LC-MS/MS Profiles and In Vitro Biological Activities of Extracts of an Endemic Species from Turkey: Stachys cretica ssp. anatolica
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Plant Material
3.2. Solvent Extraction
3.3. Total Flavonoid and Phenolic Contents
3.4. Liquid Chromatography–Electrospray Tandem Mass Spectrometry (LC–ESI–MS/MS) Analysis
3.5. Enzyme Inhibition Activity
3.6. Antioxidant Activity
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef]
- Perrino, E.; Valerio, F.; Gannouchi, A.; Trani, A.; Mezzapesa, G. Ecological and Plant Community Implication on Essential Oils Composition in Useful Wild Officinal Species: A Pilot Case Study in Apulia (Italy). Plants 2021, 10, 574. [Google Scholar] [CrossRef]
- Akçiçek, E. A new subspecies of Stachys cretica (section Eriostomum, Lamiaceae) from Turkey|Türkiye’den Stachys cretica (section Eriostomum, Lamiaceae)’ nın yeni bir alt türü. Turk. J. Bot. 2010, 34, 131–136. [Google Scholar] [CrossRef]
- Tundis, R.; Peruzzi, L.; Menichini, F. Phytochemical and biological studies of Stachys species in relation to chemotaxonomy: A review. Phytochemistry 2014, 102, 7–39. [Google Scholar] [CrossRef] [PubMed]
- Uritu, C.M.; Mihai, C.T.; Stanciu, G.-D.; Dodi, G.; Alexa-Stratulat, T.; Luca, A.; Leon-Constantin, M.-M.; Stefanescu, R.; Bild, V.; Melnic, S.; et al. Medicinal Plants of the Family Lamiaceae in Pain Therapy: A Review. Pain Res. Manag. 2018, 2018, 1–44. [Google Scholar] [CrossRef]
- Calvani, M.; Pasha, A.; Favre, C. Nutraceutical Boom in Cancer: Inside the Labyrinth of Reactive Oxygen Species. Int. J. Mol. Sci. 2020, 21, 1936. [Google Scholar] [CrossRef] [PubMed]
- Grom, L.C.; Coutinho, N.M.; Guimarães, J.T.; Balthazar, C.F.; Silva, R.; Rocha, R.S.; Freitas, M.Q.; Duarte, M.C.K.; Pimentel, T.C.; Esmerino, E.A.; et al. Probiotic dairy foods and postprandial glycemia: A mini-review. Trends Food Sci. Technol. 2020, 101, 165–171. [Google Scholar] [CrossRef]
- Kiokias, S.; Proestos, C.; Oreopoulou, V. Phenolic Acids of Plant Origin—A Review on Their Antioxidant Activity In Vitro (O/W Emulsion Systems) Along with Their in Vivo Health Biochemical Properties. Foods 2020, 9, 534. [Google Scholar] [CrossRef]
- Panzella, L.; Napolitano, A. Natural and Bioinspired Phenolic Compounds as Tyrosinase Inhibitors for the Treatment of Skin Hyperpigmentation: Recent Advances. Cosmetics 2019, 6, 57. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Rodrigues, C.F.; Sharopov, F.; Docea, A.O.; Karaca, A.C.; Sharifi-Rad, M.; Karıncaoglu, D.K.; Gülseren, G.; Özçelik, B.; Demircan, E.; et al. Diet, Lifestyle and Cardiovascular Diseases: Linking Pathophysiology to Cardioprotective Effects of Natural Bioactive Compounds. Int. J. Environ. Res. Public Health 2020, 17, 2326. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Miao, M. Dietary polyphenols modulate starch digestion and glycaemic level: A review. Crit. Rev. Food Sci. Nutr. 2020, 60, 541–555. [Google Scholar] [CrossRef] [PubMed]
