The Impact of Glycaemic Variability on Vascular Dysfunction in Diabetes
Abstract
1. Introduction
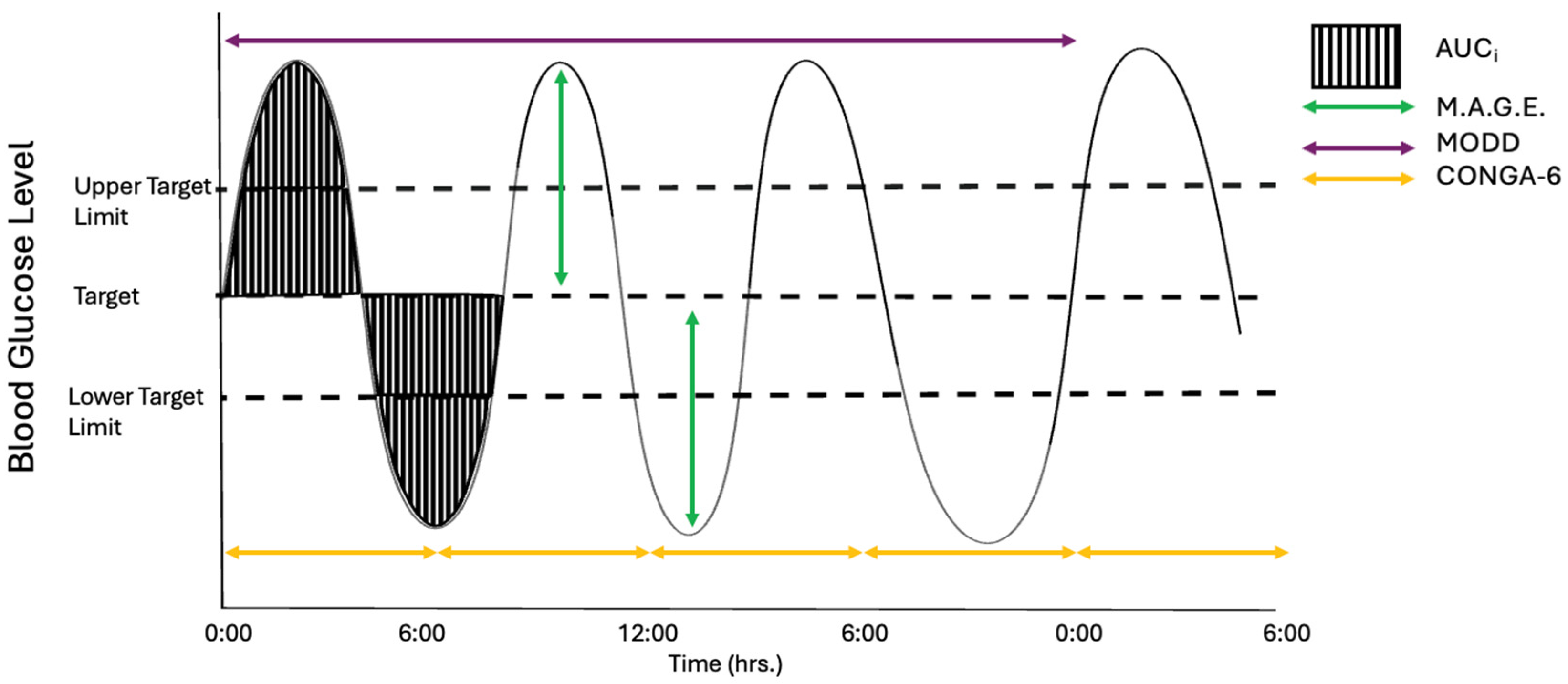
2. Glycaemic Variation
3. Models of Investigation
3.1. Animal Models
3.2. Human Models
3.3. Cellular Models
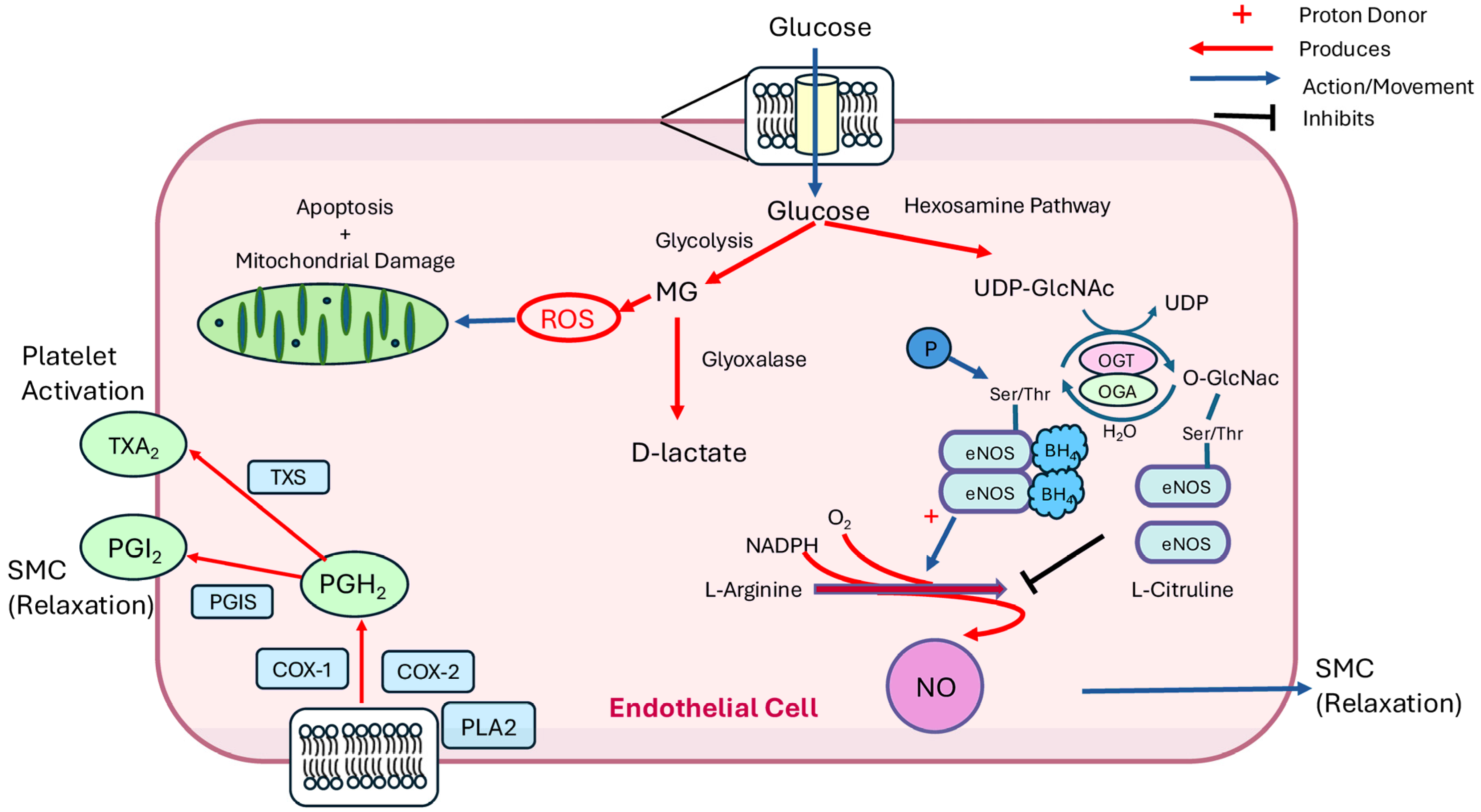
4. Potential Mechanisms of Vascular Dysfunction
4.1. Mitochondria and Apoptosis
4.2. Oxidative Stress and Reactive Oxygen Species (ROS)
4.3. Arginase and Nitric Oxide
4.4. O-GlcNAcylation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 8-ISOPGF2α | 8-Isoprostaglandin F2α |
| AGE | Advanced glycation end product |
| AICAR | 5-Aminoimidazole-4-carboxamide ribonucleotide |
| AKT | Protein kinase B |
| AMP | Adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| ATP | Adenosine triphosphate |
| AUCi | Incremental area under the curve |
| BAECs | Bovine aortic endothelial cells |
| BAX | Bcl-2-associated protein |
| Bcl-2 | B-cell Lymphoma 2 |
| CGM | Continuous glucose monitor |
| CONGA-n | Continuous overlapping net glycaemic action at n-hours |
| CSGM | Subcutaneous CGM |
| CV% | Coefficient of variance |
| CVD | Cardiovascular disease |
| DM | Diabetes mellitus |
| DNA | Deoxyribonucleic acid |
| EC | Endothelial cell |
| ED | Endothelial dysfunction |
| EDV | endothelium-dependent vasodilatation |
