PRR11 in Malignancies: Biological Activities and Targeted Therapies
Abstract
1. Introduction
2. Oncogenic role of PRR11 in Various Malignancies
2.1. Tongue Squamous Cell Carcinoma (TSCC)
2.2. Esophageal Squamous Cell Carcinoma (ESCC)
2.3. Non-small Cell Lung Cancer (NSCLC)
2.4. Breast Cancer (BRCA)
2.5. Gastric Cancer (GC) and Colorectal Cancer (CRC)
2.6. Hepatocellular Carcinoma (HCC) and Hilar Cholangiocarcinoma (HCCA)
2.7. Pancreatic Cancer
2.8. Ovarian Cancer
2.9. Osteosarcoma
3. PRR11 Involved Oncogenic Signaling Pathways and Biological Molecules
3.1. Signaling Pathways
3.2. Biological Molecules
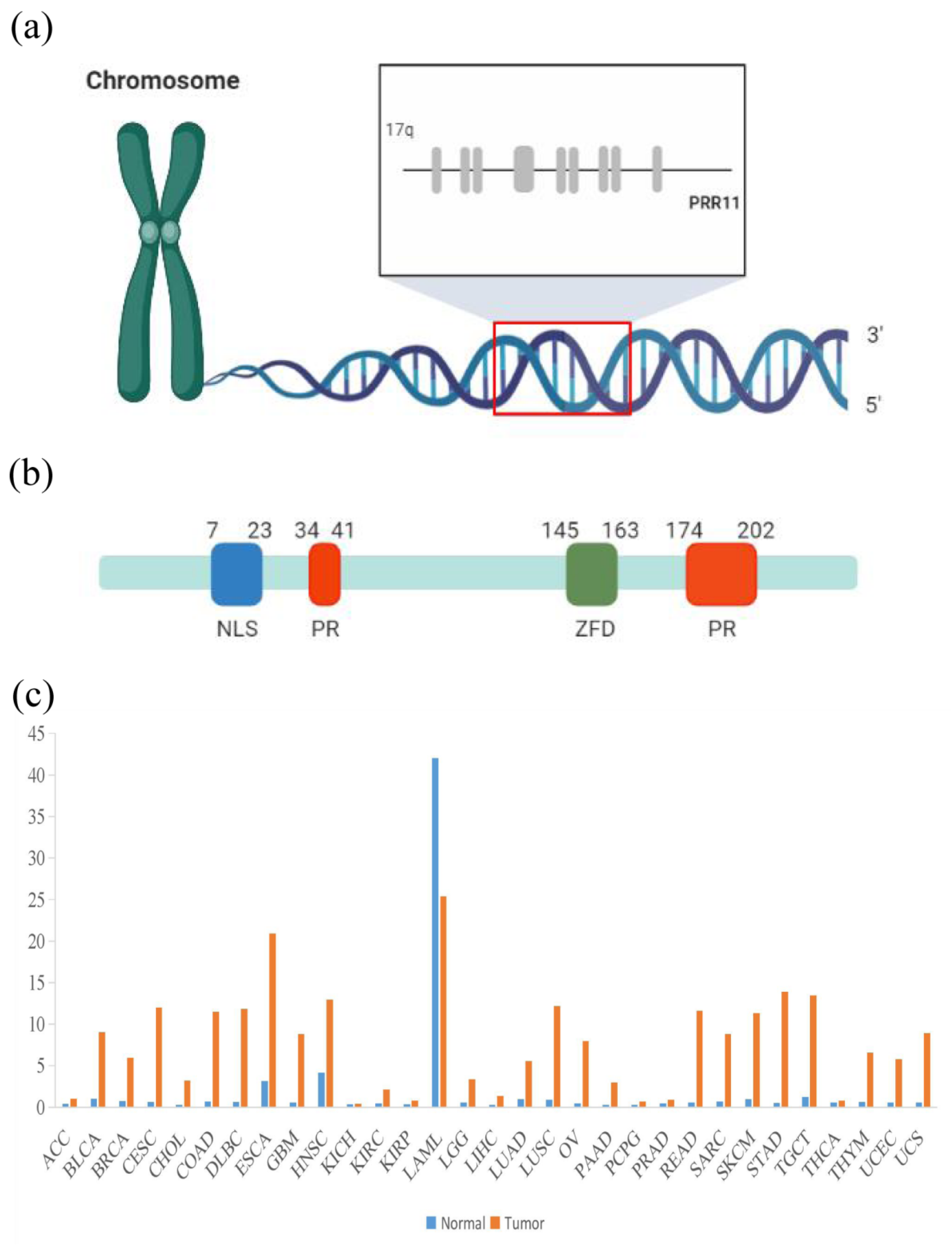
4. Biological and Clinical Significance of PRR11 in Human Cancers
4.1. PRR11 Involved in Tumorigenesis of Human Cancers
4.2. PRR11 Involved in Diagnosis and Prognosis of Malignancies
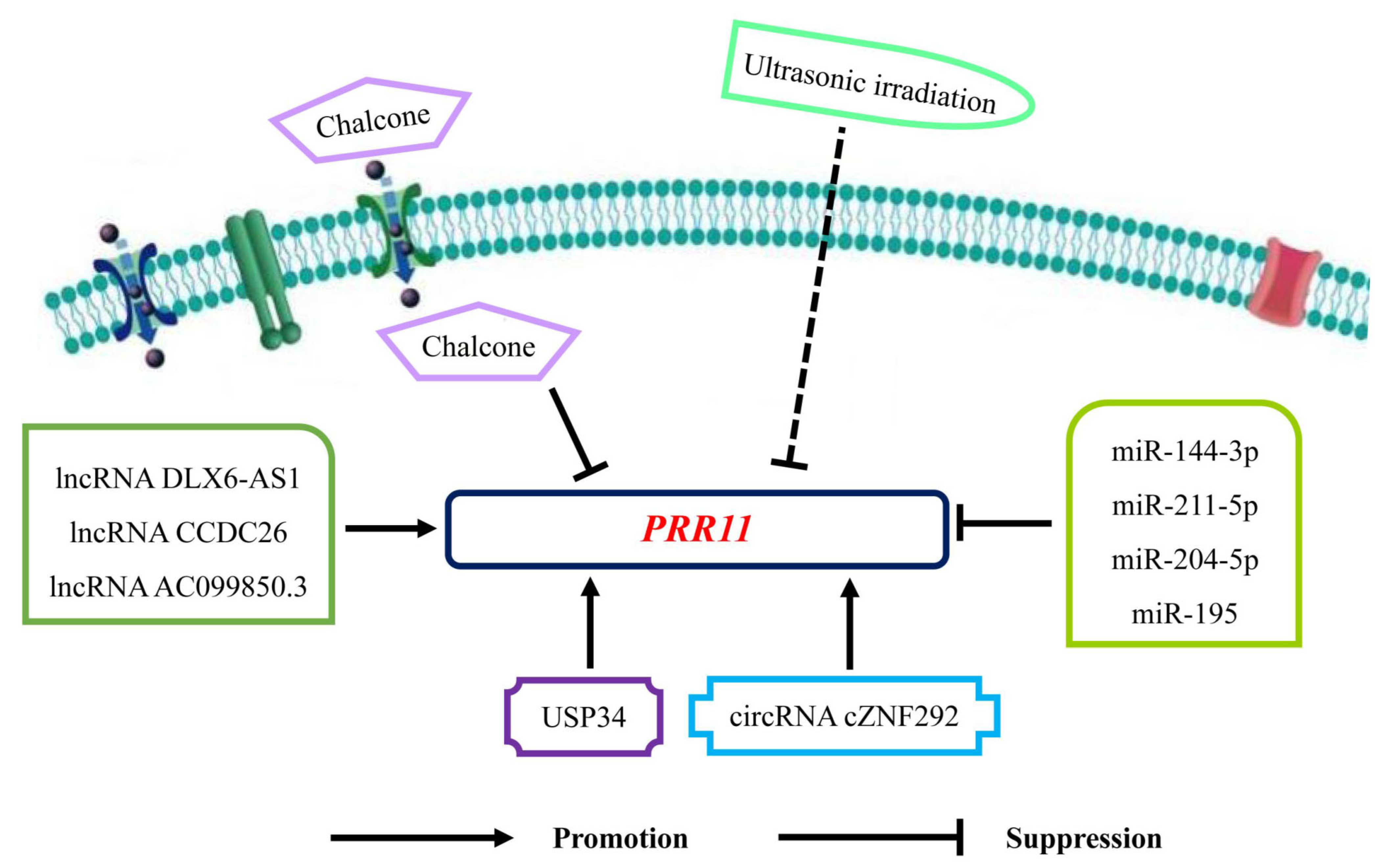
5. PRR11 Involved Targeted Therapies in Various Malignancies
5.1. Interactions between PRR11 and miRNAs
5.2. Interactions between PRR11 and lncRNAs
5.3. Interactions between PRR11 and other Molecules
6. Conclusions and Future Prospective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Lee, K.-M.; Guerrero-Zotano, A.L.; Servetto, A.; Sudhan, D.R.; Lin, C.-C.; Formisano, L.; Jansen, V.M.; González-Ericsson, P.; Sanders, M.E.; Stricker, T.P.; et al. Proline rich 11 (PRR11) overexpression amplifies PI3K signaling and promotes antiestrogen resistance in breast cancer. Nat. Commun. 2020, 11, 5488. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Xie, M.; Lan, H.; Zhang, Y.; Long, Y.; Weng, H.; Li, D.; Cai, W.; Zhu, H.; Niu, Y.; et al. PRR11 is a novel gene implicated in cell cycle progression and lung cancer. Int. J. Biochem. Cell Biol. 2013, 45, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Weng, H.; Zhang, Y.; Long, Y.; Li, Y.; Niu, Y.; Song, F.; Bu, Y. The PRR11-SKA2 Bidirectional Transcription Unit Is Negatively Regulated by p53 through NF-Y in Lung Cancer Cells. Int. J. Mol. Sci. 2017, 18, 534. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yang, H.; Zhou, H.; Peng, R.; Niu, Z.; Kang, C. PRR11 and SKA2 promote the proliferation, migration and invasion of esophageal carcinoma cells. Oncol. Lett. 2020, 20, 639–646. [Google Scholar] [CrossRef]
- Zhou, F.; Liu, H.; Zhang, X.; Shen, Y.; Zheng, D.; Zhang, A.; Lai, Y.; Li, H. Proline-rich protein 11 regulates epitheli-al-to-mesenchymal transition to promote breast cancer cell invasion. Int. J. Clin. Exp. Pathol. 2014, 7, 8692–8699. [Google Scholar]
- Hu, H.; Song, Z.; Yao, Q.; Geng, X.; Jiang, L.; Guo, C.; Li, H. Proline-Rich Protein 11 Regulates Self-Renewal and Tumorigenicity of Gastric Cancer Stem Cells. Cell. Physiol. Biochem. 2018, 47, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Hu, H.; Wang, J.; Yang, Y.; Yi, P. PRR11 Overexpression Facilitates Ovarian Carcinoma Cell Proliferation, Migration, and Invasion Through Activation of the PI3K/AKT/β-Catenin Pathway. Cell. Physiol. Biochem. 2018, 49, 696–705. [Google Scholar] [CrossRef]
