A Review on Health-Promoting, Biological, and Functional Aspects of Bioactive Peptides in Food Applications
Abstract
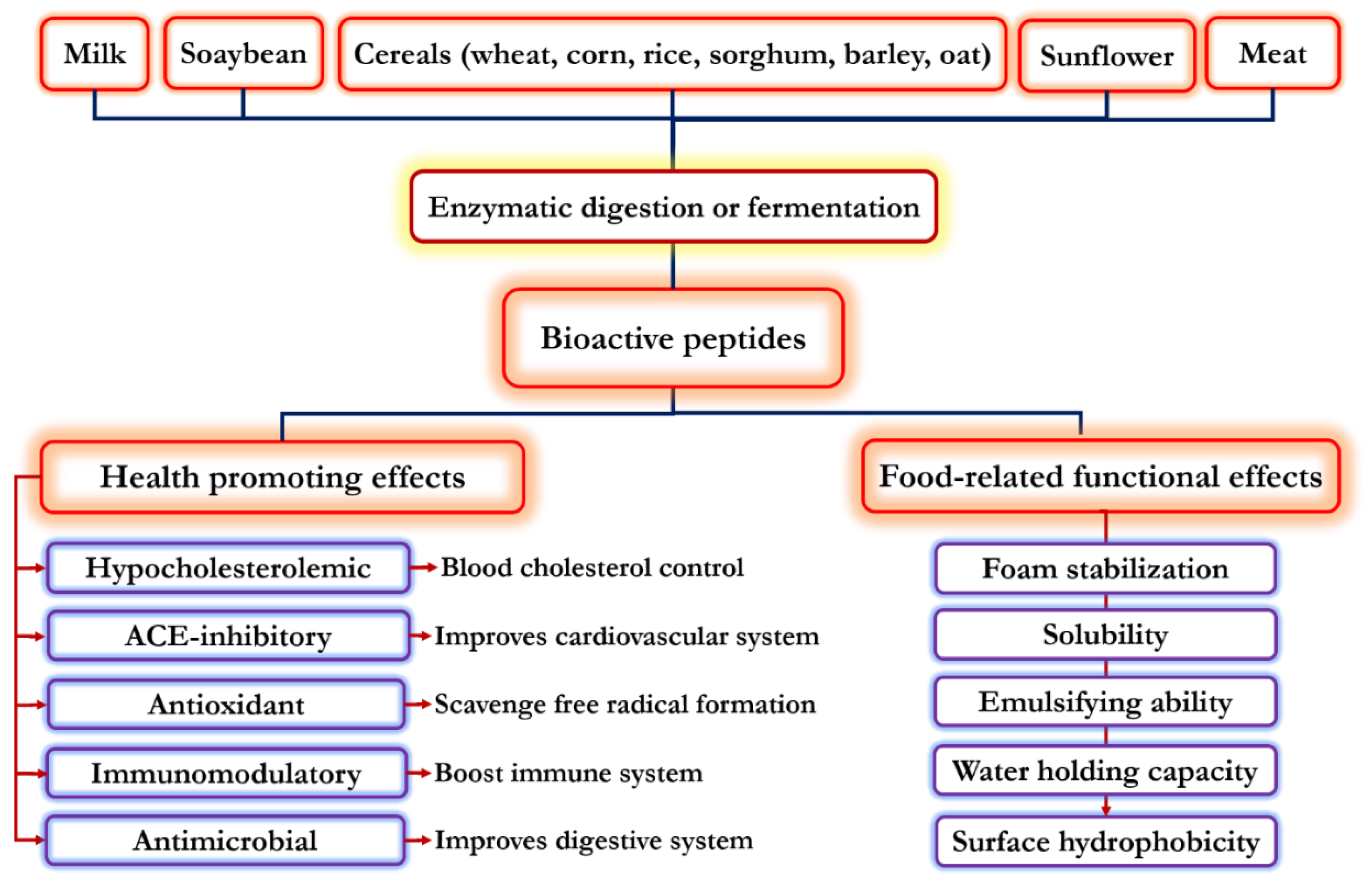
1. Introduction
2. Functional Properties of Food-Derived Bioactive Peptides
2.1. Solubility
2.2. Emulsifying Properties
2.3. Foaming Ability
2.4. Water Holding Capacity
2.5. Hydrophobicity
3. Physiological Properties of Food-Derived Bioactive Peptides
3.1. Bioactive Peptides with Antioxidant Activity
3.1.1. Milk-Derived Bioactive Peptides
3.1.2. Meat-Derived Bioactive Peptides
3.1.3. Marine-Derived Bioactive Peptides
3.1.4. Plant-Derived Bioactive Peptides
3.2. Bioactive Peptides with Angiotensin I-Converting Enzyme (ACE) Inhibitory Activity
3.2.1. Milk-Derived Bioactive Peptides
3.2.2. Meat-Derived Bioactive Peptides
3.2.3. Marine-Derived Bioactive Peptides
3.2.4. Plant-Derived Bioactive Peptides
3.3. Bioactive Peptides with Hypocholesterolemic Activity
3.3.1. Milk-Derived Bioactive Peptides
3.3.2. Meat-Derived Bioactive Peptides
3.3.3. Marine-Derived Bioactive Peptides
3.3.4. Plant-Derived Bioactive Peptides
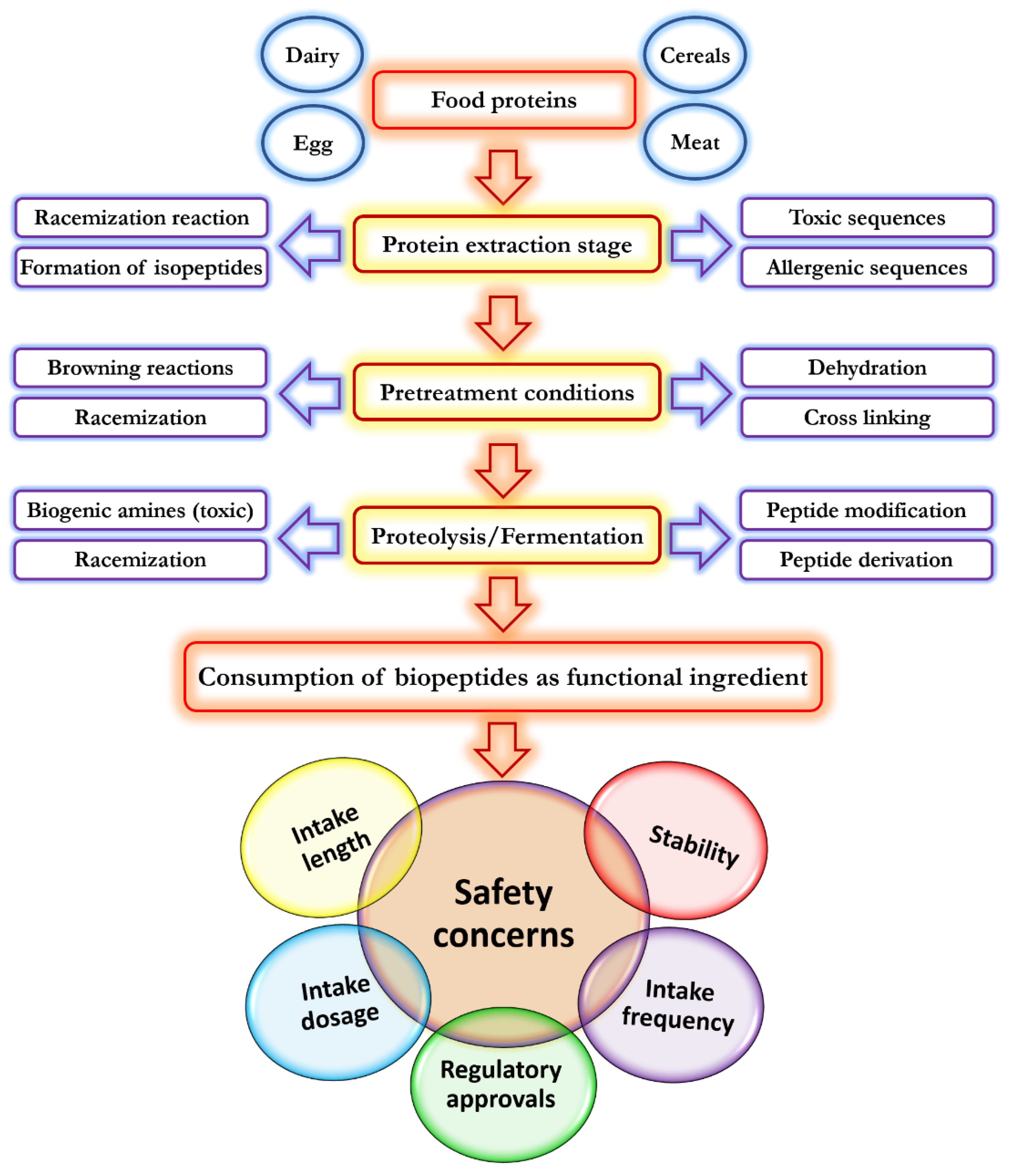
4. Application of Bioactive Peptides in Food Technology and Food Biosafety
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rezvani, M.; Hesari, J.; Peighambardoust, S.H.; Manconi, M.; Hamishehkar, H. Development and characterization of nanostructured pharmacosomal mesophases: An innovative delivery system for bioactive peptides. Adv. Pharm. Bull. 2018, 8, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Karami, Z.; Peighambardoust, S.H.; Hesari, J.; Akbari-Adergani, B.; Andreu, D. Identification and synthesis of multifunctional peptides from wheat germ hydrolysate fractions obtained by proteinase K digestion. J. Food Biochem. 2019, 43, e12800. [Google Scholar] [CrossRef] [PubMed]
- Karami, Z.; Peighambardoust, S.H.; Hesari, J.; Akbari-Adergani, B.; Andreu, D. Antioxidant, anticancer and ACE-inhibitory activities of bioactive peptides from wheat germ protein hydrolysates. Food Biosci. 2019, 32, 100450. [Google Scholar] [CrossRef]
- Borrajo, P.; Pateiro, M.; Barba, F.J.; Mora, L.; Franco, D.; Toldrá, F.; Lorenzo, J.M. Antioxidant and Antimicrobial Activity of Peptides Extracted from Meat By-products: A Review. Food Anal. Methods 2019, 12, 2401–2415. [Google Scholar] [CrossRef]
- Kim, S.K.; Wijesekara, I. Development and biological activities of marine-derived bioactive peptides: A review. J. Funct. Foods 2010, 2, 1–9. [Google Scholar] [CrossRef]
- Erdmann, K.; Cheung, B.W.Y.; Schröder, H. The possible roles of food-derived bioactive peptides in reducing the risk of cardiovascular disease. J. Nutr. Biochem. 2008, 19, 643–654. [Google Scholar] [CrossRef]
- Elias, R.J.; Kellerby, S.S.; Decker, E.A. Antioxidant activity of proteins and peptides. Crit. Rev. Food Sci. Nutr. 2008. [Google Scholar] [CrossRef]
- de Castro, R.J.S.; Sato, H.H. A response surface approach on optimization of hydrolysis parameters for the production of egg white protein hydrolysates with antioxidant activities. Biocatal. Agric. Biotechnol. 2015. [Google Scholar] [CrossRef]
- Klompong, V.; Benjakul, S.; Yachai, M.; Visessanguan, W.; Shahidi, F.; Hayes, K.D. Amino acid composition and antioxidative peptides from protein hydrolysates of yellow stripe trevally (Selaroides leptolepis). J. Food Sci. 2009. [Google Scholar] [CrossRef]
- Golshan Tafti, A.; Peighambardoust, S.H.; Hejazi, M.A.; Moosavy, M.H. Diversity of Lactobacillus strains in Iranian traditional wheat sourdough. J. Food Qual. Hazards Control 2014, 1, 41–45. [Google Scholar]
- Peighambardoust, S.H.; Beigmohammadi, F.; Peighambardoust, S.J. Application of Organoclay Nanoparticle in Low-Density Polyethylene Films for Packaging of UF Cheese. Packag. Technol. Sci. 2016, 29, 355–363. [Google Scholar] [CrossRef]
- Fasihnia, S.H.; Peighambardoust, S.H.; Peighambardoust, S.J. Nanocomposite films containing organoclay nanoparticles as an antimicrobial (active) packaging for potential food application. J. Food Process. Preserv. 2018, 42. [Google Scholar] [CrossRef]
- Klompong, V.; Benjakul, S.; Kantachote, D.; Shahidi, F. Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem. 2007. [Google Scholar] [CrossRef]
- Peighambardoust, S.H.; Tafti, A.G.; Hesari, J. Application of spray drying for preservation of lactic acid starter cultures: A review. Trends Food Sci. Technol. 2011, 22, 215–224. [Google Scholar] [CrossRef]
- Cumby, N.; Zhong, Y.; Naczk, M.; Shahidi, F. Antioxidant activity and water-holding capacity of canola protein hydrolysates. Food Chem. 2008. [Google Scholar] [CrossRef]
- Šližyte, R.; Mozuraityte, R.; Martínez-Alvarez, O.; Falch, E.; Fouchereau-Peron, M.; Rustad, T. Functional, bioactive and antioxidative properties of hydrolysates obtained from cod (Gadus morhua) backbones. Process Biochem. 2009. [Google Scholar] [CrossRef]
- Acquah, C.; Di Stefano, E.; Udenigwe, C.C. Role of hydrophobicity in food peptide functionality and bioactivity. J. Food Bioact. 2018, 4, 88–98. [Google Scholar] [CrossRef]
- Mundi, S.; Aluko, R.E. Inhibitory properties of kidney bean protein hydrolysate and its membrane fractions against renin, angiotensin converting enzyme, and free radicals. Austin J. Nutr. Food Sci. 2014, 2, 1008–1018. [Google Scholar]
