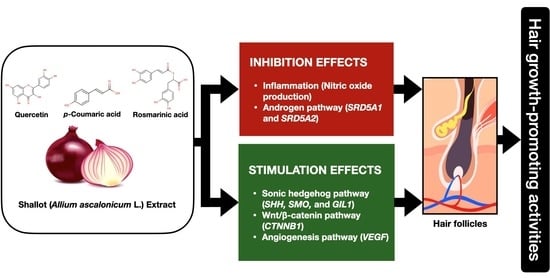
Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract
Abstract
:1. Introduction
2. Results
2.1. Extraction Yield and Bioactive Compound Estimation
2.2. Cell Viability
2.3. Anti-Inflammatory Activity
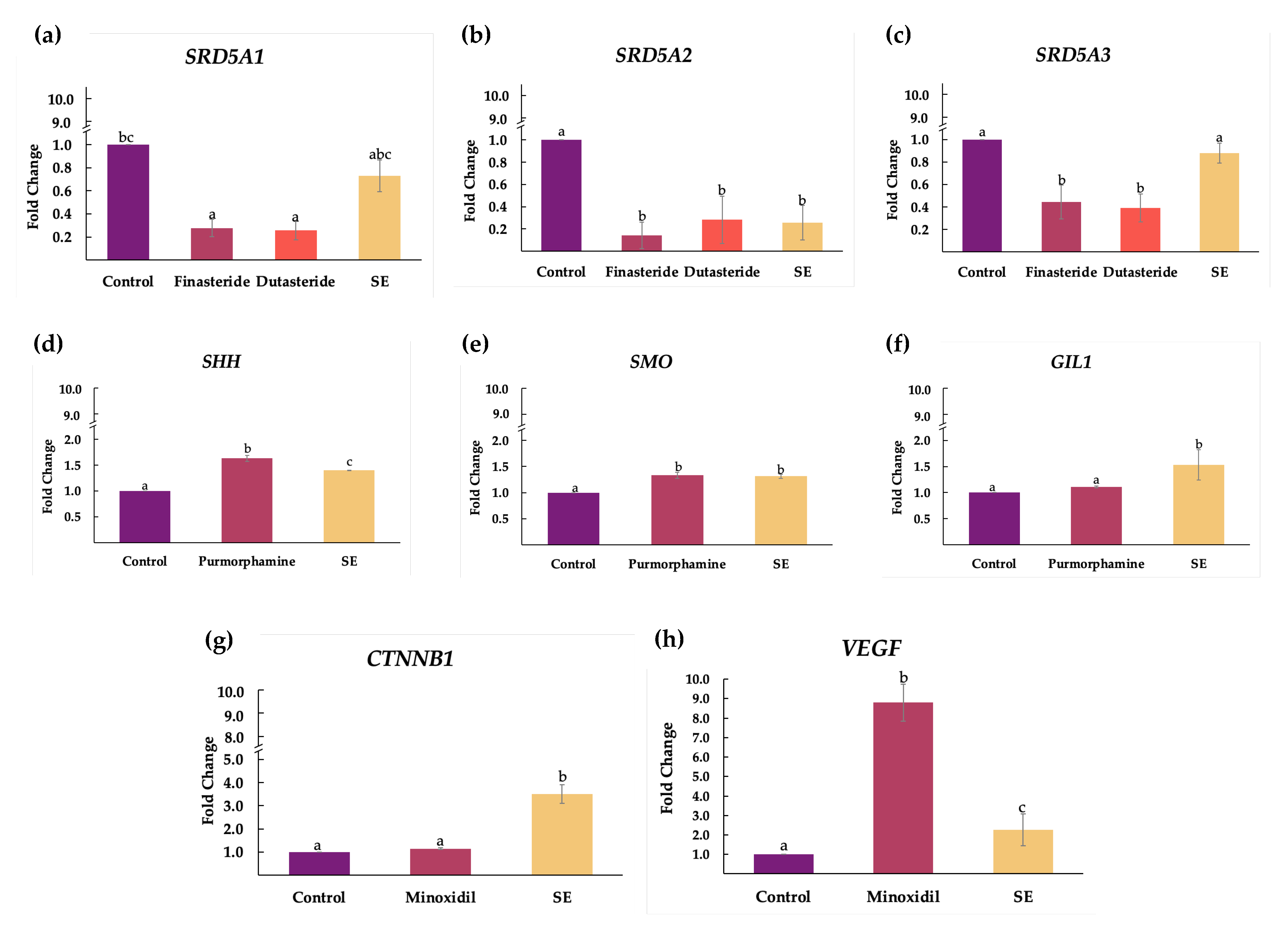
2.4. Effect of Shallot Extract on Expressions of Genes Associated with Androgenetic Alopecia
3. Discussion
4. Materials and Methods
4.1. Preparation of Extract
4.2. Phytochemical Estimations
4.2.1. Total Phenolic Content
4.2.2. Total Flavonoid Content
4.2.3. Total Polysaccharide Content
4.2.4. Total Protein Content
4.3. Determination of Phenolic Compounds by Liquid Chromatography–Mass Spectrometry (LC-MS)
4.4. Cell Viability Assay
4.5. Anti-Inflammatory Activity
4.6. Semi-Quantitative Reverse Transcription and Polymerase Chain Reaction
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sun, W.; Shahrajabian, M.H.; Cheng, Q. The insight and survey on medicinal properties and nutritive components of shallot. J. Med. Plant Res. 2019, 13, 452–457. [Google Scholar]
- Leelarungrayub, N.; Chanarat, N.; Rattanapanone, V. Potential activity of Thai shallot (Allium ascalonicum L.) extract on the prevention of hemolysis and glutathione depletion in human erythrocyte from oxidative stress. Chiang Mai J. Sci. 2004, 3, 225–234. [Google Scholar]
- Raeisi, S.; Sharifi-Rad, M.; Quek, S.Y.; Shabanpour, B.; Sharifi-Rad, J. Evaluation of antioxidant and antimicrobial effects of shallot (Allium ascalonicum L.) fruit and ajwain (Trachyspermum ammi (L.) Sprague) seed extracts in semi-fried coated rainbow trout (Oncorhynchus mykiss) fillets for shelf-life extension. LWT-Food Sci. Technol. 2016, 65, 112–121. [Google Scholar] [CrossRef]
- Owoyele, B.; Abioye, A.; Afinowi, N.; Jimoh, S.; Soladoye, A. Analgesic and anti-inflammatory effects of Allium ascalonicum. Trop. J. Health Sci. 2006, 13, 28–32. [Google Scholar] [CrossRef]
- Fattorusso, E.; Iorizzi, M.; Lanzotti, V.; Taglialatela-Scafati, O. Chemical composition of shallot (Allium ascalonicum Hort.). J. Agric. Food Chem. 2002, 50, 5686–5690. [Google Scholar] [CrossRef] [PubMed]
- Leelarungrayub, N.; Rattanapanone, V.; Chanarat, N.; Gebicki, J.M. Quantitative evaluation of the antioxidant properties of garlic and shallot preparations. Nutrition 2006, 22, 266–274. [Google Scholar] [CrossRef]
- Katzer, T.; Leite Junior, A.; Beck, R.; da Silva, C. Physiopathology and current treatments of androgenetic alopecia: Going beyond androgens and anti-androgens. Dermatol. Ther. 2019, 32, e13059. [Google Scholar] [CrossRef] [PubMed]
