Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review
Abstract
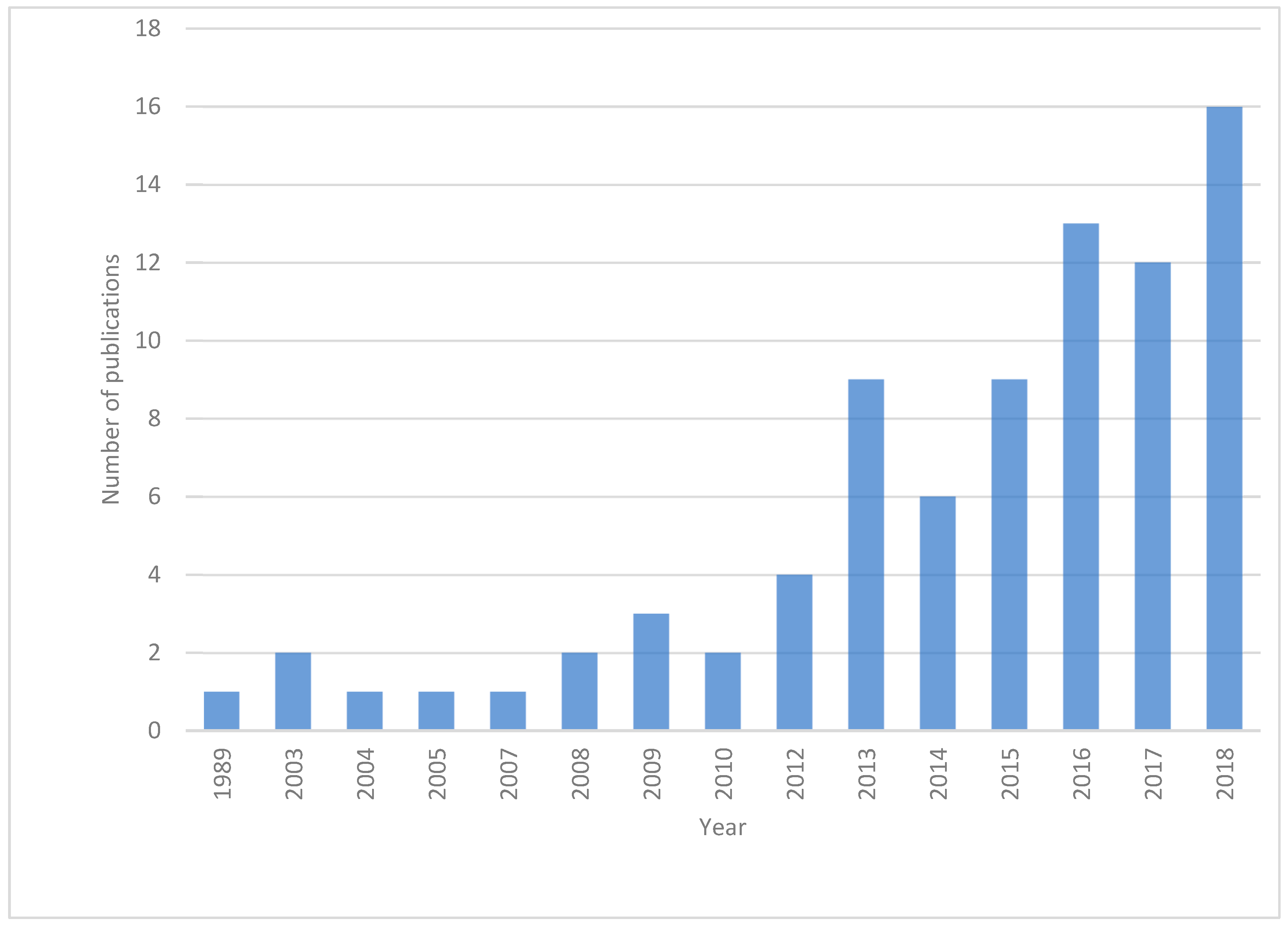
1. Introduction
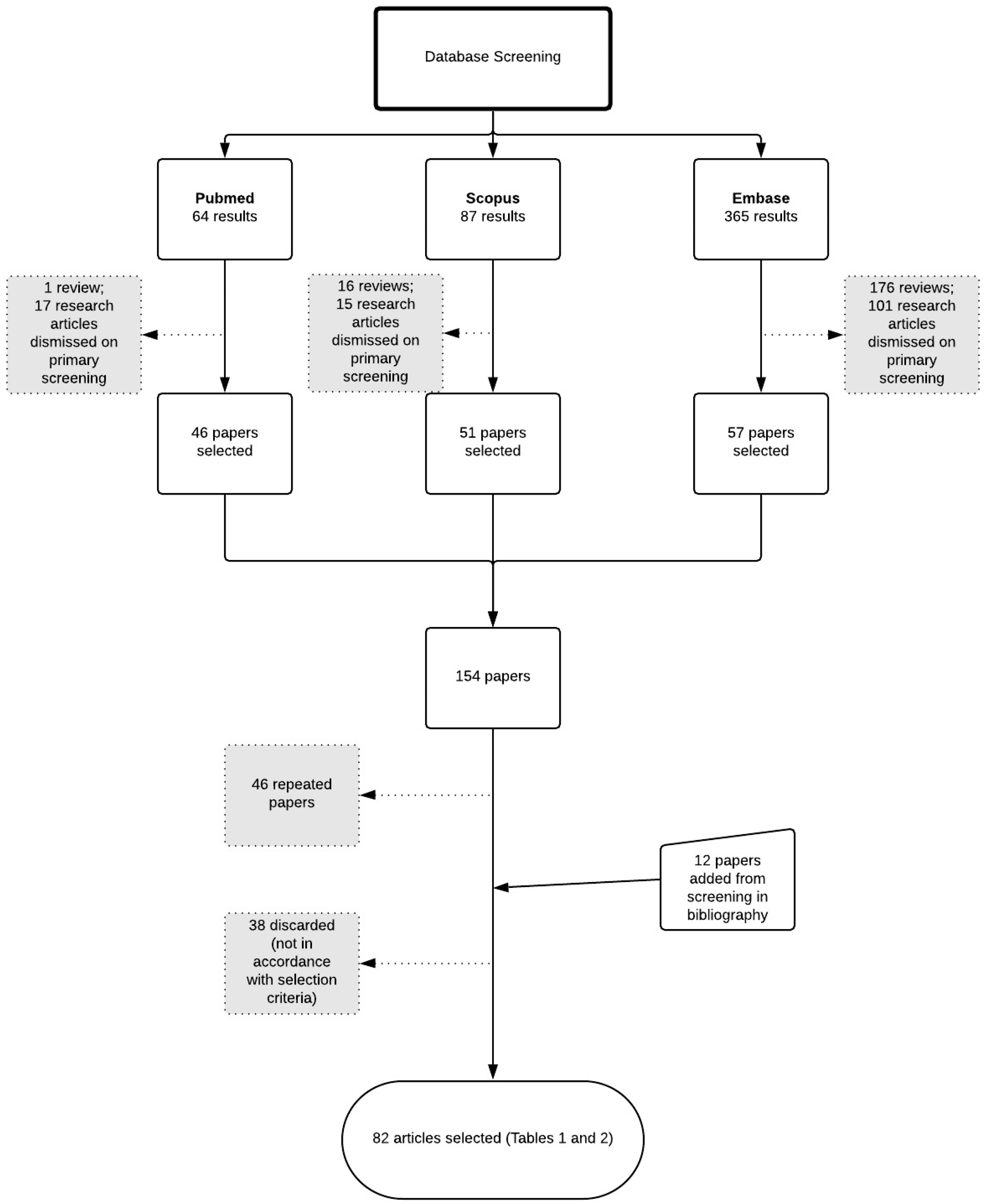
2. Literature Survey
- Original research data.
- Use of essential oil containing terpenes and/or isolated terpenes in nanostructured systems or cyclodextrins.
- Formulations for topical administration.
