Influence of Silicon on Biocontrol Strategies to Manage Biotic Stress for Crop Protection, Performance, and Improvement
Abstract
1. Introduction
2. Availability of Si in Soil
3. Action Mechanism of Si and Interaction of Biotic Stress
3.1. Silicon Resist Insect Pests’ Diseases
3.2. Effects of Silicon on Plant Fungal Diseases
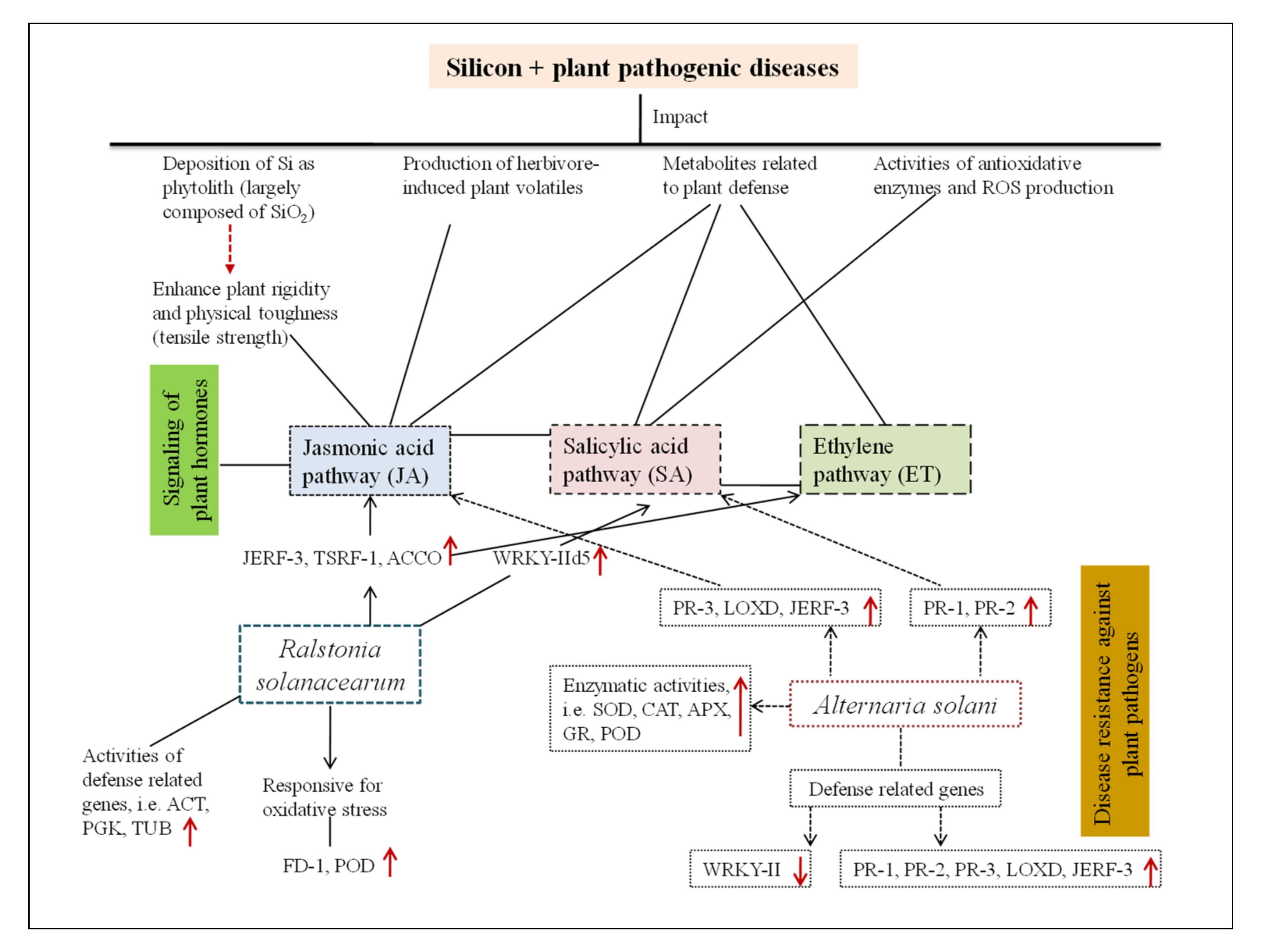
3.3. The Impact of Silicon on Plant Bacterial Infections
4. Silicon Increase Resistance Mechanism
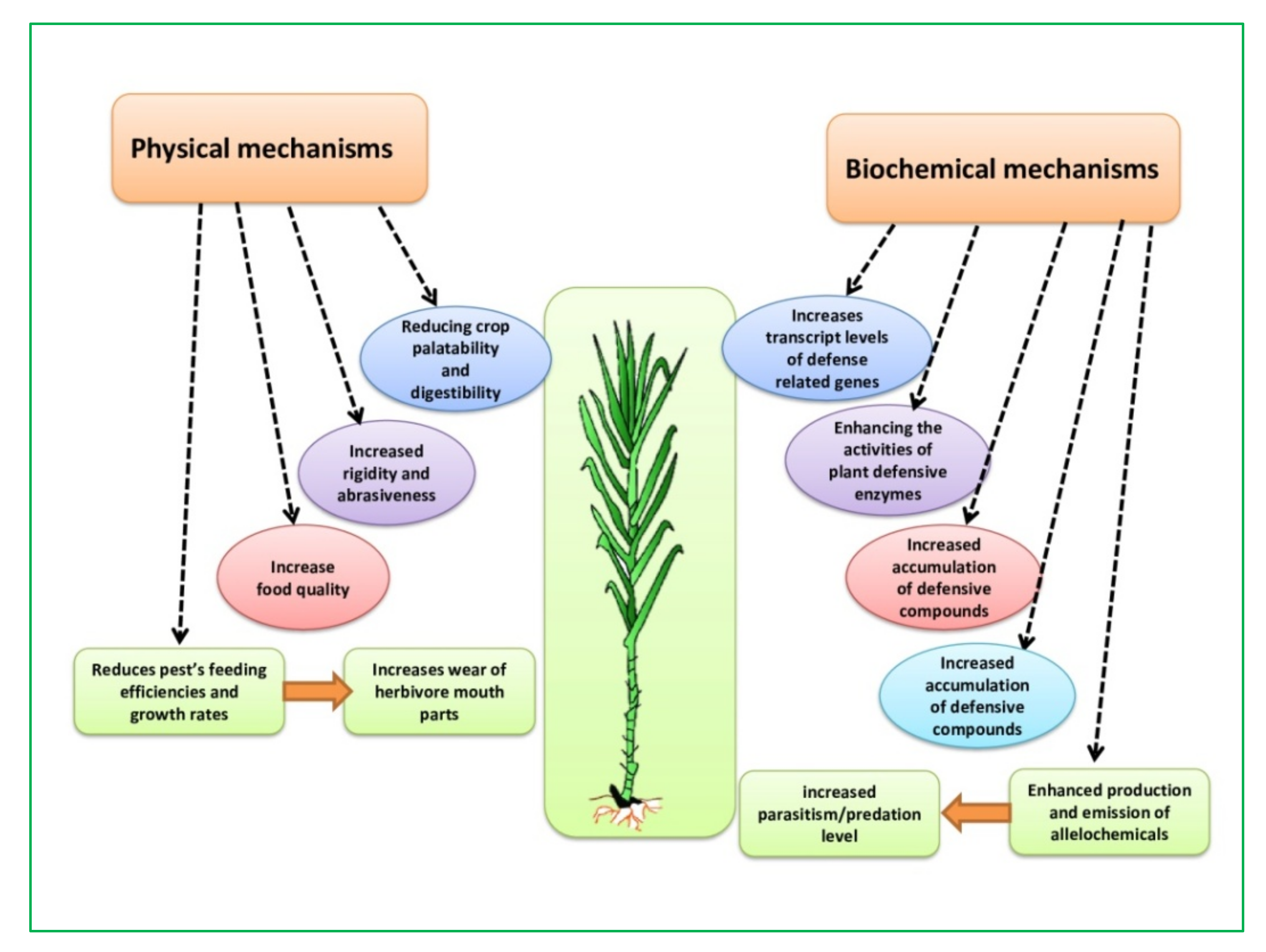
4.1. Mechanism Physical Barrier
4.2. Biochemical Mechanism
4.3. Role of Defense-Related Enzymes
4.4. Genomics and Metabolomics Prospective
5. Is Si Essential/Beneficial Element?
6. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Epstein, E. Silicon. Ann. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 641–664. [Google Scholar] [CrossRef] [PubMed]
- Coskun, D.; Deshmukh, R.; Sonah, H.; Menzies, J.G.; Reynolds, O.; Ma, J.F.; Kronzucker, H.J.; Belanger, R.R. The controversies of silicon’s role in plant biology. New Phytol. 2019, 221, 67–85. [Google Scholar] [CrossRef] [PubMed]
- Verma, K.K.; Singh, P.; Song, X.P.; Malviya, M.K.; Singh, R.K.; Chen, G.L.; Solomon, S.; Li, Y.R. Mitigating climate change for sugarcane improvement: Role of silicon in alleviating abiotic stresses. Sugar Tech. 2020, 22, 741–749. [Google Scholar] [CrossRef]
- Gulzar, N.; Ali, S.; Shah, M.A.; Kamili, A.N. Silicon supplementation improves early blight resistance in Lycopersicon esculentum Mill. by modulating the expression of defense-related genes and antioxidant enzymes. 3 Biotech. 2021, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Prabha, S.; Durgalakshmi, D.; Rajendran, S.; Lichtfouse, E. Plant-derived silica nanoparticles and composites for biosensors, bioimaging, drug delivery and supercapacitors: A review. Environ. Chem. Lett. 2021, 19, 1667–1691. [Google Scholar] [CrossRef] [PubMed]
- Cooke, J.; Leishman, M.R. Consistent alleviation of abiotic stress with silicon addition: A meta analysis. Funct. Ecol. 2016, 30, 1340–1357. [Google Scholar] [CrossRef]
- Frew, A.; Weston, L.A.; Reynolds, O.L.; Gurr, G.M. The role of silicon in plant biology: A paradigm shift in research approach. Ann. Bot. 2018, 121, 1265–1273. [Google Scholar]
- Verma, K.K.; Song, X.-P.; Lin, B.; Guo, D.-J.; Singh, M.; Rajput, V.D.; Singh, R.K.; Singh, P.; Sharma, A.; Malviya, M.K.; et al. Silicon induced drought tolerance in crop plants: Physiological adaptation strategies. Silicon 2021. [Google Scholar] [CrossRef]
- Verma, K.K.; Song, X.-P.; Verma, C.L.; Chen, Z.-L.; Rajput, V.D.; Wu, K.-C.; Liao, F.; Chen, G.-L.; Li, Y.-R. Functional relationship between photosynthetic leaf gas exchange in response to silicon application and water stress mitigation in sugarcane. Biol. Res. 2021, 54, 15. [Google Scholar] [CrossRef]
- Debona, D.; Rodrigues, F.A.; Datnoff, L.E. Silicon’s role in abiotic and biotic plantstresses. Ann. Rev. Phytopathol. 2017, 55, 85–107. [Google Scholar] [CrossRef]
- Etesami, H.; Jeong, B.R. Silicon (Si): Review and future prospects on the action mechanisms in alleviating biotic and abiotic stresses in plants. Ecotoxicol. Environ. Saf. 2018, 147, 881–896. [Google Scholar] [CrossRef]
- Savvas, D.; Ntatsi, G. Biostimulant activity of silicon in horticulture. Sci. Hortic. 2015, 196, 66–81. [Google Scholar] [CrossRef]
- Epstein, E. Silicon in Plants: Facts vs. Concepts; Elsevier Science: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Rodrigues, F.A.; Datnof, L.E. Silicon and Plant Diseases; Springer: Berlin, Germany, 2015. [Google Scholar]
- Fauteux, F.; Remus-Borel, W.; Menzies, J.; Belanger, R. Silicon and plant disease resistance against pathogenic fungi. FEMS Microbiol. Lett. 2005, 249, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Datnoff, L.E.; Rodrigues, F.A.; Seebold, K.W. Silicon and plant disease. In Mineral Nutrition and Plant Disease; Datnoff, L.E., Elmer, W.H., Huber, D.M., Eds.; The American Phytopathological Society: Saint Paul, MN, USA, 2007; pp. 233–246. [Google Scholar]
- Fortunato, A.A.; Rodrigues, F.Á.; Do Nascimento, K.J.T. Physiological and biochemical spects of the resistance of banana plants to Fusarium wilt potentiated by silicon. Phytopathology 2012, 102, 957–966. [Google Scholar] [CrossRef]
- Van Bockhaven, J.; De Vleesschauwer, D.; Hofte, M. Towards establishing roadspectrum broadspectrum disease resistance in plants: Silicon leads the way. J. Exp. Bot. 2013, 64, 1281–1293. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, M.; Wang, L.; Lin, W.; Fan, X.; Cai, K. Proteomic characterization of silicon-mediated resistance against Ralstonia solanacearum in tomato. Plant Soil 2015, 387, 425–440. [Google Scholar] [CrossRef]