- Ceruti, J.M.; Leirós, G.J.; Balañá, M.E. Androgens and androgen receptor action in skin and hair follicles. Mol. Cell. Endocrinol. 2018, 465, 122–133. [Google Scholar] [CrossRef] [Green Version]
- Anastassakis, K. Hormonal and genetic etiology of male androgenetic alopecia. In Androgenetic Alopecia From A to Z: Vol.1 Basic Science, Diagnosis, Etiology, and Related Disorders; Anastassakis, K., Ed.; Springer International Publishing: Cham, Switzerland, 2022; pp. 135–180. [Google Scholar] [CrossRef]
- Wolf, R.; Schönfelder, G.; Paul, M.; Blume-Peytavi, U. Nitric oxide in the human hair follicle: Constitutive and dihydrotestosterone-induced nitric oxide synthase expression and NO production in dermal papilla cells. J. Mol. Med. 2003, 81, 110–117. [Google Scholar] [CrossRef]
- Zgonc Škulj, A.; Poljšak, N.; Kočevar Glavač, N.; Kreft, S. Herbal preparations for the treatment of hair loss. Arch. Dermatol. 2020, 312, 395–406. [Google Scholar] [CrossRef]
- Dhariwala, M.Y.; Ravikumar, P. An overview of herbal alternatives in androgenetic alopecia. J. Cosmet. Dermatol. 2019, 18, 966–975. [Google Scholar] [CrossRef] [PubMed]
- Herman, A.; Herman, A.P. Mechanism of action of herbs and their active constituents used in hair loss treatment. Fitoterapia 2016, 114, 18–25. [Google Scholar] [CrossRef]
- Manosroi, A.; Ruksiriwanich, W.; Manosroi, W.; Abe, M.; Manosroi, J. In vivo hair growth promotion activity of gel containing niosomes loaded with the Oryza sativa bran fraction (OSF3). Adv. Sci. Lett. 2012, 16, 222–228. [Google Scholar] [CrossRef]
- Choi, B.Y. Targeting Wnt/β-catenin pathway for developing therapies for hair loss. Int. J. Mol. Sci. 2020, 21, 4915. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Nan, W.; Wang, S.; Song, X.; Si, H.; Li, T.; Li, G. Epigallocatechin-3-gallate promotes the growth of mink hair follicles through sonic hedgehog and protein kinase B signaling pathways. Front. Pharmacol. 2018, 9, 674. [Google Scholar] [CrossRef]
- Trüeb, R.M. Molecular mechanisms of androgenetic alopecia. Exp. Gerontol. 2002, 37, 981–990. [Google Scholar] [CrossRef]
- Balık, A.R.; Balık, Z.B.; Aktaş, A.; Neşelioğlu, S.; Karabulut, E.; Karabulut, A.B. Examination of androgenetic alopecia with serum biomarkers. J. Cosmet. Dermatol. 2021, 20, 1855–1859. [Google Scholar] [CrossRef]
- Sasaki, M.; Shinozaki, S.; Morinaga, H.; Kaneki, M.; Nishimura, E.; Shimokado, K. iNOS inhibits hair regeneration in obese diabetic (ob/ob) mice. Biochem. Biophys. Res. Commun. 2018, 501, 893–897. [Google Scholar] [CrossRef]
