Lipid Thermal Fingerprints of Long-term Stored Seeds of Brassicaceae
Abstract
1. Introduction
2. Results
2.1. Seed Germination
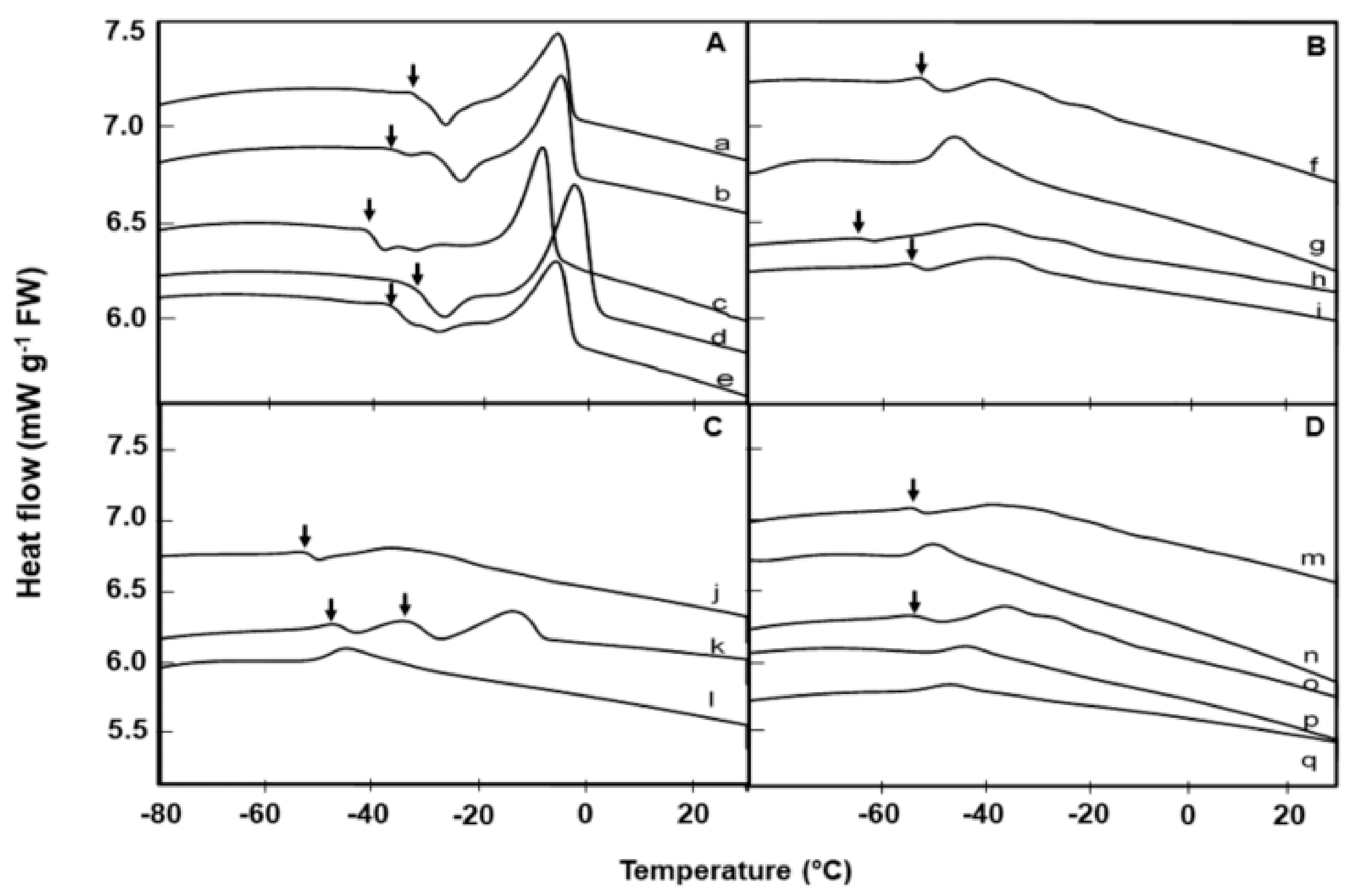
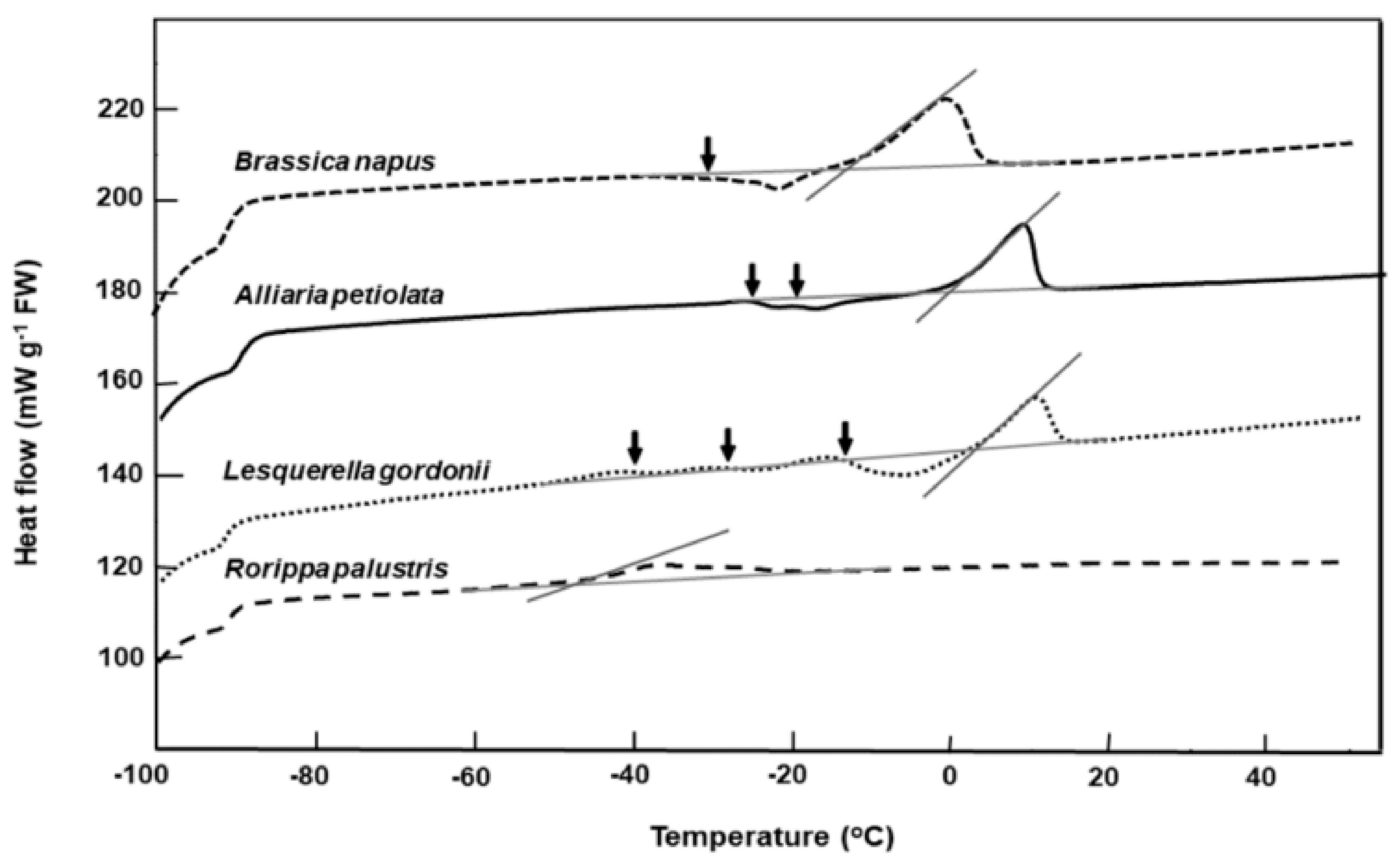
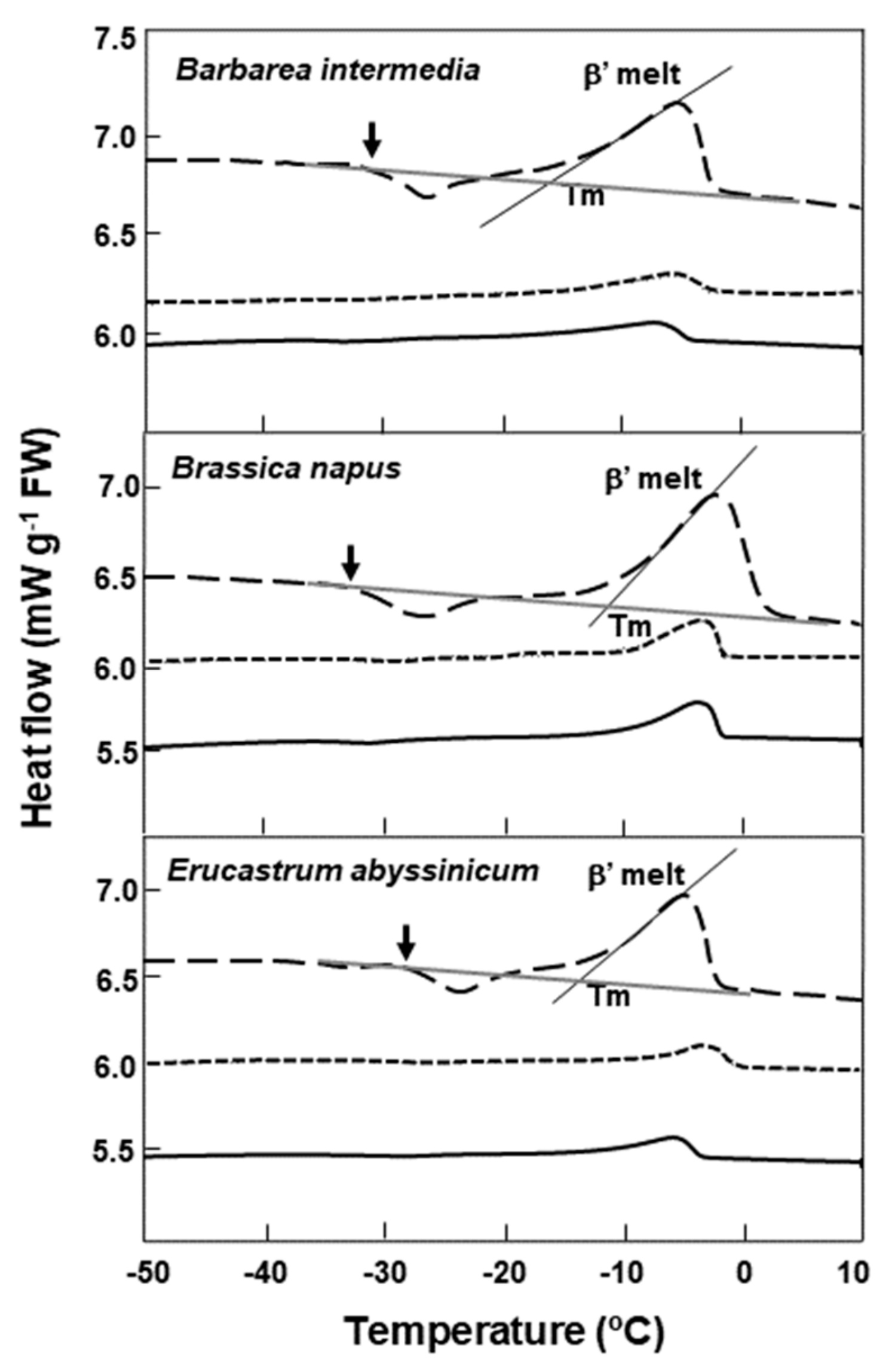
2.2. Lipid Thermal Fingerprints, Lipid Content, and Fatty Acid Compositions
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Germination Assays
4.3. Seed Lipid Thermal Analysis and Fatty Acid Compositions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ellis, R.H.; Hong, T.D.; Martín, M.C.; Pérez-García, F.; Gómez-Campo, C. The long-term storage of seeds of seventeen crucifers at very low moisture contents. Plant Var. Seeds 1993, 6, 75–81. [Google Scholar]
- Pérez-García, F.; González-Benito, M.E.; Gómez-Campo, C. High viability recorded in ultra-dry seeds of 37 species of Brassicaceae after almost 40 years of storage. Seed Sci. Technol. 2007, 35, 143–153. [Google Scholar] [CrossRef]
- Pérez-García, F.; González-Benito, M.E.; Gómez-Campo, C. Germination of fourteen endemic species from the Iberian Peninsula, Canary and Balearic Islands after 32-34 years of storage at low temperature and very low water content. Seed Sci. Technol. 2008, 36, 407–422. [Google Scholar] [CrossRef]
- Pérez-García, F.; Gómez-Campo, C.; Ellis, R.H. Successful long-term ultra-dry storage of seed of 15 species of Brassicaceae in a genebank: Variation in ability to germinate over 40 years and dormancy. Seed Sci. Technol. 2009, 37, 640–649. [Google Scholar] [CrossRef]
- Ellis, R.H.; Hong, T.D.; Roberts, E.H. An Intermediate Category of Seed Storage Behaviour? II. Effects of provenance, immaturity, and imbibition on desiccation-tolerance in coffee. J. Exp. Bot. 1991, 42, 653–657. [Google Scholar] [CrossRef]
