Arabinogalactan-Proteins from the Liverwort Marchantia polymorpha L., a Member of a Basal Land Plant Lineage, Are Structurally Different to Those of Angiosperms
Abstract
1. Introduction
2. Material and Methods
2.1. Plant Material
2.2. Isolation of Different Fractions of the Cell Wall
2.3. Isolation of AGP
2.4. Gel Diffusion Assay
2.5. Hydrolysis of AGP with Trifluoroacetic Acid (TFA)
2.6. Determination of Uronic Acids
2.7. Analysis of Neutral Monosaccharides
2.8. Structure Elucidation of Arabinogalactan Moiety
2.9. Elemental Analysis
2.10. Determination of Hyp Content
2.11. Determination of Molecular Weight
2.12. Enzyme-Linked Immunosorbent Assay (ELISA)
3. Results and Discussion
3.1. Isolation of Different Fractions of the Cell Wall
3.2. Isolation and Characterization of AGPs
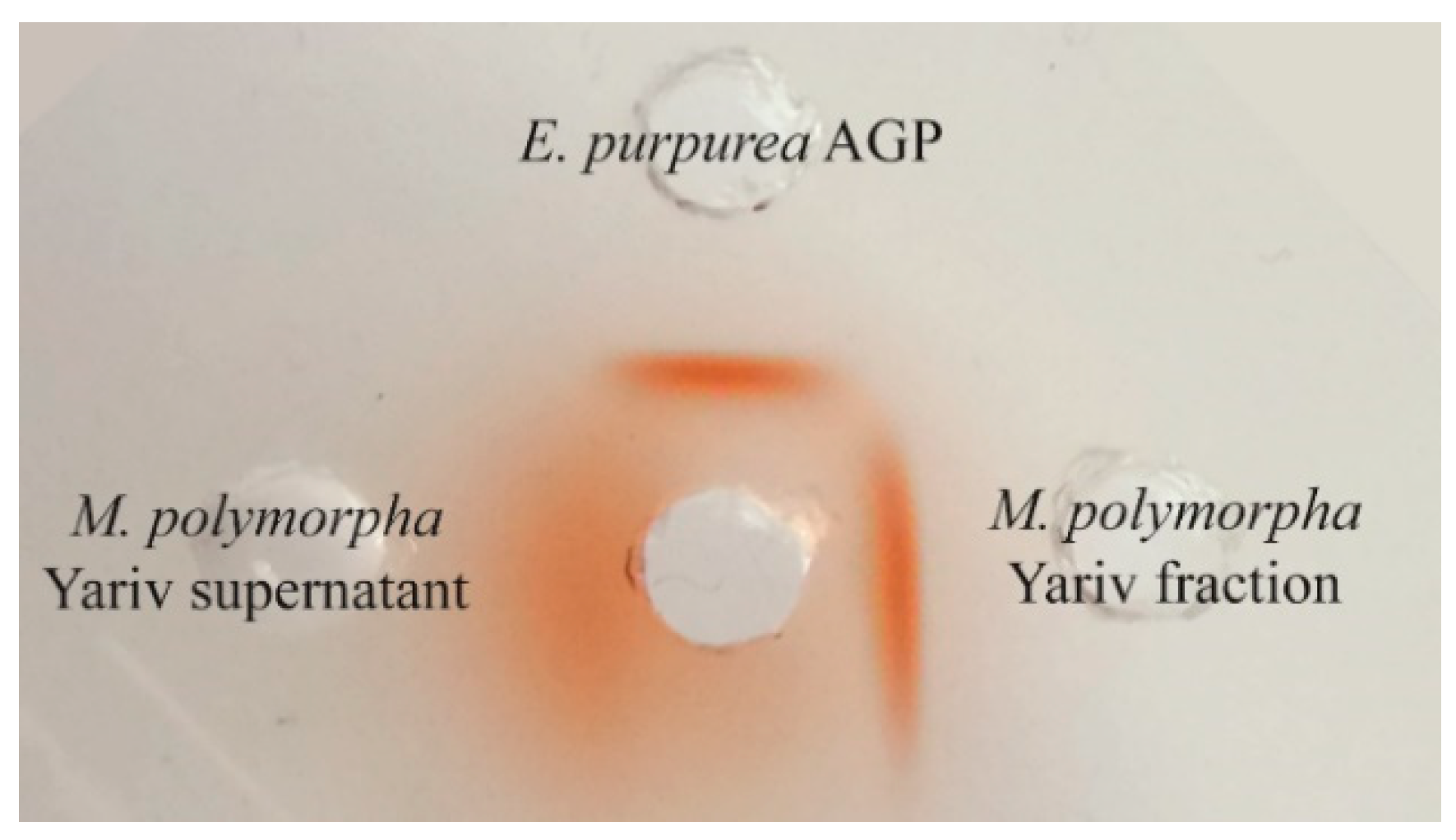
3.2.1. Yield and Gel Diffusion Assay
3.2.2. Analyses of the Protein Moiety
3.2.3. Analyses of the Carbohydrate Moiety
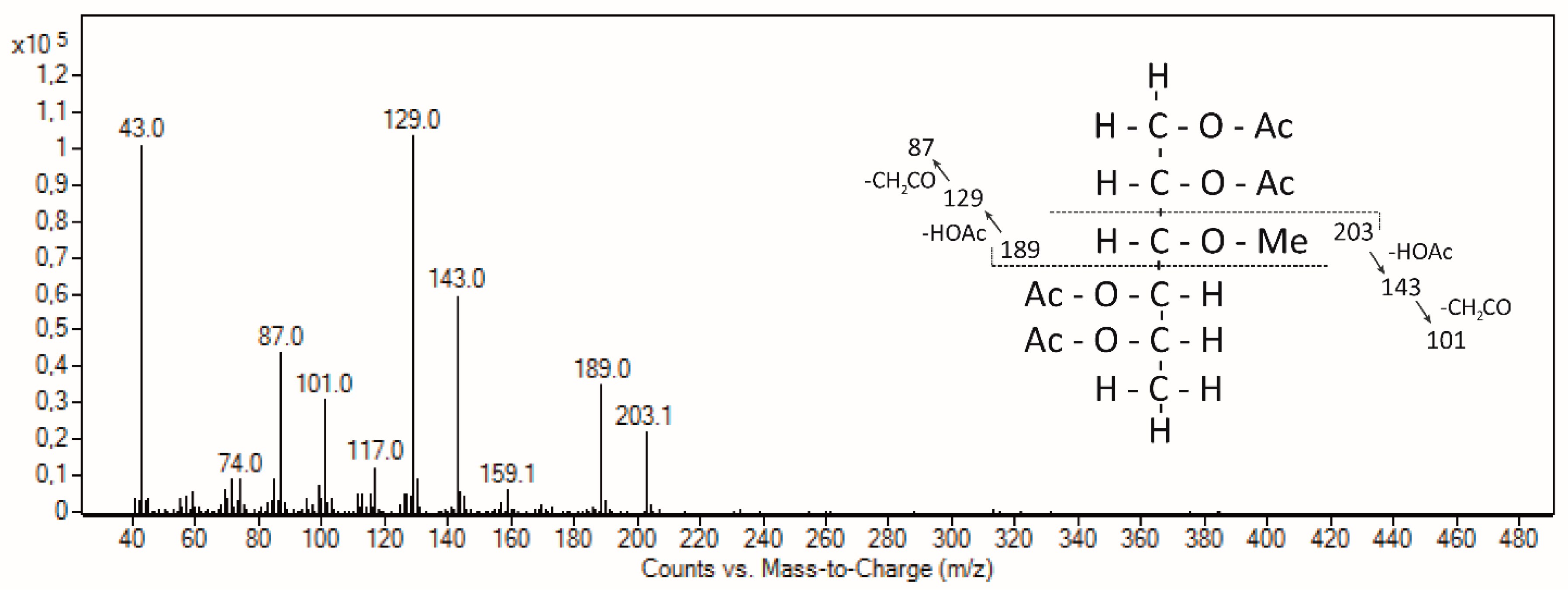
3.2.4. Structure of AGP
3.2.5. Determination of Molecular Weight by Size-Exclusion Chromatography
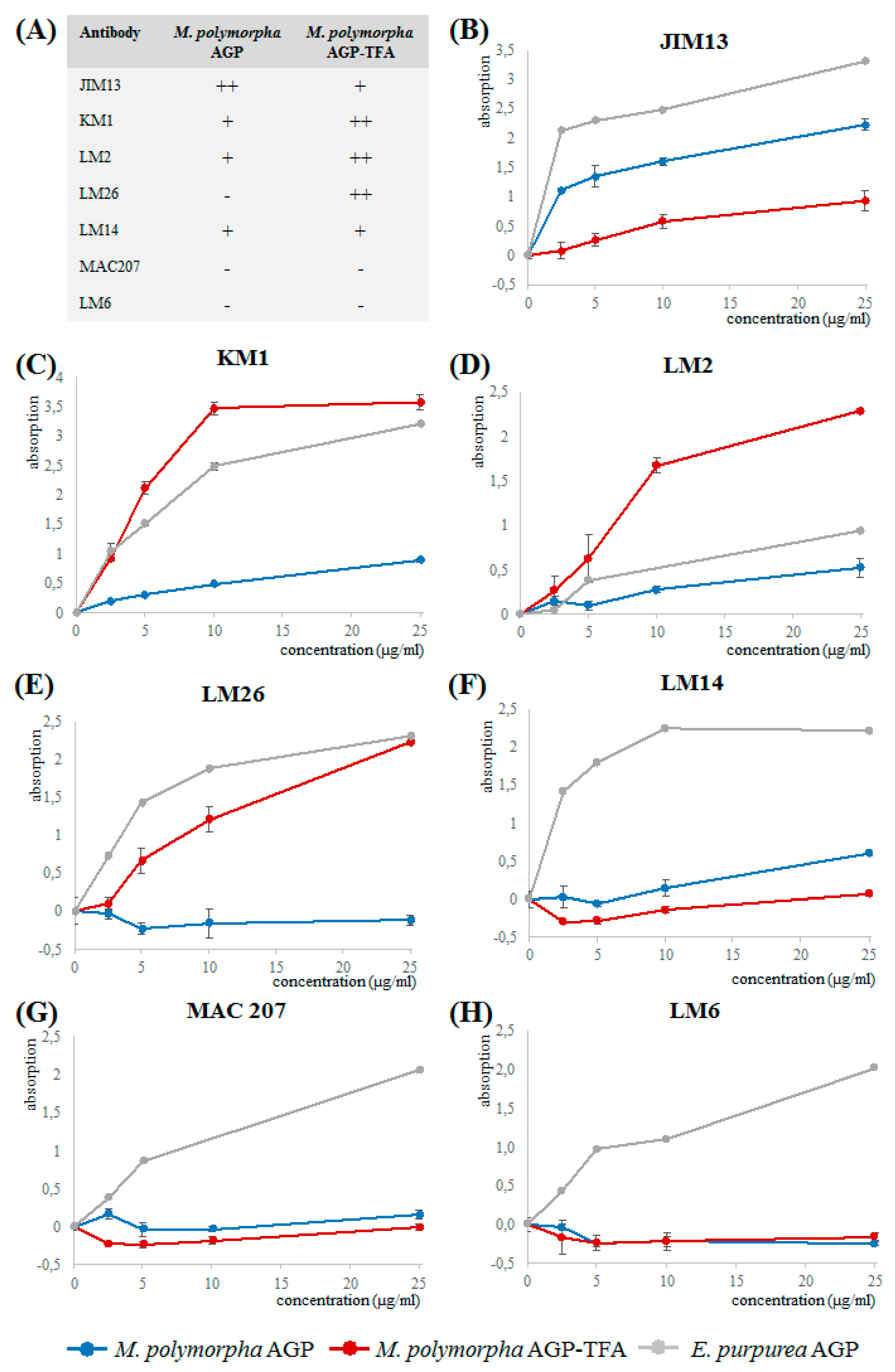
3.3. Reactivity of M. polymorpha AGP with Antibodies Directed against Angiosperm AGPs in ELISA
3.4. Functions of AGPs in Bryophytes
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Delwiche, C.F.; Cooper, E.D. The Evolutionary Origin of a Terrestrial Flora. Curr. Biol. 2015, 25, R899–R910. [Google Scholar] [CrossRef] [PubMed]
- Bowman, J.L.; Kohchi, T.; Yamato, K.T.; Jenkins, J.; Shu, S.; Ishizaki, K.; Yamaoka, S.; Nishihama, R.; Nakamura, Y.; Berger, F.; et al. Insights into Land Plant Evolution Garnered from the Marchantia polymorpha Genome. Cell 2017, 171, 287–304.e15. [Google Scholar] [CrossRef] [PubMed]
- Jill Harrison, C. Development and genetics in the evolution of land plant body plans. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2017, 372. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.; Archibald, J.M. Plant evolution: Landmarks on the path to terrestrial life. New Phytol. 2018, 217, 1428–1434. [Google Scholar] [CrossRef]
- Morris, J.L.; Puttick, M.N.; Clark, J.W.; Edwards, D.; Kenrick, P.; Pressel, S.; Wellman, C.H.; Yang, Z.; Schneider, H.; Donoghue, P.C.J. The timescale of early land plant evolution. Proc. Natl. Acad. Sci. USA 2018, 115, E2274–E2283. [Google Scholar] [CrossRef]