- Wang, C.; Yu, L.; Ren, X.; Wu, T.; Chen, X.; Huang, Y.; Cheng, B. The oncogenic potential of PRR11 gene in Tongue Squamous Cell Carcinoma cells. J. Cancer 2019, 10, 2541–2551. [Google Scholar] [CrossRef]
- Zhao, Q. RNAi-mediated silencing of praline-rich gene causes growth reduction in human lung cancer cells. Int. J. Clin. Exp. Pathol. 2015, 8, 1760–1767. [Google Scholar]
- Zhang, L.; Lei, Y.; Zhang, Y.; Li, Y.; Bu, Y.; Song, F.; Zhang, C. Silencing of PRR11 suppresses cell proliferation and induces au-tophagy in NSCLC cells. Genes Dis. 2018, 5, 158–166. [Google Scholar] [CrossRef]
- Zhan, Y.; Wu, X.; Zheng, G.; Jin, J.; Li, C.; Yu, G.; Li, W. Proline-rich protein 11 overexpression is associated with a more aggressive phenotype and poor overall survival in ovarian cancer patients. World J. Surg. Oncol. 2020, 18, 318. [Google Scholar] [CrossRef]
- Chen, Y.; Cha, Z.; Fang, W.; Qian, B.; Yu, W.; Li, W.; Yu, G.; Gao, Y. The prognostic potential and oncogenic effects of PRR11 expression in hilar cholangiocarcinoma. Oncotarget 2015, 6, 20419–20433. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Liu, W.; Xiao, Y.; Zhang, M.; Luo, Y.; Yuan, W.; Xu, Y.; Yu, G.; Hu, Y. PRR11 Is a Prognostic Marker and Potential Oncogene in Patients with Gastric Cancer. PLoS ONE 2015, 10, e0128943. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.; Jiang, Z.; Hou, A.; Wang, J.; Zhang, J.; Dai, L. Expression of PRR11 protein and its correlation with pancreatic cancer and effect on survival. Oncol. Lett. 2017, 13, 4117–4122. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Deng, Z.Z.; Li, H.Y.; Jiang, N.; Wei, Z.S.; Hong, M.F.; Wang, J.H.; Zhang, M.X.; Shi, Y.H.; Lu, Z.Q.; et al. Overex-pression of PRR11 promotes tumorigenic capability and is associated with progression in esophageal squamous cell carcinoma. Onco Targets Ther. 2019, 12, 2677–2693. [Google Scholar] [CrossRef]
- Zhang, H.; He, Z.; Qiu, L.; Wei, J.; Gong, X.; Xian, M.; Chen, Z.; Cui, Y.; Fu, S.; Zhang, Z.; et al. PRR11 promotes cell proliferation by regulating PTTG1 through interacting with E2F1 transcription factor in pan-cancer. Front. Mol. Biosci. 2022, 9, 877320. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, C.; Mai, L.; Niu, Y.; Wang, Y.; Bu, Y. PRR11 and SKA2 gene pair is overexpressed and regulated by p53 in breast cancer. BMB Rep. 2019, 52, 157–162. [Google Scholar] [CrossRef]
- Su, Q.; Shen, H.; Gu, B.; Zhu, N. miR-204-5p Hampers Breast Cancer Malignancy and Affects the Cell Cycle by Targeting PRR11. Comput. Math. Methods Med. 2022, 2022, 4010947. [Google Scholar] [CrossRef]
- Ma, H.; Yang, W.; Wang, X.; Dai, G. PRR11 Promotes Proliferation and Migration of Colorectal Cancer through Activating the EGFR/ERK/AKT Pathway via Increasing CTHRC1. Ann. Clin. Lab. Sci. 2022, 52, 86–94. [Google Scholar]
- Qiao, W.; Wang, H.; Zhang, X.; Luo, K. Proline-rich protein 11 silencing inhibits hepatocellular carcinoma growth and epitheli-al-mesenchymal transition through beta-catenin signaling. Gene 2019, 681, 7–14. [Google Scholar] [CrossRef]
- Li, K.; Yu, H.; Zhao, C.; Li, J.; Tan, R.; Chen, L. Down-regulation of PRR11 affects the proliferation, migration and invasion of osteosarcoma by inhibiting the Wnt/beta-catenin pathway. J. Cancer 2021, 12, 6656–6664. [Google Scholar] [CrossRef]
