A Two-Stage Culture Method for Zygotic Embryos Effectively Overcomes Constraints Imposed by Hypocotyl and Epicotyl Seed Dormancy in Paeonia ostii ‘Fengdan’
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Sterilization
2.2. Hypocotyl and Epicotyl Dormancy Breaking as Affected by GA3, ABA, and Endosperm
- (1)
- The percentage of hypocotyl dormancy breaking = number of embryos that release hypocotyl dormancy/the whole number of embryos
- (2)
- The percentage of epicotyl dormancy breaking = number of embryos that release epicotyl dormancy/the whole number of embryos
2.3. Hypocotyl and Epicotyl Dormancy Breaking as Affected by GA3 and 6-Benzylaminopurine (BA)
2.4. Hypocotyl and Epicotyl Dormancy Breaking as Affected by BA and GA3 in a Two-Stage Culture Method
2.5. Hypocotyl and Epicotyl Dormancy Breaking as Affected by the Light and Its Quality
2.6. Statistical Analysis
3. Results
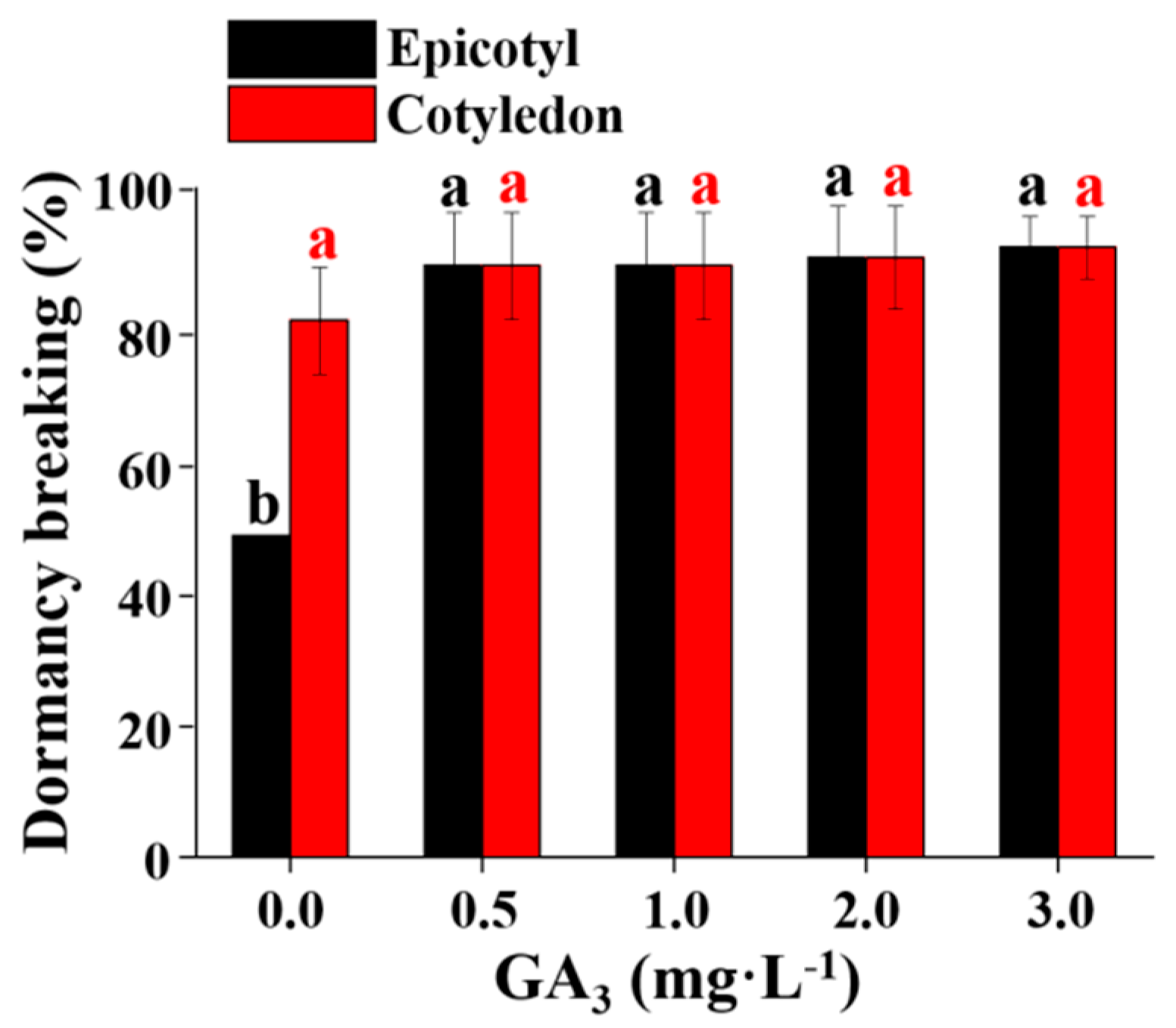
3.1. The Effect of GA3, ABA, and Endosperm on Hypocotyl Dormancy Breaking
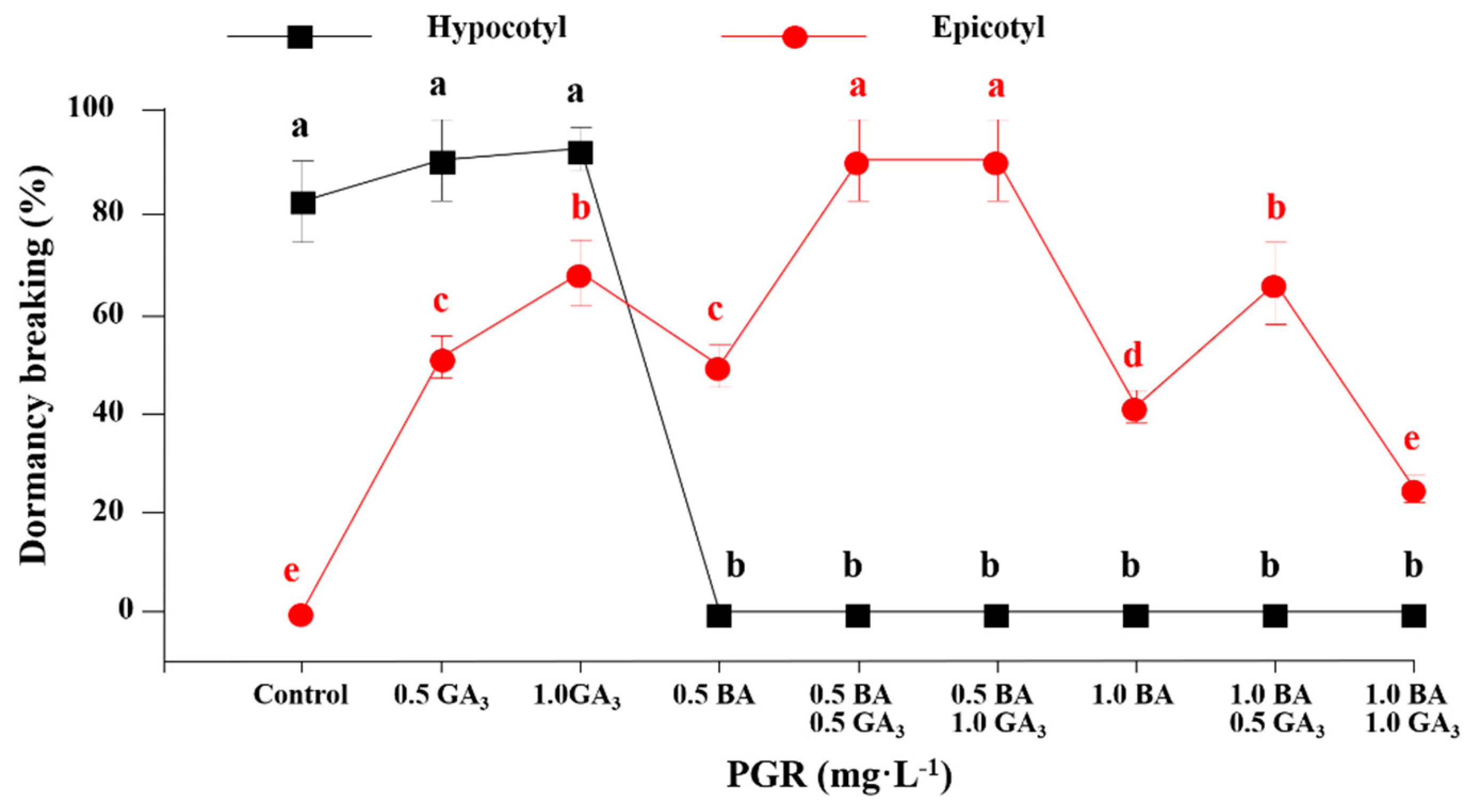
3.2. The Effect of GA3 and BA Concentration on Hypocotyl and Epicotyl Dormancy Breaking
3.3. Hypocotyl and Epicotyl Dormancy Breaking Affected by GA3 and BA in a Two-stage Culture Method
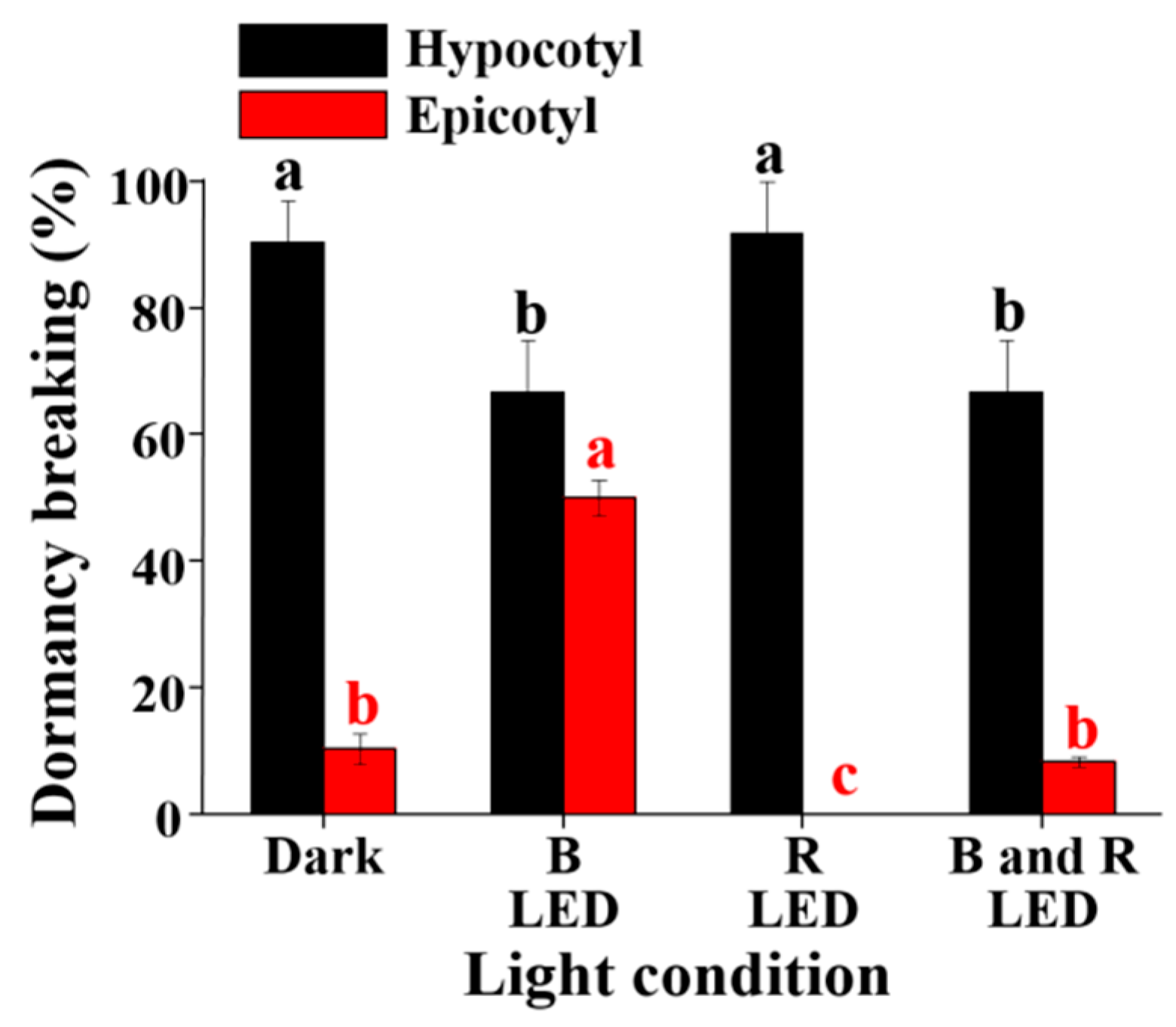
3.4. The Effect of the Light and Its Quality on Breaking Hypocotyl and Epicotyl Dormancy
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, H.F.; Li, X.F.; Wu, K.; Wang, M.K.; Liu, P.; Wang, X.S.; Deng, R.X. Antioxidant activities and chemical constituents of flavonoids from the flower of Paeonia ostii. Molecules 2017, 22, 5. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Dong, C.L.; Xue, Z.Y.; Jin, Q.J.; Xu, Y.C. De novo transcriptome sequencing and discovery of genes related to copper tolerance in Paeonia ostii. Gene 2016, 576, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Cheng, F.; Xiao, J.; Wang, Y.; Zhang, D.; Wang, Y.; Zhong, Y. Crosses of Paeonia ostii ‘Feng Dan Bai’ as maternal parents and an analysis on the potential in tree peony breeding. J. Beijing For. Univ. 2014, 36, 121–125. [Google Scholar]
