Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects
Abstract
1. Introduction
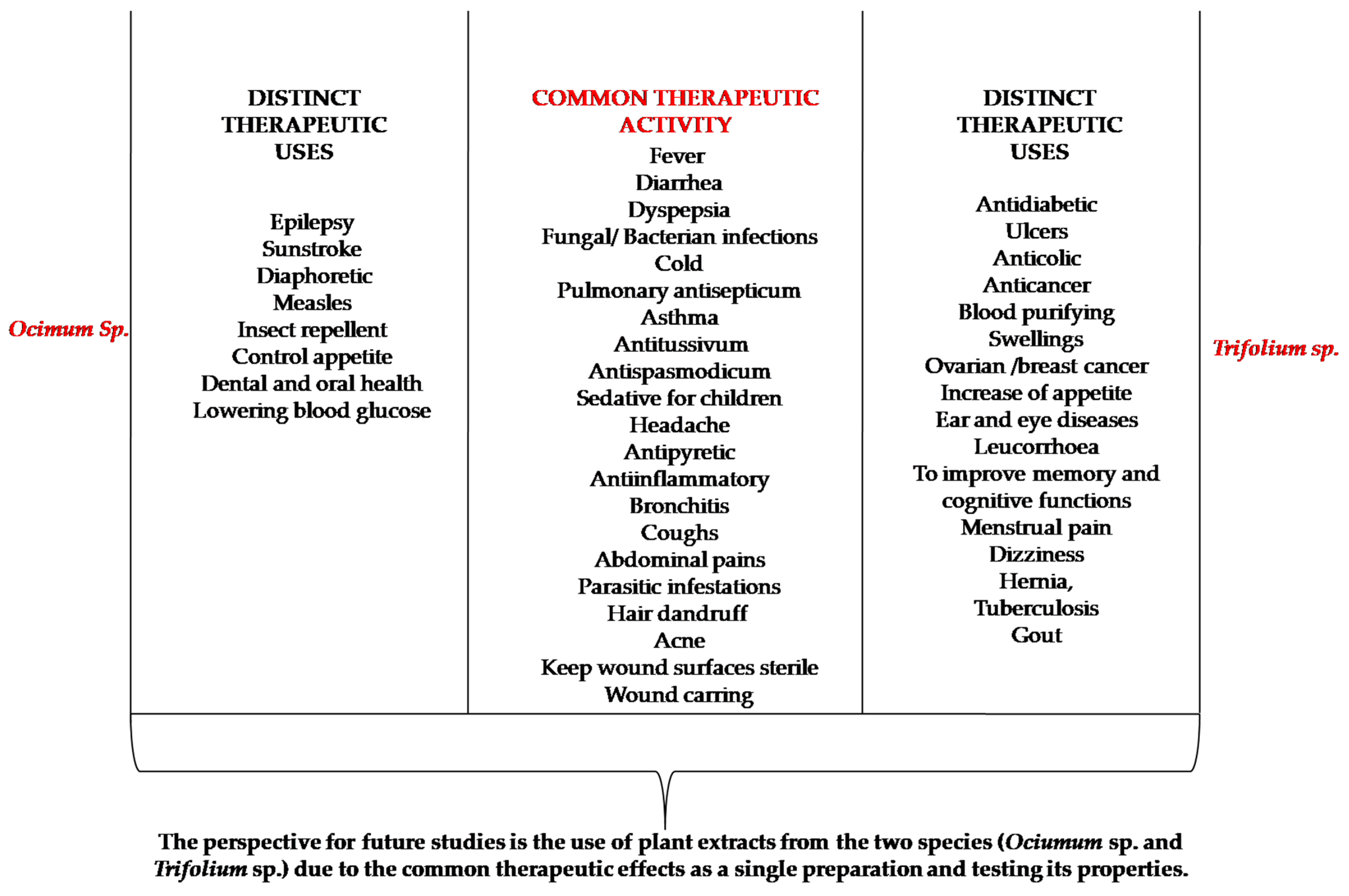
2. Common Therapeutic Activities of Ocimum and Trifolium Species
3. Phytochemistry
3.1. Phytochemical Profile of Ocimum Species
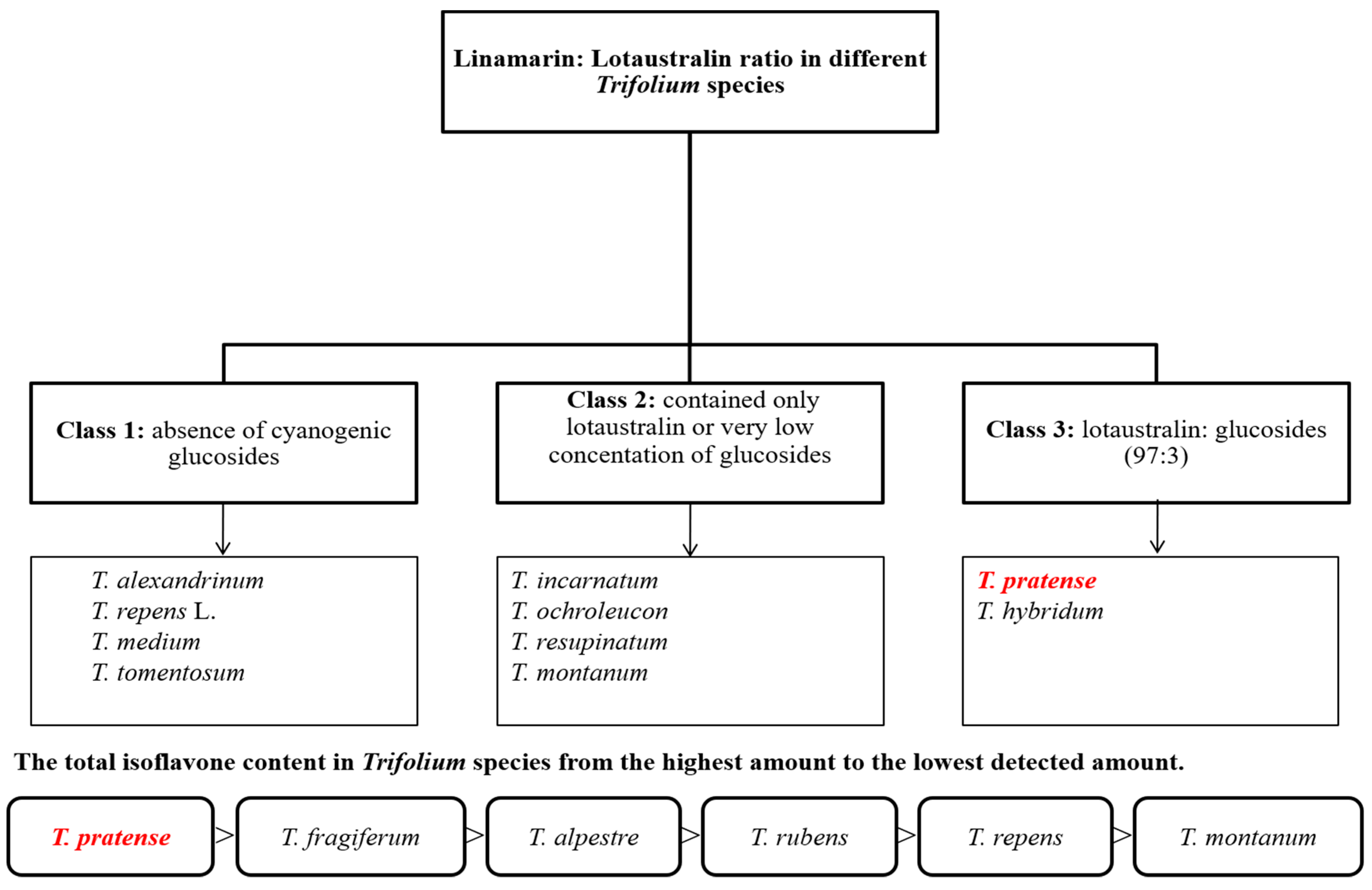
3.2. Phytochemical Profile of Trifolium Species
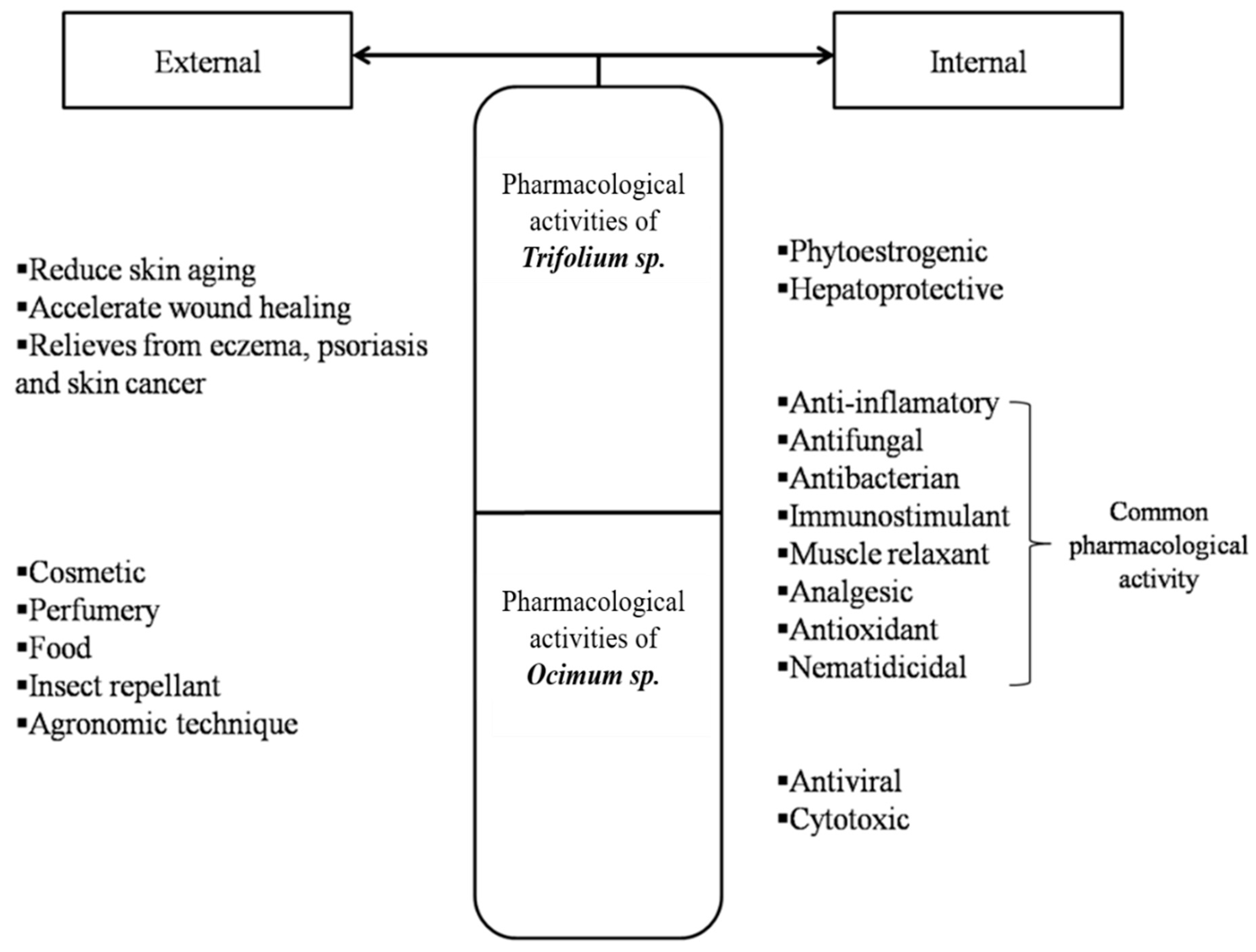
4. Pharmacological Activities
4.1. Antioxidant Capacity of Ocimum and Trifolium Species
4.2. Antimicrobial, Antiviral and Antifungal Activity of Ocimum and Trifolium Species
4.3. Anti-Inflammatory Effect of Ocimum Species
4.4. Estrogenic and Anticancer Activity of Trifolium Species
4.4.1. Estrogenic Action
4.4.2. Anticancer Activity
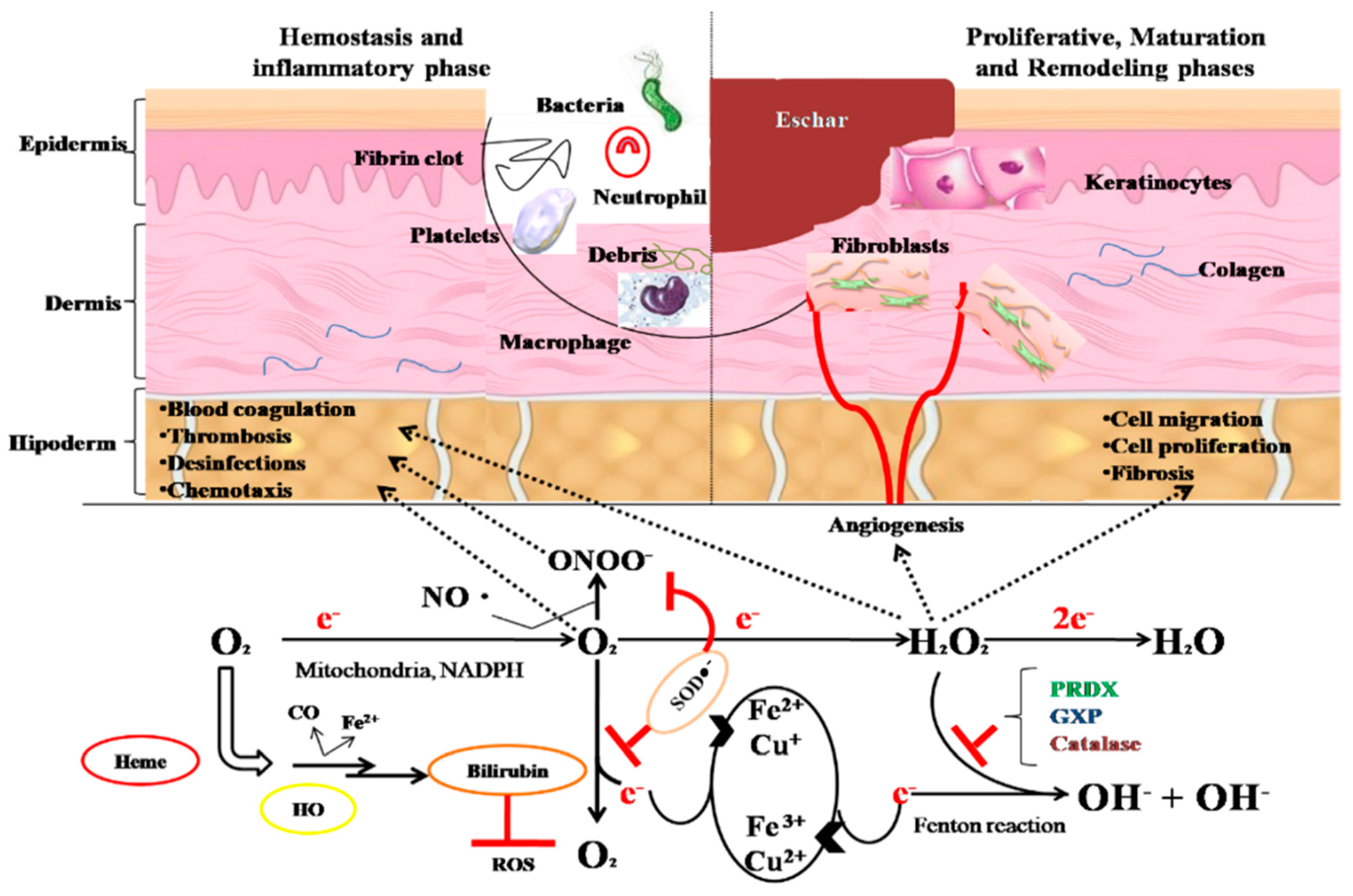
4.5. Dermal Pathology and Wound Healing Effects of Ocimum and Trifolium Species
5. Therapeutic Activities and Mechanisms of Action for Ocimum sp. and Trifolium sp. Depending on the Type of Extraction Performed
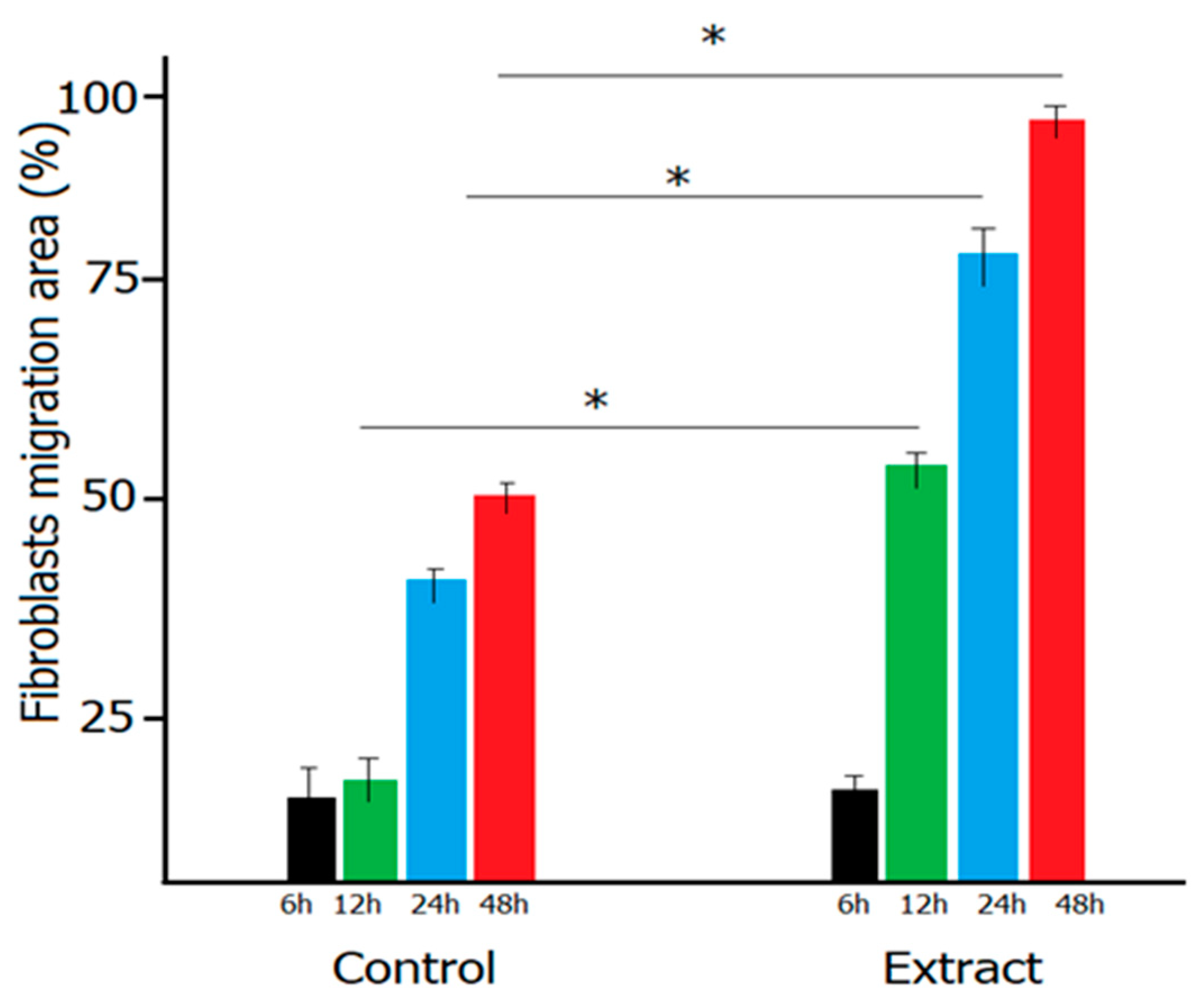
6. In Vitro Wound Healing Effect of the Mixture of Trifolium pratense and Ocimum basilicum Extracts
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tünde, J.; Vicas, L.; Tóth, I.; Braun, M.; Marian, E.; Teuşdea, A.; Vicaş, S.; Mureșan, M. Mineral Elements Profile, Bioactive Compounds and Antioxidant Capacity of Wild Blueberry and of Pharmaceutical Preparations from Blueberry (Vaccinium Myrtillus). Farmacia 2016, 64, 581–587. [Google Scholar]
- WHO|Global Status Report on Noncommunicable Diseases. 2014. Available online: http://www.who.int/nmh/publications/ncd-status-report-2014/en/ (accessed on 31 May 2021).
