Imazapic Herbigation for Egyptian Broomrape (Phelipanche aegyptiaca) Control in Processing Tomatoes—Laboratory and Greenhouse Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Phelipanche Aegyptiaca Seed
2.2. Imazapic Herbicide
2.3. LC-MS/MS Imazapic Analysis
2.4. Soil Samples Analysis
2.5. Tomato Root Samples Analysis
2.6. Dose-Response of P. aegyptiaca Seed and Seedlings to Imazapic in Petri Dish
2.7. Effect of Imazapic on P. aegyptiaca Parasitizing Tomato Roots in Polyethylene Bag Studies
2.8. Imazapic Sorption in Four Soils
2.9. Imazapic Dose-Response of Tomato and P. aegyptiaca in Pots
2.10. Tracking Imazapic Concentration in Soil and Tomato Roots
2.11. Statistical Analysis
3. Results
3.1. Dose-Response of P. aegyptiaca Seed and Seedlings to Imazapic in Petri Dish
3.2. Effect of Imazapic on P. aegyptiaca Parasitizing Tomato Roots in Polyethylene Bag Studies
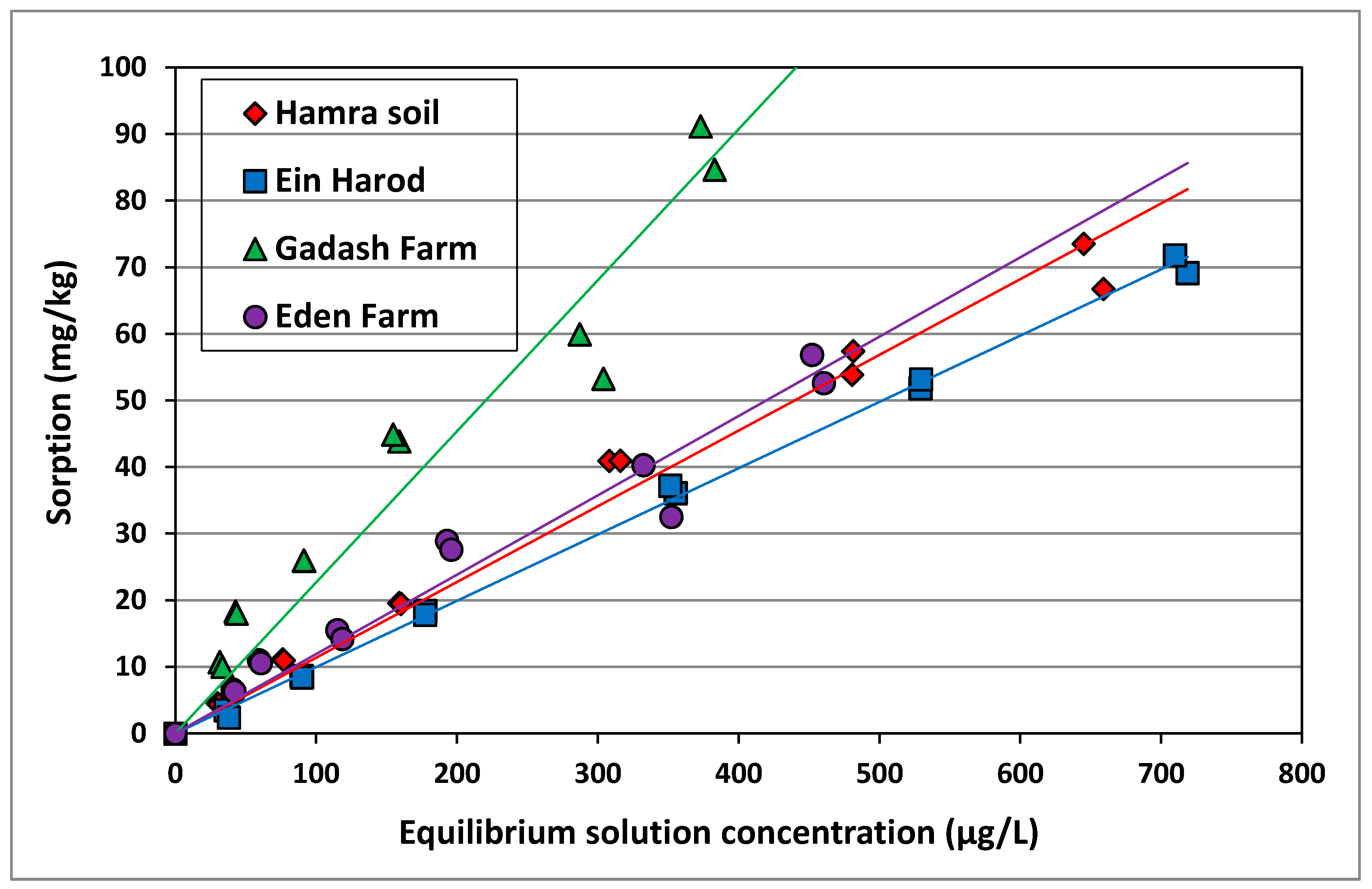
3.3. Imazapic Sorption to Soils
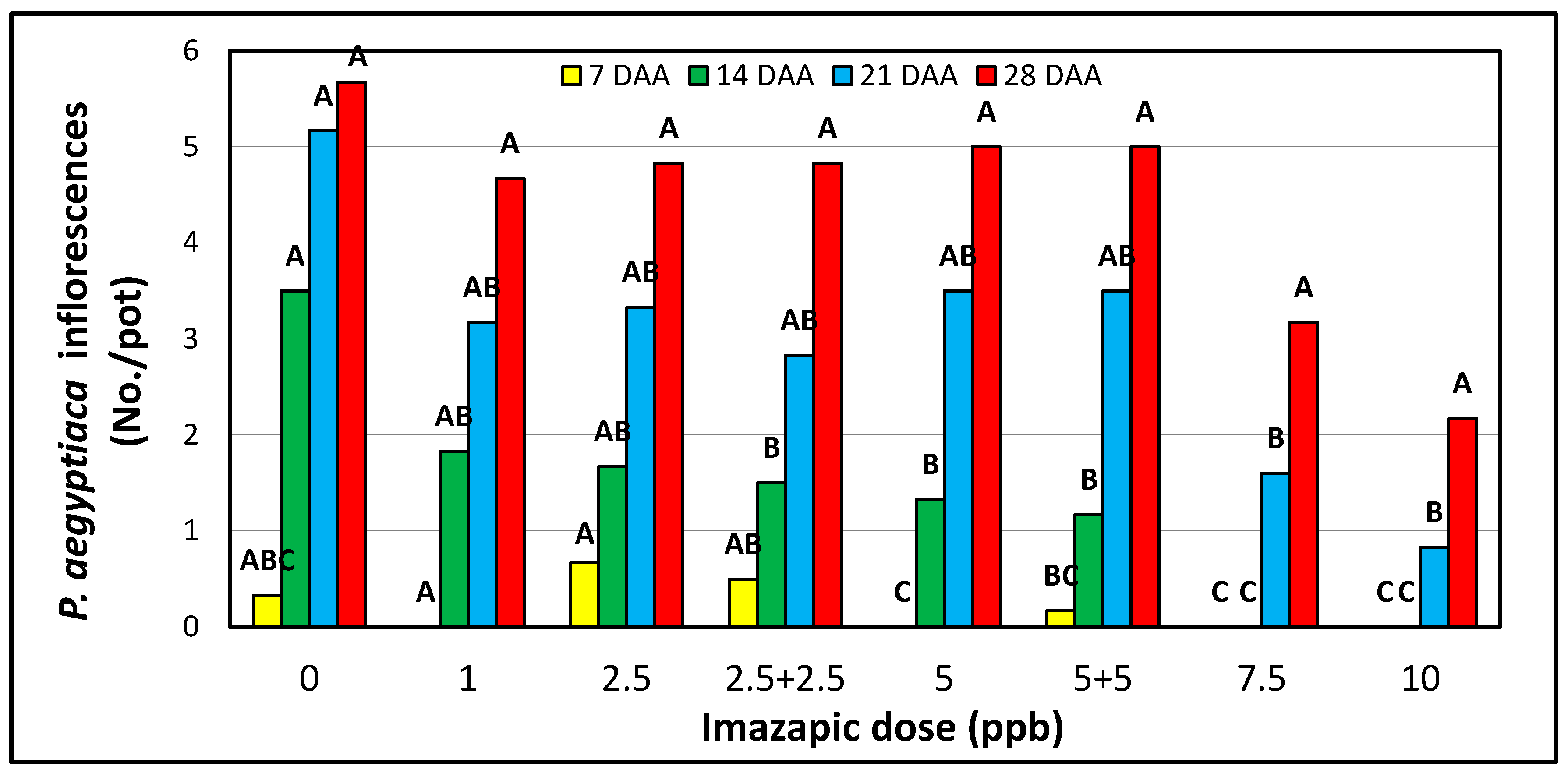
3.4. Imazapic Dose-Response of Tomato and P. aegyptiaca in Pots
3.5. Tracking Imazapic Concentration in Soil and the Tomato Plant
4. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Heide-Jorgensen, H.S. Introduction: The parasitic syndrome in higher plants. In Parasitic Orobanchaceae; Joel, D.M., Gressel, J., Musselman, L.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–18. [Google Scholar]