- Mada, S.B.; Ugwu, C.P.; Abarshi, M.M. Health Promoting Effects of Food-Derived Bioactive Peptides: A Review. Int. J. Pept. Res. Ther. 2020, 26, 831–848. [Google Scholar] [CrossRef]
- Bhat, Z.F.; Kumar, S.; Bhat, H.F. Bioactive peptides of animal origin: A review. J. Food Sci. Technol. 2015, 52, 5377–5392. [Google Scholar] [CrossRef]
- Lorenzo, J.M.; Munekata, P.E.S.; Gómez, B.; Barba, F.J.; Mora, L.; Pérez-Santaescolástica, C.; Toldrá, F. Bioactive peptides as natural antioxidants in food products—A review. Trends Food Sci. Technol. 2018, 79, 136–147. [Google Scholar] [CrossRef]
- Sarmadi, B.H.; Ismail, A. Antioxidative peptides from food proteins: A review. Peptides 2010, 31, 1949–1956. [Google Scholar] [CrossRef]
- Liu, Z.; Dong, S.; Xu, J.; Zeng, M.; Song, H.; Zhao, Y. Production of cysteine-rich antimicrobial peptide by digestion of oyster (Crassostrea gigas) with alcalase and bromelin. Food Control 2008, 19, 231–235. [Google Scholar] [CrossRef]
- Xiong, S.; Yao, X.; Li, A. Antioxidant properties of peptide from cowpea seed. Int. J. Food Prop. 2013, 16, 1245–1256. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Shyu, Y.S.; Wang, Y.T.; Hsu, C.K. Antioxidative properties of protein hydrolysate from defatted peanut kernels treated with esperase. LWT—Food Sci. Technol. 2010, 43, 285–290. [Google Scholar] [CrossRef]
- Revilla, E.; Maria, C.S.; Miramontes, E.; Bautista, J.; García-Martínez, A.; Cremades, O.; Cert, R.; Parrado, J. Nutraceutical composition, antioxidant activity and hypocholesterolemic effect of a water-soluble enzymatic extract from rice bran. Food Res. Int. 2009, 42, 387–393. [Google Scholar] [CrossRef]
- Megías, C.; Pedroche, J.; Yust, M.M.; Girón-Calle, J.; Alaiz, M.; Millán, F.; Vioque, J. Production of copper-chelating peptides after hydrolysis of sunflower proteins with pepsin and pancreatin. LWT—Food Sci. Technol. 2008, 41, 1973–1977. [Google Scholar] [CrossRef]
- Xie, Z.; Huang, J.; Xu, X.; Jin, Z. Antioxidant activity of peptides isolated from alfalfa leaf protein hydrolysate. Food Chem. 2008. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Han, L.J.; Chen, L.J. In vitro antioxidant activity of protein hydrolysates prepared from corn gluten meal. J. Sci. Food Agric. 2008, 88, 1660–1666. [Google Scholar] [CrossRef]
- Qian, Z.J.; Jung, W.K.; Kim, S.K. Free radical scavenging activity of a novel antioxidative peptide purified from hydrolysate of bullfrog skin, Rana catesbeiana Shaw. Bioresour. Technol. 2008. [Google Scholar] [CrossRef] [PubMed]
- Je, J.Y.; Lee, K.H.; Lee, M.H.; Ahn, C.B. Antioxidant and antihypertensive protein hydrolysates produced from tuna liver by enzymatic hydrolysis. Food Res. Int. 2009, 42, 1266–1272. [Google Scholar] [CrossRef]
- López-Pedrouso, M.; Franco, D.; Serrano, M.P.; Maggiolino, A.; Landete-Castillejos, T.; De Palo, P.; Lorenzo, J.M. A proteomic-based approach for the search of biomarkers in Iberian wild deer (Cervus elaphus) as indicators of meat quality. J. Proteomics 2019, 205, 103422. [Google Scholar] [CrossRef] [PubMed]
- Sakanaka, S.; Tachibana, Y. Active oxygen scavenging activity of egg-yolk protein hydrolysates and their effects on lipid oxidation in beef and tuna homogenates. Food Chem. 2006, 95, 243–249. [Google Scholar] [CrossRef]
- Liu, J.R.; Chen, M.J.; Lin, C.W. Antimutagenic and antioxidant properties of milk-kefir and soymilk-kefir. J. Agric. Food Chem. 2005, 53, 2467–2474. [Google Scholar] [CrossRef] [PubMed]
- Sheih, I.C.; Wu, T.K.; Fang, T.J. Antioxidant properties of a new antioxidative peptide from algae protein waste hydrolysate in different oxidation systems. Bioresour. Technol. 2009, 100, 3419–3425. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.H.; Peng, J.; Zhen, D.W.; Chen, Z. Physicochemical and antioxidant properties of buckwheat (Fagopyrum esculentum Moench) protein hydrolysates. Food Chem. 2009, 115, 672–678. [Google Scholar] [CrossRef]
- Wang, L.; Mao, X.; Cheng, X.; Xiong, X.; Ren, F. Effect of enzyme type and hydrolysis conditions on the in vitro angiotensin I-converting enzyme inhibitory activity and ash content of hydrolysed whey protein isolate. Int. J. Food Sci. Technol. 2010. [Google Scholar] [CrossRef]
- Ambigaipalan, P.; Al-Khalifa, A.S.; Shahidi, F. Antioxidant and angiotensin I converting enzyme (ACE) inhibitory activities of date seed protein hydrolysates prepared using Alcalase, Flavourzyme and Thermolysin. J. Funct. Foods 2015. [Google Scholar] [CrossRef]
- Wang, K.; Luo, Q.; Hong, H.; Liu, H.; Luo, Y. Novel antioxidant and ACE inhibitory peptide identified from Arthrospira platensis protein and stability against thermal/pH treatments and simulated gastrointestinal digestion. Food Res. Int. 2021, 139, 109908. [Google Scholar] [CrossRef]
- Cermeño, M.; Connolly, A.; O’Keeffe, M.B.; Flynn, C.; Alashi, A.M.; Aluko, R.E.; FitzGerald, R.J. Identification of bioactive peptides from brewers’ spent grain and contribution of Leu/Ile to bioactive potency. J. Funct. Foods 2019, 60, 103455. [Google Scholar] [CrossRef]
