Abstract
Euphorbia genus (Euphorbiaceae family), which is the third largest genus of angiosperm plants comprising ca. 2000 recognized species, is used all over the world in traditional medicine, especially in the traditional Chinese medicine. Members of this taxa are promptly recognizable by their specialized inflorescences and latex. In this review, an overview of Euphorbia-derived natural products such as essential oils, extracts, and pure compounds, active in a broad range of biological activities, and with potential usages in health maintenance, is described. The chemical composition of essential oils from Euphorbia species revealed the presence of more than 80 phytochemicals, mainly oxygenated sesquiterpenes and sesquiterpenes hydrocarbons, while Euphorbia extracts contain secondary metabolites such as sesquiterpenes, diterpenes, sterols, flavonoids, and other polyphenols. The extracts and secondary metabolites from Euphorbia plants may act as active principles of medicines for the treatment of many human ailments, mainly inflammation, cancer, and microbial infections. Besides, Euphorbia-derived products have great potential as a source of bioactive extracts and pure compounds, which can be used to promote longevity with more health.
1. Introduction
The genus Euphorbia (Euphorbiaceae) is the third major genus of flowering plants, with 1836 accepted species [1,2], subdivided into many subgenera and sections. This genus has a worldwide distribution and can be found in all temperate and tropical regions. Also, this group of plants is characterized by an extraordinary variety of forms, from small ephemerals to several forms of herbaceous annuals or perennials, big shrubs, small trees, cushion-forming subshrubs, and cactus-like succulents [3]. From the 243 Euphorbia species assessed by the IUCN Red List of Threatened species, 170 (70%) are threatened with extinction (categories vulnerable, endangered, and critically endangered) [4].
More than 5% of species of Euphorbia are used in traditional medicine, mainly as emetic and purgative agents, to treat digestive and respiratory disorders, skin and inflammatory conditions, migraine, intestinal parasites and gonorrhoea, and as wart cures [5,6,7,8,9]. The usable parts of the Euphorbia species include roots, seeds, latex, wood, barks, leaves, and whole plants [5,6,7,8,9]. A brief overview of traditional medicine applications of Euphorbia is described in Section 2.
Euphorbia species have these curative properties due to the presence of various phytochemicals, which constitute the secondary metabolites of these plants [1,10,11,12,13,14,15,16,17]. They belong mainly to the terpenoids, flavonoids and polyphenols classes which also exhibit a great variety of biological effects such as cytotoxic, mammalian mitochondrial respiratory chain inhibition, HIV-1 and bacterial infection inhibition, anti-inflammatory, multidrug resistance modulators [13,18,19,20,21,22,23]. In fact, there is a good attention in Euphorbia-derived metabolites mainly because of the diterpene ingenol mebutate identified on E. peplus L. (as well as on E. lathyris L., E. nivulia Buch.-Ham., E. esula L., E. antiquorum L., E. serpens Kunth, and E. fischeriana Steud.), and is the active ingredient of Picato® medicine used in topical therapy against the precancerous skin condition actinic keratosis [24,25,26]. However, some Euphorbia compounds are toxic, resulting from an evolutionary strategy of plant defence against predators (e.g., herbivores), compounds that have a caustic and irritating effect to the skin and promote tumours [10,27].
Euphorbia plants are easily distinguishable by their toxic and highly skin irritant milky latex and particular inflorescences, designated as cyathia [28,29], and are widely used as ornamental plants, such as E. milii Des Moul., E. tirucalli L., and E. lactea Roxb [30]. The latex is the most valuable product obtained from Euphorbia species despite being toxic, it contains several biologically active natural compounds, such as triterpenoids [31]. Besides, latex is used in commercially valuable products like paints and natural rubber (intisy rubber obtained from E. intisy Drake) [30,32].
Secondary metabolites contained in Euphorbia plants also potentiate their use for food preservation. According to Toro-Vazquez et al. [33], candelilla wax obtained from the leaves of some species of Euphorbia found in Northern Mexico and the Southwest of the United States was recognized by the Food and Drug Administration (FDA) as a food additive with gelling properties, forming oleo-gels together with vegetable oils. According to EU regulations, candelilla wax is assigned by E902 additive code, and it is also an allowed glazing agent, applied on the surface of confectionery, nuts, wafers, coffee grains, dietary supplements, and fresh fruit [34].
Taking into account the great interest of the Euphorbia plants, we aim to touch on the chemical composition of essential oils, the therapeutic potential, in vitro, in vivo, and clinical trials of Euphorbia extracts and the pure compounds. We adopt the Latin binomial taxonomic name of the Euphorbia species considered by the Plant List database. When it does not match with the taxonomic name indicated in the bibliographic reference, the synonym will be shown in parentheses.
2. Traditional Medicine Uses of Euphorbia Plants
The Euphorbia genus is well-known to involve several plants used in folk medicine in different parts of the world, especially in traditional Chinese medicine [5,7,9]. Moreover, a recent study discriminated the global geographical distribution regarding uses of Euphorbia plants in traditional medicine [6]. In this regard, three particular uses were most often detected, such as (1) treatments of digestive system disorders (very globally frequent excepting Australasia); (2) as remedies for infections/infestations (mainly in Southern Africa and America, Pacific, Asia-tropical, and Asia-temperate); and (3) for treating skin/subcutaneous cellular tissue disorders (particularly in Australasia, Europe, Asia, and Northern America). On the other hand, within the 33 species with citations in folk practices worldwide, the three most-referenced plants used as traditional medicines were E. hirta L., E. thymifolia L., and E. lathyris [6].
Euphorbia hirta whole plant has been employed in Burundi, China, Philippines, and Nigeria to manage diarrhoea [35,36,37,38], while E. hirta decoction is used in Vietnam, India, and Mozambique to treat dysentery [39,40,41] and to treat bronchitis/asthma/coughs in Nepal, Australia, the South Western United States, and Hawaii [6,39,42]. Additionally, the latex from E. hirta is also applied to treat skin diseases and fever mostly in Asia [6] and to treat gonorrhoea in Malaysia [43] and other conditions such as malaria, candidiasis, and ringworm infections [6]. Populations aroundthe Vellore District of Tamil Nadu, India, use decoction of the E. hirta whole plant to treat poisonous snakebites (topically and orally administration) [44].
Despite the registered abortifacient properties of E. thymifolia decoctions in Chile, its latex or leaf decoctions have been recorded as lactation stimulants in different continents [45]. In the case of E. lathyris, emetic and purgative actions have been described in Europe as well as its seeds used to treat snakebites, ascites, schistosomiasis, and hydropsy [38,46].
Euphorbia maculata L. in Northern America is used for the treatment of corneal opacities and warts [47], while in China, it is used to treat blood disorders (e.g., haematuria, haemoptysis, epistaxis, and hemafecia), carbuncles, and wounds [38]. Euphorbia denticulata Lam. and E. macrocarpa Boiss. & Buhse are also used for wound healing in Turkey [48], and a similar use is reported in Ethiopia for E. heterophylla L. and E. prostrata Aiton [49].
The decoction, unguent, or hot steam of other Euphorbia species are used on inflammation conditions, such as E. corollata L. (for dropsy), E. marginata Pursh, and E. antiquorum (for swellings) [6]. Similarly, E. antiquorum is utilized in Vietnam to alleviate toothache events [41] as well as for treating cutaneous dropsy, cutaneous infections, cancer, and liver ailment [50]. E. tirucalli L. and E. ingens E.Mey. ex Boiss. like E. lathyris, can be used as an emetic against snakebites [39,51]. A recent review has been published showing that E. tirucalli (whole plant and its parts individually separated) has some records in South America, India, the Middle East, and Africa regarding beneficial effects on leprosy, syphilis, cancer, asthma, and intestinal parasites [51]. The same research group [52] also published a review where they report the various applications in traditional medicine of E. neriifolia L. Its latex is used as a carminative and expectorant, as well as in the treatment of tumours, abdominal and skin problems, leprosy, asthma, and kidney stones, while the roots are used in the treatment of scorpion stings and snake bites. The leaves can also be used as carminative and in the treatment of pain, inflammation, bronchial infections and lack of appetite [52]. Euphorbia helioscopia L. is used in the traditional Chinese medicine in situations of bacillary dysentery, osteomyelitis, and malaria [53]. In Uyghur medicine, China, E. resinifera O.Berg is recurrently employed to suppress tuberculosis, toothache, and chronic pain [54], while E. fischeriana have been used as a remedy for cancer, ascites, and oedema [55], and E. granulata Forssk. is utilized against intestine worms, oedema, cough, blood impurities, and renal diseases [56,57].
However, some Euphorbia plants, especially their latex or milky sap (e.g., E. hirta, E. helioscopia, E. royleana Boiss. among others), are considered as irritating materials for skin, mouth, and throat, causing burning sensation, acute inflammation (even blisters), and nausea [58]. In veterinary medicine, E. milii Des Moul. and E. nivulia is used to treat diarrhoea and wounds in livestock, respectively, but other Euphorbia species can produce irritations [6].