- Vivancos, J.; Labbe, C.; Menzies, J.G.; Belanger, R.R. Silicon-mediated resistance of Arabidopsis against powdery mildew involves mechanisms other than the salicylic acid (SA)-dependent defence pathway. Mol. Plant Pathol. 2015, 16, 572–582. [Google Scholar] [CrossRef]
- Debona, D.; Rodrigues, F.A.; Rios, J.A.; Nascimento, K.J.T.; Silva, L.C. The effect of silicon on antioxidant metabolism of wheat leaves infected by Pyricularia oryza. Plant Pathol. 2014, 63, 581–589. [Google Scholar] [CrossRef]
- Akhtar, N.; Chandra, R.; Mazhar, Z. Silicon based defence mechanism in plants. Trends Biosci. 2018, 11, 32. [Google Scholar]
- Bayles, R.A.; Flath, K.; Hovmoller, M.S.; De Valla-vieille-Pope, C. Breakdown of the Yr17 resistance to yellow rust of wheat in northern Europe. Agronomie 2000, 20, 805–811. [Google Scholar] [CrossRef]
- Ma, Z.; Michailides, T.J. Advances in understanding molecular mechanisms of fungicide resistance and molecular detection of resistant genotypes in phytopathogenic fungi. Crop Prot. 2005, 24, 853–863. [Google Scholar] [CrossRef]
- Sakr, N. The role of silicon (Si) in increasing plant resistance against fungal diseases. Hellenic Plant Prot. J. 2016, 9, 1–15. [Google Scholar] [CrossRef]
- Ma, J.F.; Yamaji, N. Silicon uptake and accumulation in higher plants. Trends Plant Sci. 2006, 11, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Bakhat, H.F.; Bibi, N.; Zia, Z.; Abbas, S.; Hammad, H.M.; Fahad, S.; Ashraf, M.R.; Shah, G.M.; Rabbani, F.; Saeed, S. Silicon mitigates biotic stresses in crop plants: A review. Crop Prot. 2018, 104, 21–34. [Google Scholar] [CrossRef]
- Song, X.P.; Verma, K.K.; Tian, D.D.; Zhang, X.Q.; Liang, Y.J.; Huang, X.; Li, C.N.; Li, Y.R. Exploration of silicon functions to integrate with biotic stress tolerance and crop improvement. Biol. Res. 2021, 54, 19. [Google Scholar] [CrossRef]
- Epstein, E. Silicon: Its manifold roles in plants. Ann. Appl. Biol. 2009, 155, 155–160. [Google Scholar] [CrossRef]
- Verma, K.K.; Song, X.-P.; Zeng, Y.; Guo, D.-J.; Singh, M.; Rajput, V.D.; Malviya, M.K.; Wei, K.-J.; Sharma, A.; Li, D.-P.; et al. Foliar application of silicon boosts growth, photosynthetic leaf gas exchange, antioxidative response and resistance to limited water irrigation in sugarcane (Saccharum officinarum L.). Plant Physiol. Biochem. 2021, 166, 582–592. [Google Scholar] [CrossRef]
- Verma, K.K.; Song, X.P.; Tian, D.D.; Singh, M.; Verma, C.L.; Rajput, V.D.; Singh, R.K.; Sharma, A.; Singh, P.; Malviya, M.K.; et al. Investigation of defensive role of silicon during drought stress induced by irrigation capacity in sugarcane: Physiological and biochemical characteristics. ACS Omega 2021, 6, 19811–19821. [Google Scholar] [CrossRef]
- Datnoff, L.E.; Seebold, K.W.; Correa-V, F.J. The use of silicon for integrated disease management: Reducing fungicide applications and enhancing host plant resistance. Stud. Plant Sci. 2001, 8, 171–184. [Google Scholar]
- Joseph, E.K. Assessing the silicon status of rice (Oryza sativa). Master’s Thesis, Louisiana State University, Baton Rouge, LA, USA, 2009. [Google Scholar]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Academic Press: San Diego, CA, USA, 2011. [Google Scholar]
- Dematte, J.L.I.; Paggiaro, C.M.; Beltrame, J.; Ribeiro, S. Uso de silicatos em canade-açúcar. Inf. Agronômicas 2011, 133, 7–12. [Google Scholar]
- Meena, V.; Dotaniya, M.; Coumar, V.; Rajendiran, S.; Kundu, S.; Rao, A.S. A case for silicon fertilization to improve crop yields in tropical soils. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2014, 84, 505–518. [Google Scholar] [CrossRef]
- Zargar, S.M.; Mahajan, R.; Bhat, J.A.; Nazir, M.; Deshmukh, R. Role of silicon in plant stress tolerance: Opportunities to achieve a sustainable cropping system. 3 Biotech 2019, 9, 73. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Gao, L.; Dong, S.; Sun, Y.; Shen, Q.; Guo, S. Role of Silicon on Plant–Pathogen Interactions. Front. Plant Sci. 2017, 8, 701. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Schumaker, K.S.; Zhu, J.K. Cell signalling during cold, drought, and salt stress. Plant Cell 2002, 14, S165–S183. [Google Scholar] [CrossRef]
- Rabbani, M.A.; Maruyama, K.; Abe, H.; Khan, M.A.; Katsura, K.; Ito, Y.; Yoshiwara, K.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Monitoring expression profiles of rice genes under cold, drought, and high-salinity stresses and abscisic acid application using cDNA microarray and RNA gel-blot analyses. Plant Physiol. 2003, 133, 1755–1767. [Google Scholar] [CrossRef]
- Manivannan, A.; Ahn, Y.-K. Silicon regulates potential genes involved in major physiological processes in plants to combat stress. Front. Plant Sci. 2017, 8, 1346. [Google Scholar] [CrossRef]
- Sallam, M.N. Insect Damage: Damage on Post-Harvest. AGSI/FAO: INPhO. 2008. Available online: http://www.fao.org/inpho/content/compend/text/ch02–01.htm (accessed on 12 July 2021).