- Puttick, M.N.; Morris, J.L.; Williams, T.A.; Cox, C.J.; Edwards, D.; Kenrick, P.; Pressel, S.; Wellman, C.H.; Schneider, H.; Pisani, D.; et al. The Interrelationships of Land Plants and the Nature of the Ancestral Embryophyte. Curr. Biol. 2018, 28, 733–745.e2. [Google Scholar] [CrossRef]
- Kenrick, P.; Crane, P.R. The origin and early evolution of plants on land. Nature 1997, 389, 33–39. [Google Scholar] [CrossRef]
- Ligrone, R.; Duckett, J.G.; Renzaglia, K.S. Major transitions in the evolution of early land plants: A bryological perspective. Ann. Bot. 2012, 109, 851–871. [Google Scholar] [CrossRef]
- Bowman, J.L.; Araki, T.; Arteaga-Vazquez, M.A.; Berger, F.; Dolan, L.; Haseloff, J.; Ishizaki, K.; Kyozuka, J.; Lin, S.-S.; Nagasaki, H.; et al. The Naming of Names: Guidelines for Gene Nomenclature in Marchantia. Plant Cell Physiol. 2016, 57, 257–261. [Google Scholar] [CrossRef]
- Rensing, S.A. Why we need more non-seed plant models. New Phytol. 2017, 216, 355–360. [Google Scholar] [CrossRef]
- Delwiche, C.F.; Goodman, C.A.; Chang, C. Land Plant Model Systems Branch Out. Cell 2017, 171, 265–266. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, I.; Domozych, D.; Willats, W.G.T. How have plant cell walls evolved? Plant Physiol. 2010, 153, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, M.D.; Harholt, J.; Ulvskov, P.; Johansen, I.E.; Fangel, J.U.; Doblin, M.S.; Bacic, A.; Willats, W.G.T. Evidence for land plant cell wall biosynthetic mechanisms in charophyte green algae. Ann. Bot. 2014, 114, 1217–1236. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, P.; Bosneaga, E.; Auer, M. Plant cell walls throughout evolution: Towards a molecular understanding of their design principles. J. Exp. Bot. 2009, 60, 3615–3635. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zeng, W.; Bacic, A.; Johnson, K. AGPs Through Time and Space. In Annual Plant Reviews Online; American Cancer Society: Atlanta, GA, USA, 2018; pp. 1–38. [Google Scholar]
- Ellis, M.; Egelund, J.; Schultz, C.J.; Bacic, A. Arabinogalactan-proteins: Key regulators at the cell surface? Plant Physiol. 2010, 153, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Seifert, G.J.; Roberts, K. The biology of arabinogalactan proteins. Annu. Rev. Plant Biol. 2007, 58, 137–161. [Google Scholar] [CrossRef]
- Mareri, L.; Romi, M.; Cai, G. Arabinogalactan proteins: Actors or spectators during abiotic and biotic stress in plants? Plant Biosyst. 2018, 153, 173–185. [Google Scholar] [CrossRef]
- Johnson, K.L.; Jones, B.J.; Schultz, C.J.; Bacic, A. Non-enzymic cell wall (glyco) proteins. Plant Cell Wall 2003, 8, 111–154. [Google Scholar]