3. Euphorbia Plants: Essential Oil Composition and Activities
Researchers from various countries worldwide have studied the chemical composition of essential oils (EOs) from different Euphorbia species. An overview of their most abundant components (the content higher than 5%) along with the most relevant biological activities to health maintenance (when available, and when the biological activity of a positive standard compound was also presented) is given in Table 1. The chemical structure of the major constituents of EOs from Euphorbia species whose content is higher than 25% is depicted in Figure 1.

Table 1.
Chemical composition and biological activities of Euphorbia essential oils.
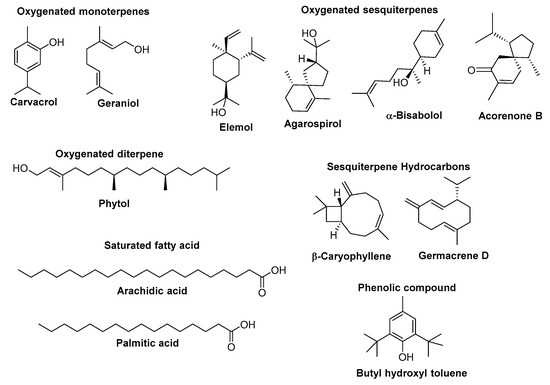
Figure 1.
Chemical structures of the constituents of Euphorbia essential oil, each one with a content exceeding 25%.
The Table 1 data show that EOs were obtained mainly from aerial parts (39%) and inflorescences (29%), in addition to leaves (18%), roots (11%), and stems (3%), by using basically two extraction methods—hydro-distillation (HD) (52%) and steam distillation (SD) (45%). The oil yield ranged from 0.07% to 1.52% (w/v) in E. cotinifolia (syn. E. caracasana) and E. fischeriana, and from 0.08% to 0.84% (w/w) in E. pilosa and E. densa. Microwave-assisted extraction (MAE) was reported only once (3%) with faster extraction time (3:1) and higher oil yield (1.2% vs. 0.7% w/v) than conventional techniques (MAE vs. HD) [74]. Qualitative and quantitative analyses were performed by gas chromatography (GC) or GC coupled to mass spectrometry (GC-MS). Samples were found to contain from 8 to 83 phytochemicals representing 81.7–99.9% of the oils content. Oxygenated sesquiterpenes (up to 86.1% of the oil in E. teheranica) characterize EOs of Euphorbia species, followed by sesquiterpene hydrocarbons (up to 34.8% in E. helioscopia) (Table 1). In general, β-caryophyllene was the most ubiquitous sesquiterpene present in 50% of the species investigated namely in E. acanthothamnos, E. apios, E. cotinifolia, E. densa, E. fischeriana, E. fragifera, E. golondrina, E. helioscopia, E. heterophylla, E. rigida, E. sanctae-caterinae, E. teheranica and E. tithymaloides constituting more than 7% of their EOs (Table 1; Figure 1).
As reported by Lokar et al. [63], different habitats can influence the quantitative composition of EO from the same species. For example, EO of E. fragifera growing in a xeric habitat was richer in aromatic terpenes than that obtained from plants collected in shady and moist soils (e.g., 61.55% vs. 3.36% of carvacrol) being the last ones characterized by great quantity of acyclic compounds (e.g., 1.24% vs. 59.65% of geraniol). Moreover, variation in the components of EOs may occur due to the season, geographical area, and date of collection [63].
From Table 1, it appears that most of the EOs of Euphorbia species studied exhibit antioxidant properties, especially by the radical scavenging mechanism. Note that some of them are more active than ascorbic acid, BHT, or BHA compounds well known for their antioxidant properties and are widely used in the food industry as a preservative.
On the other hand, the data presented also show that there are many Euphorbia species whose EOs are still not yet studied, thus evidencing a knowledge gap about the potential of these species.
4. Antibacterial and Antifungal Activity of Euphorbia Plants
Plants belonging to the genus Euphorbia are also of the great interest in the matter of their antimicrobial activity [51,52,78,79,80]. In fact, these plants are also widely used in the traditional medicine in the microbial infections [81,82], and some Euphorbia plants are believed to be a promising source of phytochemicals used in the pharmacy and food industries [83]. Since consumers prefer healthy products without synthetic raw materials, the constantly growing interest in the natural and ecologically friendly antimicrobial agents is still being observed, and therefore research on the antimicrobial activity involving Euphorbia species is relevant. The most recent studies in this subject and with greater impact is summarized in Table 2. This is not an exhaustive list of all studies performed, and only those that meet as a minimum requirement the indication of at least one statistical parameter associated with the average value of activity level (e.g., standard deviation) are included.

Table 2.
Antibacterial and antifungal activity of Euphorbia extracts.
According to Ashraf et al. [84], the hexane extract of E. royleana, when compared with methanol and water extracts, is the one with highest phenolic and flavonoid contents and the best antimicrobial agent. In fact, it exhibits antifungal activity against Aspergillus niger and antibacterial activity against the gram-positive bacteria Bacillus subtilis, about half that of the reference compounds (rifampicin and terbinafine).
The methanol extracts of E. hirta L. and E. tirucalli L. exhibit similar activity against a broad spectrum of bacteria and fungi associated with skin infections (zones of growth inhibition ranged from 9.0 mm (B. subtilis) to 14.0 mm (P. aeruginosa)) [85]. These results support the use of these species in traditional Indian medicine, and they can be used as an easily accessible source of natural antimicrobial agents [85].
The results obtained by Kumara Swamy et al. [86] showed that, independent of the extract prepared and the microorganism tested, E. neriifolia exhibits low antibacterial and antifungal activity, being the less interesting Euphorbia species as a source of antimicrobial agents.
In another study, the chloroform extract of E. paralias L. stems seems to be the most interesting extract, since it exhibits similar activity against fungi, gram-positive, and gram-negative bacteria (MIC = 15 µg/mL against C. albicans, B. subtilis, and E. coli strains, Table 2), while the chloroform extract of leaves only exhibits identical activity against C. albicans. The acetone extracts exhibit low activity against all the microorganisms tested [87].
According to the study conducted by Awaad et al. [88], the ethanolic extract of E. hirta aerial parts was the most active against all the bacteria and fungi tested when compared with E. granulata and E. helioscopia ethanol extracts, exhibiting an antifungal activity against M. canis similar to the amphotericin B in the same experimental conditions. Moreover, the authors demonstrated that heptacosan-1-ol, isolated from the active extract, could be the main metabolite responsible to the antimicrobial activity of E. hirta extract.
The study conducted by Ogbulie et al. [89] revealed that ethanolic extracts of E. hirta leaves showed higher activity against the gram-positive bacteria S. aureus (MIC = 22.55 mg/mL) than against the other microorganisms tested, but it was much less active than the ethanol extract of aerial parts against the same S. aureus strain (MIC= 1.95 µg/mL).
Unlike the ethanolic extract from the leaves of E. hirta, that does not inhibit the growth of Salmonella typhi, the ethanolic extract of aerial part from this species used in the study of Perumal et al. [90] that exhibited the strongest antimicrobial activity against Salmonella typhi with MIC value of 31 µg/mL, an activity higher than the chloramphenicol activity against the same strain (Table 2). Additionally, the same extract (the ethanolic extract of aerial part) also exhibits, against the gram-negative bacteria Proteus vulgaris, higher activity (MIC = 250 µg/mL) than the reference compounds gentamicin and chloramphenicol (MIC = 500 µg/mL) [90]. On the other hand, hexane extract was not active against all the tested microorganisms, except a weak activity against P. vulgaris [90].
Pisano et al. [91] carried out relatively extensive studies on the antimicrobial activity of the aqueous and ethanolic extracts from leaves, stems, and flowers of E. characias L. (Table 2). The results showed that most of these extracts are non-active against the organisms tested (MIC > 1.25 mg/mL), with the exception of the ethanolic extract from leaves against gram-positive bacteria Bacillus subtilis (MIC = 312.5 µg/mL).
5. Other Biological Activity of Euphorbia Plants
The Euphorbia plants have been used as medicinal plants for the treatment of many human ailments besides to those caused by bacteria and fungi. In fact, as will be demonstrated in the items below, Euphorbia extract and compounds have been evaluated as potential natural drugs with many other activities such as anti-inflammatory, antiviral, and antitumour.
5.1. In Vitro Studies
Studies on in vitro biological activity of several Euphorbia plants have been reported, involving both extracts and pure compounds.
The cytotoxicity of the crude extract of the stem bark of E. umbellata (Pax) Bruyns and its fractions (hexane, chloroform, ethanol, and methanol) were studied using in vitro assay and by applying the leukemic cells Jurkat clone E6-1 [92]. The study revealed that the CHCl3 fraction had the highest cytotoxicity (IC50 = 7.72 µg/mL), an activity value much lower than the reference compound vincristine (IC50 = 0.0031 µg/mL), but below the limit 30 µg/mL. According to NCI criteria [93], the activity of extracts is considered interesting to be studied in more detail. The established mechanism responsible for this action was associated with the promotion of cell cycle arrest at G0/G1 phase and apoptosis, which was related to two main classes of compounds in this fraction—steroids and triterpenes [92].
