Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs
Abstract
:1. Introduction
2. Material and Methods
2.1. General Methods
2.2. Sample Preparation
2.3. Dry Residue and Soluble Solids
2.4. Sugars
2.5. ElementalComposition
2.6. Nitrates
2.7. Selenium
2.8. Antioxidants
2.8.1. Ascorbic Acid
2.8.2. Polyphenols
2.8.3. Flavonoids
2.8.4. Antioxidant Activity (AOA)
2.9. Statistical Analysis
3. Results and Discussion
3.1. Mycorrhizal Index
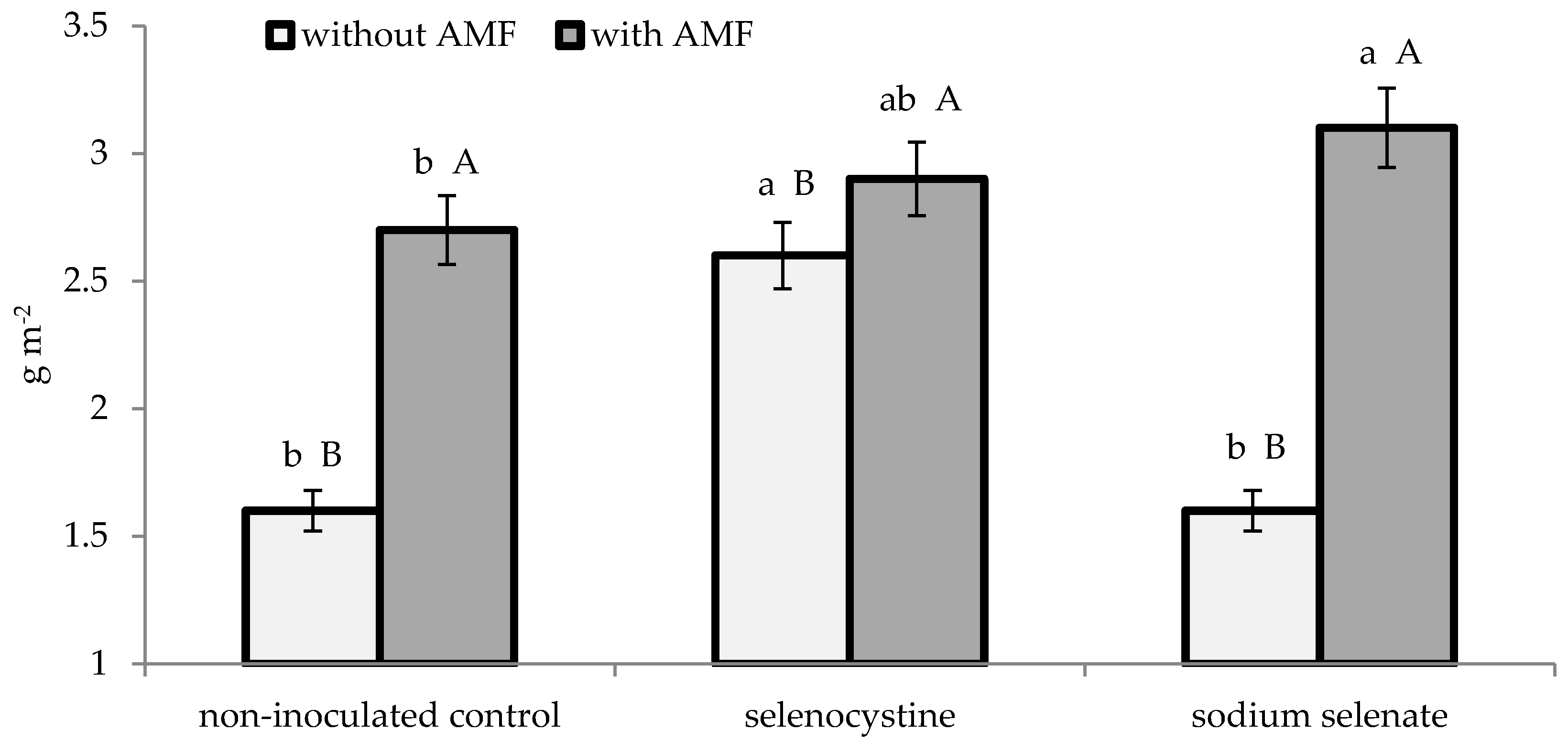
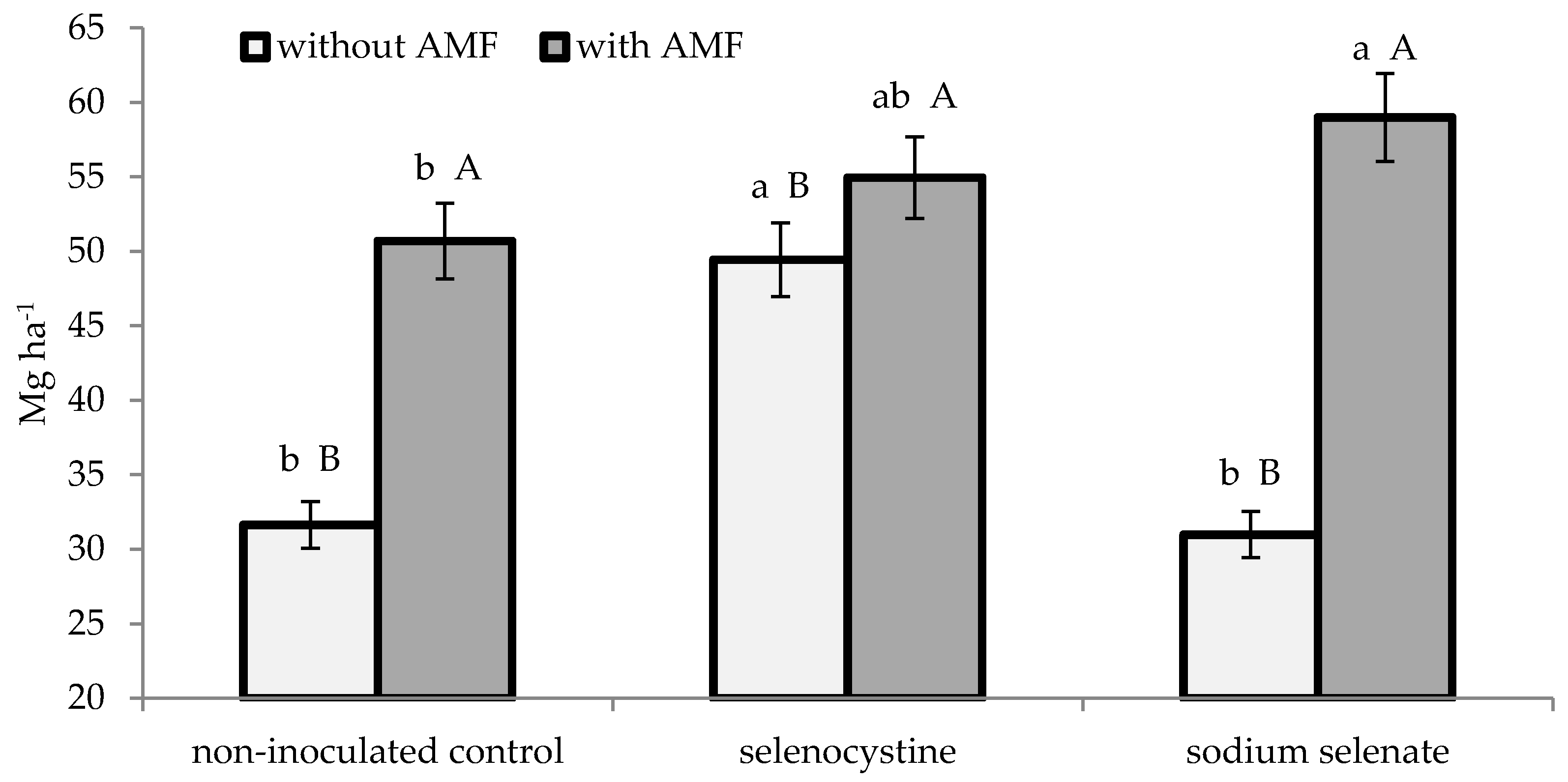
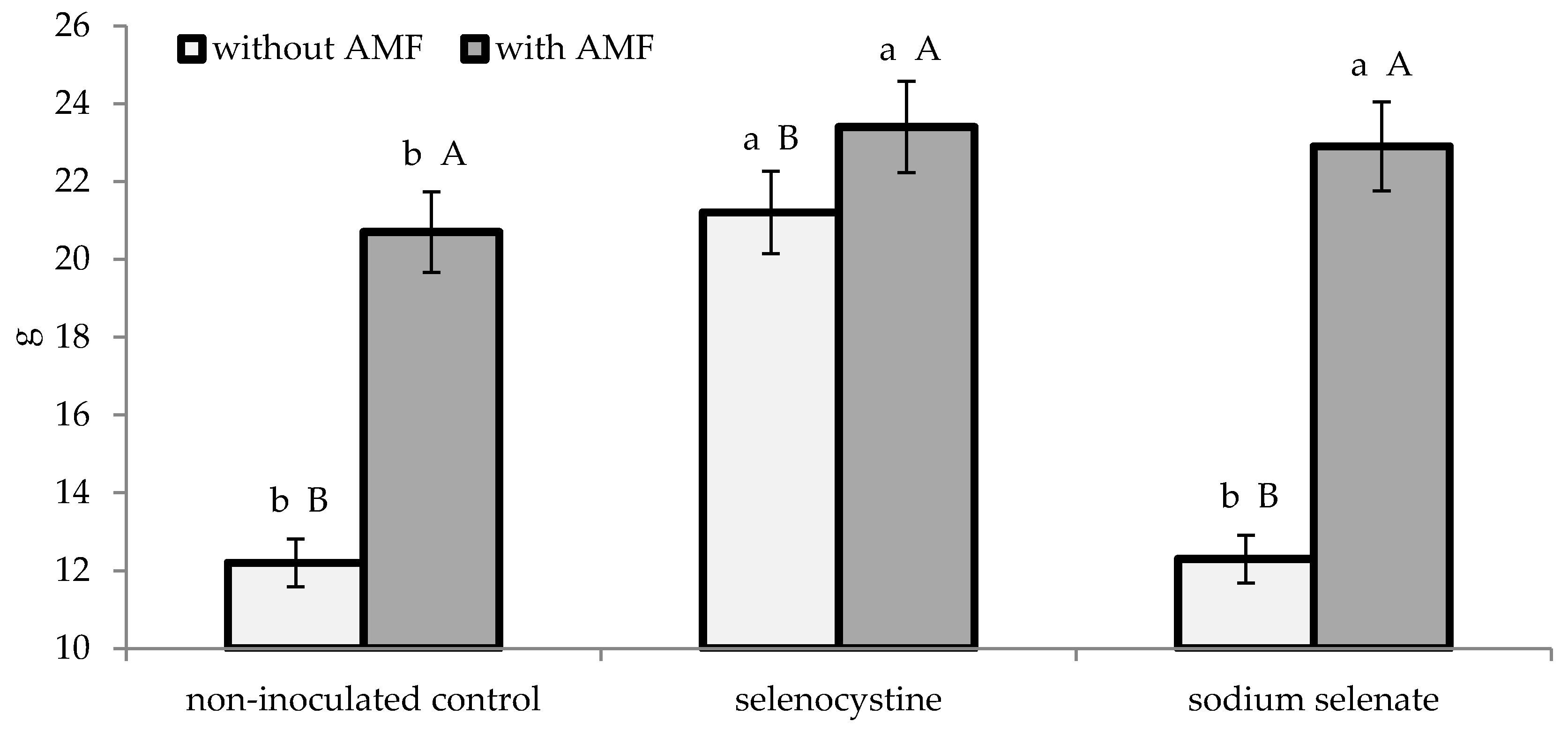
3.2. Plant Growth and Yield
3.3. Quality Indicators
3.4. Elemental Composition
3.5. Antioxidants
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Leelarungrayub, N.; Rattanapanone, V.; Chanarat, N.; Gebicki, J.M. Quantitative evaluation of the antioxidant properties of garlic and shallot preparations. Nutrition 2006, 22, 266–274. [Google Scholar] [CrossRef]
- Jalal, R.; Bagheri, S.M.; Moghimi, A.; Rasuli, M.B. Hypoglycemic effect of aqueous shallot and garlic extracts in rats with fructose-induced insulin resistance. J. Clin. Biochem. Nutr. 2007, 41, 218–223. [Google Scholar] [CrossRef]
- Tappayuthpijarn, P.; Dejatiwongse, Q.; Hincherana, T.; Suriyant, P. Effect of Allium ascalonicum on erythrocyte shape in induced hypercholesterolemia rabbits. J. Med. Assoc. Thai. 1989, 72, 448–451. [Google Scholar]
- Amin, M.; Kapadnis, B.P. Heat stable antimicrobial activity of Allium ascalonicum against bacteria and fungi. Ind. J. Exp. Biol. 2005, 43, 751–754. [Google Scholar]
- Golubkina, N.A.; Papazyan, T.T. Selenium in Nutrition. Plants, Animals, Human Beings; Moscow-Pechtny gorod: Moscow, Russia, 2006. [Google Scholar]
- Mora, M.L.; Durán, P.; Acuña, A.J.; Cartes, P.; Demanet, R.; Gianfreda, L. Improving selenium status in plant nutrition and quality. J. Soil Sci. Plant Nutr. 2015, 15, 486–503. [Google Scholar] [CrossRef]
- Alfthan, G.; Eurola, M.; Ekholm, P.; Venäläinen, E.R.; Root, T.; Korkalainen, K.; Hartikainen, H.; Salminen, P.; Hietaniemi, V.; Aspila, P.; Aro, A. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in Finland: From deficiency to optimal selenium status of the population. J. Trace Elem. Med. Biol. 2015, 31, 142–147. [Google Scholar] [CrossRef]
- Filho, J.A.C.; Sobrinho, R.R.; Rascholati, S.F. Arbuscular mycorrhizal symbiosis and its role in plant nutrition in sustainable agriculture. In Agriculturally Important Microbes for Sustainable Agriculture; Meena, V.S., Mishra, P., Bisht, J., Pattanayak, A., Eds.; Springer: Singapore, 2017. [Google Scholar]
- Gupta, A.; Gopal, M.; Tilak, K.V. Mechanism of plant growth promotion by rhizobacteria. Indian J. Exp. Biol. 2000, 38, 856–862. [Google Scholar] [PubMed]