- Classen, B.; Baumann, A.; Utermoehlen, J. Arabinogalactan-proteins in spore-producing land plants. Carbohydr. Polym. 2019, 210, 215–224. [Google Scholar] [CrossRef]
- Kremer, C.; Pettolino, F.; Bacic, A.; Drinnan, A. Distribution of cell wall components in Sphagnum hyaline cells and in liverwort and hornwort elaters. Planta 2004, 219, 1023–1035. [Google Scholar] [CrossRef]
- Raimundo, S.C.; Avci, U.; Hopper, C.; Pattathil, S.; Hahn, M.G.; Popper, Z.A. Immunolocalization of cell wall carbohydrate epitopes in seaweeds: Presence of land plant epitopes in Fucus vesiculosus L. (Phaeophyceae). Planta 2016, 243, 337–354. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, C.; Gregson, T.; Murray, L.; Sadler, I.H.; Fry, S.C. Sugar composition of the pectic polysaccharides of charophytes, the closest algal relatives of land-plants: Presence of 3-O-methyl-d-galactose residues. Ann. Bot. 2015, 116, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Classen, B.; Mau, S.L.; Bacic, A. The arabinogalactan-proteins from pressed juice of Echinacea purpurea belong to the hybrid class of hydroxyproline-rich glycoproteins. Planta Med. 2005, 71, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 34, 484–489. [Google Scholar] [CrossRef]
- Blakeney, A.B.; Harris, P.J.; Henry, R.J.; Stone, B.A. A simple and rapid preparation of alditol acetates for monosaccharide analysis. Carbohydr. Res. 1983, 113, 291–299. [Google Scholar] [CrossRef]
- Harris, P.J.; Henry, R.J.; Blakeney, A.B.; Stone, B.A. An improved procedure for the methylation analysis of oligosaccharides and polysaccharides. Carbohydr. Res. 1984, 127, 59–73. [Google Scholar] [CrossRef]
- Sweet, D.P.; Shapiro, R.H.; Albersheim, P. Quantitative analysis by various g.l.c. response-factor theories for partially methylated and partially ethylated alditol acetates. Carbohydr. Res. 1975, 40, 217–225. [Google Scholar] [CrossRef]
- Stegemann, H.; Stalder, K. Determination of Hydroxyproline. Clin. Chim. Acta 1967, 18, 267–273. [Google Scholar] [CrossRef]
- Classen, B.; Csávás, M.; Borbás, A.; Dingermann, T.; Zündorf, I. Monoclonal antibodies against an arabinogalactan-protein from pressed juice of Echinacea purpurea. Planta Med. 2004, 70, 861–865. [Google Scholar] [CrossRef]