- Wang, L.; Ma, M.; Yu, Z.; Du, S. Preparation and identification of antioxidant peptides from cottonseed proteins. Food Chem. 2021, 352, 129399. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.-L.; Ning, P.; Jiao, Y.; Xu, Z.; Cheng, Y.-H. Extraction of antioxidant peptides from rice dreg protein hydrolysate via an angling method. Food Chem. 2021, 337, 128069. [Google Scholar] [CrossRef]
- López-Pedrouso, M.; Borrajo, P.; Pateiro, M.; Lorenzo, J.M.; Franco, D. Antioxidant activity and peptidomic analysis of porcine liver hydrolysates using alcalase, bromelain, flavourzyme and papain enzymes. Food Res. Int. 2020, 137, 109389. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hao, X.; Zhang, X.; Zhang, G.; Li, X.; Liu, L.; Sun, Y.; Pan, Y. Identification of antioxidant peptides from cheddar cheese made with Lactobacillus helveticus. LWT 2021, 141, 110866. [Google Scholar] [CrossRef]
- Kudo, K.; Onodera, S.; Takeda, Y.; Benkeblia, N.; Shiomi, N. Antioxidative activities of some peptides isolated from hydrolyzed potato protein extract. J. Funct. Foods 2009, 1, 170–176. [Google Scholar] [CrossRef]
- Sompinit, K.; Lersiripong, S.; Reamtong, O.; Pattarayingsakul, W.; Patikarnmonthon, N.; Panbangred, W. In vitro study on novel bioactive peptides with antioxidant and antihypertensive properties from edible rhizomes. LWT 2020, 134, 110227. [Google Scholar] [CrossRef]
- Chai, T.-T.; Xiao, J.; Mohana Dass, S.; Teoh, J.-Y.; Ee, K.-Y.; Ng, W.-J.; Wong, F.-C. Identification of antioxidant peptides derived from tropical jackfruit seed and investigation of the stability profiles. Food Chem. 2021, 340, 127876. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, J.; Wang, Z.; Yao, Y.; Atungulu, G.G.; Ju, X.; Wang, L. Absorption and Metabolism of Peptide WDHHAPQLR Derived from Rapeseed Protein and Inhibition of HUVEC Apoptosis under Oxidative Stress. J. Agric. Food Chem. 2018, 66, 5178–5189. [Google Scholar] [CrossRef]
- Escudero, E.; Mora, L.; Toldrá, F. Stability of ACE inhibitory ham peptides against heat treatment and in vitro digestion. Food Chem. 2014, 161, 305–311. [Google Scholar] [CrossRef]
- Zhu, C.-Z.; Zhang, W.-G.; Zhou, G.-H.; Xu, X.-L. Identification of antioxidant peptides of Jinhua ham generated in the products and through the simulated gastrointestinal digestion system. J. Sci. Food Agric. 2016, 96, 99–108. [Google Scholar] [CrossRef]
- Suetsuna, K.; Ukeda, H.; Ochi, H. Isolation and characterization of free radical scavenging activities peptides derived from casein. J. Nutr. Biochem. 2000. [Google Scholar] [CrossRef]
- Hernández-Ledesma, B.; Dávalos, A.; Bartolomé, B.; Amigo, L. Preparation of Antioxidant Enzymatic Hydrolysates from α-Lactalbumin and β-Lactoglobulin. Identification of Active Peptides by HPLC-MS/MS. J. Agric. Food Chem. 2005, 53, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-X.; Wu, H.; Ling, Y.-F.; Lu, R.-R. Isolation and identification of antioxidant peptides derived from whey protein enzymatic hydrolysate by consecutive chromatography and Q-TOF MS. J. Dairy Res. 2013, 80, 367–373. [Google Scholar] [CrossRef]
- Wu, W.; He, L.; Liang, Y.; Yue, L.; Peng, W.; Jin, G.; Ma, M. Preparation process optimization of pig bone collagen peptide-calcium chelate using response surface methodology and its structural characterization and stability analysis. Food Chem. 2019, 284, 80–89. [Google Scholar] [CrossRef]
- Wattanasiritham, L.; Theerakulkait, C.; Wickramasekara, S.; Maier, C.S.; Stevens, J.F. Isolation and identification of antioxidant peptides from enzymatically hydrolyzed rice bran protein. Food Chem. 2016, 192, 156–162. [Google Scholar] [CrossRef]
- Nagpal, R.; Behare, P.; Rana, R.; Kumar, A.; Kumar, M.; Arora, S.; Morotta, F.; Jain, S.; Yadav, H. Bioactive peptides derived from milk proteins and their health beneficial potentials: An update. Food Funct. 2011, 2, 18–27. [Google Scholar] [CrossRef]
- Dullius, A.; Goettert, M.I.; de Souza, C.F.V. Whey protein hydrolysates as a source of bioactive peptides for functional foods—Biotechnological facilitation of industrial scale-up. J. Funct. Foods 2018, 42, 58–74. [Google Scholar] [CrossRef]
- Jiang, B.; Zhang, X.; Yuan, Y.; Qu, Y.; Feng, Z. Separation of Antioxidant Peptides from Pepsin Hydrolysate of Whey Protein Isolate by ATPS of EOPO Co-polymer (UCON)/Phosphate. Sci. Rep. 2017, 7, 13320. [Google Scholar] [CrossRef]
- Contreras, M.d.M.; Hernández-Ledesma, B.; Amigo, L.; Martín-Álvarez, P.J.; Recio, I. Production of antioxidant hydrolyzates from a whey protein concentrate with thermolysin: Optimization by response surface methodology. LWT—Food Sci. Technol. 2011, 44, 9–15. [Google Scholar] [CrossRef]
- Shazly, A.B.; He, Z.; El-Aziz, M.A.; Zeng, M.; Zhang, S.; Qin, F.; Chen, J. Fractionation and identification of novel antioxidant peptides from buffalo and bovine casein hydrolysates. Food Chem. 2017, 232, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Matin, A.; Otani, H. Cytotoxic and antibacterial activities of chemically synthesized κ-casecidin and its partial peptide fragments. J. Dairy Res. 2002, 69, 329. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Chouhan, V.S.; Sanghi, A.; Teotia, U.V.S. Antioxidative effect of yak milk caseinates hydrolyzed with three different proteases. Vet. World 2013. [Google Scholar] [CrossRef]