- Pritchard, H.W. Classification of Seed Storage ’Types’ for Ex Situ Conservation in Relation to Temperature and Moisture. In Ex Situ Plant Conservation; Guerrant, E.O., Havens, K., Maunder, M., Eds.; Island Press: Washington, DC, USA, 2004; pp. 139–161. [Google Scholar]
- Walters, C. Orthodoxy, recalcitrance and in-between: Describing variation in seed storage characteristics using threshold responses to water loss. Planta 2015, 242, 397–406. [Google Scholar] [CrossRef]
- Hamilton, K.N.; Ashmore, S.E.; Pritchard, H.W. Thermal analysis and cryopreservation of seeds of Australian wild citrus species (Rutaceae): Citrus australasica, C. inodora and C. garrawayi. CryoLetters 2009, 30, 268–279. [Google Scholar]
- Malik, S.K.; Chaudhury, R.; Pritchard, H.W. Long-Term, Large Scale Banking of Citrus Species Embryos: Comparisons Between Cryopreservation and other Seed Banking Temperatures. CryoLetters 2012, 33, 453–464. [Google Scholar]
- Eira, M.T.S.; Amaral da Silva, E.A.; De Castro, R.D.; Dussert, S.; Walters, C.; Bewley, J.D.; Hilhorst, H.W.M. Coffee seed physiology. Braz. J. Plant Physiol. 2006, 18, 149–163. [Google Scholar] [CrossRef]
- Crane, J.; Kovach, D.; Gardner, C.; Walters, C. Triacylglycerol phase and ‘intermediate’ seed physiology: A study of Cuphea carthagenesis. Planta 2006, 223, 1081–1089. [Google Scholar] [CrossRef]
- Sacande, M.; Golovina, E.A.; van Aelst, A.C.; Hoekstra, F.A. Viability loss of neem (Azadirachta indica) seeds associated with membrane phase behaviour. J. Exp. Bot. 2001, 52, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, H.W.; Seaton, P.T. Orchid seed storage. Historical perspective, current status and future prospects. Selbyana 1993, 14, 89–104. [Google Scholar]