- Caruso, G.; Golubkina, N.A.; Seredin, T.M.; Sellitto, V.M. Utilization of arbuscular-mycorrhyzal fungi in Allium production. Veg. Crops Russ. 2018, 3, 85–90. (In Russian) [Google Scholar]
- Larsen, E.H.; Lobinski, R.; Burger-Meÿer, K.; Hansen, M.; Ruzik, R.; Mazurowska, L.; Rasmussen, P.H.; Sloth, J.J.; Scholten, O.; Kik, C. Uptake and speciation of selenium in garlic cultivated in soil amended with symbiotic fungi (mycorrhiza) and selenate. Anal. Bioanal. Chem. 2006, 385, 1098–1108. [Google Scholar] [CrossRef]
- Duran, P.; Acuca, J.J.; Jorquera, M.A.; Azcyn, R.; Borie, F.; Cornejo, P.; Mora, M.L. Enhanced selenium content in wheat grain by coinoculation of selenobacteria and arbuscular mycorrhizal fungi: A preliminary study as a potential Se biofortification strategy. J. Cereal Sci. 2013, 57, 275–280. [Google Scholar] [CrossRef]
- Sanmartín, C.; Garmendia, I.; Romano, B.; Díaz, M.; Palop, J.A.; Goicoechea, N. Mycorrhizal inoculation affected growth, mineral composition, proteins and sugars in lettuces biofortified with organic or inorganic selenocompounds. Sci. Hort. 2014, 180, 40–51. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, S.; Wen, B.; Huang, H.; Luo, L. Accumulation and speciation of selenium in plants as affected by arbuscular mycorrhizal fungus Glomus mosseae. Biol. Trace Elem. Res. 2011, 143, 1789–1798. [Google Scholar] [CrossRef]
- Bhattacharya, A.; Toth, K.; Sen, A. Inhibition of colon cancer growth by methylselenocysteine-induced angiogenic chemomodulation is influenced by histologic characteristics of the tumor. Clin. Color. Cancer 2009, 8, 155–162. [Google Scholar] [CrossRef]
- Golubkina, N.A.; Kekina, H.G.; Caruso, G. Yield, quality and antioxidant properties of Indian mustard (Brassica juncea L.) in response to foliar biofortification with selenium and iodine. Plants 2018, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, S.; AlKhedhairy, A.A.; Ahamed, M.; Musarrat, J. Biomimetic synthesis of selenium nanospheres by bacterial strain JS-11 and its role as a biosensor for nanotoxicity assessment: A novel Se-bioassay. PLoS ONE 2013, 8, e57404. [Google Scholar] [CrossRef]
- Giovannetti, M.; Mosse, B. An evaluation of techniques for measuring vesicular-arbuscular mycorrhiza in roots. New Phytol. 1980, 84, 489–500. [Google Scholar] [CrossRef]
- Swamy, P.M. Laboratory Manual on Biotechnology; Rastogi: New Delhi, India, 2008; 617p. [Google Scholar]
- Alfthan, G.V. A micromethod for the determination of selenium in tissues and biological fluids by single-test-tube fluorimetry. Anal. Chim. Acta 1984, 65, 187–194. [Google Scholar] [CrossRef]
- Association Official Analytical Chemists (AOAC). The Official Methods of Analysis of AOAC International; 22 Vitamin C; AOAC: Rockville, MD, USA, 2012. [Google Scholar]