- Manosroi, A.; Ruksiriwanich, W.; Abe, M.; Sakai, H.; Aburai, K.; Manosroi, W.; Manosroi, J. Physico-chemical properties of cationic niosomes loaded with fraction of rice (Oryza sativa) bran extract. J. Nanosci. Nanotechnol. 2012, 12, 7339–7345. [Google Scholar] [CrossRef]
- Orellana, E.A.; Kasinski, A.L. Sulforhodamine B (SRB) assay in cell culture to investigate cell proliferation. Bio. Protoc. 2016, 6, e1984. [Google Scholar] [CrossRef] [Green Version]
- Nazir, Y.; Linsaenkart, P.; Khantham, C.; Chaitep, T.; Jantrawut, P.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R.; et al. High efficiency in vitro wound healing of Dictyophora indusiata extracts via anti-inflammatory and collagen stimulating (MMP-2 inhibition) mechanisms. J. Fungus 2021, 7, 1100. [Google Scholar] [CrossRef] [PubMed]
- Lolli, F.; Pallotti, F.; Rossi, A.; Fortuna, M.C.; Caro, G.; Lenzi, A.; Sansone, A.; Lombardo, F. Androgenetic alopecia: A review. Endocrine 2017, 57, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Madaan, A.; Verma, R.; Singh, A.T.; Jaggi, M. Review of hair follicle dermal papilla cells as in vitro screening model for hair growth. Int. J. Cosmet. Sci. 2018, 40, 429–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.; Chen, J.; Chen, F.; Wang, C.; Guo, X.; Wang, C.; Fan, Y.; Wang, Y.; Peng, Y.; Li, W. Liposomal honokiol promotes hair growth via activating Wnt3a/β-catenin signaling pathway and down regulating TGF-β1 in C57BL/6N mice. Biomed. Pharmacother. 2021, 141, 111793. [Google Scholar] [CrossRef]
- Sittisart, P.; Yossan, S.; Prasertsan, P. Antifungal property of chili, shallot and garlic extracts against pathogenic fungi, Phomopsis spp., isolated from infected leaves of para rubber (Hevea brasiliensis Muell. Arg.). Agric. Nat. Resour. 2017, 51, 485–491. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative stress and inflammation: What polyphenols can do for us? Oxid. Med. Cell. Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [Green Version]
- English Jr, R.S. A hypothetical pathogenesis model for androgenic alopecia: Clarifying the dihydrotestosterone paradox and rate-limiting recovery factors. Med. Hypotheses 2018, 111, 73–81. [Google Scholar] [CrossRef]
- Lee, M.; Rho, H.S.; Choi, K. Anti-inflammatory effects of a P-coumaric acid and kojic acid derivative in LPS-stimulated RAW264.7 macrophage cells. Biotechnol. Bioprocess. Eng. 2019, 24, 653–657. [Google Scholar] [CrossRef]
- Huang, N.; Hauck, C.; Yum, M.Y.; Rizshsky, L.; Widrlechner, M.P.; McCoy, J.A.; Murphy, P.A.; Dixon, P.M.; Nikolau, B.J.; Birt, D.F. Rosmarinic acid in Prunella vulgaris ethanol extract inhibits lipopolysaccharide-induced prostaglandin E2 and nitric oxide in RAW 264.7 mouse macrophages. J. Agric. Food Chem. 2009, 57, 10579–10589. [Google Scholar] [CrossRef] [Green Version]
