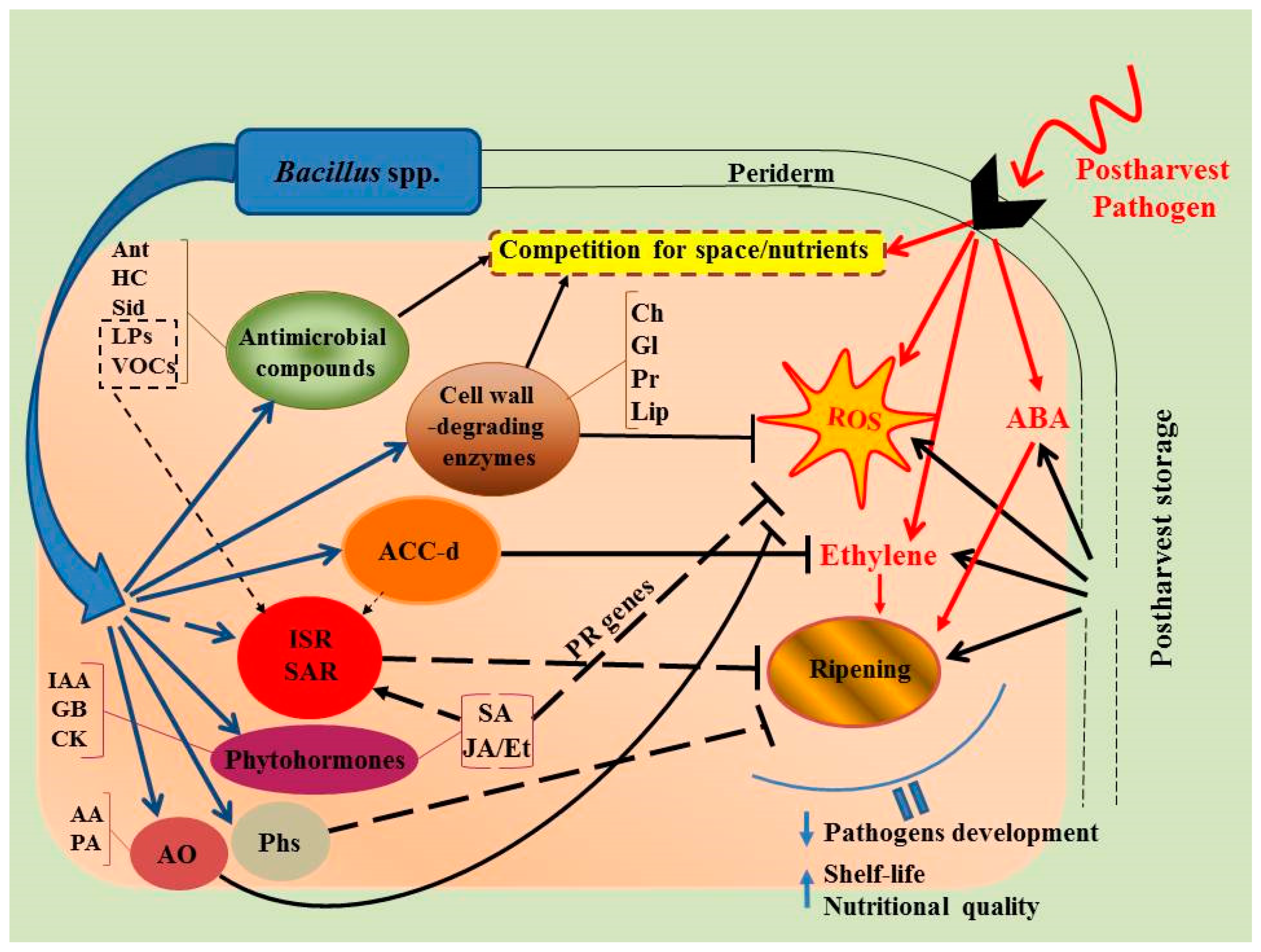
3. Potential Modes of Action of Microbial Antagonists
3.1. Competition for Space and Nutrients
3.2. Production of Antimicrobial Compounds
3.3. Synthesis of Hydrolytic Enzymes
3.4. Induction of Systemic Resistance in Host Plant Organisms
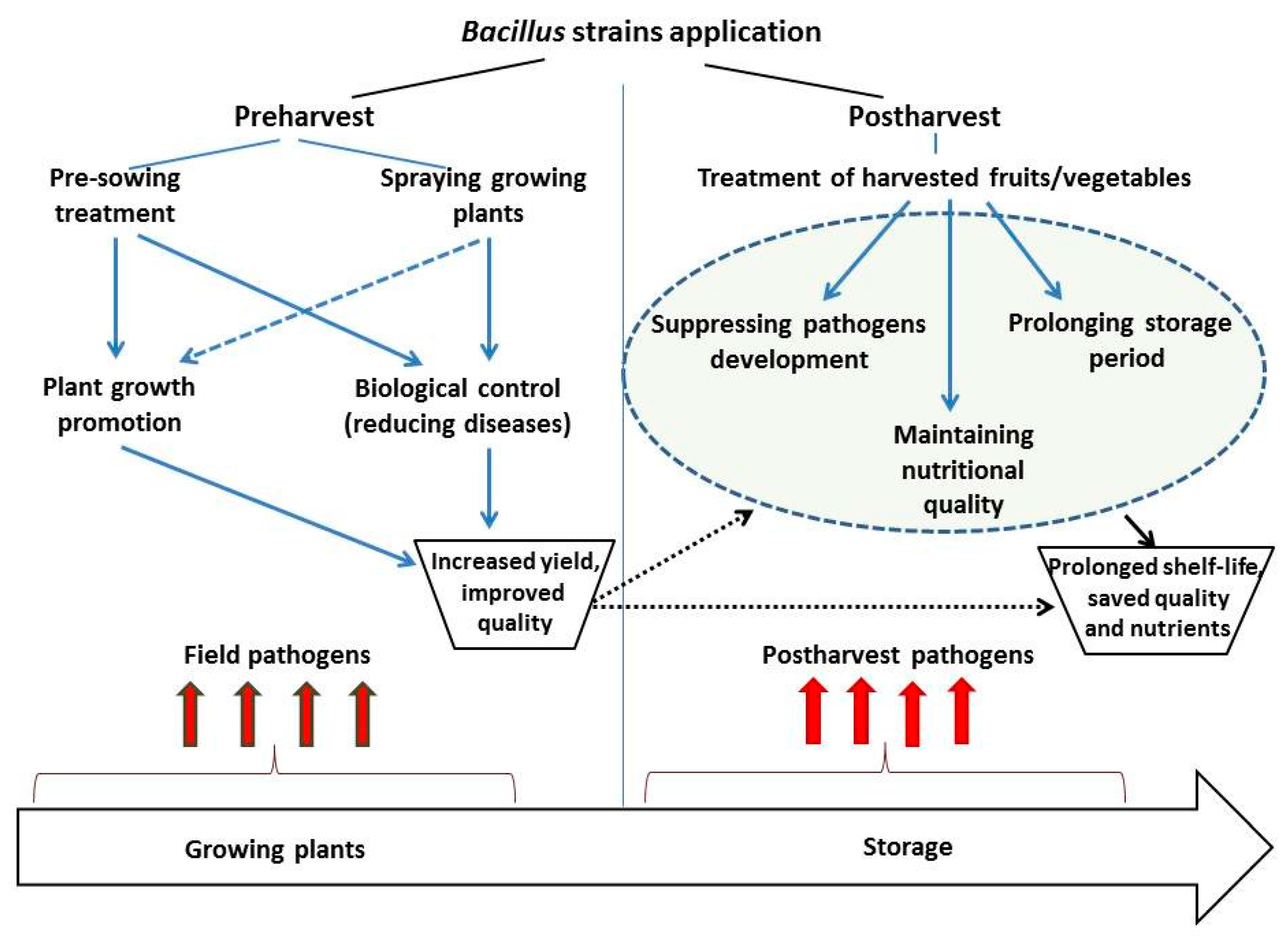
4. Methods of Application
4.1. Preharvest Application
4.2. Postharvest Application
5. Development and Commercialization of Bacillus-Based Postharvest Biocontrol Products
6. Conclusions and Future Prospects
Author Contributions
Funding
Conflicts of Interest
References
- FAO. Food Losses and Waste. 2015. Available online: http://www.fao.org/food-loss-and-food-waste/en/ (accessed on 21 September 2018).