- Raikos, V.; Dassios, T. Health-promoting properties of bioactive peptides derived from milk proteins in infant food: A review. Dairy Sci. Technol. 2014, 94, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Ledesma, B.; García-Nebot, M.J.; Fernández-Tomé, S.; Amigo, L.; Recio, I. Dairy protein hydrolysates: Peptides for health benefits. Int. Dairy J. 2014, 38, 82–100. [Google Scholar] [CrossRef]
- Singh, B.P.; Vij, S.; Hati, S. Functional significance of bioactive peptides derived from soybean. Peptides 2014, 54, 171–179. [Google Scholar] [CrossRef]
- Chatterjee, C.; Gleddie, S.; Xiao, C.W. Soybean bioactive peptides and their functional properties. Nutrients 2018, 10, 1211. [Google Scholar] [CrossRef] [PubMed]
- Dan Ramdath, D.; Padhi, E.M.T.; Sarfaraz, S.; Renwick, S.; Duncan, A.M. Beyond the cholesterol-lowering effect of soy protein: A review of the effects of dietary soy and its constituents on risk factors for cardiovascular disease. Nutrients 2017, 9, 324. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.A.; Mullen, A.M.; O’Neill, E.; Drummond, L.; Álvarez, C. Opportunities and perspectives for utilisation of co-products in the meat industry. Meat Sci. 2018, 144, 62–73. [Google Scholar] [CrossRef]
- Damgaard, T.D.; Otte, J.A.; Meinert, L.; Jensen, K.; Lametsch, R. Antioxidant capacity of hydrolyzed porcine tissues. Food Sci. Nutr. 2014, 2, 282–288. [Google Scholar] [CrossRef]
- Damgaard, T.; Lametsch, R.; Otte, J. Antioxidant capacity of hydrolyzed animal by-products and relation to amino acid composition and peptide size distribution. J. Food Sci. Technol. 2015, 52, 6511–6519. [Google Scholar] [CrossRef]
- Pateiro, M.; Borrajo, P.; Campagnol, P.C.B.; Domínguez, R.; Tomasevic, I.; Munekata, P.E.S.; Barba, F.J.; Lorenzo, J.M. Extraction of valuable compounds from meat by-products. In Green Extraction and Valorization of By-Products from Food Processing; Barba, F.J., Roselló-Soto, E., Brncic, M., Lorenzo, J.M., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 55–90. ISBN 9781138544048. [Google Scholar]
- Sarbon, N.M.; Badii, F.; Howell, N.K. Preparation and characterisation of chicken skin gelatin as an alternative to mammalian gelatin. Food Hydrocoll. 2013, 30, 143–151. [Google Scholar] [CrossRef]
- O’Sullivan, S.M.; Lafarga, T.; Hayes, M.; O’Brien, N.M. Bioactivity of bovine lung hydrolysates prepared using papain, pepsin, and Alcalase. J. Food Biochem. 2017, 41, e12406. [Google Scholar] [CrossRef]
- Borrajo, P.; Pateiro, M.; Gagaoua, M.; Franco, D.; Zhang, W.; Lorenzo, J.M. Evaluation of the antioxidant and antimicrobial activities of porcine liver protein hydrolysates obtained using Alcalase, Bromelain, and Papain. Appl. Sci. 2020, 10, 2290. [Google Scholar] [CrossRef]
- Mullen, A.M.; Álvarez, C.; Zeugolis, D.I.; Henchion, M.; O’Neill, E.; Drummond, L. Alternative uses for co-products: Harnessing the potential of valuable compounds from meat processing chains. Meat Sci. 2017, 132, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Ucak, I.; Afreen, M.; Montesano, D.; Carrillo, C.; Tomasevic, I.; Simal-Gandara, J.; Barba, F.J. Functional and Bioactive Properties of Peptides Derived from Marine Side Streams. Mar. Drugs 2021, 19, 71. [Google Scholar] [CrossRef]
- Rajapakse, N.; Mendis, E.; Jung, W.K.; Je, J.Y.; Kim, S.K. Purification of a radical scavenging peptide from fermented mussel sauce and its antioxidant properties. Food Res. Int. 2005, 38, 175–182. [Google Scholar] [CrossRef]
- Mendis, E.; Rajapakse, N.; Byun, H.G.; Kim, S.K. Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 2005, 77, 2166–2178. [Google Scholar] [CrossRef]
- Hsu, K.C. Purification of antioxidative peptides prepared from enzymatic hydrolysates of tuna dark muscle by-product. Food Chem. 2010, 122, 42–48. [Google Scholar] [CrossRef]
- Thiansilakul, Y.; Benjakul, S.; Shahidi, F. Antioxidative activity of protein hydrolysate from round scad muscle using alcalase and flavourzyme. J. Food Biochem. 2007. [Google Scholar] [CrossRef]
- Kim, S.Y.; Je, J.Y.; Kim, S.K. Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J. Nutr. Biochem. 2007, 18, 31–38. [Google Scholar] [CrossRef]
- Jo, H.Y.; Jung, W.K.; Kim, S.K. Purification and characterization of a novel anticoagulant peptide from marine echiuroid worm, Urechis unicinctus. Process Biochem. 2008. [Google Scholar] [CrossRef]
- Jung, W.K.; Kim, S.K. Isolation and characterisation of an anticoagulant oligopeptide from blue mussel, Mytilus edulis. Food Chem. 2009, 117, 687–692. [Google Scholar] [CrossRef]
- Joshi, I.; K, J.; Noorani K, P.M.; Nazeer, R.A. Isolation and characterization of angiotensin I-converting enzyme (ACE-I) inhibition and antioxidant peptide from by-catch shrimp (Oratosquilla woodmasoni) waste. Biocatal. Agric. Biotechnol. 2020, 29, 101770. [Google Scholar] [CrossRef]
- Heo, S.-J.; Park, E.-J.; Lee, K.-W.; Jeon, Y.-J. Antioxidant activities of enzymatic extracts from brown seaweeds. Bioresour. Technol. 2005, 96, 1613–1623. [Google Scholar] [CrossRef] [PubMed]
- Harnedy, P.A.; O’Keeffe, M.B.; FitzGerald, R.J. Fractionation and identification of antioxidant peptides from an enzymatically hydrolysed Palmaria palmata protein isolate. Food Res. Int. 2017. [Google Scholar] [CrossRef]