| EIDV | endothelium-independent vasodilatation |
| eNOS | Endothelial nitric oxide synthase |
| FMD | Flow-mediated dilatation |
| GLP1 | Glucagon-like peptide-1 |
| GV | Glycaemic variation |
| HbA1c | Glycosylated haemoglobin A1 |
| HUVEC | Human umbilical vein endothelial cell |
| L-NMMA | NG-Monomethyl-L-arginine |
| MAGE | Mean amplitude of glycaemic excursion |
| MCH | Methacholine chloride |
| MDA | Malondialdehyde |
| MG | Methylglyoxyl |
| MI | Myocardial infarction |
| MODD | Mean of daily differences |
| NAD | Nicotinamide adenine dinucleotide |
| NADPH | NAD phosphate |
| NO | Nitric oxide |
| NOS | Nitric oxide synthase |
| O-GlcNAc | O-linked β-N-acetylglucosamine |
| PAD | Peripheral arterial disease |
| PKC | Protein kinase C |
| PTM | Post-translation modification |
| RAGEs | AGE receptors |
| RBC | Red blood cell |
| REDOX | Reduction–oxidation |
| ROS | Reactive oxygen species |
| SGLT-2 | Sodium–glucose cotransporter-2 |
| SD | Standard deviation |
| SMC | Smooth muscle cell |
| SNP | Sodium nitroprusside |
| T1DM | Type 1 diabetes mellitus |
| T2DM | Type 2 diabetes mellitus |
| UDP-GlcNAc | Uridine-diphosphate-GlcNAc |
References
- DiabetesUK. How Many People in The UK have Diabetes? Available online: https://www.diabetes.org.uk/about-us/about-the-charity/our-strategy/statistics (accessed on 29 July 2024).