- Fajemiroye, J.O.; da Silva, D.M.; de Oliveira, D.R.; Costa, E.A. Treatment of Anxiety and Depression: Medicinal Plants in Retrospect. Fundam. Clin. Pharmacol. 2016, 30, 198–215. [Google Scholar] [CrossRef] [PubMed]
- Rezzoug, M.; Bakchiche, B.; Gherib, A.; Roberta, A.; Guido, F.; Kilinçarslan, Ö.; Mammadov, R.; Bardaweel, S.K. Chemical Composition and Bioactivity of Essential Oils and Ethanolic Extracts of Ocimum Basilicum L. and Thymus Algeriensis Boiss. & Reut. from the Algerian Saharan Atlas. BMC Complement. Altern. Med. 2019, 19, 146. [Google Scholar] [CrossRef]
- Egan, L.M.; Hofmann, R.W.; Seguin, P.; Ghamkhar, K.; Hoyos-Villegas, V. Pedigree Analysis of Pre-Breeding Efforts in Trifolium spp. Germplasm in New Zealand. BMC Genet. 2020, 21, 104. [Google Scholar] [CrossRef]
- De Araújo Couto, H.G.S.; Blank, A.F.; de Oliveira e Silva, A.M.; de Lima Nogueira, P.C.; de Fátima Arrigoni-Blank, M.; de Castro Nizio, D.A.; de Oliveira Pinto, J.A. Essential Oils of Basil Chemotypes: Major Compounds, Binary Mixtures, and Antioxidant Activity. Food Chem. 2019, 293, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Myers, S.P.; Vigar, V. Effects of a Standardised Extract of Trifolium Pratense (Promensil) at a Dosage of 80 mg in the Treatment of Menopausal Hot Flushes: A Systematic Review and Meta-Analysis. Phytomedicine 2017, 24, 141–147. [Google Scholar] [CrossRef]
- Darrah, H.H. The Cultivated Basils; Buckeye Print Co.: Buckeye, AZ, USA, 1980. [Google Scholar]