3. Discussion
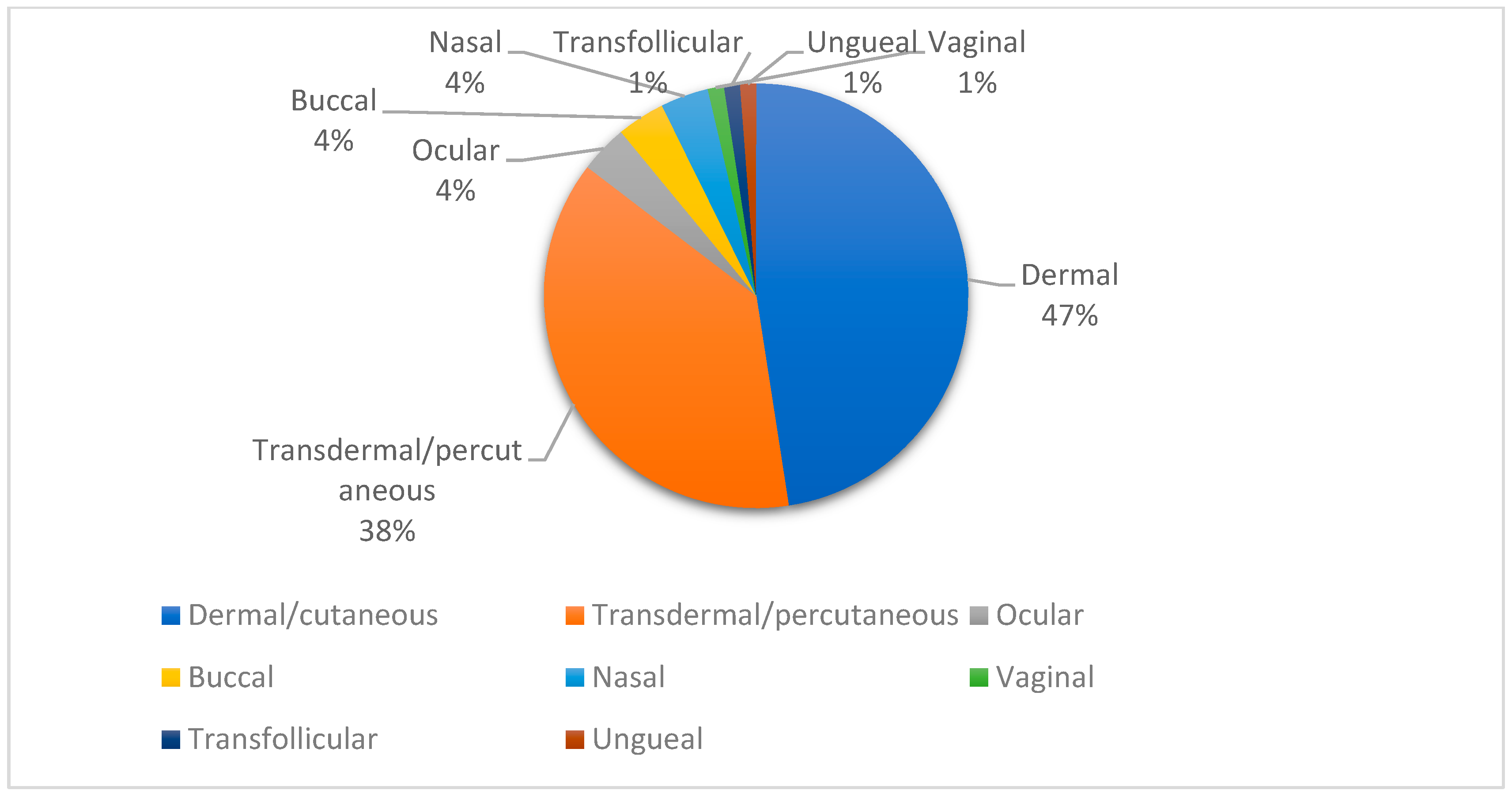
3.1. Topical Administration Routes
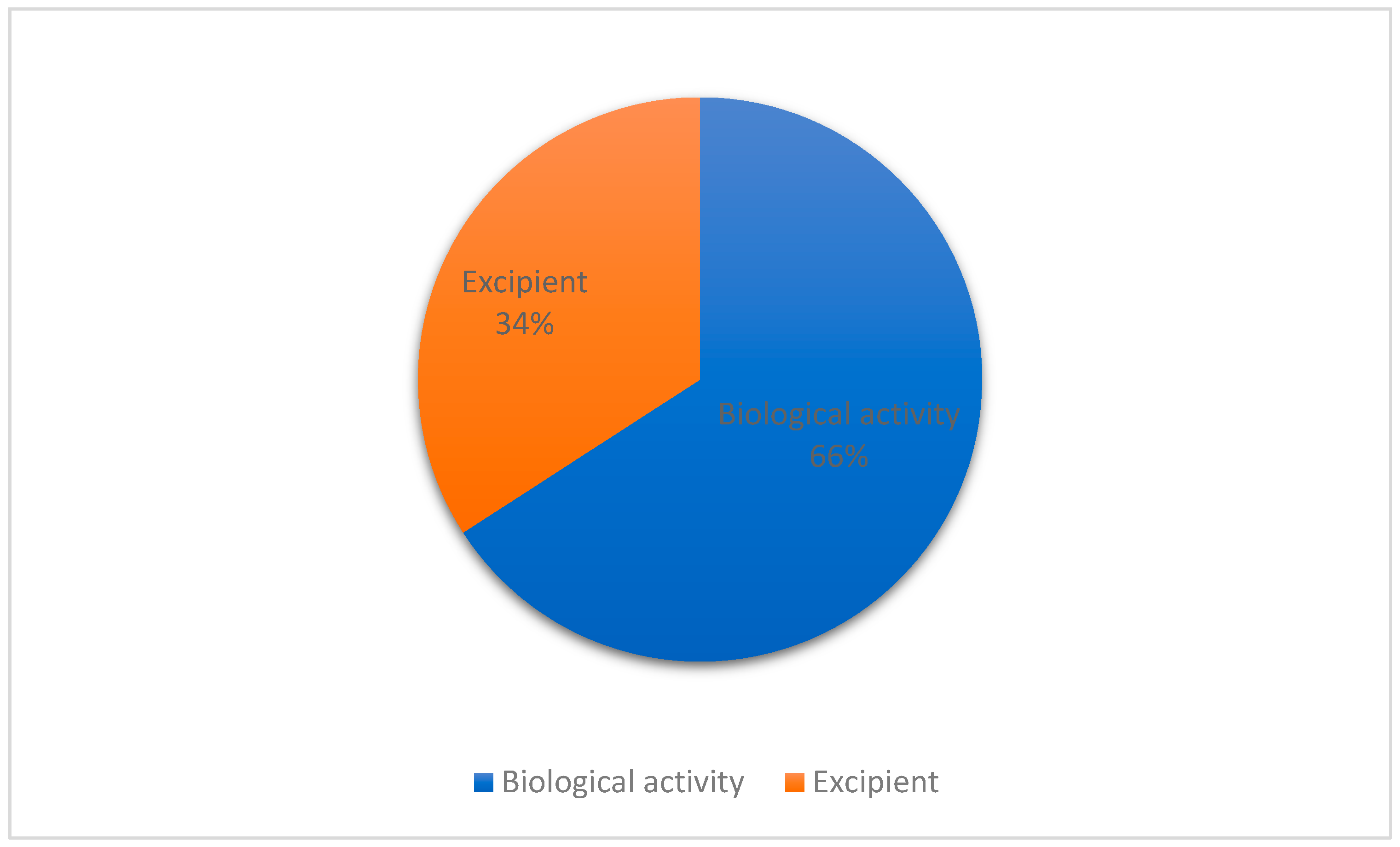
3.2. Role of Essential Oil and/or Terpenes in the Formulations
3.2.1. Nanostructured Systems Used in the Encapsulation of Terpenes and Essential Oils as Bioactive Ingredients
3.2.2. Terpenes as Excipients in Formulations for Topical Administration
3.3. Safety of Essential Oils and Terpenes in Topical Administration
3.4. Sustainability
4. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Asbahani, A.E.; Miladi, K.; Badri, W.; Sala, M.; Addi, E.H.A.; Casabianca, H.; Mousadik, A.E.; Hartmann, D.; Jilale, A.; Renaud, F.N.R.; et al. Essential oils: From extraction to encapsulation. Int. J. Pharm. 2015, 483, 220–243. [Google Scholar] [CrossRef] [PubMed]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential oils loaded in nanosystems: A developing strategy for a successful therapeutic approach. Evid. Based Complement. Alternat. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Majeed, H.; Bian, Y.-Y.; Ali, B.; Jamil, A.; Majeed, U.; Khan, Q.F.; Iqbal, K.J.; Shoemaker, C.F.; Fang, Z. Essential oil encapsulations: Uses, procedures, and trends. RSC Adv. 2015, 5, 58449–58463. [Google Scholar] [CrossRef]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007, 3, 408. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E.; Gertsch, J.; Appendino, G. Plant volatiles: Production, function and pharmacology. Nat. Prod. Rep. 2011, 28, 1359–1380. [Google Scholar] [CrossRef] [PubMed]
- Turek, C.; Stintzing, F.C. Stability of Essential Oils: A Review. Compr. Rev. Food Sci. F 2013, 12, 40–53. [Google Scholar] [CrossRef]
- Herman, A.; Herman, A.P. Essential oils and their constituents as skin penetration enhancer for transdermal drug delivery: A review. J. Pharm. Pharmacol. 2014, 67, 473–485. [Google Scholar] [CrossRef]
- Sapra, B.; Jain, S.; Tiwary, A.K. Percutaneous Permeation Enhancement by Terpenes: Mechanistic View. AAPS J. 2008, 10, 120. [Google Scholar] [CrossRef]
- Hamm, S.; Bleton, J.; Connan, J.; Tchapla, A. A chemical investigation by headspace SPME and GC–MS of volatile and semi-volatile terpenes in various Olibanum samples. Phytochemistry 2005, 66, 1499–1514. [Google Scholar] [CrossRef]
- Pivetta, T.P.; Simões, S.; Araújo, M.M.; Carvalho, T.; Arruda, C.; Marcato, P.D. Development of nanoparticles from natural lipids for topical delivery of thymol: Investigation of its anti-inflammatory properties. Colloids Surf. B Biointerfaces 2018, 164, 281–290. [Google Scholar] [CrossRef]
- Takada, H.; Yonekawa, J.; Matsumoto, M.; Furuya, K.; Sokabe, M. Hyperforin/HP-β-Cyclodextrin Enhances Mechanosensitive Ca2+Signaling in HaCaT Keratinocytes and in Atopic Skin Ex Vivo Which Accelerates Wound Healing. Biomed. Res. Int. 2017, 2017, 8701801. [Google Scholar] [CrossRef]
- Tosta, F.V.; Andrade, L.M.; Mendes, L.P.; Anjos, J.L.V.; Alonso, A.; Marreto, R.N.; Lima, E.M.; Taveira, S.F. Paclitaxel-loaded lipid nanoparticles for topical application: The influence of oil content on lipid dynamic behavior, stability, and drug skin penetration. J. Nanopart. Res. 2014, 16, 1–12. [Google Scholar] [CrossRef]
- Singh, H.; Sharma, R.; Joshi, M.; Garg, T.; Goyal, A.K.; Rath, G. Transmucosal delivery of Docetaxel by mucoadhesive polymeric nanofibers. Artif. Cells Nanomed. Biotechnol. 2015, 43, 263–269. [Google Scholar] [CrossRef]
- Bharadwaj, R.; Das, P.J.; Pal, P.; Mazumder, B. Topical delivery of paclitaxel for treatment of skin cancer. Drug Dev. Ind. Pharm. 2016, 42, 1482–1494. [Google Scholar] [CrossRef]
- Sinha, V.R.; Kaur, M.P. Permeation Enhancers for Transdermal Drug Delivery. Drug Dev. Ind. Pharm. 2000, 26, 1131–1140. [Google Scholar] [CrossRef]
- Scott, R.P.W. ESSENTIAL OILS. In Encyclopedia of Analytical Science, 2nd ed.; Worsfold, P., Townshend, A., Poole, C., Eds.; Elsevier: Oxford, UK, 2005; pp. 554–561. [Google Scholar]
- Bhowmik, D.; Gopinath, H.; Kumar, B.P.; Duraivel, S.; Kumar, K.S. Recent advances in novel topical drug delivery system. Pharma Innov. 2012, 1, 12. [Google Scholar]
- Brown, M.B.; Martin, G.P.; Jones, S.A.; Akomeah, F.K. Dermal and Transdermal Drug Delivery Systems: Current and Future Prospects. Drug Deliv. 2006, 13, 175–187. [Google Scholar] [CrossRef]
- Guilherme, V.A.; Ribeiro, L.N.M.; Tofoli, G.R.; Franz-Montan, M.; de Paula, E.; de Jesus, M.B. Current challenges and future of lipid nanoparticles formulations for topical drug application to oral mucosa, skin, and eye. Curr. Pharm. Des. 2017, 23, 6659–6675. [Google Scholar] [CrossRef]
- Sezer, A.D.; Cevher, E. Topical drug delivery using chitosan nano- and microparticles. Expert Opin. Drug Deliv. 2012, 9, 1129–1146. [Google Scholar] [CrossRef]
- Bilia, A.R.; Piazzini, V.; Guccione, C.; Risaliti, L.; Asprea, M.; Capecchi, G.; Bergonzi, M.C. Improving on Nature: The Role of Nanomedicine in the Development of Clinical Natural Drugs. Planta Med. 2017, 83, 366–381. [Google Scholar] [CrossRef]
- Barua, S.; Mitragotri, S. Challenges associated with penetration of nanoparticles across cell and tissue barriers: A review of current status and future prospects. Nano Today 2014, 9, 223–243. [Google Scholar] [CrossRef]
- Lima, P.S.S.; Lucchese, A.M.; Araújo-Filho, H.G.; Menezes, P.P.; Araújo, A.A.S.; Quintans-Júnior, L.J.; Quintans, J.S.S. Inclusion of terpenes in cyclodextrins: Preparation, characterization and pharmacological approaches. Carbohydr. Polym. 2016, 151, 965–987. [Google Scholar] [CrossRef]
- Saettone, M.F.; Burgalassi, S.; Giannaccini, B. Preparation and Evaluation in Rabbits of Topical Solutions Containing Forskolin. J. Ocul. Pharmacol. Ther. 1989, 5, 111–118. [Google Scholar] [CrossRef]