- Bartels, D.; Baumann, A.; Maeder, M.; Geske, T.; Heise, E.M.; von Schwartzenberg, K.; Classen, B. Evolution of plant cell wall: Arabinogalactan-proteins from three moss genera show structural differences compared to seed plants. Carbohydr. Polym. 2017, 163, 227–235. [Google Scholar] [CrossRef]
- Carafa, A.; Duckett, J.G.; Knox, J.P.; Ligrone, R. Distribution of cell-wall xylans in bryophytes and tracheophytes: New insights into basal interrelationships of land plants. New Phytol. 2005, 168, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Dehors, J.; Mareck, A.; Kiefer-Meyer, M.-C.; Menu-Bouaouiche, L.; Lehner, A.; Mollet, J.-C. Evolution of Cell Wall Polymers in Tip-Growing Land Plant Gametophytes: Composition, Distribution, Functional Aspects and Their Remodeling. Front. Plant Sci. 2019, 10, 441. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Ishii, T.; Matsumoto, S.; Higuchi, M.; Darvill, A.; Albersheim, P.; O’Neill, M.A. Occurrence of the primary cell wall polysaccharide rhamnogalacturonan II in pteridophytes, lycophytes, and bryophytes. Implications for the evolution of vascular plants. Plant Physiol. 2004, 134, 339–351. [Google Scholar] [CrossRef] [PubMed]
- Popper, Z.A.; Fry, S.C. Primary cell wall composition of bryophytes and charophytes. Ann. Bot. 2003, 91, 1–12. [Google Scholar] [CrossRef]
- Popper, Z.A.; Tuohy, M.G. Beyond the green: Understanding the evolutionary puzzle of plant and algal cell walls. Plant Physiol. 2010, 153, 373–383. [Google Scholar] [CrossRef]
- Roberts, A.W.; Roberts, E.M.; Haigler, C.H. Moss cell walls: Structure and biosynthesis. Front. Plant Sci. 2012, 3, 166. [Google Scholar] [CrossRef]
- O’Neill, M.A.; Ishii, T.; Albersheim, P.; Darvill, A.G. Rhamnogalacturonan II: Structure and function of a borate cross-linked cell wall pectic polysaccharide. Annu. Rev. Plant Biol. 2004, 55, 109–139. [Google Scholar] [CrossRef]
- Fu, H.; Yadav, M.P.; Nothnagel, E.A. Physcomitrella patens arabinogalactan proteins contain abundant terminal 3-O-methyl-l-rhamnosyl residues not found in angiosperms. Planta 2007, 226, 1511–1524. [Google Scholar] [CrossRef]
- Popper, Z.A.; Sadler, I.H.; Fry, S.C. 3-O-Methylrhamnose in lower land plant primary cell walls. Biochem. Syst. Ecol. 2004, 32, 279–289. [Google Scholar] [CrossRef]
- Konno, H.; Yamasaki, Y.; Katoh, K. Fractionation and partial characterization of pectic polysaccharides in cell walls from liverwort (Marchantia polymorpha) cell cultures. J. Exp. Bot. 1987, 38, 711–722. [Google Scholar] [CrossRef]