- Goldwasser, Y.; Rodenberg, J. Integrated Agronomic Management of Parasitic Weed Seed Banks. In Parasitic Orobanchaceae; Joel, D.M., Gressel, J., Musselman, L.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 393–413. [Google Scholar]
- Parker, C. The Parasitic Weeds of the Orobanchaceae. In Parasitic Orobanchaceae; Joel, D., Gressel, J., Musselman, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Parker, C.; Riches, C. Parasitic Weeds of the World: Biology and Control; CAB International: Wallingford, UK, 1993. [Google Scholar]
- Goldwasser, Y.; Kleifeld, Y. Recent approaches to Orobanche management—A review. In Weed Biology and Management; Inderjit, Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 439–466. [Google Scholar]
- Joel, D.M.; Gressel, J.; Musselman, L.J. Parasitic Orobanchaceae; Springer: Berlin/Heidelberg, Germany, 2013; 513p. [Google Scholar] [CrossRef]
- CABI Invasive Species Compendium-Orobanche aegyptiaca. Available online: https://www.cabi.org/isc/datasheet/37742#toidentity (accessed on 23 May 2021).
- Miyao, G. Egyptian broomrape eradication effort in California: A progress report on the joint effort of regulators, university, tomato growers and processors. In Proceedings of the XIV International Symposium on Processing Tomato, ISHS Acta Horticulturae 1159, Santiago, Chile, 6–9 March 2016. [Google Scholar] [CrossRef]
- Aparicio, M.F.; Delavault, P.; Timko, M.P. Management of infection by parasitic weeds: A review. Plants 2021, 9, 1184. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Wei, Q.; Shu, J.; Gan, Z.; Li, B.; Yan, D.; Huang, Z.; Guo, Y.; Wang, X.; Zhang, L.; et al. Exploration of resistance to Phelipanche aegyptiaca in tomato. Pest Manag. Sci. 2020, 76, 3806–3821. [Google Scholar] [CrossRef] [PubMed]