- Hodges, R.J.; Buzby, J.C.; Bennett, B. Postharvest losses and waste in developed and less developed countries: Opportunities to improve resource use. J. Agric. Sci. 2010, 149, 37–45. [Google Scholar] [CrossRef]
- Buchholz, F.; Kostic, T.; Sessitsch, A.; Mitter, B. The potential of plant microbiota in reducing postharvest food loss. Microb. Biotechnol. 2018, 11, 971–975. [Google Scholar] [CrossRef]
- Droby, S.; Wisniewski, M.; Teixidó, N.; Spadaro, D.; Jijakli, M.H. The science, development, and commercialization of postharvest biocontrol products. Postharvest Biol. Technol. 2016, 122, 22–29. [Google Scholar] [CrossRef]
- Wisniewski, M.; Droby, S.; John, N.; Liu, J.; Schena, L. Alternative management technologies for postharvest disease control: The journey from simplicity to complexity. Postharvest Biol. Technol. 2016, 122, 3–10. [Google Scholar] [CrossRef]
- Dimkpa, C.O.; Merten, D.; Svatos, A.; Büchel, G.; Kothe, E. Siderophores mediate reduced and increased uptake of cadmium by Streptomyces tendae F4 and sunflower (Helianthus annuus), respectively. J. Appl. Microbiol. 2009, 107, 1687–1696. [Google Scholar] [CrossRef] [PubMed]
- Arroyave-Toroa, J.J.; Mosquera, S.; Villegas-Escobar, V. Biocontrol activity of Bacillus subtilis EA-CB0015 cells and lipopeptides against postharvest fungal pathogens. Biol. Control 2017, 114, 195–200. [Google Scholar] [CrossRef]
- Sarma, B.K.; Yadav, K.S.; Singh, D.P.; Singh, H.B. Rhizobacteria mediated induced systemic tolerance in plants: Prospects for abiotic stress management. In Bacteria in Agrobiology: Stress Management; Springer: Berlin/Heidelberg, Germany, 2012; pp. 225–238. [Google Scholar]
- Van Loon, L.C. Plant responses to plant growth-promoting rhizobacteria. Eur. J. Plant Pathol. 2007, 119, 243–254. [Google Scholar] [CrossRef]
- Baez-Rogelio, A.; Morales-García, Y.E.; Quintero-Hernández, V.; Muñoz-Rojas, J. Next generation of microbial inoculants for agriculture and bioremediation. Microb. Biotechnol. 2016, 10, 19–21. [Google Scholar] [CrossRef]
- Lastochkina, O.; Pusenkova, L.; Yuldashev, R.; Babaev, M.; Garipova, S.; Blagova, D.; Khairullin, R.; Aliniaeifard, S. Effects of Bacillus subtilis on some physiological and biochemical parameters of Triticum aestivum L. (wheat) under salinity. Plant Physiol. Biochem. 2017, 121, 80–88. [Google Scholar] [CrossRef]
- Seifikalhor, M.S.; Aliniaeifard, S.; Self, M.; Javadi, E.; Bernard, F.; Li, T.; Lastochkina, O. Rhisobacteria Bacillus subtilis reduces toxic effects of high electrical conductivity in soilless culture of lettuce. Acta Hortic. 2018. [Google Scholar] [CrossRef]
- Maksimov, I.V.; Veselova, S.V.; Nuzhnaya, T.V.; Sarvarova, E.R.; Khairullin, R.M. Plant growth promoting bacteria in regulation of plant resistance to stress factors. Rus. J. Plant Physiol. 2015, 62, 715–726. [Google Scholar] [CrossRef]
- Nagórska, K.; Bikowski, M.; Obuchowski, M. Multicellular behaviour and production of a wide variety of toxic substances support usage of Bacillus subtilis as a powerful biocontrol agent. Acta Biochim. Pol. 2007, 54, 495–508. [Google Scholar]
- Ongena, M.; Jacques, P. Bacillus lipopeptides: Versatile weapons for plant disease biocontrol. Trends Microbiol. 2008, 16, 115–125. [Google Scholar] [CrossRef]
- Gao, H.; Xu, X.; Dai, Y.; He, H. Isolation, identification and characterization of Bacillus subtilis CF-3, a bacterium from fermented bean curd for controlling postharvest diseases of peach fruit. Food Sci. Technol. Res. 2016, 22, 377–385. [Google Scholar] [CrossRef][Green Version]
- Knox, O.G.G.; Killham, K.; Leifert, C. Effects of increased nitrate availability on the control of plant pathogenic fungi by the soil bacterium Bacillus subtilis. Appl. Soil Ecol. 2000, 15, 227–231. [Google Scholar] [CrossRef]
- Mannanov, R.N.; Sattarova, R.K. Antibiotics produced by Bacillus bacteria. Chem. Nat. Compd. 2001, 37, 117–123. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Zhu, X.R.; Li, Y.B. Postharvest control of litchi fruit rot by Bacillus subtilis. Food Sci. Technol. 2001, 34, 430–436. [Google Scholar] [CrossRef]
- Leelasuphakul, W.; Sivanunsakul, P.; Phongpaichit, S. Purification, characterization and synergistic activity of b1,3-glucanase and antibiotic extract from an antagonistic Bacillus subtilis NSRS 89-24 against rice blast and sheath blight pathogens. Enzyme Microb. Technol. 2006, 38, 990–997. [Google Scholar] [CrossRef]
- Aouadhi, C.; Rouissi, Z.; Kmiha, S.; Mejri, S.; Maaroufi, A. Effect of sporulation conditions on the resistance of Bacillus sporothermodurans spores to nisin and heat. Food Microbiol. 2016, 54, 6–10. [Google Scholar] [CrossRef]
- Gupta, V.; Bochow, H.; Dolej, S.; Dolej, S.; Fischer, I. Plant growth-promoting Bacillus subtilis strain as potential inducer of systemic resistance in tomato against Fusarium wilt. J. Plant Dis. Protect. 2000, 107, 145–154. [Google Scholar]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L. Plant growth-promoting rhizobacteria (PGPR): Their potential as antagonists and biocontrol agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- García-Gutiérrez, L.; Zeriouh, H.; Romero, D.; Cubero, J.; Vicente, A.; Pérez-García, A. The antagonistic strain Bacillus subtilis UMAF6639 also confers protection to melon plants against cucurbit powdery mildew by activation of jasmonate—And salicylic acid-dependent defense responses. Microb. Biotechnol. 2013, 6, 264–274. [Google Scholar] [CrossRef]
- Pusenkova, L.I.; Il’yasova, E.Y.; Lastochkina, O.V.; Maksimov, I.V.; Leonova, S.A. Changes in the species composition of the rhizosphereand phyllosphere of sugar beet under the impact of biological preparations based on endophytic bacteria and their metabolites. Eurasian Soil Sci. 2016, 49, 1136–1144. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Wirth, S.J.; Shurigin, V.V.; Hashem, A.; Abd Allah, E.F. Endophytic bacteria improve plant growth, symbiotic performance of chickpea (Cicer arietinum L.) and induce suppression of root rot caused by Fusarium solani under salt stress. Front. Microbiol. 2017, 8, 1887. [Google Scholar] [CrossRef]
- Shafi, O.; Tian, H.; Ji, M. Bacillus species as versatile weapons for plant pathogens: A review. Biotechnol. Biotechnol. Equip. 2017, 31, 446–459. [Google Scholar] [CrossRef]
- Bochow, H.; El-Sayed, S.F.; Junge, H.; Stavropoulou, A.; Schmiedeknecht, G. Use of Bacillus subtilis as biocontrol agent. IV. Salt-stress tolerance induction by Bacillus subtilis FZB24 seed treatment in tropical vegetable field crops, and its mode of action. Zeitschrift fur Pflanzenkrankheiten und Pflanzenschutz. 2001, 108, 21–30. [Google Scholar]
- Saleh, S.A.; Heuberger, H.; Schnitzler, W.H. Alleviation of salinity effect on artichoke productivity by Bacillus subtilis FZB24, supplemental Ca and micronutrients. J. Appl. Bot. Food Qual. 2005, 79, 24–32. [Google Scholar]