More recently, ethanolic extract of latex from E. umbellata has been evaluated for its anti-HIV properties. The study demonstrated in several models (in resting CD4+T cells, in cells from HIV-1 + individuals, and in primary non-human primate CD4+T cells) that this extract is able to reactivate the HIV virus in latency, acting on three factors with synergistic effect [94].
The ethanolic extract of E. lunulata Bunge (this name is a synonym of E. esula L.), at 10–20 µg/mL, had a significant anti-proliferative, anti-migration, and anti-invasion effect on multidrug resistant human gastric SGC7901/ADR cells, being able to arrest cell cycle progression and to induce cell apoptosis [95]. The same study further revealed that the cell cycle was arrested in G2/M phase, while significant apoptotic morphological changes was observed.
Methanolic extract of E. hirta aerial parts exhibited 90% growth inhibition against Plasmodium falciparum at 5 µg/mL and low toxicity against KB 3-1 cells, demonstrating its potential as antimalarial agent [96]. From this extract and by a bio-guided methodology, the authors isolated three flavonol glycosides—afzelin, quercitrin, and myricitrin (Figure 2)—that exhibit the ability to significantly inhibit the proliferation of the protozoan responsible for malaria disease Plasmodium falciparum strains FCR-3 (cycloguanil-resistant from Gambia) and CDC1 (chloroquine sensitive), with similar IC50 values 2.5 to 11.6 µM [96]. Quercitrin is able to arrest irreversibly the life cycle of the parasite at the trophozoite stage [96].
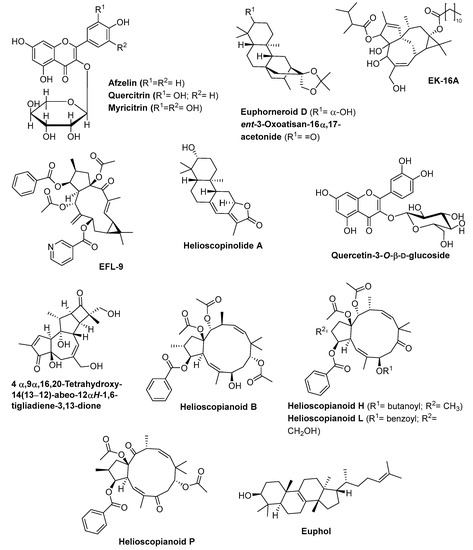
Figure 2.
Chemical structures of in vitro bioactive Euphorbia compounds.
Several diterpenes isolated from stem bark of E. neriifolia exhibit anti-HIV-I activity [97]. Among these diterpenes, euphorneroid D and ent-3-oxoatisan-16α,17-acetonide (Figure 2) were the most active compounds, with EC50 values of 34 µM and 24 µM respectively, although much less active than the reference compound (zidovudine, EC50 = 0.0019 µM). Besides these diterpenes, the ingenol derivative EK-16A (Figure 2), isolated from the E. kansui S.L.Liou ex S.B.Ho, can also be useful as antiviral agent against HIV-1 [98]. In fact, this compound is a PKCγ activator, 200-fold more potent (IC50 = 3.53 to 4.06 nM) than prostratin (IC50 = 768 to 865 nM) in reactivating latent HIV-1 without exerting detectable cytotoxicity on C11 and the J-Lat 10.6 cells viability and T cell activation. In addition, this compound exhibits a synergistic effect with other activators of HIV-1 from latency like prostratin, 5-azacytidine, and romidepsin [98].
The seeds of E. lathyris contain many natural macrocyclic diterpenes with a lathyrane skeleton [46,99], five of which were evaluated against A549, MDA-MB-231, MCF-7, KB, and KB-VIN cancer cell lines [99]. The compound EFL-9 (Figure 2) exhibits the strongest cytotoxicity against all cell lines (IC50 values ranging from 5.7 to 8.4 µM against four of the five cell lines tested). The most important is its similar toxicity against the multidrug-resistant cancer cell line KB-VIN and the parental cell KB line (IC50 values of 5.7 and 6.1 µM, respectively), while the anticancer taxol exhibits a highly differentiated toxicity to these cell lines (IC50 values of 1.9 µM and 6.7 nM, respectively) [99]. The nicotinate ester at C-7 on the EFL-9 compound seems to play a crucial role in the cytotoxicity against the KB-VIN cell line. Additionally, the cytotoxicity of EFL-9 is due to its action on actin filament aggregation as well as on partial disruption of microtubules networks [99].
Helioscopinolide A (Figure 2), one jolkinolide-type diterpenoid isolated from the ethanolic extract of E. helioscopia whole plant exhibits a strong cytotoxic activity against HeLa cells line (IC50 = 0.11 µM), being even more active than the adriamycin used as positive control (IC50 = 0.41 µM) [100]. Helioscopinolide A exhibits moderate activity against MDA-MB-231 cells line (IC50 = 2.1 µM) and is much lesser active than Adriamycin (IC50 = 0.34 µM) [100].
According Safwat et al. [101], quercetin-3-O-β-d-glucoside (Figure 2), a safe compound isolated from 70% methanol extract of E. paralias whole plant, in addition to exhibiting moderate toxicity against HepG2 and A549 cancer cell lines (IC50 values of 41 and 36 µM, respectively), exhibits the ability to inhibit the glutamine synthetase enzyme (IC50 = 0.048 µM). Taking into account that this enzyme is identified as a potential target in the development of new antimycobacterial agents, once it plays a significant role as virulence factor of Mycobacterium tuberculosis, Safwat et al. [101] showed that quercetin-3-O-β-d-glucoside could be used as an antituberculotic agent.
Several isolated diterpenoids from the roots of E. ebracteolata Hayata were reported to inhibit human carboxylesterase 2 (hCE-2), a human enzyme able to metabolize clinical drugs, resulting in adverse clinical reactions such as the reduction of biological availability of the drugs [102]. The compound 4αβ,9α,16,20-tetrahydroxy-14(13→12)-abeo-12αH-1,6-tigliadiene-3,13-dione (Figure 2) exhibits the strongest competitive inhibitory activity (IC50 = 3.88 µM, with Ki = 4.94 µM), a higher activity than the hCE-2 inhibitor BNNP (IC50 = 5.60 µM). On the other hand, this compound has non-toxic effect against HCT-116, HepG2, BGC-823, H460, and SK-OV-3 cell lines (IC50 >100 µM) [102], which is an advantage in its application as an hCE-2 inhibitor.
Helioscopianoid P and helioscopianoid H (Figure 2) are two jatrophane-type diterpenoids isolated from the whole plants of E. helioscopia, at 20 µM, exhibit inhibitory effects on P-glycoprotein in adriamycin (ADM)-resistant human breast adenocarcinoma cell line (MCF-7/ADR), while helioscopianoid L, helioscopianoid H and helioscopianoid B (Figure 2), also jatrophane-type diterpenoids isolated from the same species, exhibit neuroprotective effects against serum deprivation-induced PC12 cell damage and rotenone-induced PC12 cell damage [103].
The main component of E. umbellata and E. tirucalli latex is the tetracyclic triterpene alcohol euphol (Figure 2) [104,105]. This compound exhibits cytotoxic effect against several leukemia, colorectal, melanoma and glioma cancer cell lines [104,105]. When tested against Jurkat, HL-60, K-562 B16F10 and HRT-18 cell lines, euphol is toxic mainly against K-562 cell line (34.44 µM) by apoptosis induction, with non-toxicity to blood cells (SI > 2), but low selectivity to 3T3 cell line (SI = 0.55) [104]. On the other hand, euphol is particularly active against glioma cells (including primary, paediatric and adult glioma cell lines), with IC50 values range from 5.98 to 31.05 µM, much more active than the temozolomide, a clinical drug used to treat some brain cancers (IC50 values range from 97.00 µM to 1 mM) and with higher selective cytotoxicity index (0.64–3.36) than temozolomide (0.11–1.13) [105].
5.2. In Vivo Studies
The first significant in vivo studies reported on Euphorbia extracts or pure compounds were published in the 1990s. For example, the lyophilized E. hirta decoction exhibits antidiarrheal activity on diarrhoea induced by castor oil, arachidonic acid, and prostaglandin E2, but is inactive when the diarrhoea is induced by magnesium sulphate [106]. The flavonoid quercitrin (Figure 2) isolated from the previously reported lyophilized E. hirta decoction, exhibits antidiarrheal activity (at doses of 50 mg/kg), in castor oil- and PGE2-induced diarrhoea in mice [107], although the same authors showed that quercitrin activity is due to the glycone quercetin released in the intestine [107]. On the other hand, the water extract of E. hirta leaves, in adult Wister rats at 50–100 mg/kg dose, exhibits diuretic effect and increase the excretion of Na+, K+ and HCO3 [108].