- Golubkina, N.A.; Kosheleva, O.V.; Krivenkov, L.V.; Dobrutskaya, H.G.; Nadezhkin, S.; Caruso, G. Intersexual differences in plant growth, yield, mineral composition and antioxidants of spinach (Spinacia oleracea L.) as affected by selenium form. Sci. Hortic. 2017, 225, 350–358. [Google Scholar] [CrossRef]
- Da Silva, L.A.L.; Pezzini, B.R.; Soares, L. Spectrophotometric determination of the total flavonoid content in OcimumbasilicumL. (Lamiaceae) leaves. Pharmacogn. Mag. 2015, 11, 96–101. [Google Scholar] [CrossRef]
- Maximova, T.V.; Nikulina, I.N.; Pakhomov, V.P.; Shkarina, H.I.; Chumakova, Z.V.; Arzamastsev, A.P. Method of Antioxidant Activity Determination. RU Patent No 2.170,930, 20 July 2001. [Google Scholar]
- Srivastava, S.; Adholeya, A.; Conlan, X.A.; Cahill, D.M. Acidic potassium permanganate chemiluminescence for the determination of antioxidant potential in three cultivars of Ocimumbasilicum. Plant Foods Hum. Nutr. 2015, 70. [Google Scholar] [CrossRef]
- Zhan, M.G.; Liu, N.; Liu, H. Determination of the total mass of antioxidant substances and antioxidant capacity per unit mass in serum using redox titration. Bioinorg. Chem. Appl. 2014, 2014, 928595. [Google Scholar] [CrossRef]
- Barea, J.M.; Pozo, M.J.; Azcón, R.; Azcón-Aguilar, C. Microbial cooperation in the rhizosphere. J. Exp. Bot. 2005, 56, 1761–1778. [Google Scholar] [CrossRef]
- Priyadharsini, P.S.; Pandey, R.R.; Muthukumar, T. Arbuscular mycorrhizal and dark septate fungal associations in shallot (Allium cepa L. var. aggregatum) under conventional agriculture. Acta Bot. Croat. 2012, 71, 159–175. [Google Scholar] [CrossRef]
- Hayman, D.S.; Mosse, B. Plant growth responses to vesicular-arbuscular mycorrhiza. Growth of endogone-inoculated plants in phosphate-deficient soils. New Phytol. 1971, 70, 19–27. [Google Scholar] [CrossRef]
- Turakainen, M.; Hartikainen, H.; Seppänen, M.M. Effects of selenium treatments on potato (Solanum tuberosum L.) growth and concentrations of soluble sugars and starch. J. Agric. Food Chem. 2004, 52, 5378–5382. [Google Scholar] [CrossRef] [PubMed]
- Bona, E.; Cantamessa, S.; Massa, N.; Manassero, P.; Marsano, F.; Copetta, A.; Lingua, G.; D’Agostino, G.; Gamalero, E.; Berta, G. Arbuscular mycorrhizal fungi and plant growth-promoting Pseudomonads improve yield, quality and nutritional value of tomato: A field study. Mycorrhiza 2016, 27. [Google Scholar] [CrossRef]
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and novel mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709. [Google Scholar] [CrossRef]
- Sekara, A.; Pokluda, R.; Del Vacchio, L.; Somma, S.; Caruso, G. Interactions among genotype, environment and agronomic practices on production and quality of storage onion (Allium cepa L.)—A review. Hort. Sci. 2017, 44, 21–42. [Google Scholar] [CrossRef]