- El-Nabarawi, M.A.; Shamma, R.N.; Farouk, F.; Nasralla, S.M. Dapsone-Loaded Invasomes as a Potential Treatment of Acne: Preparation, Characterization, and In Vivo Skin Deposition Assay. AAPS PharmSciTech 2018, 19, 2174–2184. [Google Scholar] [CrossRef]
- Ahmad, A.; Abuzinadah, M.F.; Alkreathy, H.M.; Banaganapalli, B.; Mujeeb, M. Ursolic acid rich ocimum sanctum L leaf extract loaded nanostructured lipid carriers ameliorate adjuvant induced arthritis in rats by inhibition of COX-1, COX-2, TNF-α and IL-1: Pharmacological and docking studies. PLoS ONE 2018, 13, e0193451. [Google Scholar] [CrossRef]
- Abdel-Hafez, S.M.; Hathout, R.M.; Sammour, O.A. Curcumin-loaded ultradeformable nanovesicles as a potential delivery system for breast cancer therapy. Colloids Surf. B Biointerfaces 2018, 167, 63–72. [Google Scholar] [CrossRef]
- Cui, Y.; Mo, Y.; Zhang, Q.; Tian, W.; Xue, Y.; Bai, J.; Du, S. Microneedle-Assisted Percutaneous Delivery of Paeoniflorin-Loaded Ethosomes. Molecules 2018, 23, 3371. [Google Scholar] [CrossRef]
- Song, K.; Xin, M.; Yu, H.; Zheng, Z.; Li, J.; Li, M.; Guo, H.; Tan, Y.; Wu, X. Novel ultra-small micelles based on rebaudioside A: A potential nanoplatform for ocular drug delivery. Int. J. Pharm. 2018, 552, 265–276. [Google Scholar] [CrossRef]
- Sims, K.R.; Liu, Y.; Hwang, G.; Jung, H.I.; Koo, H.; Benoit, D.S.W. Enhanced design and formulation of nanoparticles for anti-biofilm drug delivery. Nanoscale 2019, 11, 219–236. [Google Scholar] [CrossRef]
- Abd, E.; Benson, H.A.E.; Roberts, M.S.; Grice, J.E. Follicular Penetration of Caffeine from Topically Applied Nanoemulsion Formulations Containing Penetration Enhancers: In vitro Human Skin Studies. Skin Pharmacol. Physiol. 2018, 31, 252–260. [Google Scholar] [CrossRef]
- Nagai, N.; Ogata, F.; Otake, H.; Nakazawa, Y.; Kawasaki, N. Design of a transdermal formulation containing raloxifene nanoparticles for osteoporosis treatment. Int. J. Nanomed. 2018, 13, 5215–5229. [Google Scholar] [CrossRef]
- Freag, M.S.; Saleh, W.M.; Abdallah, O.Y. Laminated chitosan-based composite sponges for transmucosal delivery of novel protamine-decorated tripterine phytosomes: Ex-vivo mucopenetration and in-vivo pharmacokinetic assessments. Carbohydr. Polym. 2018, 188, 108–120. [Google Scholar] [CrossRef]
- Khan, K.; Aqil, M.; Imam, S.S.; Ahad, A.; Moolakkadath, T.; Sultana, Y.; Mujeeb, M. Ursolic acid loaded intra nasal nano lipid vesicles for brain tumour: Formulation, optimization, in-vivo brain/plasma distribution study and histopathological assessment. Biomed. Pharmacother. 2018, 106, 1578–1585. [Google Scholar] [CrossRef]
- Bonaccorso, A.; Musumeci, T.; Carbone, C.; Vicari, L.; Lauro, M.R.; Puglisi, G. Revisiting the role of sucrose in PLGA-PEG nanocarrier for potential intranasal delivery. Pharm. Dev. Technol. 2018, 23, 265–274. [Google Scholar] [CrossRef]
- Lason, E.; Sikora, E.; Miastkowska, M.; Escribano, E.; Garcia-Celma, M.J.; Solans, C.; Llinas, M.; Ogonowski, J. NLCs as a potential carrier system for transdermal delivery of forskolin. Acta Biochim. Pol. 2018, 65, 437–442. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Fei, Y.R.; Fang, L.; Zheng, H.S.; Mu, C.F.; Li, F.Z.; Zhang, Y.S. Preparation of mixed monoterpenes edge activated PEGylated transfersomes to improve the in vivo transdermal delivery efficiency of sinomenine hydrochloride. Int. J. Pharm. 2017, 533, 266–274. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, Y.; Li, Z.; Li, N.; Feng, N. Essential oil-mediated glycerosomes increase transdermal paeoniflorin delivery: Optimization, characterization, and evaluation in vitro and in vivo. Int. J. Nanomed. 2017, 12, 3521–3532. [Google Scholar] [CrossRef]
- Duangjit, S.; Nimcharoenwan, T.; Chomya, N.; Locharoenrat, N.; Ngawhirunpat, T. Computational design strategy: An approach to enhancing the transdermal delivery of optimal capsaicin-loaded transinvasomes. Drug Dev. Ind. Pharm. 2017, 43, 98–107. [Google Scholar] [CrossRef]
- Rangsimawong, W.; Obata, Y.; Opanasopit, P.; Ngawhirunpat, T.; Takayama, K. Enhancement of Galantamine HBr Skin Permeation Using Sonophoresis and Limonene-Containing PEGylated Liposomes. AAPS PharmSciTech 2017, 19, 1093–1104. [Google Scholar] [CrossRef]
- Yang, M.; Gu, Y.; Yang, D.; Tang, X.; Liu, J. Development of triptolide-nanoemulsion gels for percutaneous administration: Physicochemical, transport, pharmacokinetic and pharmacodynamic characteristics. J. Nanobiotechnol. 2017, 15, 88. [Google Scholar] [CrossRef]
- Qadri, G.R.; Ahad, A.; Aqil, M.; Imam, S.S.; Ali, A. Invasomes of isradipine for enhanced transdermal delivery against hypertension: Formulation, characterization, and in vivo pharmacodynamic study. Artif. Cells Nanomed. Biotechnol. 2017, 45, 139–145. [Google Scholar] [CrossRef]
- Teixeira, G.F.D.; Vieira-Neto, A.E.; da Costa, F.N.; e Silva, A.R.A.; Campos, A.R. Antinociceptive effect of (-)-α-bisabolol in nanocapsules. Biomed. Pharmacother. 2017, 91, 946–950. [Google Scholar] [CrossRef]
- Abdelsamie, S.M.; Kamel, A.O.; Sammour, O.A.; Ibrahim, S.M. Terbinafine hydrochloride nanovesicular gel: In vitro characterization, ex vivo permeation and clinical investigation. Eur. J. Pharm. Sci. 2016, 88, 91–100. [Google Scholar] [CrossRef]
- Kamran, M.; Ahad, A.; Aqil, M.; Imam, S.S.; Sultana, Y.; Ali, A. Design, formulation and optimization of novel soft nano-carriers for transdermal olmesartan medoxomil delivery: In vitro characterization and in vivo pharmacokinetic assessment. Int. J. Pharm. 2016, 505, 147–158. [Google Scholar] [CrossRef]