- Hämäläinen, M.; Nieminen, R.; Vuorela, P.; Heinonen, M.; Moilanen, E. Anti-inflammatory effects of flavonoids: Genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-κB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-κB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat. Inflamm. 2007, 2007, 45673. [Google Scholar]
- Khantham, C.; Linsaenkart, P.; Chaitep, T.; Jantrawut, P.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R.; Prom-u-thai, C.; et al. Antioxidation, anti-inflammation, and regulation of SRD5A gene expression of Oryza sativa cv. Bue Bang 3 CMU husk and bran extracts as androgenetic alopecia molecular treatment substances. Plants 2022, 11, 330. [Google Scholar] [CrossRef] [PubMed]
- Sawaya, M.E.; Price, V.H. Different levels of 5α-reductase type I and II, aromatase, and androgen receptor in hair follicles of women and men with androgenetic alopecia. J. Invest. Dermatol. 1997, 109, 296–300. [Google Scholar] [CrossRef] [Green Version]
- Sánchez, P.; Serrano-Falcón, C.; Torres, J.; Serrano, S.; Ortega, E. 5α-Reductase isozymes and aromatase mRNA levels in plucked hair from young women with female pattern hair loss. Arch. Dermatol. 2018, 310, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Asada, Y.; Sonoda, T.; Ojiro, M.; Kurata, S.; Sato, T.; Ezaki, T.; Takayasu, S. 5α-reductase type 2 is constitutively expressed in the dermal papilla and connective tissue sheath of the hair follicle in vivo but not during culture in vitro. J. Clin. Endocrinol. Metab. 2001, 86, 2875–2880. [Google Scholar] [PubMed] [Green Version]
- Li, J.; Ding, Z.; Wang, Z.; Lu, J.-F.; Maity, S.N.; Navone, N.M.; Logothetis, C.J.; Mills, G.B.; Kim, J. Androgen regulation of 5α-reductase isoenzymes in prostate cancer: Implications for prostate cancer prevention. PLoS ONE 2011, 6, e28840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Song, L.; Wang, H.; Wang, J.; Xu, Z.; Xing, N. Quercetin in prostate cancer: Chemotherapeutic and chemopreventive effects, mechanisms and clinical application potential. Oncol. Rep. 2015, 33, 2659–2668. [Google Scholar] [CrossRef] [Green Version]
- Hiipakka, R.A.; Zhang, H.-Z.; Dai, W.; Dai, Q.; Liao, S. Structure–activity relationships for inhibition of human 5α-reductases by polyphenols. Biochem. Pharmacol. 2002, 63, 1165–1176. [Google Scholar] [CrossRef]