- Silva, M.G.V.; Vieira, I.G.P.; Mendes, F.N.P.; Albuquerque, I.L.; dos Santos, R.N.; Silva, F.O.; Morais, S.M. Variation of Ursolic Acid Content in Eight Ocimum Species from Northeastern Brazil. Molecules 2008, 13, 2482–2487. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Callahan, A.; Cantrell, C.L. Yield and Oil Composition of 38 Basil (Ocimum Basilicum L.) Accessions Grown in Mississippi. J. Agric. Food Chem. 2008, 56, 241–245. [Google Scholar] [CrossRef]
- Sestili, P.; Ismail, T.; Calcabrini, C.; Guescini, M.; Catanzaro, E.; Turrini, E.; Layla, A.; Akhtar, S.; Fimognari, C. The Potential Effects of Ocimum Basilicum on Health: A Review of Pharmacological and Toxicological Studies. Expert Opin. Drug Metab. Toxicol. 2018, 14, 679–692. [Google Scholar] [CrossRef]
- Akgül, A. Volatile Oil Composition of Sweet Basil (Ocimum Basilicum L.) Cultivating in Turkey (Short Communication). Food Nahr. 1989, 33, 87–88. [Google Scholar] [CrossRef]
- Simon, J.E.; Quinn, J.; Murray, R.G. Basil: A Source of Essential Oils. Advances in new crops. In Proceedings of the First National Symposium “New Crops: Research, Development, Economics”, Indianapolis, IN, USA, 23–26 October 1988; pp. 484–489. [Google Scholar]
- Lachowicz, K.J.; Jones, G.P.; Briggs, D.R.; Bienvenu, F.E.; Palmer, M.V.; Ting, S.S.T.; Hunter, M. Characteristics of Essential Oil from Basil (Ocimum Basilicum L.) Grown in Australia. J. Agric. Food Chem. 1996, 44, 877–881. [Google Scholar] [CrossRef]
- Handbook of Arabian Medicinal Plants. Available online: https://www.routledge.com/Handbook-of-Arabian-Medicinal-Plants/Ghazanfar/p/book/9780849305399 (accessed on 28 April 2021).