- Ellis, R.H.; Hong, T.D.; Roberts, E.H. Effect of storage temperature and moisture on the germination of papaya seeds. Seed Sci. Res. 1991, 1, 69–72. [Google Scholar] [CrossRef]
- Chen, H.; Pritchard, H.W.; Seal, C.; Nadarajan, J.; Li, W.; Yang, S.; Kranner, I. Post desiccation germination of mature seeds of tea (Camellia sinensis L.) can be enhanced by pro-oxidant treatment, but partial desiccation tolerance does not ensure survival at −20 °C. Plant Sci. 2012, 184, 36–44. [Google Scholar] [CrossRef]
- Priestley, D.A. Seed aging. In Implications of Seed Storage and Persistence in the Soil; Comstock Publishing: Ithaca, NY, USA; London, UK, 1986; p. 304. [Google Scholar]
- Mira, S.; Estrelles, E.; Gonzalez-Benito, M.E.; Corbineau, F. Biochemical changes induced in seeds of Brassicaceae wild species during ageing. Acta Physiol. Plant. 2011, 33, 1803–1809. [Google Scholar] [CrossRef]
- Medeiros, A.C.S.; Probert, R.J.; Sader, R.; Smith, R.D. The moisture relations of seed longevity in Astronium urundeuva (Fr. All.) Engl. Seed Sci. Technol. 1998, 26, 289–298. [Google Scholar]
- Pritchard, H.W.; Dickie, J.B. Predicting seed longevity: The use and abuse of seed viability equations. In Seed Conservation: Turning Science into Practice; Smith, R.D., Dickie, J.B., Linington, S.H., Pritchard, H.W., Probert, R.J., Eds.; Royal Botanic Gardens, Kew: London, UK, 2003; pp. 653–721. [Google Scholar]
- Probert, R.J.; Daws, M.I.; Hay, F.R. Ecological correlates of ex situ seed longevity: A comparative study on 195 species. Ann. Bot. 2009, 104, 57–69. [Google Scholar] [CrossRef]
- Walters, C.; Wheeler, L.J.; Grotenhuis, J.M. Longevity of seeds stored in a genebank: Species characteristics. Seed Sci. Res. 2005, 15, 1–20. [Google Scholar] [CrossRef]