However, the highest incidence of in vivo studies involving Euphorbia species has occurred in the last few years.
The 80% hydroethanolic extract of E. tirucalli latex at 0.250 mg (daily for 21 and 35 days) seems to be able to induce significant increases in TH1 cytokines (GM-CSF, IL-2), TH2 cytokines (IL-6), and chemokines (IL-1β, RANTES) and thus incite immunological stimulation and improve the immune system on adult male Sprague Dawley rats [109].
A steroid and terpenoid-rich fractions were isolated from the hydroethanolic extract of E. tirucalli root that, at 60 mg/kg, exhibits better protection against peripheral nociceptive pain on acetic acid induced abdominal constrictions mice model than the analgesic aceclofenac sodium and better anti-inflammatory activity in carrageenan-induced mice model than indomethacin [110].
On the other hand, the 70% hydroethanolic extract of E. supina Raf. (this name is a synonym of E. maculata L.), at 10 mg/mL, is able to significantly reduce the ear thickness and number of inflammatory cells on the Propionibacterium acnes-induced skin inflammation by inhibition of pro-inflammatory cytokines expression and the MAPK signalling pathway [111].
The wound-healing activity of different extracts (n-hexane, ethyl acetate, and methanol) from aerial parts of different Euphorbia species (E. helioscopia, E. characias subsp. Wulfenii (Hoppe ex W.D.J.Koch) Radcl.-Sm., E. macroclada Boiss., E. seguieriana subsp. seguieriana (this name is a synonym of E. seguieriana Neck.), and E. virgata Waldst. & Kit. (this name is a synonym of E. esula subsp. tommasiniana (Bertol.) Kuzmanov), was evaluated in vivo by Özbilgin et al. [112]. The authors showed that the methanol extract of E. characias subsp. wulfenii was the most active, significantly reducing the linear incision wound (43.04%) while the circular excision wound decreased 65.24%, with a significant decrease in wound inflammation (34.74%) in relation to the control groups. These activities were further linked with the quercetin glycosides identified in the extract [113].
A comparative study with several species, including E. helioscopia, E. lactea Haw., and E. nivulia Buch.-Ham. showed that the ethanol extract from the latter (at 100 mg/kg) was the one that caused the greatest anticonvulsant effects and highest reduction in the progression of epileptogenesis in pentylenetetrazole-induced kindling model of epilepsy in mice [114].
It was well demonstrated that the lyophilized decoction of fresh whole E. hirta had great potential against dengue virus since, after 14 days of administration at 100 mg/kg dose, it significantly increased platelet counts in rats, also causing a decrease in coagulation time and bleeding [115].
As shown above and in a recent review on the subject [116], several reports have suggested that Euphorbia plant possesses, in in vitro assays, considerable cytotoxic potential. Some in vivo studies also corroborate the anticancer potential and safety of Euphorbia extracts and compounds. For example, the methanolic extract of E. triaculeata Forssk., when tested in albino mice at 250 to 1000mg/kg body weight, is not genotoxic or clastogenic, while the pre-treatment with this extract inhibits the clastogenicity induced by cyclophosphamide without impairing its cytotoxic potential [117].
The tetracyclic triterpene alcohol, euphol (Figure 2), showed antitumoural effects in GAMG glioma model through the activation of autophagy-associated cell death [105].
Euphornin L (Figure 3), a relatively abundant jatrophane diterpenoid from E. helioscopia, exhibits outstanding lipid-lowering effect on golden Syrian hamsters fed with a high-fat diet at a well-tolerated dose of 30mg/kg (p.o.) [118]. In fact, the authors observed a significant decrease on CHOL and LDL-C levels in the treatment group when compared with control group, while other parameters (HDLC, TG, and body weight) did not suffer significant changes [119].
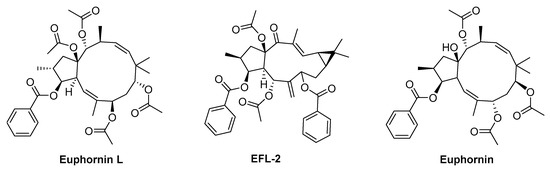
Figure 3.
Chemical structures of some in vivo bioactive Euphorbia compounds.
Euphorbia factor L2 (EFL-2, Figure 3), a secondary metabolite isolated from seeds of E. lathyris, was tested to treat lipopolysaccharide (LPS) induced acute lung injury (ALI) in mice [119]. EFL-2 at concentration of 40 mg/kg causes an attenuation of the pathological changes in the lung by significant suppression of the recruitment and transmigration of inflammatory cells, specifically neutrophils [119]. The same compound was tested, using SMMC7721 xenograft BALB/c nude mice, to evaluate its effect on human hepatocellular carcinoma [120]. EFL-2, administered at 25 and 50 mg/kg for 14 days, is able to inhibit the growth of hepatocellular carcinoma in BALB/c athymic nude mice, causing a decrease in tumour volume and weight, mainly by targeting STAT3 and AKT inactivation during TGF-β-induced EMT and metastasis [120].
From E. helioscopia aerial parts were isolated several secondary metabolites that exhibit vasodepressive activity, being the euphornin (Figure 3) the most active, causing a significant reduction in direct blood pressure (42 mmHg) in an adult male Wistar albino rat model during 70 min [121].
5.3. Clinical Studies
There are very few clinical studies involving Euphorbia species. In a clinical trial whose results were published in 2011 [122], skin cancer lesions (basal cell carcinomas, squamous cell carcinomas, and intraepidermal carcinomas) were treated topically with 100–300 µL of E. peplus latex (daily for three days). The results showed this latex exhibits a clinical response that is comparable to existing non-surgical treatments used in clinical therapeutics against human non-melanoma skin cancers. The active ingredient of E. peplus latex has been identified as ingenol mebutate, the active principle of the clinical medicine Picato® used in the treatment of actinic keratosis [24].
The E. hirta extract is able to cause an increase in the number of platelets in dengue patients from 30–55 age group and a decrease in flu-like symptoms in 70% of the patients. However, no identical effect was observed in the 14–25-age group [123].
A clinical study on phase 2 to determine the safe and efficacious dose of E. prostrata for control of per rectal bleeding in patients with first- and second-degree haemorrhoids is now complete (Clinical Trials Identifier: NCT01041911) [124]. In that trial, 82% of the patients had complete cessation of bleeding at the end of two weeks and thus demonstrate that E. prostrata can be used as an effective therapeutic agent and without adverse effects in the treatment of early stage haemorrhoids [125].
A clinical trial involving E. kansui (an E. kansui extract powder prepared as tea) is currently in the recruitment phase and intends to evaluate the immune response of HIV+ individuals to this herbal supplement and its dependence with the dose applied. Also, the safety of the herbal preparation is evaluated (Clinical Trial Identifier: NCT02531295) [124].
6. Conclusions
The Euphorbia species are plants well known for their applications, especially its latex, in traditional medicine around the world. Their chemical composition may vary according to the species, to the part of the plant, and the applied extraction methodology. This variability could also be influenced by the different habitats, seasons, and dates of collection. Quantitative and qualitative analysis have been developed for the compounds’ identification, being the major constituents of essential oils oxygenated sesquiterpenes followed by sesquiterpene hydrocarbons.
Additionally, several studies reported and discussed above, confirmed, in vitro, in vivo, and in clinical trials, the biological activities of Euphorbia extracts and pure compounds. These compounds and extracts could be applied to the treatment of different diseases mainly related to microbial infections, as well as inflammation and cancer.
Euphorbia plants have great potential as a source of bioactive extracts and pure compounds, which may lead to the development of new drugs for clinical use.
Author Contributions
All authors contributed to the manuscript. Conceptualization, B.S. and J.S.-R.; validation investigation, resources, data curation, writing first draft, S.V., H.A., E.P., D.K., S.I.O., K.C., M.S., L.C., E.C.-B., A.S.-C., R.C., and W.C.C.; review and editing, J.S.-R., B.S., M.d.l.L.C.-G., M.I., A.O.A., and A.M.L.S.; All the authors read and approved the final manuscript.
Funding
This research was funded by FCT/MCT, supporting the cE3c centre (FCT Unit, UID/BIA/00329/2013, 2015-2018, and UID/BIA/00329/2019), and the QOPNA research Unit (FCT UID/QUI/00062/2019).
Acknowledgments
Thanks to the University of Azores and University of Aveiro.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Shi, Q.W.; Su, X.H.; Kiyota, H. Chemical and pharmacological research of the plants in genus Euphorbia. Chem. Rev. 2008, 108, 4295–4327. [Google Scholar] [CrossRef] [PubMed]
- The Plant List. Available online: http://www.theplantlist.org/1.1/browse/A/Compositae/Inula/ (accessed on 3 June 2019).