- Wang, C.; Yu, L.; Hu, F.; Wang, J.; Chen, X.; Tai, S.; Cheng, B. Upregulation of proline rich 11 is an independent unfavorable prognostic factor for survival of tongue squamous cell carcinoma patients. Oncol. Lett. 2017, 14, 4527–4534. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Noguchi, S.; Hattori, A.; Tanimoto, N.; Nishida, R.; Hirano, K.; Wada, Y.; Matsuyama, S.; Shimada, T.; Akiyoshi, H. Establishing cell lines for canine tonsillar and non-tonsillar oral squamous cell carcinoma and identifying characteristics associated with ma-lignancy. Tissue Cell. 2020, 67, 101408. [Google Scholar] [CrossRef] [PubMed]
- Karatas, O.F.; Oner, M.; Abay, A.; Diyapoglu, A. MicroRNAs in human tongue squamous cell carcinoma: From pathogenesis to therapeutic implications. Oral Oncol. 2017, 67, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Hararah, M.K.; Stokes, W.A.; Oweida, A.; Patil, T.; Amini, A.; Goddard, J.; Bowles, D.W.; Karam, S.D. Epidemiology and treatment trends for primary tracheal squamous cell carcinoma. Laryngoscope 2019, 130, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Kurumi, H.; Isomoto, H. Current Topics in Esophageal Squamous Cell Carcinoma. Cancers 2020, 12, 2898. [Google Scholar] [CrossRef]
- Hall, T.M.; Tétreault, M.-P.; Hamilton, K.E.; Whelan, K.A. Autophagy as a cytoprotective mechanism in esophageal squamous cell carcinoma. Curr. Opin. Pharmacol. 2018, 41, 12–19. [Google Scholar] [CrossRef]
- Macedo-Silva, C.; Miranda-Goncalves, V.; Lameirinhas, A.; Lencart, J.; Pereira, A.; Lobo, J.; Guimaraes, R.; Martins, A.T.; Hen-rique, R.; Bravo, I.; et al. JmjC-KDMs KDM3A and KDM6B modulate radioresistance under hypoxic conditions in esoph-ageal squamous cell carcinoma. Cell Death Dis. 2020, 11, 1068. [Google Scholar] [CrossRef]
- Chen, N.; Zhang, G.; Fu, J.; Wu, Q. Identification of Key Modules and Hub Genes Involved in Esophageal Squamous Cell Car-cinoma Tumorigenesis Using WCGNA. Cancer Control. 2020, 27, 1073274820978817. [Google Scholar] [CrossRef]
- Chen, J.; Kang, C.-Y.; Niu, Z.-X.; Zhou, H.-C.; Yang, H.-M. A chalcone inhibits the growth and metastasis of KYSE-4 esophageal cancer cells. J. Int. Med. Res. 2020, 48, 300060520928831. [Google Scholar] [CrossRef]
- Socinski, M.A.; Obasaju, C.; Gandara, D.; Hirsch, F.R.; Bonomi, P.; Bunn, P.; Kim, E.S.; Langer, C.J.; Natale, R.B.; Novello, S.; et al. Clinicopathologic Features of Advanced Squamous NSCLC. J. Thorac. Oncol. 2016, 11, 1411–1422. [Google Scholar] [CrossRef]
- Bansal, P.; Osman, D.; Gan, G.N.; Simon, G.R.; Boumber, Y. Recent Advances in Immunotherapy in Metastatic NSCLC. Front. Oncol. 2016, 6, 239. [Google Scholar] [CrossRef] [PubMed]
- Friedlaender, A.; Addeo, A.; Russo, A.; Gregorc, V.; Cortinovis, D.; Rolfo, C.D. Targeted Therapies in Early Stage NSCLC: Hype or Hope? Int. J. Mol. Sci. 2020, 21, 6329. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Ohbayashi, C.; Yanagita, E.; Jimbo, N.; Kajimoto, K.; Sakuma, T.; Hirose, T.; Yoshimura, M.; Maniwa, Y.; Itoh, T. PRR11 immunoreactivity is a weak prognostic factor in non-mucinous invasive adenocarcinoma of the lung. Pathologica 2017, 109, 133–139. [Google Scholar] [PubMed]