- Gogoll, K.; Stein, P.; Lee, K.; Arnold, P.; Peters, T.; Schild, H.; Radsak, M.; Langguth, P. Solid nanoemulsion as antigen and immunopotentiator carrier for transcutaneous immunization. Cell. Immunol. 2016, 308, 35–43. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.; Liu, B.; Du, L.; Jia, X.; Han, L.; Jin, Y. Tea tree oil nanoemulsions for inhalation therapies of bacterial and fungal pneumonia. Colloids Surf. B Biointerfaces 2016, 141, 408–416. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, X.; Du, L.; Jin, Y. Preparation of asiaticoside-loaded coaxially electrospinning nanofibers and their effect on deep partial-thickness burn injury. Biomed. Pharmacother. 2016, 83, 33–40. [Google Scholar] [CrossRef]
- Shen, C.Y.; Xu, P.H.; Shen, B.D.; Min, H.Y.; Li, X.R.; Han, J.; Yuan, H.L. Nanogel for dermal application of the triterpenoids isolated from Ganoderma lucidum (GLT) for frostbite treatment. Drug Deliv. 2016, 23, 610–618. [Google Scholar] [CrossRef][Green Version]
- Ahmad, M.; Rai, S.M.; Mahmood, A. Hydrogel Microparticles as an Emerging Tool in Pharmaceutical Field: A Review. Adv. Polym. Technol. 2016, 35, 121–128. [Google Scholar] [CrossRef]
- Jamal, M.; Imam, S.S.; Aqil, M.; Amir, M.; Mir, S.R.; Mujeeb, M. Transdermal potential and anti-arthritic efficacy of ursolic acid from niosomal gel systems. Int. Immunopharmacol. 2015, 29, 361–369. [Google Scholar] [CrossRef]
- Horev, B.; Klein, M.I.; Hwang, G.; Li, Y.; Kim, D.; Koo, H.; Benoit, D.S.W. pH-activated Nanoparticles for Controlled Topical Delivery of Farnesol to Disrupt Oral Biofilm Virulence. ACS Nano 2015, 9, 2390–2404. [Google Scholar] [CrossRef]
- Alvarado, H.; Abrego, G.; Souto, E.B.; Garduño-Ramirez, M.L.; Clarés, B.; García, M.; Calpena, A. Nanoemulsions for dermal controlled release of oleanolic and ursolic acids: In vitro, ex vivo and in vivo characterization. Colloids Surf. B Biointerfaces 2015, 130, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.-C.; Chiang, B.-H.; Huang, D.-W.; Li, P.-H. Skin permeation of d-limonene-based nanoemulsions as a transdermal carrier prepared by ultrasonic emulsification. Ultrason. Sonochem. 2014, 21, 826–832. [Google Scholar] [CrossRef]
- Rangsimawong, W.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T. Terpene-Containing PEGylated Liposomes as Transdermal Carriers of a Hydrophilic Compound. Boil. Pharm. Bull. 2014, 37, 1936–1943. [Google Scholar] [CrossRef]
- Charoenputtakun, P.; Pamornpathomkul, B.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T. Terpene Composited Lipid Nanoparticles for Enhanced Dermal Delivery of All-trans-Retinoic Acids. Boil. Pharm. Bull. 2014, 37, 1139–1148. [Google Scholar] [CrossRef]
- Dehelean, C.A.; Feflea, S.; Gheorgheosu, D.; Ganta, S.; Cimpean, A.M.; Muntean, D.; Amiji, M.M. Anti-Angiogenic and Anti-Cancer Evaluation of Betulin Nanoemulsion in Chicken Chorioallantoic Membrane and Skin Carcinoma in Balb/c Mice. J. Biomed. Nanotechnol. 2013, 9, 577–589. [Google Scholar] [CrossRef]
- Sandig, A.G.; Campmany, A.C.; Campos, F.F.; Villena, M.M.; Naveros, B.C. Transdermal delivery of imipramine and doxepin from newly oil-in-water nanoemulsions for an analgesic and anti-allodynic activity: Development, characterization and in vivo evaluation. Colloids Surf. B Biointerfaces 2013, 103, 558–565. [Google Scholar] [CrossRef]
- Chen, X.; Peng, L.-H.; Shan, Y.-H.; Li, N.; Wei, W.; Yu, L.; Li, Q.-M.; Liang, W.-Q.; Gao, J.-Q. Astragaloside IV-loaded nanoparticle-enriched hydrogel induces wound healing and anti-scar activity through topical delivery. Int. J. Pharm. 2013, 447, 171–181. [Google Scholar] [CrossRef]
- Subongkot, T.; Wonglertnirant, N.; Songprakhon, P.; Rojanarata, T.; Opanasopit, P.; Ngawhirunpat, T. Visualization of ultradeformable liposomes penetration pathways and their skin interaction by confocal laser scanning microscopy. Int. J. Pharm. 2013, 441, 151–161. [Google Scholar] [CrossRef]
- Barros, N.B.; Migliaccio, V.; Facundo, V.A.; Ciancaglini, P.; Stabeli, R.G.; Nicolete, R.; Silva-Jardim, I. Liposomal-lupane system as alternative chemotherapy against cutaneous leishmaniasis: Macrophage as target cell. Exp. Parasitol. 2013, 135, 337–343. [Google Scholar] [CrossRef]
- Patel, P.A.; Patil, S.C.; Kalaria, D.R.; Kalia, Y.N.; Patravale, V.B. Comparative in vitro and in vivo evaluation of lipid based nanocarriers of Huperzine A. Int. J. Pharm. 2013, 446, 16–23. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, L.; Yuan, L.; Zhang, Z.-H.; Liu, X.; Wu, Q. Formulation, characterization, and evaluation of in vitro skin permeation and in vivo pharmacodynamics of surface-charged tripterine-loaded nanostructured lipid carriers. Int. J. Nanomed. 2012, 7, 3023–3033. [Google Scholar]
- Paolino, D.; Cosco, D.; Cilurzo, F.; Trapasso, E.; Morittu, V.M.; Celia, C.; Fresta, M. Improved in vitro and in vivo collagen biosynthesis by asiaticoside-loaded ultradeformable vesicles. J. Control. 2012, 162, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-G.; Liu, Y.-F.; Gao, T.-W. Preparation and anti-inflammatory activity of triptolide ethosomes in an erythema model. J. Liposome Res. 2010, 20, 297–303. [Google Scholar] [CrossRef]
- Fang, J.-Y. Combination of calcipotriol and methotrexate in nanostructured lipid carriers for topical delivery. Int. J. Nanomed. 2010, 5, 117. [Google Scholar] [CrossRef]
- Mura, S.; Manconi, M.; Sinico, C.; Valenti, D.; Fadda, A.M. Penetration enhancer-containing vesicles (PEVs) as carriers for cutaneous delivery of minoxidil. Int. J. Pharm. 2009, 380, 72–79. [Google Scholar] [CrossRef]
- Dragicevic-Curic, N.; Gräfe, S.; Albrecht, V.; Fahr, A. Topical application of temoporfin-loaded invasomes for photodynamic therapy of subcutaneously implanted tumours in mice: A pilot study. J. Photochem. Photobiol. B Boil. 2008, 91, 41–50. [Google Scholar] [CrossRef]