- Ruksiriwanich, W.; Khantham, C.; Linsaenkart, P.; Chaitep, T.; Jantrawut, P.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R. In vitro and in vivo regulation of SRD5A mRNA expression of supercritical carbon dioxide extract from Asparagus racemosus Willd. Root as anti-sebum and pore-minimizing active ingredients. Molecules 2022, 27, 1535. [Google Scholar] [CrossRef]
- Chittasupho, C.; Manthaisong, A.; Okonogi, S.; Tadtong, S.; Samee, W. Effects of quercetin and curcumin combination on antibacterial, antioxidant, in vitro wound healing and migration of human dermal fibroblast cells. Int. J. Mol. Sci. 2022, 23, 142. [Google Scholar] [CrossRef]
- Rondanelli, M.; Perna, S.; Peroni, G.; Guido, D. A bibliometric study of scientific literature in Scopus on botanicals for treatment of androgenetic alopecia. J. Cosmet. Dermatol. 2016, 15, 120–130. [Google Scholar] [CrossRef]
- Murata, K.; Noguchi, K.; Kondo, M.; Onishi, M.; Watanabe, N.; Okamura, K.; Matsuda, H. Promotion of hair growth by Rosmarinus officinalis leaf extract. Phytother. Res. 2013, 27, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, J.; Burgeson, R.E.; Morgan, B.A. Wnt signaling maintains the hair-inducing activity of the dermal papilla. Genes Dev. 2000, 14, 1181–1185. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, Y.; Jia, K.; Xu, X.; Li, Y.; Zhao, Y.; Zhang, X.; Zhang, J.; Liu, G.; Deng, S.; et al. Crosstalk between androgen and Wnt/β-catenin leads to changes of wool density in FGF5-knockout sheep. Cell Death Dis. 2020, 11, 407. [Google Scholar] [CrossRef] [PubMed]
- Leiros, G.J.; Attorresi, A.I.; Balaña, M.E. Hair follicle stem cell differentiation is inhibited through cross-talk between Wnt/β-catenin and androgen signalling in dermal papilla cells from patients with androgenetic alopecia. Br. J. Dermatol. Suppl. 2012, 166, 1035–1042. [Google Scholar] [CrossRef] [PubMed]
- Kwack, M.H.; Ahn, J.S.; Kim, M.K.; Kim, J.C.; Sung, Y.K. Preventable effect of L-threonate, an ascorbate metabolite, on androgen-driven balding via repression of dihydrotestosteroneinduced dickkopf-1 expression in human hair dermal papilla cells. BMB Rep. 2010, 43, 688–692. [Google Scholar] [CrossRef]
- Kwack, M.H.; Sung, Y.K.; Chung, E.J.; Im, S.U.; Ahn, J.S.; Kim, M.K.; Kim, J.C. Dihydrotestosterone-inducible Dickkopf 1 from balding dermal papilla cells causes apoptosis in follicular keratinocytes. J. Invest. Dermatol. 2008, 128, 262–269. [Google Scholar] [CrossRef] [Green Version]
- Zhang, N.n.; Park, D.K.; Park, H.J. Hair growth-promoting activity of hot water extract of Thuja orientalis. BMC Complement. Altern. Med. 2013, 13, 9. [Google Scholar] [CrossRef] [Green Version]