- Ouzounidou, G.; Giannakoula, A.; Asfi, M.; Ilias, I. Differential Responses of onion and garlic against plant growth regulators. Pak. J. Bot. 2011, 43, 2051–2057. [Google Scholar]
- Yang, J.; Kloepper, J.W.; Ryu, C.M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci. 2009, 14, 1–4. [Google Scholar] [CrossRef]
- Baslam, M.; Garmendia, I.; Goicoechea, N. The arbuscular mycorrhizal symbiosis can overcome reductions in yield and nutritional quality in greenhouse-lettuces cultivated at inappropriate growing seasons. Sci. Hort. 2013, 164, 145–154. [Google Scholar] [CrossRef]
- Nori, M.; Aali, J.; Shafizi, R. Effect of different sources and levels of nitrogen fertilizer on yield and nitrate accumulation in garlic (Allium sativum L.). Int. J. Agric. Crop. Sci. 2012, 4, 1878–1880. [Google Scholar]
- Mohamed, A.A.; Eweda, W.E.E.; Heggo, A.M.; Hassan, E.A. Effect of dual inoculation with arbuscular mycorrhizal fungi and sulphur-oxidising bacteria on onion (Allium cepa L.) and maize (Zea mays L.) grown in sandy soil under green house conditions. Ann. Agric. Sci. 2014, 59, 109–118. [Google Scholar] [CrossRef]
- Kučová, L. Effect of mycorrhizal inoculation of leek A. porrum L. on mineral nitrogen leaching. Hort. Sci. 2016, 43, 195–202. [Google Scholar] [CrossRef]
- Copetta, A.; Bardi, L.; Bertolone, E.; Berta, G. Fruit production and quality of tomato plants (Solanum lycopersicum L.) are affected by green compost and arbuscular mycorrhizal fungi. Plant Biosyst. 2011, 145, 106–115. [Google Scholar] [CrossRef]
- Bücking, H.; Kafle, A. Role of arbuscular mycorrhizal fungi in the nitrogen uptake of plants: Current knowledge and research gaps. Agronomy 2015, 5, 587–612. [Google Scholar] [CrossRef]
- Chung, M.J.; Lee, S.H.; Sung, N.J. Inhibitory effect of whole strawberries, garlic juice or kale juice on endogenous formation of N-nitrosodimethylamine in humans. Cancer Lett. 2002, 182, 1–10. [Google Scholar] [CrossRef]
- Commission Regulation (EC). Setting maximum levels for certain contaminants in foodstuffs. Commission Regulation (EC) No. 1881/2006. Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- Pontigo, S.; Godoy, K.; Jiménez, H.; Gutiérrez-Moraga, A.; Mora, M.L.; Cartes, P. Silicon-mediated alleviation of aluminum toxicity by modulation of Al/Si uptake and antioxidant performance in ryegrass plants. Front. Plant Sci. 2017, 8, 642. [Google Scholar] [CrossRef]
- Jansen, S.; Broadley, M.R.; Robbrecht, E.; Smets, E. Aluminum hyperaccumulation in angiosperms: A review of its phylogenetic significance. Bot. Rev. 2002, 68, 235–269. [Google Scholar] [CrossRef]
- Zhang, Y.; Yao, Q.; Li, J.; Wang, Y.; Liu, X.; Hu, Y.; Chen, J. Contributions of an arbuscular mycorrhizal fungus to growth and physiology of loquat (Eriobotrya japonica) plants subjected to drought stress. Mycol. Prog. 2015, 14, 84–94. [Google Scholar] [CrossRef]