- Zhang, L.; Zhang, Y.; Lei, Y.; Wei, Z.; Li, Y.; Wang, Y.; Bu, Y.; Zhang, C. Proline-rich 11 (PRR11) drives F-actin assembly by recruiting the actin-related protein 2/3 complex in human non-small cell lung carcinoma. J. Biol. Chem. 2020, 295, 5335–5349. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.; Zhang, C.; Weng, H.; Li, Y.; Cai, W.; Xie, M.; Long, Y.; Ai, Q.; Liu, Z.; et al. The gene pair PRR11 and SKA2 shares a NF-Y-regulated bidirectional promoter and contributes to lung cancer devel-opment. Biochim. Biophys. Acta 2015, 1849, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ni, R.; Wang, J.; Liu, Y. Knockdown of lncRNA DLX6-AS1 inhibits cell proliferation, migration and invasion while promotes apoptosis by downregulating PRR11 expression and upregulating miR-144 in non-small cell lung cancer. Biomed. Pharmacother. 2019, 109, 1851–1859. [Google Scholar] [CrossRef] [PubMed]
- Jafari, S.H.; Saadatpour, Z.; Salmaninejad, A.; Momeni, F.; Mokhtari, M.; Nahand, J.S.; Rahmati, M.; Mirzaei, H.; Kianmehr, M. Breast cancer diagnosis: Imaging techniques and biochemical markers. J. Cell. Physiol. 2018, 233, 5200–5213. [Google Scholar] [CrossRef]
- Barzaman, K.; Karami, J.; Zarei, Z.; Hosseinzadeh, A.; Kazemi, M.H.; Moradi-Kalbolandi, S.; Safari, E.; Farahmand, L. Breast cancer: Biology, biomarkers, and treatments. Int. Immunopharmacol. 2020, 84, 106535. [Google Scholar] [CrossRef]
- Yang, H.; Zhou, L.; Chen, J.; Su, J.; Shen, W.; Liu, B.; Zhou, J.; Yu, S.; Qian, J. A four-gene signature for prognosis in breast cancer patients with hypermethylated IL15RA. Oncol. Lett. 2019, 17, 4245–4254. [Google Scholar] [CrossRef]
- Luo, H.; Li, J.; Lin, Q.; Xiao, X.; Shi, Y.; Ye, X.; Wei, Z.; Liu, Y.; Xu, J. Ultrasonic irradiation and SonoVue microbubbles-mediated RNA interference targeting PRR11 inhibits breast cancer cells proliferation and metastasis, but promotes apoptosis. Biosci. Rep. 2020, 40, BSR20201854. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Tahara, T.; Shiroeda, H.; Minato, T.; Matsue, Y.; Hayashi, R.; Matsunaga, K.; Otsuka, T.; Nakamura, M.; Toshikuni, N.; et al. Influence of HRH2 promoter polymorphism on aberrant DNA methylation of DAPK and CDH1in the gastric epithelium. BMC Gastroenterol. 2013, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Raja, U.M.; Gopal, G.; Shirley, S.; Ramakrishnan, A.S.; Rajkumar, T. Immunohistochemical expression and localization of cyto-kines/chemokines/growth factors in gastric cancer. Cytokine 2017, 89, 82–90. [Google Scholar] [CrossRef]
- Dekker, E.; Tanis, P.J.; Vleugels, J.L.A.; Kasi, P.M.; Wallace, M.B. Colorectal cancer. Lancet 2019, 394, 1467–1480. [Google Scholar] [CrossRef] [PubMed]
- Barbáchano, A.; Fernández-Barral, A.; Bustamante-Madrid, P.; Prieto, I.; Rodríguez-Salas, N.; Larriba, M.; Muñoz, A. Organoids and Colorectal Cancer. Cancers 2021, 13, 2657. [Google Scholar] [CrossRef]