- McDonald, M.B. Seed deterioration: Physiology, repair and assessment. Seed Sci. Technol. 1999, 27, 177–237. [Google Scholar]
- Walters, C. Understanding the mechanisms and kinetics of seed aging. Seed Sci. Res. 1998, 8, 223–244. [Google Scholar] [CrossRef]
- Vertucci, C.W. A calorimetric study of the changes in lipids during seed storage under dry conditions. Plant Physiol. 1992, 99, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Walters, C. Temperature dependency of molecular mobility in preserved seeds. Biophys. J. 2004, 86, 1253–1258. [Google Scholar] [CrossRef]
- Crane, J.; Miller, A.L.; Van Roekel, J.W.; Walters, C. Triacylglycerols determine the unusual storage physiology of Cuphea seed. Planta 2003, 217, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Fielder, H.; Brotherton, P.; Hosking, J.; Hopkins, J.J.; Ford-Lloyd, B.; Maxted, N. Enhancing the Conservation of Crop Wild Relatives in England. PLoS ONE 2015, 10, e0130804. [Google Scholar] [CrossRef]
- Tanksley, S.D.; McCouch, S.R. Seed Banks and Molecular Maps: Unlocking Genetic Potential from the Wild. Science 1997, 277, 1063–1066. [Google Scholar] [CrossRef]
- Branca, F.; Lapichino, G. Some wild and cultivated Brassicaceae exploited in Sicily as vegetables. FAO/IPGRI Plant Genet. Resour. Newsl. 1997, 110, 22–28. [Google Scholar]
- Gray, A.R. Taxonomy and evolution of broccoli (Brassica oleracea var. italica). Econ. Bot. 1982, 36, 397–410. [Google Scholar] [CrossRef]
- Massie, I.H.; Astley, D.; King, G.J. Patterns of genetic diversity and relationships between regional groups and populations of Italian landrace cauliflower and broccoli. (Brassica oleracea L. var. botrytis L. and var. italica Plenck). Acta Hortic. 1996, 407, 45–53. [Google Scholar]