- Huelsken, J.; Vogel, R.; Erdmann, B.; Cotsarelis, G.; Birchmeier, W. beta-Catenin controls hair follicle morphogenesis and stem cell differentiation in the skin. Cell 2001, 105, 533–545. [Google Scholar] [CrossRef] [Green Version]
- Mi, Y.; Zhong, L.; Lu, S.; Hu, P.; Pan, Y.; Ma, X.; Yan, B.; Wei, Z.; Yang, G. Quercetin promotes cutaneous wound healing in mice through Wnt/β-catenin signaling pathway. J. Ethnopharmacol. 2022, 290, 115066. [Google Scholar] [CrossRef]
- Jin, Z.; Ke, J.; Guo, P.; Wang, Y.; Wu, H. Quercetin improves blood-brain barrier dysfunction in rats with cerebral ischemia reperfusion via Wnt signaling pathway. Am. J. Transl. Res. 2019, 11, 4683–4695. [Google Scholar]
- Gritli-Linde, A.; Hallberg, K.; Harfe, B.D.; Reyahi, A.; Kannius-Janson, M.; Nilsson, J.; Cobourne, M.T.; Sharpe, P.T.; McMahon, A.P.; Linde, A. Abnormal hair development and apparent follicular transformation to mammary gland in the absence of hedgehog signaling. Dev. Cell 2007, 12, 99–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Veltri, A.; Lang, C.; Lien, W.H. Concise review: Wnt signaling pathways in skin development and epidermal stem cells. Stem Cells 2018, 36, 22–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, S.H.; Kim, Y.; Jung, N.; Hwang, J.W.; Kim, N.; Ha, J.C.; Kim, M.J.; Lee, Y.; Choi, Y.S.; Han, K.; et al. Hair growth-promoting effect of recombinant human sonic hedgehog proteins. Biomed. Dermatol. 2019, 3, 7. [Google Scholar] [CrossRef] [Green Version]
- Paladini, R.D.; Saleh, J.; Qian, C.; Xu, G.X.; Rubin, L.L. Modulation of hair growth with small molecule agonists of the hedgehog signaling pathway. J. Invest. Dermatol. 2005, 125, 638–646. [Google Scholar] [CrossRef] [Green Version]
- St-Jacques, B.; Dassule, H.R.; Karavanova, I.; Botchkarev, V.A.; Li, J.; Danielian, P.S.; McMahon, J.A.; Lewis, P.M.; Paus, R.; McMahon, A.P. Sonic hedgehog signaling is essential for hair development. Curr. Biol. 1998, 8, 1058–1068. [Google Scholar] [CrossRef] [Green Version]
- Naser, B.; Bodinet, C.; Tegtmeier, M.; Lindequist, U. Thuja occidentalis (Arbor vitae): A review of its pharmaceutical, pharmacological and clinical properties. Evid. Based Complement. Altern. 2005, 2, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Lin, L.; Ni, B.; Lin, H.; Zhang, M.; Li, X.; Yin, X.; Qu, C.; Ni, J. Traditional usages, botany, phytochemistry, pharmacology and toxicology of Polygonum multiflorum Thunb.: A review. J. Ethnopharmacol. 2015, 159, 158–183. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Zhang, N.; Park, D.K. Topical application of Polygonum multiflorum extract induces hair growth of resting hair follicles through upregulating Shh and β-catenin expression in C57BL/6 mice. J. Ethnopharmacol. 2011, 135, 369–375. [Google Scholar] [CrossRef]