- Booth, N.L.; Overk, C.R.; Yao, P.; Burdette, J.E.; Nikolic, D.; Chen, S.-N.; Bolton, J.L.; van Breemen, R.B.; Pauli, G.F.; Farnsworth, N.R. The Chemical and Biological Profile of a Red Clover (Trifolium Pratense) Phase II Clinical Extract. J. Altern. Complement. Med. 2006, 12, 133–139. [Google Scholar] [CrossRef]
- Zohary, M.; Heller, D. The Genus Trifolium; Israel Academy of Sciences and Humanities: Jerusalem, Israel, 1984; ISBN 978-965-208-056-1. [Google Scholar]
- Shang, H.; Li, R.; Wu, H.; Sun, Z. Polysaccharides from Trifolium Repens L. Extracted by Different Methods and Extraction Condition Optimization. Sci. Rep. 2019, 9, 6353. [Google Scholar] [CrossRef]
- Sundararajan, B.; Moola, A.K.; Vivek, K.; Kumari, B.D.R. Formulation of Nanoemulsion from Leaves Essential Oil of Ocimum Basilicum L. and Its Antibacterial, Antioxidant and Larvicidal Activities (Culex Quinquefasciatus). Microb. Pathog. 2018, 125, 475–485. [Google Scholar] [CrossRef]
- Makri, O.; Kintzios, S. Ocimum sp. (Basil): Botany, Cultivation, Pharmaceutical Properties, and Biotechnology. J. Herbs Spices Med. Plants 2008, 13, 123–150. [Google Scholar] [CrossRef]
- Flanigan, P.M.; Niemeyer, E.D. Effect of Cultivar on Phenolic Levels, Anthocyanin Composition, and Antioxidant Properties in Purple Basil (Ocimum Basilicum L.). Food Chem. 2014, 164, 518–526. [Google Scholar] [CrossRef]
- Teofilović, B.; Grujić-Letić, N.; Goločorbin-Kon, S.; Stojanović, S.; Vastag, G.; Gadžurić, S. Experimental and Chemometric Study of Antioxidant Capacity of Basil (Ocimum Basilicum) Extracts. Ind. Crops Prod. 2017, 100, 176–182. [Google Scholar] [CrossRef]
- Antonescu, A.I.; Jurca, T.; Gligor, F.; Craciun, I.; Fritea, L.; Patay, E.B.; Muresan, M.; Udeanu, D.I.; Ioniță, C.A.; Antonescu, A.; et al. Comparative phytochemical and antioxidative characterization of Trifolium pratense L. and Ocimum basilicum L. Farmacia 2019, 67. [Google Scholar] [CrossRef]
- Benedec, D.; Pârvu, A.E.; Oniga, I.; Toiu, A.; Tiperciuc, B. Effects of Ocimum Basilicum L. Extract on Experimental Acute Inflammation. Rev. Med. Chir. Soc. Med. Nat. Iasi 2007, 111, 1065–1069. [Google Scholar] [PubMed]
- Dhama, K.; Sharun, K.; Gugjoo, M.B.; Tiwari, R.; Alagawany, M.; Yatoo, M.I.; Thakur, P.; Iqbal, H.M.N.; Chaicumpa, W.; Michalak, I.; et al. A Comprehensive Review on Chemical Profile and Pharmacological Activities of Ocimum Basilicum. Food Rev. Int. 2021. [Google Scholar] [CrossRef]
- Kolodziejczyk-Czepas, J. Trifolium Species-Derived Substances and Extracts—Biological Activity and Prospects for Medicinal Applications. J. Ethnopharmacol. 2012, 143, 14–23. [Google Scholar] [CrossRef]
- Muzashvili, T.; Moniuszko-Szajwaj, B.; Pecio, L.; Oleszek, W.; Stochmal, A. Ultraperformance Liquid Chromatography Tandem Mass Spectrometry Determination of Cyanogenic Glucosides in Trifolium Species. J. Agric. Food Chem. 2014, 62, 1777–1782. [Google Scholar] [CrossRef]
- Kolodziejczyk-Czepas, J. Trifolium Species—The Latest Findings on Chemical Profile, Ethnomedicinal Use and Pharmacological Properties. J. Pharm. Pharmacol. 2016, 68, 845–861. [Google Scholar] [CrossRef]
- Ahmad, S.; Zeb, A. Phytochemical Profile and Pharmacological Properties of Trifolium Repens. J. Basic Clin. Physiol. Pharmacol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dabkevičienė, G.; Butkutė, B.; Lemežienė, N.; Jakštas, V.; Vilčinskas, E.; Janulis, V. Distribution of Formononetin, Daidzein and Genistein in Trifolium Species and Their Aerial Plant Parts. Chemija 2012, 23, 306–311. [Google Scholar]
- Yokoyama, S.-I.; Kodera, M.; Hirai, A.; Nakada, M.; Ueno, Y.; Osawa, T. Red Clover (Trifolium Pratense L.) Sprout Prevents Metabolic Syndrome. J. Nutr. Sci. Vitaminol. 2020, 66, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Zgonc Škulj, A.; Poljšak, N.; Kočevar Glavač, N.; Kreft, S. Herbal Preparations for the Treatment of Hair Loss. Arch. Dermatol. Res. 2020, 312, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Kubes, J.; Skalicky, M.; Tumova, L.; Martin, J.; Hejnak, V.; Martinkova, J. Vanadium Elicitation of Trifolium Pratense L. Cell Culture and Possible Pathways of Produced Isoflavones Transport across the Plasma Membrane. Plant Cell Rep. 2019, 38, 657–671. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Xu, B.; Yu, Z.; He, Q.; Hu, Z.; Zhou, S.; Chen, M.; Zhu, L. Trifolium Flavonoids Overcome Gefitinib Resistance of Non-Small-Cell Lung Cancer Cell by Suppressing ERK and STAT3 Signaling Pathways. Biomed. Res. Int. 2020, 2020, 2491304. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Ino, Y.; Hirai, A.; Okamura, A.; Ishikawa, H.; Yokoyama, S.-I.; Osawa, T. Effects of Isoflavone-Rich Red Clover Extract on Blood Glucose Level: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Food Sci. 2021, 86, 1393–1399. [Google Scholar] [CrossRef]
- Lee, J.; Scagel, C.F. Chicoric Acid Levels in Commercial Basil (Ocimum Basilicum) and Echinacea Purpurea Products. J. Funct. Foods 2010, 2, 77–84. [Google Scholar] [CrossRef]
- Politeo, O.; Jukic, M.; Milos, M. Chemical Composition and Antioxidant Capacity of Free Volatile Aglycones from Basil (Ocimum Basilicum L.) Compared with Its Essential Oil. Food Chem. 2007, 101, 379–385. [Google Scholar] [CrossRef]
- Fritea, L.; Banica, F.; Costea, T.O.; Moldovan, L.; Iovan, C.; Cavalu, S. A gold nanoparticles—Graphene based electrochemical sensor for sensitive determination of nitrazepam. J. Electroanal. Chem. 2018, 830–831, 63–71. [Google Scholar] [CrossRef]
- Miere, F.; Teusdea, A.C.; Laslo, V.; Fritea, L.; Moldovan, L.; Costea, T.; Uivarosan, D.; Vicas, S.I.; Pallag, A. Natural Polymeric Beads for Encapsulation of Stellaria Media Extract with Antioxidant Properties. Mater. Plast. 2019, 56, 671–679. [Google Scholar] [CrossRef]
- Mastaneh, M.; Ahamd, M.; Taher, N.; Mehrdad, H. Antioxidant Effect of Purple Basil (Lamiaceae) Phenolics. Orient. J. Chem. 2014, 30, 1965–1969. [Google Scholar] [CrossRef]
- Dobjanschi, L.; Luminita, F.; Patay, E.; Tamas, M. Comparative Study of the Morphological and Phytochemical Characterization of Romanian Solidago Species. Pak. J. Pharm. Sci. 2019, 32, 1571–1579. [Google Scholar]