- Liu, K.; Eastwood, R.J.; Flynn, S.; Turner, R.M.; Stuppy, W.H. Seed Information Database (Release 7.1, May 2008). Available online: http://www.kew.org/data/sid (accessed on 8 October 2015).
- Mira, S.; Estrelles, E.; Gonzalez-Benito, M.E. Effect of water content and temperature on seed longevity of seven Brassicaceae species after 5 years of storage. Plant Biol. 2015, 17, 153–162. [Google Scholar] [CrossRef]
- Nagel, M.; Borner, A. The longevity of crop seeds stored under ambient conditions. Seed Sci. Res. 2010, 20, 1–12. [Google Scholar] [CrossRef]
- Gómez-Campo, C. Preservation of West Mediterranean members of the cruciferous tribe Brassiceae. Biol. Conserv. 1972, 4, 355–360. [Google Scholar] [CrossRef]
- Nilsson, P.; Johansson, S.A.; Merker, A. Variation in seed oil composition of species from the genera Barbarea and Lepidium. Acta Agric. Scand. B-Soil Plant Sci. 1998, 48, 159–164. [Google Scholar]
- Azimova, S.S.; Glushenkova, A.I.; Vinogradova, V.I. (Eds.) Lipids, Lipophilic Components and Essential Oils from Plant Sources; Springer: London, UK, 2011; Volume 1. [Google Scholar] [CrossRef]
- Vertucci, C.W. Effects of cooling rate on seeds exposed to liquid nitrogen temperatures. Plant Physiol. 1989, 90, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- Larsson, K. Alternation of melting points in homologous series of long-chain compounds. J. Am. Oil Chem. Soc. 1996, 43, 559–562. [Google Scholar] [CrossRef]
- Small, D.M. (Ed.) The Physical Chemistry of Lipids: From Alkanes to Phospholipids; Plenum Press: New York, NY, USA, 1986; p. 672. [Google Scholar]
- Rustan, A.C.; Drevon, C.A. (Eds.) Fatty Acids: Structures and Properties. Encyclopedia of Life Sciences; Nature Publishing: London, UK, 2005; Available online: http://www.els.net (accessed on 8 October 2015).