- Esfandi, R.; Walters, M.E.; Tsopmo, A. Antioxidant properties and potential mechanisms of hydrolyzed proteins and peptides from cereals. Heliyon 2019, 5, e01538. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Du, M.; Shen, M.; Wu, T.; Lin, L. Physico-chemical properties, antioxidant activities and angiotensin-I converting enzyme inhibitory of protein hydrolysates from Mung bean (Vigna radiate). Food Chem. 2019, 270, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Görgüç, A.; Bircan, C.; Yılmaz, F.M. Sesame bran as an unexploited by-product: Effect of enzyme and ultrasound-assisted extraction on the recovery of protein and antioxidant compounds. Food Chem. 2019, 283, 637–645. [Google Scholar] [CrossRef]
- Zou, Z.; Wang, M.; Wang, Z.; Aluko, R.E.; He, R. Antihypertensive and antioxidant activities of enzymatic wheat bran protein hydrolysates. J. Food Biochem. 2020, 44, e13090. [Google Scholar] [CrossRef] [PubMed]
- Girgih, A.T.; He, R.; Malomo, S.; Offengenden, M.; Wu, J.; Aluko, R.E. Structural and functional characterization of hemp seed (Cannabis sativa L.) protein-derived antioxidant and antihypertensive peptides. J. Funct. Foods 2014, 6, 384–394. [Google Scholar] [CrossRef]
- Rho, S.J.; Lee, J.S.; Chung, Y.I.; Kim, Y.W.; Lee, H.G. Purification and identification of an angiotensin I-converting enzyme inhibitory peptide from fermented soybean extract. Process Biochem. 2009, 44, 490–493. [Google Scholar] [CrossRef]
- Ahmad, I.; Yanuar, A.; Mulia, K.; Mun’Im, A. Review of angiotensin-converting enzyme inhibitory assay: Rapid method in drug discovery of herbal plants. Pharmacogn. Rev. 2017, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Balasuriya, B.W.N.; Rupasinghe, H.P.V. Plant flavonoids as angiotensin converting enzyme inhibitors in regulation of hypertension. Funct. Foods Heal. Dis. 2011, 1, 172–188. [Google Scholar] [CrossRef]
- Matsui, T.; Tanaka, M. Antihypertensive Peptides and Their Underlying Mechanisms. In Bioactive Proteins and Peptides as Functional Foods and Nutraceuticals; Mine, Y., Li-Chan, E., Jiang, B., Eds.; Blackwell Publishing Ltd.: Ames, IA, USA, 2010; pp. 43–54. [Google Scholar]
- Daskaya-Dikmen, C.; Yucetepe, A.; Karbancioglu-Guler, F.; Daskaya, H.; Ozcelik, B. Angiotensin-I-Converting Enzyme (ACE)-Inhibitory Peptides from Plants. Nutrients 2017, 9, 316. [Google Scholar] [CrossRef]
- Kaur, A.; Kehinde, B.A.; Sharma, P.; Sharma, D.; Kaur, S. Recently isolated food-derived antihypertensive hydrolysates and peptides: A review. Food Chem. 2021, 346, 128719. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Wan, J.; Le, G.; Shi, Y. Novel angiotensin I-converting enzyme inhibitory peptides isolated from Alcalase hydrolysate of mung bean protein. J. Pept. Sci. Off. Publ. Eur. Pept. Soc. 2006, 12, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Sonklin, C.; Alashi, M.A.; Laohakunjit, N.; Kerdchoechuen, O.; Aluko, R.E. Identification of antihypertensive peptides from mung bean protein hydrolysate and their effects in spontaneously hypertensive rats. J. Funct. Foods 2020, 64, 103635. [Google Scholar] [CrossRef]
- Connolly, A.; O’Keeffe, M.B.; Piggott, C.O.; Nongonierma, A.B.; FitzGerald, R.J. Generation and identification of angiotensin converting enzyme (ACE) inhibitory peptides from a brewers’ spent grain protein isolate. Food Chem. 2015, 176, 64–71. [Google Scholar] [CrossRef]
- Abdel-Hamid, M.; Otte, J.; De Gobba, C.; Osman, A.; Hamad, E. Angiotensin I-converting enzyme inhibitory activity and antioxidant capacity of bioactive peptides derived from enzymatic hydrolysis of buffalo milk proteins. Int. Dairy J. 2017, 66, 91–98. [Google Scholar] [CrossRef]
- Salampessy, J.; Reddy, N.; Kailasapathy, K.; Phillips, M. Functional and potential therapeutic ACE-inhibitory peptides derived from bromelain hydrolysis of trevally proteins. J. Funct. Foods 2015, 14, 716–725. [Google Scholar] [CrossRef]
- So, P.B.T.; Rubio, P.; Lirio, S.; Macabeo, A.P.; Huang, H.-Y.; Corpuz, M.J.-A.T.; Villaflores, O.B. In vitro angiotensin I converting enzyme inhibition by a peptide isolated from Chiropsalmus quadrigatus Haeckel (box jellyfish) venom hydrolysate. Toxicon 2016, 119, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, A.; Vázquez, A. Bioactive peptides: A review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Ganguly, A.; Sharma, K.; Majumder, K. Food-Derived Bioactive Peptides and Their Role in Ameliorating Hypertension and Associated Cardiovascular Diseases. In Advances in Food and Nutrition Research; Toldrá, F., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 89, pp. 165–207. ISBN 978-0-12-817171-4. [Google Scholar]
- Muguerza, B.; Ramos, M.; Sánchez, E.; Manso, M.A.; Miguel, M.; Aleixandre, A.; Delgado, M.A.; Recio, I. Antihypertensive activity of milk fermented by Enterococcus faecalis strains isolated from raw milk. Int. Dairy J. 2006. [Google Scholar] [CrossRef]
- Ibrahim, H.R.; Ahmed, A.S.; Miyata, T. Novel angiotensin-converting enzyme inhibitory peptides from caseins and whey proteins of goat milk. J. Adv. Res. 2017, 8, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Parmar, H.; Hati, S.; Sakure, A. In Vitro and In Silico Analysis of Novel ACE-Inhibitory Bioactive Peptides Derived from Fermented Goat Milk. Int. J. Pept. Res. Ther. 2018, 24, 441–453. [Google Scholar] [CrossRef]