- Goldwasser, Y.; Kleifeld, Y.; Plakhine, D.; Rubin, B. Variation in vetch (Vicia spp.) response to Orobanche aegyptiaca. Weed Sci. 1997, 45, 756–762. [Google Scholar] [CrossRef]
- Shaner, D.L. Herbicide Handbook, 10th ed.; Weed Science Society of America: Champaign, IL, USA, 2014. [Google Scholar]
- Eizenberg, H.; Goldwasser, Y. Control of Egyptian Broomrape in Processing Tomato: A Summary of 20 Years of Research and Successful Implementation. Plant Dis. 2018, 102, 1477–1488. [Google Scholar] [CrossRef] [PubMed]
- Eizenberg, H.; Aly, R.; Cohen, Y. Technologies for smart chemical control of broomrape (Orobanche spp. and Phelipanche spp.). Weed Sci. 2012, 60, 316–323. [Google Scholar] [CrossRef]
- Hershenhorn, Y.; Eizenberg, H.; Dor, E.; Kapulnik, Y.; Goldwasser, Y. Phelipanche aegyptiaca management in tomato. Weed Res. 2009, 49 (Suppl. 1), 34–37. [Google Scholar] [CrossRef]
- Lewis, K.A.; Tzilivakis, J.; Warner, D.; Green, A. An international database for pesticide risk assessments and management. Hum. Ecol. Risk Assess. Int. J. 2016, 22, 1050–1064. [Google Scholar] [CrossRef]
- Tu, M.; Hurd, C.; Randall, J.M. Weed Control Methods Handbook; The Nature Conservancy: Arlington County, VA, USA, 2001; Available online: http://tncweeds.ucdavis.edu (accessed on 8 June 2021).