- Dragicevic-Curic, N.; Scheglmann, D.; Albrecht, V.; Fahr, A. Temoporfin-loaded invasomes: Development, characterization and in vitro skin penetration studies. J. Control. 2008, 127, 59–69. [Google Scholar] [CrossRef]
- Dragicevic-Curic, N.; Scheglmann, D.; Albrecht, V.; Fahr, A. Development of different temoporfin-loaded invasomes—Novel nanocarriers of temoporfin: Characterization, stability and in vitro skin penetration studies. Colloids Surf. B Biointerfaces 2009, 70, 198–206. [Google Scholar] [CrossRef]
- Mei, Z.; Wu, Q.; Hu, S.; Lib, X.; Yang, X. Triptolide Loaded Solid Lipid Nanoparticle Hydrogel for Topical Application. Drug Dev. Ind. Pharm. 2005, 31, 161–168. [Google Scholar] [CrossRef]
- Rode, T. Complex formation of sericoside with hydrophilic cyclodextrins: Improvement of solubility and skin penetration in topical emulsion-based formulations. Eur. J. Pharm. Biopharm. 2003, 55, 191–198. [Google Scholar] [CrossRef]
- Mei, Z. Solid lipid nanoparticle and microemulsion for topical delivery of triptolide. Eur. J. Pharm. Biopharm. 2003, 56, 189–196. [Google Scholar] [CrossRef]
- Afifah, S.N.; Azhar, S.; Ashari, S.E.; Salim, N. Development of a kojic monooleate-enriched oil-in-water nanoemulsion as a potential carrier for hyperpigmentation treatment. Int. J. Nanomed. 2018, 13, 6465–6479. [Google Scholar]
- Barradas, T.N.; Senna, J.P.; Cardoso, S.A.; Silva, K.G.D.E.; Mansur, C.R.E. Formulation characterization and in vitro drug release of hydrogel-thickened nanoemulsions for topical delivery of 8-methoxypsoralen. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 92, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.A.; Gouvêa, M.M.; Antunes, G.R.; Freitas, Z.M.F.; Marques, F.F.C.; Ricci-Junior, E. Nanoemulsion containing 8-methoxypsoralen for topical treatment of dermatoses: Development, characterization and ex vivo permeation in porcine skin. Int. J. Pharm. 2018, 547, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Patel, D.K.; Rai, V.K.; Pal, A.; Yadav, N.P. Development of Emulgel formulation for vaginal candidiasis: Pharmaceutical characterization, in vitro and in vivo evaluation. J. Drug Deliv. Sci. Technol. 2018, 48, 490–498. [Google Scholar] [CrossRef]
- Alotaibi, F.O.; Mustafa, G.; Ahuja, A. Study of Enhanced Anti-Inflammatory Potential of Nigella Sativa in Topical Nanoformulation. Int. J. Pharm. Pharm. Sci. 2018, 10, 41. [Google Scholar] [CrossRef]
- Barradas, T.N.; Senna, J.P.; Cardoso, S.A.; Nicoli, S.; Padula, C.; Santi, P.; Rossi, F.; Silva, K.G.D.E.; Mansur, C.R.E. Hydrogel-thickened nanoemulsions based on essential oils for topical delivery of psoralen: Permeation and stability studies. Eur. J. Pharm. Biopharm. 2017, 116, 38–50. [Google Scholar] [CrossRef]
- Kalita, S.; Kandimalla, R.; Devi, B.; Kalita, B.; Kalita, K.; Deka, M.; Kataki, A.C.; Sharma, A.; Kotoky, J. Dual delivery of chloramphenicol and essential oil by poly-ε-caprolactone-Pluronic nanocapsules to treat MRSA-Candida co-infected chronic burn wounds. RSC Adv. 2017, 7, 1749–1758. [Google Scholar] [CrossRef]
- Montenegro, L.; Pasquinucci, L.; Zappalà, A.; Chiechio, S.; Turnaturi, R.; Parenti, C. Rosemary Essential Oil-Loaded Lipid Nanoparticles: In Vivo Topical Activity from Gel Vehicles. Pharmaceutics 2017, 9, 48. [Google Scholar] [CrossRef]
- Navayan, A.; Moghimipour, E.; Khodayar, M.J.; Vazirianzadeh, B.; Siahpoosh, A.; Valizadeh, M.; Mansourzadeh, Z. Evaluation of the Mosquito Repellent Activity of Nano-sized Microemulsion of Eucalyptus globulus Essential Oil Against Culicinae. Jundishapur J. Nat. Pharm. Prod. 2017, 12. [Google Scholar] [CrossRef]
- Alam, P.; Ansari, M.J.; Anwer, M.K.; Raish, M.; Kamal, Y.K.T.; Shakeel, F. Wound healing effects of nanoemulsion containing clove essential oil. Artif. Cells Nanomed. Biotechnol. 2017, 45, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.; Srivastava, S.; Mishra, N.; Singh, D.K.; Luqman, S.; Chanda, D.; Yadav, N.P. Development, optimization, and characterization of a novel tea tree oil nanogel using response surface methodology. Drug Dev. Ind. Pharm. 2016, 42, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sawatdee, S.; Choochuay, K.; Chanthorn, W.; Srichana, T. Evaluation of the topical spray containing Centella asiatica extract and its efficacy on excision wounds in rats. Acta Pharm. 2016, 66, 233–244. [Google Scholar] [CrossRef]
- Botelho, M.A.; Barros, G.; Queiroz, D.B.; Carvalho, C.F.; Gouvea, J.; Patrus, L.; Bannet, M.; Patrus, D.; Rego, A.; Silva, I.; et al. Nanotechnology in Phytotherapy: Antiinflammatory Effect of a Nanostructured Thymol Gel from Lippia sidoides in Acute Periodontitis in Rats. Phytother. Res. 2016, 30, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, D.M.; El-Alim, S.H.A.; Asfour, M.H.; Al-Okbi, S.Y.; Mohamed, D.A.; Awad, G.; El-Alim, S.A. Transdermal nanoemulsions of Foeniculum vulgare Mill. essential oil: Preparation, characterization and evaluation of antidiabetic potential. J. Drug Deliv. Sci. Technol. 2015, 29, 99–106. [Google Scholar] [CrossRef]
- Aldawsari, H.M.; Badr-Eldin, S.M.; Labib, G.S.; El-Kamel, A.H. Design and formulation of a topical hydrogel integrating lemongrass-loaded nanosponges with an enhanced antifungal effect: In vitro/in vivo evaluation. Int. J. Nanomed. 2015, 10, 893–902. [Google Scholar]
- Lucca, L.G.; De Matos, S.P.; Borille, B.T.; Dias, D.D.O.; Teixeira, H.F.; Veiga, V.F.; Limberger, R.P.; Koester, L.S. Determination of β-caryophyllene skin permeation/retention from crude copaiba oil (Copaifera multijuga Hayne) and respective oil-based nanoemulsion using a novel HS-GC/MS method. J. Pharm. Biomed. Anal. 2015, 104, 144–148. [Google Scholar] [CrossRef]