- Galloway, A.F.; Pedersen, M.J.; Merry, B.; Marcus, S.E.; Blacker, J.; Benning, L.G.; Field, K.J.; Knox, J.P. Xyloglucan is released by plants and promotes soil particle aggregation. New Phytol. 2018, 217, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- Peña, M.J.; Darvill, A.G.; Eberhard, S.; York, W.S.; O’Neill, M.A. Moss and liverwort xyloglucans contain galacturonic acid and are structurally distinct from the xyloglucans synthesized by hornworts and vascular plants. Glycobiology 2008, 18, 891–904. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.L.; Cassin, A.M.; Lonsdale, A.; Wong, G.K.-S.; Soltis, D.E.; Miles, N.W.; Melkonian, M.; Melkonian, B.; Deyholos, M.K.; Leebens-Mack, J.; et al. Insights into the Evolution of Hydroxyproline-Rich Glycoproteins from 1000 Plant Transcriptomes. Plant Physiol. 2017, 174, 904–921. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Zhao, H.; Cheng, Z.; Ke, Y.; Liu, J.; Ma, H. Evolution Analysis of the Fasciclin-Like Arabinogalactan Proteins in Plants Shows Variable Fasciclin-AGP Domain Constitutions. Int. J. Mol. Sci. 2019, 20, 1945. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.E.; Gleeson, P.A.; Jermyn, M.A.; Knox, R.B. Characterization and Localization of β-Lectins in Lower and Higher Plants. Aust. J. Plant Phy. 1978, 5, 707. [Google Scholar] [CrossRef]
- Ligrone, R.; Vaughn, K.C.; Renzaglia, K.S.; Knox, J.P.; Duckett, J.G. Diversity in the distribution of polysaccharide and glycoprotein epitopes in the cell walls of bryophytes: New evidence for the multiple evolution of water-conducting cells. New Phytol. 2002, 156, 491–508. [Google Scholar] [CrossRef]
- Shibaya, T.; Kaneko, Y.; Sugawara, Y. Involvement of arabinogalactan proteins in protonemata development from cultured cells of Marchantia polymorpha. Physiol. Plant. 2005, 124, 504–514. [Google Scholar] [CrossRef]
- Shibaya, T.; Sugawara, Y. Induction of multinucleation by beta-glucosyl Yariv reagent in regenerated cells from Marchantia polymorpha protoplasts and involvement of arabinogalactan proteins in cell plate formation. Planta 2009, 230, 581–588. [Google Scholar] [CrossRef]
- Lee, K.J.D.; Sakata, Y.; Mau, S.-L.; Pettolino, F.; Bacic, A.; Quatrano, R.S.; Knight, C.D.; Knox, J.P. Arabinogalactan proteins are required for apical cell extension in the moss Physcomitrella patens. Plant Cell 2005, 17, 3051–3065. [Google Scholar] [CrossRef]
- Bartels, D.; Classen, B. Structural investigations on arabinogalactan-proteins from a lycophyte and different monilophytes (ferns) in the evolutionary context. Carbohydr. Polym. 2017, 172, 342–351. [Google Scholar] [CrossRef]