- Zayed, A.M.; Lytle, C.M.; Terry, N. Accumulation and volatilization of different chemical species of selenium by plants. Planta 1998, 206, 284–292. [Google Scholar] [CrossRef]
- Poluboyarinov, P.A.; Sharonov, G.I.; Leshenko, P.P. Method of production of biologically active compound-selenocystine. RU Patent No 2537166, 24 May 2013. [Google Scholar]
- de Souza, M.P.; Chu, D.; Zhao, M.; Zayed, A.M.; Ruzin, S.E.; Schichne, D.; Terry, N. Rhizosphere bacteria enhance selenium accumulation and volatilization by Indian mustard. Plant Physiol. 1999, 119, 565–574. [Google Scholar] [CrossRef]
- Lone, R.; Shuab, R.; Sharma, V.; Kumar, V.; Mir, R.; Koul, K.K. Effect of arbuscular mycorrhizal fungi on growth and development of potato (Solanum tuberosum) plant. Asian J. Crop Sci. 2015, 7, 233–243. [Google Scholar]
- Albrechtova, J.; Latr, A.; Nedorost, L.; Pokluda, R.; Posta, K.; Vosatka, M. Dual inoculation with mycorrhizal and saprotrophic fungi applicable in sustainable cultivation improves the yield and nutritive value of onion. Sci. World J. 2012, 2012, 374091. [Google Scholar] [CrossRef]
- Konieczny, A.; Kowalska, I. Effect of arbuscular mycorrhizal fungi on the content of zinc in lettuce grown at two phosphorus levels and an elevated zinc level in a nutrient solution. J. Elem. 2016, 22, 761–772. [Google Scholar] [CrossRef]
- Duc, N.H. Impact of Arbuscular Mycorrhizal Fungi on Plant Tolerance to Some Abiotic Stresses and Phytopathogens. Ph.D. Dissertation, SzentIstván University, Godollo, Hungary, 2017. [Google Scholar]
- Smirnoff, N.; Wheeler, G.L. Ascorbic acid in plants: Biosynthesis and function. CRC Crit. Rev. Biochem. Mol. Biol. 2000, 35, 291–314. [Google Scholar]
- Kyle, J.A.M.; Duthie, G.G. Flavonoids in foods. In Flavonoids; Chemistry, Biochemistry and Health Implications; Andersen, O.M., Markham, K., Eds.; Taylor and Francis Group: New York, NY, USA, 2006; pp. 219–263. [Google Scholar]
- Ariyanti, N.A.; Torikai, K.; Kirana, R.P.; Hirata, S.; Sulistyaningsih, E.; Ito, S.; Yamauchi, N.; Kobayashi, N.; Shigyo, M. Comparative study on phytochemical variations in Japanese F1 varieties of bulb onions and south-east Asian shallot landraces. Hort. J. 2018, 87, 63–72. [Google Scholar] [CrossRef]
- Poblocka-Olech, L.; Glod, D.; Żebrowska, M.; Sznitowska, M.; Krause-Baranowsk, M. TLC determination of flavonoids from different cultivars of Allium cepa and Allium ascalonicum. Acta Pharm. 2016, 66, 543–554. [Google Scholar] [CrossRef]
- Ruairidh, J.; Sawers, H.; Ramírez-Flores, M.R.; Olalde-Portugal, V.; Paszkowsk, U. The impact of domestication and crop improvement on arbuscular mycorrhizal symbiosis in cereals: Insights from genetics and genomics. New Phytol. 2018, 220, 1135–1139. [Google Scholar]