- Whicher, C.A.; O’Neill, S.; Holt, R.I.G. Diabetes in the UK: 2019. Diabet. Med. 2020, 37, 242–247. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Swerdlow, M.A.; Armstrong, A.A.; Conte, M.S.; Padula, W.V.; Bus, S.A. Five year mortality and direct costs of care for people with diabetic foot complications are comparable to cancer. J. Foot Ankle Res. 2020, 13, 16. [Google Scholar] [CrossRef] [PubMed]
- Soyoye, D.O.; Abiodun, O.O.; Ikem, R.T.; Kolawole, B.A.; Akintomide, A.O. Diabetes and peripheral artery disease: A review. World J. Diabetes 2021, 12, 827–838. [Google Scholar] [CrossRef]
- Wukich, D.K.; Raspovic, K.M.; Suder, N.C. Patients With Diabetic Foot Disease Fear Major Lower-Extremity Amputation More Than Death. Foot Ankle Spec. 2018, 11, 17–21. [Google Scholar] [CrossRef]
- Marx, N.; Federici, M.; Schutt, K.; Muller-Wieland, D.; Ajjan, R.A.; Antunes, M.J.; Christodorescu, R.M.; Crawford, C.; Di Angelantonio, E.; Eliasson, B.; et al. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes. Eur. Heart J. 2023, 44, 4043–4140. [Google Scholar] [CrossRef]
- Boussageon, R.; Bejan-Angoulvant, T.; Saadatian-Elahi, M.; Lafont, S.; Bergeonneau, C.; Kassai, B.; Erpeldinger, S.; Wright, J.M.; Gueyffier, F.; Cornu, C. Effect of intensive glucose lowering treatment on all cause mortality, cardiovascular death, and microvascular events in type 2 diabetes: Meta-analysis of randomised controlled trials. BMJ 2011, 343, d4169. [Google Scholar] [CrossRef]
- Giorgino, F.; Leonardini, A.; Laviola, L. Cardiovascular disease and glycemic control in type 2 diabetes: Now that the dust is settling from large clinical trials. Ann. N. Y. Acad. Sci. 2013, 1281, 36–50. [Google Scholar] [CrossRef]
- UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998, 352, 837–853. [Google Scholar] [CrossRef]
- Marx, N.; Husain, M.; Lehrke, M.; Verma, S.; Sattar, N. GLP-1 Receptor Agonists for the Reduction of Atherosclerotic Cardiovascular Risk in Patients With Type 2 Diabetes. Circulation 2022, 146, 1882–1894. [Google Scholar] [CrossRef]
- Cosentino, F.; Hishikawa, K.; Katusic, Z.S.; Luscher, T.F. High glucose increases nitric oxide synthase expression and superoxide anion generation in human aortic endothelial cells. Circulation 1997, 96, 25–28. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Arima, H.; Zoungas, S.; Ninomiya, T.; Cooper, M.; Hamet, P.; Mancia, G.; Poulter, N.; Harrap, S.; Woodward, M.; et al. Impact of visit-to-visit glycemic variability on the risks of macrovascular and microvascular events and all-cause mortality in type 2 diabetes: The ADVANCE trial. Diabetes Care 2014, 37, 2359–2365. [Google Scholar] [CrossRef]
- Caprnda, M.; Mesarosova, D.; Ortega, P.F.; Krahulec, B.; Egom, E.; Rodrigo, L.; Kruzliak, P.; Mozos, I.; Gaspar, L. Glycemic Variability and Vascular Complications in Patients with Type 2 Diabetes Mellitus. Folia Med. 2017, 59, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Shen, Y.; Liu, Y.H.; Dai, Y.; Wu, Z.M.; Wang, X.Q.; Yang, C.D.; Li, L.Y.; Liu, J.M.; Zhang, L.P.; et al. Impact of glycemic control on the association of endothelial dysfunction and coronary artery disease in patients with type 2 diabetes mellitus. Cardiovasc. Diabetol. 2021, 20, 64. [Google Scholar] [CrossRef] [PubMed]
- Akasaka, T.; Sueta, D.; Tabata, N.; Takashio, S.; Yamamoto, E.; Izumiya, Y.; Tsujita, K.; Kojima, S.; Kaikita, K.; Matsui, K.; et al. Effects of the Mean Amplitude of Glycemic Excursions and Vascular Endothelial Dysfunction on Cardiovascular Events in Nondiabetic Patients With Coronary Artery Disease. J. Am. Heart Assoc. 2017, 6, e004841. [Google Scholar] [CrossRef] [PubMed]
- Seufert, J. Addressing glycaemic variation. Br. J. Diabetes Vasc. Dis. 2011, 11, 2–5. [Google Scholar] [CrossRef]
- Dandona, P.; Aljada, A.; Chaudhuri, A.; Mohanty, P. Endothelial dysfunction, inflammation and diabetes. Rev. Endocr. Metab. Disord. 2004, 5, 189–197. [Google Scholar] [CrossRef]
- Klimontov, V.V.; Saik, O.V.; Korbut, A.I. Glucose Variability: How Does It Work? Int. J. Mol. Sci. 2021, 22, 7783. [Google Scholar] [CrossRef]
- Ciechanowska, A.; Gora, I.M.; Sabalinska, S.; Ladyzynski, P. The Effect of High and Variable Glucose on the Viability of Endothelial Cells Co-Cultured with Smooth Muscle Cells. Int. J. Mol. Sci. 2022, 23, 6704. [Google Scholar] [CrossRef]
- Costantino, S.; Paneni, F.; Battista, R.; Castello, L.; Capretti, G.; Chiandotto, S.; Tanese, L.; Russo, G.; Pitocco, D.; Lanza, G.A.; et al. Impact of Glycemic Variability on Chromatin Remodeling, Oxidative Stress, and Endothelial Dysfunction in Patients With Type 2 Diabetes and With Target HbA(1c) Levels. Diabetes 2017, 66, 2472–2482. [Google Scholar] [CrossRef]
