Wound Healing and Antioxidant Capabilities of Zizyphus mauritiana Fruits: In-Vitro, In-Vivo, and Molecular Modeling Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plants Material
2.2. In-Vitro Antioxidant Activity
2.3. In-Vivo Wound Healing Activity
2.4. LC-MS Analysis
2.5. Molecular Docking
2.6. Prediction of the Antioxidant activity
3. Results and Discussion
3.1. In Vitro Antioxidant Activity
3.1.1. Hydrogen Peroxide Scavenging Activity
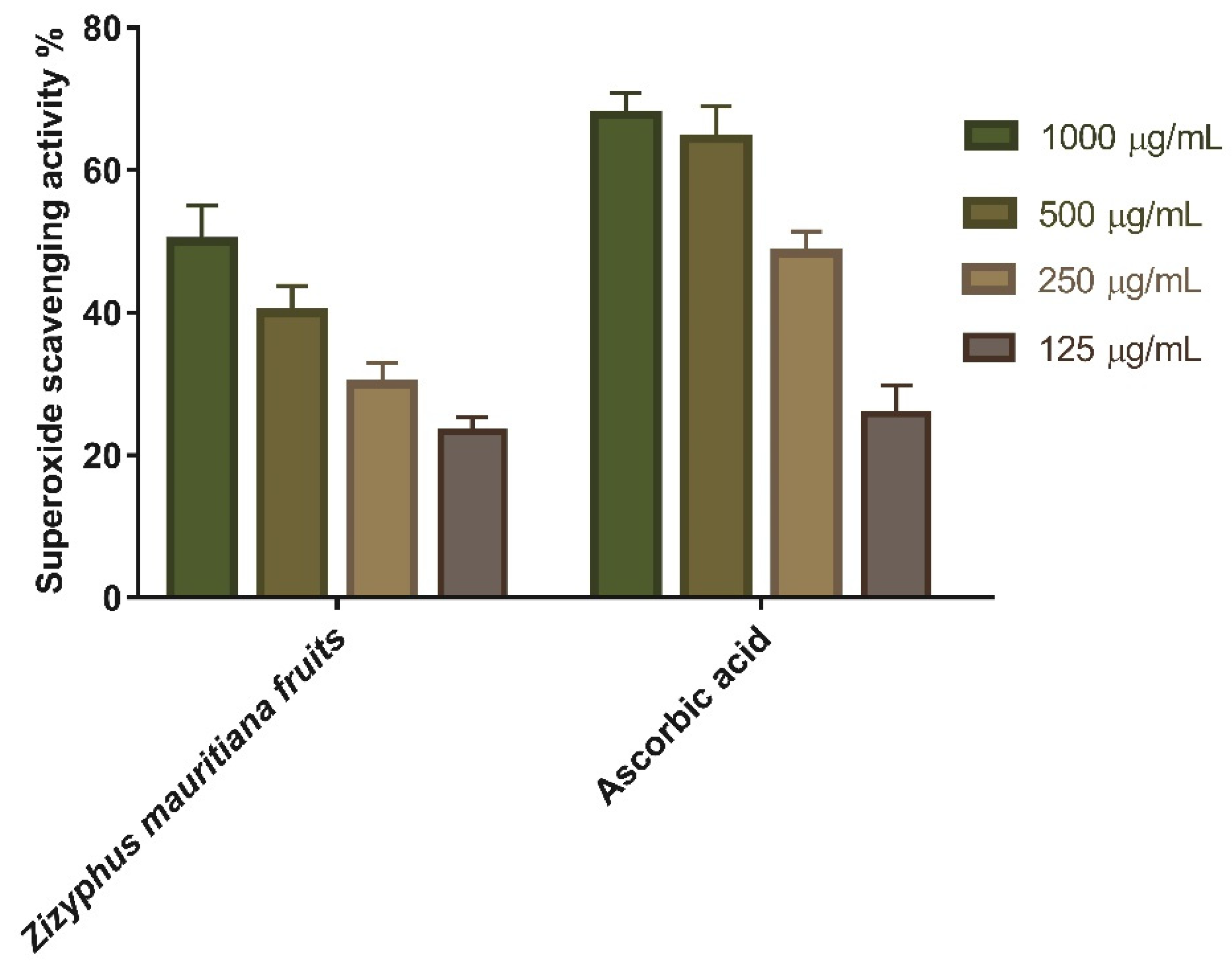
3.1.2. Superoxide Radical Scavenging Activity
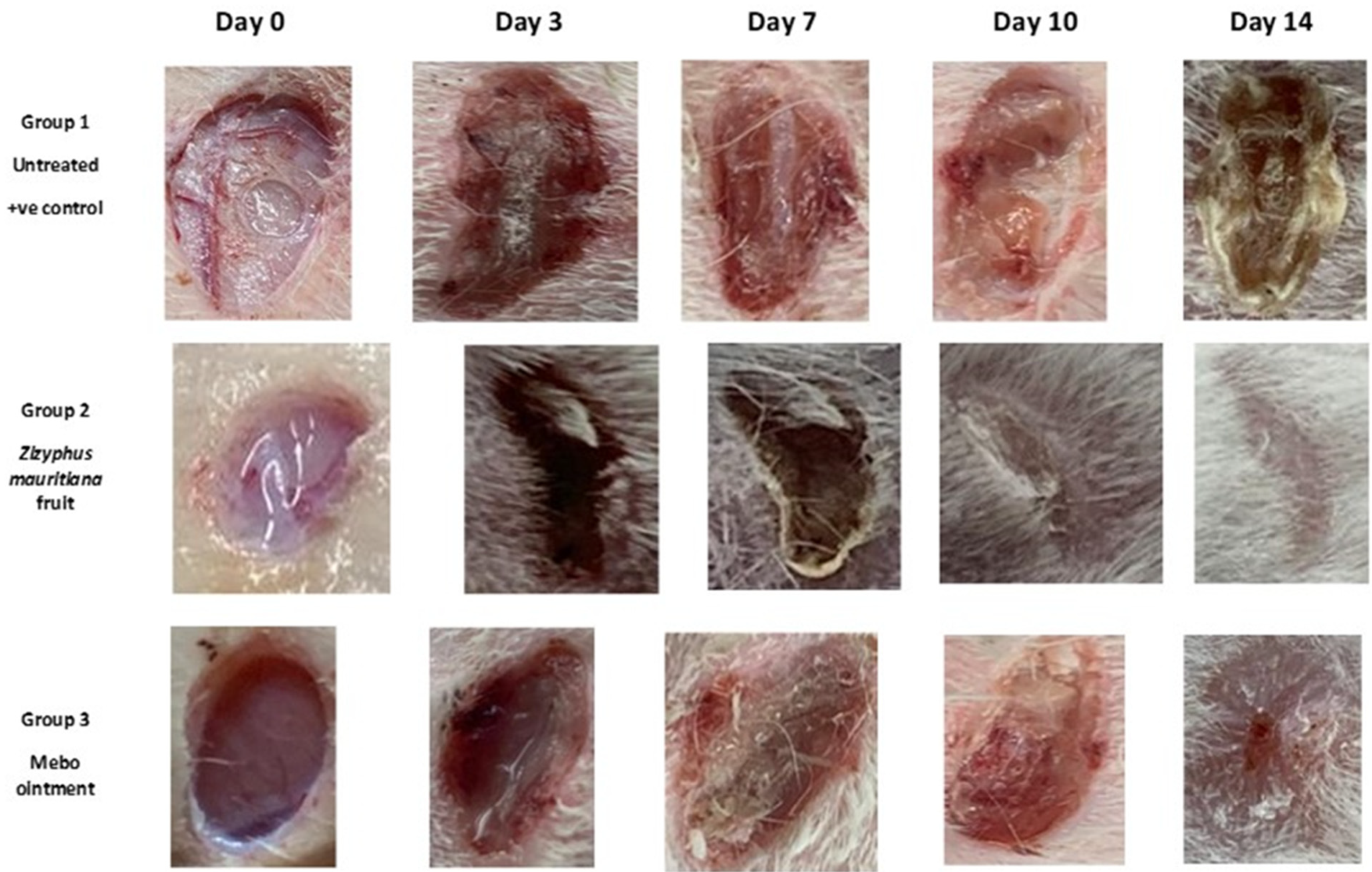
3.2. Wound Healing Activity
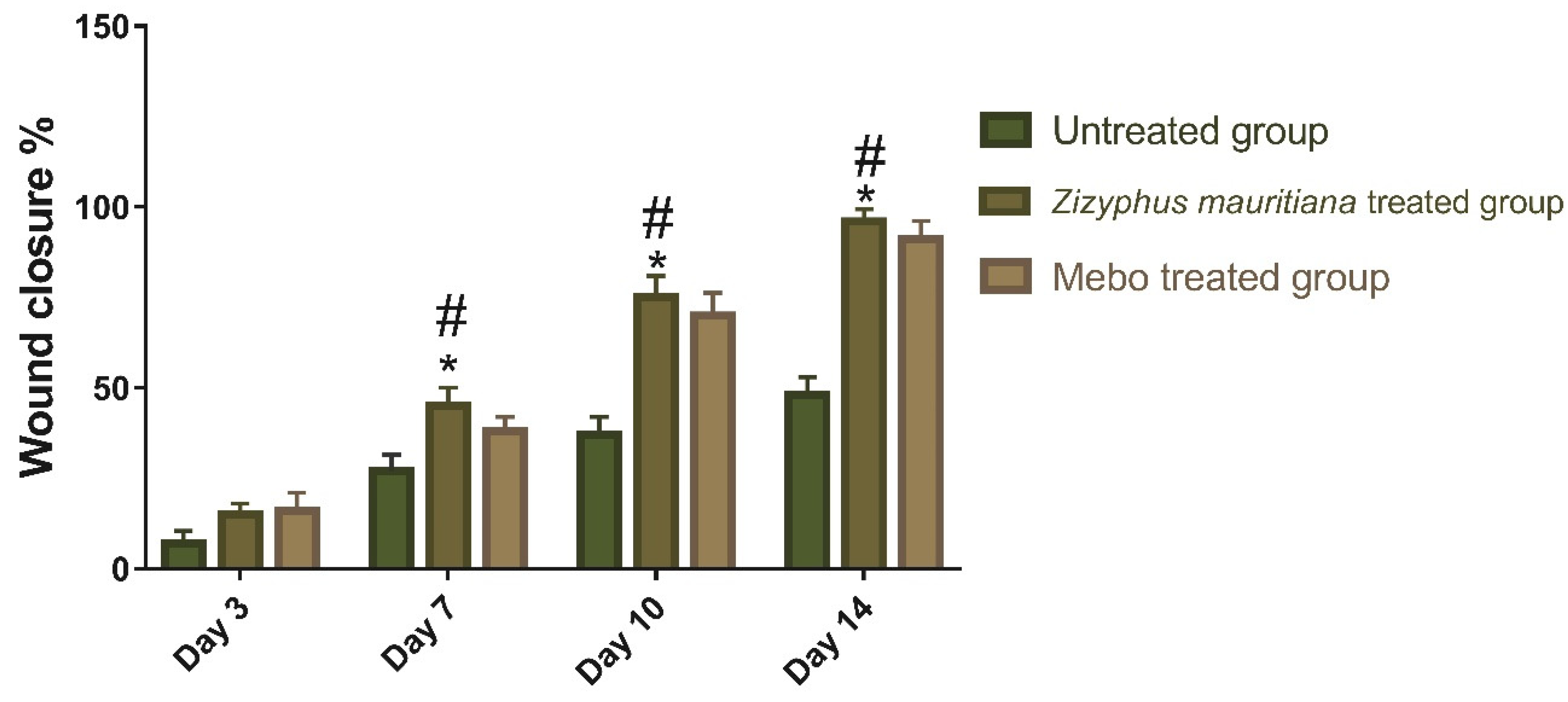
3.2.1. Wound Closure Rate Estimation
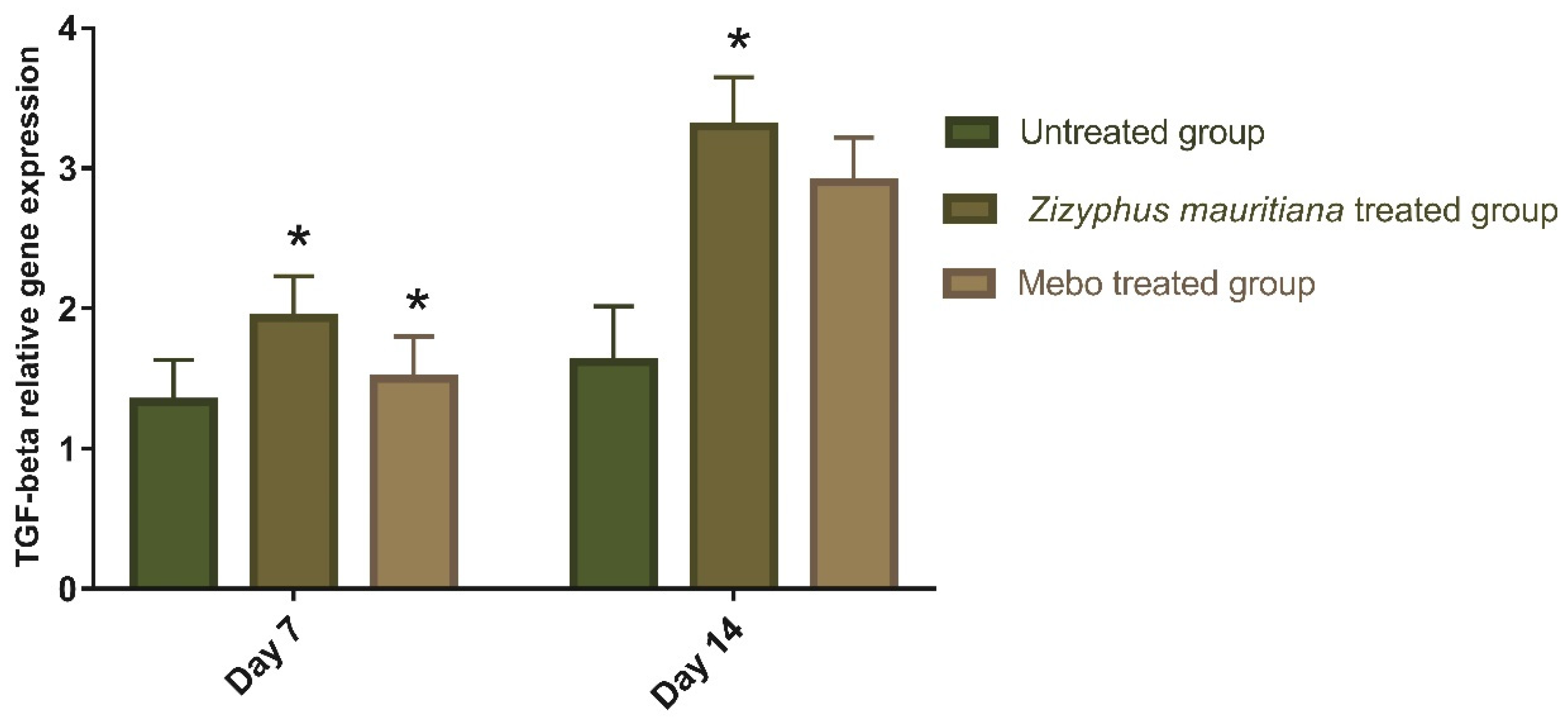
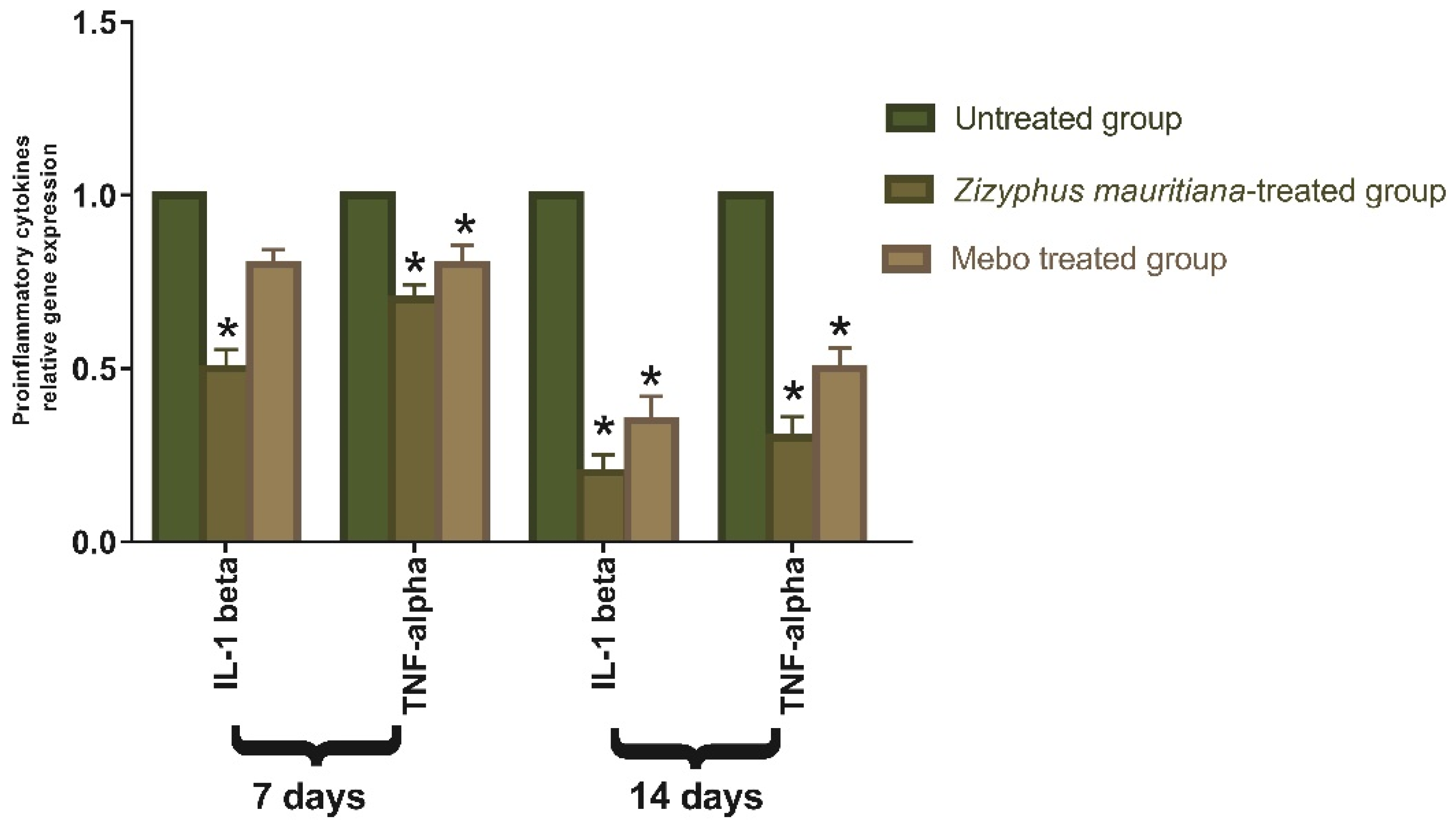
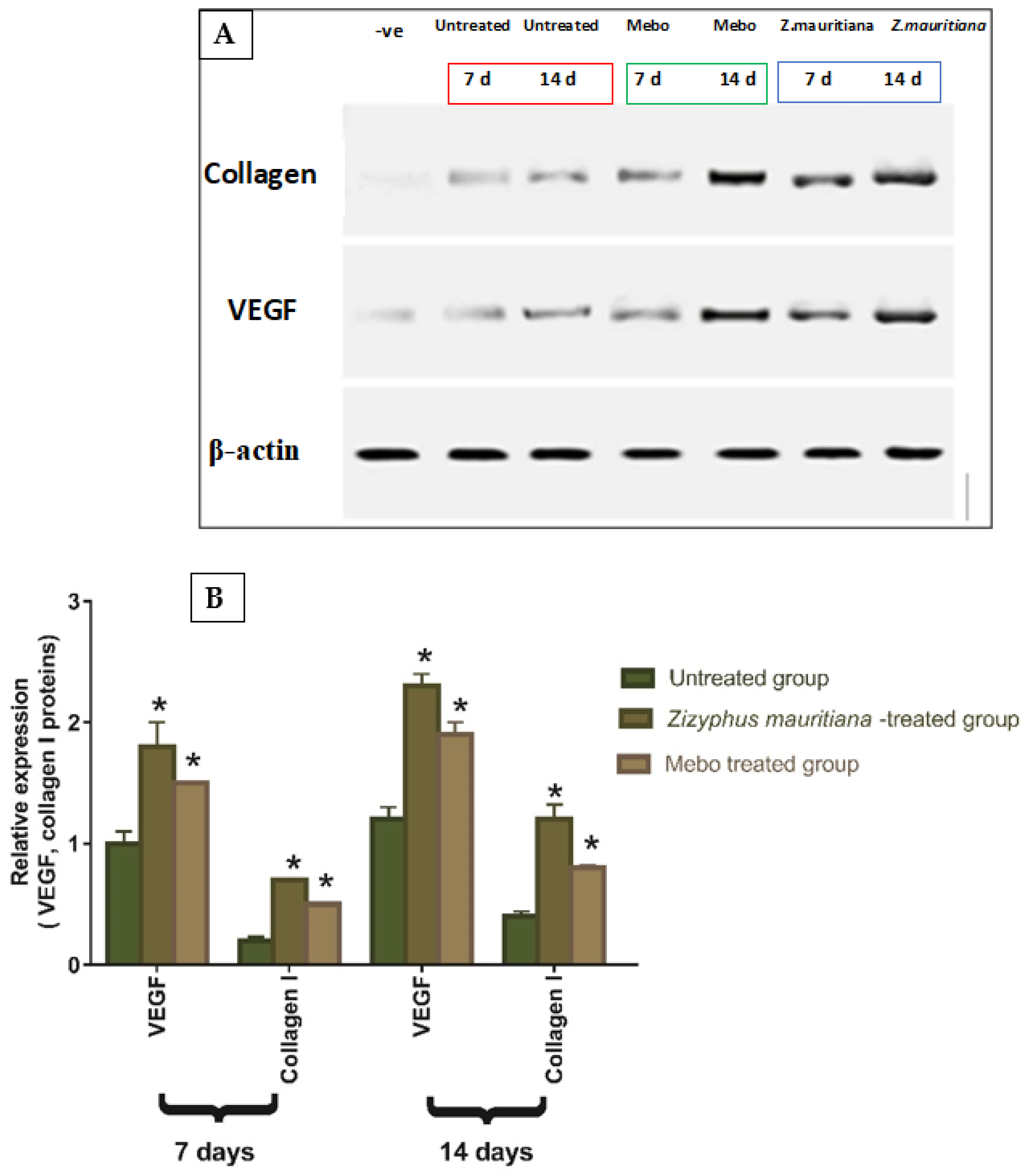
3.2.2. Effect of Z. mauritiana Fruit Extract on Expression of TGF-β, TNF-α, IL-1β, Collagen Type I, and VEGF
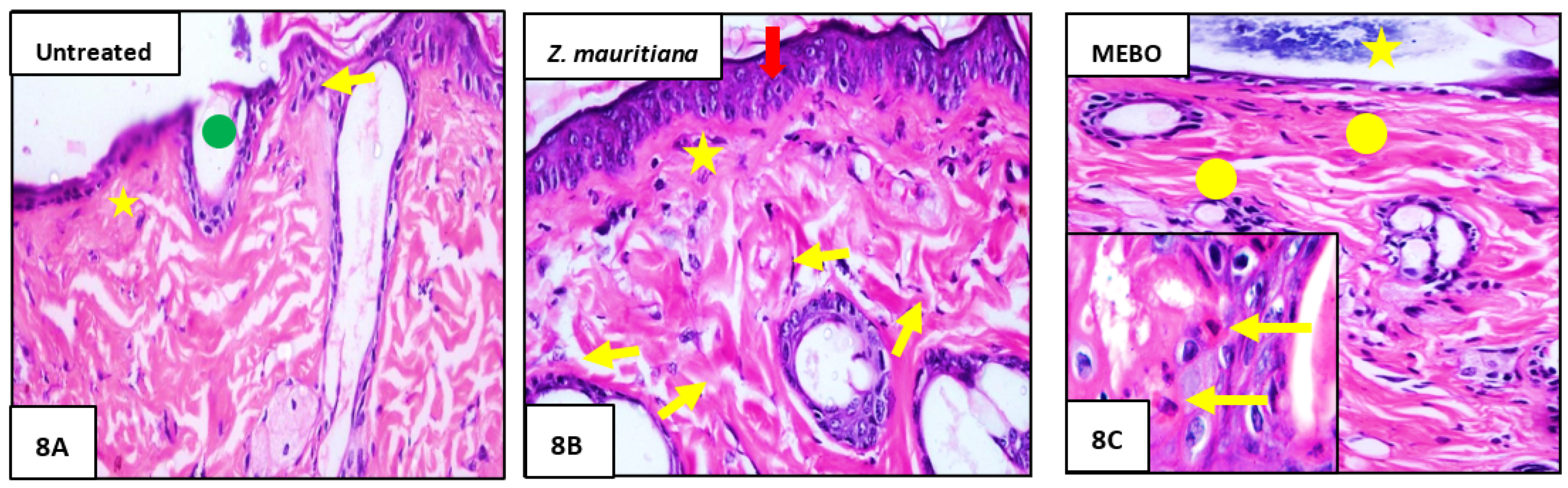
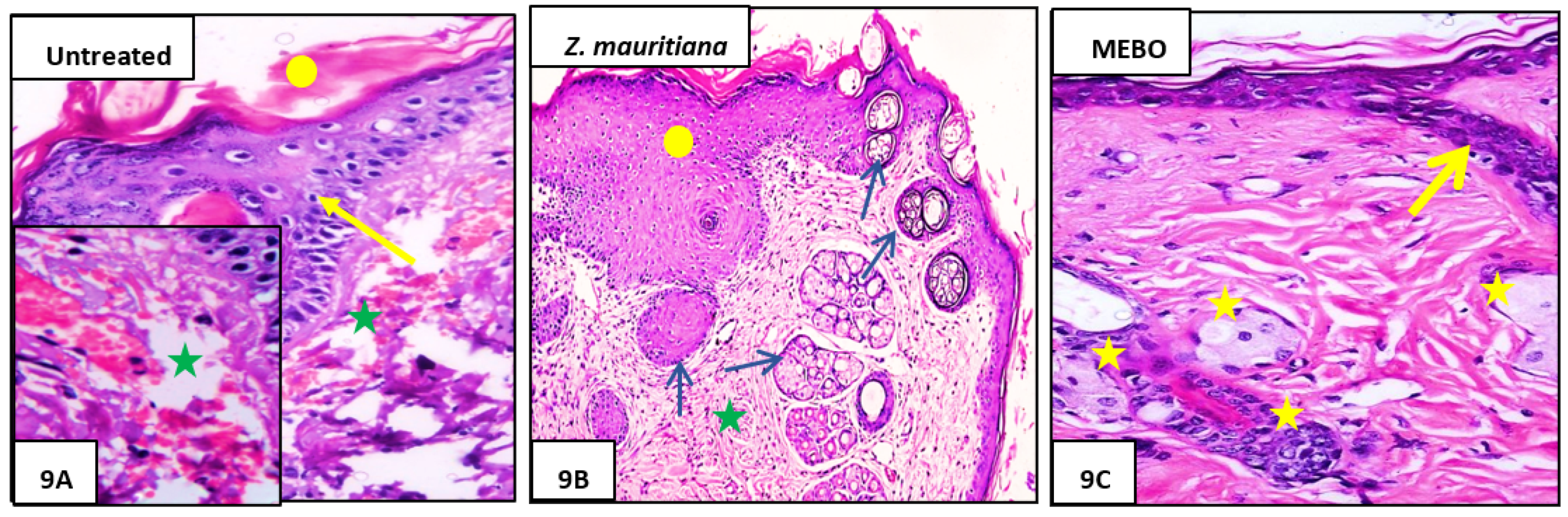
3.2.3. Histopathological Study
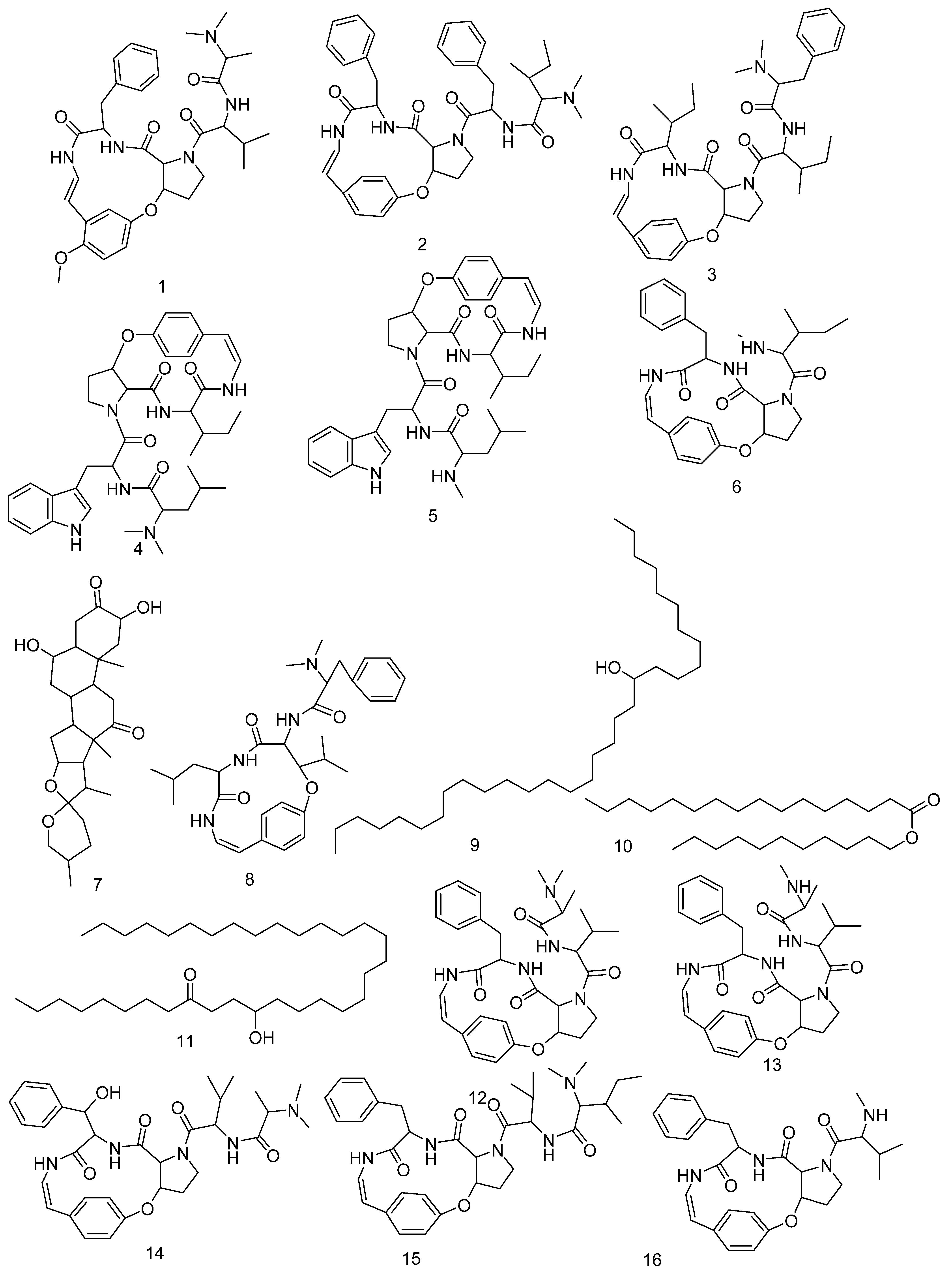
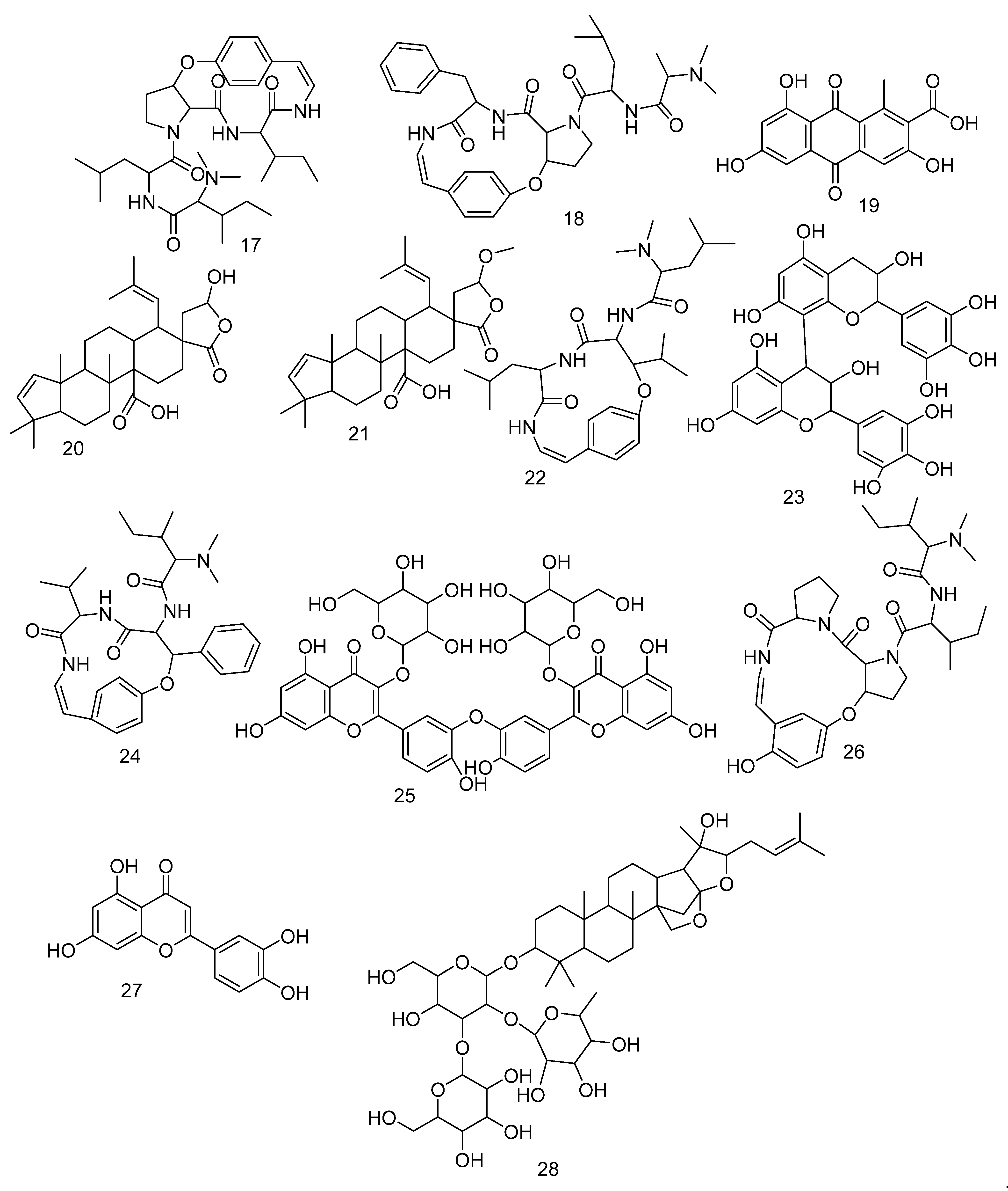
3.3. LC-HRMS Chemical Profiling
3.4. Computational Analysis
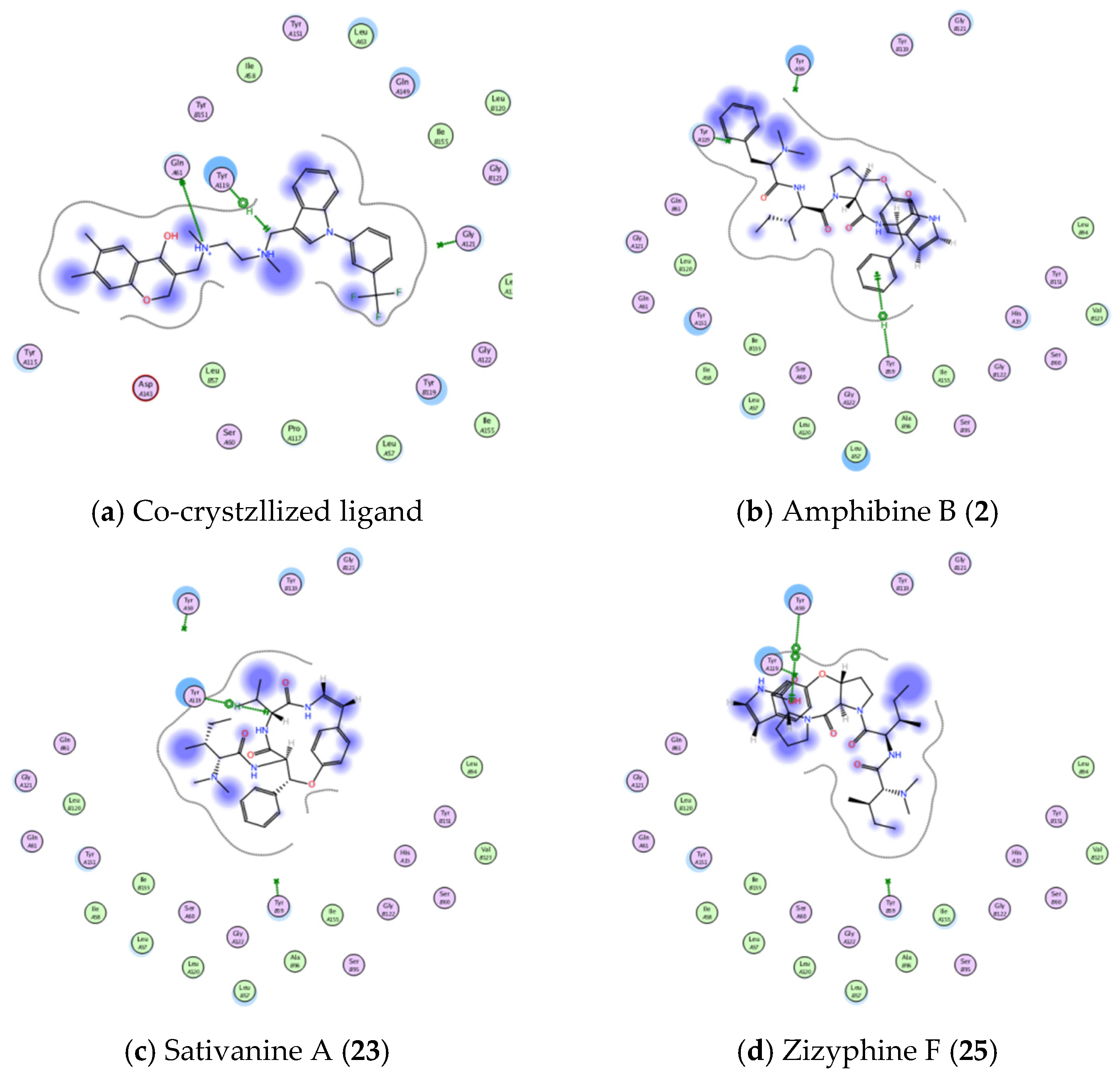
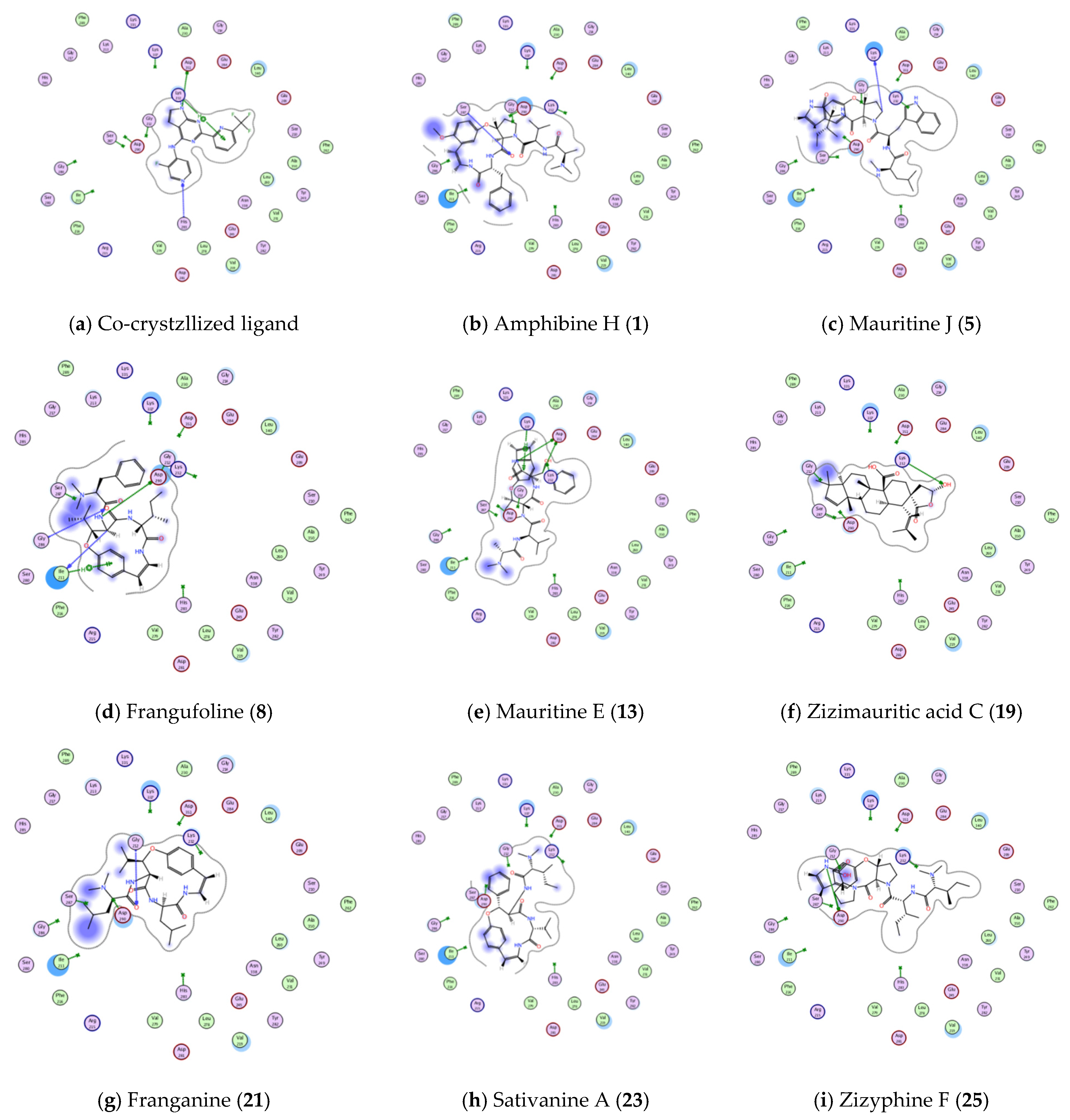
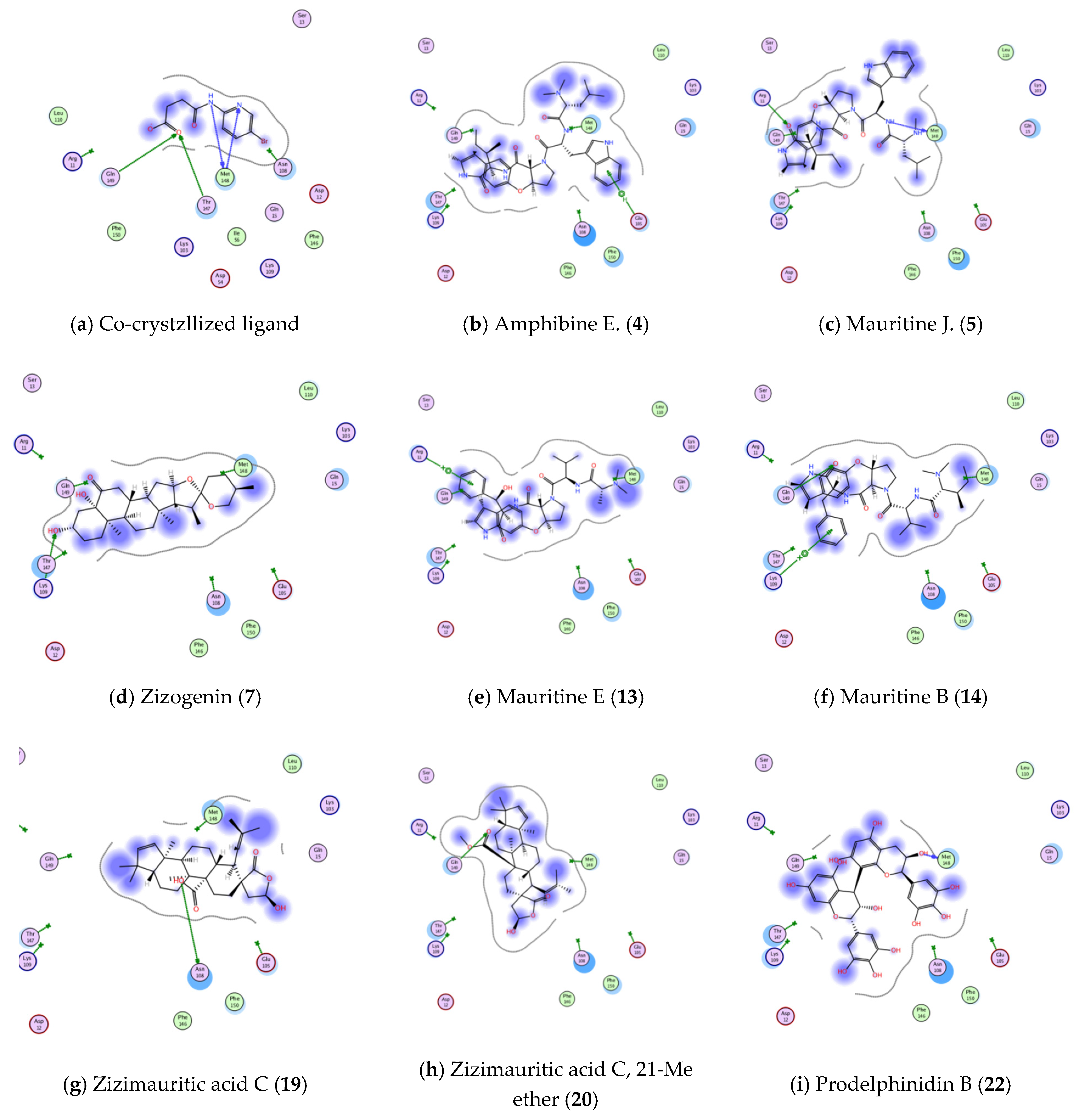
3.4.1. Molecular Docking Studies
3.4.2. Antioxidant Activity Prediction
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moeini, A.; Pedram, P.; Makvandi, P.; Malinconico, M.; d’Ayala, G.G. Wound healing and antimicrobial effect of active secondary metabolites in chitosan-based wound dressings: A review. Carbohydr. Polym. 2020, 233, 115839. [Google Scholar] [CrossRef] [PubMed]
- Alnusaire, T.S.; Sayed, A.M.; Elmaidomy, A.H.; Al-Sanea, M.M.; Albogami, S.; Albqmi, M.; Alowaiesh, B.F.; Mostafa, E.M.; Musa, A.; Youssif, K.A.; et al. An In Vitro and In Silico Study of the Enhanced Antiproliferative and Pro-Oxidant Potential of Olea europaea L. cv. Arbosana Leaf Extract via Elastic Nanovesicles (Spanlastics). Antioxidants 2021, 10, 1860. [Google Scholar] [CrossRef] [PubMed]
- Musa, A.; Shady, N.H.; Ahmed, S.R.; Alnusaire, T.; Sayed, A.; Alowaiesh, B.; Sabouni, I.; Al-Sanea, M.; Mostafa, E.; Youssif, K.; et al. Antiulcer Potential of Olea europea L. cv. Arbequina Leaf Extract Supported by Metabolic Profiling and Molecular Docking. Antioxidants 2021, 10, 644. [Google Scholar] [CrossRef] [PubMed]
- Hisham Shady, N.; Youssif, K.A.; Sayed, A.M.; Belbahri, L.; Oszako, T.; Hassan, H.M.; Abdelmohsen, U.R. Sterols and triterpenes: Antiviral potential supported by in-silico analysis. Plants 2021, 10, 41. [Google Scholar] [CrossRef]
- Shady, N.H.; Hayallah, A.M.; Mohamed, M.F.; Ghoneim, M.M.; Chilingaryan, G.; Al-Sanea, M.M.; Fouad, M.A.; Kamel, M.S.; Abdelmohsen, U.R. Targeting 3CLpro and SARS-CoV-2 RdRp by Amphimedon sp. Metabolites: A Computational Study. Molecules 2021, 26, 3775. [Google Scholar] [CrossRef]
- Shady, N.H.; Tawfike, A.F.; Yahia, R.; Fouad, M.A.; Brachmann, A.O.; Piel, J.; Abdelmohsen, U.R.; Kamel, M.S. Cytotoxic activity of actinomycetes Nocardia sp. and Nocardiopsis sp. associated with marine sponge Amphimedon sp. Nat. Prod. Res. (AHEAD-OF-PRINT). 2021, 1–6. [Google Scholar] [CrossRef]