- Vertucci, C.W. Relationship between thermal transitions and freezing injury in pea and soybean seeds. Plant Physiol. 1989, 90, 1121–1128. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, D.; Hill, M.H.; Lynch, R.T.; Pritchard, H.W.; Walters, C. Longevity of Preserved Germplasm: The Temperature Dependency of Aging Reactions in Glassy Matrices of Dried Fern Spores. Plant Cell Physiol. 2019, 60, 376–392. [Google Scholar] [CrossRef] [PubMed]
- Himawan, C.; Starov, V.M.; Stapley, A.G.F. Thermodynamic and kinetic aspects of fat crystallization. Adv. Colloid Interface Sci. 2006, 122, 3–33. [Google Scholar] [CrossRef]
- Dickie, J.B.; Ellis, R.H.; Kraak, H.L.; Ryder, K.; Tompsett, P.B. Temperature and seed storage longevity. Ann. Bot. 1990, 65, 197–204. [Google Scholar] [CrossRef]
- Volk, G.M.; Crane, J.; Caspersen, A.M.; Hill, L.M.; Gardner, C.; Walters, C. Massive cellular disruption occurs during early imbibition of Cuphea seeds containing crystallized triacylglycerols. Planta 2006, 224, 1415–1426. [Google Scholar] [CrossRef]
- Dussert, S.; Chabrillange, N.; Rocquelin, G.; Engelmann, F.; Lopez, M.; Hamon, S. Tolerance of coffee (Coffea spp.) seeds to ultra-low temperature exposure in relation to calorimetric properties of tissue water, lipid composition and cooling procedure. Physiol. Plant. 2001, 112, 495–504. [Google Scholar] [CrossRef]
- Liu, X.S. Sample size for the Z test and its confidence interval. Int. J. Math. Educ. Sci. Technol. 2011, 43, 266–270. [Google Scholar]
- Nadarajan, J.; Marzalina, M.; Krishnapillay, B.; Staines, H.J.; Benson, E.E.; Harding, K. Applications of differential scanning calorimetry in developing cryopreservation strategies for Parkia speciosa, a tropical tree producing recalcitrant seeds. CryoLetters 2008, 29, 95–110. [Google Scholar] [PubMed]
- FAO. Genebank Standards for Plant Genetic Resources for Food and Agriculture; FAO: Rome, Italy, 2014; Available online: http://www.fao.org/3/a-i3704e.pdf (accessed on 8 October 2015).
| Years of Storage in UPM (or RBG Kew) Bank | Germination (%) | |||||
|---|---|---|---|---|---|---|
| Before Storage at UPM *1 | After Storage at UPM and Pre-DSC | After Storage at UPM and Post-DSC | Before Storage at RBG Kew | After Storage at RBG Kew | ||
| Alliaria petiolata Cavara & Grande. | NA (32) | NA | NA | NA | 92 S | 24 |
| Alyssoides utriculata Medik. | 43 (NA) | 100 | 100 § NS | 100 § NS | ||
| Alyssum saxatile L. | 43 (25) | 100 | 100 NS | 100 NS | 100 NS | 100 |
| Arabis turrita L. | 43 (NA) | 0 | 4 ‡ NS | 24 ‡ S | ||
| Barbarea intermedia Boreau. | 43 (NA) | 95 | 100 NS | 93 NS | ||
| Brassica napus L. | 43 (33) | 100 | 100 NS | 100 NS | 100 NS | 100 |