- Turan, M.; Ekinci, M.; Yıldırım, E.; Güneş, K.; Karagöz, K.; Kotan, R.; Dursun, A. Plant growth-promoting rhizobacteria improved growth, nutrient, and hormone content in cabbage (Brassica oleracea) seedlings. Turk. J. Agric. For. 2014, 38, 327–333. [Google Scholar] [CrossRef]
- Berg, G. Plant-microbe interactions promoting plant growth and health: Perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Biotechnol. 2009, 84, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.K.; Singh, M.C.; Singh, S.S.; Kumar, A.K.; Pathak, M.M.; Shakywar, R.C.; Pandey, A.K. Inside the plants: Endophytic bacteria and their functional attributes for plant growth promotion. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 11–21. [Google Scholar] [CrossRef]
- Jiang, Y.M.; Chen, F.; Li, Y.B.; Liu, S.X. A preliminary study on the biological control of postharvest diseases of Litchi fruit. J. Fruit Sci. 2001, 14, 185–186. [Google Scholar]
- Wang, Y.; Xu, Z.; Zhu, P.; Liu, P.; Zhang, Z.; Mastuda, Y.; Toyoda, H.; Xu, L. Postharvest biological control of melon pathogens using Bacillus subtilis EXWB1. J. Plant Pathol. 2010, 92, 645–652. [Google Scholar]
- Kim, G.H.; Koh, Y.J.; Jung, J.S.; Hur, J.S. Control of postharvest fruit rot diseases of kiwifruit by antagonistic bacterium Bacillus subtilis. Acta Hortic. 2015, 1096, 377–382. [Google Scholar] [CrossRef]
- Kim, Y.S.; Balaraju, K.; Jeon, Y. Effects of rhizobacteria Paenibacillus polymyxa APEC136 and Bacillus subtilis APEC170 on biocontrol of postharvest pathogens of apple fruits. J. Zhejiang Univ. Sci. B 2016, 17, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Deverall, B.J. Bacillus subtilis as a control agent against fungal pathogens of citrus fruit. Trans. Br. Mycol. Soc. 1984, 83, 487–490. [Google Scholar] [CrossRef]
- Mari, M.; Guizzardi, M.; Pratella, G.C. Biological control of gray mold in pears by antagonistic bacteria. Biol. Control 1996, 7, 30–37. [Google Scholar] [CrossRef]
- Touré, Y.; Ongena, M.; Jacques, P.; Guiro, A.; Thonart, P. Role of lipopeptides produced by Bacillus subtilis GA1 in the reduction of grey mould disease caused by Botrytis cinerea on apple. J. Appl. Microbiol. 2004, 96, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shao, X.F.; Tu, K.; Chen, J.K. Inhibitory effect of Bacillus subtilis B10 on the diseases of postharvest strawberry. J. Fruit Sci. 2007, 24, 339–343. [Google Scholar]
- Jamalizadeh, M.; Etebarian, H.R.; Aminian, H.; Alizadeh, A. Biological control of Botrytis mali on apple fruit by use of Bacillus bacteria, isolated from the rhizosphere of wheat. Arch. Phytopathol. Plant Protect. 2010, 43, 1836–1845. [Google Scholar]
- Kilani-Feki, O.; Ben Khedher, S.; Dammak, M.; Kamoun, A.; Jabnoun-Khiareddine, H.; Daami-Remadi, M.; Touns, S. Improvement of antifungal metabolites production by Bacillus subtilis V26 for biocontrol of tomato postharvest disease. Biol. Control 2016, 95, 73–82. [Google Scholar] [CrossRef]
- Miller, A.R. Harvest and Handling Injury: Physiology, Biochemistry, and Detection. In Postharvest Physiology and Pathology of Vegetables; Marcel Dekker Inc.: New York, NY, USA, 2003. [Google Scholar]
- Tronsmo, A.; Denis, C. The use of Trichoderma species to control strawberry fruit rots. Neth. J. Plant Pathol. 1977, 83, 449–455. [Google Scholar] [CrossRef]
- Pusey, P.L.; Wilson, C.L. Postharvest biological control of stone fruit brown rot by Bacillus subtilis. Plant Dis. 1984, 68, 753–756. [Google Scholar] [CrossRef]
- Mohammadi, P.; Tozlu, E.; Kotan, R.; Şenol Kotan, M. Potential of some bacteria for biological control of postharvest citrus green mould caused by Penicillium digitatum. Plant Protect. Sci. 2017, 53, 134–143. [Google Scholar]
- Jamalizadeh, M.; Etebarian, H.R.; Alizadeh, A.A.; Aminian, H. Biological control of gray mold on apple fruits by Bacillus licheniformis (EN74-1). Phytoparasitica 2008, 36, 23. [Google Scholar] [CrossRef]
- Alfonzo, A.; Conigliaro, G.; Torta, L.; Burruano, S.; Moschetti, G. Antagonism of Bacillus subtilis strain AG1 against vine wood fungal pathogens. Phytopathol. Mediterr. 2009, 48, 155–158. [Google Scholar]
- Fan, H.; Ru, J.; Zhang, Y.; Wang, Q.; Li, Y. Fengycin produced by Bacillus subtilis 9407 plays a major role in the biocontrol of apple ring rot disease. Microbiol. Res. 2017, 199, 89–97. [Google Scholar] [CrossRef]
- Okigbo, R.N. Biological control of postharvest fungal rot of yam (Dioscorea spp.) with Bacillus subtilis. Mycopathologia 2005, 159, 307–314. [Google Scholar] [CrossRef]
- Qi, D.; Hui, M.; Liang, Q.; Niu, T. Postharvest biological control of blue mold and black spot on apple-pear (Pyrus bretschneideri Rehd.) fruit by Bacillus subtilis H110. Chin. J. Appl. Environ. Biol. 2005, 11, 171–174. [Google Scholar]
- Utkhede, R.S.; Sholberg, P.L. In vitro inhibition of plant pathogens: Bacillus subtilis and Enterobacter aerogenes in vivo control of two postharvest cherry diseases. Can. J. Microbiol. 1986, 32, 963–967. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J.; Jin, P.; Zheng, Y. Investigating the efficacy of Bacillus subtilis SM21 on controlling Rhizopus rot in peach fruit. Int. J. Food Microbiol. 2013, 164, 141–147. [Google Scholar] [CrossRef]
- Korsten, L.; De Villiers, E.E.; De Jager, E.S.; Cook, N.; Kotzé, J.M. Biological control of avocado postharvest diseases. In South African Avocado Growers’ Association Yearbook; University of Pretoria: Tzaneen, South Africa, 1991; Volume 14, pp. 57–59. [Google Scholar]
- Chebotar, V.K.; Kiprushkina, E.I. Application of microbial preparations in potato storage technologies. Dostizheniya Nauki i Tekhniki APK 2015, 29, 33–35. (In Russian) [Google Scholar]
- Kavino, M.; Manoranjitham, S.K.; Vijayakumar, N.K.R. Plant growth stimulation and biocontrol of Fusarium wilt (Fusarium oxysporium f. sp. cubene) by coinoculation of banana (Musa sp.) plantlets with PGPR and endophytes. In Recent Trends in PGPR Research for Sustainable Crop Productivity, Proceedings of the 4th Asian PGPR Conference, Hanoi, Vietnam, 3–6 May 2016; Scientific Publishers: Jodhpur, India, 2016. [Google Scholar]
- Maksimov, I.V.; Khairullin, R.M. The role of Bacillus bacterium in formation of plant defense: Mechanism and reaction. In The Handbook of Microbial Bioresourses; Gupta, V.K., Sharma, G.D., Tuohy, M.G., Gaur, R., Eds.; CAB International: Galway, Irish Republic, 2016; Chapter 4; pp. 56–80. [Google Scholar]
- Rahman, S.; Rahman, L.; Khalil, A.T.; Ali, N.; Zia, D.; Ali, M.; Shinwari, Z.K. Endophyte-mediated synthesis of silver nanoparticles and their biological applications. Appl. Microbiol. Biotechnol. 2019, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Maksimov, I.V.; Pusenkova, L.I.; Abizgildina, R.R. Biopreparation with endophytic bacterium Bacillus subtilis 26D created postharvest protecting effect in potato tubers. Agrochemistry 2011, 6, 43–48. (In Russian) [Google Scholar]