- Aetiba, J.P.N. Field Evaluation of Levo 2.4 sl (Botanical) for the Management of Insect Pests of Eggplant (Solanum Melongena L.) and Okra (Abelmoschus esculentus L. Moench). Master’s Thesis, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana, 2015. [Google Scholar]
- Korth, K.L. Medicago truncatula mutants demonstrate the role of plant calcium oxalate crystals as an effective defense against chewing insects. Plant Physiol. 2006, 141, 188–195. [Google Scholar] [CrossRef]
- Damalas, C. Understanding benefits and risks of pesticide use. Sci. Res. Essay 2009, 4, 945–949. [Google Scholar]
- Dos Santos, M.; Junqueira, A.R.; De Sá, V.M.; Zanúncio, J.; Serrão, J. Effect of silicon on the morphology of the midgut and mandible of tomato leaf miner Tuta absoluta (Lepidoptera: Gelechiidae) larvae. ISJ 2015, 12, 158–165. [Google Scholar]
- Huberty, A.F.; Denno, R.F. Plant water stress and its consequences for herbivorous insects: A new synthesis. Ecology 2004, 85, 1383–1398. [Google Scholar] [CrossRef]
- Meyer, J.; Keeping, M. Impact of silicon in alleviating biotic stress in sugarcane in South Africa. Proc. S. Afr. Sugar Technol. Assoc. 2005, 23, 14–18. [Google Scholar]
- Ma, J.F.; Takahashi, E. Soil, fertilizer, and plant silicon research in Japan. Soil Fertil. Plant Silicon Res. Jpn. 2002, 107–180. [Google Scholar]
- Nakata, Y.; Ueno, M.; Kihara, J.; Ichii, M.; Taketa, S.; Arase, S. Rice blast disease and susceptibility to pests in a silicon uptake-deficient mutant lsi1 of rice. Crop Prot. 2008, 27, 865–868. [Google Scholar] [CrossRef]
- Hunt, J.W.; Dean, A.P.; Webster, R.E.; Johnson, G.N.; Ennos, A.R. A novel mechanism by which silica defends grasses against herbivory. Ann. Bot. 2008, 102, 653–656. [Google Scholar] [CrossRef]
- Liang, Y.; Nikolic, M.; Bélanger, R.; Gong, H.; Song, A. Silicon in Agriculture; Springer: Dordrecht, The Netherlands, 2015. [Google Scholar]
- Forkpah, C.; Dixon, L.R.; Fahrbach, S.E.; Rueppell, O. Xenobiotic effects on intestinal stem cell proliferation in adult honey bee (Apis mellifera L) workers. PLoS ONE 2014, 9, e91180. [Google Scholar] [CrossRef] [PubMed]
- Lehane, M.; Billingsley, P. Biology of the Insect Midgut; Chapman & Hall: London, UK, 1996. [Google Scholar]
- Massey, F.P.; Ennos, A.R.; Hartley, S.E. Silica in grasses as a defense against insect herbivores: Contrasting effects on folivores and a phloem feeder. J. Anim. Ecol. 2006, 75, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Harrewijn, P. Resistance Mechanisms of Plant Genotypes to Various Aphid Species. In Aphid-Plant Genotype Interactions; Campbell, R.K., Eikenbary, R.D., Eds.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 117–130. [Google Scholar]
- Dreyer, D.; Campbell, B. Chemical basis of host-plant resistance to aphids. Plant Cell Environ. 1987, 10, 353–361. [Google Scholar]
- Fawe, A.; Abou-Zaid, M.; Menzies, J.G.; Bélanger, R.R. Silicon mediated accumulation of flavonoid phytoalexins in cucumber. Phytopathology 1998, 88, 396–401. [Google Scholar] [CrossRef]
- Hodson, M.; Sangster, A. Observations on the distribution of mineral elements in the leaf of wheat (Triticum aestivum L.), with particular reference to silicon. Ann. Bot. 1988, 62, 463–471. [Google Scholar] [CrossRef]
- Ramirez, C.C.; Niemeyer, H.M. Salivation into sieve elements in relation to plant chemistry: The case of the aphid Sitobion fragariae and the wheat, Triticum aestivum. Entomol. Exp. Appl. 1999, 91, 111–114. [Google Scholar] [CrossRef]
- Oliveira, M.R.V.; Henneberry, T.J.; Anderson, P. History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot. 2001, 20, 709–723. [Google Scholar] [CrossRef]
- Correa, R.S.; Moraes, J.C.; Aaud, A.M.; Carvalho, G.A. Silicon and acibenzolar-S-methyl as resistance inducers in cucumber, against the whitefly Bemisia tabaci (Hemiptera: Aleyrodidae) biotype B. Neotrop. Entomol. 2005, 34, 429–433. [Google Scholar] [CrossRef]
- Reynolds, O.L.; Keeping, M.G.; Meyer, J.H. Silicon-augmented resistance of plants to herbivorous insects: A review. Ann. Appl. Biol. 2009, 155, 171–186. [Google Scholar] [CrossRef]
- Gomes, F.B.; Moraes, J.C.D.; Santos, C.D.D.; Goussain, M.M. Resistance induction in wheat plants by silicon and aphids. Sci. Agric. 2005, 62, 547–551. [Google Scholar] [CrossRef]
- Yang, L.; Han, Y.; Li, P.; Li, F.; Ali, S.; Hou, M. Silicon amendment is involved in the induction of plant defense responses to a phloem feeder. Sci. Rep. 2017, 7, 4232. [Google Scholar] [CrossRef] [PubMed]
- Tayabi, K.; Azizi, P. Influence of silica on rice yield and stem-borer (Chilo supremain) in Rasht/Iran. Pestcides 1984, 18, 20–22. [Google Scholar]
- Costa, R.R.; Moraes, J.C. Effects of silicon acid and of acibenzolar-S-methyl on Schizaphis graminum (Rondani) (Hemiptera: Aphididae) in wheat plants. Neotrop. Entomol. 2006, 35, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Moraes, J.C.; Goussain, M.M.; Basagli, M.A.; Carvalho, G.A.; Ecole, C.C.; Sampaio, M.V. Silicon influence on the tritrophic interaction: Wheat plants, the greenbug Schizaphis graminum (Rondani) (Hemiptera: Aphididae), and its natural enemies, Chrysoperla externa (Hagen) (Neuroptera: Chrysopidae) and Aphidius colemani Viereck (Hymenoptera: Aphidiidae). Neotrop. Entomol. 2004, 33, 619–624. [Google Scholar]
- Salim, M.; Saxena, R. Iron, silica, and aluminum stresses and varietal resistance in rice: Effects on white backed plant hopper. Crop Sci. 1992, 32, 212–219. [Google Scholar] [CrossRef]
- Moraes, J.C.; Goussain, M.M.; Carvalho, G.A.; Costa, R.R. Feeding non-preference of the corn leaf aphid rhopalosiphum maidis (fitch, 1856) (hemiptera: Aphididae) to corn plants (Zea mays L.) treated with silicon. Cienc. E Agrotecnol. 2005, 29, 761–766. [Google Scholar] [CrossRef]
- Calatayud, P.A.; Njuguna, E.; Mwalusepo, S.; Gathara, M.; Okuku, G.; Kibe, A.; Musyoka, B.; Williamson, D.; Ongamo, G.; Juma, G.; et al. Can climate-driven change influence silicon assimilation by cereals and hence the distribution of lepidopteran stem borers in East Africa? Agric. Ecosyst. Environ. 2016, 224, 95–103. [Google Scholar] [CrossRef]
- Redmond, C.T.; Potter, D.A. Silicon fertilization does not enhance creeping bentgrass resistance to cutworms and white grubs. Appl. Turfgrass Sci. 2007, 6, 1–7. [Google Scholar]
- Calatayud, P.A.; Njuguna, E.; Juma, G. Silica in insect-plant interactions. Entomol. Ornithol. Herpetol. 2016, 5, e125. [Google Scholar]
- Gatarayiha, M.C.; Laing, M.D.; Miller, R.M. Combining applications of potassium silicate and Beauveria bassiana to four crops to control two spotted spider mite, Tetranychus urticae Koch. Int. J. Pest Manag. 2010, 56, 291–297. [Google Scholar] [CrossRef]
- Dicke, M.; Van Loon, J.J.A.; Soler, R. Chemical complexity of volatiles from plants induced by multiple attack. Nat. Chem. Biol. 2009, 5, 317–324. [Google Scholar] [CrossRef]
- Kvedaras, O.L.; An, M.; Choi, Y.S.; Gurr, G.M. Silicon enhances natural enemy attraction and biological control through induced plant defences. Bull. Entomol. Res. 2010, 100, 367–371. [Google Scholar] [CrossRef]
- Connick, V.J. The Impact of Silicon Fertilisation on the Chemical Ecology of Grapevine, Vitis Vinifera Constitutive and Induced Chemical Defences against Arthropod Pests and Their Natural Enemies. Master’s Thesis, Charles Sturt University, Albury–Wodonga, Bathurst, NSW, Australia, 2011. [Google Scholar]
- Reynolds, O.L.; Padula, M.P.; Zeng, R.; Gurr, G.M. Silicon: Potential to promote direct and indirect effects on plant defense against arthropod pests in agriculture. Front. Plant Sci. 2016, 7, 1–13. [Google Scholar] [CrossRef]
- Rodrigues, F.A.; McNally, D.J.; Datnoff, L.E.; Jones, J.B.; Labbe, C.; Benhamou, N.; Menzies, J.; Belanger, R. Silicon enhances the accumulation of diterpenoid phytoalexins in rice: A potential mechanism for blast resistance. Phytopathology 2004, 94, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Remus-Borel, W.; Menzies, J.G.; Bélanger, R.R. Silicon induces antifungal compounds in powdery mildew-infected wheat. Physiol. Mol. Plant Pathol. 2005, 66, 108–115. [Google Scholar] [CrossRef]
- Cai, K.; Gao, D.; Luo, S.; Zeng, R.; Yang, J.; Zhu, X. Physiological and cytological mechanisms of silicon-induced resistance in rice against blast disease. Physiol. Plant 2008, 134, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Chen, Q.i.n.; Liu, Q.; Zhang, W.; Ding, R. Exogenous silicon (Si) increases antioxidant enzyme activity and reduces lipid peroxidation in roots of salt-stressed barley (Hordeum vulgare L.). J. Plant Physiol. 2003, 160, 1157–1164. [Google Scholar] [CrossRef]
- Ye, M.; Song, Y.; Long, J.; Wang, R.; Baerson, S.R.; Pan, Z.; Zhu-Salzman, K.; Xie, J.; Cai, K.; Luo, S.; et al. Priming of jasmonate-mediated antiherbivore defense responses in rice by silicon. Proc. Natl. Acad. Sci. USA 2013, 110, E3631–E3639. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Lei, W.; Wen, L.; Hou, M. Silicon-mediated resistance in a susceptible rice variety to the rice leaf folder, Cnaphalocrocis medinalis Guenée (Lepidoptera: Pyralidae). PLoS ONE 2015, 10, e0120557. [Google Scholar] [CrossRef] [PubMed]