- Harrington, J.; Pena, A.S.; Wilson, L.; Gent, R.; Dowling, K.; Baghurst, P.; Couper, J. Vascular function and glucose variability improve transiently following initiation of continuous subcutaneous insulin infusion in children with type 1 diabetes. Pediatr. Diabetes 2013, 14, 504–511. [Google Scholar] [CrossRef]
- Hoffman, R.P.; Dye, A.S.; Huang, H.; Bauer, J.A. Effects of glucose control and variability on endothelial function and repair in adolescents with type 1 diabetes. ISRN Endocrinol. 2013, 2013, 876547. [Google Scholar] [CrossRef]
- Maeda, M.; Hayashi, T.; Mizuno, N.; Hattori, Y.; Kuzuya, M. Intermittent high glucose implements stress-induced senescence in human vascular endothelial cells: Role of superoxide production by NADPH oxidase. PLoS ONE 2015, 10, e0123169. [Google Scholar] [CrossRef]
- Quagliaro, L.; Piconi, L.; Assaloni, R.; Martinelli, L.; Motz, E.; Ceriello, A. Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells: The role of protein kinase C and NAD(P)H-oxidase activation. Diabetes 2003, 52, 2795–2804. [Google Scholar] [CrossRef]
- Suh, S.; Kim, J.H. Glycemic Variability: How Do We Measure It and Why Is It Important? Diabetes Metab. J. 2015, 39, 273–282. [Google Scholar] [CrossRef]
- Otowa-Suematsu, N.; Sakaguchi, K.; Komada, H.; Nakamura, T.; Sou, A.; Hirota, Y.; Kuroda, M.; Shinke, T.; Hirata, K.I.; Ogawa, W. Comparison of the relationship between multiple parameters of glycemic variability and coronary plaque vulnerability assessed by virtual histology-intravascular ultrasound. J. Diabetes Investig. 2017, 9, 610–615. [Google Scholar] [CrossRef]
- Rodbard, D. New and improved methods to characterize glycemic variability using continuous glucose monitoring. Diabetes Technol. Ther. 2009, 11, 551–565. [Google Scholar] [CrossRef]
- Faerch, K.; Alssema, M.; Mela, D.J.; Borg, R.; Vistisen, D. Relative contributions of preprandial and postprandial glucose exposures, glycemic variability, and non-glycemic factors to HbA (1c) in individuals with and without diabetes. Nutr. Diabetes 2018, 8, 38. [Google Scholar] [CrossRef]
- Verges, B.; Pignol, E.; Rouland, A.; Bouillet, B.; Baillot-Rudoni, S.; Quilot, E.; Djeffal, A.; Petit, J.M. Glycemic Variability Assessment with a 14-Day Continuous Glucose Monitoring System: When and How Long to Measure MAGE (Mean Amplitude of Glucose Excursion) for Optimal Reliability? J. Diabetes Sci. Technol. 2022, 16, 982–987. [Google Scholar] [CrossRef]
- Di Flaviani, A.; Picconi, F.; Di Stefano, P.; Giordani, I.; Malandrucco, I.; Maggio, P.; Palazzo, P.; Sgreccia, F.; Peraldo, C.; Farina, F.; et al. Impact of glycemic and blood pressure variability on surrogate measures of cardiovascular outcomes in type 2 diabetic patients. Diabetes Care 2011, 34, 1605–1609. [Google Scholar] [CrossRef]
- Buscemi, S.; Verga, S.; Cottone, S.; Azzolina, V.; Buscemi, B.; Gioia, D.; Cerasola, G. Glycaemic variability and inflammation in subjects with metabolic syndrome. Acta Diabetol. 2009, 46, 55–61. [Google Scholar] [CrossRef]
- Derr, R.; Garrett, E.; Stacy, G.A.; Saudek, C.D. Is HbA(1c) affected by glycemic instability? Diabetes Care 2003, 26, 2728–2733. [Google Scholar] [CrossRef]
- Sherwani, S.I.; Khan, H.A.; Ekhzaimy, A.; Masood, A.; Sakharkar, M.K. Significance of HbA1c Test in Diagnosis and Prognosis of Diabetic Patients. Biomark Insights 2016, 11, 95–104. [Google Scholar] [CrossRef]
- John, W.G.; Mosca, A.; Weykamp, C.; Goodall, I. HbA1c standardisation: History, science and politics. Clin. Biochem. Rev. 2007, 28, 163–168. [Google Scholar]
- Joshi, A.; Mitra, A.; Anjum, N.; Shrivastava, N.; Khadanga, S.; Pakhare, A.; Joshi, R. Patterns of Glycemic Variability During a Diabetes Self-Management Educational Program. Med. Sci. 2019, 7, 52. [Google Scholar] [CrossRef]
- Tsang, H.G.; Rashdan, N.A.; Whitelaw, C.B.; Corcoran, B.M.; Summers, K.M.; MacRae, V.E. Large animal models of cardiovascular disease. Cell Biochem. Funct. 2016, 34, 113–132. [Google Scholar] [CrossRef]
- Gomez-Salinero, J.M.; Redmond, D.; Rafii, S. Microenvironmental determinants of endothelial cell heterogeneity. Nat. Rev. Mol. Cell Biol. 2025, 26, 476–495. [Google Scholar] [CrossRef]
- Heather, L.C.; Hafstad, A.A.-O.; Halade, G.A.-O.; Harmancey, R.A.-O.; Mellor, K.A.-O.; Mishra, P.A.-O.; Mulvihill, E.A.-O.; Nabben, M.A.-O.; Nakamura, M.A.-O.; Rider, O.J.; et al. Guidelines on models of diabetic heart disease. Am. J. Physiol. Heart Circ. Physiol. 2022, 323, H176–H200. [Google Scholar] [CrossRef]