- Knox, J.P.; Linstead, P.J.; Peart, J.; Cooper, C.; Roberts, K. Developmentally regulated epitopes of cell surface arabinogalactan proteins and their relation to root tissue pattern formation. Plant J. 1991, 1, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Yates, E.A.; Valdor, J.F.; Haslam, S.M.; Morris, H.R.; Dell, A.; Mackie, W.; Knox, J.P. Characterization of carbohydrate structural features recognized by anti-arabinogalactan-protein monoclonal antibodies. Glycobiology 1996, 6, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Pattathil, S.; Avci, U.; Baldwin, D.; Swennes, A.G.; McGill, J.A.; Popper, Z.; Bootten, T.; Albert, A.; Davis, R.H.; Chennareddy, C.; et al. A comprehensive toolkit of plant cell wall glycan-directed monoclonal antibodies. Plant Physiol. 2010, 153, 514–525. [Google Scholar] [CrossRef] [PubMed]
- Pennell, R.I.; Knox, J.P.; Scofield, G.N.; Selvendran, R.R.; Roberts, K. A family of abundant plasma membrane-associated glycoproteins related to the arabinogalactan proteins is unique to flowering plants. J. Cell Biol. 1989, 108, 1967–1977. [Google Scholar] [CrossRef]
- Ruprecht, C.; Bartetzko, M.P.; Senf, D.; Dallabernadina, P.; Boos, I.; Andersen, M.C.F.; Kotake, T.; Knox, J.P.; Hahn, M.G.; Clausen, M.H.; et al. A Synthetic Glycan Microarray Enables Epitope Mapping of Plant Cell Wall Glycan-Directed Antibodies. Plant Physiol. 2017, 175, 1094–1104. [Google Scholar] [CrossRef]
- Smallwood, M.; Yates, E.A.; Willats, W.G.T.; Martin, H.; Knox, J.P. Immunochemical comparison of membrane-associated and secreted arabinogalactan-proteins in rice and carrot. Planta 1996, 198, 452–459. [Google Scholar] [CrossRef]
- Willats, W.G.; Marcus, S.E.; Knox, J.P. Generation of monoclonal antibody specific to (1--5)-alpha-l-arabinan. Carbohydr. Res. 1998, 308, 149–152. [Google Scholar] [CrossRef]
- Verhertbruggen, Y.; Marcus, S.E.; Haeger, A.; Verhoef, R.; Schols, H.A.; McCleary, B.V.; McKee, L.; Gilbert, H.J.; Knox, J.P. Developmental complexity of arabinan polysaccharides and their processing in plant cell walls. Plant J. 2009, 59, 413–425. [Google Scholar] [CrossRef]
- Moller, I.; Marcus, S.E.; Haeger, A.; Verhertbruggen, Y.; Verhoef, R.; Schols, H.; Ulvskov, P.; Mikkelsen, J.D.; Knox, J.P.; Willats, W. High-throughput screening of monoclonal antibodies against plant cell wall glycans by hierarchical clustering of their carbohydrate microarray binding profiles. Glycoconj. J. 2008, 25, 37–48. [Google Scholar] [CrossRef]
