Anti-Angiogenic Effects of Phytochemicals on miRNA Regulating Breast Cancer Progression
Abstract
1. Introduction
2. Tumor Angiogenesis
2.1. Angiogenic Signaling in EC
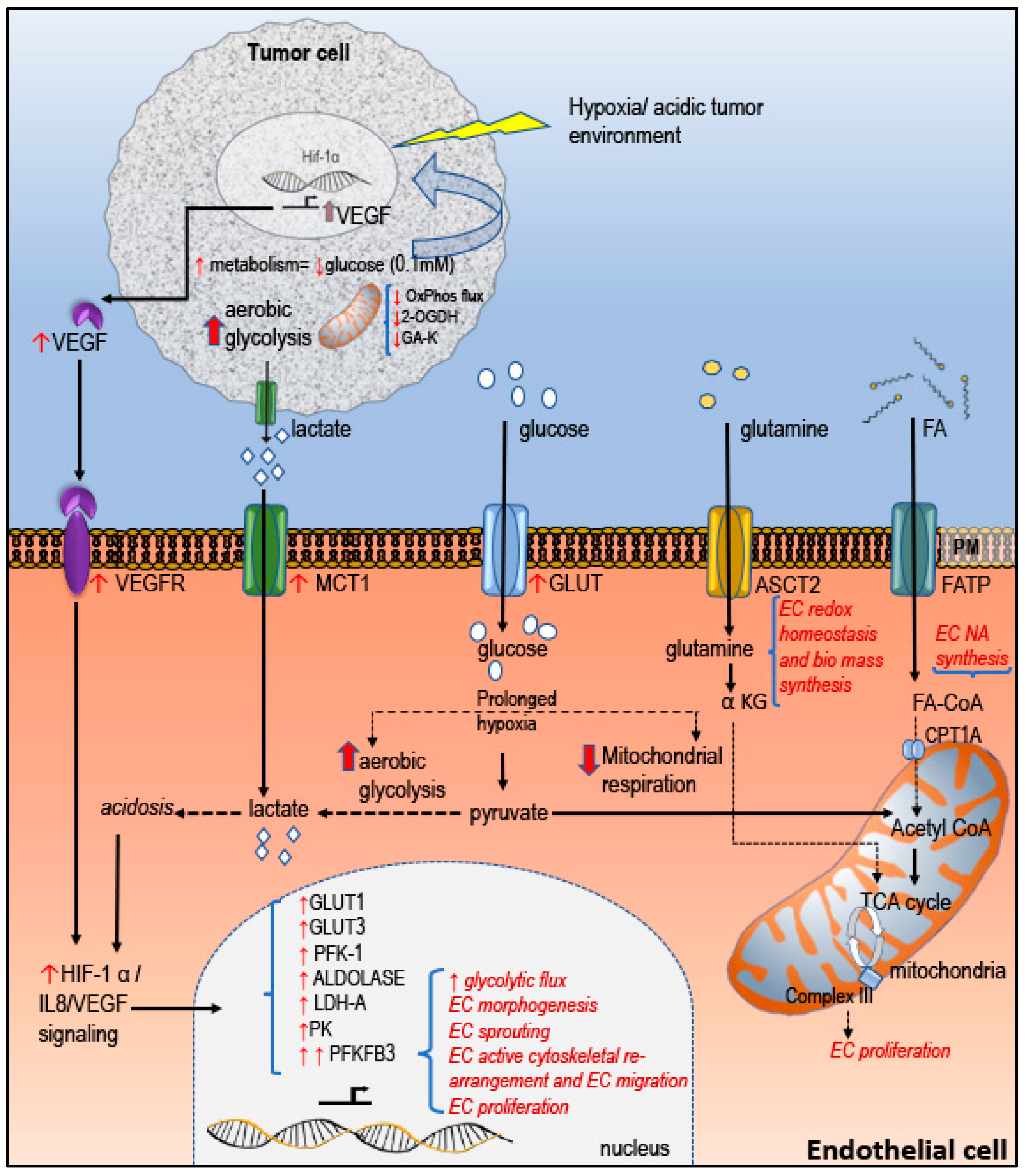
2.2. Tumor EC Metabolism in Regulating Tumor Angiogenesis
3. Central Role of miRNA in the Regulation of Tumor Angiogenesis: The Role of Phytochemicals
3.1. miRNA in EC VEGF Signaling
Plant Compounds Targeting VEGF Regualting miRNA
3.2. AngiomiRs in EC Metabolism
Plant Compounds Targeting Metabolism Regulating miRNA
3.3. AngiomiRs Responsive to Oxygen Level
Plant Compounds Targeting HIF-1α Regualting miRNA
3.4. ROS Sensing miRNAs and Tumor Angiogenesis
Plant Compounds Targeting ROS Sensing miRNAs
4. Clinical significance of miRNA in BC
5. Discussion and Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALK4 | Activin receptor-like kinase 4 |
| ASCT2 | System ASC amino acid transporters 2 |
| BACH1 | BTB Domain And CNC Homolog 1 |
| bFGF | Basic fibroblast growth factor |
| BM-MSC | Human bone marrow-derived mesenchymal stem cell |
| CPT1A | Carnitine palmitoyl transferase 1A |
| CXCL8 | Chemokine (C-X-C motif) ligand 8 |
| DNMT | DNA methyltransferase |
| EMT | Epithelial–mesenchymal transition |
| EPC | Bone marrow–derived endothelial progenitor cells |
| FA | Fatty acid |
| FASN | Fatty acid synthase |
| FATP | Fatty acid transporter protein |
| FGF2 | Fibroblast grow factor-2 |
| FZD | Frizzled receptor |
| G6PD | Glucose-6-phosphate dehydrogenase |
| GPCR | G-protein-coupled receptor |
| HIF | Hypoxia inducible factor |
| IDC | Infiltrating ductal carcinoma |
| IGF-I | Insulin-like growth factor I |
| IRS1 | Insulin receptor substrate 1 |
| LAT1 | L-type amino acid transporters 1 |
| LATS2 | Large tumor suppressor, homology 2 |
| LDHA/B | Lactate dehydrogenase A and B |
| Let-7a | Lethal-7a |
| LZTFL1 | Leucine zipper transcription factor-like 1 |
| MLC II | Myosin light chain II |
| MMP11 | Matrix metalloproteinase 11 |
| MMP9 | Matrix metalloproteinase 9 |
| MTDH | Metadherin |
| NgBR | Nogo-B receptor |
| NO | Nitric oxide |