- Haque, E.; Chand, R. Antihypertensive and antimicrobial bioactive peptides from milk proteins. Eur. Food Res. Technol. 2008. [Google Scholar] [CrossRef]
- Daliri, E.B.M.; Lee, B.H.; Park, B.J.; Kim, S.H.; Oh, D.H. Antihypertensive peptides from whey proteins fermented by lactic acid bacteria. Food Sci. Biotechnol. 2018, 27, 1781–1789. [Google Scholar] [CrossRef]
- Gonzalez-Gonzalez, C.R.; Machado, J.; Correia, S.; McCartney, A.L.; Stephen Elmore, J.; Jauregi, P. Highly proteolytic bacteria from semi-ripened Chiapas cheese elicit angiotensin-I converting enzyme inhibition and antioxidant activity. LWT 2019, 111, 449–456. [Google Scholar] [CrossRef]
- Panayotova, T.; Pashova-Baltova, K.; Dimitrov, Z. Production of ACE-inhibitory peptides in milk fermented with selected lactic acid bacteria. J. BioSci. Biotechnol. 2018, 7, 31–37. [Google Scholar]
- Gómez-Ruiz, J.A.; Recio, I.; Belloque, J. ACE-Inhibitory Activity and Structural Properties of Peptide Asp-Lys-Ile-His-Pro [β-CN f(47−51)]. Study of the Peptide Forms Synthesized by Different Methods. J. Agric. Food Chem. 2004, 52, 6315–6319. [Google Scholar] [CrossRef]
- Mora, L.; Gallego, M.; Reig, M.; Toldrá, F. Challenges in the quantitation of naturally generated bioactive peptides in processed meats. Trends Food Sci. Technol. 2017, 69, 306–314. [Google Scholar] [CrossRef]
- Escudero, E.; Aristoy, M.-C.; Nishimura, H.; Arihara, K.; Toldrá, F. Antihypertensive effect and antioxidant activity of peptide fractions extracted from Spanish dry-cured ham. Meat Sci. 2012, 91, 306–311. [Google Scholar] [CrossRef]
- Escudero, E.; Mora, L.; Fraser, P.D.; Aristoy, M.-C.; Arihara, K.; Toldrá, F. Purification and Identification of antihypertensive peptides in Spanish dry-cured ham. J. Proteomics 2013, 78, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Dellafiora, L.; Paolella, S.; Dall’Asta, C.; Dossena, A.; Cozzini, P.; Galaverna, G. Hybrid in Silico/in vitro Approach for the Identification of Angiotensin I Converting Enzyme Inhibitory Peptides from Parma Dry-Cured Ham. J. Agric. Food Chem. 2015, 63, 6366–6375. [Google Scholar] [CrossRef] [PubMed]
- Gu, R.-Z.; Liu, W.-Y.; Lin, F.; Jin, Z.-T.; Chen, L.; Yi, W.-X.; Lu, J.; Cai, M.-Y. Antioxidant and angiotensin I-converting enzyme inhibitory properties of oligopeptides derived from black-bone silky fowl (Gallus gallus domesticus Brisson) muscle. Food Res. Int. 2012, 49, 326–333. [Google Scholar] [CrossRef]
- Choe, J.; Seol, K.-H.; Kim, H.-J.; Hwang, J.-T.; Lee, M.; Jo, C. Isolation and identification of angiotensin I-converting enzyme inhibitory peptides derived from thermolysin-injected beef M. longissimus. Asian-Australas J. Anim. Sci. 2019, 32, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Qian, Z.J.; Kim, S.K. A novel angiotensin I converting enzyme inhibitory peptide from tuna frame protein hydrolysate and its antihypertensive effect in spontaneously hypertensive rats. Food Chem. 2010. [Google Scholar] [CrossRef]
- Gildberg, A.; Arnesen, J.A.; Sæther, B.-S.; Rauø, J.; Stenberg, E. Angiotensin I-converting enzyme inhibitory activity in a hydrolysate of proteins from Northern shrimp (Pandalus borealis) and identification of two novel inhibitory tri-peptides. Process Biochem. 2011, 46, 2205–2209. [Google Scholar] [CrossRef]
- Lee, S.Y.; Hur, S.J. Antihypertensive peptides from animal products, marine organisms, and plants. Food Chem. 2017, 228, 506–517. [Google Scholar] [CrossRef]
- Vieira, E.F.; Ferreira, I.M. Antioxidant and antihypertensive hydrolysates obtained from by-products of cannery sardine and brewing industries. Int. J. Food Prop. 2017, 20, 662–673. [Google Scholar] [CrossRef]
- Tian, L.; Liu, J.; Ma, L.; Zhang, L.; Wang, S.; Yan, E.; Zhu, H. Isolation and Purification of Antioxidant and ACE-Inhibitory Peptides from Yak (Bos grunniens) Skin. J. Food Process. Preserv. 2017, 41, e13123. [Google Scholar] [CrossRef]
- Nazeer, R.A.; Deeptha, R. Antioxidant Activity and Amino Acid Profiling of Protein Hydrolysates from the Skin of Sphyraena barracuda and Lepturacanthus savala. Int. J. Food Prop. 2013, 16, 500–511. [Google Scholar] [CrossRef][Green Version]
- Connolly, A.; O’Keeffe, M.B.; Nongonierma, A.B.; Piggott, C.O.; FitzGerald, R.J. Isolation of peptides from a novel brewers spent grain protein isolate with potential to modulate glycaemic response. Int. J. Food Sci. Technol. 2017, 52, 146–153. [Google Scholar] [CrossRef]
- Alashi, A.M.; Blanchard, C.L.; Mailer, R.J.; Agboola, S.O.; Mawson, A.J.; He, R.; Malomo, S.A.; Girgih, A.T.; Aluko, R.E. Blood pressure lowering effects of Australian canola protein hydrolysates in spontaneously hypertensive rats. Food Res. Int. 2014, 55, 281–287. [Google Scholar] [CrossRef]
- Karami, Z.; Akbari-adergani, B. Bioactive food derived peptides: A review on correlation between structure of bioactive peptides and their functional properties. J. Food Sci. Technol. 2019, 56, 535–547. [Google Scholar] [CrossRef]
- Wu, J.; Aluko, R.E.; Nakai, S. Structural Requirements of Angiotensin I-Converting Enzyme Inhibitory Peptides: Quantitative Structure-Activity Relationship Modeling of Peptides Containing 4-10 Amino Acid Residues. QSAR Comb. Sci. 2006, 25, 873–880. [Google Scholar] [CrossRef]