- Estes, C.; Razavi, H.; Loomba, R.; Younossi, Z.; Sanyal, A.J. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology 2018, 67, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Faivre, S.; Rimassa, L.; Finn, R.S. Molecular therapies for HCC: Looking outside the box. J. Hepatol. 2020, 72, 342–352. [Google Scholar] [CrossRef]
- Kim, D.W.; Talati, C.; Kim, R. Hepatocellular carcinoma (HCC): Beyond sorafenib—Chemotherapy. J. Gastrointest. Oncol. 2017, 8, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Li, J.; Guo, D.; Chen, B.; Liu, P.; Xiao, Y.; Yang, K.; Liu, Z.; Liu, Q. Identification of 13 Key Genes Correlated With Progression and Prognosis in Hepatocellular Carcinoma by Weighted Gene Co-expression Network Analysis. Front. Genet. 2020, 11, 153. [Google Scholar] [CrossRef] [PubMed]
- Rerknimitr, R.; Angsuwatcharakon, P.; Ratanachu-ek, T.; Khor, C.J.; Ponnudurai, R.; Moon, J.H.; Seo, D.W.; Pantongrag-Brown, L.; Sangchan, A.; Pisespongsa, P.; et al. Asia-Pacific Working Group on Hepatobiliary, Asia-Pacific consensus recom-mendations for endoscopic and interventional management of hilar cholangiocarcinoma. J. Gastroenterol. Hepatol. 2013, 28, 593–607. [Google Scholar] [CrossRef]
- Moris, D.; Kostakis, I.D.; Machairas, N.; Prodromidou, A.; Tsilimigras, D.I.; Ravindra, K.V.; Sudan, D.L.; Knechtle, S.J.; Barbas, A.S. Comparison between liver transplantation and resection for hilar cholangiocarcinoma: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0220527. [Google Scholar] [CrossRef] [PubMed]
- Kremer, D.M.; Nelson, B.S.; Lin, L.; Yarosz, E.L.; Halbrook, C.J.; Kerk, S.A.; Sajjakulnukit, P.; Myers, A.; Thurston, G.; Hou, S.W.; et al. GOT1 inhibition promotes pancreatic cancer cell death by ferroptosis. Nat. Commun. 2021, 12, 4860. [Google Scholar] [CrossRef] [PubMed]
- Jentzsch, V.; Davis, J.; Djamgoz, M. Pancreatic Cancer (PDAC): Introduction of Evidence-Based Complementary Measures into Integrative Clinical Management. Cancers 2020, 12, 3096. [Google Scholar] [CrossRef] [PubMed]
- Hau, S.O.; Wahlin, S.; Cervin, S.; Falk, V.; Nodin, B.; Elebro, J.; Eberhard, J.; Moran, B.; Gallagher, W.M.; Karnevi, E.; et al. PRR11 unveiled as a top candidate biomarker within the RBM3 -regulated transcriptome in pancreatic cancer. J. Pathol. Clin. Res. 2021, 8, 65–77. [Google Scholar] [CrossRef]
- Lin, C.; Xia, J.; Gu, Z.; Meng, Y.; Gao, D.; Wei, S. Downregulation of USP34 Inhibits the Growth and Migration of Pancreatic Cancer Cells via Inhibiting the PRR11. Onco Targets Ther. 2020, 13, 1471–1480. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, P.; Yue, Z.; Zhang, D.; You, K.; Wang, J. miR-144-3p Induces Cell Cycle Arrest and Apoptosis in Pancreatic Cancer Cells by Targeting Proline-Rich Protein 11 Expression via the Mitogen-Activated Protein Kinase Signaling Pathway. DNA Cell Biol. 2017, 36, 619–626. [Google Scholar] [CrossRef]
- Odunsi, K. Immunotherapy in ovarian cancer. Ann. Oncol. 2017, 28 (Suppl. S8), viii1–viii7. [Google Scholar] [CrossRef] [PubMed]
- Roett, M.A.; Evans, P. Ovarian cancer: An overview. Am. Fam. Physician 2009, 80, 609–616. [Google Scholar] [PubMed]