- Lastochkina, O.V.; Yuldashev, R.A.; Pusenkova, L.I. Assessment of the influence of Bacillus subtilis bacterial strains in mix with salicylic acid on productivity and infection of potato tubers. Agric. Sci. Innov. Dev. AIC 2015, 1, 112–117. (In Russian) [Google Scholar]
- Aghdam, M.S.; Asghari, M.; Babalar, M.; Sarcheshmeh, M.A.A. Impact of salicylic acid on postharvest physiology of fruits and vegetables. In Eco-Friendly Technology for Postharvest Produce Quality; Academic Press: Bihar, India, 2016; pp. 243–268. [Google Scholar] [CrossRef]
- Fung, R.; Wang, C.; Smith, D.; Gross, K.; Tian, M. MeSA and MeJA increase steady-state transcript levels of alternative oxidase and resistance against chilling injury in sweet peppers (Capsicum annuum L.). Plant Sci. 2004, 166, 711–719. [Google Scholar] [CrossRef]
- Cai, C.; Xu, C.J.; Li, X.; Ferguson, I.; Chen, K.S. Accumulation of lignin in relation to change in activities of lignification enzymes in loquat fruit flesh after harvest. Postharvest Biol. Technol. 2006, 40, 163–169. [Google Scholar] [CrossRef]
- Demoz, B.T.; Korsten, L. Bacillus subtilis attachment, colonization, and survival on avocado flowers and its mode of action on stem-end rot pathogens. Biol. Control 2006, 37, 68–74. [Google Scholar] [CrossRef]
- Yang, D.M.; Bi, Y.; Chen, X.R.; Ge, Y.H.; Zhao, J. Biological control of postharvest diseases with Bacillus subtilis (B1 strain) on muskmelons (Cucumis melo L. cv. Yindi). Acta Hortic. 2006, 712, 735–739. [Google Scholar] [CrossRef]
- Punjia, Z.K.; Rodriguez, G.; Tirajoh, A. Effects of Bacillus subtilis strain QST 713 and storage temperatures on post-harvest disease development on greenhouse tomatoes. Crop Prot. 2016, 84, 98–104. [Google Scholar] [CrossRef]
- Zhou, T.; Schneider, K.E.; Li, X. Development of biocontrol agents from food microbial isolates for controlling post-harvest peach brown rot caused by Monilinia fructicola. Int. J. Food Microbiol. 2008, 126, 180–185. [Google Scholar] [CrossRef]
- Yánez-Mendizábal, V.; Zeriouh, H.; Viñas, I.; Torres, R.; Usall, J.; de Vicente, A.; Pérez-García, A.; Teixidó, N. Biological control of peach brown rot (Monilinia spp.) by Bacillus subtilis CPA-8 is based on production of fengycin-like lipopeptides. Eur. J. Plant Pathol. 2012, 132, 609–619. [Google Scholar] [CrossRef]
- Gotor-Vila, A.; Usall, J.; Torres, R.; Solsona, C.; Teixidó, N. Biocontrol products based on Bacillus amyloliquefaciens CPA-8 using fluid-bed spray-drying process to control postharvest brown rot in stone fruit. LWT Food Sci. Technol. 2017, 82, 274–282. [Google Scholar] [CrossRef][Green Version]
- Govender, V.; Korsten, L.; Sivakumar, D. Semi-commercial evaluation of Bacillus licheniformis to control mango postharvest diseases in South Africa. Postharvest Biol. Technol. 2005, 38, 57–65. [Google Scholar] [CrossRef]
- Droby, S.; Chalutz, E.; Wilson, C.L.; Wisniewski, M.E. Biological control of postharvest diseases: A promising alternative to the use of synthetic fungicides. Phytoparasitica 1992, 20, 1495–1503. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Tworkoski, T.J.; Sharer, C. Characterizing the mechanism of biological control of postharvest diseases on fruit with a simple method to study competition for nutrients. Phytopathology 2000, 90, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Jijakli, M.H.; Grevesse, C.; Lepoivre, P. Modes of action of biocontrol agents of postharvest diseases: Challenges and difficulties. Bulletin-OILB/SROP 2001, 24, 317–318. [Google Scholar]
- El-Ghaouth, A.; Wilson, C.L.; Wisniewski, M.E. Biologically based alternatives to synthetic fungicides for the postharvest diseases of fruit and vegetables. In Diseases of Fruit and Vegetables; Samh, N., Ed.; Kluwen Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 511–535. [Google Scholar]
- Cawoy, H.; Debois, D.; Franzil, L.; De Pauw, E.; Thonart, P.; Ongena, M. Lipopeptides as main ingredients for inhibition of fungal phytopathogens by Bacillus subtilis/amyloliquefaciens. Microb. Biotechnol. 2015, 8, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Baindara, P.; Korpole, S. Lipopeptides: Status and strategies to control fungal infection. In Recent Trends in Antifungal Agents and Antifungal Therapy; Springer: New Delhi, India, 2016; pp. 97–121. [Google Scholar]
- Van der Ent, S.S.; Van Wees, S.C.M.; Pieterse, C.M.J. Jasmonate signaling in plant interactions with resistance-inducing beneficial microbes. Phytochemistry 2009, 70, 1581–1588. [Google Scholar] [CrossRef] [PubMed]
- Maksimov, I.V.; Abizgildina, R.R.; Pusenkova, L.I. Plant growth promoting rhizobacteria as alternative to chemical crop protectors from pathogens (Review). Appl. Biochem. Microbiol. 2011, 47, 333–345. [Google Scholar] [CrossRef]
- De Vleesschauwer, D.; Höfte, M. Rhizobacteria-Induced Systemic Resistance. Adv. Bot. Res. 2009, 51, 223–281. [Google Scholar] [CrossRef]
- Bacon, W.C.; Yates, E.I.; Hinton, M.D.; Meredith, F. Biological control of Fusarium moniliforme in maize. Environ. Health Perspect. 2001, 109, 325–332. [Google Scholar]
- Porcel, R.; Zamarreño, A.M.; García-Mina, J.M.; Aroca, R. Involvement of plant endogenous ABA in Bacillus megaterium PGPR activity in tomato plants. BMC Plant Biol. 2014, 14, 36. [Google Scholar] [CrossRef]
- Wilson, C.L.; Wisniewski, M.E. Biological control of postharvest diseases of fruit and vegetables: An emerging technology. Annu. Rev. Phytopathol. 1989, 27, 425–441. [Google Scholar] [CrossRef]
- Droby, S.; Chalutz, E. Mode of action of biological agents of postharvest diseases. In Biological Control of Postharvest Diseases—Theory and Practice; Wilson, C.L., Wisniewski, M.E., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 63–75. [Google Scholar]
- Droby, S.; Chalutz, E.; Wilson, C.L.; Wisniewski, M. Characterization of the biocontrol activity of Debaryomyces hansenii in the control of Penicillium digitatum on grapefruit. Can. J. Microbiol. 1989, 35, 794–800. [Google Scholar] [CrossRef]
- Castoria, R.; de Curtis, F.; Lima, G.; Caputo, L.; Pacifico, S.; de Cicco, V. Aureobasidium pullulans (LS-30), an antagonist of postharvest pathogens of fruits: Study on its mode of action. Postharvest Biol. Technol. 2001, 32, 717–724. [Google Scholar] [CrossRef]
- Mardanova, A.M.; Hadieva, G.F.; Lutfullin, M.T.; Khilyas, I.V.; Minnullina, L.F.; Gilyazeva, A.G.; Bogomolnaya, L.M.; Sharipova, M.R. Bacillus subtilis strains with antifungal activity against the phytopathogenic fungi. Agric. Sci. 2017, 8, 1–20. [Google Scholar] [CrossRef]
- Stein, T. Bacillus subtilis antibiotics: Structures, syntheses and specific functions. Mol. Microbiol. 2005, 56, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Kloepper, J.W.; Gutierrez Estrada, A.; McInroy, J.A. Photoperiod regulates elicitation of growth promotion but not induced resistance by plant growth promoting rhizobacteria. Can. J. Microbiol. 2009, 53, 159–167. [Google Scholar] [CrossRef]