- Takeda, N.; Handa, Y.; Tsuzuki, S.; Kojima, M.; Sakakibara, H.; Kawaguchi, M. Gibberellin regulates infection and colonization of host roots by arbuscular mycorrhizal fungi. Plant Signal. Behav. 2015, 10, e1028706. [Google Scholar] [CrossRef]
- Soldatov, S.A.; Khryanin, V.N. Sodium selenite effect on hormonal status and sex in hemp. Doklady RAAS 2006, 2, 13–17. (In Russian) [Google Scholar]
- Berhow, M.A. Effects of early plant growth regulator treatments on flavonoid levels in grapefruit. Plant Growth Reg. 2000, 30, 225–232. [Google Scholar] [CrossRef]
| Experimental Factor | Mycorrhizal Index | Growth Indices | Marketable Bulbs | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| % | LAI (m2·m−2) | Dry Matter (g·m−2) | Yield (Mg·ha−1) | Mean Weight (g) | Number per Plant | |||||
| Microorganism inoculation | ||||||||||
| AMF formulate | 65.7 ± 2.6 | 1.25 ± 0.11 | 2.91 ± 0.23 | 54.9 ± 2.4 | 22.3 ± 1.4 | 6.0 ± 0.3 | ||||
| Non-inoculated control | 26.4 ± 1.1 | 0.82 ± 0.06 | 1.92 ± 0.15 | 37.3 ± 1.7 | 15.2 ± 0.9 | 6.1 ± 0.3 | ||||
| * | * | * | * | * | n.s. | |||||
| Selenium treatment | ||||||||||
| Selenocystine | 47.5 ± 1.8 | 1.18 ± 0.11 | a | 2.77 ± 0.23 | a | 52.2 ± 2.1 | a | 22.3 ± 1.5 | a | 5.8 ± 0.3 |
| Sodium selenate | 46.0 ± 1.5 | 1.00 ± 0.07 | b | 2.34 ± 0.16 | b | 45.0 ± 1.7 | b | 17.6 ± 1.1 | b | 6.3 ± 0.4 |
| Non-treated control | 44.7 ± 1.6 | 0.92 ± 0.07 | b | 2.14 ± 0.13 | b | 41.2 ± 2.3 | b | 16.5 ± 1.2 | b | 6.2 ± 0.3 |
| n.s. | n.s. | |||||||||
| Experimental Factor | DM (%) | SS (°Brix) | TS (g·100 g−1d.w.) | MS(%) | TA mg Citric Acid·100 g−1d.w. | TP (g·100 g−1d.w.) | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Microorganism inoculation | |||||||||||
| AMF formulate | 18.6 ± 0.06 | 15.3 ± 0.07 | 70.2 ± 0.39 | 10.2 ± 0.1 | 2.10 ± 0.08 | 10.2 ± 0.30 | |||||
| Non-inoculated control | 18.2 ± 0.06 | 14.4 ± 0.08 | 64.5 ± 0.67 | 8.4 ± 0.1 | 1.77 ± 0.03 | 9.3 ± 0.23 | |||||
| n.s. | * | * | * | * | * | ||||||
| Selenium treatment | |||||||||||
| Selenocystine | 18.7 ± 0.09 | 15.0 ± 0.06 | ab | 67.5 ± 0.94 | ab | 9.0 ± 0.16 | b | 2.05 ± 0.08 | a | 10.2 ± 0.18 | a |
| Sodium selenate | 18.5 ± 0.13 | 15.3 ± 0.15 | a | 70.6 ± 1.25 | a | 10.3 ± 0.35 | a | 1.95 ± 0.12 | a | 9.8 ± 0.10 | ab |
| Non-treated control | 18.2 ± 0.05 | 14.4 ± 0.20 | b | 64.2 ± 1.18 | b | 8.7 ± 0.23 | b | 1.80 ± 0.09 | b | 9.3 ± 0.25 | b |
| n.s. | |||||||||||
| Experimental Factor | K | Ca | Mg | Na | P | S | Cl | NO3− | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Microorganism inoculation | ||||||||||||||
| AMF formulate | 8981 ± 850 | 1364 ± 135 | 1043 ± 28 | 986 ± 134 | 3078 ± 145 | 706 ± 42 | 1704 ± 98 | 716 ± 117 | ||||||
| Non-inoculated control | 8195 ± 718 | 1003 ± 105 | 938 ± 52 | 935 ± 112 | 2716 ± 122 | 608 ± 46 | 1488 ± 85 | 551 ± 55 | ||||||