- Link, T.; Kuhlmann, J.D.; Kobelt, D.; Herrmann, P.; Vassileva, Y.D.; Kramer, M.; Frank, K.; Gockenjan, M.; Wimberger, P.; Stein, U. Clinical relevance of circulating MACC1 and S100A4 transcripts for ovarian cancer. Mol. Oncol. 2019, 13, 1268–1279. [Google Scholar] [CrossRef]
- Czarnecka, A.M.; Synoradzki, K.; Firlej, W.; Bartnik, E.; Sobczuk, P.; Fiedorowicz, M.; Grieb, P.; Rutkowski, P. Molecular Biology of Osteosarcoma. Cancers 2020, 12, 2130. [Google Scholar] [CrossRef] [PubMed]
- Rickel, K.; Fang, F.; Tao, J. Molecular genetics of osteosarcoma. Bone 2017, 102, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Pyun, W.Y.; Park, H.W. Cancer Metabolism: Phenotype, Signaling and Therapeutic Targets. Cells 2020, 9, 2308. [Google Scholar] [CrossRef] [PubMed]
- Zhan, T.; Rindtorff, N.; Boutros, M. WNT signaling in cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.; Giancotti, F.G. Integrin Signaling in Cancer: Mechanotransduction, Stemness, Epithelial Plasticity, and Therapeutic Resistance. Cancer Cell 2019, 35, 347–367. [Google Scholar] [CrossRef] [PubMed]
- Kulkoyluoglu-Cotul, E.; Arca, A.; Madak-Erdogan, Z. Crosstalk between Estrogen Signaling and Breast Cancer Metabolism. Trends Endocrinol. Metab. 2018, 30, 25–38. [Google Scholar] [CrossRef]
- Liu, K.; Gao, X.; Kang, B.; Liu, Y.; Wang, D.; Wang, Y. The Role of Tumor Stem Cell Exosomes in Cancer Invasion and Metasta-sis. Front. Oncol. 2022, 12, 836548. [Google Scholar] [CrossRef]
- Lee, Y.T.; Tan, Y.J.; Oon, C.E. Molecular targeted therapy: Treating cancer with specificity. Eur. J. Pharmacol. 2018, 834, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; Ryder, M.; Jimenez, C. Targeted Therapy for Advanced Thyroid Cancer: Kinase Inhibitors and Beyond. Endocr. Rev. 2019, 40, 1573–1604. [Google Scholar] [CrossRef]
- Syeda, Z.A.; Langden, S.S.S.; Munkhzul, C.; Lee, M.; Song, S.J. Regulatory Mechanism of MicroRNA Expression in Cancer. Int. J. Mol. Sci. 2020, 21, 1723. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, J.; Hayder, H.; Zayed, Y.; Peng, C. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol. 2018, 9, 402. [Google Scholar] [CrossRef]
- Yi, L.; Liu, Y.; Xu, A.; Li, S.; Zhang, H.; Peng, M.; Li, Z.; Ren, H.; Dai, J.; Luo, C.; et al. MicroRNA-26b-5p sup-presses the proliferation of tongue squamous cell carcinoma via targeting proline rich 11 (PRR11). Bioengineered 2021, 12, 5830–5838. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Yang, K.; Wang, W.; Tian, R.; Wang, H.; Wang, K. MicroRNA-211-5p promotes apoptosis and inhibits the migration of osteosarcoma cells by targeting proline-rich protein PRR11. Biochem. Cell Biol. 2020, 98, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; He, H.; Duan, X.; Wu, W.; Mai, Z.; Zhang, T.; Fan, J.; Deng, T.; Zhong, W.; Liu, Y.; et al. miR-195 inhibits cell proliferation and angiogenesis in human prostate cancer by downregulating PRR11 expression. Oncol. Rep. 2018, 39, 1658–1670. [Google Scholar] [CrossRef] [PubMed]