- Duffy, B.; Schouten, A.; Raaijmakers, J.M. Pathogen selfdefense: Mechanisms to counteract microbial antagonism. Annu. Rev. Phytopathol. 2003, 41, 501–538. [Google Scholar] [CrossRef]
- Chen, X.-H.; Scholz, R.; Borriss, M.; Junge, H.; Mögel, G.; Kunz, S.; Borris, R. Difficidin and bacilysin produced by plant-associated Bacillus amyloliquefaciens are efficient in controlling fire blight disease. J. Biotechnol. 2009, 140, 38–44. [Google Scholar] [CrossRef]
- Ongena, M.; Jourdan, E.; Adam, A.; Paquot, M.; Brans, A.; Joris, B.; Arpigny, J.L.; Thonart, P. Surfactin and fengycin lipopeptides of Bacillus subtilis as elicitors of induced systemic resistance in plants. Environ. Microbiol. 2007, 9, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Ongena, M.; Henry, G.; Thonart, P. The role of cyclic lipopeptides in the biocontrol activity of Bacillus subtilis. In Recent Developments in Management of Plant Diseases (Plant Pathology in the 21st Century); Gisi, U., Chet, I., Gullino, M., Eds.; Springer Science + Business Media B.V.: Dordrecht, The Netherlands; Heidelberg, Germany; London, UK; New York, NY, USA, 2010; pp. 59–69. [Google Scholar] [CrossRef]
- Ahmad, Z.; Wu, J.; Chen, L.; Dong, W. Isolated Bacillus subtilis strain 330-2 and its antagonistic genes identified by the removing PCR. Sci. Rep. 2017, 7, 1777. [Google Scholar] [CrossRef] [PubMed]
- Loeffler, W.; Kratzer, W.; Kremer, S.; Kugler, M.; Petersen, F.; Jung, G.; Rapp, C.; Tschen, J.S.M. Gegen pilze wirksame antibiotika der Bacillus subtilis-gruppe. Forum Mikrobiol. 1990, 3, 156–163. [Google Scholar]
- Arrebola, E.; Jacobs, R.; Korsten, L. Iturin A is the principal inhibitor in the biocontrol activity of Bacillus amyloliquefaciens PPCB004 against postharvest fungal pathogens. J. Appl. Microbiol. 2010, 108, 386–395. [Google Scholar] [CrossRef]
- Raaijmakers, J.M.; De Bruijn, I.; Nybroe, O.; Ongena, M. Natural functions of lipopeptides from Bacillus and Pseudomonas: More than surfactants and antibiotics. FEMS Microbiol. Rev. 2010, 34, 1037–1062. [Google Scholar] [CrossRef] [PubMed]
- Mora, I.; Cabrefiga, J.; Montesinos, E. Antimicrobial peptide genes in Bacillus strains from plant environments. Int. Microbiol. 2011, 14, 213–223. [Google Scholar] [PubMed]
- Ongena, M.; Jacques, P.; Touré, Y.; Destain, J.; Jabrane, A.; Thonart, P. Involvement of fengycin-type lipopeptides in the multifaceted biocontrol potential of Bacillus subtilis. Appl. Microbiol. Biotechnol. 2005, 69, 29–38. [Google Scholar] [CrossRef]
- Luo, C.; Liu, X.; Zhou, H.; Wang, X.; Chen, Z. Nonribosomal peptide synthase gene clusters for lipopeptide biosynthesis in Bacillus subtilis 916 and their phenotypic functions. Appl. Environ. Microbiol. 2015, 81, 422–431. [Google Scholar] [CrossRef]
- Cho, S.J.; Lee, S.K.; Cha, B.J.; Kim, Y.H.; Shin, K.-S. Detection and characterization of the Gloeosporium gloeosporioides growth inhibitory compound iturin A from Bacillus subtilis strain KS03. FEMS Microbiol. Lett. 2003, 223, 47–51. [Google Scholar] [CrossRef]
- Romero, D.; de Vicente, A.; Rakotoaly, R.H.; Dufour, S.E.; Veening, J.-W.; Arrebola, E.; Cazorla, F.M.; Kuipers, O.P.; Paquot, M.; PérezGarcía, A. The iturin and fengycin families of lipopeptides are key factors in antagonism of Bacillus subtilis toward Podosphaera fusca. Mol. Plant Microbe Interact. 2007, 20, 430–440. [Google Scholar] [CrossRef]
- Jones, T.S. Chemical evidence for the multiplicity of the antibiotics produced by Bacillus polymyxa. Ann. N. Y. Acad. Sci. 1949, 51, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Falardeau, J.; Wise, C.; Novitsky, L.; Avis, T.J. Ecological and mechanistic insights into the direct and indirect antimicrobial properties of Bacillus subtilis lipopeptides on plant pathogens. J. Chem. Ecol. 2013, 39, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Jasim, B.; Sreelakshmi, K.S.; Mathew, J.; Radhakrishnan, E.K. Surfactin, iturin and fengycin biosynthesis by endophytic Bacillus sp. from Bacopa monnieri. Microb. Ecol. 2016, 72, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Peypoux, F.; Bonmatin, J.M.; Wallach, J. Recent trends in the biochemistry of surfactin. Appl. Microbiol. Biotechnol. 1999, 51, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Mihalache, G.; Balaes, T.; Gostin, I.; Stefan, M.; Coutte, F.; Krier, F. Lipopeptides produced by Bacillus subtilis as new biocontrol products against fusariosis in ornamental plants. Environ. Sci. Pollut. Res. 2017, 25, 29784. [Google Scholar] [CrossRef]
- Aranda, F.J.; Teruel, J.A.; Ortiz, A. Further aspects on the hemolytic activity of the antibiotic lipopeptide iturin A. Biochim. Biophys. Acta 2005, 1713, 51–56. [Google Scholar] [CrossRef]
- Dixon, R.A.; Achnine, L.; Kota, P.; Liu, C.J.; Reddy, M.S.; Wang, L. The phenylpropanoid pathway and plant defense—A genomics perspective. Mol. Plant Pathol. 2002, 3, 371–390. [Google Scholar] [CrossRef]
- Rahman, A.; Uddin, W.; Wenner, N.G. Induced systemic resistance responses in perennial ryegrass against Magnaporthe oryzae elicited by semi-purified surfactin lipopeptides and live cells of Bacillus amyloliquefaciens. Mol. Plant Pathol. 2015, 16, 546–558. [Google Scholar] [CrossRef]
- Kong, H.G.; Kim, J.C.; Choi, G.J.; Lee, K.Y.; Kim, H.J.; Hwang, E.C.; Moon, B.-J.; Lee, S.W. Production of surfactin and iturin by Bacillus licheniformis N1 responsible for plant disease control activity. Plant Pathol. J. 2012, 26, 170–177. [Google Scholar] [CrossRef]
- Waewthongrak, W.; Pisuchpen, S.; Leelasuphakul, W. Effect of Bacillus subtilis and chitosan applications on green mold (Penicilium digitatum Sacc.) decay in citrus fruit. Postharvest Biol. Technol. 2015, 99, 44–49. [Google Scholar] [CrossRef]
- Krebs, B.; Ockhardt, A.; Hoeding, B.; Bendzko, P.; Maximov, J.; Etzel, W. Cyclic Peptides from Bacillus amyloliquefaciens Useful Antimycotics, Antivirals, Fungicides, Nematicides etc. German Patent DE19641213, 26 September 1996. [Google Scholar]
- Banat, I.M.; Makkar, R.S.; Cameotra, S.S. Potential commercial applications of microbial surfactants. Appl. Microbiol. Biotechnol. 2000, 53, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Marrone, P.G. An effective biofungicide with novel modes of action. Pestic. Outlook 2002, 13, 193–194. [Google Scholar] [CrossRef]
- Singh, P.; Cameotra, S.S. Enhancement of metal bioremediation by use of microbial surfactants. Biochem. Biophys. Res. Commun. 2004, 319, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Gueldner, R.C.; Reilly, C.C.; Pussey, P.L.; Costello, C.E.; Arrendale, R.F.; Cox, R.H.; Himmelsbach, D.S.; Crumley, F.G.; Culter, H.G. Isolation and identification of iturins as antifungal peptides in biological control of peach brown rot with Bacillus subtilis. J. Agric. Food Chem. 1988, 36, 366–370. [Google Scholar] [CrossRef]