- Chien, S. Effects of disturbed flow on endothelial cells. Ann. Biomed. Eng. 2008, 36, 554–562. [Google Scholar] [CrossRef]
- Zaragoza, C.; Gomez-Guerrero, C.; Martin-Ventura, J.L.; Blanco-Colio, L.; Lavin, B.; Mallavia, B.; Tarin, C.; Mas, S.; Ortiz, A.; Egido, J. Animal models of cardiovascular diseases. J. Biomed. Biotechnol. 2011, 2011, 497841. [Google Scholar] [CrossRef]
- Bellinger, D.A.; Merricks, E.P.; Nichols, T.C. Swine models of type 2 diabetes mellitus: Insulin resistance, glucose tolerance, and cardiovascular complications. ILAR J. 2006, 47, 243–258. [Google Scholar] [CrossRef]
- Diemar, S.S.; Sejling, A.S.; Iversen, K.K.; Engstrom, T.; Honge, J.L.; Tonder, N.; Vejlstrup, N.; Idorn, M.; Ekstrom, K.; Pedersen-Bjergaard, U.; et al. Influence of acute glycaemic level on measures of myocardial infarction in non-diabetic pigs. Scand. Cardiovasc. J. 2015, 49, 376–382. [Google Scholar] [CrossRef]
- Ajjan, R.A. The clinical importance of measuring glycaemic variability: Utilising new metrics to optimise glycaemic control. Diabetes Obes. Metab. 2024, 26 (Suppl. 7), 3–16. [Google Scholar] [CrossRef]
- Inaishi, J.; Saisho, Y.; Watanabe, Y.; Tsuchiya, T.; Sasaki, H.; Masaoka, T.; Itoh, H. Changes in glycemic variability, gastric emptying and vascular endothelial function after switching from twice-daily to once-weekly exenatide in patients with type 2 diabetes: A subpopulation analysis of the twin-exenatide study. BMC Endocr. Disord. 2022, 22, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.G.; Zhang, Y.Q.; Zhao, D.K.; Wu, J.X.; Zhao, J.; Jiao, X.M.; Chen, B.; Lv, X.F. Relationship between blood glucose fluctuation and macrovascular endothelial dysfunction in type 2 diabetic patients with coronary heart disease. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 3593–3600. [Google Scholar] [PubMed]
- Kuricová, K.; Pácal, L.; Šoupal, J.; Prázný, M.; Kaňková, K. Effect of glucose variability on pathways associated with glucotoxicity in diabetes: Evaluation of a novel in vitro experimental approach. Diabetes Res. Clin. Pract. 2016, 114, 1–8. [Google Scholar] [CrossRef]
- Ido, Y.; Carling, D.; Ruderman, N. Hyperglycemia-induced apoptosis in human umbilical vein endothelial cells: Inhibition by the AMP-activated protein kinase activation. Diabetes 2002, 51, 159–167. [Google Scholar] [CrossRef]
- Mohajan, D.; Mohajan, H.K. Hyperosmolar Hyperglycaemic State: A Life-Threatening Complication of Type 2 Diabetes Patients. J. Innov. Med. Res. 2023, 2, 30–35. [Google Scholar] [CrossRef]
- Lemmey, H.A.L.; Ye, X.; Ding, H.C.; Triggle, C.R.; Garland, C.J.; Dora, K.A. Hyperglycaemia disrupts conducted vasodilation in the resistance vasculature of db/db mice. Vascul. Pharmacol. 2018, 103–105, 29–35. [Google Scholar] [CrossRef]
- Kitada, M.; Zhang, Z.; Mima, A.; King, G.L. Molecular mechanisms of diabetic vascular complications. J. Diabetes Investig. 2010, 1, 77–89. [Google Scholar] [CrossRef]
- Madonna, R.; Pieragostino, D.; Rossi, C.; Confalone, P.; Cicalini, I.; Minnucci, I.; Zucchelli, M.; del Boccio, P.; de Caterina, R. Simulated hyperglycemia impairs insulin signaling in endothelial cells through a hyperosmolar mechanism. Vascul. Pharmacol. 2020, 130, 106678. [Google Scholar] [CrossRef]
- He, A.; Hu, S.; Pi, Q.; Guo, Y.; Long, Y.; Luo, S.; Xia, Y. Regulation of O-GlcNAcylation on endothelial nitric oxide synthase by glucose deprivation and identification of its O-GlcNAcylation sites. Sci. Rep. 2020, 10, 19364. [Google Scholar] [CrossRef]
- Calis, P.; Vojtech, L.; Hladik, F.; Gravett, M.G. A review of ex vivo placental perfusion models: An underutilized but promising method to study maternal-fetal interactions. J. Matern. Fetal Neonatal. Med. 2022, 35, 8823–8835. [Google Scholar] [CrossRef] [PubMed]
- Kwok, J.C.; Huang, W.; Leung, W.C.; Chan, S.K.; Chan, K.Y.; Leung, K.M.; Chu, A.C.; Lam, A.K. Human placenta as an ex vivo vascular model for neurointerventional research. J. Neurointerv. Surg. 2014, 6, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Deng, J.; Hao, S.; Wang, B. A Potential In Vitro 3D Cell Model to Study Vascular Diseases by Simulating the Vascular Wall Microenvironment and Its Application. Life 2022, 12, 427. [Google Scholar] [CrossRef] [PubMed]
- Jenny, L.; Melmer, A.; Laimer, M.; Hardy, E.T.; Lam, W.A.; Schroeder, V. Diabetes affects endothelial cell function and alters fibrin clot formation in a microvascular flow model: A pilot study. Diab. Vasc. Dis. Res. 2020, 17, 1479164120903044. [Google Scholar] [CrossRef]