- Rashid, F.; Shah, A.; Shan, G. Long Non-coding RNAs in the Cytoplasm. Genom. Proteom. Bioinform. 2016, 14, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Mu, J.; Shi, Y.; Xin, H. LncRNA CCDC26 Interacts with CELF2 Protein to Enhance Myeloid Leukemia Cell Proliferation and Invasion via the circRNA_ANKIB1/miR-195-5p/PRR11 Axis. Cell Transplant. 2021, 30, 963689720986080. [Google Scholar] [CrossRef]
- Zhong, F.; Liu, S.; Hu, D.; Chen, L. LncRNA AC099850.3 promotes hepatocellular carcinoma proliferation and invasion through PRR11/PI3K/AKT axis and is associated with patients prognosis. J. Cancer 2022, 13, 1048–1060. [Google Scholar] [CrossRef]
- Yang, P.; Qiu, Z.; Jiang, Y.; Dong, L.; Yang, W.; Gu, C.; Li, G.; Zhu, Y. Silencing of cZNF292 circular RNA suppresses human glioma tube formation via the Wnt/beta-catenin signaling pathway. Oncotarget 2016, 7, 63449–63455. [Google Scholar] [CrossRef]
- Lin, J.; Yang, J.; Xu, X.; Wang, Y.; Yu, M.; Zhu, Y. A robust 11-genes prognostic model can predict overall survival in bladder cancer patients based on five cohorts. Cancer Cell Int. 2020, 20, 402. [Google Scholar] [CrossRef]
- Atambayeva, S.; Niyazova, R.; Ivashchenko, A.; Pyrkova, A.; Pinsky, I.; Akimniyazova, A.; Labeit, S. The Binding Sites of miR-619-5p in the mRNAs of Human and Orthologous Genes. BMC Genom. 2017, 18, 428. [Google Scholar] [CrossRef]
- Sun, Z.; Fu, X.; Zhang, L.; Yang, X.; Liu, F.; Hu, G. A protein chip system for parallel analysis of multi-tumor markers and its application in cancer detection. Anticancer Res. 2004, 24, 1159–1165. [Google Scholar]

| Human Cancers | Assessed Cell Lines | Targets | Related Biological Activities | Ref |
|---|---|---|---|---|
| Tongue squamous cell carcinoma (TSCC) | SCC15, HSC3 | p21, p27, CDK2, cyclin A | ↑proliferation, ↑invasion, ↑cell-cycle progression, ↑tumorigenicity | [8] |
| Esophageal squamous cell carcinoma (ESCC) | EC9706, EC109 | Akt, EMT signaling | ↑proliferation, ↑migration, ↑invasion | [4] |
| KYSE30, ECA109 | Wnt/β-catenin signaling | ↑stem cell-like traits, ↑tumorigenicity | [15] | |
| Non-small cell lung cancer (NSCLC) | H1299 | \ | ↑proliferation, ↑migration, ↑cell-cycle progression, ↑invasion, ↑tumorigenicity | [2] |
| H1299, A549 | \ | ↑proliferation, ↑cell-cycle progression, ↑tumorigenicity | [9] | |
| H1299, A549 | Akt/mTOR signaling | ↑proliferation, ↓apoptosis, ↓autophagy | [10] | |
| NCI-H460 | E2F1/PTTG1 | ↑proliferation, ↑migration | [16] | |
| Breast cancer (BRCA) | MCF7, HCC1428 | p85, PI3K signaling | ↑proliferation, ↑antiestrogen resistance | [1] |
| HS578T, MDA-MB-231 | EMT signaling | ↑proliferation, ↑invasion | [5] | |
| MCF7, MDA-MB-231 | \ | ↑proliferation, ↑migration, ↑invasion | [17] | |
| AU565, MDA-MB-231 | \ | ↑proliferation, ↑migration, ↑cell-cycle progression, ↑invasion | [18] | |
| Gastric cancer (GC) | SGC-7901, HGC-27 | MAPK signaling | ↑self-renewal, ↑stemness, ↑tumorigenicity | [6] |
| SGC-7901 | CTHRC1, LXN | ↑proliferation, ↑colony formation, ↑tumorigenicity | [13] | |
| Colorectal cancer (CRC) | SW480, HCT116 | EGFR/ERK/AKT signaling CTHRC1 | ↑proliferation, ↑migration, ↑invasion, ↑xenograft tumor growth | [19] |
| Hepatocellular carcinoma (HCC) | HepG2 | E2F1/PTTG1 | ↑proliferation, ↑migration | [16] |