- Dubin, H.J.; Rajaram, S. Breeding disease resistant wheats for tropical highlands and lowlands. Ann. Rev. Phytopathol. 1996, 34, 503–526. [Google Scholar] [CrossRef] [PubMed]
- Shephard, M.C. Screening for fungicides. Annu. Rev. Phytopathol. 1997, 25, 189–206. [Google Scholar] [CrossRef]
- Pozza, E.A.; Pozza, A.A.A.; Botelho, D.M.D.S. Silicon in plant disease control. Rev. Ceres 2015, 62, 323–331. [Google Scholar] [CrossRef]
- Menzies, J.; Bowen, P.; Ehret, D.; Glass, A.D.M. Foliar applications of potassium silicate reduce severity of powdery mildew on cucumber, muskmelon, and zucchini squash. J. Am. Soc. Hortic. Sci. 1992, 117, 902–905. [Google Scholar] [CrossRef]
- Miyake, Y.; Takahashi, E. Effect of silicon on the growth of solution-cultured cucumber plant comparative studies on silica nutrition in plants. Comparative studies on silica nutrition in plants. J. Soil Sci. Plant Nutr. 1982, 53, 23–29. [Google Scholar]
- Brugiére, T.; Exley, C. Callose-associated silica deposition in Arabidopsis. J. Trace Elem. Med. Biol. 2017, 39, 86–90. [Google Scholar] [CrossRef]
- Chen, X.-Y.; Kim, J.-Y. Callose synthesis in higher plants. Plant Signal. Behav. 2009, 4, 489–492. [Google Scholar] [CrossRef]
- Ma, J.F. Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci. Plant Nutr. 2004, 50, 11–18. [Google Scholar] [CrossRef]
- Chérif, M.; Asselin, A.; Belanger, R.R. Defense responses induced by soluble silicon in cucumber roots infected by Pythium spp. Phytopathology 1994, 84, 236–242. [Google Scholar] [CrossRef]
- Rahman, A.; Wallis, C.; Uddin, W. Silicon induced systemic defense responses in perennial ryegrass against infection by Magnaporthe oryzae. Phytopathology 2015, 105, 748–757. [Google Scholar] [CrossRef]
- White, W.; White, P. Sugarcane borer resistance in sugarcane as affected by silicon applications in potting medium. J. Am. Soc. Sugar Cane Technol. 2013, 33, 38–54. [Google Scholar]
- Sidhu, J.K.; Stout, M.J.; Blouin, D.C.; Datnoff, L.E. Effect of silicon soil amendment on performance of sugarcane borer, Diatraea saccharalis (Lepidoptera: Crambidae) on rice. Bull. Entomol. Res. 2013, 103, 656–664. [Google Scholar] [CrossRef]
- Vilela, M.; Moraes, J.C.; Alves, E.; Santos-Cividanes, T.M.; Santos, F.A. Induced resistance to Diatraea saccharalis (Lepidoptera: Crambidae) via silicon application in sugarcane. Rev. Colomb. Entomol. 2014, 40, 44–48. [Google Scholar]
- Kvedaras, O.L.; Keeping, M.G. Silicon impedes stalk penetration by the borer Eldana saccharina in sugarcane. Entomol. Exp. Appl. 2007, 125, 103–110. [Google Scholar] [CrossRef]
- Keeping, M.G.; Kvedaras, O.L.; Bruton, A.G. Epidermal silicon in sugarcane: Cultivar differences and role in resistance to sugarcane borer Eldana saccharina. Environ. Exp. Bot. 2009, 66, 54–60. [Google Scholar] [CrossRef]
- Keeping, M.G.; Meyer, J.H.; Sewpersad, C. Soil silicon amendments increase resistance of sugarcane to stalk borer Eldana saccharina Walker (Lepidoptera: Pyralidae) under field conditions. Plant Soil 2013, 363, 297–318. [Google Scholar] [CrossRef]
- Nikpay, A. Improving biological control of stalk borers in sugarcane by applying silicon as a soil amendment. J. Plant Prot. Res. 2016, 56, 394–401. [Google Scholar] [CrossRef]
- Nikpay, A.; Soleyman Nejadian, E.; Goldasteh, S.; Farazmand, H. Response of sugarcane and sugarcane stalk borers Sesamia spp. (Lepidoptera: Noctuidae) to calcium silicate fertilization. Neotrop. Entomol. 2015, 44, 498–503. [Google Scholar] [CrossRef]
- Nikpay, A.; Soleyman Nejadian, E.; Goldasteh, S.; Farazmand, H. Efficacy of silicon formulations on sugarcane stalk borers, quality characteristics and parasitism rate on five commercial varieties. Proc. Nat. Acad. Sci. India Sec. B Biol. Sci. 2017, 87, 289–297. [Google Scholar] [CrossRef]
- Korndörfer, A.P.; Grisoto, E.; Vendramim, J.D. Induction of insect plant resistance to the spittlebug Mahanarva fimbriolata Stål (Hemiptera: Cercopidae) in sugarcane by silicon application. Neotrop. Entomol. 2011, 40, 387–392. [Google Scholar] [PubMed]
- Indhumathi, V.S.; Chandramani, P.; Jayaraj, J.; Chinniah, C.; Mahendran, P.P.; Kumutha, K. Effect of silicon sources against sugarcane early shoot borer Chilo infuscatellus Snellen. Ind. J. Entomol. 2019, 81, 206–209. [Google Scholar] [CrossRef]
- Indhumathi, V.S.; Chandramani, P.; Jayaraj, J.; Chinniah, C.; Mahendran, P.P.; Kumutha, K. Impact of silicon on sugarcane leafhopper, Pyrilla perpusilla Walker by enhancing parasitism of Epiricania melanoleuca (Fletcher). J. Biol. Control 2018, 32, 155–159. [Google Scholar] [CrossRef]
- Nikpay, A.; Soleyman Nejadian, E. Field applications of silicon-based fertilizers against sugarcane yellow mite Oligonychus sacchari. Sugar Tech 2014, 16, 319–324. [Google Scholar] [CrossRef]
- Nikpay, A.; Laane, H.M. Foliar amendment of silicic acid on population of yellow mite, Oligonychus sacchari (Acari: Tetranychidae) and its predatory beetle, Stethorus gilvifrons (Col.: Coccinellidae) on two sugarcane commercial varieties. Persian J. Acarol. 2020, 9, 57–66. [Google Scholar]
- Atencio, R.; Goebel, F.R.; Guerra, A. Effect of Silicon and Nitrogen on Diatraea tabernella Dyar in Sugarcane in Panama. Sugar Tech 2019, 21, 113–121. [Google Scholar] [CrossRef]
- Hou, M.; Han, Y. Silicon-mediated rice plant resistance to the asiatic rice borer (Lepidoptera: Crambidae): Effects of silicon amendment and rice varietal resistance. J. Econ. Entomol. 2010, 103, 1412–1419. [Google Scholar] [CrossRef]
- Han, Y.Q.; Wen, G.H.; Peng, Z.; Zhang, D.Y.; Hou, M.L. Effects of silicon amendment on the occurrence of rice insect pests and diseases in a field test. J. Integr. Agric. 2018, 17, 2172–2181. [Google Scholar] [CrossRef]
- He, W.; Yang, M.; Li, Z.; Qiu, J.; Liu, F.; Qu, X.; Qiu, Y.; Li, R. High levels of silicon provided as a nutrient in hydroponic culture enhances rice plant resistance to brown planthopper. Crop Prot. 2015, 67, 20–25. [Google Scholar] [CrossRef]
- Yang, G.Q.; Zhu, Z.F.; Hu, W.F.; Ge, L.Q.; Wu, J.C. Effects of foliar spraying of silicon and phosphorus on rice (Oryza sativa) plants and their resistance to the white-backed planthopper, Sogatella furcifera (Hemiptera: Delphacidae). Acta Entomol. Sin. 2014, 57, 927–934. [Google Scholar]
- Jeer, M.; Telugu, U.M.; Voleti, S.R.; Padmakumari, A.P. Soil application of silicon reduces of yellow stem borer, Scirpophaga incertullas (Walker) damage in rice. J. Appl. Entomol. 2017, 141, 189–201. [Google Scholar] [CrossRef]
- Catalani, C.G.; Vieira, M.R.; Camargos, L.S.D.; Bomfim, N.C.P.; Agustini, J.A. Effects of potassium silicate application on papaya plants in the two spotted spider mite population. Rev. Bras. Frutic. 2017, 39, e840. [Google Scholar]
- Ramirez-Godoy, A.; Vera-Hoyos, M.P.; Jimenez-Beltran, N.; Restrepo-Diaz, H. Effect of Potassium Silicate Application on Populations of Asian Citrus Psyllid in Tahiti Lime. Am. Soc. Horticul. Sci. 2018, 28, 684–691. [Google Scholar] [CrossRef]
- Dogramaci, M.; Arthurs, S.P.; Chen, J.; Osborne, L. Silicon applications have minimal effects on Scirtothrips dorsalis (Thysanoptera: Thripidae) populations on pepper plant, Capsicum annum L. Fla. Entomol. 2013, 96, 48–54. [Google Scholar] [CrossRef]
- Ribeiro, E.B.; Castellani, M.A.; Andrade Júnior, D.; Prado, R.M.; Souza, L.; Freire, D.S.; Savi, P.J. Biological aspects of the two- spotted spider mite on strawberry plants under silicon application. Hortic. Bras. 2021, 39, 5–10. [Google Scholar] [CrossRef]
- Ranger, C.M.; Singh, A.P.; Frantz, J.M.; Cañas, L.; Locke, J.C.; Reding, M.E.; Vorsa, N. Influence of silicon on resistance of Zinnia elegans to Myzus persicae (Hemiptera: Aphididae). Environ. Entomol. 2009, 38, 129–136. [Google Scholar] [CrossRef]
- Alyousuf, A.; Hamid, D.; Desher, M.A.; Nikpay, A.; Laane, H.M. Effect of Silicic Acid Formulation (Silicon 0.8%) on Two Major Insect Pests of Tomato under Greenhouse Conditions. Silicon 2021. [Google Scholar] [CrossRef]
- Almeida, G.D.; Pratissoli, D.; Zanuncio, J.C.; Vicentini, V.B.; Holtz, A.M.; Serrão, J.E. Calcium silicate and organic mineral fertilizer increase the resistance of tomato plants to Frankliniella schultzei. Phytoparasitica 2009, 37, 225–230. [Google Scholar] [CrossRef]
- Teixeira, N.C.; Valim, J.O.S.; Oliveira, M.G.A.; Campos, W.G. Combined effects of soil silicon and drought stress on host plant chemical and ultrastructural quality for leaf-chewing and sap-sucking insects. J. Agron. Crop Sci. 2020, 206, 187–201. [Google Scholar] [CrossRef]
- Johnson, S.N.; Rowe, R.C.; Hall, C.R. Silicon is an inducible and effective herbivore defence against Helicoverpa punctigera (Lepidoptera: Noctuidae) in soybean. Bull. Entomol. Res. 2020, 110, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.; Moraes, J. Silicon influence on resistance induction against Bemisia tabaci biotype B (Genn.) (Hemiptera: Aleyrodidae) and on vegetative development in two soybean cultivars. Neotrop. Entomol. 2011, 40, 495–500. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jeer, M.; Yele, Y.; Sharma, K.C.; Prakash, N.B. Exogenous application of different silicon sources and potassium reduces pink stem borer damage and improves photosynthesis, yield and related parameters in wheat. Silicon 2021, 13, 901–910. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.L.; Su, J.W.; Wang, Y.L.; Tan, J.F.; Han, Y.L. Effect of nitrogen application combined with silicon on density of Sitobion avenae and contents of biochemical materials of winter wheat at the late growth stage. J. Plant Nutr. Fert. 2013, 19, 832–839. [Google Scholar]