- Hu, S.Y.; Xue, C.D.; Li, Y.J.; Li, S.; Gao, Z.N.; Qin, K.R. Microfluidic investigation for shear-stress-mediated repair of dysglycemia-induced endothelial cell damage. Mechanobiol. Med. 2024, 2, 100069. [Google Scholar] [CrossRef]
- Hu, S.Y.; Zhu, T.T.; Yin, X.Y.; Li, S.S.; Xue, C.D.; Li, Y.J.; Hu, X.Q.; Qin, K.R. An integrated shear-glucose system to study how shear stress repairs oscillating glucose-induced endothelial cell damage via ROS/Ca2+/ eNOS signaling. Microchem. J. 2025, 215, 114415. [Google Scholar] [CrossRef]
- Austin, S.K. Haemostasis. Medicine 2017, 45, 204–208. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef]
- Huang, Y.; Yue, L.; Qiu, J.; Gao, M.; Liu, S.; Wang, J. Endothelial Dysfunction and Platelet Hyperactivation in Diabetic Complications Induced by Glycemic Variability. Horm. Metab. Res. 2022, 54, 419–428. [Google Scholar] [CrossRef]
- Dagamajalu, S.; Rex, D.A.B.; Gopalakrishnan, L.; Karthikkeyan, G.; Gurtoo, S.; Modi, P.K.; Mohanty, V.; Mujeeburahiman, M.; Soman, S.; Raju, R.; et al. A network map of endothelin mediated signaling pathway. J. Cell Commun. Signal. 2021, 15, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Kakizawa, H.; Itoh, M.; Itoh, Y.; Imamura, S.; Ishiwata, Y.; Matsumoto, T.; Yamamoto, K.; Kato, T.; Ono, Y.; Nagata, M. The relationship between glycemic control and plasma vascular endothelial growth factor and endothelin-1 concentration in diabetic patients. Metabolism 2004, 53, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, N.M.S.; Mahmud, A.M.; Maulood, I.M. Vascular actions of Ang 1-7 and Ang 1-8 through EDRFs and EDHFs in non-diabetes and diabetes mellitus. Nitric Oxide 2025, 156, 9–26. [Google Scholar] [PubMed]
- Das, S.; Chandrasekhar, S.; Yadav, J.S.; Grée, R. Recent developments in the synthesis of prostaglandins and analogues. Chem. Rev. 2007, 107, 3286–3337. [Google Scholar] [CrossRef]
- Fadini, G.P.; Albiero, M.; Menegazzo, L.; Boscaro, E.; Pagnin, E.; Iori, E.; Cosma, C.; Lapolla, A.; Pengo, V.; Stendardo, M.; et al. The redox enzyme p66Shc contributes to diabetes and ischemia-induced delay in cutaneous wound healing. Diabetes 2010, 59, 2306–2314. [Google Scholar] [CrossRef]
- Fadini, G.P.; Albiero, M.; Bonora, B.M.; Poncina, N.; Vigili de Kreutzenberg, S.; Avogaro, A. p66Shc gene expression in peripheral blood mononuclear cells and progression of diabetic complications. Cardiovasc. Diabetol. 2018, 17, 16. [Google Scholar] [CrossRef]
- Berry, A.; Cirulli, F. The p66(Shc) gene paves the way for healthspan: Evolutionary and mechanistic perspectives. Neurosci. Biobehav. Rev. 2013, 37, 790–802. [Google Scholar] [CrossRef]
- Camici, G.G.; Schiavoni, M.; Francia, P.; Bachschmid, M.; Martin-Padura, I.; Hersberger, M.; Tanner, F.C.; Pelicci, P.; Volpe, M.; Anversa, P.; et al. Genetic deletion of p66(Shc) adaptor protein prevents hyperglycemia-induced endothelial dysfunction and oxidative stress. Proc. Natl. Acad. Sci. USA 2007, 104, 5217–5222. [Google Scholar] [CrossRef]
- Grant, B.; Sandelson, M.; Agyemang-Prempeh, B.; Zalin, A. Managing obesity in people with type 2 diabetes. Clin. Med. 2021, 21, e327–e331. [Google Scholar] [CrossRef]
- Liu, T.S.; Pei, Y.H.; Peng, Y.P.; Chen, J.; Jiang, S.S.; Gong, J.B. Oscillating high glucose enhances oxidative stress and apoptosis in human coronary artery endothelial cells. J. Endocrinol. Investig. 2014, 37, 645–651. [Google Scholar] [CrossRef]
- Lorenzo, O.; Ramirez, E.; Picatoste, B.; Egido, J.; Tunon, J. Alteration of energy substrates and ROS production in diabetic cardiomyopathy. Mediators Inflamm. 2013, 2013, 461967. [Google Scholar] [CrossRef]
- Risso, A.; Mercuri, F.; Quagliaro, L.; Damante, G.; Ceriello, A. Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E924–E930. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Shen, H.; Liu, H.; Wang, Y.; Bai, Y.; Han, P. Acute blood glucose fluctuation enhances rat aorta endothelial cell apoptosis, oxidative stress and pro-inflammatory cytokine expression in vivo. Cardiovasc. Diabetol. 2016, 15, 109. [Google Scholar] [CrossRef] [PubMed]
- Culic, O.; Gruwel, M.L.; Schrader, J. Energy turnover of vascular endothelial cells. Am. J. Physiol. 1997, 273, C205–C213. [Google Scholar] [CrossRef] [PubMed]
- Darley-Usmar, V. The powerhouse takes control of the cell; the role of mitochondria in signal transduction. Free Radic. Biol. Med. 2004, 37, 753–754. [Google Scholar] [CrossRef]