| * | * | * | * | * | * | * | * | |||||||
| Selenium treatment | ||||||||||||||
| Selenocystine | 9228 ± 304 | a | 1332 ± 207 | a | 1001 ± 27 | 1049 ± 47 | a | 2932 ± 124 | 702 ± 55 | a | 1663 ± 112 | a | 729 ± 114 | a |
| Sodium selenate | 9125 ± 581 | a | 1216 ± 176 | a | 997 ± 18 | 1057 ± 22 | a | 2845 ± 97 | 658 ± 51 | ab | 1615 ± 129 | ab | 667 ± 98 | b |
| Non-treated control | 7412 ± 294 | b | 1003 ± 158 | b | 974 ± 112 | 776 ± 9 | b | 2915 ± 382 | 612 ± 67 | b | 1511 ± 104 | b | 505 ± 37 | c |
| n.s. | n.s. | |||||||||||||
| Element | Non-Inoculated | AMF Formulate | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Non-Se | Selenocystine | Sodium Selenate | Non-Se | Selenocystine | Sodium Selenate | |||||||
| K | 7118 ± 594 | c | 8924 ± 931 | ab | 8544 ± 810 | bc | 7706 ± 789 | bc | 9531 ± 938c | a | 9706 ± 873c | a |
| NO3 | 469 ± 35 | d | 615 ± 59 | bc | 569 ± 44 | bc | 541 ± 32 | cd | 842 ± 79c | a | 765 ± 73c | a |
| Experimental Factor | Cu | Fe | Zn | Se | B | Mn | Si | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Microorganism inoculation | ||||||||||||
| AMF formulate | 4.31 ± 0.28 | 65.4 ± 11.2 | 18.7 ± 3.4 | 4.66 ± 2.38 | 8.56 ± 1.11 | 881 ± 0.83 | 15.7 ± 1.2 | |||||
| Non-inoculated control | 3.75 ± 0.36 | 29.8 ± 2.9 | 12.4 ± 1.0 | 0.88 ± 0.57 | 6.84 ± 0.42 | 7.19 ± 1.03 | 11.8 ± 1.1 | |||||
| * | * | * | * | * | * | |||||||
| Selenium treatment | ||||||||||||
| Selenocystine | 4.10 ± 0.20 | 43.7 ± 15.9 | b | 16.1 ± 2.2 | a | 5.14 ± 3.64 | a | 7.47 ± 0.35 | b | 7.99 ± 0.02 | 14.4 ± 2.5 | b |
| Sodium selenate | 4.00 ± 0.12 | 40.9 ± 13.6 | b | 13.8 ± 1.49 | b | 3.03 ± 1.91 | b | 7.42 ± 0.23 | b | 8.16 ± 0.21 | 12.7 ± 2.7 | c |
| Non-treated control | 3.97 ± 0.76 | 58.2 ± 24.1 | a | 13.5 ± 2.5 | b | 0.14 ± 0.11 | c | 8.22 ± 2.01 | a | 7.85 ± 2.20 | 18.6 ± 5.24 | a |
| n.s. | n.s. | |||||||||||
| Element | Non-Inoculated Control | AMF Formulate | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Non-Se | Selenocystine | Sodium Selenate | Non-Se | Selenocystine | Sodium Selenate | |||||||
| B | 6.21 ± 0.62 | c | 7.12 ± 0.71 | b | 7.19 ± 0.72 | b | 10.23 ± 1.02 | a | 7.81 ± 0.78 | b | 7.65 ± 0.76 | b |
| Cu | 3.21 ± 0.32 | b | 3.91 ± 0.39 | ab | 4.14 ± 0.41 | a | 4.73 ± 0.47 | a | 4.30 ± 0.43 | a | 3.90 ± 0.39 | ab |
| Fe | 34.1 ± 3.4 | c | 27.8 ± 2.8 | c | 27.3 ± 2.7 | c | 82.3 ± 8.2 | a | 59.5 ± 6.0 | b | 54.5 ± 5.5 | b |
| Mn | 5.65 ± 0.56 | c | 7.97 ± 0.80 | b | 7.95 ± 0.80 | b | 10.05 ± 1.00 | a | 8.01 ± 0.80 | b | 8.37 ± 0.84 | ab |
| Zn | 11.00 ± 1.10 | d | 13.91 ± 1.39 | bc | 12.33 ± 1.23 | cd | 15.92 ± 1.59 | ab | 18.28 ± 1.83 | a | 15.31 ± 1.53 | ab |
| Si | 13.36 ± 1.34 | cd | 11.87 ± 1.19 | de | 10.04 ± 1.00 | e | 23.84 ± 2.38 | a | 16.86 ± 1.69 | b | 15.36 ± 1.54 | bc |