- Toma, L.; Sanda, G.M.; Niculescu, L.S.; Deleanu, M.; Sima, A.V.; Stancu, C.S. Phenolic Compounds Exerting Lipid-Regulatory, Anti-Inflammatory and Epigenetic Effects as Complementary Treatments in Cardiovascular Diseases. Biomolecules 2020, 10, 641. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Kirkan, B.; Sarikurkcu, C.; Ceylan, O. Metabolite profiling and health benefits of Stachys cretica subsp. mersinaea as a medicinal food. Ind. Crop. Prod. 2019, 131, 85–89. [Google Scholar] [CrossRef]
- Kirkan, B. Antioxidant potential, enzyme inhibition activity, and phenolic profile of extracts from Stachys cretica subsp. vacillans. Ind. Crop. Prod. 2019, 140. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Kirkan, B.; Sarikurkcu, C. Phenolic ingredients and therapeutic potential of Stachys cretica subsp. smyrnaea for the management of oxidative stress, Alzheimer’s disease, hyperglycemia, and melasma. Ind. Crop. Prod. 2019, 127, 82–87. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Menichini, F. An Overview on Chemical Aspects and Potential Health Benefits of Limonoids and Their Derivatives. Crit. Rev. Food Sci. Nutr. 2013, 54, 225–250. [Google Scholar] [CrossRef] [PubMed]
- Picot, M.C.N.; Mahomoodally, M.F. Effects of Aphloia theiformis on key enzymes related to diabetes mellitus. Pharm. Biol. 2017, 55, 864–872. [Google Scholar] [CrossRef]
- Elfalleh, W.; Kirkan, B.; Sarikurkcu, C. Antioxidant potential and phenolic composition of extracts from Stachys tmolea: An endemic plant from Turkey. Ind. Crop. Prod. 2019, 127, 212–216. [Google Scholar] [CrossRef]
- Kocak, M.; Uren, M.; Calapoglu, M.; Tepe, A.S.; Mocan, A.; Rengasamy, K.; Sarikurkcu, C. Phenolic profile, antioxidant and enzyme inhibitory activities of Stachys annua subsp. annua var. annua. S. Afr. J. Bot. 2017, 113, 128–132. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Kocak, M.S.; Uren, M.C.; Calapoglu, M.; Tepe, A.S. Potential sources for the management global health problems and oxidative stress: Stachys byzantina and S. iberica subsp. iberica var. densipilosa. Eur. J. Integr. Med. 2016, 8, 631–637. [Google Scholar] [CrossRef]
- Ertas, A.; Yener, I. A comprehensive study on chemical and biological profiles of three herbal teas in Anatolia; rosmarinic and chlorogenic acids. S. Afr. J. Bot. 2020, 130, 274–281. [Google Scholar] [CrossRef]