- Govaerts, R.; Frodin, D.G.; Radcliffe-Smith, A. World Checklist and Bibliography of Euphorbiaceae (with Pandaceae); Royal Botanic Gardens: Kew, UK, 2000; Volume 2. [Google Scholar]
- IUCN Red List. The IUCN Red List of Threatened Species. Available online: https://www.iucnredlist.org/ (accessed on 1 June 2019).
- Kumar, S.; Malhotra, R.; Kumar, D. Euphorbia hirta: Its chemistry, traditional and medicinal uses, and pharmacological activities. Pharmacogn. Rev. 2010, 4, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Ernst, M.; Grace, O.M.; Saslis-Lagoudakis, C.H.; Nilsson, N.; Simonsen, H.T.; Rønsted, N. Global medicinal uses of Euphorbia L. (Euphorbiaceae). J. Ethnopharmacol. 2015, 176, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Pascal, O.A.; Bertrand, A.E.V.; Esaïe, T.; Sylvie, H.A.M.; Eloi, A.Y. A review of the ethnomedicinal uses, phytochemistry and pharmacology of the Euphorbia genus. Pharma Innov. J. 2017, 6, 34–39. [Google Scholar]
- Webster, G.L. Classification of the Euphorbiaceae. Ann. Mo. Bot. Gard. 2006, 81, 3. [Google Scholar] [CrossRef]
- Özbilgin, S.; Saltan Çitoǧlu, G. Uses of some Euphorbia species in traditional medicine in Turkey and their biological activities. Turk. J. Pharm. Sci. 2012, 9, 241–256. [Google Scholar]
- Jassbi, A.R. Chemistry and biological activity of secondary metabolites in Euphorbia from Iran. Phytochemistry 2006, 67, 1977–1984. [Google Scholar] [CrossRef]
- Ma, Q.G.; Liu, W.Z.; Wu, X.Y.; Zhou, T.X.; Qin, G.W. Chemical studies of Lang-Du, a traditional Chinese medicine.1. Diterpenoids from Euphorbia fischeriana. Phytochemistry 1997, 44, 663–666. [Google Scholar]
- Hassan, A.; Yaqoob, U.; Nawchoo, I.A.; Gulzar, S.; Mohi-Ud-Din, G.; Nazir, S.; Ashraf, A. Conspectus of phytochemical constituents of Euphorbia wallichii Hook. F.: A Review. Res. Rev. J. Bot. 2016, 5, 24–31. [Google Scholar]
- Ravikanth, V.; Niranjan Reddy, V.L.; Prabhakar Rao, T.; Diwan, P.V.; Ramakrishna, S.; Venkateswarlu, Y. Macrocyclic diterpenes from Euphorbia nivulia. Phytochemistry 2002, 59, 331–335. [Google Scholar] [CrossRef]
- Öksüz, S.; Gürek, F.; Gil, R.R.; Pengsuparp, T.; Pezzuto, J.M.; Cordell, G.A. Four diterpene esters from Euphorbia myrsinites. Phytochemistry 1995, 38, 1457–1462. [Google Scholar] [CrossRef]
- Hohmann, J.; Rédei, D.; Evanics, F.; Kálmán, A.; Argay, G.; Bartók, T. Serrulatin A and B, new diterpene polyesters from Euphorbia serrulata. Tetrahedron 2000, 56, 3619–3623. [Google Scholar] [CrossRef]
- Abdelgaleil, S.A.M.; Kassem, S.M.I.; Doe, M.; Baba, M.; Nakatani, M. Diterpenoids from Euphorbia paralias. Phytochemistry 2001, 58, 1135–1139. [Google Scholar] [CrossRef]
- Lima, E.M.; Medeiros, J.M.; Davin, L.B. Pentacyclic triterpenes from Euphorbia stygiana. Phytochemistry 2003, 63, 421–425. [Google Scholar] [CrossRef]
- Fatope, M.O.; Zeng, L.; Ohayaga, J.E.; Shi, G.; McLaughlin, J.L. Selectively cytotoxic diterpenes from Euphorbia poisonii. J. Med. Chem. 1996, 39, 1005–1008. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Wang, N.L.; Yao, X.S.; Miyata, S.; Kitanaka, S. Diterpenes from the roots of Euphorbia kansui and their in vitro effects on the cell division of xenopus. J. Nat. Prod. 2002, 65, 1246–1251. [Google Scholar] [CrossRef]
- Betancur-Galvis, L.; Palomares, E.; Marco, J.A.; Estornell, E. Tigliane diterpenes from the latex of Euphorbia obtusifolia with inhibitory activity on the mammalian mitochondrial respiratory chain. J. Ethnopharmacol. 2003, 85, 279–282. [Google Scholar] [CrossRef]
- Hezareh, M. Prostratin as a new therapeutic agent targeting HIV viral reservoirs. Drug News Perspect. 2006, 18, 496–500. [Google Scholar] [CrossRef]
- Geng, D.; Yi, L.T.; Shi, Y.; Min, Z.D. Structure and antibacterial property of a new diterpenoid from Euphorbia helioscopia. Chin. J. Nat. Med. 2015, 13, 704–706. [Google Scholar] [CrossRef]
- Hohmann, J.; Rédei, D.; Forgo, P.; Molnár, J.; Dombi, G.; Zorig, T. Jatrophane diterpenoids from Euphorbia mongolica as modulators of the multidrug resistance of L5128 mouse lymphoma cells. J. Nat. Prod. 2003, 66, 976–979. [Google Scholar] [CrossRef]
- Berman, B. New developments in the treatment of actinic keratosis: Focus on ingenol mebutate gel. Clin. Cosmet. Investig. Dermatol. 2012, 5, 111–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frezza, C.; Venditti, A.; Sciubba, F.; Tomai, P.; Antonetti, M.; Franceschin, M.; Di Cocco, M.E.; Gentili, A.; Delfini, M.; Serafini, M.; et al. Phytochemical profile of Euphorbia peplus L. collected in Central Italy and NMR semi-quantitative analysis of the diterpenoid fraction. J. Pharm. Biomed. Anal. 2018, 160, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Seca, A.M.L.; Pinto, D.C.G.A. Plant secondary metabolites as anticancer agents: Successes in clinical trials and therapeutic application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef] [PubMed]
- Machado, M.M.; de Oliveira, L.F.; Zuravski, L.; de Souza, R.O.; Fischer, P.; Duarte, J.A.; Rocha, M.O.; Güez, C.M.; Boligon, A.A.; Athayde, M.L. Evaluation of genotoxic and cytotoxic effects of hydroalcoholic extract of Euphorbia tirucalli (Euphorbiaceae) in cell cultures of human leukocytes. An. Acad. Bras. Cienc. 2016, 88, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Prenner, G.; Rudall, P.J. Comparative ontogeny of the cyathium in Euphorbia (Euphorbiaceae) and its allies: Exploring the organ–flower–inflorescence boundary. Am. J. Bot. 2007, 94, 1612–1629. [Google Scholar] [CrossRef] [PubMed]
- Horn, J.W.; van Ee, B.W.; Morawetz, J.J.; Riina, R.; Steinmann, V.W.; Berry, P.E.; Wurdack, K.J. Phylogenetics and the evolution of major structural characters in the giant genus Euphorbia L. (Euphorbiaceae). Mol. Phylogenet. Evol. 2012, 63, 305–326. [Google Scholar] [CrossRef] [PubMed]
- Rizk, A.F.M. The chemical constituents and economic plants of the Euphorbiaceae. Bot. J. Linn. Soc. 1987, 94, 293–326. [Google Scholar] [CrossRef]
- Jing, S.X.; Hua, J.; Li, S.H.; Liu, Y.; Luo, S.H.; Xiao, C.J. Chemical profile and defensive function of the latex of Euphorbia peplus. Phytochemistry 2017, 136, 56–64. [Google Scholar]
- Dabholkar, D.A.; Kaicker, P.K.; Diwan, R.K. Euphorbia latex—Its chemistry and industrial applications. 1. Res. Ind. 1991, 36, 126–131. [Google Scholar]
- Toro-Vazquez, J.F.; Morales-Rueda, J.A.; Dibildox-Alvarado, E.; Charó-Alonso, M.; Alonzo-Macias, M.; González-Chávez, M.M. Thermal and textural properties of organogels developed by candelilla wax in safflower oil. JAOCS J. Am. Oil Chem. Soc. 2007, 84, 989–1000. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food. Scientific opinion on the re-evaluation of candelilla wax (E 902) as a food additive. EFSA J. 2012, 10, 2946. Available online: www.efsa.europa.eu/efsajournal.htm (accessed on 28 January 2013). [CrossRef]