- Barja, G. Mitochondrial oxygen consumption and reactive oxygen species production are independently modulated: Implications for aging studies. Rejuvenation Res. 2007, 10, 215–224. [Google Scholar] [CrossRef]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef]
- Dowling, D.K.; Simmons, L.W. Reactive oxygen species as universal constraints in life-history evolution. Proc. Biol. Sci. 2009, 276, 1737–1745. [Google Scholar] [CrossRef]
- Juan, C.A.; Perez de la Lastra, J.M.; Plou, F.J.; Perez-Lebena, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef]
- Endemann, D.H.; Schiffrin, E.L. Nitric oxide, oxidative excess, and vascular complications of diabetes mellitus. Curr. Hypertens. Rep. 2004, 6, 85–89. [Google Scholar] [CrossRef]
- Amina, B.S.G.; Nassima, M.S.; Hafida, M.; Sid Ahmed, M.; Ahmed, S.B. Elevation of oxidative stress markers in Type 1 diabetic children. J. Diabetes Endocrinol. 2015, 6, 5–11. [Google Scholar] [CrossRef]
- Dominguez, C.; Ruiz, E.; Gussinye, M.; Carrascosa, A. Oxidative stress at onset and in early stages of type 1 diabetes in children and adolescents. Diabetes Care 1998, 21, 1736–1742. [Google Scholar] [CrossRef] [PubMed]
- El-Din Elshalkami, N.M.M.; Salem, N.A.B.; ElShabrawy, W.O.; Abou-Elhassan, S.M. Oxidative status and its relationship with glycemic state in children with type 1 diabetes mellitus. Alex. J. Pediatr. 2022, 35, 201–207. [Google Scholar] [CrossRef]
- Ogawa, S.; Nakayama, K.; Nakayama, M.; Mori, T.; Matsushima, M.; Okamura, M.; Senda, M.; Nako, K.; Miyata, T.; Ito, S. Methylglyoxal is a predictor in type 2 diabetic patients of intima-media thickening and elevation of blood pressure. Hypertension 2010, 56, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Sena, C.M.; Matafome, P.; Crisostomo, J.; Rodrigues, L.; Fernandes, R.; Pereira, P.; Seica, R.M. Methylglyoxal promotes oxidative stress and endothelial dysfunction. Pharmacol. Res. 2012, 65, 497–506. [Google Scholar] [CrossRef]
- Vidal, N.; Cavaille, J.P.; Graziani, F.; Robin, M.; Ouari, O.; Pietri, S.; Stocker, P. High throughput assay for evaluation of reactive carbonyl scavenging capacity. Redox Biol. 2014, 2, 590–598. [Google Scholar] [CrossRef]
- Bourajjaj, M.; Stehouwer, C.D.; van Hinsbergh, V.W.; Schalkwijk, C.G. Role of methylglyoxal adducts in the development of vascular complications in diabetes mellitus. Biochem. Soc. Trans. 2003, 31, 1400–1402. [Google Scholar] [CrossRef]
- Schalkwijk, C.G.; Micali, L.R.; Wouters, K. Advanced glycation endproducts in diabetes-related macrovascular complications: Focus on methylglyoxal. Trends Endocrinol. Metab. 2023, 34, 49–60. [Google Scholar] [CrossRef]
- Chen, X.; Mori, T.; Guo, Q.; Hu, C.; Ohsaki, Y.; Yoneki, Y.; Zhu, W.; Jiang, Y.; Endo, S.; Nakayama, K.; et al. Carbonyl stress induces hypertension and cardio-renal vascular injury in Dahl salt-sensitive rats. Hypertens. Res. 2013, 36, 361–367. [Google Scholar] [CrossRef]
- Hayashi, T.; Juliet, P.A.; Miyazaki, A.; Ignarro, L.J.; Iguchi, A. High glucose downregulates the number of caveolae in monocytes through oxidative stress from NADPH oxidase: Implications for atherosclerosis. Biochim. Biophys. Acta 2007, 1772, 364–372. [Google Scholar] [CrossRef]
- Patente, T.A.; Mohammedi, K.; Bellili-Munoz, N.; Driss, F.; Sanchez, M.; Fumeron, F.; Roussel, R.; Hadjadj, S.; Correa-Giannella, M.L.; Marre, M.; et al. Allelic variations in the CYBA gene of NADPH oxidase and risk of kidney complications in patients with type 1 diabetes. Free Radic. Biol. Med. 2015, 86, 16–24. [Google Scholar] [CrossRef]
- Hayashi, T.; Yano, K.; Matsui-Hirai, H.; Yokoo, H.; Hattori, Y.; Iguchi, A. Nitric oxide and endothelial cellular senescence. Pharmacol. Ther. 2008, 120, 333–339. [Google Scholar] [CrossRef]
- Konior, A.; Schramm, A.; Czesnikiewicz-Guzik, M.; Guzik, T.J. NADPH oxidases in vascular pathology. Antioxid. Redox Signal. 2014, 20, 2794–2814. [Google Scholar] [CrossRef] [PubMed]
- Kassan, M.; Choi, S.-K.; Galán, M.; Lee, Y.-H.; Trebak, M.; Matrougui, K. Enhanced p22PHOX expression impairs vascular function through p38 and ERK1/2 MAP kinase-dependent mechanisms in type 2 diabetic mice. Am. J. Physiol.-Heart Circ. Physiol. 2014, 306, H972–H980. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Pittman, R.N.; Popel, A.S. Nitric oxide in the vasculature: Where does it come from and where does it go? A quantitative perspective. Antioxid. Redox Signal. 2008, 10, 1185–1198. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.; Garcia, T.; Aniqa, M.; Ali, S.; Ally, A.; Nauli, S.M. Endothelial Nitric Oxide Synthase (eNOS) and the Cardiovascular System: In Physiology and in Disease States. Am. J. Biomed. Sci. Res. 2022, 15, 153–177. [Google Scholar] [CrossRef]