- Castangia, I.; Manca, M.L.; Caddeo, C.; Maxia, A.; Murgia, S.; Pons, R.; Demurtas, D.; Pando, D.; Falconieri, D.; Peris, J.E.; et al. Faceted phospholipid vesicles tailored for the delivery of Santolina insularis essential oil to the skin. Colloids Surf. B Biointerfaces 2015, 132, 185–193. [Google Scholar] [CrossRef]
- Flores, F.C.; de Lima, J.A.; da Silva, C.R.; Benvegnú, D.; Ferreira, J.; Burger, M.E.; Beck, R.C.R.; Rolim, C.M.B.; Rocha, M.I.U.M.; da Veiga, M.L.; et al. Hydrogels Containing Nanocapsules and Nanoemulsions of Tea Tree Oil Provide Antiedematogenic Effect and Improved Skin Wound Healing. J. Nanosci. Nanotechnol. 2015, 15, 800–809. [Google Scholar] [CrossRef]
- Sugumar, S.; Ghosh, V.; Nirmala, M.J.; Mukherjee, A.; Chandrasekaran, N. Ultrasonic emulsification of eucalyptus oil nanoemulsion: Antibacterial activity against Staphylococcus aureus and wound healing activity in Wistar rats. Ultrason. Sonochem. 2014, 21, 1044–1049. [Google Scholar] [CrossRef] [PubMed]
- Tonglairoum, P.; Chuchote, T.; Ngawhirunpat, T.; Rojanarata, T.; Opanasopit, P. Encapsulation of plai oil/2-hydroxypropyl-β-cyclodextrin inclusion complexes in polyvinylpyrrolidone (PVP) electrospun nanofibers for topical application. Pharm. Dev. Technol. 2014, 19, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Danielli, L.J.; Dos Reis, M.; Bianchini, M.; Camargo, G.S.; Bordignon, S.A.; Guerreiro, I.K.; Fuentefria, A.; Apel, M.A. Antidermatophytic activity of volatile oil and nanoemulsion of Stenachaenium megapotamicum (Spreng.) Baker. Ind. Crop. Prod. 2013, 50, 23–28. [Google Scholar] [CrossRef]
- Ortan, A.; Ferdes, M.; Rodino, S.; Pirvu, C.D.; Draganescu, D. Liposomally encapsulated volatile oil of Anethum graveolens. Farmacia 2013, 61, 361–370. [Google Scholar]
- Flores, F.C.; Lima, J.A.; Ribeiro, R.F.; Alves, S.H.; Rolim, C.M.B.; Beck, R.C.R.; Silva, C.B.; De Lima, J.A.; Da Silva, C.B. Antifungal Activity of Nanocapsule Suspensions Containing Tea Tree Oil on the Growth of Trichophyton rubrum. Mycopathologia 2013, 175, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.D.O.; Colombo, M.; Kelmann, R.G.; De Souza, T.P.; Bassani, V.L.; Teixeira, H.F.; Veiga, V.F., Jr.; Limberger, R.P.; Koester, L.S. Optimization of headspace solid-phase microextraction for analysis of β-caryophyllene in a nanoemulsion dosage form prepared with copaiba (Copaifera multijuga Hayne) oil. Anal. Chim. 2012, 721, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.S.; Alam, M.S.; Imam, F.I.; Siddiqui, M.R. Topical nanoemulsion of turmeric oil for psoriasis: Characterization, ex vivo and in vivo assessment. Int. J. Drug Deliv. 2012, 4. [Google Scholar] [CrossRef]
- Sakulku, U.; Nuchuchua, O.; Uawongyart, N.; Puttipipatkhachorn, S.; Soottitantawat, A.; Ruktanonchai, U. Characterization and mosquito repellent activity of citronella oil nanoemulsion. Int. J. Pharm. 2009, 372, 105–111. [Google Scholar] [CrossRef]
- Lai, F.; Sinico, C.; De Logu, A.; Zaru, M.; Müller, R.H.; Fadda, A.M. SLN as a topical delivery system for Artemisia arborescens essential oil: In vitro antiviral activity and skin permeation study. Int. J. Nanomed. 2007, 2, 419–425. [Google Scholar]
- Suslina, S.N.; Alekseev, K.V.; Starodubova, M.V.; Bagirova, V.L. Drug synthesis methods and manufacturing technology: Development and comparative evaluation of a new liposomal dermatological preparation based on Viton oil. Pharm. Chem. J. 2004, 38, 492–497. [Google Scholar] [CrossRef]
- Heather, A.E.B. Transdermal Drug Delivery: Penetration Enhancement Techniques. Curr. Drug Deliv. 2005, 2, 23–33. [Google Scholar]
- Schnitzler, P.; Suschke, U.; Reichling, J.; Saller, R. Essential Oils of Aromatic Plants with Antibacterial, Antifungal, Antiviral, and Cytotoxic Properties—An Overview. Complement. Med. Res. 2009, 16, 79–90. [Google Scholar]
- De Matos, S.P.; Lucca, L.G.; Koester, L.S. Essential oils in nanostructured systems: Challenges in preparation and analytical methods. Talanta 2019, 195, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, M.; Wang, H.; Du, S. Increased cutaneous wound healing effect of biodegradable liposomes containing madecassoside: Preparation optimization, in vitro dermal permeation, and in vivo bioevaluation. Int. J. Nanomed. 2016, 11, 2995–3007. [Google Scholar] [CrossRef] [PubMed]
- Lohani, A.; Verma, A. Vesicles: Potential nano carriers for the delivery of skin cosmetics. J. Cosmet. Ther. 2017, 19, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Conte, R.; Marturano, V.; Peluso, G.; Calarco, A.; Cerruti, P.; Andrade, P.; Valentão, P. Recent Advances in Nanoparticle-Mediated Delivery of Anti-Inflammatory Phytocompounds. Int. J. Mol. Sci. 2017, 18, 709. [Google Scholar] [CrossRef]
- Cal, K.; Centkowska, K. Use of cyclodextrins in topical formulations: Practical aspects. Eur. J. Pharm. Biopharm. 2008, 68, 467–478. [Google Scholar] [CrossRef]
- Wadhwa, G.; Kumar, S.; Chhabra, L.; Mahant, S.; Rao, R. Essential oil–cyclodextrin complexes: An updated review. J. Incl. Phenom. Macrocycl. Chem. 2017, 89, 39–58. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Mura, S.; Manconi, M.; Fadda, A.M.; Sala, M.C.; Perricci, J.; Pini, E.; Sinico, C. Penetration enhancer-containing vesicles (PEVs) as carriers for cutaneous delivery of minoxidil: In vitro evaluation of drug permeation by infrared spectroscopy. Pharm. Dev. Technol. 2013, 18, 1339–1345. [Google Scholar] [CrossRef]
- Fox, L.T.; Gerber, M.; Du Plessis, J.; Hamman, J.H. Transdermal Drug Delivery Enhancement by Compounds of Natural Origin. Molecules 2011, 16, 10507–10540. [Google Scholar] [CrossRef]