- Polygenis-bigendako, M.J.; Lejoly, J. Plantes employées dans le traitement des diarrhées en médecine traditionnelle au Burundi occidental. Bull. Société R. Bot. Belg. 1989, 122, 87–97. [Google Scholar]
- Osemeobo, G.J. Effects of common property resource utilization on wildlife conservation in Nigeria. GeoJournal 1991, 23, 241–248. [Google Scholar] [CrossRef]
- Gaioni, D.T. Medical choices in a Philippine highland community. Ethnomedical and biomedical dimensions of Bauko clinical reality. Anthropos 2002, 97, 505–518. [Google Scholar]
- Lai, X.Z.; Yang, Y.B.; Shan, X.L. The investigation of Euphorbiaceous medicinal plants in southern China. Econ. Bot. 2004, 28, S307. [Google Scholar] [CrossRef]
- Hargreaves, B.J. The spurges of Botswana. Botsw. Notes Rec. 1991, 23, 115–130. [Google Scholar]
- Manandhar, N.P. An inventory of some herbal drugs of Myagdi district, Nepal. Econ. Bot. 1995, 49, 371–379. [Google Scholar] [CrossRef]
- Van Sam, H.; Baas, P.; Keßler, P.J.A. Traditional medicinal plants in Ben En National Park, Vietnam. Blumea 2008, 53, 569–601. [Google Scholar] [CrossRef]
- Baslas, R.K. Agarwal, R. Isolation and characterisation of different constituents of Euphorbia hirta Linn. Curr. Sci. 1980, 49, 311–312. [Google Scholar]
- Colley, F.C. Traditional Indian medicine in Malaysia. J. Malays. Branch R. Asiat. Soc. 1978, 51, 77–109. [Google Scholar]
- Gopi, K.; Renu, K.; Sannanaik Vishwanath, B.; Jayaraman, G. Protective effect of Euphorbia hirta and its components against snake venom induced lethality. J. Ethnopharmacol. 2015, 165, 180–190. [Google Scholar] [CrossRef]
- Lammers, T.G. Systematics of Clermontia (Campanulaceae, Lobelioideae). Syst. Bot. Monogr. 1992, 32, 1–97. [Google Scholar] [CrossRef]
- Lu, J.; Li, G.; Huang, J.; Zhang, C.; Zhang, L.; Zhang, K.; Li, P.; Lin, R.; Wang, J. Lathyrane-type diterpenoids from the seeds of Euphorbia lathyris. Phytochemistry 2014, 104, 79–88. [Google Scholar] [CrossRef]
- Bard, C.L. A contribution to the history of medicine in southern California. J. Calif. Gt. Basin Anthropol. 2006, 26, 95–108. [Google Scholar]
- Kaval, I.; Behcet, L.; Cakilcioglu, U. Ethnobotanical study on medicinal plants in Gecitli and its surrounding (Hakkari-Turkey). J. Ethnopharmacol. 2014, 155, 171–184. [Google Scholar] [CrossRef]
- Mummed, B.; Abraha, A.; Feyera, T.; Nigusse, A.; Assefa, S. In vitro antibacterial activity of selected medicinal plants in the traditional treatment of skin and wound infections in eastern Ethiopia. BioMed Res. Int. 2018, 2018, 1862401. [Google Scholar] [CrossRef]
- Hsieh, W.T.; Lin, H.Y.; Chen, J.H.; Lin, W.C.; Kuo, Y.H.; Wood, W.G.; Lu, H.F.; Chung, J.G. Latex of Euphorbia antiquorum-induced S-phase arrest via active ATM kinase and MAPK pathways in human cervical cancer HeLa cells. Environ. Toxicol. 2015, 30, 1205–1215. [Google Scholar] [CrossRef]
- Mali, P.Y.; Panchal, S.S. Euphorbia tirucalli L.: Review on morphology, medicinal uses, phytochemistry and pharmacological activities. Asian Pac. J. Trop. Biomed. 2017, 7, 603–613. [Google Scholar]
- Mali, P.Y.; Panchal, S.S. Euphorbia neriifolia L.: Review on botany, ethnomedicinal uses, phytochemistry and biological activities. Asian Pac. J. Trop. Biomed. 2017, 10, 430–438. [Google Scholar]
- Chen, H.; Wang, H.; Yang, B.; Jin, D.Q.; Yang, S.; Wang, M.; Xu, J.; Ohizumi, Y.; Guo, Y. Diterpenes inhibiting NO production from Euphorbia helioscopia. Fitoterapia 2014, 95, 133–138. [Google Scholar] [CrossRef]
- Wang, S.Y.; Li, G.Y.; Zhang, K.; Wang, H.Y.; Liang, H.G.; Huang, C.; Huang, J.; Wang, J.H.; Yang, B.F. New ingol-type diterpenes from the latex of Euphorbia resinifera. J. Asian Nat. Prod. Res. 2019, 1–8. [Google Scholar] [CrossRef]
- Jian, B.; Zhang, H.; Liu, J. Structural diversity and biological activities of diterpenoids derived from Euphorbia fischeriana Steud. Molecules 2018, 23, 935. [Google Scholar] [CrossRef]
- Ahmad, S.; Perveen, S.; Arshad, M.A.; Rehman, T. Pharmacological and nutritive potential of Euphorbia granulata. J. Complement. Integr. Med. 2019, in press. [Google Scholar] [CrossRef]
- Malik, S.; Ahmad, S.; Sadiq, A.; Alam, K.; Wariss, H.M.; Ahmad, I.; Hayat, M.K.; Anjum, S.; Mukhtar, M. A comparative ethno-botanical study of Cholistan (an arid area) and Pothwar (a semi-arid area) of Pakistan for traditional medicines. J. Ethnobiol. Ethnomed. 2015, 11, 31. [Google Scholar] [CrossRef]
- Bhatia, H.; Manhas, R.K.; Kumar, K.; Magotra, R. Traditional knowledge on poisonous plants of Udhampur district of Jammu and Kashmir, India. J. Ethnopharmacol. 2014, 152, 207–216. [Google Scholar] [CrossRef]
- Fokialakis, N.; Melliou, E.; Magiatis, P.; Harvala, C.; Mitaku, S. Composition of the steam volatiles of six Euphorbia spp. from Greece. Flavour Fragr. J. 2003, 18, 39–42. [Google Scholar] [CrossRef]
- Rojas, J.; Baldovino, S.; Vizcaya, M.; Rojas, L.B.; Morales, A. The chemical composition of the essential oils of Euphorbia caracasana and E. cotinifolia (Euphorbiaceae) from Venezuela. Nat. Prod. Commun. 2009, 4, 571–572. [Google Scholar] [CrossRef]
- Merza, J. Chemical composition of essential oil extracted from Euphorbia densa Schrenk and evaluation its antioxidant activity. Food Sci. Qual. Manag. 2018, 76, 31–34. [Google Scholar]
- Cui, J.; Yang, X.; Dong, A.; Cheng, D.; Wang, J.; Zhao, H.; Xu, R. Chemical composition and antioxidant activity of Euphorbia fischeriana essential oil from China. J. Med. Plants Res. 2015, 19, 4794–4798. [Google Scholar]
- Lokar, L.C.; Maurich, V.; Poldini, L. Chemical aspect of floral biology in Euphorbia fragifera. Folia Geobot. Phytotaxon. 1986, 21, 277–285. [Google Scholar] [CrossRef]
- Ertas, A.; Yilmaz, M.A.; Firat, M. Chemical profile by LC–MS/MS, GC/MS and antioxidant activities of the essential oils and crude extracts of two Euphorbia species. Nat. Prod. Res. 2015, 29, 529–534. [Google Scholar] [CrossRef]
- Ndam, L.M.; Mih, A.M.; Tening, A.S.; Fongod, A.G.N.; Temenu, N.A.; Fujii, Y. Phytochemical analysis, antimicrobial and antioxidant activities of Euphorbia golondrina L.C. Wheeler (Euphorbiaceae Juss.): An unexplored medicinal herb reported from Cameroon. Springerplus 2016, 5, 264. [Google Scholar] [CrossRef][Green Version]
- Akhgar, M.R.; Rajaei, P.; Aieen, S. Constituents of the essential oil of Euphorbia hebecarpa. Chem. Nat. Compd. 2014, 50, 929–930. [Google Scholar] [CrossRef]
- Deveci, E.; Tel-Çayan, G.; Duru, M.E. Investigation of chemical composition, antioxidant, anticholinesterase and anti-urease activities of Euphorbia helioscopia. Int. J. Second. Metab. 2018, 5, 259–269. [Google Scholar]
- Adedoyin, B.J.; Okeniyi, S.O.; Garba, S.; Salihu, L. Cytotoxicity, antioxidant and antimicrobial activities of essential oil extracted from Euphorbia heterophylla plant. Topclass J. Herb. Med. 2013, 2, 84–89. [Google Scholar]