- Xie, L.H.; Niu, L.X.; Zhang, Y.L.; Jin, M.; Ji, D.; Zhang, X.X. Pollen sources influence the traits of seed and seed oil in Paeonia ostii ‘Feng Dan’. HortScience 2017, 52, 700–705. [Google Scholar] [CrossRef]
- Cui, H.L.; Cheng, F.Y.; Peng, L.P. Determination of the fatty acid composition in tree peony seeds using near-infrared spectroscopy. J. Am. Oil Chem. Soc. 2016, 93, 943–952. [Google Scholar]
- Peng, L.P.; Cai, C.F.; Zhong, Y.; Xu, X.X.; Xian, H.L.; Cheng, F.Y.; Mao, J.F. Genetic analyses reveal independent domestication origins of the emerging oil crop Paeonia ostii, a tree peony with a long-term cultivation history. Sci. Rep. 2017, 7, 5340. [Google Scholar] [CrossRef]
- Zhang, K.; Yao, L.; Zhang, Y.; Baskin, J.M.; Baskin, C.C.; Xiong, Z.; Tao, J. A review of the seed biology of Paeonia species (Paeoniaceae), with particular reference to dormancy and germination. Planta 2019, 249, 291–303. [Google Scholar] [CrossRef]
- Ren, X.X.; Xue, J.Q.; Wang, S.L.; Xue, Y.Q.; Zhang, P.; Jiang, H.D.; Zhang, X.X. Proteomic analysis of tree peony (Paeonia ostii ‘Feng Dan’) seed germination affected by low temperature. J. Plant Physiol. 2018, 224–225, 56–67. [Google Scholar] [CrossRef]
- Porceddu, M.; Mattana, E.; Pritchard, H.W.; Bacchetta, G. Sequential temperature control of multi-phasic dormancy release and germination of Paeonia corsica seeds. J. Plant Ecol. 2015, 9, 464–473. [Google Scholar] [CrossRef]
- Yu, X.; Zhao, R.; Cheng, F. Seed germination of tree and herbaceous peonies: A mini-review. Seed Sci. Biotechnol. 2014, 1, 11–14. [Google Scholar]