- Sinico, C.; Fadda, A.M. Vesicular carriers for dermal drug delivery. Opin. Drug Deliv. 2009, 6, 813–825. [Google Scholar] [CrossRef] [PubMed]
- Lalko, J.; Api, A. Investigation of the dermal sensitization potential of various essential oils in the local lymph node assay. Chem. Toxicol. 2006, 44, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Aqil, M.; Ahad, A.; Sultana, Y.; Ali, A.; Aqil, M. Status of terpenes as skin penetration enhancers. Drug Discov. 2007, 12, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Koul, O. Essential Oils as Green Pesticides: Potential and Constraints. Biopestic. Int. 2008, 4, 63–84. [Google Scholar]
- Tanzi, C.D.; Vian, M.A.; Giniès, C.; Elmaataoui, M.; Chemat, F. Terpenes as Green Solvents for Extraction of Oil from Microalgae. Molecules 2012, 17, 8196–8205. [Google Scholar] [CrossRef]
- Marturano, V.; Bizzarro, V.; De Luise, A.; Calarco, A.; Ambrogi, V.; Giamberini, M.; Tylkowski, B.; Cerruti, P. Essential oils as solvents and core materials for the preparation of photo-responsive polymer nanocapsules. Nano Res. 2018, 11, 2783–2795. [Google Scholar] [CrossRef]
- Mashwani, Z.-U.-R.; Khan, M.A.; Khan, T.; Nadhman, A. Applications of plant terpenoids in the synthesis of colloidal silver nanoparticles. Adv. Sci. 2016, 234, 132–141. [Google Scholar] [CrossRef]
- Périno-Issartier, S.; Giniès, C.; Cravotto, G.; Chemat, F. A comparison of essential oils obtained from lavandin via different extraction processes: Ultrasound, microwave, turbohydrodistillation, steam and hydrodistillation. J. Chromatogr. A 2013, 1305, 41–47. [Google Scholar] [CrossRef]
- Anastas, P.T.; Kirchhoff, M.M. Origins, Current Status, and Future Challenges of Green Chemistry. Acc. Chem. Res. 2002, 35, 686–694. [Google Scholar] [CrossRef]
- Tucker, J.L. Green Chemistry, a Pharmaceutical Perspective. Org. Process. Res. Dev. 2006, 10, 315–319. [Google Scholar] [CrossRef]
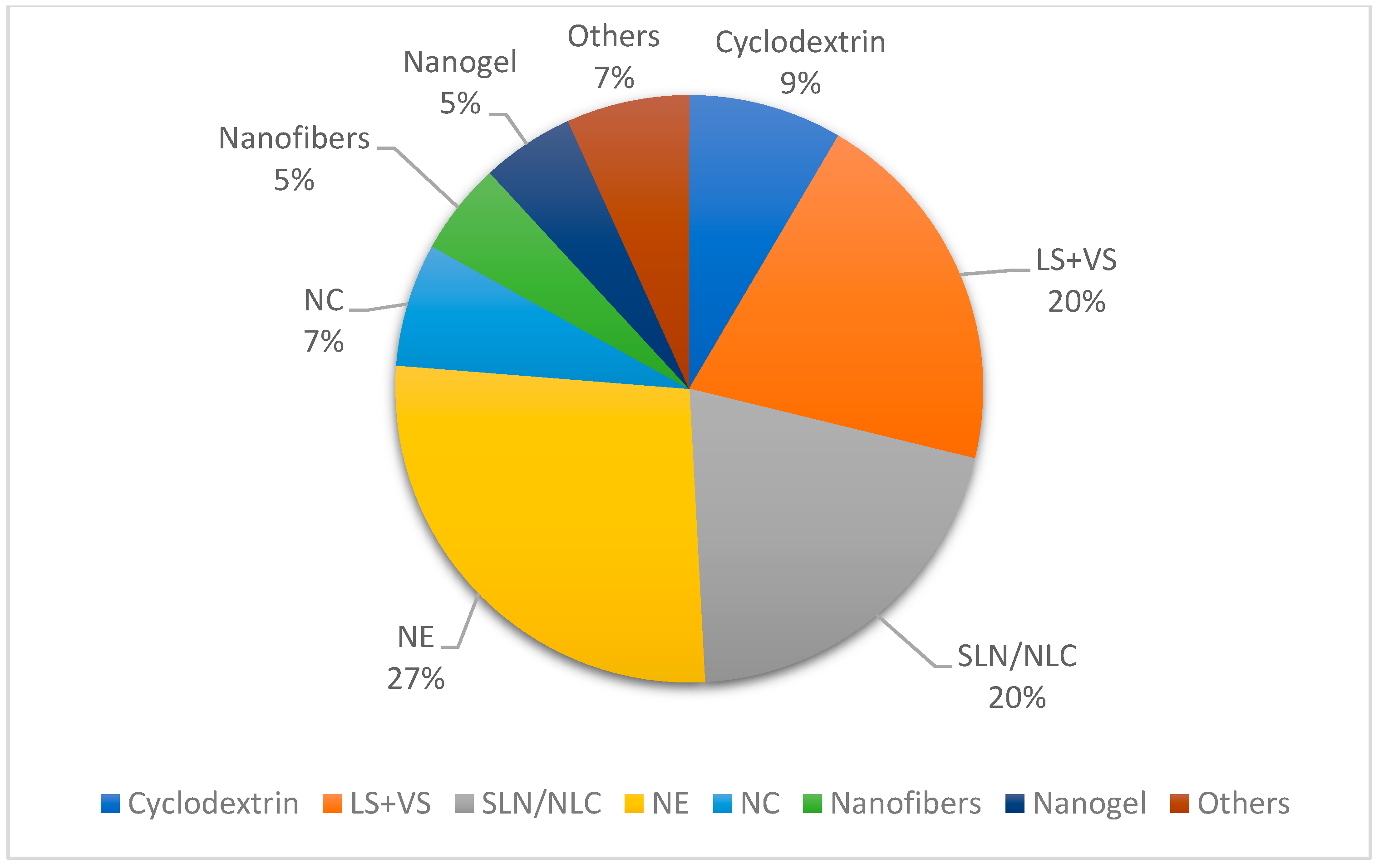
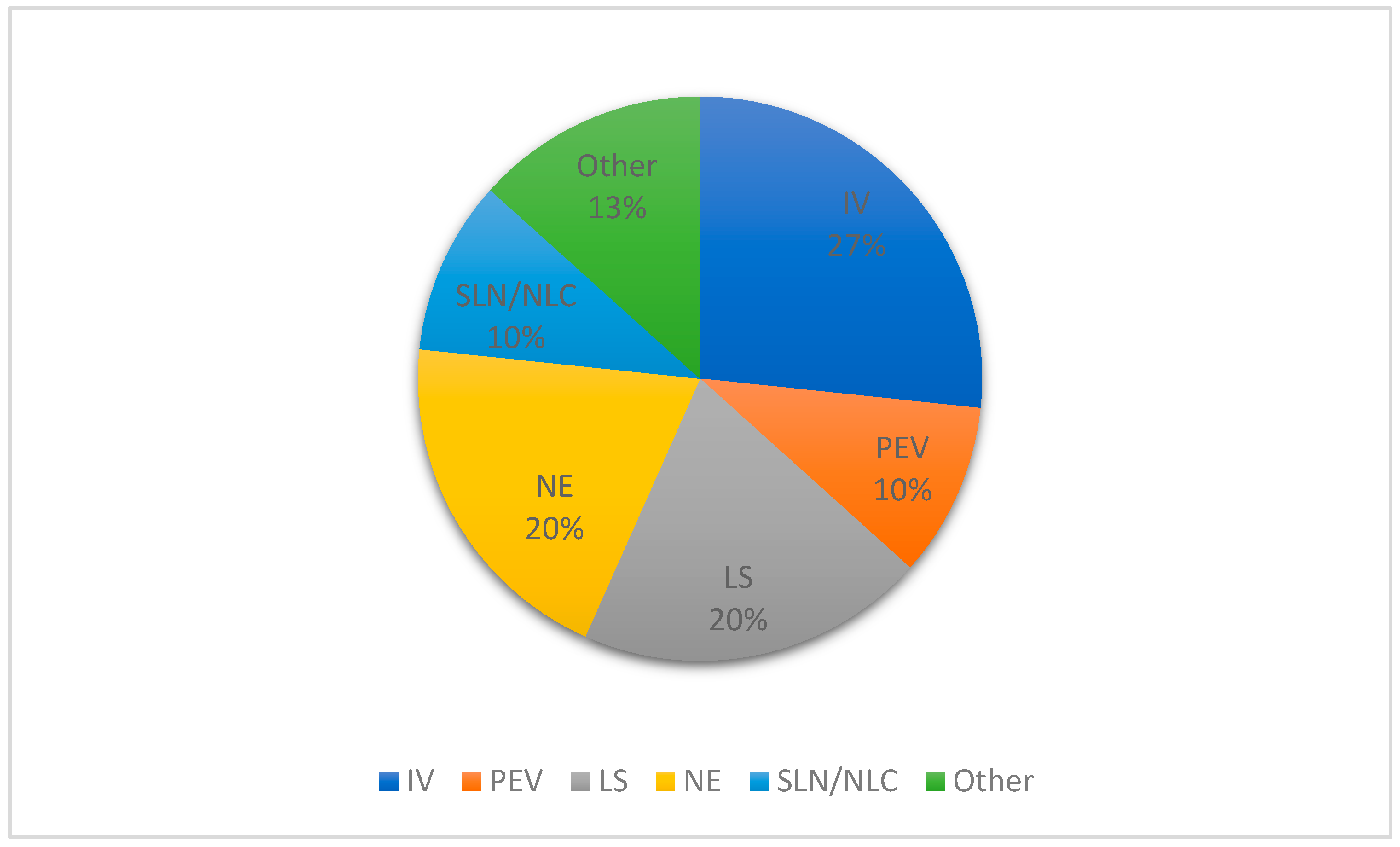
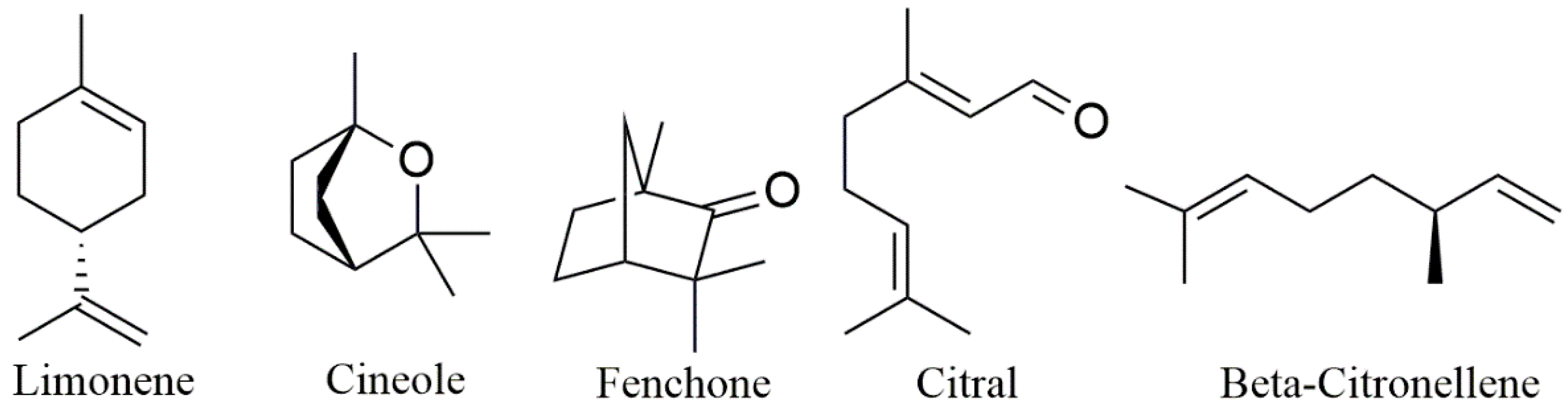
| Year | Terpene | System | Administration Route | Biological Activity | Terpene Role in the Nanosystem | |
|---|---|---|---|---|---|---|
| [25] | 2018 | Limonene, cineole, fenchone, and citral | Invasomes | Cutaneous | Anti-acne | Excipient |
| [26] | 2018 | Ursolic acid | Solid lipid nanoparticles | Cutaneous | Antiarthritic | Bioactive |
| [27] | 2018 | Limonene | Nanovesicles | Transdermal | Antineoplastic | Excipient |
| [28] | 2018 | Paeoniflorin | Ethosomes | Transdermal | Antiarthritic | Bioactive |
| [29] | 2018 | Rebaudioside A | Ultra-small micelles | Ocular | NA | Excipient |
| [30] | 2018 | Farnesol | Nanoparticles | Oral | Antibiofilm | Bioactive |
| [31] | 2018 | Eucaliptol | Nanoemulsion | Transfollicular | NA | Excipient |
| [32] | 2018 | Menthol | Nanoparticles | Transdermal | Osteoporosis treatment | Excipient |
| [33] | 2018 | Tripterine | Phytosomes | Oral | Antineoplastic | Bioactive |
| [34] | 2018 | Ursolic acid and anethole | Liposomes | Nasal | Antineoplastic | Bioactive and excipient |
| [10] | 2018 | Thymol | Solid lipid nanoparticles | Cutaneous | Anti-inflammatory | Bioactive |
| [35] | 2018 | Docetaxel | Polymeric nanoparticles | Nasal | Antineoplastic | Bioactive |
| [36] | 2018 | Forskolin | Nanostructures lipid carriers | Transdermal | Photoprotector | Bioactive |
| [37] | 2017 | Citral and limonene | Transferosomes and liposomes | Transdermal | Antiarthritic | Excipient |
| [38] | 2017 | Paeoniflorin | Glycerosomes | Transdermal | Anti-inflammatory | Bioactive and Excipient |
| [39] | 2017 | Limonene | Transinvasomes | Transdermal | NA | Excipient |
| [40] | 2017 | Limonene | PEGylated liposomes | Transdermal | Alzheimer’s treatment | Excipient |
| [41] | 2017 | Triptolide | Nanoemulsion | Percutaneous | Anti-inflammatory and analgesic | Bioactive |
| [42] | 2017 | β-citronellene | Invasomes | Transdermal | Hypertension treatment | Excipient |
| [43] | 2017 | α-bisabolol | Nanocapsules | Ocular | Antinociceptive | Bioactive |
| [11] | 2017 | Hyperforin | Hydroxypropyl-β-cyclodextrin | Cutaneous | Wound healing | Bioactive |
| [44] | 2016 | Cineole and limonene | Penetration enhancer vehicle | Transdermal | Antifungal | Excipient |
| [45] | 2016 | β-citronellene | Invasomes | Transdermal | Hypertension treatment | Excipient |
| [46] | 2016 | Squalene | Solid nanoemulsion | Transdermal | Immunization | Excipient |
| [14] | 2016 | Paclitaxel | Solid Lipid Nanoparticles | Cutaneous | Antineoplastic | Bioactive |
| [47] | 2016 | Madecassoside | Liposomes | Cutaneous | Wound healing | Bioactive |
| [48] | 2016 | Asiaticoside | Nanofibers | Cutaneous | Wound healing | Bioactive |
| [49] | 2016 | Triterpenoids of Ganoderma l. | Nanogel | Cutaneous | Frostbite treatment | Bioactive |
| [50] | 2016 | Safranal | Nanoemulsion | Nasal | Cerebral ischemia treatment | Bioactive |
| [51] | 2015 | Ursolic acid | Niosomal gel | Transdermal | Antiarthritic | Bioactive |
| [52] | 2015 | Farnesol | Polymeric nanoparticles | Oral | Antibiofilm | Bioactive |
| [53] | 2015 | Ursolic acid and oleanoic acid | Nanoemulsion | Cutaneous | Anti-inflammatory | Bioactive |
| [13] | 2015 | Docetaxel | Nanofibers | Cutaneous | Antineoplastic | Bioactive |
| [54] | 2014 | Limonene | Nanoemulsion | Transdermal | NA | Excipient |
| [55] | 2014 | Limoneno | PEGilated liposomes | Transdermal | NA | Excipient |
| [56] | 2014 | Limonene and 1,8-cineole | Nanoemulsion and solid lipid nanoparticles and nanostructures lipid carriers | Cutaneous | Cutaneous lesions treatment | Excipient |
| [12] | 2014 | Paclitaxel | Solid lipid nanoparticles and Nanostructures lipid carriers | Cutaneous | Hyperkeratosis treatment | Bioactive |