| B | Cu | Fe | Mn | Zn | Si | Ca | K | Mg | Na | P | NO3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cu | 0.89 | 1 | ||||||||||
| Fe | 0.88 | 0.69 | 1 | |||||||||
| Mn | 0.91 ** | 0.93 ** | 0.70 | 1 | ||||||||
| Zn | 0.60 | 0.70 | 0.71 | 0.63 | 1 | |||||||
| Si | 0.89 | 0.64 | 0.97 ** | 0.66 | 0.60 | 1 | ||||||
| Ca | 0.35 | 0.51 | 0.50 | 0.48 | 0.93 ** | 0.33 | 1 | |||||
| K | −0.02 | 0.25 | 0.02 | 0.29 | 0.62 | −0.16 | 0.85 | 1 | ||||
| Mg | 0.89 | 0.95 ** | 0.76 | 0.97 ** | 0.80 | 0.68 | 0.67 | 0.43 | 1 | |||
| Na | −0.21 | 0.16 | −0.21 | 0.13 | 0.44 | −0.40 | 0.71 | 0.95 ** | 0.27 | 1 | ||
| P | 0.95 ** | 0.91 ** | 0.86 | 0.95 ** | 0.78 | 0.83 | 0.57 | 0.25 | 0.96 ** | 0.04 | 1 | |
| NO3 | 0.08 | 0.29 | 0.26 | 0.25 | 0.80 | 0.077 | 0.96 ** | 0.92 ** | 0.46 | 0.83 | 0.33 | |
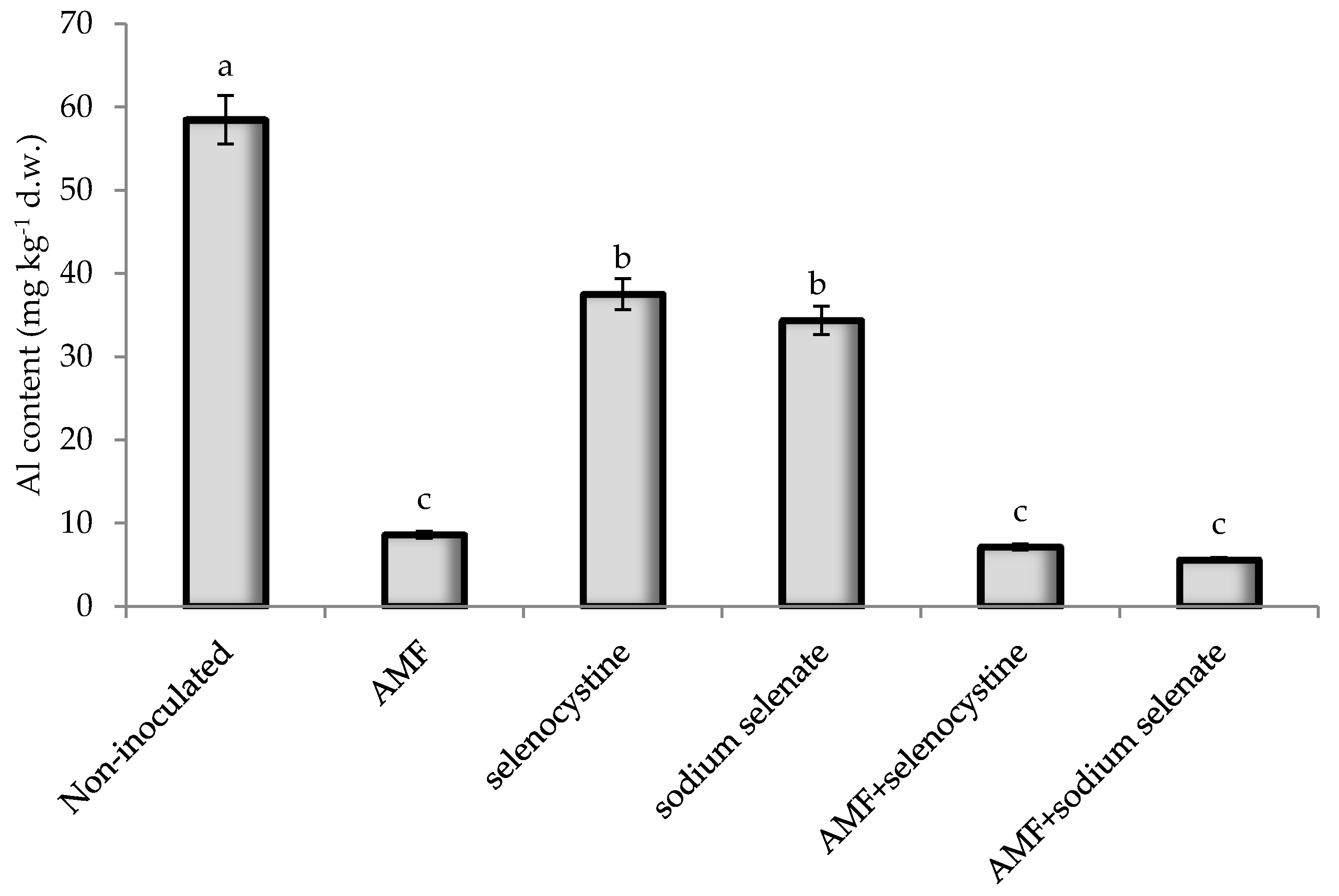
| Al | −0.72 | −0.76 | −0.77 | −0.80 | −0.89 | −0.62 | −0.87 | −0.63 | 0.91 ** | 0.45 | −0.84 | 0.72 |
| Experimental Factor | Polyphenols mg·g−1d.w. | Flavonoids mg Quercetin·g−1d.w. | Ascorbic Acid mg·g−1d.w. | AOA mg Gallic Acid·g−1d.w. | |
|---|---|---|---|---|---|
| Microorganism inoculation | |||||
| AMF formulate | 10.0 ± 0.12 | 0.23 ± 0.01 | 0.51 ± 0.01 | 17.5 ± 0.53 | |
| Non-inoculated control | 9.7 ± 0.12 | 0.21 ± 0.01 | 0.45 ± 0.02 | 16.2 ± 0.47 | |
| n.s. | n.s. | * | * | ||
| Selenium treatment | |||||
| Selenocystine | 10.1 ± 0.19 | 0.22 ± 0.03 | 0.50 ± 0.03 | a | 17.1 ± 1.49 |
| Sodium selenate | 9.9 ± 0.18 | 0.23 ± 0.02 | 0.53 ± 0.01 | a | 16.8 ± 1.58 |
| Non-treated control | 9.6 ± 0.10 | 0.22 ± 0.03 | 0.43 ± 0.01 | b | 16.7 ± 1.26 |
| n.s. | n.s. | n.s. | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golubkina, N.; Zamana, S.; Seredin, T.; Poluboyarinov, P.; Sokolov, S.; Baranova, H.; Krivenkov, L.; Pietrantonio, L.; Caruso, G. Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs. Plants 2019, 8, 102. https://doi.org/10.3390/plants8040102
Golubkina N, Zamana S, Seredin T, Poluboyarinov P, Sokolov S, Baranova H, Krivenkov L, Pietrantonio L, Caruso G. Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs. Plants. 2019; 8(4):102. https://doi.org/10.3390/plants8040102
Chicago/Turabian StyleGolubkina, Nadezhda, Svetlana Zamana, Timofei Seredin, Pavel Poluboyarinov, Sergei Sokolov, Helene Baranova, Leonid Krivenkov, Laura Pietrantonio, and Gianluca Caruso. 2019. "Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs" Plants 8, no. 4: 102. https://doi.org/10.3390/plants8040102
APA StyleGolubkina, N., Zamana, S., Seredin, T., Poluboyarinov, P., Sokolov, S., Baranova, H., Krivenkov, L., Pietrantonio, L., & Caruso, G. (2019). Effect of Selenium Biofortification and Beneficial Microorganism Inoculation on Yield, Quality and Antioxidant Properties of Shallot Bulbs. Plants, 8(4), 102. https://doi.org/10.3390/plants8040102