- Stein, T.; Entian, K.-D. Maturation of the lantibiotic subtilin: Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry to monitor precursors and their proteolytic processing in crude bacterial cultures. Rapid Commun. Mass Spectrom. 2002, 16, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Phister, T.G.; O’Sullivan, D.J.; McKay, L.L. Identification of bacilysin, chlorotetaine, and iturin A produced by Bacillus sp. strain CS93 isolated from pozol, a Mexican fermented maize dough. Appl. Environ. Microbiol. 2004, 70, 631–634. [Google Scholar] [CrossRef] [PubMed]
- Bongers, R.S.; Veening, J.-W.; Van Wieringen, M.; Kuipers, O.P.; Kleerebezem, M. Development and characterization of a subtilin-regulated expression system in Bacillus subtilis: Strict control of gene expression by addition of subtilin. Appl. Environ. Microbiol. 2005, 71, 8818–8824. [Google Scholar] [CrossRef] [PubMed]
- Rajavel, M.; Mitra, A.; Gopal, B. Role of Bacillus subtilis BacB in the synthesis of bacilysin. J. Biol. Chem. 2009, 46, 31882–31892. [Google Scholar] [CrossRef]
- Lee, H.; Kim, H.-Y. Lantibiotics, class I bacteriocins from the genus Bacillus. J. Microbiol. Biotechnol. 2011, 21, 229–235. [Google Scholar]
- Chung, S.; Kong, H.; Buyer, J.S.; Lakshman, D.K.; Lydon, J.; Kim, S.-D.; Roberts, D.P. Isolation and partial characterization of Bacillus subtilis ME488 for suppression of soilborne pathogens of cucumber and pepper. Appl. Microbiol. Biotechnol. 2008, 80, 115–123. [Google Scholar] [CrossRef]
- Joshi, R.; McSpadden-Gardener, B.B. Identification and characterization of novel genetic markers associated with biological control activities in Bacillus subtilis. Phytopathology 2006, 96, 145–154. [Google Scholar] [CrossRef] [PubMed]
- González-Sánchez, M.A.; Pérez-Jimenez, R.M.; Pliego, C.; Ramos, C.; de Vicente, A.; Cazorla, F.M. Biocontrol bacteria selected by a direct plant protection strategy against avocado white root rot show antagonism as a prevalent trait. J. Appl. Microbiol. 2010, 109, 65–78. [Google Scholar] [PubMed]
- Haggag, W.M. Isolation of bioactive antibiotic peptides from Bacillus brevis and Bacillus polymyxa against Botrytis grey mold in strawberry. Arch. Phytopathol. Plant Protect. 2008, 41, 477–491. [Google Scholar] [CrossRef]
- Gong, Q.; Zhang, C.; Lu, F.; Zhao, H.; Bie, X.; Lu, Z. Identification of bacillomycin D from Bacillus subtilis fmbJ and its inhibition effects against Aspergillus flavus. Food Control 2013, 36, 8–14. [Google Scholar] [CrossRef]
- Bull, C.T.; Wadsworth, M.L.K.; Sorenson, K.N.; Takemoto, J.; Austin, R.; Smilanick, J.L. Syringomycin E produced by biological agents controls green mold on lemons. Biol. Control 1998, 12, 89–95. [Google Scholar] [CrossRef]
- Bais, H.P.; Fall, R.; Vivanco, J.M. Biocontrol of Bacillus subtilis against infection of arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant Physiol. 2004, 134, 307–319. [Google Scholar] [CrossRef]
- Asaka, O.; Shoda, M. Biocontrol of Rhizoctonia solani damping-off of tomato with Bacillus subtilis RB14. Appl. Environ. Microbiol. 1996, 62, 4081–4085. [Google Scholar]
- Kajimura, Y.; Sugiayama, M.; Kaneda, M. Bacillopeptins, new cyclic lipopeptide antibiotics from Bacillus subtilis FR-2. J. Antibiot. 1995, 48, 1095–1103. [Google Scholar] [CrossRef]
- Yánez-Mendizábal, V.; Usall, J.; Viñas, I.; Casals, C.; Marín, S.; Solsona, C.; Teixidó, N. Potential of a new strain of Bacillus subtilis CPA-8 to control the major postharvest diseases of fruit. Biocontrol Sci. Technol. 2011, 21, 409–426. [Google Scholar] [CrossRef]
- Singh, D.; Sharma, R.R. Postharvest diseases of fruit and vegetables and their management. In Sustainable Pest Management; Prasad, D., Ed.; Daya Publishing House: New Delhi, India, 2009. [Google Scholar]
- Rodrigues, L.; Banat, I.M.; Teixeira, J.; Oliveira, R. Biosurfactants: Potential applications in medicine. J. Antimicrob. Chemother. 2006, 57, 609–618. [Google Scholar] [CrossRef]
- Bakker, P.A.H.M.; Pieterse, C.M.J.; Van Loon, L.C. Induced systemic resistance by fluorescent Pseudomonas spp. Phytopathology 2007, 97, 239–243. [Google Scholar] [CrossRef]
- Lorito, M.; Harman, G.E.; Hayes, C.K.; Broadway, R.M.; Trosomo, A.; Woo, S.L.; Di-Pietro, A. Chitolytic enzymes produced by Trichoderma harzianum: Antifungal activity of purified endochitinase and chitobiase. Phytopathology 1993, 83, 302–307. [Google Scholar] [CrossRef]
- Chernin, L.; Chet, I. Microbial enzymes in the biocontrol of plant pathogens and pests. In Enzymes in the Environment: Activity, Ecology, and Applications; Burns, R.G., Dick, R.P., Eds.; Marcel Dekker Inc.: New York, NY, USA, 2002; pp. 171–226. [Google Scholar]
- Sharma, R.R.; Singh, D.; Singh, R. Biological control of postharvest diseases of fruits and vegetables by microbial antagonists: A review. Biol. Control 2009, 50, 205–222. [Google Scholar] [CrossRef]
- Aktuganov, G.E.; Galimzyanova, N.F.; Melent’ev, A.I.; Kuzmina, L.Y. Extracellular hydrolases of strain Bacillus sp. 739 and their involvement in the lysis of micromycete cell walls. Microbiology (Mikrobiologiya) 2007, 76, 413–420. [Google Scholar] [CrossRef]
- Kavitha, K.; Nakkeeran, S.; Chandrasekar, G. Rhizobacterial mediated induction of defense enzymes to enhance the resistance of turmeric (Curcuma longa L.) to Pythium aphanidermatum causing rhizome rot. Arch. Phytopathol. Plant Protect. 2012, 45, 199–219. [Google Scholar] [CrossRef]
- Torres, M.A. ROS in biotic interactions. Physiol. Plant. 2010, 138, 414–429. [Google Scholar] [CrossRef]
- Sewelam, N.; Kazan, K.; Thomas Hall, S.R.; Kidd, B.N.; Manners, J.M.; Schenk, P.M. Ethylene response factor 6 is a regulator of reactive oxygen species signaling in Arabidopsis. PLoS ONE 2013, 8, e70289. [Google Scholar] [CrossRef]
- Maksimov, I.V.; Abisgildina, R.R.; Yusupova, Z.R.; Khairullin, R.M. Effect of Bacillus subtilis 26D on the hydrogen peroxide level and peroxidase activity in spring wheat plants. Agrochemistry 2010, 1, 55–60. (In Russian) [Google Scholar]
- Schoonbeek, H.J.; Jacquat Bovet, A.C.; Mascher, F.; Metraux, J.P. Oxalate degrading bacteria can protect Arabidopsis thaliana and crop plants against Botrytis cinerea. Mol. Plant Microbe Interact. 2007, 20, 1535–1544. [Google Scholar] [CrossRef]
- Kumar, P.; Dubey, R.C.; Maheshwari, D.K. Bacillus strains isolated from rhizosphere showed plant growth promoting and antagonistic activity against phytopathogens. Microbiol. Res. 2012, 167, 493–499. [Google Scholar] [CrossRef]
- Swain, P.; Nayak, S.K.; Nanda, P.K.; Dash, S. Biological effects of bacterial lipopolysaccharide (endotoxin) in fish: A review. Fish Shellfish Immunol. 2008, 25, 191–201. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Korsten, L. Biological control of postharvest diseases of fruits. Annu. Rev. Phytopathol. 2002, 40, 411–441. [Google Scholar] [CrossRef]