- Xue, J.Q.; Wang, S.L.; Zhang, P.; Zhu, F.Y.; Ren, X.X.; Liu, C.J.; Zhang, X.X. On the role of physiological substances, abscisic acid and its biosynthetic genes in seed maturation and dormancy of tree peony (Paeonia ostii ‘Feng Dan’). Sci. Hortic. 2015, 182, 92–101. [Google Scholar] [CrossRef]
- Jing, X.; Zheng, G. The characteristics in seed germination and dormancy of four wild species of tree peonies and their bearing on endangerment. Acta Phytophysiol. Sin. 1999, 25, 214–221. [Google Scholar]
- Cheng, F.Y.; Du, X.J. Effects of chilling and gibberellic acid on the seed germination and seedling growth in Paeonia ostii ‘Feng Dan’. Acta Hortic. Sin. 2008, 35, 553. [Google Scholar]
- Bewley, J.D. Seed germination and dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Finch-Savage, W.E.; Leubner-Metzger, G. Seed dormancy and the control of germination. New Phytol. 2006, 171, 501–523. [Google Scholar] [CrossRef]
- Vleeshouwers, L.; Bouwmeester, H.; Karssen, C. Redefining seed dormancy: An attempt to integrate physiology and ecology. J. Ecol. 1995, 83, 1031–1037. [Google Scholar] [CrossRef]
- Finch-Savage, W.E.; Cadman, C.S.C.; Toorop, P.E.; Lynn, J.R.; Hilhorst, H.W.M. Seed dormancy release in Arabidopsis Cvi by dry after-ripening, low temperature, nitrate and light shows common quantitative patterns of gene expression directed by environmentally specific sensing. Plant J. 2007, 51, 60–78. [Google Scholar] [CrossRef]
- Bentsink, L.; Koornneef, M. Seed dormancy and germination. Arabidopsis Book 2008, 6, e0103. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. A classification system for seed dormancy. Seed Sci. Res. 2004, 14, 1–16. [Google Scholar] [CrossRef]
- Koornneef, M.; Bentsink, L.; Hilhorst, H. Seed dormancy and germination. Curr. Opin. Plant Biol. 2002, 5, 33–36. [Google Scholar] [CrossRef]
- Kucera, B.; Cohn, M.A.; Leubner-Metzger, G. Plant hormone interactions during seed dormancy release and germination. Seed Sci. Res. 2005, 15, 281–307. [Google Scholar] [CrossRef]