- Cebe-Suarez, S.; Zehnder-Fjällman, A.; Ballmer-Hofer, K. The role of VEGF receptors in angiogenesis; complex partnerships. Cell. Mol. Life Sci. 2006, 63, 601–615. [Google Scholar] [CrossRef] [Green Version]
- Yano, K.; Brown, L.F.; Detmar, M. Control of hair growth and follicle size by VEGF-mediated angiogenesis. J. Clin. Investig. 2001, 107, 409–417. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.Y.; Su, C.H.; Chiang, C.Y.; Wu, C.N.; Kuan, Y.H. Observation of the expression of vascular endothelial growth factor and the potential effect of promoting hair growth treated with chinese herbal BeauTop. Evid. Based Complement. Altern. 2021, 2021, 6667011. [Google Scholar] [CrossRef] [PubMed]
- Kubanov, A.; Gallyamova, Y.A.; Korableva, O. The study of growth factors in patients with androgenic alopecia. Biomed. Pharmacol. J. 2017, 10, 1219–1228. [Google Scholar] [CrossRef]
- Formiga, R.d.O.; Alves Júnior, E.B.; Vasconcelos, R.C.; Araújo, A.A.; de Carvalho, T.G.; de Araújo Junior, R.F.; Guerra, G.B.C.; Vieira, G.C.; de Oliveira, K.M.; Diniz, M.d.F.F.M.; et al. Effect of p-cymene and rosmarinic acid on gastric ulcer healing–involvement of multiple endogenous curative mechanisms. Phytomedicine 2021, 86, 153497. [Google Scholar] [CrossRef] [PubMed]
- Leksawasdi, N.; Taesuwan, S.; Prommajak, T.; Techapun, C.; Khonchaisri, R.; Sittilop, N.; Halee, A.; Jantanasakulwong, K.; Phongthai, S.; Nunta, R.; et al. Ultrasonic extraction of bioactive compounds from green soybean pods and application in green soybean milk antioxidants fortification. Foods 2022, 11, 588. [Google Scholar] [CrossRef]
- Arjin, C.; Hongsibsong, S.; Pringproa, K.; Seel-Audom, M.; Ruksiriwanich, W.; Sutan, K.; Sommano, S.R.; Sringarm, K. Effect of ethanolic Caesalpinia sappan fraction on in vitro antiviral activity against porcine reproductive and respiratory syndrome virus. Vet. Sci. 2021, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Wisetkomolmat, J.; Arjin, C.; Satsook, A.; Seel-Audom, M.; Ruksiriwanich, W.; Prom-u-Thai, C.; Sringarm, K. Comparative analysis of nutritional components and phytochemical attributes of selected Thai rice bran. Front. Nutr. 2022, 9. [Google Scholar] [CrossRef]
- Manosroi, A.; Chankhampan, C.; Kietthanakorn, B.O.; Ruksiriwanich, W.; Chaikul, P.; Boonpisuttinant, K.; Sainakham, M.; Manosroi, W.; Tangjai, T.; Manosroi, J. Pharmaceutical and cosmeceutical biological activities of hemp (Cannabis sativa L. var. sativa) leaf and seed extracts. Chiang Mai J. Sci. 2019, 46, 180–195. [Google Scholar]