| Coincya rupestris L. | 43 (NA) | 92 | 86 NS | 100 S | ||
| Conringia orientalis (L.) C.Presl. | 44 (NA) | 0 | 100 ‡ S | 100 ‡ NS | ||
| Descurainia sophia L. | 44 (23) | 2 | 100 ‡ S | 96 ‡ NS | 100 S | 90 |
| Erucastrum abyssinicum O.E.Schulz. | 44 (NA) | 100 | 90 S | 98 NS | ||
| Erysimum cheiri Crantz. | 43 (34) | 100 | 100 NS | 100 NS | 100 NS | 100 |
| Erysimum odoratum Baumg. | 43 (NA) | 100 | 100 NS | 100 NS | ||
| Erysimum repandum L. | 43 (NA) | 100 | 100 NS | 100 NS | ||
| Lesquerella gordonii Watson. | NA (8) | NA | NA | NA | 96 S | 0 |
| Matthiola incana (L.) W.T.Aiton. | 43 (35) | 95 | 100 NS | 100 NS | 100 NS | 100 |
| Matthiola sinuata (L.) W.T.Aiton. | 43 (22) | 100 | 100 § NS | 40 § S | 100 NS | 100 |
| Moricandia arvensis (L.) DC. | 44 (15) | 20 | 96 † S | 96 † NS | 100 NS | 94 |
| Rorippa palustris (L.) Besser | NA (11) | NA | NA | NA | 100 NS | 100 |
| Sinapis arvensis Sinapis arvensis L. | 43 (12) | 3 | 73 † S | 85 † S | 100 S | 90 |
| Sisymbrium orientale L. | 43 (24) | 3 | 100 ‡ S | 99 ‡ NS | 100 S | 81 |
| Species | Lipid Content (%) | Melt Onset T (°C) | Peak Mid-point (°C) | Melt End T (°C) | Melt Enthalpy (ΔH) (mJ g−1DW) | 16:0 (Pal-mitic Acid) | 18:0 (Stearic Acid) | 18:1 (Oleic Acid) | 18:2 (Linoleic Acid) | 18:3 (Lino-lenic Acid) | 20:1 (Gondoic Acid) | 21:0 (HeneiCosylic Acid) | 22:1 (Erucic Acid) | 24:1 (Nervonic Acid) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alliaria petiolata | 28.0 (± 2.6) 5,7 | −2.1 (± 0.7) | 12.7 (± 0.1) | 17.5 (± 0.8) | 18.41 (± 0.2) | 3.5 | 7.0 | 9.0 | 24.8 | 7.2 | 5.3 | - | 44.2 | 6.1 |
| Alyssoides utriculata | 12.7 (± 0.5) 2,7 | −49.8 (± 0.0) | −42.7 (± 0.6) | −36.4 (± 0.8) | 2.33 (± 0.6) | 7 | 3 | 17 | 11 | 71.1 | 0.6 | - | 2.7 | 0.4 |
| Alyssum saxatile | 18.2 (± 4.7) 2,7 | −50.9 (± 0.2) | −44.9 (± 0.1) | −35.3 (± 0.8) | 5.98 (± 0.1) | 5 | 1 | 12 | 20 | 58 | 0 | - | 0 | 0 |
| Arabis turrita | NA | −50.3 (± 0.1) | −45.3 (± 0.0) | −40.3 (± 0.2) | 3.22 (± 0.8) | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Barbarea intermedia | 30.0 3 | −14.4 (± 0.2) | −5.1 (± 0.3) | −2.3 (± 0.2) | 19.37 (± 0.3) | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Brassica napus | 40.5 (± 2.3) 4,7 | −10.1 (± 0.4) [−12.9] * (± 0.2) | −2.1 (± 0.1) [−4.2] * (± 0.1) | 1.7 (0) [−1.7] * (± 0.1) | 30.92 (± 0.8) [31.12] * (± 0.4) | 3 | 0.9 | 14 | 15 | 8 | 10 | - | 55 | 0 |
| Coincya rupestris | 17.45 7 | −41.1 (± 0.8) | −33.2 (± 0.3) | −21.7 (± 0.0) | 7.63 (± 0.2) | 3 | 1.5 | 13.5 | 16.3 | 25.7 | 6.1 | 0.5 | 30.5 | 3.5 |
| Conringia orientalis | 26.5 (± 1.1) 4,7 | −15.0 (± 0.0) | −8.3 (± 0.2) | −5.9 (± 0.1) | 20.92 (± 0.2) | 2.9 | 0.3 | 7 | 33 | 3.5 | 24.2 | - | 25.4 | 3.5 |