- Assis, F.A.; Moraes, J.C.; Assis, G.A.; Parolin, F.J.T. Induction of caterpillar resistance in sunflower using silicon and Acibenzolar-S-Methyl. J. Agric. Sci. Technol. 2015, 17, 543–550. [Google Scholar]
- Callis-Duehl, K.; Mcauslane, H.; Duehl, A.J.; Levey, D.J. The effects of silica fertilizer as an anti-herbivore defense in cucumber. J. Hort. Res. 2018, 25, 89–98. [Google Scholar] [CrossRef]
- Islam, T.; Moore, B.D.; Johnson, S.N. Novel evidence for systemic induction of silicon defences in cucumber following attack by a global insect herbivore. Ecol. Entomol. 2020, 45, 1373–1381. [Google Scholar] [CrossRef]
- Pinto, D.G.; Aguilar, M.A.G.; Souza, C.A.S.; Silva, D.M.; Siqueira, P.R.; Cao, J.R.; Zanetti, L.V. Alterações isiológicas após aplicação de silício em cacau e sua inluência na preferência por pulgões. Rev. Ceres 2012, 59, 360–367. [Google Scholar] [CrossRef]
- Peixoto, M.L.; Moraes, J.C.; Silva, A.A.; Assis, F.A. Efeito do silício na preferência para oviposição de Bemisia tabaci biótipo B (Genn.) (Hemiptera: Aleyrodidae) em plantas de feijão (Phaseolus vulgaris L.). Cienc. Agrotecnologia 2011, 35, 478–481. [Google Scholar] [CrossRef]
- Islam, T.; Moore, B.D.; Johnson, S.N. Silicon suppresses a ubiquitous mite herbivore and promotes natural enemy attraction by altering plant volatile blends. J. Pest Sci. 2021, 93, 41–48. [Google Scholar]
- Da Silva, V.F.; Moraes, J.C.; Melo, B.A. Influence of silicon on the development, productivity and infestation by insect pests in potato crops. Cienc. Agrotecnologia 2010, 34, 1465–1469. [Google Scholar] [CrossRef]
- Nagaratna, W.; Kalleshwaraswamy, C.M.; Dhananjaya, B.C.; Sharanabasappa; Prakash, N.B. Effect of silicon and plant growth regulators on the biology and fitness of fall armyworm, Spodoptera frugiperda, a recently invaded pest of maize in India. Silicon 2021. [Google Scholar] [CrossRef]
- Moise, E.R.D.; McNeil, J.N.; Hartley, S.E.; Henry, H.A.L. Plant silicon effects on insect feeding dynamics are influenced by plant nitrogen availability. J. Appl. Entomol. 2019, 167, 91–97. [Google Scholar] [CrossRef]
- Mir, S.H.; Rashid, I.; Hussain, B.; Reshi, Z.A.; Assad, R.; Sofi, I.A. Silicon Supplementation of Rescuegrass Reduces Herbivory by a Grasshopper. Front. Plant Sci. 2019, 10, 671. [Google Scholar] [CrossRef] [PubMed]
- Hogan, B.; McDermott, F.; Schmidt, O. Effects of silicon-rich soil amendments on growth, mortality and bark feeding damage of Sitka spruce (Picea sitchensis) seedlings under field conditions. Scand. J. For. Res. 2018, 33, 550–559. [Google Scholar] [CrossRef]
- Massey, F.P.; Hartley, S.E. Physical defences wear you down: Progressive and irreversible impacts of silica on insect herbivores. J. Anim. Ecol. 2009, 78, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Belanger, R.R.; Benhamou, N.; Menzies, J.G. Cytological evidence of an active role of silicon in wheat resistance to powdery mildew (Blumeria graminis f. sp. tritici). Phytopathology 2003, 93, 402–412. [Google Scholar] [CrossRef]
- Hao, Z.; Fayolle, L.; Van Tuinen, D.; Chatagnier, O.; Li, X.; Gianinazzi, S.; Gianinazzi-Pearson, V. Local and systemic mycorrhiza-induced protection against the ectoparasitic nematode Xiphinema index involves priming of defence gene responses in grapevine. J. Exp. Bot. 2012, 63, 3657–3672. [Google Scholar] [CrossRef]
- Belanger, R.R.; Vivancos, J.; Wilkinson, J.A.; Belzile, F.; Menzies, J.G. Silicon infl uence on biotic stress in plants. In Proceedings of the 6th International Conference on Silicon in Agriculture, Stockholm, Sweden, 26–30 August 2014; p. 42. [Google Scholar]
- Heckman, J. Silicon: A Benefi cial Substance. Better Crops 2013, 97, 14–16. [Google Scholar]
- Datnoff, L.E.; Heckman, J.R. Silicon fertilizers for plant disease protection. In Proceedings of the 16th World Fertilizer Congress of CIECRio, Rio de Janeiro, Brazil, 20–24 October 2014; pp. 37–38. [Google Scholar]
- Epstein, E. The anomaly of silicon in plant biology. Proc. Natl. Acad. Sci. USA 1994, 91, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Hattori, T.; Inanaga, S.; Araki, H.; An, P.; Morita, S.; Luxová, M.; Lux, A. Application of silicon enhanced drought tolerance in Sorghum bicolor. Physiol. Plant 2005, 123, 459–466. [Google Scholar] [CrossRef]
- Dannon, E.A.; Wydra, K. Interaction between silicon amendment, bacterial wilt development and phenotype of Ralstonia solanacearum in tomato genotypes. Physiol. Mol. Plant Pathol. 2004, 64, 233–243. [Google Scholar] [CrossRef]
- Diogo, R.V.C.; Wydra, K. Silicon-induced basal resistance in tomato against Ralstonia solanacearum is related to modification of pectic cell wall polysaccharide structure. Physiol. Mol. Plant Pathol. 2007, 70, 120–129. [Google Scholar] [CrossRef]
- Yang, M.; Gusev, E.P.; Ieong, M.; Gluschenkov, O.; Boyd, D.; Chan, K.K.; Kozlowski, P.; D’Emic, C.; Sicina, R.; Jamison, P.; et al. Performance dependence of CMOS on silicon substrate orientation for ultrathin oxynitride and H2O2 gate dielectrics. IEEE Electron. Device Lett. 2003, 24, 339–341. [Google Scholar] [CrossRef]
- Liang, Y.; Si, J.; Römheld, V. Silicon uptake and transport is an active process in Cucumis sativus. New Phytol. 2005, 167, 797–804. [Google Scholar] [CrossRef]
- Liang, Y.; Wong, J.W.C.; Wei, L. Silicon-mediated enhancement of cadmium tolerance in maize (Zea mays L.) grown in cadmium contaminated soil. Chemosphere 2005, 58, 475–483. [Google Scholar] [CrossRef]
- Ghareeb, H.; Becker, A.; Iven, T.; Feussner, I.; Schirawski, J. Sporisorium reilianum infection changes inflorescence and branching architectures of maize. Plant Physiol. 2011, 156, 2037–2052. [Google Scholar] [CrossRef]
- Ghareeb, H.; Bozsó, Z.; Ott, P.G.; Repenning, C.; Stahl, F.; Wydra, K. Transcriptome of silicon-induced resistance against Ralstonia solanacearum in the silicon non-accumulator tomato implicates priming effect. Physiol. Mol. Plant Pathol. 2011, 75, 83–89. [Google Scholar] [CrossRef]
- Ghareeb, H.; Bozsó, Z.; Ott, P.G.; Wydra, K. Silicon and Ralstonia solanacearum modulate expression stability of housekeeping genes in tomato. Physiol. Mol. Plant Pathol. 2011, 75, 176–179. [Google Scholar] [CrossRef]
- Brunings, A.M.; Datnoff, L.E.; Ma, J.F.; Mitani, N.; Nagamura, Y.; Rathinasabapathi, B.; Kirst, M. Differential gene expression of rice in response to silicon and rice blast fungus Magnaporthe oryzae. Ann. Appl. Biol. 2009, 155, 161–170. [Google Scholar] [CrossRef]
- Chain, F.; Côté-Beaulieu, C.; Belzile, F.; Menzies, J.; Bélanger, R. A comprehensive transcriptomic analysis of the effect of silicon on wheat plants under control and pathdogen stress conditions. Mol. Plant Microbe Interact. 2009, 22, 1323–1330. [Google Scholar] [CrossRef]
- Tesfagiorgis, H.B.; Laing, M.D.; Annegarn, H.J. Evaluation of biocontrol agents and potassium silicate for the management of powdery mildew of zucchini. Biol. Control 2014, 73, 8–15. [Google Scholar] [CrossRef]
- Conceicao, C.S.; Felix, K.C.S.; Mariano, R.L.R.; Medeiros, E.V.; Souza, E.B. Combined effect of yeast and silicon on the control of bacterial fruit blotch in melon. Sci. Hort. 2014, 174, 164–170. [Google Scholar] [CrossRef]
- Kiirika, L.M.; Stahl, F.; Wydra, K. Phenotypic and molecular characterization of resistance induction by single and combined application of chitosan and silicon in tomato against Ralstonia solanacearum. Physiol. Mol. Plant Pathol. 2013, 81, 1–12. [Google Scholar] [CrossRef]
- Tesfagiorgis, H.B.; Annegarn, H.J. Evaluation of adjuvants for enhanced management of Podosphaera xanthii using potassium silicate and biocontrol agents. Crop Prot. 2013, 48, 69–75. [Google Scholar] [CrossRef]
- Chang, S.J.; Tzeng, D.D.S.; Li, C.C. Effect of silicon nutrient on bacterial blight resistance of rice (Oryza sativa L.). In Second Silicon in Agriculture Conference; Matoh, T., Ed.; Press-Net: Kyoto, Japan, 2002; pp. 31–33. [Google Scholar]
- Dasgupta, N.; Nandy, P.; Sengupta, C.; Das, S. Protein and enzymes regulations towards salt tolerance of some Indian mangroves in relation to adaptation. Trees Struct. Funct. 2012, 26, 377–391. [Google Scholar] [CrossRef]
- Sahebi, M.; Hanafi, M.M.; Akmar, A.S.N.; Rafii, M.Y.; Azizi, P.; Tengoua, F.F.; Azwa, J.N.M.; Shabanimofrad, M. Importance of silicon and mechanisms of biosilica formation in plants. Biomed. Res. Int. 2015, 2015, 396010. [Google Scholar] [CrossRef] [PubMed]
- Kelman, A. One hundred and one years of research on bacterial wilt. In Bacterial Wilt: Molecular and Ecological Aspects; Prior, P., Allen, C., Elphinstone, J., Eds.; INRA Editions: Paris, France, 1998; pp. 1–5. [Google Scholar] [CrossRef]