- Karaboduk, K.; Karabacak, O.; Yiyit Dogan, S.; Karaboduk, H.; Gunduzer, E.; Tekinay, T. Comparison of antimicrobial, antioxidant capacities and hplc analysis of three stachys species in Turkey. J. Environ. Prot. Ecol. 2014, 15, 1293–1302. [Google Scholar]
- Janabi, A.H.W.; Kamboh, A.A.; Saeed, M.; Xiaoyu, L.; BiBi, J.; Majeed, F.; Naveed, M.; Mughal, M.J.; Korejo, N.A.; Kamboh, R.; et al. Flavonoid-rich foods (FRF): A promising nutraceutical approach against lifespan-shortening diseases. Iran. J. Basic Med. Sci. 2020, 23, 140–153. [Google Scholar]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modarresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Sarikurkcu, C.; Ozer, M.S.; Cakir, A.; Eskici, M.; Mete, E. GC/MS Evaluation and In Vitro Antioxidant Activity of Essential Oil and Solvent Extracts of an Endemic Plant Used as Folk Remedy in Turkey:Phlomis bourgaeiBoiss. Evid.-Based Complement. Altern. Med. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, K.U.; Ali, S.A.; Ali, A.; Naaz, I. Natural Tyrosinase Inhibitors: Role of Herbals in the Treatment of Hyperpigmentary Disorders. Mini-Rev. Med. Chem. 2019, 19, 796–808. [Google Scholar] [CrossRef] [PubMed]
- Sarikurkcu, C.; Kirkan, B.; Ozer, M.S.; Ceylan, O.; Atilgan, N.; Cengiz, M.; Tepe, B. Chemical characterization and biological activity of Onosma gigantea extracts. Ind. Crop. Prod. 2018, 115, 323–329. [Google Scholar] [CrossRef]
- Cittan, M.Ç.A. Development and Validation of an Analytical Methodology Based on Liquid Chromatography-Electrospray Tandem Mass Spectrometry for the Simultaneous Determination of Phenolic Compounds in Olive Leaf Extract. J. Chromatogr. Sci. 2018, 56, 336–343. [Google Scholar] [CrossRef]
- Tlili, N.; Kirkan, B.; Sarikurkcu, C. LC–ESI–MS/MS characterization, antioxidant power and inhibitory effects on α-amylase and tyrosinase of bioactive compounds from hulls of Amygdalus communis: The influence of the extracting solvents. Ind. Crop. Prod. 2019, 128, 147–152. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Dinparast, L.; Zengin, G.; Sarikurkcu, C.; Bahadori, S.; Asghari, B.; Movahhedin, N. Functional components, antidiabetic, anti-Alzheimer’s disease, and antioxidant activities of Salvia syriaca L. Int. J. Food Prop. 2016, 20, 1761–1772. [Google Scholar] [CrossRef]
- Zengin, G.; Sarıkürkçü, C.; Aktümsek, A.; Ceylan, R. Antioxidant potential and inhibition of key enzymes linked to Alzheimer’s diseases and diabetes mellitus by monoterpene-rich essential oil from Sideritis galatica Bornm. Endemic to Turkey. Rec. Nat. Prod. 2015, 10, 195–206. [Google Scholar]