- Miere, F.; Fritea, L.; Cavalu, S.; Vicaș, S.I. Formulation, characterization, and advantages of using liposomes in multiple therapies. Pharmacophore 2020, 11, 1–12. [Google Scholar]
- Kwee, E.M.; Niemeyer, E.D. Variations in Phenolic Composition and Antioxidant Properties among 15 Basil (Ocimum Basilicum L.) Cultivars. Food Chem. 2011, 128, 1044–1050. [Google Scholar] [CrossRef]
- Oza, M.J.; Kulkarni, Y.A. Trifolium Pratense (Red Clover) Improve SIRT1 Expression and Glycogen Content in High Fat Diet-Streptozotocin Induced Type 2 Diabetes in Rats. Chem. Biodivers. 2020, 17, e2000019. [Google Scholar] [CrossRef] [PubMed]
- Bahcesular, B.; Yildirim, E.D.; Karaçocuk, M.; Kulak, M.; Karaman, S. Seed Priming with Melatonin Effects on Growth, Essential Oil Compounds and Antioxidant Activity of Basil (Ocimum Basilicum L.) under Salinity Stress. Ind. Crops Prod. 2020, 146, 112165. [Google Scholar] [CrossRef]
- Miere, F.; Vicas, S.I.; Timar, A.V.; Ganea, M.; Zdrinca, M.; Cavalu, S.; Fritea, L.; Vicas, L.; Muresan, M.; Pallag, A.; et al. Preparation and Characterization of Two Different Liposomal Formulations with Bioactive Natural Extract for Multiple Applications. Processes 2021, 9, 432. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological Effects of Essential Oils—A Review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, I.; Elmastaş, M.; Aboul-Enein, H.Y. Determination of Antioxidant and Radical Scavenging Activity of Basil (Ocimum Basilicum L. Family Lamiaceae) Assayed by Different Methodologies. Phytother. Res. 2007, 21, 354–361. [Google Scholar] [CrossRef]
- Hinneburg, I.; Damien Dorman, H.J.; Hiltunen, R. Antioxidant Activities of Extracts from Selected Culinary Herbs and Spices. Food Chem. 2006, 97, 122–129. [Google Scholar] [CrossRef]
- Pichersky, E.; Gershenzon, J. The Formation and Function of Plant Volatiles: Perfumes for Pollinator Attraction and Defense. Curr. Opin. Plant Biol. 2002, 5, 237–243. [Google Scholar] [CrossRef]
- Salas, J.J.; Sánchez, C.; García-González, D.L.; Aparicio, R. Impact of the Suppression of Lipoxygenase and Hydroperoxide Lyase on the Quality of the Green Odor in Green Leaves. J. Agric. Food Chem. 2005, 53, 1648–1655. [Google Scholar] [CrossRef]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of Plant Volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef]
- Vuorinen, T.; Reddy, G.V.P.; Nerg, A.-M.; Holopainen, J.K. Monoterpene and Herbivore-Induced Emissions from Cabbage Plants Grown at Elevated Atmospheric CO2 Concentration. Atmos. Environ. 2004, 38, 675–682. [Google Scholar] [CrossRef]
- Vianna, E.; Ebeler, S.E. Monitoring Ester Formation in Grape Juice Fermentations Using Solid Phase Microextraction Coupled with Gas Chromatography-Mass Spectrometry. J. Agric. Food Chem. 2001, 49, 589–595. [Google Scholar] [CrossRef]
- Pedraza-Chaverri, J.; Cárdenas-Rodríguez, N.; Orozco-Ibarra, M.; Pérez-Rojas, J.M. Medicinal Properties of Mangosteen (Garcinia Mangostana). Food Chem. Toxicol. 2008, 46, 3227–3239. [Google Scholar] [CrossRef]
- Jurca, T.; Baldea, I.; Filip, G.A.; Olteanu, D.; Clichici, S.; Pallag, A.; Vicas, L.; Marian, E.; Micle, O.; Muresan, M. The Effect of Tropaeolum Majus L. on Bacterial Infections and in Vitro Efficacy on Apoptosis and DNA Lesions in Hyperosmotic Stress. J. Physiol. Pharmacol. 2018, 69. [Google Scholar] [CrossRef]
- Vlaisavljevic, S.; Kaurinovic, B.; Popovic, M.; Djurendic-Brenesel, M.; Vasiljevic, B.; Cvetkovic, D.; Vasiljevic, S. Trifolium Pratense L. as a Potential Natural Antioxidant. Molecules 2014, 19, 713–725. [Google Scholar] [CrossRef]
- Reis, A.; Boutet-Mercey, S.; Massot, S.; Ratet, P.; Zuanazzi, J.A.S. Isoflavone Production in Hairy Root Cultures and Plantlets of Trifolium Pratense. Biotechnol. Lett. 2019, 41, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, P. Skin Cancer and Role of Herbal Medicines. Asian J. Pharm. Pharmacol. 2018, 4, 404–412. [Google Scholar] [CrossRef]
- Nacar, S.; Tansi, S. Chemical Components of Different Basil (Ocimum Basilicum L.) Cultivars Grown in Mediterranean Regions in Turkey. Isr. J. Plant Sci. 2000, 48, 109–112. [Google Scholar] [CrossRef]
- Al-Maskri, A.Y.; Hanif, M.A.; Al-Maskari, M.Y.; Abraham, A.S.; Al-Sabahi, J.N.; Al-Mantheri, O. Essential Oil from Ocimum Basilicum (Omani Basil): A Desert Crop. Nat. Prod. Commun. 2011, 6, 1487–1490. [Google Scholar] [PubMed]
- Politeo, O.; Jukic, M.; Milos, M. Comparison of Chemical Composition and Antioxidant Activity of Glycosidically Bound and Free Volatiles from Clove (Eugenia Caryophyllata Thunb.). J. Food Biochem. 2010, 34, 129–141. [Google Scholar] [CrossRef]
- Sullivan, M.L.; Quesenberry, K.H. Clover, Red (Trifolium Pratense). Methods Mol. Biol. 2015, 1223, 237–254. [Google Scholar] [CrossRef] [PubMed]
- Mouradov, A.; Panter, S.; Labandera, M.; Ludlow, E.; Emmerling, M.; Spangenberg, G. Clovers (Trifolium spp.). Methods Mol. Biol. 2006, 343, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, H.; Papadopoulou, C. Antimicrobial Activity of Basil, Oregano, and Thyme Essential Oils. J. Microbiol. Biotechnol. 2017, 27, 429–438. [Google Scholar] [CrossRef]
- Araújo Silva, V.; Pereira da Sousa, J.; de Luna Freire Pessôa, H.; Fernanda Ramos de Freitas, A.; Douglas Melo Coutinho, H.; Beuttenmuller Nogueira Alves, L.; Oliveira Lima, E. Ocimum Basilicum: Antibacterial Activity and Association Study with Antibiotics against Bacteria of Clinical Importance. Pharm. Biol. 2016, 54, 863–867. [Google Scholar] [CrossRef]
- Chiang, L.-C.; Ng, L.-T.; Cheng, P.-W.; Chiang, W.; Lin, C.-C. Antiviral Activities of Extracts and Selected Pure Constituents of Ocimum Basilicum. Clin. Exp. Pharmacol. Physiol. 2005, 32, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Zahran, E.; Abdelmohsen, U.; Khalil, H.; Desoukey, S.; Ahmed, M.; Kamel, M. Diversity, Phytochemical and Medicinal Potential of the Genus Ocimum L. (Lamiaceae). Phytochem. Rev. 2020, 19. [Google Scholar] [CrossRef]