- Seo, M.; Nambara, E.; Choi, G.; Yamaguchi, S. Interaction of light and hormone signals in germinating seeds. Plant Mol. Biol. 2008, 69, 463. [Google Scholar] [CrossRef]
- Benech-Arnold, R.L.; Sánchez, R.A.; Forcella, F.; Kruk, B.C.; Ghersa, C.M. Environmental control of dormancy in weed seed banks in soil. Field Crops Res. 2000, 67, 105–122. [Google Scholar] [CrossRef]
- Raghavan, V. One hundred years of zygotic embryo culture investigations. In Vitro Cell Dev. Biol. Plant 2003, 39, 437–442. [Google Scholar] [CrossRef]
- Buchheim, J.A.T.; Burkhart, L.F.; Meyer, M.M. Effect of exogenous gibberellic acid, abscisic acid, and benzylaminopurine on epicotyl dormancy of cultured herbaceous peony embryos. Plant Cell Tissue Organ Cult. 1994, 36, 35–43. [Google Scholar] [CrossRef]
- Yang, H.; Pei, D. Study on embryo culture of peony (Paeonia L.) seed. Guangxi Agric. Sci. 2006, 37, 108–110. [Google Scholar]
- Wang, H.; van Staden, J. Establishment of in vitro cultures of tree peonies. S. Afr. J. Bot. 2001, 67, 358–361. [Google Scholar] [CrossRef][Green Version]
- Wang, Y.; He, G.M.; Han, L.X. Study on embryo-culture and seedling growth for Paeonia rockii. Hunan Agric. Sci. 2012, 9, 103–106. [Google Scholar]
- Villiers, T.A.; Wareing, P.F. The possible role of low temperature in breaking the dormancy of seeds of Fraxinus excelsior L. J. Exp. Bot. 1965, 16, 519–531. [Google Scholar] [CrossRef]
- Nyachiro, J.M.; Clarke, F.R.; DePauw, R.M.; Knox, R.E.; Armstrong, K.C. Temperature effects on seed germination and expression of seed dormancy in wheat. Euphytica 2002, 126, 123–127. [Google Scholar] [CrossRef]
- Penfield, S.; Josse, E.-M.; Kannangara, R.; Gilday, A.D.; Halliday, K.J.; Graham, I.A. Cold and light control seed germination through the bHLH transcription factor SPATULA. Curr. Biol. 2005, 15, 1998–2006. [Google Scholar] [CrossRef] [PubMed]