- Ram-Kishun, S.; Kishun, R. Loss in yield of tomato due to bacterial wilt caused by Pseudomonas solanacearum. Ind. Phytopathol. 1987, 40, 152–155. [Google Scholar]
- Yabuuchi, E.; Kosako, Y.; Yano, I.; Hotta, H.; Nishiuchi, Y. Transfer of two Burkholderia and an alcaligenes species to Ralstonia gen. Nov.: Proposal of Ralstonia pickettii (Ralston, palleroni and Doudoroff 1973) comb. Nov., Ralstonia solanacearum (Smith 1896) comb. Nov. and Ralstonia eutropha (Davis 1969) comb. Nov. Microbiol. Immunol. 1995, 39, 897–904. [Google Scholar] [CrossRef]
- Samuels, A.L.; Glass, A.D.M.; Ehret, D.L.; Menzies, J.G. Mobility and deposition of silicon in cucumber plants. Plant Cell Environ. 1991, 14, 485–492. [Google Scholar] [CrossRef]
- Kurabachew, H.; Stahl, F.; Wydra, K. Global gene expression of rhizobacteria-silicon mediated induced systemic resistance in tomato (Solanum lycopersicum) against Ralstonia solanacearum. Physiol. Mol. Plant Pathol. 2013, 84, 44–52. [Google Scholar] [CrossRef]
- Ghanmi, D.; Mcnally, D.J.; Benhamou, N.; Menzies, J.G.; Bélanger, R.R. Powdery mildew of Arabidopsis thaliana: A pathosystem for exploring the role of silicon in plant-microbe interactions. Physiol. Mol. Plant Pathol. 2004, 64, 189–199. [Google Scholar] [CrossRef]
- Yoshida, S.; Ohnishi, Y.; Kitagishi, K. Histochemistry of silicon in rice plant III: The presence of cuticle-silica double layer in the epidermal tissue. Soil Sci. Plant Nutr. 1962, 8, 1–5. [Google Scholar] [CrossRef]
- Inanaga, S.; Okasaka, A.; Tanaka, S. Does silicon exist in association with organic compounds in rice plant? Jpn. Soc. Soil Sci. Plant Nutr. 1995, 11, 111–117. [Google Scholar] [CrossRef]
- Kim, S.; Kim, W.; Park, E.; Choi, D. Silicon-induced cell wall fortification of rice leaves: A possible cellular mechanism of enhanced host resistance to blast. Phytopathology 2002, 92, 1095–1110. [Google Scholar] [CrossRef] [PubMed]
- Hayasaka, T.; Fujii, H.; Ishiguro, K. The role of silicon in preventing appressorial penetration by the rice blast fungus. Phytopathology 2008, 98, 1038–1044. [Google Scholar] [CrossRef][Green Version]
- Heine, G.; Tikum, G.; Horst, W. The effect of silicon on the infection by and spread of Pythium aphanidermatum in single roots of tomato and bitter gourd. J. Exp. Bot. 2007, 58, 569–577. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Van der Does, D.; Zamioudis, C.; Leon-Reyes, A.; Van Wees, S.C.M. Hormonal modulation of plant immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 489–521. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, D.; Chen, J.; Yang, Y.; Huang, Z.; Huang, D.; Wang, X.C.; Huang, R. Tomato stress-responsive factor TSRF1 interacts with ethylene responsive element GCC box and regulates pathogen resistance to Ralstonia solanacearum. Plant Mol. Biol. 2004, 55, 825–834. [Google Scholar] [CrossRef]
- Fauteux, F.; Chain, F.; Belzile, F.; Menzies, J.G.; Bélanger, R.R. The protective role of silicon in the Arabidopsis–powdery mildew pathosystem. Proc. Natl. Acad. Sci. USA 2006, 103, 17554–17559. [Google Scholar] [CrossRef]
- Iwai, T.; Miyasaka, A.; Seo, S.; Ohashi, Y. Contribution of ethylene biosynthesis for resistance to blast fungus infection in young rice plants. Plant Physiol. 2006, 142, 1202. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Lin, Y.M.; Chao, T.C.; Wang, J.F.; Liu, A.C.; Ho, F.I.; Cheng, C.P. Virus-induced gene silencing reveals the involvement of ethylene-, salicylic acid- and mitogen-activated protein kinase-related defense pathways in the resistance of tomato to bacterial wilt. Physiol. Plant. 2009, 136, 324–335. [Google Scholar] [CrossRef]
- Kim, Y.H.; Khan, A.L.; Kim, D.H.; Lee, S.Y.; Kim, K.M.; Waqas, M.; Jung, H.Y.; Shin, J.H.; Kim, J.G.; Lee, I.J. Silicon mitigates heavy metal stress by regulating P-type heavy metal ATPases, Oryza sativa low silicon genes, and endogenous phytohormones. BMC Plant Biol. 2014, 14, 13. [Google Scholar] [CrossRef]
- De Vos, M.; Van Oosten, V.R.; Van Poecke, R.M.P.; Van Pelt, J.A.; Pozo, M.J.; Mueller, M.J.; Buchala, A.J.; Metraux, J.-P.; Van Loon, L.C.; Dicke, M.; et al. Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol. Plant Microbe Interact. 2005, 18, 923–937. [Google Scholar] [CrossRef] [PubMed]
- Jadhao, K.R.; Bansal, A.; Rout, G.R. Silicon amendment induces synergistic plant defense mechanism against pink stem borer (Sesamia inferens Walker.) in finger millet (Eleusine coracana Gaertn.). Sci. Rep. 2020, 10, 4229. [Google Scholar] [CrossRef]
- Conrath, U. Systemic acquired resistance. Plant Signal. Behav. 2006, 4, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Cai, K.; Gao, D.; Chen, J.; Luo, S. Probing the mechanisms of silicon-mediated pathogen resistance. Plant Signal. Behav. 2009, 4, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, A.A.; Debona, D.; Bernardeli, A.M.A.; Rodrigues, F.A. Defense-related enzymes in soybean resistance to target spot. J. Phytopathol. 2015, 163, 731–742. [Google Scholar] [CrossRef]
- Shetty, R.; Jensen, B.; Shetty, N.P.; Hansen, M.; Hansen, C.W.; Starkey, K.R.; Jorgensen, H.J.L. Silicon induced resistance against powdery mildew of roses caused by Podosphaera pannosa. Plant Pathol. 2012, 61, 120–131. [Google Scholar] [CrossRef]
- Silva, W.L.D.; Cruz, M.F.A.; Fortunato, A.A.; Rodrigues, F. Histochemical aspects of wheat resistance to leaf blast mediated by silicon. Sci. Agric. 2015, 72, 322–327. [Google Scholar] [CrossRef]
- Rodrigues, F.A.; Jurick, W.M.; Datnoff, L.E.; Jones, J.B.; Rollins, J.A. Silicon infl uences cytological and molecular events in compatible and incompatible rice-Magnaporthe grisea interactions. Physiol. Mol. Plant Pathol. 2005, 66, 144–159. [Google Scholar] [CrossRef]
- Song, A.; Xue, G.; Cui, P.; Fan, F.; Liu, H.; Yin, C.; Sun, W.; Liang, Y. The role of silicon in enhancing resistance to bacterial blight of hydroponic- and soil-cultured rice. Sci. Rep. 2016, 6, 24640. [Google Scholar] [CrossRef] [PubMed]
- Brisson, L.F.; Tenhaken, R.; Lamb, C. Function of oxidative crosslinking of cell wall structural proteins in plant disease resistance. Plant Cell 1994, 6, 1703–1712. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.; Ye, X. Induction of chitinases in tobacco plants systemically protected against blue mold by Peronospora tabacina or tobacco mosaic virus. Phytopathology 1992, 82, 119–123. [Google Scholar] [CrossRef]
- Shewry, P.R.; Lucas, J.A. Plant proteins that confer resistance to pests and pathogens. Adv. Bot. Res. 1997, 26, 135–192. [Google Scholar]
- Qin, G.Z.; Tian, S.P. Enhancement of biocontrol activity of Cryptococcus laurentii by silicon and the possible mechanisms involved. Phytopathology 2005, 95, 69–75. [Google Scholar] [CrossRef]
- Bi, Y.; Tian, S.P.; Guo, Y.R.; Ge, Y.H.; Qin, G.Z. Sodium silicate reduces postharvest decay on Hami melons: Induced resistance and fungistatic effects. Plant Dis. 2006, 90, 279–283. [Google Scholar] [CrossRef]
- Dallagnol, L.J.; Rodrigues, F.A.; DaMatta, F.M.; Mielli, M.V.B.; Pereira, S.C. Deficiency in silicon uptake aff ects cytological, physiological, and biochemical events in the rice-Bipolaris oryzae interaction. Phytopathology 2011, 101, 92–104. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, L.; Zhao, J.; Bi, Y. Use of silicon oxide and sodium silicate for controlling Trichothecium roseum postharvest rot in Chinese cantaloupe (Cucumis melo L.). Int. J. Food Sci. Technol. 2007, 42, 1012–1018. [Google Scholar] [CrossRef]
- Xavier, M.S.F.; Rodrigues, F.A.; Domiciano, G.B.; Oliveira, H.V.; Silveira, P.R.; Moreira, W.R. Wheat resistance to leaf blast mediated by silicon. Aust. Plant Pathol. 2011, 40, 28–38. [Google Scholar]
- Polanco, L.R.; Rodrigues, F.A.; Nascimento, K.J.; Cruz, M.F.; Curvelo, C.R.; DaMatta, F.M.; Vale, F.X.R. Photosynthetic gas exchange and antioxidative system in common bean plants infected by Colletotrichum lindemuthianum and supplied with silicon. Trop. Plant Pathol. 2014, 39, 35–42. [Google Scholar] [CrossRef]
- Domiciano, G.P.; Cacique, I.S.; Freitas, C.C.; Filippi, M.C.C.; DaMatta, F.M.; Do Vale, F.X.R.; Rodrigues, F.A. Alterations in gas exchange and oxidative metabolism in rice leaves infected by Pyricularia oryzae are attenuated by silicon. Phytopathology 2015, 105, 738–747. [Google Scholar] [CrossRef]
- Schurt, D.A.; Cruz, M.F.; Nascimento, K.J.; Filippi, M.C.; Rodrigues, F.A. Silicon potentiates the activities of defense enzymes in the leaf sheaths of rice plants infected by Rhizoctonia solani. Trop. Plant Pathol. 2014, 39, 457–463. [Google Scholar] [CrossRef]
- Dallagnol, L.; Rodrigues, F.; Pascholati, S.; Fortunato, A.; Camargo, L. Comparison of root and foliar applications of potassium silicate in potentiating post-infection defences of melon against powdery mildew. Plant Pathol. 2015, 64, 1085–1093. [Google Scholar] [CrossRef]
- Nwugo, C.C.; Huerta, A.J. The effect of silicon on the leaf proteome of rice (Oryza sativa L.) plants under cadmium-stress. J. Proteome Res. 2011, 10, 518–528. [Google Scholar] [CrossRef]