- Zengin, G.; Sarikurkcu, C.; Gunes, E.; Uysal, A.; Ceylan, R.; Uysal, S.; Gungor, H.; Aktumsek, A. Two Ganoderma species: Profiling of phenolic compounds by HPLC–DAD, antioxidant, antimicrobial and inhibitory activities on key enzymes linked to diabetes mellitus, Alzheimer’s disease and skin disorders. Food Funct. 2015, 6, 2794–2802. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E.; Erçağ, E. The cupric ion reducing antioxidant capacity and polyphenolic content of some herbal teas. Int. J. Food Sci. Nutr. 2006, 57, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Kocak, M.S.; Sarikurkcu, C.; Cengiz, M.; Kocak, S.; Uren, M.C.; Tepe, B. Salvia cadmica: Phenolic composition and biological activity. Ind. Crop. Prod. 2016, 85, 204–212. [Google Scholar] [CrossRef]
- Kose, E.O.; Aktaş, O.; Deniz, I.G.; Sarikürkçü, C. Chemical composition, antimicrobial and antioxidant activity of essential oil of endemic ferula lycia boiss. J. Med. Plants Res. 2010, 4, 1698–1703. [Google Scholar]
- Sarikurkcu, C. Antioxidant activities of solvent extracts from endemic Cyclamen mirabile Hildebr. tubers and leaves. Afr. J. Biotechnol. 2011, 10, 831–839. [Google Scholar]
- Zengin, G.; Sarikurkcu, C.; Uyar, P.; Aktumsek, A.; Uysal, S.; Kocak, M.S.; Ceylan, R. Crepis foetida L. subsp. rhoeadifolia (Bieb.) Celak. as a source of multifunctional agents: Cytotoxic and phytochemical evaluation. J. Funct. Foods 2015, 17, 698–708. [Google Scholar] [CrossRef]
- Tepe, B.; Sarikurkcu, C.; Berk, S.; Alim, A.; Akpulat, H.A. Chemical composition, radical scavenging and antimicrobial activity of the essential oils of Thymus boveii and Thymus hyemalis. Rec. Nat. Prod. 2011, 5, 208–220. [Google Scholar]
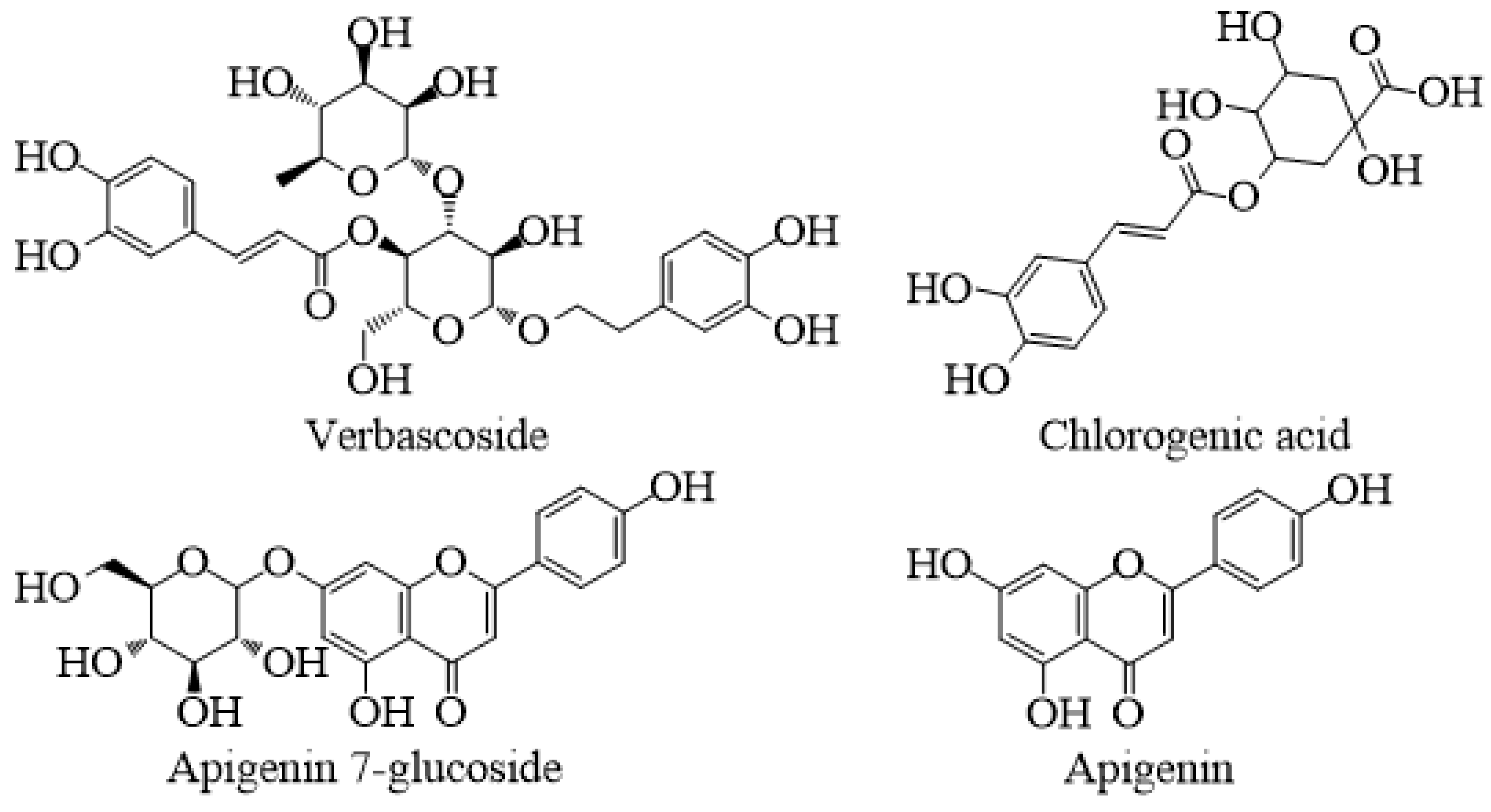
| Compound | Water | Methanol |
|---|---|---|
| Gallic acid | 5.60 ± 0.10 b | 33.06 ± 0.93 a |
| Protocatechuic acid | 363.46 ± 0.09 a | 193.92 ± 1.31 b |
| 3,4-Dihydroxyphenylacetic acid | 18.57 ± 0.07 a | 6.70 ± 0.24 b |
| (+)-Catechin | nd | nd |
| Pyrocatechol | nd | nd |
| Chlorogenic acid | 7252.19 ± 466.89 b | 9437.77 ± 407.11 a |
| 2,5-Dihydroxybenzoic acid | 9.04 ± 0.49 a | 8.38 ± 0.52 a |
| 4-Hydroxybenzoic acid | 43.37 ± 0.06 b | 97.26 ± 1.07 a |
| (-)-Epicatechin | nd | nd |
| Caffeic acid | 967.83 ± 10.86 a | 124.56 ± 0.23 b |
| Vanillic acid | nd | 857.97 ± 31.36 |
| Syringic acid | 91.45 ± 5.44 b | 512.43 ± 9.85 a |
| 3-Hydroxybenzoic acid | 4.86 ± 0.17 b | 10.11 ± 0.14 a |
| Vanillin | nd | 81.02 ± 2.15 |
| Verbascoside | 15447.91 ± 102.41 b | 25402.03 ± 163.12 a |
| Taxifolin | nd | nd |