- Derkx, M.P.M.; Karssen, C.M. Effects of light and temperature on seed dormancy and gibberellin-stimulated germination in Arabidopsis thaliana: Studies with gibberellin-deficient and -insensitive mutants. Physiol. Plant 1993, 89, 360–368. [Google Scholar] [CrossRef]
- Taylorson, R.B. Phytochrome controlled changes in dormancy and germination of buried weed seeds. Weed Sci. 2017, 20, 417–422. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. Seed germination ecophysiology of the woodland herb Asarum canadense. Am. Midl. Nat. 1986, 116, 132–139. [Google Scholar] [CrossRef]
- Baskin, C.C. Breaking physical dormancy in seeds–focussing on the lens. New Phytol. 2003, 158, 229–232. [Google Scholar] [CrossRef]
- Hao, H.P.; He, Z.; Li, H.; Shi, L.; Tang, Y.D. Effect of root length on epicotyl dormancy release in seeds of Paeonia ludlowii, Tibetan peony. Ann. Bot. 2013, 113, 443–452. [Google Scholar] [CrossRef]
- Riefler, M.; Novak, O.; Strnad, M.; Schmülling, T. Arabidopsis cytokinin receptor mutants reveal functions in shoot growth, leaf senescence, seed size, germination, root development, and cytokinin metabolism. Plant Cell 2006, 18, 40–54. [Google Scholar] [CrossRef]
- Chory, J.; Reinecke, D.; Sim, S.; Washburn, T.; Brenner, M. A role for cytokinins in de-etiolation in Arabidopsis (det mutants have an altered response to cytokinins). Plant Physiol. 1994, 104, 339–347. [Google Scholar] [CrossRef]
- Müller, K.; Tintelnot, S.; Leubner-Metzger, G. Endosperm-limited Brassicaceae seed germination: Abscisic acid inhibits embryo-induced endosperm weakening of Lepidium sativum (cress) and endosperm rupture of cress and Arabidopsis thaliana. Plant Cell Physiol. 2006, 47, 864–877. [Google Scholar] [CrossRef]
- Ogawa, M.; Hanada, A.; Yamauchi, Y.; Kuwahara, A.; Kamiya, Y.; Yamaguchi, S. Gibberellin biosynthesis and response during Arabidopsis seed germination. Plant Cell 2003, 15, 1591–1604. [Google Scholar] [CrossRef]