- Shady, N.H.; Khattab, A.R.; Ahmed, S.; Liu, M.; Quinn, R.J.; Fouad, M.A.; Kamel, M.S.; Bin Muhsinah, A.; Krischke, M.; Mueller, M.J. Hepatitis C Virus NS3 Protease and Helicase Inhibitors from Red Sea Sponge (Amphimedon) Species in Green Synthesized Silver Nanoparticles Assisted by in Silico Modeling and Metabolic Profiling. Int. J. Nanomed. 2020, 15, 3377. [Google Scholar] [CrossRef]
- Al-Sanea, M.M.; Abelyan, N.; Abdelgawad, M.A.; Musa, A.; Ghoneim, M.M.; Al-Warhi, T.; Aljaeed, N.; Alotaibi, O.J.; Alnusaire, T.S.; Abdelwahab, S.F.; et al. Strawberry and ginger silver nanoparticles as potential inhibitors for SARS-CoV-2 assisted by in silico modeling and metabolic profiling. Antibiotics 2021, 10, 824. [Google Scholar] [CrossRef]
- Orfali, R.; Rateb, M.E.; Hassan, H.M.; Alonazi, M.; Gomaa, M.; Mahrous, N.; GabAllah, M.; Kandeil, A.; Perveen, S.; Abdelmohsen, U.; et al. Sinapic Acid Suppresses SARS CoV-2 Replication by Targeting Its Envelope Protein. Antibiotics 2021, 10, 420. [Google Scholar] [CrossRef]
- Alsenani, F.; Ashour, A.M.; Alzubaidi, M.A.; Azmy, A.F.; Hetta, M.H.; Abu-Baih, D.H.; Elrehany, M.A.; Zayed, A.; Sayed, A.M.; Abdelmohsen, U.R.; et al. Wound Healing Metabolites from Peters’ Elephant-Nose Fish Oil: An In Vivo Investigation Supported by In Vitro and In Silico Studies. Mar. Drugs 2021, 19, 605. [Google Scholar] [CrossRef]
- Namchaiw, P.; Jaisin, Y.; Niwaspragrit, C.; Malaniyom, K.; Auvuchanon, A.; Ratanachamnong, P. The Leaf Extract of Coccinia grandis (L.) Voigt Accelerated In Vitro Wound Healing by Reducing Oxidative Stress Injury. Oxidative Med. Cell. Longev. 2021, 2021, 3963510. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, G.S.; Singh, A.K.; Thaloor, D.; Banaudha, K.K.; Patnaik, G.K.; Srimal, R.C.; Maheshwari, R.K. Enhancement of wound healing by curcumin in animals. Wound Repair Regen. 1998, 6, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Ghayempour, S.; Montazer, M.; Rad, M.M. Encapsulation of Aloe Vera extract into natural Tragacanth Gum as a novel green wound healing product. Int. J. Biol. Macromol. 2016, 93, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Sayed, U.; Deshmukh, I. Application of Herbs for Wound Dressings-Review. Int. J. Adv. Sci. Eng. 2021, 7, 1843–1848. [Google Scholar] [CrossRef]
- Plastina, P.; Bonofiglio, D.; Vizza, D.; Fazio, A.; Rovito, D.; Giordano, C.; Barone, I.; Catalano, S.; Gabriele, B. Identification of bioactive constituents of Ziziphus jujube fruit extracts exerting antiproliferative and apoptotic effects in human breast cancer cells. J. Ethnopharmacol. 2012, 140, 325. [Google Scholar] [CrossRef]
- Prakash, O.; Usmani, S.; Singh, R.; Singh, N.; Gupta, A.; Ved, A. A panoramic view on phytochemical, nutritional, and therapeutic attributes of Ziziphus mauritiana Lam.: A comprehensive review. Phytother. Res. 2021, 35, 63. [Google Scholar] [CrossRef]
- Kundu, A.; Barik, B.; Mondal, D.; Dey, A.; Banerji, A. Zizyberanalic acid, a pentacyclic triterpenoid of Zizyphus jujuba. Phytochemistry 1989, 28, 3155. [Google Scholar] [CrossRef]