- Torode, T.A.; O’Neill, R.; Marcus, S.E.; Cornuault, V.; Pose, S.; Lauder, R.P.; Kračun, S.K.; Rydahl, M.G.; Andersen, M.C.F.; Willats, W.G.T.; et al. Branched Pectic Galactan in Phloem-Sieve-Element Cell Walls: Implications for Cell Mechanics. Plant Physiol. 2018, 176, 1547–1558. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Motose, H.; Iwamoto, K.; Fukuda, H. Expression and genome-wide analysis of the xylogen-type gene family. Plant Cell Physiol. 2011, 52, 1095–1106. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Hu, J.; Guo, S.; Wang, J.; Cheng, Y.; Dang, X.; Wu, L.; He, Y. Proteome analysis of Physcomitrella patens exposed to progressive dehydration and rehydration. J. Exp. Bot. 2012, 63, 711–726. [Google Scholar] [CrossRef] [PubMed]
- Shibaya, T.; Sugawara, Y. Involvement of arabinogalactan proteins in the regeneration process of cultured protoplasts of Marchantia polymorpha. Physiol. Plant 2007, 130, 271–279. [Google Scholar] [CrossRef]
| Neutral Monosaccharide | M. polymorpha HMF (n = 3) | M. polymorpha (NH4)2C2O4 (n = 3) | M. polymorpha Na2CO3 (n = 3) | M. polymorpha KOH (n = 3) | ||||
|---|---|---|---|---|---|---|---|---|
| 3-O-Me-Rha | 1.6 | ±0.3 | 1.1 | ±0.0 | 1.0 | ±0.1 | trace | |
| Rha | 4.8 | ±0.1 | 5.7 | ±1.3 | 11.5 | ±0.7 | 7.0 | ±0.1 |
| Fuc | 5.7 | ±0.0 | 5.3 | ±0.1 | 3.2 | ±0.3 | 2.4 | ±0.0 |
| Rib (?) | trace | 4.6 | ±0.0 | trace | trace | |||
| Ara | 17.7 | ±0.1 | 16.0 | ±0.3 | 24.7 | ±0.8 | 17.3 | ±0.1 |
| Xyl | 11.2 | ±0.3 | 10.7 | ±0.1 | 4.9 | ±4.1 | 15.1 | ±0.4 |
| Man | 6.8 | ±0.2 | 7.8 | ±0.1 | 8.9 | ±0.4 | 10.6 | ±0.2 |
| Gal | 20.1 | ±0.5 | 17.5 | ±0.3 | 33.0 | ±1.7 | 24.1 | ±0.1 |
| Glc | 32.2 | ±0.3 | 31.3 | ±0.3 | 12.9 | ±0.6 | 23.3 | ±0.3 |
| M. polymorpha | Uronic Acids |
|---|---|
| HMF | 4.2 ± 0.3 |
| (NH4)2C2O4 | 11.4 ± 0.2 |
| Na2CO3 | 4.9 ± 0.2 |
| KOH | 7.8 ± 0.0 |
| Neutral Monosaccharide | M. polymorpha Yariv Fraction (n = 3) | M. polymorpha Yariv Supernatant (n = 3) | M. polymorpha Yariv Fraction TFA Hydrolysis (n = 1) | ||
|---|---|---|---|---|---|
| Gal | 46.9 | ± 0.3 | 23.4 | ± 0.1 | 79.6 |
| Ara | 31.6 | ± 0.5 | 22.7 | ± 0.1 | 2.1 |
| Glc | 9.5 | ± 0.7 | 9.4 | ± 0.2 | 10.3 |
| 3-O-Me-Rha | 2.5 | ± 0.2 | 2.1 | ± 0.2 | 1.5 |
| Rha | 2.4 | ± 0.1 | 8.0 | ± 0.1 | 1.6 |
| Rib (?) | 2.1 | ± 0.1 | trace | - | |
| Fuc | 1.8 | ± 0.1 | 8.9 | ± 0.1 | trace |
| Xyl | 1.6 | ± 0.1 | 17.8 | ± 0.2 | 2.7 |