- Pieterse, C.M.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; van Wees, S.C.; Bakker, P.A. Induced systemic resistance by beneficial microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Van der Does, D.; Zamioudis, C.; Leon Reyes, A.; van Wees, S.C.M. Hormonal modulation of plant immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 1–28. [Google Scholar] [CrossRef]
- Dodd, I.C.; Zinovkina, N.Y.; Safronova, V.I.; Belimov, A.A. Rhizobacterial mediation of plant hormone status. Ann. Appl. Biol. 2010, 157, 361–379. [Google Scholar] [CrossRef]
- Dobbelaere, S.; Vanderleyden, J.; Okon, Y. Plant growthpromoting effects of diazotrophs in the rhizosphere. Crit. Rev. Plant Sci. 2003, 22, 107–149. [Google Scholar] [CrossRef]
- Sivasakthi, S.; Kanchana, D.; Usharani, G.; Saranraj, P. Production of plant growth promoting substance by Pseudomonas fluorescens and Bacillus subtilis isolates from paddy rhizosphere soil of Cuddalore district, Tamil Nadu, India. Int. J. Microbiol. Res. 2013, 4, 227–233. [Google Scholar]
- Kudoyarova, G.R.; Melentiev, A.I.; Martynenko, E.V.; Timergalina, L.N.; Arkhipova, T.N.; Shendel, G.V.; Kuz’mina, L.Y.; Dodd, I.C.; Veselov, S.Y. Cytokinin producing bacteria stimulate amino acid deposition by wheat roots. Plant Physiol. Biochem. 2014, 83, 285–291. [Google Scholar] [CrossRef]
- Belimov, A.A.; Dodd, I.C.; Safronova, V.I.; Dumova, V.A.; Shaposhnikov, A.I.; Ladatko, A.G.; Davies, W.J. Abscisic acid metabolizing rhizobacteria decrease ABA concentrations in planta and alter plant growth. Plant Physiol. Biochem. 2014, 74, 84–91. [Google Scholar] [CrossRef]
- Cohen, A.C.; Travaglia, C.N.; Bottini, R.; Piccoli, P.N. Participation of abscisic acid and gibberellins produced by endophytic Azospirillum in the alleviation of drought effects in maize. Botany 2009, 87, 455–462. [Google Scholar] [CrossRef]
- Zhang, H.; Kim, M.S.; Krishnamachari, V.; Payton, P.; Sun, Y.; Grimson, M.; Farag, M.A.; Ryu, C.M.; Allen, R.; Melo, I.S.; et al. Rhizobacterial volatile emissions regulate auxin homeostasis and cell expansion in Arabidopsis. Planta 2007, 226, 839–851. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Xie, X.; Kim, M.S.; Kornyeyev, D.A.; Holaday, S.; Paré, P.W. Soil bacteria augment Arabidopsis photosynthesis by decreasing glucose sensing and abscisic acid levels in planta. Plant J. 2008, 56, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Pertry, I.; Václavíková, K.; Depuydt, S.; Galuszka, P.; Spíchal, L.; Temmerman, W.; Stes, E.; Schmülling, T.; Kakimoto, T.; van Montagu, M.C.E.; et al. Identification of Rhodococcus fascians cytokinins and their modus operandi to reshape the plant. Proc. Natl. Acad. Sci. USA 2006, 106, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Gutierrez, L.; Romero, D.; Zeriouh, H.; Cazorla, F.M.; Torés, J.A.; de Vicente, A.; Pérez-García, A. Isolation and selection of plant growth-promoting rhizobacteria as inducers of systemic resistance in melon. Plant Soil 2012, 358, 201–212. [Google Scholar] [CrossRef]
- Spadaro, D.; Gullino, M.L. State of the art and future prospects of the biological control of postharvest fruit diseases. Int. J. Food Microbiol. 2004, 91, 185–194. [Google Scholar] [CrossRef]
- Razzaq, K.; Khan, A.S.; Malik, A.U.; Shahid, M. Ripening period influences fruit softening and antioxidative system of ‘Samar Bahisht Chaunsa’ mango. Sci. Hortic. 2013, 160, 108–114. [Google Scholar] [CrossRef]
- Li, T.; Yun, Z.; Zhang, D.D.; Yang, C.W.; Zhu, H.; Jiang, Y.M.; Duan, X.W. Proteomic analysis of differentially expressed proteins involved in ethylene-induced chilling tolerance in harvested banana fruit. Front. Plant Sci. 2015, 6, 845. [Google Scholar] [CrossRef] [PubMed]
- Belimov, A.A.; Dodd, I.C.; Hontzeas, N.; Theobald, J.C.; Safronova, V.I.; Davies, W.J. Rhizosphere bacteria containing ACC deaminase increase yield of plants grown in drying soil via both local and systemic hormone signaling. New Phytol. 2009, 181, 413–423. [Google Scholar] [CrossRef]
- Gamalero, E.; Glick, B.R. Bacterial modulation of plant ethylene levels. Plant Physiol. 2015, 169, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Farace, G.; Fernandez, O.; Jacquens, L.; Coutte, F.; Krier, F.; Jacques, P.; Clément, C.; Barka, E.A.; Jacquard, C.; Dorey, S. Cyclic lipopeptides from Bacillus subtilis activate distinct patterns of defense responses in grapevine. Mol. Plant Pathol. 2015, 16, 177–187. [Google Scholar] [CrossRef]
- Sarosh, B.R.; Danielsson, J.; Meijer, J. Transcript profiling of oilseed rape (Brassica napus) primed for biocontrol differentiate genes involved in microbial interactions with beneficial Bacillus amyloliquefaciens from pathogenic Botrytis cinerea. Plant Mol. Biol. 2009, 70, 31–45. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.P.; Uhl, J.; Grosch, R.; Alquéres, S.; Pittroff, S.; Dietel, K.; Schmitt-Kopplin, P.; Borriss, R.; Hartmann, A. Cyclic lipopeptides of Bacillus amyloliquefaciens subsp. plantarum colonizing the lettuce rhizosphere enhance plant defense responses toward the bottom rot rathogen Rhizoctonia solani. Mol. Plant Microbe Interact. 2015, 28, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Chandler, S.; Van Hese, N.; Coutte, F.; Jacques, P.; Hofte, M.; De Vleesschauwer, D. Role of cyclic lipopeptides produced by Bacillus subtilis in mounting induced immunity in rice (Oryza sativa L.). Physiol. Mol. Plant Pathol. 2015, 91, 20–30. [Google Scholar] [CrossRef]
- Zhang, S.; Jiang, W.; Li, J.; Meng, L.; Cao, X.; Hu, J.; Liu, Y.; Chen, J.; Sha, C. Whole genome shotgun sequence of Bacillus amyloliquefaciens TF28, a biocontrol entophytic bacterium. Stand. Genomic Sci. 2016, 11, 73. [Google Scholar] [CrossRef] [PubMed]
- Rodov, V.; Ben-Yehoshua, S.; Fang, D.Q.; D’hallewin, G.; Castia, T. Accumulation of phytoalexins scoparone and scopoletin in citrus fruits subjected to various postharvest treatments. Acta Hortic. 1994, 381, 517–523. [Google Scholar] [CrossRef]
- Arras, G. Mode of action of an isolate of Candida famata in biological control of Penicillium digitatum in orange fruits. Postharvest Biol. Technol. 1996, 8, 191–198. [Google Scholar] [CrossRef]
- Ahn, I.P.; Lee, S.W.; Kim, M.G.; Park, S.R.; Hwang, D.J.; Bae, S.C. Priming by rhizobacterium protects tomato plants from biotrophic and necrotrophic pathogen infections through multiple defense mechanisms. Mol. Cells 2011, 32, 7–14. [Google Scholar] [CrossRef]
- Nakkeeran, S.; Kavitha, K.; Chandrasekar, G.; Renukadevi, P.; Fernando, W.G.D. Induction of plant defense compounds by Pseudomonas chlororaphis PA23 and Bacillus subtilis BSCBE4 in controlling damping-off of hot pepper caused by Pythium aphanidermatum. Biocontrol Sci. Technol. 2006, 16, 403–416. [Google Scholar] [CrossRef]
- Thilagavathi, R.; Saravanakumar, D.; Ragupathi, N.; Samiyappan, R. A combination of biocontrol agents improves the management of dry root rot (Macrophomina phaseolina) in greengram. Phytopathol. Mediterr. 2007, 46, 157–167. [Google Scholar]