- Cheng, G.; Bai, Y.; Zhao, Y.; Tao, J.; Liu, Y.; Tu, G.; Ma, L.; Liao, N.; Xu, X. Flavonoids from Ziziphus jujuba Mill var. spinosa. Tetrahedron 2000, 56, 8915. [Google Scholar] [CrossRef]
- Abalaka, M.; Daniyan, S.; Mann, A. Evaluation of the antimicrobial activities of two Ziziphus species (Ziziphus mauritiana L. and Ziziphus spinachristi L.) on some microbial pathogens. Afr. J. Pharm. Pharmacol. 2010, 4, 135. [Google Scholar]
- Tripathi, M.; Pandey, M.; Jha, R.; Pandey, V.; Tripathi, P.; Singh, J. Cyclopeptide alkaloids from Zizyphus jujuba. Fitoterapia 2001, 72, 507. [Google Scholar] [CrossRef]
- Reyes, A.; PMiclat, A.; Bañares, A.; Dela Peña, R. Lack of antibacterial activity of aqueous and ethanolic leaf extracts of Ziziphus talanai (Blanco) Merr. J. Pharm. Negat. Results 2018, 9, 44. [Google Scholar] [CrossRef]
- Elaloui, M.; Laamouri, A.; Ennajah, A.; Cerny, M.; Mathieu, C.; Vilarem, G.; Chaar, H.; Hasnaoui, B. Phytoconstituents of leaf extracts of Ziziphus jujuba Mill. plants harvested in Tunisia. Ind. Crops Prod. 2016, 83, 133. [Google Scholar] [CrossRef] [Green Version]
- Jarald, E.; Joshi, S.; Jain, D. Antidiabetic activity of extracts and fraction of Zizyphus mauritiana. Pharm. Biol. 2009, 47, 328. [Google Scholar] [CrossRef]
- Deshpande, M.S.; Tondare, P.R.; Paygude, S.V.; Apte, K.G.; Parab, P.B. Evaluation of antioxidant, anti-inflammatory and adipocyte differentiation inhibitory potential of Ziziphus mauritiana bark extract. Pharmacogn. J. 2013, 5, 205. [Google Scholar] [CrossRef]
- Talmale, S.; Bhujade, A.; Patil, M. Anti-allergic and anti-inflammatory properties of Zizyphus mauritiana root bark. Food Funct. 2015, 6, 2975. [Google Scholar] [CrossRef]
- Saiah, H.; Mokhtar, M.; Saiah, W.; Aichouni, A.; ELKebir, F.Z.; Allem, R. In vivo wound healing potential and HPLC-PDA-ESI-MS profiling of Zizyphus lotus L.(Desf.) leaves methanol extract. J. Food Biochem. 2018, 42, e12570. [Google Scholar] [CrossRef]
- Hovaneţ, M.-V.; Oprea, E.; Ancuceanu, R.V.; Duţu, L.E.; Budura, E.A.; Şeremet, O.; Ancu, I.; Moroşan, E. Wound healing properties of Ziziphus jujuba Mill. leaves. Rom. Biotechnol. Lett. 2016, 21, 11842. [Google Scholar]
- Yassien, E.E.; Hamed, M.M.; Abdelmohsen, U.R.; Hassan, H.M.; Gazwi, H.S. In vitro antioxidant, antibacterial, and antihyperlipidemic potential of ethanolic Avicennia marina leaves extract supported by metabolic profiling. Environ. Sci. Pollut. Res. 2021, 28, 27207. [Google Scholar] [CrossRef]
- Shady, N.H.; Hassan, H.A.; Elrehany, M.A.; Kamel, M.S.; Saber, E.A.; Maher, S.A.; Abo-Elsoud, F.A.; Sayed, A.M.; Abdelmohsen, U.R.; Gaber, S.S. Hyphaene thebaica (doum)-derived extract alleviates hyperglycemia in diabetic rats: A comprehensive in silico, in vitro and in vivo study. Food Funct. 2021, 12, 11303. [Google Scholar] [CrossRef]
- Alhadrami, H.A.; Alkhatabi, H.; Abduljabbar, F.H.; Abdelmohsen, U.R.; Sayed, A.M. Anticancer Potential of Green Synthesized Silver Nanoparticles of the Soft Coral Cladiella pachyclados Supported by Network Pharmacology and In Silico Analyses. Pharmaceutics 2021, 13, 1846. [Google Scholar] [CrossRef]