- Wergedahl, H.; Liaset, B.; Gudbrandsen, O.A.; Lied, E.; Espe, M.; Muna, Z.; Mørk, S.; Berge, R.K. Fish protein hydrolysate reduces plasma total cholesterol, increases the proportion of HDL cholesterol, and lowers Acyl-CoA:cholesterol acyltransferase activity in liver of Zucker rats. J. Nutr. 2004. [Google Scholar] [CrossRef]
- Pak, V.V.; Koo, M.; Lee, N.; Kim, M.S.; Kwon, D.Y. Structure—Activity relationships of the peptide Ile-Ala-Val-Pro and its derivatives revealed using the semi-empirical AM1 method. Chem. Nat. Compd. 2005. [Google Scholar] [CrossRef]
- Zhong, F.; Zhang, X.; Ma, J.; Shoemaker, C.F. Fractionation and identification of a novel hypocholesterolemic peptide derived from soy protein Alcalase hydrolysates. Food Res. Int. 2007, 40, 756–762. [Google Scholar] [CrossRef]
- Aiello, G.; Ferruzza, S.; Ranaldi, G.; Sambuy, Y.; Arnoldi, A.; Vistoli, G.; Lammi, C. Behavior of three hypocholesterolemic peptides from soy protein in an intestinal model based on differentiated Caco-2 cell. J. Funct. Foods 2018, 45, 363–370. [Google Scholar] [CrossRef]
- Wakasa, Y.; Tamakoshi, C.; Ohno, T.; Hirose, S.; Goto, T.; Nagaoka, S.; Takaiwa, F. The Hypocholesterolemic Activity of Transgenic Rice Seed Accumulating Lactostatin, a Bioactive Peptide Derived from Bovine Milk β-Lactoglobulin. J. Agric. Food Chem. 2011, 59, 3845–3850. [Google Scholar] [CrossRef]
- Nagaoka, S.; Nakamura, A.; Shibata, H.; Kanamaru, Y. Soystatin (VAWWMY), a Novel Bile Acid-Binding Peptide, Decreased Micellar Solubility and Inhibited Cholesterol Absorption in Rats. Biosci. Biotechnol. Biochem. 2010, 74, 1738–1741. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Tsai, J.-S.; Chen, G.-W. Purification and identification of hypocholesterolemic peptides from freshwater clam hydrolysate with in vitro gastrointestinal digestion. J. Food Biochem. 2017, 41, e12385. [Google Scholar] [CrossRef]
- Nagaoka, S.; Futamura, Y.; Miwa, K.; Awano, T.; Yamauchi, K.; Kanamaru, Y.; Tadashi, K.; Tamotsu, K. Identification of Novel Hypocholesterolemic Peptides Derived from Bovine Milk β-Lactoglobulin. Biochem. Biophys. Res. Commun. 2001, 281, 11–17. [Google Scholar] [CrossRef]
- Morikawa, K.; Ishikawa, K.; Kanamaru, Y.; Hori, G.; Nagaoka, S. Effects of Dipeptides Having a C-Terminal Lysine on the Cholesterol 7α-Hydroxylase mRNA Level in HepG2 Cells. Biosci. Biotechnol. Biochem. 2007, 71, 821–825. [Google Scholar] [CrossRef]
- Nagaoka, S. Structure–function properties of hypolipidemic peptides. J. Food Biochem. 2019, 43, e12539. [Google Scholar] [CrossRef]
- Katsuda, S.; Ito, M.; Waseda, Y.; Morimatsu, F.; Taguchi, Y.; Hasegawa, M.; Takaichi, S.; Yamada, R.; Furukawa, Y.; Shimizu, T. Papain-Hydrolyzed Pork Meat Reduces Serum Cholesterol Level and Premature Atherosclerosis in Dietary-Induced Hypercholesterolemic Rabbits. J. Nutr. Sci. Vitaminol. 2000, 46, 180–187. [Google Scholar] [CrossRef]
- Wang, Y.; Jones, P.J.H.; Ausman, L.M.; Lichtenstein, A.H. Soy protein reduces triglyceride levels and triglyceride fatty acid fractional synthesis rate in hypercholesterolemic subjects. Atherosclerosis 2004, 173, 269–275. [Google Scholar] [CrossRef]
- Pak, V.V.; Koo, M.S.; Kasymova, T.D.; Kwon, D.Y. Isolation and Identification of Peptides from Soy 11S-Globulin with Hypocholesterolemic Activity. Chem. Nat. Compd. 2005, 41, 710–714. [Google Scholar] [CrossRef]
- Coelho, M.S.; Soares-Freitas, R.A.M.; Arêas, J.A.G.; Gandra, E.A.; Salas-Mellado, M.d.l.M. Peptides from Chia Present Antibacterial Activity and Inhibit Cholesterol Synthesis. Plant Foods Hum. Nutr. 2018, 73, 101–107. [Google Scholar] [CrossRef]
- Maestri, E.; Marmiroli, M.; Marmiroli, N. Bioactive peptides in plant-derived foodstuffs. J. Proteomics 2016, 147, 140–155. [Google Scholar] [CrossRef] [PubMed]
- Marques, M.R.; Fontanari, G.G.; Pimenta, D.C.; Soares-Freitas, R.M.; Arêas, J.A.G. Proteolytic hydrolysis of cowpea proteins is able to release peptides with hypocholesterolemic activity. Food Res. Int. 2015, 77, 43–48. [Google Scholar] [CrossRef]
- Ulug, S.K.; Jahandideh, F.; Wu, J. Novel technologies for the production of bioactive peptides. Trends Food Sci. Technol. 2021, 108, 27–39. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, R.; Rana, S. Bioactive peptides: A review. Int. J. Bioautomation 2011, 15, 223–250. [Google Scholar]
- Raveschot, C.; Cudennec, B.; Coutte, F.; Flahaut, C.; Fremont, M.; Drider, D.; Dhulster, P. Production of bioactive peptides by lactobacillus species: From gene to application. Front. Microbiol. 2018, 9, 2354. [Google Scholar] [CrossRef] [PubMed]
- Hafeez, Z.; Cakir-Kiefer, C.; Roux, E.; Perrin, C.; Miclo, L.; Dary-Mourot, A. Strategies of producing bioactive peptides from milk proteins to functionalize fermented milk products. Food Res. Int. 2014, 63, 71–80. [Google Scholar] [CrossRef]
- Przybylski, R.; Firdaous, L.; Châtaigné, G.; Dhulster, P.; Nedjar, N. Production of an antimicrobial peptide derived from slaughterhouse by-product and its potential application on meat as preservative. Food Chem. 2016, 211, 306–313. [Google Scholar] [CrossRef]