- Richards, D.E.; King, K.E.; Ait-Ali, T.; Harberd, N.P. How gibberellin regulates plant growth and development: A molecular genetic analysis of gibberellin signaling. Annu. Rev. Plant Biol. 2001, 52, 67–88. [Google Scholar] [CrossRef] [PubMed]
- Groot, S.P.C.; Karssen, C.M. Gibberellins regulate seed germination in tomato by endosperm weakening: A study with gibberellin-deficient mutants. Planta 1987, 171, 525–531. [Google Scholar] [CrossRef]
- Leubner-Metzger, G. Brassinosteroids and gibberellins promote tobacco seed germination by distinct pathways. Planta 2001, 213, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S.; Kamiya, Y. Gibberellins and light-stimulated seed germination. J. Plant Growth Regul. 2001, 20, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Amen, R.D. A model of seed dormancy. Bot. Rev. 1968, 34, 1–31. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Ogawa, M.; Kuwahara, A.; Hanada, A.; Kamiya, Y.; Yamaguchi, S. Activation of gibberellin biosynthesis and response pathways by low temperature during imbibition of Arabidopsis thaliana seeds. Plant Cell 2004, 16, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Hennig, L.; Stoddart, W.M.; Dieterle, M.; Whitelam, G.C.; Schäfer, E. Phytochrome E controls light-induced germination of Arabidopsis. Plant Physiol. 2002, 128, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Barrero, J.M.; Jacobsen, J.V.; Talbot, M.J.; White, R.G.; Swain, S.M.; Garvin, D.F.; Gubler, F. Grain dormancy and light quality effects on germination in the model grass Brachypodium distachyon. New Phytol. 2012, 193, 376–386. [Google Scholar] [CrossRef]
- Goggin, D.E.; Powles, S.B.; Toorop, P.E.; Steadman, K.J. Dark-mediated dormancy release in stratified Lolium rigidum seeds is associated with higher activities of cell wall-modifying enzymes and an apparent increase in gibberellin sensitivity. J. Plant Physiol. 2011, 168, 527–533. [Google Scholar] [CrossRef]