- Vanhoutte, P.M.; Shimokawa, H.; Feletou, M.; Tang, E.H. Endothelial dysfunction and vascular disease-a 30th anniversary update. Acta Physiol. 2017, 219, 22–96. [Google Scholar] [CrossRef]
- Caldwell, R.W.; Rodriguez, P.C.; Toque, H.A.; Narayanan, S.P.; Caldwell, R.B. Arginase: A Multifaceted Enzyme Important in Health and Disease. Physiol. Rev. 2018, 98, 641–665. [Google Scholar] [CrossRef]
- Mahdi, A.; Kovamees, O.; Checa, A.; Wheelock, C.E.; von Heijne, M.; Alvarsson, M.; Pernow, J. Arginase inhibition improves endothelial function in patients with type 2 diabetes mellitus despite intensive glucose-lowering therapy. J. Intern. Med. 2018, 284, 388–398. [Google Scholar] [CrossRef]
- Pernow, J.; Jung, C. Arginase as a potential target in the treatment of cardiovascular disease: Reversal of arginine steal? Cardiovasc. Res. 2013, 98, 334–343. [Google Scholar] [CrossRef]
- Liao, Y.; Gou, L.; Chen, L.; Zhong, X.; Zhang, D.; Zhu, H.; Lu, X.; Zeng, T.; Deng, X.; Li, Y. NADPH oxidase 4 and endothelial nitric oxide synthase contribute to endothelial dysfunction mediated by histone methylations in metabolic memory. Free Radic. Biol. Med. 2018, 115, 383–394. [Google Scholar] [CrossRef]
- El-Osta, A.; Brasacchio, D.; Yao, D.; Pocai, A.; Jones, P.L.; Roeder, R.G.; Cooper, M.E.; Brownlee, M. Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J. Exp. Med. 2008, 205, 2409–2417. [Google Scholar] [CrossRef]
- Wautier, J.L.; Wautier, M.P. Endothelial Cell Participation in Inflammatory Reaction. Int. J. Mol. Sci. 2021, 22, 6341. [Google Scholar] [CrossRef]
- Rodriguez-Manas, L.; Lopez-Doriga, P.; Petidier, R.; Neira, M.; Solis, J.; Pavon, I.; Peiro, C.; Sanchez-Ferrer, C.F. Effect of glycaemic control on the vascular nitric oxide system in patients with type 1 diabetes. J. Hypertens. 2003, 21, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Bolanle, I.O.; Riches-Suman, K.; Williamson, R.; Palmer, T.M. Emerging roles of protein O-GlcNAcylation in cardiovascular diseases: Insights and novel therapeutic targets. Pharmacol. Res. 2021, 165, 105467. [Google Scholar] [CrossRef] [PubMed]
- Wells, L.; Vosseller, K.; Hart, G.W. Glycosylation of nucleocytoplasmic proteins: Signal transduction and O-GlcNAc. Science 2001, 291, 2376–2378. [Google Scholar] [CrossRef] [PubMed]
- Du, X.L.; Edelstein, D.; Dimmeler, S.; Ju, Q.; Sui, C.; Brownlee, M. Hyperglycemia inhibits endothelial nitric oxide synthase activity by posttranslational modification at the Akt site. J. Clin. Investig. 2001, 108, 1341–1348. [Google Scholar] [CrossRef]
- Michell, B.J.; Griffiths, J.E.; Mitchelhill, K.I.; Rodriguez-Crespo, I.; Tiganis, T.; Bozinovski, S.; de Montellano, P.R.; Kemp, B.E.; Pearson, R.B. The Akt kinase signals directly to endothelial nitric oxide synthase. Curr. Biol. 1999, 9, 845–848. [Google Scholar] [CrossRef]
- Du, X.L.; Edelstein, D.; Rossetti, L.; Fantus, I.G.; Goldberg, H.; Ziyadeh, F.; Wu, J.; Brownlee, M. Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc. Natl. Acad. Sci. USA 2000, 97, 12222–12226. [Google Scholar] [CrossRef]
- Fulton, D.; Gratton, J.P.; McCabe, T.J.; Fontana, J.; Fujio, Y.; Walsh, K.; Franke, T.F.; Papapetropoulos, A.; Sessa, W.C. Regulation of endothelium-derived nitric oxide production by the protein kinase Akt. Nature 1999, 399, 597–601. [Google Scholar] [CrossRef]
- Beleznai, T.; Bagi, Z. Activation of hexosamine pathway impairs nitric oxide (NO)-dependent arteriolar dilations by increased protein O-GlcNAcylation. Vascul. Pharmacol. 2012, 56, 115–121. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Offler, L.J.; Wells, L.K.; Palmer, T.M. The Impact of Glycaemic Variability on Vascular Dysfunction in Diabetes. Biomolecules 2025, 15, 1544. https://doi.org/10.3390/biom15111544
Offler LJ, Wells LK, Palmer TM. The Impact of Glycaemic Variability on Vascular Dysfunction in Diabetes. Biomolecules. 2025; 15(11):1544. https://doi.org/10.3390/biom15111544
Chicago/Turabian StyleOffler, Laura J., Liz K. Wells, and Timothy M. Palmer. 2025. "The Impact of Glycaemic Variability on Vascular Dysfunction in Diabetes" Biomolecules 15, no. 11: 1544. https://doi.org/10.3390/biom15111544
APA StyleOffler, L. J., Wells, L. K., & Palmer, T. M. (2025). The Impact of Glycaemic Variability on Vascular Dysfunction in Diabetes. Biomolecules, 15(11), 1544. https://doi.org/10.3390/biom15111544