- Da, K.; Nowak, J.; Flinn, B. Potato cytosine methylation and gene expression changes induced by a beneficial bacterial endophyte, Burkholderia phytofirmans strain PsJN. Plant Physiol. Biochem. 2012, 50, 24–34. [Google Scholar] [CrossRef]
- Coates, L.; Johnson, G.; Dale, M. Postharvest diseases of fruit and vegetables. In Plant Pathogens and Plant Diseases; Brown, J.F., Ogle, H.J., Eds.; Rockvale Publications: Armidale, Australia, 1997; pp. 533–548. [Google Scholar]
- Ippolito, A.; Nigro, F. Impact of preharvest application of biological control agents on postharvest diseases of fresh fruit and vegetables. Crop Prot. 2000, 19, 715–723. [Google Scholar] [CrossRef]
- Ippolito, A.; Schena, L.; Pentimone, I.; Nigro, F. Control of postharvest rots of sweet cherries by pre- and postharvest applications of Aureobasidium pullulans in combination with calcium chloride or sodium bicarbonate. Postharvest Biol. Technol. 2005, 36, 245–252. [Google Scholar] [CrossRef]
- Irtwange, S.V. Application of biological control agents in pre- and postharvest operations. Agric. Eng. Int. CIGR J. 2006, 8, 1–12. [Google Scholar]
- Leibinger, W.; Breuker, B.; Hahn, M.; Mendgen, K. Control of postharvest pathogens and colonization of the apple surface by antagonistic microorganisms in the field. Phytopathology 1997, 87, 1103–1110. [Google Scholar] [CrossRef] [PubMed]
- Narayanasamy, P. Ecology of Postharvest Microbial Pathogens. Postharvest Pathogens and Disease Management; John Wiley & Sons, Inc.: Coimbatore, India, 2005; pp. 79–116. [Google Scholar]
- Korsten, L.; Bezuidenhout, J.J.; Kotzé, J.M. Biological control of postharvest diseases of avocado. In South African Avocado Growers’ Association Yearbook; University of Pretoria: Tzaneen, South Africa, 1988. [Google Scholar]
- Barkai-Golan, R. Postharvest diseases of fruit and vegetables. In Development and Control; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Droby, S. Improving quality and safety of fresh fruit and vegetables after harvest by the use of biocontrol agents and natural materials. Acta Hortic. 2006, 709, 45–51. [Google Scholar] [CrossRef]
- Pusey, P.L.; Hotchkiss, M.W.; Dulmage, H.T.; Banumgardner, R.A.; Zehr, E.I. Pilot tests for commercial production and application of Bacillus subtilis (B-3) for postharvest control of peach brown rot. Plant Dis. 1988, 72, 622–626. [Google Scholar] [CrossRef]
- Anon. AgraQuest Introduces New Soil Fungicide for Potatoes and Other Crops. 2010. Available online: http://agraquest.com/news/2010/01/agraquest-introduces-newsoil-fungicide-for-potatoes-and-other-crops/ (accessed on 25 September 2018).
- Walton, D. New Zealand Agrichemical Manual; WHAM Media: Wellington, New Zealand, 2002. [Google Scholar]
| Bacterial Inoculant | Disease/Pathogen | Fruits/ Vegetables | Reference(s) |
|---|---|---|---|
| B. subtilis | Brown rot (Lasiodiplodia theobromae) | Apricot | [45] |
| Stem and rot (Botryodiplodia theobromae Pat.) | Avocado | [64] | |
| Botrytis rot (Botrytis cinerea) | Cherry | [52] | |
| Green mold (Penicillium digitatum) | Citrus | [37] | |
| Sour rot (Geotrichum candidum Link) | Citrus | [37] | |
| Stem and rot (Botryodiplodia theobromae, Phomopsis citri H.S. Fawc., Alternaria citri Ell., and Pierce) | Citrus | [37] | |
| Alternaria rot (A. alternata (Fr.) Keissler) | Litchi | [33] | |
| Brown rot (Lasiodiplodia theobromae) | Nectarine | [45] | |
| Brown rot (L. theobromae) | Peach | [45] | |
| Brown rot (L. theobromae) | Plum | [45] | |
| Fungal rot (Aspergillus niger, Botryodiploidia theobromae, Penicillium oxalicum) | Yam | [50] | |
| Gray mold (Botrytis cinerea) | Strawberry | [40] | |
| Alternaria rot (Alternaria alternata) | Muskmelon | [65] | |
| A. alternata | Melon | [34] | |
| Aspergillus, Pennicillium, Alternaria, Cladosporium, Fusarium, Mucor | Potato | [60] | |
| Colletotrichum acutatum | Apple | [35] | |
| Botrytis cinerea | Apple | [39] | |
| Botryosphaeria dothidea, Diaporthe actinidiae, Botrytis cinerea | Kiwifruit | [35] | |
| Anthracnose (Colletotrichum gloeosporioides, C. acutatum), white rot (B. dothidea) | Apple | [36] | |
| Ring rot (B. dothidea) | Apple | [49] | |
| Rot (Penicillium sp., Rhizopus stolonifer) | Tomato | [66] | |
| Grey mold (Botrytis cinerea) | Tomato | [42] | |
| Brown rot (Monilinia fructicola) | Peach | [67] | |
| Brown rot (Monilinia spp.) | Peach | [68] | |
| Rot (Rhizopus) | Peach | [53] | |
| Green mold (Penicillium digitatum) | Citrus | [46] | |
| B. amyloliquefaciens | Brown rot (Monilinia spp.) | Peaches | [69] |
| Nectarines | |||
| Flat peaches | |||
| Cherries | |||
| Apricots | |||
| Plums | |||
| B. licheniformis (Weigmann) Verhoeven | Anthracnose (Colletotrichum gloeosporioides) and stem end rot (Dothiorella gregaria Sacc.) | Mango | [70] |
| B. licheniformis | Grey mold (Botrytis mali) | Apple | [47] |
| B. pumilus | Gray mold (B. cinerea) | Pear | [38] |
| Product | Microbial Agent | Fruits/Vegetables | Target Disease(s) | Manufacturer/Distributor |
|---|---|---|---|---|
| Rhio-plus | B. subtilis FZB 24 | Potatoes and other vegetables | Powdery mildew and root rots | KFZB Biotechnick, Germany |
| Serenade | B. subtilis QST713 | Apple, pear, grapes, tomato, potato | Powdery mildew, late blight, brown rot, and fire blight | AgraQuest. Inc., USA |
| Phytosporin-M Golden Authum, AntiGnil Phytosporin M | B. subtilis 26D | Carrot, tomato, cabbage, sugar beet, potato | Rots, mold | Bashinkom, Russia |
| Rhapsody® | B. subtilis QST 713 | Tomato | Rots | Bayer, Canada |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lastochkina, O.; Seifikalhor, M.; Aliniaeifard, S.; Baymiev, A.; Pusenkova, L.; Garipova, S.; Kulabuhova, D.; Maksimov, I. Bacillus Spp.: Efficient Biotic Strategy to Control Postharvest Diseases of Fruits and Vegetables. Plants 2019, 8, 97. https://doi.org/10.3390/plants8040097
Lastochkina O, Seifikalhor M, Aliniaeifard S, Baymiev A, Pusenkova L, Garipova S, Kulabuhova D, Maksimov I. Bacillus Spp.: Efficient Biotic Strategy to Control Postharvest Diseases of Fruits and Vegetables. Plants. 2019; 8(4):97. https://doi.org/10.3390/plants8040097
Chicago/Turabian StyleLastochkina, Oksana, Maryam Seifikalhor, Sasan Aliniaeifard, Andrey Baymiev, Ludmila Pusenkova, Svetlana Garipova, Darya Kulabuhova, and Igor Maksimov. 2019. "Bacillus Spp.: Efficient Biotic Strategy to Control Postharvest Diseases of Fruits and Vegetables" Plants 8, no. 4: 97. https://doi.org/10.3390/plants8040097
APA StyleLastochkina, O., Seifikalhor, M., Aliniaeifard, S., Baymiev, A., Pusenkova, L., Garipova, S., Kulabuhova, D., & Maksimov, I. (2019). Bacillus Spp.: Efficient Biotic Strategy to Control Postharvest Diseases of Fruits and Vegetables. Plants, 8(4), 97. https://doi.org/10.3390/plants8040097