- Loing, E.; Lachance, R.; Ollier, V.; Hocquaux, M. A New Strategy to Modulate Alopecia Using a Combination of Two Specific and Unique Ingredients. J. Cosmet. Sci. 2013, 64, 45–58. [Google Scholar]
- Ghitea, T.C.; El-Kharoubi, A.; Ganea, M.; Bimbo-Szuhai, E.; Nemeth, T.S.; Ciavoi, G.; Foghis, M.; Dobjanschi, L.; Pallag, A.; Micle, O. The Antimicrobial Activity of Origanum Vulgare L. Correlated with the Gastrointestinal Perturbation in Patients with Metabolic Syndrome. Molecules 2021, 26, 283. [Google Scholar] [CrossRef] [PubMed]
- Marian, E.; Vicas, L.G.; Jurca, T.; Muresan, M.; Pallag, A.; Stan, R.L.; Sevastre, B.; Diaconeasa, Z.; Ionescu, C.M.L.; Hangan, A.C. Salivia Officinalis L. and Verbascum Phlomoides L. Chemical, Antimicrobial, Antioxidant and Antitumor Investigations. Rev. Chim. 2018, 69, 365–370. [Google Scholar] [CrossRef]
- Harlow, B.E.; Flythe, M.D.; Kagan, I.A.; Goodman, J.P.; Klotz, J.L.; Aiken, G.E. Isoflavone Supplementation, via Red Clover Hay, Alters the Rumen Microbial Community and Promotes Weight Gain of Steers Grazing Mixed Grass Pastures. PLoS ONE 2020, 15, e0229200. [Google Scholar] [CrossRef]
- Singh, S.; Majumdar, D.K. Evaluation of Antiinflammatory Activity of Fatty Acids of Ocimum Sanctum Fixed Oil. Indian J. Exp. Biol. 1997, 35, 380–383. [Google Scholar]
- Singh, S.; Taneja, M.; Majumdar, D.K. Biological Activities of Ocimum Sanctum L. Fixed Oil—An Overview. Indian J. Exp. Biol. 2007, 45, 403–412. [Google Scholar]
- Okoye, F.B.C.; Obonga, W.O.; Onyegbule, F.A.; Ndu, O.O.; Ihekwereme, C.P. Chemical composition and anti-inflammatory activity of essential oils from the leaves of Ocimum basilicum L. and Ocimum gratissimum L. (Lamiaceae). Int. J. Pharm. Sci. Res. 2014, 5, 2174–2180. [Google Scholar]
- Mediratta, P.K.; Sharma, K.K.; Singh, S. Evaluation of Immunomodulatory Potential of Ocimum Sanctum Seed Oil and Its Possible Mechanism of Action. J. Ethnopharmacol. 2002, 80, 15–20. [Google Scholar] [CrossRef]
- Maul, R.; Kulling, S.E. Absorption of Red Clover Isoflavones in Human Subjects: Results from a Pilot Study. Br. J. Nutr. 2010, 103, 1569–1572. [Google Scholar] [CrossRef]
- Lutter, S.; Schmalbach, K.; Esch, H.L.; Lehmann, L. The Isoflavone Irilone Contributes to the Estrogenic Potential of Dietary Supplements Containing Red Clover. Arch. Toxicol. 2014, 88, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, P.; Rasini, E.; Luini, A.; Legnaro, M.; Luzzani, M.; Casareto, E.; Carreri, M.; Paracchini, S.; Marino, F.; Cosentino, M. Isoflavone Content and Estrogenic Activity of Different Batches of Red Clover (Trifolium Pratense L.) Extracts: An in Vitro Study in MCF-7 Cells. Fitoterapia 2014, 94, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Finley, J.W. Proposed Criteria for Assessing the Efficacy of Cancer Reduction by Plant Foods Enriched in Carotenoids, Glucosinolates, Polyphenols and Selenocompounds. Ann. Bot. 2005, 95, 1075–1096. [Google Scholar] [CrossRef] [PubMed]
- Karakaş, F.; Yildirim, A.; Bayram, R.; Yavuz, M.; Gepdiremen, A.; Turker, A. Antiproliferative Activity of Some Medicinal Plants on Human Breast and Hepatocellular Carcinoma Cell Lines and Their Phenolic Contents. Trop. J. Pharm. Res. 2015, 14, 1787. [Google Scholar] [CrossRef]
- Orafidiya, L.O.; Agbani, E.O.; Oyedele, A.O.; Babalola, O.O.; Onayemi, O. Preliminary Clinical Tests on Topical Preparations of Ocimum Gratissimum Linn Leaf Essential Oil for the Treatment of Acne Vulgaris. Clin. Drug Investig. 2002. [Google Scholar] [CrossRef]
- Orafidiya, L.O.; Agbani, E.O.; Adelusola, K.A.; Iwalewa, E.O.; Adebanji, O.A.; Adediran, E.A.F.; Agbani, N.T. A Study on the Effect of the Leaf Essential Oil of Ocimum Gratissimum Linn. on Cyclophosphamide-Induced Hair Loss. Int. J. Aromather. 2004, 14, 119–128. [Google Scholar] [CrossRef]
- Pansang, S.; Maphanta, S.; Tuntijarukorn, P.; Viyoch, J. Skin Irritation Test of a Microemulsion Containing Essential Oil Isolated from Ocimum Basilicum. ScienceAsia 2010, 36, 355–358. [Google Scholar] [CrossRef]
- Renda, G.; Yalçın, F.N.; Nemutlu, E.; Akkol, E.K.; Süntar, I.; Keleş, H.; Ina, H.; Çalış, I.; Ersöz, T. Comparative Assessment of Dermal Wound Healing Potentials of Various Trifolium L. Extracts and Determination of Their Isoflavone Contents as Potential Active Ingredients. J. Ethnopharmacol. 2013, 148, 423–432. [Google Scholar] [CrossRef]
- Lipovac, M.; Chedraui, P.; Gruenhut, C.; Gocan, A.; Kurz, C.; Neuber, B.; Imhof, M. Effect of Red Clover Isoflavones over Skin, Appendages, and Mucosal Status in Postmenopausal Women. Obstet. Gynecol. Int. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Dweck, A.C. The Internal and External Use of Medicinal Plants. Clin. Dermatol. 2009, 27, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; An, X.; Wang, S.; Sun, M.; Zhou, H. Basil Polysaccharides: A Review on Extraction, Bioactivities and Pharmacological Applications. Bioorg. Med. Chem. 2020, 28, 115179. [Google Scholar] [CrossRef]
- Shahrajabian, M.H.; Sun, W.; Cheng, Q. Chemical Components and Pharmacological Benefits of Basil (Ocimum Basilicum): A Review. Int. J. Food Prop. 2020, 23, 1961–1970. [Google Scholar] [CrossRef]
- Hussain, A.I.; Anwar, F.; Hussain Sherazi, S.T.; Przybylski, R. Chemical Composition, Antioxidant and Antimicrobial Activities of Basil (Ocimum Basilicum) Essential Oils Depends on Seasonal Variations. Food Chem. 2008, 108, 986–995. [Google Scholar] [CrossRef]
- Goel, A.; Kumar, S.; Singh, D.K.; Bhatia, A.K. Wound Healing Potential of Ocimum Sanctum Linn. with Induction of Tumor Necrosis Factor-Alpha. Indian J. Exp. Biol. 2010, 48, 402–406. [Google Scholar] [PubMed]
- Manosroi, J.; Dhumtanom, P.; Manosroi, A. Anti-Proliferative Activity of Essential Oil Extracted from Thai Medicinal Plants on KB and P388 Cell Lines. Cancer Lett. 2006, 235, 114–120. [Google Scholar] [CrossRef]
- Huang, X.; Sun, J.; Chen, G.; Niu, C.; Wang, Y.; Zhao, C.; Sun, J.; Huang, H.; Huang, S.; Liang, Y.; et al. Resveratrol Promotes Diabetic Wound Healing via SIRT1-FOXO1-c-Myc Signaling Pathway-Mediated Angiogenesis. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef]