- Shady, N.H.; Abdelmohsen, U.R.; AboulMagd, A.M.; Amin, M.N.; Ahmed, S.; Fouad, M.A.; Kamel, M.S. Cytotoxic potential of the Red Sea sponge Amphimedon sp. supported by in silico modelling and dereplication analysis. Nat. Prod. Res. 2021, 35, 6093. [Google Scholar] [CrossRef] [PubMed]
- Siwik, D.A.; Pagano, P.J.; Colucci, W.S. Oxidative stress regulates collagen synthesis and matrix metalloproteinase activity in cardiac fibroblasts. Am. J. Physiol.-Cell Physiol. 2001, 280, 53. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Menocal, L.; Shareef, S.; Salgado, M.; Shabbir, A.; Van Badiavas, E. Role of whole bone marrow, whole bone marrow cultured cells, and mesenchymal stem cells in chronic wound healing. Stem Cell Res. Ther. 2015, 6, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singh, K.; Agrawal, N.K.; Gupta, S.K.; Sinha, P.; Singh, K. Increased expression of TLR9 associated with pro-inflammatory S100A8 and IL-8 in diabetic wounds could lead to unresolved inflammation in type 2 diabetes mellitus (T2DM) cases with impaired wound healing. J. Diabetes Its Complicat. 2016, 30, 99. [Google Scholar] [CrossRef]
- Kim, D.-O.; Chun, O.K.; Kim, Y.J.; Moon, H.-Y.; Lee, C.Y. Quantification of polyphenolics and their antioxidant capacity in fresh plums. J. Agric. Food Chem. 2003, 51, 6509. [Google Scholar] [CrossRef] [PubMed]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861. [Google Scholar] [CrossRef] [Green Version]
- Krzyszczyk, P.; Schloss, R.; Palmer, A.; Berthiaume, F. The role of macrophages in acute and chronic wound healing and interventions to promote pro-wound healing phenotypes. Front. Physiol. 2018, 9, 419. [Google Scholar] [CrossRef]
- Pachuau, L. Recent developments in novel drug delivery systems for wound healing. Expert Opin. Drug Deliv. 2015, 12, 1895. [Google Scholar] [CrossRef]
- Suguna, L.; Singh, S.; Sivakumar, P.; Sampath, P.; Chandrakasan, G. Influence of Terminalia chebula on dermal wound healing in rats. Phytother. Res. 2002, 16, 227. [Google Scholar] [CrossRef]
- Tang, T.; Yin, L.; Yang, J.; Shan, G. Emodin, an anthraquinone derivative from Rheum officinale Baill, enhances cutaneous wound healing in rats. Eur. J. Pharmacol. 2007, 567, 177. [Google Scholar] [CrossRef]
- Wankell, M.; Munz, B.; Hübner, G.; Hans, W.; Wolf, E.; Goppelt, A.; Werner, S. Impaired wound healing in transgenic mice overexpressing the activin antagonist follistatin in the epidermis. EMBO J. 2001, 20, 5361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beer, H.-D.; Gassmann, M.G.; Munz, B.; Steiling, H.; Engelhardt, F.; Bleuel, K.; Werner, S. Expression and function of keratinocyte growth factor and activin in skin morphogenesis and cutaneous wound repair. J. Investig. Dermatol. Symp. Proc. 2000, 5, 34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pastar, I.; Stojadinovic, O.; Yin, N.C.; Ramirez, H.; Nusbaum, A.G.; Sawaya, A.; Patel, S.B.; Khalid, L.; Isseroff, R.R.; Tomic-Canic, M. Epithelialization in wound healing: A comprehensive review. Adv. Wound Care 2014, 3, 445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haroon, Z.A.; Amin, K.; Saito, W.; Wilson, W.; Greenberg, C.S.; Dewhirst, M.W. SU5416 delays wound healing through inhibition of TGF-β activation. Cancer Biol. Ther. 2002, 1, 121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feinberg, T.Y.; Zheng, H.; Liu, R.; Wicha, M.S.; Yu, S.M.; Weiss, S.J. Divergent Matrix-Remodeling Strategies Distinguish Developmental from Neoplastic Mammary Epithelial Cell Invasion Programs. Dev. Cell 2018, 47, 145. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, M.; Kashima, M.; Ito, T.; Watanabe, A.; Izumiyama, N.; Sano, M.; Kagaya, M.; Shioya, T.; Miura, M. Differential regulation of metalloproteinase production, proliferation and chemotaxis of human lung fibroblasts by PDGF, interleukin-1β and TNF-α. Mediat. Inflamm. 2000, 9, 155. [Google Scholar] [CrossRef]
- Sano, C.; Shimizu, T.; Tomioka, H. Effects of secretory leukocyte protease inhibitor on the tumor necrosis factor-alpha production and NF-κB activation of lipopolysaccharide-stimulated macrophages. Cytokine 2003, 21, 38. [Google Scholar] [CrossRef]
- Schultz, G.S.; Ladwig, G.; Wysocki, A. Extracellular matrix: Review of its roles in acute and chronic wounds. World Wide Wounds 2005, 2005, 1. [Google Scholar]
- Ferrara, N. Vascular endothelial growth factor: Basic science and clinical progress. Endocr. Rev. 2004, 25, 581. [Google Scholar] [CrossRef]
- Carmeliet, P. VEGF as a key mediator of angiogenesis in cancer. Oncology 2005, 69, 4. [Google Scholar] [CrossRef]
- Hochstein, A.; Bhatia, A. Collagen: Its role in wound healing. Wound Manag. 2014, 4, 104. [Google Scholar]
- Brett, D. A review of collagen and collagen-based wound dressings. Wounds 2008, 20, 347. [Google Scholar] [PubMed]
- Abdel-galil, F.M.; El-Jissry, M.A. Cyclopeptide alkaloids from Zizyphus spina-christi. Phytochemistry 1991, 30, 1348. [Google Scholar] [CrossRef]
- Tschesche, R.; Kaußmann, E.U.; Fehlhaber, H.W. Alkaloide aus Rhamnaceen, XIII. Amphibin-B,-C,-D and-E, vier Peptidalkaloide aus Ziziphus amphibia A. Cheval. Chem. Ber. 1972, 105, 3094. [Google Scholar] [CrossRef] [PubMed]
- Jossang, A.; Zahir, A.; Diakite, D. Mauritine J, a cyclopeptide alkaloid from Zizyphus mauritiana. Phytochemistry 1996, 42, 565. [Google Scholar] [CrossRef]
- Panseeta, P.; Lomchoey, K.; Prabpai, S.; Kongsaeree, P.; Suksamrarn, A.; Ruchirawat, S.; Suksamrarn, S. Antiplasmodial and antimycobacterial cyclopeptide alkaloids from the root of Ziziphus mauritiana. Phytochemistry 2011, 72, 909. [Google Scholar] [CrossRef]
- Tripathi, S. Ziziphus jujuba: A phytopharmacological review. Int. J. Res. Dev. Pharm. Life Sci. 2014, 3, 959–966. [Google Scholar]
- Alves, R.J.; Pinto, A.C.; da Costa, A.V.; Rezende, C.M. Zizyphus mauritiana Lam.(Rhamnaceae) and the chemical composition of its floral fecal odor. J. Braz. Chem. Soc. 2005, 16, 654–656. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, S.K.; Verma, S.; Singh, S.S.; Sammal, S.; Kumar, S. New aliphatic ester and alcohol from the leaves of Ziziphus mauritiana. Indian J. Chem.-Sect. B 2002, 41, 878–880. [Google Scholar]