| [57] | 2013 | Betulin | Nanoemulsion | Cutaneous | Antineoplastic | Bioactive |
| [58] | 2013 | Limonene | Nanoemulsion | Transdermal | Analgesic | Excipient |
| [59] | 2013 | Astragaloside IV | Solid lipid nanoparticles | Cutaneous | Wound healing | Bioactive |
| [60] | 2013 | Limonene | Liposomes | Transdermal | NA | Excipient |
| [61] | 2013 | Lupane | Liposomes | Cutaneous | Leishmanicidal | Bioactive |
| [62] | 2013 | Hurpezine A | Solid lipid nanoparticles, Nanostructures lipid carriers and Microemulsion | Transdermal | Alzheimer’s treatment | Bioactive |
| [63] | 2012 | Tripterine | Nanostructures lipid carriers | Cutaneous | Antineoplastic | Bioactive |
| [64] | 2012 | Asiaticoside | Liposomes | Transdermal | Stimulation of collagen synthesis | Bioactive |
| [65] | 2010 | Triptolide | Ethosomes | Transdermal | Anti-inflammatory | Bioactive |
| [66] | 2010 | Squalene | Nanostructures lipid carriers | Cutaneous | Psoriasis treatment | Excipient |
| [67] | 2009 | Cineole | Penetration enhancer vehicle | Transdermal | Alopecia treatment | Excipient |
| [68,69,70] | 2009, 2008 | Limonene, citral and cineole | Invasomes | Transdermal | Photosensitization | Excipient |
| [71] | 2005 | Triptolide | Solid lipid nanoparticles | Cutaneous | Anti-inflammatory | Bioactive |
| [72] | 2003 | Sericoside | derivates of β- and γ-cyclodextrins | Cutaneous | Anti-inflammatory | Bioactive |
| [73] | 2003 | Triptolide | Solid lipid nanoparticles | Cutaneous | Anti-inflammatory | Bioactive |
| [24] | 1989 | Forskolin | β- and γ-cyclodextrins | Ocular | Treatment of intraocular hypertension | Bioactive |
| Year | Essential Oil/Fixed Oil/Plant Extract | System | Administration Route | Biological Activity | Essential Oil/Fixed Oil/Plant Extract Role in the System | |
|---|---|---|---|---|---|---|
| [74] | 2018 | Lemon EO | Nanoemulsion | Cutaneous | Hyperpigmentation treatment | Excipient |
| [75] | 2018 | Clove EO and sweet fennel EO | Nanoemulsion | Cutaneous | Autoimmune dermatoses | Excipient |
| [76] | 2018 | Clove EO and sweet fennel EO | Nanoemulsion | Cutaneous | Autoimmune dermatoses | Excipient |
| [77] | 2018 | Mentha EO | Nanogel | Vaginal | Antifungal | Bioactive |
| [78] | 2018 | Nigella Sativa EO | Nanoemulsions | Cutaneous | Anti-inflammatory | Bioactive |
| [79] | 2017 | Clove EO and Sweet Fennel EO | Nanoemulsion | Transdermal | Autoimmune dermatoses | Excipient |
| [80] | 2017 | Cymbopogon Flexuous EO | Nanocapsules | Cutaneous | Antimicrobial | Bioactive |
| [81] | 2017 | Rosemary EO | Lipid Nanoparticles | Cutaneous | Skin hydration | Bioactive |
| [82] | 2017 | Eucaliptus globulosus EO | Nanosized-microemulsion | Cutaneous | Repellent | Bioactive |
| [83] | 2017 | Syzygium aromaticum EO | Nanoemulsion | Cutaneous | Wound healing and antidermatophytic | Bioactive |
| [84] | 2016 | Tea Tree Oil | Nanoemulsion | Cutaneous | Antimicrobial | Bioactive |
| [85] | 2016 | Centella asiatica extract | Hydroxypropyl-β-cyclodextrin | Cutaneous | Wound healing | Bioactive |
| [86] | 2016 | Lippia sidoides EO | Nanogel | Oral | Periodontitis treatment | Bioactive |
| [87] | 2015 | Foeniculum vulgare EO | Nanoemulsion | Transdermal | Antidiabetic | Bioactive |
| [88] | 2015 | Lemongrass oil | Nanosponges | Cutaneous | Antifungal | Bioactive |
| [89] | 2015 | Copaifera multijuga EO | Nanoemulsion | Percutaneous | Anti-inflammatory | Bioactive |
| [90] | 2015 | Santolina insularis EO | Penetration enhancing vehicle | Percutaneous | Antimicrobial | Bioactive and excipient |
| [91] | 2015 | Melaleuca alternifolia EO | Nanoemulsion and Nanocapsules | Cutaneous | Wound healing and antidermatotophytic | Bioactive |
| [92] | 2014 | Eucalyptus oil | Nanoemulsion | Cutaneous | Antibacterial and Wound healing | Bioactive |
| [93] | 2014 | Plai oil | Hydroxypropyl-β-cyclodextrin and Nanofibers | Cutaneous | Anti-inflammatory | Bioactive |
| [94] | 2013 | Stenachaenium megapotamicum EO | Nanoemulsion | Cutaneous | Antidermatophytic | Bioactive |
| [95] | 2013 | Anethum graveolens EO | Liposomes | Cutaneous | Antifungal | Bioactive |
| [96] | 2013 | M. alternifolia EO | Nanoemulsion and nanocapsules | Ungueal | Onychomycosis treatment | Bioactive |
| [97] | 2012 | C. multijuga EO | Nanoemulsion | Percutaneous | Anti-inflammatory | Bioactive |
| [98] | 2012 | Turmeric oil | Nanoemulsion | Cutaneous | Psoriasis treatment | Bioactive |
| [99] | 2009 | Citronella oil | Nanoemulsion | Cutaneous | Repellent | Bioactive |
| [100] | 2007 | Artemisia arborescens EO | Solid Lipid Nanoparticles | Cutaneous | Antiviral | Bioactive |
| [101] | 2004 | Viton oil | Liposomes | Cutaneous | Anti-inflammatory | Bioactive |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Matos, S.P.; Teixeira, H.F.; de Lima, Á.A.N.; Veiga-Junior, V.F.; Koester, L.S. Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review. Biomolecules 2019, 9, 138. https://doi.org/10.3390/biom9040138
de Matos SP, Teixeira HF, de Lima ÁAN, Veiga-Junior VF, Koester LS. Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review. Biomolecules. 2019; 9(4):138. https://doi.org/10.3390/biom9040138
Chicago/Turabian Stylede Matos, Sheila P., Helder F. Teixeira, Ádley A. N. de Lima, Valdir F. Veiga-Junior, and Letícia S. Koester. 2019. "Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review" Biomolecules 9, no. 4: 138. https://doi.org/10.3390/biom9040138
APA Stylede Matos, S. P., Teixeira, H. F., de Lima, Á. A. N., Veiga-Junior, V. F., & Koester, L. S. (2019). Essential Oils and Isolated Terpenes in Nanosystems Designed for Topical Administration: A Review. Biomolecules, 9(4), 138. https://doi.org/10.3390/biom9040138