- Saha, S.; Mukhopadhyay, M.K.; Ghosh, P.D.; Nath, D. Effect of Methanolic Leaf Extract of Ocimum Basilicum L. on Benzene-Induced Hematotoxicity in Mice. Evid. Based Complement. Altern. Med. 2012, 2012, e176385. [Google Scholar] [CrossRef]
- Monga, J.; Sharma, M.; Tailor, N.; Ganesh, N. Antimelanoma and Radioprotective Activity of Alcoholic Aqueous Extract of Different Species of Ocimum in C(57)BL Mice. Pharm. Biol. 2011, 49, 428–436. [Google Scholar] [CrossRef]
- Rastogi, S.; Shukla, Y.; Paul, B.N.; Chowdhuri, D.K.; Khanna, S.K.; Das, M. Protective Effect of Ocimum Sanctum on 3-Methylcholanthrene, 7,12-Dimethylbenz(a)Anthracene and Aflatoxin B1 Induced Skin Tumorigenesis in Mice. Toxicol. Appl. Pharmacol. 2007, 224, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Bravo, E.; Amrani, S.; Aziz, M.; Harnafi, H.; Napolitano, M. Ocimum Basilicum Ethanolic Extract Decreases Cholesterol Synthesis and Lipid Accumulation in Human Macrophages. Fitoterapia 2008, 79, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, P.; Rai, V.; Singh, R.B. Randomized Placebo-Controlled, Single Blind Trial of Holy Basil Leaves in Patients with Noninsulin-Dependent Diabetes Mellitus. Int. J. Clin. Pharmacol. Ther. 1996, 34, 406–409. [Google Scholar] [PubMed]
- Amrani, S.; Harnafi, H.; Gadi, D.; Mekhfi, H.; Legssyer, A.; Aziz, M.; Martin-Nizard, F.; Bosca, L. Vasorelaxant and Anti-Platelet Aggregation Effects of Aqueous Ocimum Basilicum Extract. J. Ethnopharmacol. 2009, 125, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Alipour, G.; Dashti, S.; Hosseinzadeh, H. Review of Pharmacological Effects of Myrtus Communis L. and Its Active Constituents. Phytother. Res. 2014, 28, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Tchimene, M.K.; Okoli, C.O.; Iwu, M.M. Antidiabetic Property of Some Nigerian Medicinal Plants. J. Med. Plants Res. 2016, 10, 139–148. [Google Scholar] [CrossRef][Green Version]
- Jain, R.; Aqil, M.; Ahad, A.; Ali, A.; Khar, R.K. Basil Oil Is a Promising Skin Penetration Enhancer for Transdermal Delivery of Labetolol Hydrochloride. Drug Dev. Ind. Pharm. 2008, 34, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Hozayen, W.G.; El-Desouky, M.A.; Soliman, H.A.; Ahmed, R.R.; Khaliefa, A.K. Antiosteoporotic Effect of Petroselinum Crispum, Ocimum Basilicum and Cichorium Intybus L. in Glucocorticoid-Induced Osteoporosis in Rats. BMC Complement. Altern. Med. 2016, 16, 165. [Google Scholar] [CrossRef]
- Nweze, E.I.; Eze, E.E. Justification for the Use of Ocimum Gratissimum L. in Herbal Medicine and Its Interaction with Disc Antibiotics. BMC Complement. Altern. Med. 2009, 9, 37. [Google Scholar] [CrossRef]
- Viyoch, J.; Pisutthanan, N.; Faikreua, A.; Nupangta, K.; Wangtorpol, K.; Ngokkuen, J. Evaluation of in Vitro Antimicrobial Activity of Thai Basil Oils and Their Micro-Emulsion Formulas against Propionibacterium Acnes. Int. J. Cosmet. Sci. 2006, 28, 125–133. [Google Scholar] [CrossRef]
- Rashidian, A.; Roohi, P.; Mehrzadi, S.; Ghannadi, A.R.; Minaiyan, M. Protective Effect of Ocimum Basilicum Essential Oil Against Acetic Acid–Induced Colitis in Rats. J. Evid. Based Complement. Altern. Med. 2016, 21, NP36–NP42. [Google Scholar] [CrossRef] [PubMed]
- Taie, H.A.A.; Salama, Z.A.E.-R.; Samir, R. Potential Activity of Basil Plants as a Source of Antioxidants and Anticancer Agents as Affected by Organic and Bio-Organic Fertilization. Not. Bot. Horti Agrobot. Cluj Napoca 2010, 38, 119–127. [Google Scholar] [CrossRef]
- Nguyen, P.M.; Niemeyer, E.D. Effects of Nitrogen Fertilization on the Phenolic Composition and Antioxidant Properties of Basil (Ocimum Basilicum L.). J. Agric. Food Chem. 2008, 56, 8685–8691. [Google Scholar] [CrossRef] [PubMed]
- Chokechaijaroenporn, O.; Bunyapraphatsara, N.; Kongchuensin, S. Mosquito Repellent Activities of Ocimum Volatile Oils. Phytomedicine 1994, 1, 135–139. [Google Scholar] [CrossRef]
- Ueda-Nakamura, T.; Mendonça-Filho, R.R.; Morgado-Díaz, J.A.; Korehisa Maza, P.; Prado Dias Filho, B.; Aparício Garcia Cortez, D.; Alviano, D.S.; do Socorro, M.S.R.; Lopes, A.H.C.S.; Alviano, C.S.; et al. Antileishmanial Activity of Eugenol-Rich Essential Oil from Ocimum Gratissimum. Parasitol. Int. 2006, 55, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Wagay, N. Medicinal Flora and Ethno-Botanical Knowledge of Baramulla Tehsil in Jammu and Kashmir, India. Int. J. Adv. Biotechnol. Res. 2014, 5, 539–546. [Google Scholar]
- Chen, T.; Zhong, F.-J.; Hong, Y.-M.; Su, W.-J.; Zhuang, L.-L.; Qiu, L.-X. Effect of Trifolium Pratense Extract on Methionine-Choline-Deficient Diet-Induced Steatohepatitis in C57BL/6 Mice. Chin. J. Nat. Med. 2014, 12, 194–198. [Google Scholar] [CrossRef]
- Tundis, R.; Marrelli, M.; Conforti, F.; Tenuta, M.; Bonesi, M.; Menichini, F.; Loizzo, M. Trifolium Pratense and T. Repens (Leguminosae): Edible Flower Extracts as Functional Ingredients. Foods 2015, 4, 338–348. [Google Scholar] [CrossRef]
- Jung, E.H.; Kim, S.R.; Hwang, I.K.; Ha, T.Y. Hypoglycemic Effects of a Phenolic Acid Fraction of Rice Bran and Ferulic Acid in C57BL/KsJ-Db/Db Mice. J. Agric. Food Chem. 2007, 55, 9800–9804. [Google Scholar] [CrossRef]
- Circosta, C.; De Pasquale, R.; Palumbo, D.R.; Samperi, S.; Occhiuto, F. Effects of Isoflavones from Red Clover (Trifolium Pratense) on Skin Changes Induced by Ovariectomy in Rats. Phytother. Res. 2006, 20, 1096–1099. [Google Scholar] [CrossRef] [PubMed]
- Ijaz, F.; Iqbal, Z.; Alam, J.; Khan, S.M.; Afzal, A.; Rahman, I.U.; Afzal, M.; Islam, M. Ethno Medicinal Study upon Folk Recipes Against Various Human Diseases in Sarban Hills, Abbottabad, Pakistan. World J. Zool. 2015, 10, 41–46. [Google Scholar]
- Ghazanfarpour, M.; Sadeghi, R.; Latifnejad Roudsari, R.; Mirzaii Najmabadi, K.; Mousavi Bazaz, M.; Abdolahian, S.; Khadivzadeh, T. Effects of Red Clover on Hot Flash and Circulating Hormone Concentrations in Menopausal Women: A Systematic Review and Meta-Analysis. Avicenna J. Phytomed. 2015, 5, 498–511. [Google Scholar] [PubMed]