- Goyal, M.; Nagori, B.P.; Sasmal, D. Review on ethnomedicinal uses, pharmacological activity and phytochemical constituents of Ziziphus mauritiana (Z. jujuba Lam., non Mill). Spatula DD 2012, 2, 107–116. [Google Scholar] [CrossRef]
- Tschesche, R.; Wilhelm, H.; Kaußmann, E.U.; Eckhardt, G. Alkaloide aus Rhamnaceen, XVII1) Mauritin-C,-D,-E und-F; neue Peptidalkaloide aus Ziziphus mauritiana Lam. Justus Liebigs Ann. Chem. 1974, 1974, 1694–1701. [Google Scholar] [CrossRef]
- Ji, C.-J.; Zeng, G.-Z.; Han, J.; He, W.-J.; Zhang, Y.-M.; Tan, N.-H. Zizimauritic acids A–C, three novel nortriterpenes from Ziziphus mauritiana. Bioorg. Med. Chem. Lett. 2012, 22, 6377–6380. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, R.; Chopda, M. Phyto-Pharmacology of Ziziphus jujuba Mill-A plant review. Pharmacogn. Rev. 2009, 3, 320. [Google Scholar]
- Zozio, S.; Servent, A.; Cazal, G.; Mbéguié-A-Mbéguié, D.; Ravion, S.; Pallet, D.; Abel, H. Changes in antioxidant activity during the ripening of jujube (Ziziphus mauritiana Lamk). Food Chem. 2014, 150, 448. [Google Scholar] [CrossRef]
- Mostafa, M.; Takano, F.; Ohta, T. P-54 New biflavonoyl ether glycosides from Zizyphus spina-christi: Symmetrical ether derived from two different flavonoids. Symp. Chem. Nat. Prod. Symp. Pap. 2010, 52, 631. [Google Scholar]
- Prashith, K.T.; Vinayaka, K.; Mallikarjun, N.; Bharath, A.C.; Shailendra, K.B.; Mc Rakesh, K.; Vinod, K.H. Antibacterial, Insecticidal and Free radical scavenging activity of methanol extract of Ziziphus rugosa Lam.(Rhamnaceae) fruit pericarp. Pharmacogn. J. 2011, 2, 65–69. [Google Scholar] [CrossRef]
- Filimonov, D.A.; Lagunin, A.A.; Gloriozova, T.A.; Rudik, A.V.; Druzhilovskii, D.S.; Pogodin, P.V.; Poroikov, V.V. Prediction of the biological activity spectra of organic compounds using the PASS online web resource. Chem. Heterocycl. Compd. 2014, 50, 444. [Google Scholar] [CrossRef]
- Hassan, H.; Abdel-Aziz, A. Evaluation of free radical-scavenging and anti-oxidant properties of black berry against fluoride toxicity in rats. Food Chem. Toxicol. 2010, 48, 1999–2004. [Google Scholar] [CrossRef]
- Srinivasan, R.; Chandrasekar, M.; Nanjan, M.J.; Suresh, B. Antioxidant activity of Caesalpinia digyna root. J. Ethnopharmacol. 2007, 113, 284–291. [Google Scholar] [CrossRef]
- Afzal, S.; Batool, M.; Ch, B.A.; Ahmad, A.; Uzair, M.; Afzal, K. Immunomodulatory, Cytotoxicity, and Antioxidant Activities of Roots of Ziziphus mauritiana. Pharmacogn. Mag. 2017, 13, S262–S265. [Google Scholar] [CrossRef] [Green Version]
- Yoshikawa, K.; Shimono, N.; Arihara, S. Antisweet Natural Products. VI. Jujubasaponins IV, V and Vi from Zizyphus jujuba mill. Chem. Pharm. Bull. 1992, 40, 2275–2278. [Google Scholar] [CrossRef] [Green Version]


| Sample | IC50 |
|---|---|
| Zizyphus mauritiana extract | 189.2 ± 1.6 µg/mL |
| Ascorbic acid | 194.2 ± 0.8 µg/mL |
| Sample | IC50 |
|---|---|
| Zizyphus mauritiana extract | 146.7 ± 2.1 µg/mL |
| Ascorbic acid | 154.4 ± 1.5 µg/mL |
| # | Molecule | Energy Score (kcal/mol) | RMSD (Å) |
|---|---|---|---|
| - | 2AZ5 Co-crystallized ligand | −5.5254 | 1.3787 |
| 1 | Amphibine H. | −6.6756 | 1.4343 |
| 2 | Amphibine B. | −6.7857 | 2.0384 |