- Khantham, C.; Yooin, W.; Sringarm, K.; Sommano, S.R.; Jiranusornkul, S.; Carmona, F.D.; Nimlamool, W.; Jantrawut, P.; Rachtanapun, P.; Ruksiriwanich, W. Effects on steroid 5-alpha reductase gene expression of Thai rice bran extracts and molecular dynamics study on SRD5A2. Biology 2021, 10, 319. [Google Scholar] [CrossRef]
- Wang, F. Semi-Quantitative RT-PCR: An Effective Method to Explore The Regulation of Gene Transcription Level Affected by Environmental Pollutants. In Environmental Toxicology and Toxicogenomics: Principles, Methods, and Applications; Pan, X., Zhang, B., Eds.; Springer: New York, NY, USA, 2021; pp. 95–103. [Google Scholar]
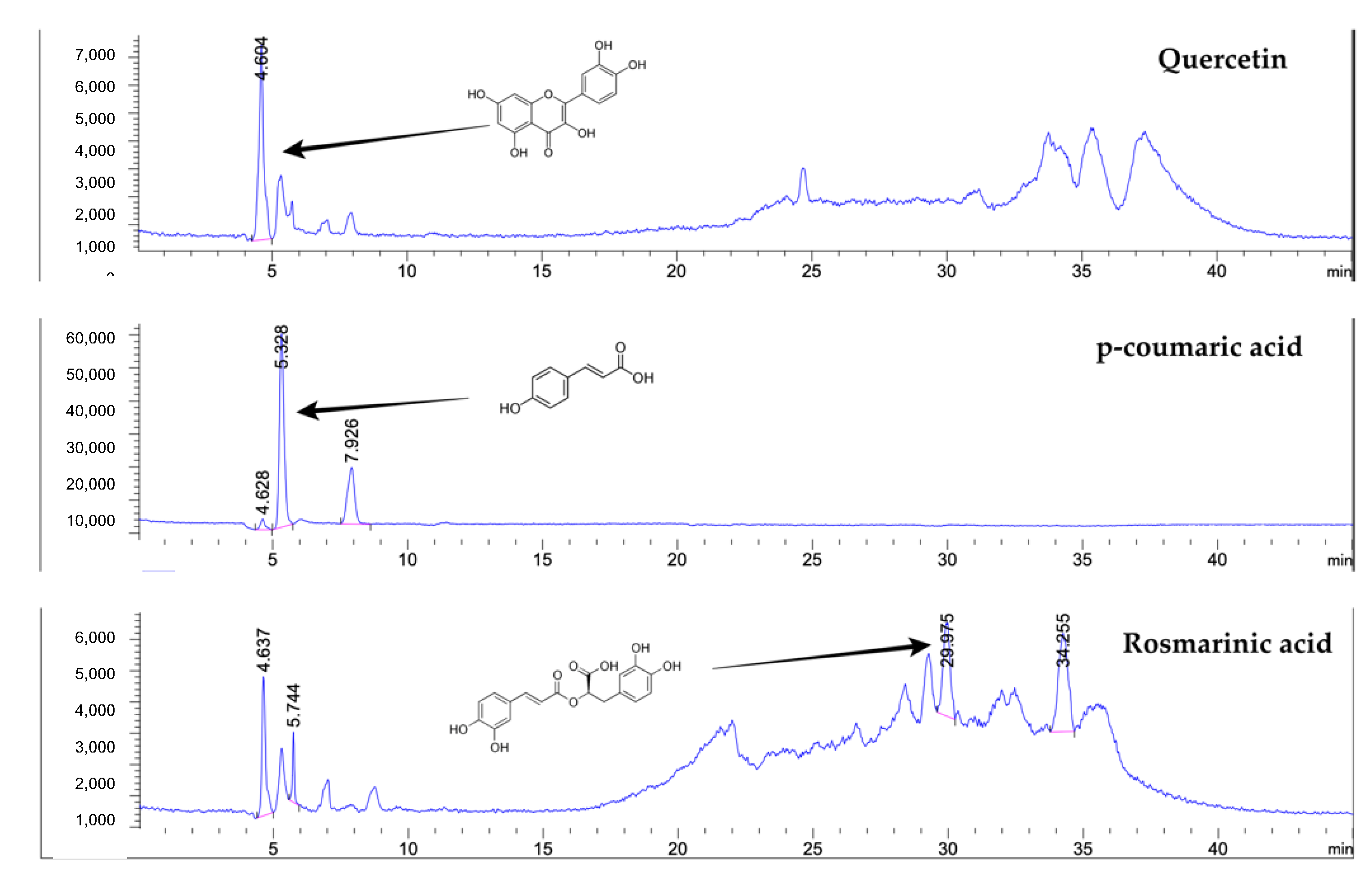


| Bioactive Compounds | Content | |
|---|---|---|
| Total polysaccharide content | 0.90 ± 0.06 | mg D-glucose/g |
| Total protein content | 1.01 ± 0.04 | mg BSAE/g |
| Total phenolic content | 4.69 ± 0.42 | mg GAE/g |
| Total flavonoid content | <0.003 | mg EGCGE/g |
| Phenolic compounds | ||
| p-Coumaric acid | 1.091 ± 0.011 | mg/g |
| Quercetin | 0.029 ± 0.002 | mg/g |
| Rosmarinic acid | 0.234 ± 0.007 | mg/g |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruksiriwanich, W.; Khantham, C.; Muangsanguan, A.; Chittasupho, C.; Rachtanapun, P.; Jantanasakulwong, K.; Phimolsiripol, Y.; Sommano, S.R.; Sringarm, K.; Ferrer, E.; et al. Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract. Plants 2022, 11, 1499. https://doi.org/10.3390/plants11111499
Ruksiriwanich W, Khantham C, Muangsanguan A, Chittasupho C, Rachtanapun P, Jantanasakulwong K, Phimolsiripol Y, Sommano SR, Sringarm K, Ferrer E, et al. Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract. Plants. 2022; 11(11):1499. https://doi.org/10.3390/plants11111499
Chicago/Turabian StyleRuksiriwanich, Warintorn, Chiranan Khantham, Anurak Muangsanguan, Chuda Chittasupho, Pornchai Rachtanapun, Kittisak Jantanasakulwong, Yuthana Phimolsiripol, Sarana Rose Sommano, Korawan Sringarm, Emilia Ferrer, and et al. 2022. "Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract" Plants 11, no. 11: 1499. https://doi.org/10.3390/plants11111499
APA StyleRuksiriwanich, W., Khantham, C., Muangsanguan, A., Chittasupho, C., Rachtanapun, P., Jantanasakulwong, K., Phimolsiripol, Y., Sommano, S. R., Sringarm, K., Ferrer, E., & Barba, F. J. (2022). Phytochemical Constitution, Anti-Inflammation, Anti-Androgen, and Hair Growth-Promoting Potential of Shallot (Allium ascalonicum L.) Extract. Plants, 11(11), 1499. https://doi.org/10.3390/plants11111499