- American Cyanamid Company. Plateau Herbicide, for Weed Control, Native Grass Establishment and Turf Growth Suppression on Roadsides and Other Non-Crop Areas, PE-47015; American Cyanamid Company: Parsippany, NJ, USA, 2000. [Google Scholar]
- Gerstl, Z.; Yaron, B. Behavior of bromacil and napropamide in soils. II. Distribution after application from a point source. Soil Sci. Soc. Am. J. 1983, 47, 478–483. [Google Scholar] [CrossRef]
- Gerstl, Z.; Albasel, N. Field distribution of pesticides applied via a drip irrigation system. Irrig. Sci. 1984, 5, 181–193. [Google Scholar] [CrossRef]
- Ephrath, J.E.; Hershenhorn, J.; Achdari, G.; Bringer, S.; Eizenberg, H. Use of logistic equation for detection of the initial parasitism phase of Egyptian broomrape (Phelipanche aegyptiaca) in tomato. Weed Sci. 2012, 60, 57–63. [Google Scholar] [CrossRef]



| Soil Source | pH | Clay (%) | Silt (%) | Sand (%) | OC (%) |
|---|---|---|---|---|---|
| Eden Farm | 7.4 | 47 | 27 | 26 | 1.2 |
| Gadash Farm | 7.5 | 59 | 31 | 10 | 0.94 |
| Ein Harod | 8.0 | 57 | 29 | 14 | 0.41 |
| Bet Dagan | 7.6 | 10 | 3 | 87 | 0.40 |
| Treatment | Imazapic Application (ppb) | |
|---|---|---|
| 27 DAP | 51 DAP | |
| Control | ||
| 1 | 1.0 | |
| 2 | 2.5 | |
| 3 | 2.5 | 2.5 |
| 4 | 5.0 | |
| 5 | 5.0 | 5.0 |
| 6 | 7.5 | |
| 7 | 10.0 | |
| Imazapic Concentration (ppb) | Seed Germination (% of Control) | Seedling Vigor (% of Control) |
|---|---|---|
| 0 | 100.0 ± 0.01 | 100.0 ± 0.02 |
| 500 | 97.90 ± 0.04 | 94.98 ± 0.02 |
| 1000 | 96.54 ± 0.02 | 96.45 ± 0.03 |
| 5000 | 97.28 ± 0.01 | 98.45 ± 0.04 |
| Soil | Kd (L/kg) | Koc | r2 |
|---|---|---|---|
| Gadash Farm | 0.23 a | 24.4 | 0.938 |
| Eden Farm | 0.13 b | 10.8 | 0.957 |
| Hamra | 0.11 bc | 27.5 | 0.983 |
| Ein Harod | 0.10 c | 26.8 | 0.998 |
| Days after Treatment | Imazapic Soil Concentration (ppb w/w) | Imazapic Tomato Root Concentration (ppb w/w) |
|---|---|---|
| 1 | 7.5 ± 2.4 | 263.6 ± 30.5 |
| 3 | 2.6 ± 0.1 | 344.3 ± 93.9 |
| 7 | 0.4 ± 0.2 | 234.6 ± 87.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goldwasser, Y.; Rabinovitz, O.; Gerstl, Z.; Nasser, A.; Paporisch, A.; Kuzikaro, H.; Sibony, M.; Rubin, B. Imazapic Herbigation for Egyptian Broomrape (Phelipanche aegyptiaca) Control in Processing Tomatoes—Laboratory and Greenhouse Studies. Plants 2021, 10, 1182. https://doi.org/10.3390/plants10061182
Goldwasser Y, Rabinovitz O, Gerstl Z, Nasser A, Paporisch A, Kuzikaro H, Sibony M, Rubin B. Imazapic Herbigation for Egyptian Broomrape (Phelipanche aegyptiaca) Control in Processing Tomatoes—Laboratory and Greenhouse Studies. Plants. 2021; 10(6):1182. https://doi.org/10.3390/plants10061182
Chicago/Turabian StyleGoldwasser, Yaakov, Onn Rabinovitz, Zev Gerstl, Ahmed Nasser, Amit Paporisch, Hadar Kuzikaro, Moshe Sibony, and Baruch Rubin. 2021. "Imazapic Herbigation for Egyptian Broomrape (Phelipanche aegyptiaca) Control in Processing Tomatoes—Laboratory and Greenhouse Studies" Plants 10, no. 6: 1182. https://doi.org/10.3390/plants10061182
APA StyleGoldwasser, Y., Rabinovitz, O., Gerstl, Z., Nasser, A., Paporisch, A., Kuzikaro, H., Sibony, M., & Rubin, B. (2021). Imazapic Herbigation for Egyptian Broomrape (Phelipanche aegyptiaca) Control in Processing Tomatoes—Laboratory and Greenhouse Studies. Plants, 10(6), 1182. https://doi.org/10.3390/plants10061182