| Man | 1.6 | ± 0.0 | 7.6 | ± 0.1 | 2.0 |
| Liverwort | Mosses | |||
|---|---|---|---|---|
| Neutral Monosaccharide % (w/w) | Marchantia polymorpha | Sphagnum sp. * | Physcomitrella patens * | Polytrichastrum formosum * |
| Gal | 46.9 | 65.4 | 41.0 | 64.1 |
| Ara | 31.6 | 9.7 | 33.3 | 15.6 |
| Rha | 2.4 | 3.3 | 1.5 | 8.4 |
| 3-O-Me-Rha | 2.5 | 11.9 | 11.9 | 2.6 |
| others | 16.6 | 9.7 | 12.3 | 9.3 |
| Ara:Gal | 1:1.5 | 1:6.7 | 1:1.2 | 1:4.1 |
| Monosaccharide | Linkage Type | M. polymorpha AGP | M. polymorpha AGP after Partial Acid (TFA) Hydrolysis |
|---|---|---|---|
| Galp | 1,3,6- | 27.1 | 20.6 |
| 1,6- | - | 25.0 | |
| 1,4- | 4.3 | 17.4 | |
| 1,3- | 19.2 | 14.3 | |
| 1- | - | 22.7 | |
| Araf | 1,5- | 2.7 | - |
| 1,3- | 4.3 | - | |
| 1- | 36.3 | - | |
| Rhap | 1,2,4- | 1.5 | - |
| 1,4- | 2.4 | - | |
| 1- | 2.2 | - |
| Antibody | Epitope | Key References |
|---|---|---|
| JIM13 | AGP glycan, e.g., β-D-GlcAp-(1→3)-α-D-GalAp-(1→2)-α-L-Rha | [52,53,54] |
| MAC 207 | AGP glycan, e.g., β-D-GlcAp-(1→3)-α-D-GalAp-(1→2)-α-L-Rha | [53,54,55] |
| KM1 | (1→6)-β-D-Galp units in AGs type II | [30,56] |
| LM2 | (1→6)-β-D-Galp units with terminal ß-D-GlcAp in AGP | [53,56,57] |
| LM6 | (1→5)-α-L-Araf oligomers in arabinan or AGP | [50,56,58,59] |
| LM14 | Type II AG in pectin or AGP glycan | [60] |
| LM26 | Branched (1→4)-β-D-galactan | [61] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Happ, K.; Classen, B. Arabinogalactan-Proteins from the Liverwort Marchantia polymorpha L., a Member of a Basal Land Plant Lineage, Are Structurally Different to Those of Angiosperms. Plants 2019, 8, 460. https://doi.org/10.3390/plants8110460
Happ K, Classen B. Arabinogalactan-Proteins from the Liverwort Marchantia polymorpha L., a Member of a Basal Land Plant Lineage, Are Structurally Different to Those of Angiosperms. Plants. 2019; 8(11):460. https://doi.org/10.3390/plants8110460
Chicago/Turabian StyleHapp, Kathrin, and Birgit Classen. 2019. "Arabinogalactan-Proteins from the Liverwort Marchantia polymorpha L., a Member of a Basal Land Plant Lineage, Are Structurally Different to Those of Angiosperms" Plants 8, no. 11: 460. https://doi.org/10.3390/plants8110460
APA StyleHapp, K., & Classen, B. (2019). Arabinogalactan-Proteins from the Liverwort Marchantia polymorpha L., a Member of a Basal Land Plant Lineage, Are Structurally Different to Those of Angiosperms. Plants, 8(11), 460. https://doi.org/10.3390/plants8110460