| Sinapic acid | 2.93 ± 0.47 b | 5.71 ± 0.01 a |
| p-Coumaric acid | 177.34 ± 2.32 a | 22.21 ± 0.59 b |
| Ferulic acid | 461.08 ± 5.84 b | 665.44 ± 7.72 a |
| Luteolin 7-glucoside | 67.08 ± 1.95 b | 225.19 ± 3.39 a |
| Hesperidin | nd | nd |
| Hyperoside | 101.51 ± 0.72 b | 252.49 ± 2.74 a |
| Rosmarinic acid | 68.22 ± 0.97 a | 6.58 ± 1.72 b |
| Apigenin 7-glucoside | 2116.10 ± 16.02 b | 2313.87 ± 0.48 a |
| 2-Hydroxycinnamic acid | nd | nd |
| Pinoresinol | 12.58 ± 0.89 b | 204.85 ± 16.38 a |
| Eriodictyol | 0.69 ± 0.01 b | 3.52 ± 0.15 a |
| Quercetin | 5.22 ± 0.40 a | 8.73 ± 1.22 a |
| Luteolin | 62.71 ± 3.01 b | 859.93 ± 31.06 a |
| Kaempferol | nd | 4.65 ± 0.71 |
| Apigenin | 163.04 ± 5.53 b | 2004.75 ± 59.35 a |
| Assays | Water | Methanol |
|---|---|---|
| Ferrous ion chelating (mg EDTAE/g extract) | 68.38 ± 0.69 a | 53.64 ± 1.35 b |
| CUPRAC reducing power (mg TE/g extract) | 532.56 ± 5.83 a | 246.13 ± 2.80 b |
| FRAP reducing power (mg TE/g extract) | 303.28 ± 1.48 a | 127.20 ± 7.18 b |
| DPPH radical scavenging (mg TE/g extract) | 249.89 ± 1.52 a | 80.44 ± 3.04 b |
| ABTS radical scavenging (mg TE/g extract) | 276.68 ± 12.35 a | 112.19 ± 4.31 b |
| Phosphomolybdenum (mg TE/g extract) | 801.69 ± 31.26 a | 546.31 ± 13.99 b |
| Assays | Water | Methanol |
|---|---|---|
| Yield (%) | 12.25 | 5.45 |
| Total flavonoids (mg QE/g extract) | 30.20 ± 0.01 a | 24.62 ± 0.05 b |
| Total phenolics (mg GAE/g extract) | 42.13 ± 2.04 a | 27.13 ± 0.68 b |
| Tyrosinase inhibition (mg KAE/g extract) | 163.14 ± 10.55 b | 306.05 ± 4.94 a |
| α-Amylase inhibition (mg ACE/g extract) | 5.12 ± 0.59 b | 471.19 ± 28.62 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carev, I.; Sarikurkcu, C. LC-MS/MS Profiles and In Vitro Biological Activities of Extracts of an Endemic Species from Turkey: Stachys cretica ssp. anatolica. Plants 2021, 10, 1054. https://doi.org/10.3390/plants10061054
Carev I, Sarikurkcu C. LC-MS/MS Profiles and In Vitro Biological Activities of Extracts of an Endemic Species from Turkey: Stachys cretica ssp. anatolica. Plants. 2021; 10(6):1054. https://doi.org/10.3390/plants10061054
Chicago/Turabian StyleCarev, Ivana, and Cengiz Sarikurkcu. 2021. "LC-MS/MS Profiles and In Vitro Biological Activities of Extracts of an Endemic Species from Turkey: Stachys cretica ssp. anatolica" Plants 10, no. 6: 1054. https://doi.org/10.3390/plants10061054
APA StyleCarev, I., & Sarikurkcu, C. (2021). LC-MS/MS Profiles and In Vitro Biological Activities of Extracts of an Endemic Species from Turkey: Stachys cretica ssp. anatolica. Plants, 10(6), 1054. https://doi.org/10.3390/plants10061054