- Elshamy, A.I.; Abd-ElGawad, A.M.; El Gendy, A.E.N.G.; Assaeed, A.M. Chemical characterization of Euphorbia heterophylla L. essential oils and their antioxidant activity and allelopathic potential on Cenchrus echinatus L. Chem. Biodivers. 2019, 16, 1900051. [Google Scholar] [CrossRef]
- Ogunlesi, M.; Okiei, W.; Ofor, E.; Osibote, A. Analysis of the essential oil from the dried leaves of Euphorbia hirta Linn (Euphorbiaceae), a potential medication for asthma. Afr. J. Biotechnol. 2009, 8, 7042–7050. [Google Scholar]
- Lin, J.; Dou, J.; Xu, J.; Aisa, H.A. Chemical composition, antimicrobial and antitumor activities of the essential oils and crude extracts of Euphorbia macrorrhiza. Molecules 2012, 17, 5030–5039. [Google Scholar] [CrossRef]
- Li, X.; Bai, G.; Wang, R.; Yang, J.; Yuan, M.; An, Y.; Wu, X. Study on the chemical components of volatile oil from Euphorbia pekinensis radix. Zhong Yao Cai 2013, 36, 237–239. [Google Scholar]
- Ram, C.; Joshi, P.; Prasad, K. Chemical composition of the essential oil of Euphorbia pilosa from Munsiari, Pithoragarh, India. Int. J. Res. Pharm. Pharm. Sci. 2018, 3, 1–4. [Google Scholar]
- Reda, E.; Saleh, I.; El Gendy, A.N.; Talaat, Z.; Hegazy, M.E.; Haggag, E. Chemical constituents of Euphorbia sanctae-catharinae Fayed essential oil: A comparative study of hydro-distillation and microwave-assisted extraction. J. Adv. Pharm. Res. 2017, 1, 155–159. [Google Scholar] [CrossRef][Green Version]
- Feizbakhsh, A.; Bighdeli, M.; Tehrani, M.S.; Rustaiyan, A.; Masoudi, S. Chemical constituents of the essential oil of Euphorbia teheranica Boiss., a species endemic to Iran. J. Essent. Oil Res. 2004, 16, 40–41. [Google Scholar] [CrossRef]
- Prasad, K.; Bisht, G. Evaluation of nutritive minerals and antioxidants values of Euphorbia thymifolia Linn. Curr. Res. Chem. 2011, 3, 98–105. [Google Scholar] [CrossRef]
- Rahman, A.; Rahman, M.; Demirtas, I. Chemical composition and antioxidant potential of essential oil and organic extracts of Euphorbia tithymaloides L. from Kushtia Region. Anticancer Agents Med. Chem. 2018, 18, 1482–1488. [Google Scholar] [CrossRef]
- Sharanappa, R.; Vidyasagar, G.M. Anti-Candida activity of medicinal plants. A review. Int. J. Pharm. Pharm. Sci. 2013, 5, 9–16. [Google Scholar]
- Kumari, I. A study of the antimicrobial effect of Euphorbia hirta L. against Escherichia coli. Vegetos 2018, 31, 123–125. [Google Scholar] [CrossRef]
- Siritapetawee, J.; Limphirat, W.; Wongviriya, W.; Maneesan, J.; Samosornsuk, W. Isolation and characterization of a galactose-specific lectin (EantH) with antimicrobial activity from Euphorbia antiquorum L. latex. Int. J. Biol. Macromol. 2018, 120, 1846–1854. [Google Scholar] [CrossRef]
- Diop, E.A.; Queiroz, E.F.; Kicka, S.; Rudaz, S.; Diop, T.; Soldati, T.; Wolfender, J.L. Survey on medicinal plants traditionally used in Senegal for the treatment of tuberculosis (TB) and assessment of their antimycobacterial activity. J. Ethnopharmacol. 2018, 216, 71–78. [Google Scholar] [CrossRef]
- Shamsabadipour, S.; Ghanadian, M.; Saeedi, H.; Rahimnejad, M.R.; Mohammadi-Kamalabadi, M.; Ayatollahi, S.M.; Salimzadeh, L. Triterpenes and Steroids from Euphorbia denticulata Lam. With Anti-Herpes Symplex Virus Activity. Iran. J. Pharm. Res. 2013, 12, 759–767. [Google Scholar]
- Zengin, G.; Uysal, A.; Aktumsek, A.; Mocan, A.; Mollica, A.; Locatelli, M.; Custodio, L.; Neng, N.R.; Nogueira, J.M.F.; Aumeeruddy-Elalfi, Z.; et al. Euphorbia denticulata Lam.: A promising source of phyto-pharmaceuticals for the development of novel functional formulations. Biomed. Pharmacother. 2017, 87, 27–36. [Google Scholar] [CrossRef]
- Ashraf, A.; Sarfraz, R.A.; Rashid, M.A.; Shahid, M. Antioxidant, antimicrobial, antitumor, and cytotoxic activities of an important medicinal plant (Euphorbia royleana) from Pakistan. J. Food Drug Anal. 2015, 23, 109–115. [Google Scholar] [CrossRef]
- Chanda, S.; Baravalia, Y. Screening of some plant extracts against some skin diseases caused by oxidative stress and microorganisms. Afr. J. Biotechnol. 2010, 9, 3210–3217. [Google Scholar]
- Kumara Swamy, M.; Pokharen, N.; Dahal, S.; Anuradha, M. Phytochemical and antimicrobial studies of leaf extract of Euphorbia neriifolia. J. Med. Plant Res. 2011, 5, 5785–5788. [Google Scholar]
- Hlila, M.B.; Majouli, K.; Ben Jannet, H.; Aouni, M.; Mastouri, M.; Selmi, B. Antimicrobial activity of Tunisian Euphorbia paralias L. Asian Pac. J. Trop. Biomed. 2017, 7, 629–632. [Google Scholar] [CrossRef]
- Awaad, A.S.; Alothman, M.R.; Zain, Y.M.; Zain, G.M.; Alqasoumi, S.I.; Hassan, D.A. Comparative nutritional value and antimicrobial activities between three Euphorbia species growing in Saudi Arabia. Saudi Pharm. J. 2017, 25, 1226–1230. [Google Scholar] [CrossRef]
- Ogbulie, B.J.N.; Ogueke, C.C.; Okoli, I.C.; Anyanwu, B.N. Antibacterial activities and toxicological potentials of crude ethanolic extracts of Euphorbia hirta. J. Am. Sci. 2007, 3, 11–16. [Google Scholar]
- Perumal, S.; Mahmud, R.; Pillai, S.; Lee, W.C.; Ramanathan, S. Antimicrobial activity and cytotoxicity evaluation of Euphorbia hirta (L.) extracts from Malaysia. APCBEE Procedia 2012, 2, 80–85. [Google Scholar] [CrossRef]
- Pisano, M.B.; Cosentino, S.; Viale, S.; Spanò, D.; Corona, A.; Esposito, F.; Tramontano, E.; Montoro, P.; Tuberoso, C.I.G.; Medda, R.; et al. Biological activities of aerial parts extracts of Euphorbia characias. BioMed Res. Int. 2016, 2016, 1538703. [Google Scholar] [CrossRef]
- Kanunfre, C.C.; Leffers, T.; Cruz, L.S.; Luz, L.E.C.; Crisma, A.R.; Wang, M.; Avula, B.; Khan, I.A.; Beltrame, F.L. Euphorbia umbellata bark extracts—An in vitro cytotoxic study. Braz. J. Farmacogn. 2017, 27, 206–213. [Google Scholar] [CrossRef]
- Suffness, M.; Pezzuto, J.M. Assays related to cancer drug discovery. In Methods in Plant Biochemistry: Assays for Bioactivity; Hostettmann, K., Ed.; Academic Press: London, UK, 1990; pp. 71–133. [Google Scholar]
- Valadão, A.L.C.; Pezzuto, P.; Silva, V.A.O.; Gonçalves, B.S.; Rossi, Á.D.; da Cunha, R.D.; Siani, A.C.; Tostes, J.B.F.; Trovó, M.; Damasco, P.; et al. Reactivation of latent HIV-1 in vitro using an ethanolic extract from Euphorbia umbellata (Euphorbiaceae) latex. PLoS ONE 2018, 13, 0207664. [Google Scholar] [CrossRef]
- Fu, Z.; Han, X.; Du, J.; Han, X.; Liu, W.; Shao, S.; Liu, X. Euphorbia lunulata extract acts on multidrug resistant gastric cancer cells to inhibit cell proliferation, migration and invasion, arrest cell cycle progression, and induce apoptosis. J. Ethnopharmacol. 2018, 212, 8–17. [Google Scholar] [CrossRef]
- Liu, Y.; Murakami, N.; Ji, H.; Abreu, P.; Zhang, S. Antimalarial flavonol glycosides from Euphorbia hirta. Pharm. Biol. 2007, 45, 278–281. [Google Scholar] [CrossRef]
- Yan, S.L.; Li, Y.H.; Chen, X.Q.; Liu, D.; Chen, C.H.; Li, R.T. Diterpenes from the stem bark of Euphorbia neriifolia and their in vitro anti-HIV activity. Phytochemistry 2018, 145, 40–47. [Google Scholar] [CrossRef]