| 3 | Amphibine D. | - | - |
| 4 | Amphibine E. | −5.9343 | 1.5565 |
| 5 | Amphibine E., N-DeMe or Mauritine J. | −6.8964 | 1.6562 |
| 6 | Amphibine F | - | - |
| 7 | Zizogenin or 2,6-Dihydroxyspirostane-3,12-dione | −5.7141 | 1.8788 |
| 8 | Frangufoline | −6.1900 | 1.8342 |
| 11 | Mauritine A. | - | - |
| 12 | Mauritine F. | - | - |
| 13 | Mauritine E. | −6.6522 | 1.7973 |
| 14 | Mauritine B. | −6.4077 | 1.4182 |
| 15 | Mauritine C. | - | - |
| 16 | Mauritine D. | - | - |
| 17 | Mauritine H. | −6.3098 | 1.6508 |
| 19 | Zizimauritic acid C. | −5.5227 | 1.7836 |
| 20 | Zizimauritic acid C., 21-Me ether | −5.2907 | 1.4763 |
| 21 | Franganine | −5.7895 | 1.6281 |
| 22 | Prodelphinidin B. or Gallocatechin(4α→8)gallocatechin | −4.9952 | 1.6455 |
| 23 | Sativanine A. | −5.9033 | 1.9644 |
| 25 | Zizyphine F. | −5.5143 | 2.0778 |
| # | Molecule | Energy Score (S; kcal/mol) | RMSD (Å) |
|---|---|---|---|
| - | 6B8Y co-crystallized ligand | −5.102 | 1.1231 |
| 1 | Amphibine H. | −6.6039 | 1.5284 |
| 2 | Amphibine B. | - | - |
| 3 | Amphibine D. | - | - |
| 4 | Amphibine E. | - | - |
| 5 | Amphibine E., N-DeMe or Mauritine J. | −4.2228 | 1.5592 |
| 6 | Amphibine F. | - | - |
| 7 | Zizogenin or 2,6-Dihydroxyspirostane-3,12-dione | −1.4649 | 1.5668 |
| 8 | Frangufoline | −4.3323 | 1.5238 |
| 11 | Mauritine A. | - | - |
| 12 | Mauritine F. | - | - |
| 13 | Mauritine E. | −1.8020 | 1.4485 |
| 14 | Mauritine B. | - | - |
| 15 | Mauritine C. | - | - |
| 16 | Mauritine D. | - | - |
| 17 | Mauritine H. | −8.0128 | 1.3190 |
| 19 | Zizimauritic acid C. | −3.9723 | 1.6145 |
| 20 | Zizimauritic acid C., 21-Me ether | - | - |
| 21 | Franganine | −5.6339 | 1.3131 |
| 22 | Prodelphinidin B. or Gallocatechin(4α→8)gallocatechin | −5.4270 | 1.2301 |
| 23 | Sativanine A. | −3.9078 | 1.3504 |
| 25 | Zizyphine F. | −4.9981 | 1.4643 |
| # | Molecule | Energy Score (S; kcal/mol) | RMSD (Å) |
|---|---|---|---|
| - | 6Y8M co-crystallized ligand | −4.2536 | 1.0950 |
| 1 | Amphibine H. | −5.2842 | 1.5363 |
| 2 | Amphibine B. | −4.8328 | 1.8557 |
| 3 | Amphibine D. | - | - |
| 4 | Amphibine E. | −4.9107 | 1.8266 |
| 5 | Amphibine E., N-DeMe or Mauritine J. | −5.4092 | 1.5702 |
| 6 | Amphibine F. | - | - |
| 7 | Zizogenin or 2,6-Dihydroxyspirostane-3,12-dione | −4.3238 | 1.7200 |
| 8 | Frangufoline | −5.3185 | 1.3011 |
| 11 | Mauritine A. | - | - |
| 12 | Mauritine F. | - | - |
| 13 | Mauritine E. | −4.2738 | 1.8927 |
| 14 | Mauritine B. | −5.6825 | 1.4982 |
| 15 | Mauritine C. | - | - |
| 16 | Mauritine D. | - | - |
| 17 | Mauritine H. | - | - |
| 19 | Zizimauritic acid C. | −4.3469 | 1.4960 |
| 20 | Zizimauritic acid C., 21-Me ether | −4.3448 | 1.7335 |
| 21 | Franganine | −4.6395 | 1.3794 |
| 22 | Prodelphinidin B. or Gallocatechin(4α→8)gallocatechin | −3.8771 | 1.2185 |
| 23 | Sativanine A. | - | - |
| 25 | Zizyphine F. | −4.6249 | 1.9028 |
| Active Site | Ligand | Binding Energy Score (S; kcal/mol) | Ligand—Active Site Interactions | ||
|---|---|---|---|---|---|
| a. a. Residue | Bond Type | Bond Length (Å) | |||
| TNFα (PDB ID: 2AZ5) | Co-crystallized ligand | −5.5254 | GLN 61 | H-donor | 2.97 |
| TYR 119 | H-pi | 4.08 | |||