- Long, R.L.; Stevens, J.C.; Griffiths, E.M.; Adamek, M.; Powles, S.B.; Merritt, D.J. Detecting karrikinolide responses in seeds of the Poaceae. Aust. J. Bot. 2011, 59, 610–620. [Google Scholar] [CrossRef]
- Zhao, X.; Yu, X.; Foo, E.; Symons, G.M.; Lopez, J.; Bendehakkalu, K.T.; Xiang, J.; Weller, J.L.; Liu, X.; Reid, J.B.; et al. A study of gibberellin homeostasis and cryptochrome-mediated blue light inhibition of hypocotyl elongation. Plant Physiol. 2007, 145, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.V.; Barrero, J.M.; Hughes, T.; Julkowska, M.; Taylor, J.M.; Xu, Q.; Gubler, F. Roles for blue light, jasmonate and nitric oxide in the regulation of dormancy and germination in wheat grain (Triticum aestivum L.). Planta 2013, 238, 121–138. [Google Scholar] [CrossRef] [PubMed]
- Gubler, F.; Hughes, T.; Waterhouse, P.; Jacobsen, J. Regulation of dormancy in barley by blue light and after-ripening: Effects on abscisic acid and gibberellin metabolism. Plant Physiol. 2008, 147, 886–896. [Google Scholar] [CrossRef] [PubMed]
- Goggin, D.E.; Steadman, K.J.; Powles, S.B. Green and blue light photoreceptors are involved in maintenance of dormancy in imbibed annual ryegrass (Lolium rigidum) seeds. New Phytol. 2008, 180, 81–89. [Google Scholar] [CrossRef] [PubMed]

| First Culture Stage | Second Culture Stage | Abbreviation | ||||
|---|---|---|---|---|---|---|
| PGR (mg·L−1) | Temperature (°C) | PGR (mg·L−1) | Temperature (°C) | |||
| GA3 | BA | GA3 | BA | |||
| 0.0 | 0.0 | 25 | 0.0 | 0.0 | 25 | Control (no PGR, 25 °C) |
| 0.0 | 0.0 | 25 | 1.0 | 0.0 | 25 | Control → 1.0 GA3 (25 °C) |
| 0.0 | 0.0 | 25 | 0.0 | 0.5 | 25 | Control → 0.5 BA (25 °C) |
| 0.0 | 0.0 | 25 | 1.0 | 0.5 | 25 | Control → 1.0 GA3 and 0.5 BA (25 °C) |
| 1.0 | 0.0 | 25 | 1.0 | 0.0 | 25 | 1.0 GA3 (25 °C) |
| 1.0 | 0.5 | 25 | 1.0 | 0.5 | 25 | 1.0 GA3 and 0.5 BA (25 °C) |
| Explant | Temperature (°C) | Medium | PGR (mg·L−1) | Hypocotyl Dormancy Breaking (%) | |