- da Silva Malheiros, P.; Daroit, D.J.; Brandelli, A. Food applications of liposome-encapsulated antimicrobial peptides. Trends Food Sci. Technol. 2010, 21, 284–292. [Google Scholar] [CrossRef]
- Alvarado, Y.; Muro, C.; Illescas, J.; Díaz, M.d.C.; Riera, F. Encapsulation of antihypertensive peptides from whey proteins and their releasing in gastrointestinal conditions. Biomolecules 2019, 9, 164. [Google Scholar] [CrossRef]
- Alvarado Pérez, Y.; Muro Urista, C.; Maciel Cerda, A.; Álvarez Sánchez, J.; Riera Rodríguez, F. Antihypertensive and Antioxidant Properties from Whey Protein Hydrolysates Produced by Encapsulated Bacillus subtilis Cells. Int. J. Pept. Res. Ther. 2019. [Google Scholar] [CrossRef]
- McClements, D.J. Encapsulation, protection, and delivery of bioactive proteins and peptides using nanoparticle and microparticle systems: A review. Adv. Colloid Interface Sci. 2018, 253, 1–22. [Google Scholar] [CrossRef]
- Mohan, A.; Rajendran, S.R.C.K.; He, Q.S.; Bazinet, L.; Udenigwe, C.C. Encapsulation of food protein hydrolysates and peptides: A review. RSC Adv. 2015, 5, 79270–79278. [Google Scholar] [CrossRef]
- Liu, L.; Li, S.; Zheng, J.; Bu, T.; He, G.; Wu, J. Safety considerations on food protein-derived bioactive peptides. Trends Food Sci. Technol. 2020, 96, 199–207. [Google Scholar] [CrossRef]
- Lafarga, T.; Hayes, M. Bioactive protein hydrolysates in the functional food ingredient industry: Overcoming current challenges. Food Rev. Int. 2017, 33, 217–246. [Google Scholar] [CrossRef]

| Protein Source | Enzyme | Peptide Amino Acid Sequence | Ref. |
|---|---|---|---|
| Arthrospira platensis protein | Alcalase | VTAGLVGGGAGK | [39] |
| Brewers’ spent grain | Alcalase | IPY and LPY | [40] |
| Cotton seed | Alcalase | YSNQNGRF | [41] |
| Rice dreg protein | Alcalase | GDMNP and LLLRW | [42] |
| Porcine liver | Alcalase | ALFQDVQKPSQDEWGK, APAAIGPYSQAVLVDR, FLANVSTVLTSK, FLEQQNQVLQTK, LGEHNIDVLEGNEQFINAAK, REATQPEVDTTLGR | [32] |
| Porcine liver | Alcalase, bromelain, flavourzyme, and papain | ALFQDVQKPSQDEWGK, APAAIGPYSQAVLVDR, GLNQALVDLHALGSAR, LSGPQAGLGEYLFER | [43] |
| Cheddar cheese | Microbial protease (Lactobacillus helveticus) | EMPFPK, KEMPFPK, SDIPNPIGSENSEK | [44] |
| Potato waste | Pancreatin and Amano-P | FGER, FDRR, FGERR | [45] |
| Rhizome proteins of ginger | Pepsin and trypsin | VTYM | [46] |
| Wheat germ | Pepsin | KELPPSDADW | [3] |
| Jackfruit seed | Trypsin | VGPWQK | [47] |
| Rapeseed | Alkali protease | WDHHAPQLR | [48] |
| Myosin, Spanish dry-cured ham | Pancreatin | SNAAC | [49] |
| Jinhua dry-cured ham | Pepsin-trypsin | LPGGGHGDL, LPGGGT, KEER | [50] |
| Bovine casein | pepsin | YFYPEL | [51] |
| β-lactoglobulin | Corolase PP | WYSLAMAASDI | [52] |
| Whey protein concentrate | Alcalase | WYSL | [53] |
| Pig bone collagen | Alcalase and neutrase | AGPAGPAGAR, AGPHGPPGKDGR, GPAGPHGPPGKDGR | [54] |
| Rice bran | papain and trypsin | VAGAEDAAK, AAVQGQVEK, GGHELSK, CQHHHDQWK | [55] |
| Protein Source | Enzyme | Peptide Amino Acid Sequence | Ref. |
|---|---|---|---|
| Mung bean | Alcalase | KDYRL, VTPALR, KLPAGTLF | [98] |
| Brewers’ spent grain | ILDL, and ILLPGAQDGL | [40] | |
| Mung bean | Bromelain | LPRL, YADLVE, LRLESF, HLNVVHEN, and PGSGCAGTDL | [99] |
| Brewers’ spent grain | Flavourzyme | IPLQP and LPLQP | [100] |
| Rhizome proteins of turmeric | Pepsin and trypsin | CGVGAA, DVDP, and CACGGV | [46] |
| Rhizome proteins of ginger | VTYM | [46] | |
| Wheat germ | Proteinase K | SGGSYADELVSTAK | [2] |
| Arthrospira platensis protein | Trypsin | PTGNPLSP | [39] |
| β-lactoglobulin | Thermolysin | LDTDYKK | [59] |
| Buffaloe skim milk | Papain, pepsin, and trypsin | FPGPIPK IPPK QPPQ | [101] |
| Trevally (Pseudocaranx sp.) | Bromelain | AR, AV, and APER | [102] |
| Haeckel (box jellyfish) venom | Pepsin | ACPGPNPGRP | [103] |
| Protein Source | Enzyme | Peptide Amino Acid Sequence | Ref. |
|---|---|---|---|
| Soy protein | Alcalase | WGAPSL | [132] |
| Trypsin and pepsin | IAVPGEVA, IAVPTGVA, and LPYP | [133] | |
| Bovine milk β-lactoglobulin | Trypsin | IIAEK | [134] |
| Soybean glycinin | VAWWMY | [135] | |
| Freshwater clam (Corbicula fluminea) | Protamex | VKP, VKK | [136] |
| Soybean | YVVNPDNDEN, YVVNPDNNEN | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peighambardoust, S.H.; Karami, Z.; Pateiro, M.; Lorenzo, J.M. A Review on Health-Promoting, Biological, and Functional Aspects of Bioactive Peptides in Food Applications. Biomolecules 2021, 11, 631. https://doi.org/10.3390/biom11050631
Peighambardoust SH, Karami Z, Pateiro M, Lorenzo JM. A Review on Health-Promoting, Biological, and Functional Aspects of Bioactive Peptides in Food Applications. Biomolecules. 2021; 11(5):631. https://doi.org/10.3390/biom11050631
Chicago/Turabian StylePeighambardoust, Seyed Hadi, Zohreh Karami, Mirian Pateiro, and José M. Lorenzo. 2021. "A Review on Health-Promoting, Biological, and Functional Aspects of Bioactive Peptides in Food Applications" Biomolecules 11, no. 5: 631. https://doi.org/10.3390/biom11050631
APA StylePeighambardoust, S. H., Karami, Z., Pateiro, M., & Lorenzo, J. M. (2021). A Review on Health-Promoting, Biological, and Functional Aspects of Bioactive Peptides in Food Applications. Biomolecules, 11(5), 631. https://doi.org/10.3390/biom11050631