| Amphibine B. | −6.7857 | TYR 59 | pi-H | 4.02 | |
| Sativanine A. | −5.9033 | TYR 119 | H-pi | 3.95 | |
| Zizyphine F. | −5.5143 | TYR 59 | pi-pi | 3.85 | |
| TGFBR1 kinase (PDB ID: 6B8Y) | Co-crystallized ligand | −5.102 | ASP 351 | H-donor | 2.72 |
| HIS 283 | H-acceptor | 2.89 | |||
| LYS 232 | pi-H | 3.94 | |||
| Amphibine H. | −6.6039 | SER 287 | H-acceptor | 3.07 | |
| Mauritine J. | −5.4092 | LYS 337 | H-donor | 3.12 | |
| Frangufoline | −4.3323 | ILE 211 | H-donor | 3.22 | |
| ASP 290 | H-donor | 3.07 | |||
| GLY 286 | H-acceptor | 3.43 | |||
| ILE 211 | pi-H | 4.35 | |||
| Mauritine E. | −1.8020 | ASP 351 | H-donor | 3.35 | |
| ASP 351 | H-donor | 2.88 | |||
| LYS 232 | H-acceptor | 3.00 | |||
| LYS 337 | pi-H | 3.89 | |||
| Zizimauritic acid C. | −3.9723 | ASP 290 | H-donor | 3.39 | |
| Franganine | −5.6339 | LYS 232 | H-acceptor | 2.70 | |
| Sativanine A. | −3.9078 | GLY 212 | H-acceptor | 3.22 | |
| Zizyphine F. | −4.9981 | ASP 290 | H-donor | 3.13 | |
| ASP290 | H-donor | 2.92 | |||
| Interleukin 1β (PDB ID: 6Y8M) | Co-crystallized ligand | −4.2536 | MET 148 | H-donor | 2.73 |
| MET 148 | H-acceptor | 2.94 | |||
| THR 147 | H-acceptor | 2.62 | |||
| GLN 149 | H-acceptor | 2.46 | |||
| Amphibine E. | −4.9107 | GLU 105 | pi-H | 4.25 | |
| Mauritine J. | −5.4092 | MET 148 | H-donor | 3.20 | |
| ARG 11 | H-acceptor | 2.97 | |||
| Zizogenin | −4.3238 | LYS 109 | H-acceptor | 3.09 | |
| Mauritine E. | −4.2738 | ARG 11 | pi-cation | 4.34 | |
| Mauritine B. | −5.6825 | GLN 149 | H-acceptor | 3.32 | |
| LYS 109 | pi-cation | 4.06 | |||
| Zizimauritic acid C. | −4.3469 | ASN 108 | H-donor | 2.81 | |
| Zizimauritic acid C., 21-Me ether | −4.3448 | GLN 149 | H-acceptor | 3.04 | |
| Prodelphinidin B. | −4.8631 | MET 148 | H-donor | 2.76 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shady, N.H.; Soltane, R.; Maher, S.A.; Saber, E.A.; Elrehany, M.A.; Mostafa, Y.A.; Sayed, A.M.; Abdelmohsen, U.R. Wound Healing and Antioxidant Capabilities of Zizyphus mauritiana Fruits: In-Vitro, In-Vivo, and Molecular Modeling Study. Plants 2022, 11, 1392. https://doi.org/10.3390/plants11111392
Shady NH, Soltane R, Maher SA, Saber EA, Elrehany MA, Mostafa YA, Sayed AM, Abdelmohsen UR. Wound Healing and Antioxidant Capabilities of Zizyphus mauritiana Fruits: In-Vitro, In-Vivo, and Molecular Modeling Study. Plants. 2022; 11(11):1392. https://doi.org/10.3390/plants11111392
Chicago/Turabian StyleShady, Nourhan Hisham, Raya Soltane, Sherif A. Maher, Entesar Ali Saber, Mahmoud A. Elrehany, Yaser A. Mostafa, Ahmed M. Sayed, and Usama Ramadan Abdelmohsen. 2022. "Wound Healing and Antioxidant Capabilities of Zizyphus mauritiana Fruits: In-Vitro, In-Vivo, and Molecular Modeling Study" Plants 11, no. 11: 1392. https://doi.org/10.3390/plants11111392
APA StyleShady, N. H., Soltane, R., Maher, S. A., Saber, E. A., Elrehany, M. A., Mostafa, Y. A., Sayed, A. M., & Abdelmohsen, U. R. (2022). Wound Healing and Antioxidant Capabilities of Zizyphus mauritiana Fruits: In-Vitro, In-Vivo, and Molecular Modeling Study. Plants, 11(11), 1392. https://doi.org/10.3390/plants11111392