- Cegieła, U.; Folwarczna, J.; Pytlik, M.; Zgórka, G. Effects of Extracts from Trifolium Medium L. and Trifolium Pratense L. on Development of Estrogen Deficiency-Induced Osteoporosis in Rats. Evid. Based Complement. Altern. Med. 2012, 2012, e921684. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ali Khan, B.; Ullah, S.; Khan, M.K.; Alshahrani, S.M.; Braga, V.A. Formulation and Evaluation of Ocimum Basilicum-Based Emulgel for Wound Healing Using Animal Model. Saudi Pharm. J. 2020, 28, 1842–1850. [Google Scholar] [CrossRef] [PubMed]
- Habibi Zadeh, S.K.; Farahpour, M.-R.; Kar, H.H. The Effect of Topical Administration of an Ointment Prepared from Trifolium Repens Hydroethanolic Extract on the Acceleration of Excisional Cutaneous Wound Healing. Wounds 2020, 32, 253–261. [Google Scholar] [PubMed]
- Kurahashi, T.; Fujii, J. Roles of Antioxidative Enzymes in Wound Healing. J. Dev. Biol. 2015, 3, 57–70. [Google Scholar] [CrossRef]

| Extract Type | Therapeutic Effect | Mechanism of Action | Ref. |
|---|---|---|---|
| Methanol extract | Wound healing effect | Angiogenesis stimulation (by cytokine activity modulation (TNF- α); Antimicrobial activity and antifungal activity; Antioxidative properties (Ferullic and Chlorogenic Acid). | [91,92,93,94] |
| Hepatoprotection | Modulatory effect in hepatocytes comparable to oleanolic and ursolic acids | [67,74] | |
| Ethanol extract | Dermatological effects | Strong antiviral activity against DNA viruses and RNA viruses; Effect on lipid accumulation in human macrophage | [95] |
| Anti-cancer | Cytotoxic effect: increase of Glutathione S-Transferase (GST) activity and protection in carcinogenicity or toxicity (antioxidant activity, antiproliferative effect). | [96,97,98] | |
| Hypocholesterolemia | Lowering the lipid accumulation in human macrophage. | [99] | |
| Hydroalcoholic extract | Adjuvant in diabetes treatment | Anti-hyperglycemic effect (antioxidant activity and inhibition of α-glucosidase and α-amylase activities). | [93] |
| Vasorelaxant/anti-platelet effect | Anti-thrombotic effect (inhibits ADP and thrombin induced platelet aggregation) | [24,100,101,102] | |
| Neuro-psycho effects | Anxiolytic and sedative effect (action of malic, caffeic, kaempferol and oleanolic acids) | [101] | |
| Antiosteoporotic effect | Bone protection against osteoporosis induced by glucocorticoids. | [103] | |
| Anti-inflammatory effect | slight effect on Nitrogen Oxide synthesis, reduced leukocytes and monocytes, activation of phagocytes circulation | [104] | |
| Essential oils | Treatment of different skin pathologies/antiaging | Enhancing the skin penetration in vitro animal experiments. Antioxidant capacity (major oil compounds: linalool, isoanethole, eugenol) comparable to tocopherol. | [105,106] |
| Complementary with antibiotics | Synergic effect of Basil with some antibiotics for the treatment of certain bacterial infection (ex. Propionibacterium acne) | [107] | |
| Antitumoral effect | Cytotoxic activity (higher inhibition of the viability of Ehrlich ascites carcinoma cells due to linalool) | [108] | |
| Anti-Colitis treatment | Protective effect against colitis induced by acetic acid (significant decrease of myeloperoxidase). | [109] |
| Extract Type | Therapeutic Effect | Mechanism of Action | Ref. |
|---|---|---|---|
| Methanolic extract | Wound healing effect | The genistein present in the extract stimulates angiogenesis by activating the beta estrogen receptor, both by mechanisms dependent on this receptor and by independent mechanisms regulating wound healing. Antioxidant (isoflavones: triterpene saponins and flavonoids). Anti-inflammatory: genistein achieved by the downregulation of proinflammatory mediator activity (inactivation of nuclear factor-κB (NF-κB) and reduction in the expression levels of TNF-α Antimicrobial Antifungal effect against: Aspergillus niger, C. albicans and Fusarium verticillioides. | [28,110,111,112,113,114] |
| Antiaging | high concentration of phenolic compounds and flavonoids have the ability to reduce and neutralize free radicals in the skin | [115] | |
| Antiplatelet aggregation | activates the antiplatelet factor nitric oxide synthesis in the cells | [116] | |
| Ethanolic extract | Antispasmodic | in laryngitis, whooping cough, bronchitis and tuberculosis causes relaxation of the smooth muscles of the airways with relief of spasms | [112] |
| Hydroalcoholic extract | Hepatoprotective | increases the level of methionine in hepatic steatosis | [100] |
| Anti-diabetic | Ferulic acid inhibits the enzymes involved in the digestion of carbohydrates (α-amylase and α-glucosidase) and has anti-lipase activity. | [63,117] | |
| Anticancer | The dimeric alkaloids vinblastine and vincristine have anticancer properties due to their activity in destroying cancer cells. Polyphenolic compounds have a protective role and induce a reduction in the number of human tumor cells or an increase in them. | [57,118] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antonescu, A.-I.; Miere, F.; Fritea, L.; Ganea, M.; Zdrinca, M.; Dobjanschi, L.; Antonescu, A.; Vicas, S.I.; Bodog, F.; Sindhu, R.K.; et al. Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants 2021, 10, 1390. https://doi.org/10.3390/plants10071390
Antonescu A-I, Miere F, Fritea L, Ganea M, Zdrinca M, Dobjanschi L, Antonescu A, Vicas SI, Bodog F, Sindhu RK, et al. Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants. 2021; 10(7):1390. https://doi.org/10.3390/plants10071390
Chicago/Turabian StyleAntonescu (Mintas), Andreea-Ina, Florina Miere (Groza), Luminita Fritea, Mariana Ganea, Mihaela Zdrinca, Luciana Dobjanschi, Angela Antonescu, Simona Ioana Vicas, Florin Bodog, Rakesh K. Sindhu, and et al. 2021. "Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects" Plants 10, no. 7: 1390. https://doi.org/10.3390/plants10071390
APA StyleAntonescu, A.-I., Miere, F., Fritea, L., Ganea, M., Zdrinca, M., Dobjanschi, L., Antonescu, A., Vicas, S. I., Bodog, F., Sindhu, R. K., & Cavalu, S. (2021). Perspectives on the Combined Effects of Ocimum basilicum and Trifolium pratense Extracts in Terms of Phytochemical Profile and Pharmacological Effects. Plants, 10(7), 1390. https://doi.org/10.3390/plants10071390