|---|---|---|---|---|---|
| GA3 | ABA | ||||
| Embryo | 25 | MS | 0.0 | 0.0 | 80.0 ± 2.1 a z |
| Embryo | 25 | MS | 1.0 | 0.0 | 81.7 ± 3.2 a |
| Embryo | 25 | MS | 0.0 | 1.0 | 0.0 ± 0.0 b |
| Embryo | 25 | MS | 1.0 | 1.0 | 0.0 ± 0.0 b |
| Embryo and endosperm | 25 | MS | 1.0 | 0.0 | 0.0 ± 0.0 b |
| - | - | - | - | - | *** |
| Treatments (mg·L−1) | Dormancy Breaking (%) | |
|---|---|---|
| Hypocotyl | Epicotyl | |
| Control (no PGR, 25 °C) | 80.0 ± 2.1 a z | 10.3 ± 0.8 d |
| Control → 1.0 GA3 (25 °C) | 80.0 ± 2.1 a | 30.4 ± 2.2 b |
| Control → 0.5 BA (25 °C) | 80.0 ± 2.1 a | 30.1 ± 1.8 b |
| Control → 1.0 GA3 and 0.5 BA (25 °C) | 80.0 ± 2.1 a | 92.5 ± 3.8 a |
| 1.0 GA3 (25 °C) | 80.0 ± 3.2 a | 30.0 ± 2.0 b |
| 1.0 GA3 and 0.5 BA (25 °C) | 0.0 ± 0.0 b | 91.7 ± 1.9 a |
| F-test | *** | *** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, X.; Liu, Y.; Jeong, B.R. A Two-Stage Culture Method for Zygotic Embryos Effectively Overcomes Constraints Imposed by Hypocotyl and Epicotyl Seed Dormancy in Paeonia ostii ‘Fengdan’. Plants 2019, 8, 356. https://doi.org/10.3390/plants8100356
Ren X, Liu Y, Jeong BR. A Two-Stage Culture Method for Zygotic Embryos Effectively Overcomes Constraints Imposed by Hypocotyl and Epicotyl Seed Dormancy in Paeonia ostii ‘Fengdan’. Plants. 2019; 8(10):356. https://doi.org/10.3390/plants8100356
Chicago/Turabian StyleRen, Xiuxia, Ya Liu, and Byoung Ryong Jeong. 2019. "A Two-Stage Culture Method for Zygotic Embryos Effectively Overcomes Constraints Imposed by Hypocotyl and Epicotyl Seed Dormancy in Paeonia ostii ‘Fengdan’" Plants 8, no. 10: 356. https://doi.org/10.3390/plants8100356
APA StyleRen, X., Liu, Y., & Jeong, B. R. (2019). A Two-Stage Culture Method for Zygotic Embryos Effectively Overcomes Constraints Imposed by Hypocotyl and Epicotyl Seed Dormancy in Paeonia ostii ‘Fengdan’. Plants, 8(10), 356. https://doi.org/10.3390/plants8100356