| Huh7 | EMT, β-catenin signaling | ↑proliferation, ↑migration, ↑invasion, ↑xenograft tumor growth | [20] | |
| Hilar cholangiocarcinoma (HCCA) | QBC939 | EMT signalingUCHL1, SNAT1, EGR1 | ↑proliferation, ↑migration, ↑cell-cycle progression, ↑xenograft tumor growth | [12] |
| Pancreatic cancer | Capan-1 | \ | ↑proliferation, ↑migration | [14] |
| BxPC3 | E2F1/PTTG1 | ↑proliferation, ↑migration | [16] | |
| Ovarian cancer | Caov3, HO-8910 | PI3K/AKT/β-catenin signaling, c-myc, cyclin D1, MMP2, TIMP-2 | ↑proliferation, ↑migration, ↑invasion | [7] |
| SKOV3, HO-8910 | EGR1, N-cadherin | ↑proliferation, ↑migration, ↑xenograft tumor growth | [11] | |
| Osteosarcoma | SAOS2, MG63, U2OS | Wnt/β-catenin, EMT signaling | ↑proliferation, ↑migration, ↑invasion, ↓apoptosis | [21] |
| Human Cancers | Clinical Cases or Tissues | Clinical Significance | Ref |
|---|---|---|---|
| Tongue squamous cell carcinoma (TSCC) | 126 TSCC and 12 non-cancerous tongue tissue samples | ↑clinical stage, ↑T classification, ↑N classification, ↓vital status, ↓survival time | [22] |
| Non-small cell lung cancer (NSCLC) | 40 lung cancer tissues, 8 normal tissues and 246 lung cancer patients | ↑clinical stage, ↓overall survival | [2,9] |
| Breast cancer (BRCA) | 260 ER+ BRCA patients | ↓recurrence free survival, ↓relapse-free survival (RFS), ↑Ki67, ↑endocrine resistance | [1] |
| 109 BRCA patients | ↑lymph node metastasis, ↓overall survival | [5] | |
| Gastric cancer (GC) | 216 GC patients | ↑T stage, ↑TNM stage, ↑tumor differentiation, ↓overall survival | [13] |
| Hepatocellular carcinoma (HCC) | 80 pairs of HCC tissues and adjacent non-tumor liver tissues | ↑tumor size, ↑TNM stage, ↓overall survival | [20] |
| Hilar cholangiocarcinoma (HCCA) | 49 HC patients | ↑invasion, ↑lymph node metastasis, ↑CA199, ↑recurrence, ↓disease-free survival | [12] |
| Pancreatic cancer | 38 pancreatic cancer samples and 10 normal pancreatic tissues | ↑invasion, ↑tumor differentiation, ↓overall survival | [14] |
| Ovarian cancer | 51 pairs of OC tissues and normal ovarian surface epithelium tissues | ↑FIGO stage, ↑tumor size, ↑lymph node metastasis | [7] |
| 49 primary invasive OC patients | ↑FIGO stage, ↓overall survival | [11] | |
| Osteosarcoma | 62 pairs of osteosarcoma tissues and adjacent non-tumor tissues | ↑tumor size, ↑Enneking stage, ↑lymph node metastasis | [21] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, W.; Chen, L. PRR11 in Malignancies: Biological Activities and Targeted Therapies. Biomolecules 2022, 12, 1800. https://doi.org/10.3390/biom12121800
Han W, Chen L. PRR11 in Malignancies: Biological Activities and Targeted Therapies. Biomolecules. 2022; 12(12):1800. https://doi.org/10.3390/biom12121800
Chicago/Turabian StyleHan, Wei, and Liang Chen. 2022. "PRR11 in Malignancies: Biological Activities and Targeted Therapies" Biomolecules 12, no. 12: 1800. https://doi.org/10.3390/biom12121800
APA StyleHan, W., & Chen, L. (2022). PRR11 in Malignancies: Biological Activities and Targeted Therapies. Biomolecules, 12(12), 1800. https://doi.org/10.3390/biom12121800