- Wang, P.; Lu, P.; Qu, X.; Shen, Y.; Zeng, H.; Zhu, X.; Zhu, Y.; Li, X.; Wu, H.; Xu, J.; et al. Reactivation of HIV-1 from latency by an ingenol derivative from Euphorbia kansui. Sci. Rep. 2017, 7, 9451. [Google Scholar] [CrossRef]
- Teng, Y.N.; Wang, Y.; Hsu, P.L.; Xin, G.; Zhang, Y.; Morris-Natschke, S.L.; Goto, M.; Lee, K.H. Mechanism of action of cytotoxic compounds from the seeds of Euphorbia lathyris. Phytomedicine 2018, 41, 62–66. [Google Scholar] [CrossRef]
- Lu, Z.Q.; Guan, S.H.; Li, X.N.; Chen, G.T.; Zhang, J.Q.; Huang, H.L.; Liu, X.; Guo, D.A. Cytotoxic diterpenoids from Euphorbia helioscopia. J. Nat. Prod. 2008, 71, 873–876. [Google Scholar] [CrossRef]
- Safwat, N.A.; Kashef, M.T.; Aziz, R.K.; Amer, K.F.; Ramadan, M.A. Quercetin 3-O-glucoside recovered from the wild Egyptian Sahara plant, Euphorbia paralias L., inhibits glutamine synthetase and has antimycobacterial activity. Tuberculosis 2018, 108, 106–113. [Google Scholar] [CrossRef]
- Wang, A.H.; Tian, X.G.; Cui, Y.L.; Huo, X.K.; Zhang, B.J.; Deng, S.; Feng, L.; Ma, X.C.; Jia, J.M.; Wang, C. Diterpenoids from the roots of Euphorbia ebracteolata and their inhibitory effects on human carboxylesterase 2. Phytochemistry 2018, 146, 82–90. [Google Scholar] [CrossRef]
- Mai, Z.; Ni, G.; Liu, Y.; Zhang, Z.; Li, L.; Chen, N.; Yu, D. Helioscopianoids A–Q, bioactive jatrophane diterpenoid esters from Euphorbia helioscopia. Acta Pharm. Sin. B 2018, 8, 805–817. [Google Scholar] [CrossRef]
- Cruz, L.S.; de Oliveira, T.L.; Kanunfre, C.C.; Paludo, K.S.; Minozzo, B.R.; Prestes, A.P.; Wang, M.; Fernandes, D.; Santos, F.A.D.; Manda, V.K.; et al. Pharmacokinetics and cytotoxic study of euphol from Euphorbia umbellata (Bruyns) Pax latex. Phytomedicine 2018, 47, 105–112. [Google Scholar] [CrossRef]
- Silva, V.A.O.; Rosa, M.N.; Miranda-Gonçalves, V.; Costa, A.M.; Tansini, A.; Evangelista, A.F.; Martinho, O.; Carloni, A.C.; Jones, C.; Lima, J.P.; et al. Euphol, a tetracyclic triterpene, from Euphorbia tirucalli induces autophagy and sensitizes temozolomide cytotoxicity on glioblastoma cells. Investig. New Drugs 2019, 37, 223–237. [Google Scholar] [CrossRef]
- Galvez, J.; Zarzuelo, A.; Crespo, M.E.; Lorente, M.D.; Ocete, M.A.; Jimenez, J. Antidiarrhoeic activity of Euphorbia hirta extract and isolation of an active flavonoid constituent. Planta Med. 1993, 59, 333–336. [Google Scholar] [CrossRef]
- Gálvez, J.; Crespo, M.E.; Jiménez, J.; Suárez, A.; Zarzuelo, A. Antidiarrhoeic activity of quercitrin in mice and rats. J. Pharm. Pharmacol. 1993, 45, 157–159. [Google Scholar] [CrossRef]
- Johnson, P.B.; Abdurahman, E.M.; Tiam, E.A.; Abdu-Aguye, I.; Hussaini, I.M. Euphorbia hirta leaf extracts increase urine output and electrolytes in rats. J. Ethnopharmacol. 1999, 65, 63–69. [Google Scholar] [CrossRef]
- Ibrahim, E.H.; Shaker, K.H.; Kilany, M.; Ghramh, H.A. Cytokines/chemokines profile in rats treated with Euphorbia tirucalli extract. Arab. J. Sci. Eng. 2018, 43, 3443–3451. [Google Scholar] [CrossRef]
- Palit, P.; Mukherjee, D.; Mahanta, P.; Shadab, M.; Ali, N.; Roychoudhury, S.; Asad, M.; Mandal, S.C. Attenuation of nociceptive pain and inflammatory disorders by total steroid and terpenoid fraction of Euphorbia tirucalli Linn root in experimental in vitro and in vivo model. Inflammopharmacology 2018, 26, 235–250. [Google Scholar] [CrossRef]
- Lim, H.J.; Jeon, Y.D.; Kang, S.H.; Shin, M.K.; Lee, K.M.; Jung, S.E.; Cha, J.Y.; Lee, H.Y.; Kim, B.R.; Hwang, S.W.; et al. Inhibitory effects of Euphorbia supina on Propionibacterium acnes-induced skin inflammation in vitro and in vivo. BMC Complement. Altern. Med. 2018, 18, 263. [Google Scholar] [CrossRef]
- Özbilgin, S.; Akkol, E.K.; Süntar, İ.; Tekin, M.; İşcan, G.S. Wound-healing activity of some species of Euphorbia L. Rec. Nat. Prod. 2019, 13, 104–113. [Google Scholar] [CrossRef]
- Özbilgin, S.; Acıkara, Ö.B.; Akkol, E.K.; Süntar, I.; Keleş, H.; İşcan, G.S. In vivo wound-healing activity of Euphorbia characias subsp. wulfenii: Isolation and quantification of quercetin glycosides as bioactive compounds. J. Ethnopharmacol. 2018, 224, 400–408. [Google Scholar]
- Rehman, A.H.; Al Sharari, S.D.; Ahmad, M.; Akhtar, M.; Khan, Y.; Ashraf, M.N. Evaluation of anticonvulsant and antiepileptogenic activity of Euphorbia nivulia in PTZ-induced kindling model of epilepsy in mice. Pak. J. Pharm. Sci. 2019, 32, 675–681. [Google Scholar]
- Perera, S.D.; Jayawardena, U.A.; Jayasinghe, C.D. Potential use of Euphorbia hirta for dengue: A systematic review of scientific evidence. J. Trop. Med. 2018, 2018, 2048530. [Google Scholar] [CrossRef]
- Aleksandrov, M.; Maksimova, V.; Koleva Gudeva, L. Review of the anticancer and cytotoxic activity of some species from genus Euphorbia. Agric. Conspec. Sci. 2019, 84, 1–5. [Google Scholar]
- Mohamed, H.M.; Aly, M.S. Evaluation of genotoxicity of Euphorbia triaculeata Forssk. extract on mice bone marrow cells in vivo. Toxicol. Rep. 2018, 5, 625–631. [Google Scholar] [CrossRef]
- Li, J.; Li, H.H.; Wang, W.Q.; Song, W.B.; Wang, Y.P.; Xuan, L.J. Jatrophane diterpenoids from Euphorbia helioscopia and their lipid-lowering activities. Fitoterapia 2018, 128, 102–111. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, S.; Cheng, X.; Lu, C.; Tao, W.; Zhang, Y.; William, B.C.; Cao, X.; Yi, S.; Liu, Y.; et al. Euphorbia factor L2 alleviates lipopolysaccharide-induced acute lung injury and inflammation in mice through the suppression of NF-κB activation. Biochem. Pharmacol. 2018, 155, 444–454. [Google Scholar] [CrossRef]
- Lu, F.; Huayun, Z.; Weiwei, T.; Li, L.; Xin, S.; Ming, Z.; Dongdong, S. Euphorbia factor L2 inhibits TGF-β-induced cell growth and migration of hepatocellular carcinoma through AKT/STAT3. Phytomedecine 2019, 62, 152931. [Google Scholar]
- Barla, A.; Birman, H.B.; Kültür, S.; Öksüz, S. Secondary metabolites from Euphorbia helioscopia and their vasodepressor activity. Turk. J. Chem. 2006, 30, 325–332. [Google Scholar]
- Ramsay, J.R.; Suhrbier, A.; Aylward, J.H.; Ogbourne, S.; Cozzi, S.J.; Poulsen, M.G.; Baumann, K.C.; Welburn, P.; Redlich, G.L.; Parsons, P.G. The sap from Euphorbia peplus is effective against human nonmelanoma skin cancers. Br. J. Dermatol. 2011, 164, 633–636. [Google Scholar] [CrossRef]
- Mir, M.; Khurshid, R.; Aftab, R. Management of thrombocytopenia and flu-like symptoms in dengue patients with herbal water of Euphorbia hirta. J. Ayub Med. Coll. 2012, 24, 6–9. [Google Scholar]
- ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ct2/results?term=euphorbia&draw=1&rank=1#rowId0 (accessed on 22 June 2019).
- Gupta, P.J. The efficacy of Euphorbia prostrata in early grades of symptomatic haemorrhoids—A pilot study. Eur. Rev. Med. Pharmacol. Sci. 2011, 15, 199–203. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).