| Descurainia sophia | 37.1 (± 2.3) 4,7 | −49.6 (± 1.2) | −36.7 (± 0.4) | −27.3 (± 0.4) | 7.79 (± 0.8) | 6 | 2 | 14 | 17 | 37 | 12.5 | - | 9 | 0 |
| Erucastrum abyssinicum | 34.0 5 | −12.4 (± 0.0) | −4.7 (± 0.1) | −2.1 (± 0.1) | 21.04 (± 1.2) | 3 | 1 | 10 | 18 | 13 | 8 | - | 40 | 2 |
| Erysimum cheiri | 29.5 4 | −42.7 (± 0.5) | −34.1 (± 0.2) | −27.4 (± 1.2) | 5.97 (± 0.8) | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Erysimum odoratum | 30.5 (± 0.8) 2,7 | −45.6 (± 0.1) | −33.5 (± 0.2) | −24.6 (± 0.4) | 7.86 (± 0.3) | 3.1 | 0.8 | 3.6 | 18 | 30.2 | 5.4 | - | 28.6 | 3.6 |
| Erysimum repandum | 26.7 (± 6.3) 2,7 | −46.9 (± 0.8) | −35.8 (± 0.5) | −23.4 (± 2.4) | 6.45 (± 0.4) | 5.9 | 1.7 | 9.3 | 17 | 33.0 | 2 | - | 17 | - |
| Lesquerella gordonii | 27.3 (± 0.1) 5,7 | −5.6 (± 0.2) | 12.2 (± 0.3) | 17.4 (± 0.1) | 5.85 (± 0.1) | 2 | 2 | 22 | 4 | 7 | 1.0 | 57.9 | - | - |
| Matthiola incana | 20.8 2 | −44.3 (± 0.5) | −38.0 (± 1.0) | −31.7 (± 1.4) | 2.31 (± 0.2) | 8 | 2.5 | 13.6 | 9 | 62 | 0.5 | - | 0.8 | 0 |
| Matthiola sinuata | 29.0 5 | −47.0 (± 0.3) | −41.2 (± 0.1) | −34.2 (± 0.4) | 9.46 (± 0.2) | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Moricandia arvensis | 32.4 (± 0.9) 6,7 | −40.7 (± 0.7) | -32.8 (± 0.3) | −27.5 (± 0.4) | 3.95 (± 0.2) | 10.4 | 1.7 | 11.5 | 19.6 | 32.6 | 5.3 | - | 20 | 0.5 |
| Rorippa palustris | 21.0 4 | −48.3 (± 1.2) | −38.6 (± 0.8) | −20.7 (± 0.7) | 5.63 (± 0.4) | NA | NA | NA | NA | NA | NA | NA | NA | NA |
| Sinapis arvensis | 28.4 (± 1.1) 4,7 | −15.5 (± 1.0) | −6.3 (± 0.7) | −2.8 (± 0.4) | 20.00 (± 0.8) | 3.9 | 0.9 | 19.9 | 16 | 19.3 | 10.8 | - | 34.2 | 1.2 |
| Sisymbrium orientale | 22.1 (± 0.1) 2,7 | −42.9 (± 0.2) | −34.8 (± 0.3) | −25.1 (± 0.4) | 3.61 (± 0.2) | 9.4 | 0.9 | 6.4 | 13.3 | 41.4 | 3.6 | 17.3 | - | 0.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mira, S.; Nadarajan, J.; Liu, U.; González-Benito, M.E.; Pritchard, H.W. Lipid Thermal Fingerprints of Long-term Stored Seeds of Brassicaceae. Plants 2019, 8, 414. https://doi.org/10.3390/plants8100414
Mira S, Nadarajan J, Liu U, González-Benito ME, Pritchard HW. Lipid Thermal Fingerprints of Long-term Stored Seeds of Brassicaceae. Plants. 2019; 8(10):414. https://doi.org/10.3390/plants8100414
Chicago/Turabian StyleMira, Sara, Jayanthi Nadarajan, Udayangani Liu, Maria Elena González-Benito, and Hugh W. Pritchard. 2019. "Lipid Thermal Fingerprints of Long-term Stored Seeds of Brassicaceae" Plants 8, no. 10: 414. https://doi.org/10.3390/plants8100414
APA StyleMira, S., Nadarajan, J., Liu, U., González-Benito, M. E., & Pritchard, H. W. (2019). Lipid Thermal Fingerprints of Long-term Stored Seeds of Brassicaceae. Plants, 8(10), 414. https://doi.org/10.3390/plants8100414