- Watanabe, S.; Shimoi, E.; Ohkama, N.; Hayashi, H.; Yoneyama, T.; Yazaki, J.; Fujii, F.; Shinbo, K.; Yamamoto, K.; Sakata, K.; et al. Identifi cation of several rice genes regulated by Si nutrition. Soil Sci. Plant Nutr. 2004, 50, 1273–1276. [Google Scholar] [CrossRef]
- Kauss, K.; Franke, R.; Gilbert, S.; Dietrich, A.; Kroger, N. Silica deposition by a strongly cationic proline-rich protein from systemically resistant cucumber plants. Plant J. 2003, 33, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Van Bockhaven, J.; Spíchal, L.; Novák, O.; Strnad, M.; Asano, T.; Kikuchi, S.; Höfte, M.; DeVleesschauwer, D. Silicon induces resistance to the brown spot fungusCochliobolus miyabeanus by preventing the pathogen from hijacking the rice ethylenepathway. New Phytol. 2015, 206, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.P.; Chao, D.Y.; Lin, H.X. Understanding abiotic stress tolerance mechanisms: Recent studies on stress response in rice. J. Integr. Plant Biol. 2007, 49, 742–750. [Google Scholar] [CrossRef]
- Lucas, S.; Durmaz, E.; Akpinar, B.A.; Budak, H. The drought response displayed by a DRE-binding protein from Triticum dicoccoides. Plant Physiol. Biochem. 2011, 49, 346–351. [Google Scholar] [CrossRef]
- Qin, F.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Achievements and challenges in understanding plant abiotic stress responses and tolerance. Plant Cell Physiol. 2011, 52, 1569–1582. [Google Scholar] [CrossRef]
- Gregoire, C.; Remus-Borel, W.; Vivancos, J.; Labbe, C.; Belzile, F.; Belanger, R.R. Discovery of a multigene family of aquaporin silicon transporters in the primitive plant Equisetum arvense. Plant J. 2012, 72, 320–330. [Google Scholar] [CrossRef]
- Wang, H.S.; Yu, C.; Fan, P.-P.; Bao, B.-F.; Li, T.; Zhu, Z.-J. Identification of two cucumber putative silicon transporter genes in Cucumis sativus. J. Plant Growth Regul. 2015, 34, 332–338. [Google Scholar] [CrossRef]
- Mitani, N.; Yamaji, N.; Ago, Y.; Iwasaki, K.; Ma, J.F. Isolation and functional characterization of an influx silicon transporter in two pumpkin cultivars contrasting in silicon accumulation. Plant J. 2011, 66, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Yamaji, N.; Mitani-Ueno, N. Transport of silicon from roots to panicles in plants. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2011, 87, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Epstein, E.; Bloom, A.J. Mineral Nutrition of Plants: Principles and Perspectives, 2nd ed.; Sinuar Associates Inc. Publishers: Saunderland, MA, USA, 2005; pp. 201–240. [Google Scholar]
- Guntzer, F.; Keller, C.; Meunier, J.D. Benefits of plant silicon for crops: A review. Agron. Sustain. Dev. 2012, 32, 201–213. [Google Scholar] [CrossRef]
- Verma, K.K.; Song, X.-P.; Li, D.-M.; Singh, M.; Rajput, V.D.; Malviya, M.K.; Minkina, T.; Singh, R.K.; Singh, P.; Li, Y.-R. Interactive role of silicon and plant—Rhizobacteria mitigating abiotic stresses: A new approach for sustainable agriculture and climate change. Plants 2020, 9, 1055. [Google Scholar] [CrossRef]
| Crop | Pests | Pest Species | Adaptive Mechanisms | Source |
| Sugarcane (Saccharum officinarum L.) | Stalk borer | Diatraea saccharalis (Lepidoptera: Crambidae) | Enhanced Si accumulation and relative growth rate and decreased boring success of sugarcane borer larvae and feeding injury. Upgraded cuticle thickening and crystals accumulation on the leaf stomata. | [95,96,97] |
| African stalk borer | Eldana saccharina (Lepidoptera: Pyralidae) | Si-amended plants significantly enhanced the accumulation of Si in soil and plant organs relative to normal plants, and the outer rind was harder than the control. Treated plants reduced borer penetration, stalk injury, and gain of larval mass. Si directly supported the resistance of E. saccharina through a decreased larval growth rate and feeding injury to the crop plants and indirectly supported it by delayed stalk penetration, resulting mostly in an enhanced exposure time frequency of mature larvae to natural enemies. | [98,99,100] | |
| Stalk borer | Sesamia spp. (Lepidoptera: Noctuidae) | Si increased the tolerance efficiency of sugarcane against stalk borers. The significant loss on borer population and damage but the major loss in the stalk injury (%), bored internodes, moth exit holes, and length of borer tunnel and number of larvae and pupae per 100 stalks were monitored in the sensitive cultivar. It enhanced cane and juice quality parameters and efficiency of parasitism. | [101,102,103] | |
| Spittlebug | Mahanarva fimbriolata (Hem.: Cercopidae) | Si enhanced the uptake, accumulation, and nymphal mortality. It totally depended on the sugarcane cultivars. The duration of pre-oviposition, fecundity, and egg viability were found to be unchanged by Si amendment. | [104] | |
| Internode borer | Chilo infuscatellus (Lepidoptera: Crambidae) | Si reduced the damage incidences and was significantly effective against early shoot borers. | [105] | |
| Leafhopper | Pyrilla perpusilla | The Pyrilla population was less in the Si-applied field, and parasitism (%) increased. The Pyrilla population reduced by an increment of E. melanoleuca parasitism with Si amendment. | [106] | |
| Yellow mite | Oligonychus sacchari (Acari: Tetranychidae) | Significant differences were found in Si and control groups of mite and predatory beetle populations. The population density of mites decreased in all the Si-applied categories as compared to control plants. It is the potential element for the management of mite injury and should be applied with other management approaches. | [107,108] | |
| Stalk borer | Diatraea tabernella (Lepidoptera: Pyralidae) | The amendment of Si-based products decreased internodes borer (about 50%) loss. | [109] | |
| Rice (Oryza sativa L.) | Asiatic stem borer | Chilo suppressalis (Lepidoptera: Crambidae) | Si-applied plants enhanced Si concentration relative to normal plants and reduced borer penetration, weight increase, stem injury, and prolonged penetration time and larval behavior. Plant mortality by stem borer, leaf folder, and population size of the plant hopper were positively reduced. The results showed that the application of Si may provide substantial protective capacity from a few of the rice pests during field conditions. | [110,111] |
| Brown planthopper | Nilaparvata lugens (Hemiptera: Delphacidae) | The higher dose of Si had no symptoms on the morphological traits. It is the major element that restricts brown planthopper (BPH) response in rice–BPH interactions, and it is more beneficial for non-pesticide BPH control. | [112] | |
| White-backed planthopper | Sogatella furcifera (Hemiptera: Delphacidae) | Increased Si content in the upper and lower sides of rice leaves in the foliar spray of Si. Sufficient Si cells were found around the stomata. The oxalic acid and soluble sugar content were enhanced significantly. The number of eggs laid by per female of S. furciferafed was reduced. | [113] | |
| Yellow stem borer | Scirpophaga incertulas (Lepidoptera:Crambidae) | All the soil treatments reduced damage by YSB at vegetative and reproductive phases across five varieties as compared to the control. Si revealed the enhanced deposition of Si in cell walls and a two- to five-fold increase in Si content across treatments. The histological studies showed the rupture of the peritrophic membrane, increased vacuolation, disintegration of columnar cells, and discharge of cellular contents into the gut lumen due to abrasion of midgut epithelium, as compared to the control where the columnar cells and midgut lining were intact. | [114] | |
| Papaya (Carica papaya L.) | Spotted spider mite | Tetranychus urticae (Acari: Tetranychidae) | Plant leaves were performed to investigate the physiological parameters that indicate the activation of the defense strategy of plants. Si induced the formation of plant defense substances decreasing, the net reproduction rate. | [115] |
| Tahiti Lime (Citrus spp.) | Asian Citrus Psyllid | Diaphorina citri (Homoptera: Liviidae) | The use of Si in seedlings and trees infected Asian citrus psyllid (ACP) oviposition, causing a loss of about 60%. It did not affect the macro-micro nutrient profile of plants, with the exception of the foliar application. | [116] |
| Pepper (Capsicum annum L.) | Chilli Thrips | Scirtothrips dorsalis (Thysanoptera: Thripidae) | A very low impact of Si on the leaf morphological injury and numbers of thrips restored from diseased plants were observed. Jasmonic acid as a plant defense elicitor did not change the proportion of the leaves that sustained thrips injury. Plant roots absorb Si in the soil but are not distributed or translocated to the other plant organs, i.e., leaf and shoot. No significant effects were observed in the plant biomass. | [117] |
| Strawberry (Fragaria × ananassa) | Spotted spider mite | Tetranychus urticae (Acari: Tetranychidae) | Si prolonged the frequency of some immature phases of the mites in parental and F1 generations; no changes were found at the complete biological cycle. The time of pre-oviposition and oviposition and the longevity of the parental generation and the longevity and oviposition of the F1 generation of the two-spotted spider mite were negatively affected by the addition of Si. | [118] |
| Zinnia elegans | Aphid | Myzus persicae (Hemiptera: Aphididae) | No changes were found at the duration of the pre-reproductive and survivorship of M. persicae by Si, but the total cumulative fecundity and the intrinsic rate of increase (r(m)) were slightly decreased on Z. elegans plants subjected to Si. Si content increased in plant leaves. Phenolics compounds and guaiacol peroxidase (GPX) activity were slightly affected. | [119] |
| Tomato (Solanum lycopersicum L.) | Silver whitefly | Bemisia tabaci (Hemiptera: Aleyrodidae) | Si reduced the population of immature whiteflies on tomato plants. Foliar spray was more efficient in decreasing the density of population of these pests as compared to Si irrigation. | [120] |
| Leaf miner | Tuta absoluta (Lepidoptera: Gelechiidae) | A potential impact of Si on crops for increasing plant vigor and tolerance to pest injury was observed. Si reduced the population of immature tomato leaf miners on tomato crops. | [120] | |
| Tomato (Lycopersicon esculentum Mill.) | Cotton thrips | Frankliniella schultzei (Thysanoptera: Thripidae) | Si enhanced the number of lesions and the mortality of nymphs, reduced the injury on tomato leaves, and increased the tolerance strategy to pests. | [121] |
| Collard greens (Brassica oleracea) | Diamond back moth | Plutella xylostella (Lepidoptera: Plutellidae) | Nutritional variations mediated by stress and Si in fiber, LWC, soluble N, and glucosinolates did not enhance insect activities in any feeding guild. | [122] |
| Cabbage aphid | Brevicoryne brassicae (Hemiptera: Aphididae) | Si improved the resistance capacity of stress and herbivore stresses. | [122] | |
| Soybean (Glycine max L.) | Budworm | Helicoverpa punctigera (Lepidoptera: Noctuidae) | Herbivory decreased leaf biomass in Si-applied and normal plants compared to herbivore-free plants. Si and herbivory enhanced the Si level. It decreased H. punctigera relative growth rates. | [123] |
| Silver whitefly | Bemisia tabaci (Hemiptera: Aleyrodidae) | No effects were found on silverleaf whitefly oviposition, but significant mortality in nymphs was found. Si enhanced the resistance degree to silverleaf whitefly and down-regulated the phenolic compounds, but no effect on lignin formation and the vegetative growth phase was observed. However, an enhanced tolerance capacity to plants was observed. | [124] | |
| Wheat (Triticum aestivum L.) | Pink stem borer | Sesamia inferens (Lepidoptera: Noctuidae) | Si enhanced the photosynthetic performance, biomass, and productivity. | [125] |
| Aphid | Schizaphis graminum (Hemiptera: Aphididae) | The aphid’s intrinsic rate of population increased after seedling emergence and the enzymatic activities, i.e., POD, PPLO, and PAL associated in the plant defense mechanisms. | [64] | |
| Grain Aphid | Sitobion avenae (Hemiptera: Aphididae) | The density of wheat aphids was enhanced during N application, which closely correlates to the losses of the average soluble sugar and total phenolic content. The effects of the Si on the reduction in population density of aphids would be associated to the increment of the average contents of soluble sugar, phenolic compounds, and tannin contents of wheat leaves and ears. | [126] | |
| Sunflower (Helianthus annuus L.) | Bordered patch | Chlosyne lacinia (Lepidoptera: Nymphalidae) | Reduced weight of the caterpillars at the first and second week of age was observed. Si increased the distribution of the element and decreased lignin content. Negative correlations were found in Si and larval weight. It is an alternative strategy that can effectively integrate into the management of pest in crops. | [127] |
| Cucumber (Cucumis sativa L.) | Silver whitefly | Bemisia tabaci (Hemiptera: Aleyrodidae) | Si-treated plant leaves were less injured as compared to normal plants. No positive signs were found in treated and normal plants regarding lignin content, nutritional elements, water status, trichome density, and carbon and nitrogen levels. Volatile organic compounds and indole content increased for plant defense priming, and cellulose content was reduced. | [62,128] |
| Cucumber (Cucumis sativus L.) | Cotton Bollworm | Helicoverpa armigera (Lepidoptera: Noctuidae) | Herbivory positively enhanced the accumulation of Si in infected plant leaves. The use of Si upregulated Si and the C:N ratio while reducing the larval relative consumption and the relative growth rate in the in situ assays. | [129] |
| Cocoa (Toxoptera aurantii) | Aphid | Toxoptera aurantii (Aphididae) | The efficiency of the chlorophyll fluorescence yield of PSII (Fv/Fm), photosynthetic responses, and total soluble phenol activities were significantly enhanced. The amendment of Si did not affect the morphological performance index. | [130] |
| Bean (Phaseolus vulgaris L.) | Silver whitefly | Bemisia tabaci (Hemiptera: Aleyrodidae) | No changes were observed in the oviposition of the whitefly and the nymph development as well as the phenol levels after Si amendment. | [131] |
| Bean (Phaseolus vulgaris L.) | Spider mite | Tetranychus urticae (Acari: Tetranychidae) | Si suppressed the T. urticae egg-laying, population growth, and leaflet damage and slightly mitigated T. urticae-induced losses in photosynthetic responses. | [132] |
| Potato (Solanum tuberosum L.) | Beetle | Diabrotica speciosa (Chrysomelidae) | No significant interactions were found between Si and crop parameters. The incidence of beetles and aphids was not influenced by Si application and neither was the growth, development, and final output of the crop plants. | [133] |
| Grape (Vitis vinifera L.) | Grapevine moth | Phalaenoides glycinae (Lepidoptera: Noctuidae) | Application of Si may also indirectly affect plant pests through induced chemical defenses by altering and increasing the production of herbivore-induced plant volatiles (HIPVs). It plays a major role in induced plant defense strategies activated by herbivore feeding or oviposition. | [77] |
| Maize (Zea mays L.) | Fall armyworm | Spodoptera frugiperda (Lepidoptera: Noctuidae) | Si reduced the larval weight, pre-pupal weight, pupal weight and larval survival, and fecundity and fertility. The biological characteristics of S. frugiperda were non-significantly correlated with increasing levels of Si, phenols, tannins, and potassium levels in plant leaves. | [134] |
| True armyworm | Pseudeletia unipuncta (Lepi-doptera: Noctuidae) | Effectively decreased the palatability and digestibility of the plant leaves and thus impacted nutrient uptake by insect herbivores. The addition of Si increased larval mortality as compared to the control because early instars with poorly developed mandibles could not feed effectively. | [135] | |
| Rescuegrass (Bromus catharticus) | Grasshopper | Oxya grandis (Orthoptera: Acrididae) | Si enhanced more than 12 times the higher supplementation treatments. The maximum dose of Si in Si-rich plants did not affect the morphological structure of the phytoliths. | [136] |
| Sitka spruce (Picea sitchensis) | Large pine weevil | Hylobius abietis (Coleoptera: Curculionidae) | No significant effects were shown on the growth or mortality of plants after Si application. Bark Si content was found to be similar as compared to normal seedlings. | [137] |
| Ryegrass (Lolium perenne L.) | African Armyworm | Spodoptera exempta (Lepidoptera: Noctuidae) | Si decreased the digestibility of plant leaves and decreased the functionality with S. exempta-ingested food to body mass and the amount of nitrogen absorbed from their food, leading to a decreased rate of insect growth. | [138] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verma, K.K.; Song, X.-P.; Tian, D.-D.; Guo, D.-J.; Chen, Z.-L.; Zhong, C.-S.; Nikpay, A.; Singh, M.; Rajput, V.D.; Singh, R.K.; et al. Influence of Silicon on Biocontrol Strategies to Manage Biotic Stress for Crop Protection, Performance, and Improvement. Plants 2021, 10, 2163. https://doi.org/10.3390/plants10102163
Verma KK, Song X-P, Tian D-D, Guo D-J, Chen Z-L, Zhong C-S, Nikpay A, Singh M, Rajput VD, Singh RK, et al. Influence of Silicon on Biocontrol Strategies to Manage Biotic Stress for Crop Protection, Performance, and Improvement. Plants. 2021; 10(10):2163. https://doi.org/10.3390/plants10102163
Chicago/Turabian StyleVerma, Krishan K., Xiu-Peng Song, Dan-Dan Tian, Dao-Jun Guo, Zhong-Liang Chen, Chang-Song Zhong, Amin Nikpay, Munna Singh, Vishnu D. Rajput, Rupesh Kumar Singh, and et al. 2021. "Influence of Silicon on Biocontrol Strategies to Manage Biotic Stress for Crop Protection, Performance, and Improvement" Plants 10, no. 10: 2163. https://doi.org/10.3390/plants10102163
APA StyleVerma, K. K., Song, X.-P., Tian, D.-D., Guo, D.-J., Chen, Z.-L., Zhong, C.-S., Nikpay, A., Singh, M., Rajput, V. D., Singh, R. K., Minkina, T., & Li, Y.-R. (2021). Influence of Silicon on Biocontrol Strategies to Manage Biotic Stress for Crop Protection, Performance, and Improvement. Plants, 10(10), 2163. https://doi.org/10.3390/plants10102163