| PDCD4 | Protein programmed cell death 4 |
| PFKFB3 | Phosphofructokinase-2/fructose-2,6-bisphosphatase |
| TIMP2 | Tissue inhibitor of metalloproteinases 2 |
| TME | Tumor microenvironment |
| TNFα | Tumor necrosis factor α |
| VASH1 | Vasohibin-1 |
| WNK1 | WNK lysine deficient protein kinase 1 |
| ZO-1 | Zonula occludens 1 |
| α KG | α keto glutarate |
References
- Engels, K.; Fox, S.B.; Whitehouse, R.M.; Gatter, K.C.; Harris, A.L. Distinct angiogenic patterns are associated with high-grade in situ ductal carcinomas of the breast. J. Pathol. 1997, 181, 207–212. [Google Scholar] [CrossRef]
- Bosari, S.; Lee, A.K.C.; DeLellis, R.A.; Wiley, B.D.; Heatley, G.J.; Silverman, M.L. Microvessel quantitation and prognosis in invasive breast carcinoma. Hum. Pathol. 1992, 23, 755–761. [Google Scholar] [CrossRef]
- Guidi, A.J.; Fischer, L.; Harris, J.R.; Schnitt, S.J. Microvessel Density and Distribution in Ductal Carcinoma In Situ of the Breast. J. Natl. Cancer Inst. 1994, 86, 614–619. [Google Scholar] [CrossRef]
- Brem, S.S.; Jensen, H.M.; Gullino, P.M. Angiogenesis as a marker of preneoplastic lesions of the human breast. Cancer 1978, 41, 239–244. [Google Scholar] [CrossRef]
- Dirkx, A.E.M.; oude Egbrink, M.G.A.; Wagstaff, J.; Griffioen, A.W. Monocyte/macrophage infiltration in tumors: Modulators of angiogenesis. J. Leukocyte Biol. 2006, 80, 1183–1196. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. What Is the Evidence That Tumors Are Angiogenesis Dependent? J. Natl. Cancer Inst. 1990, 82, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J. Tumor angiogenesis: Therapeutic implications. New Engl. J. Med. 1971, 285, 1182–1186. [Google Scholar] [CrossRef]
- Pearson, J.D. Endothelial cell biology. Radiology 1991, 179, 9–14. [Google Scholar] [CrossRef]
- Dudley, A.C. Tumor endothelial cells. Cold Spring Harb. Perspect. Med. 2012, 2, a006536. [Google Scholar] [CrossRef]
- Karar, J.; Maity, A. PI3K/AKT/mTOR Pathway in Angiogenesis. Front. Mol Neurosci 2011, 4, 51. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, J.; Fang, H.; Tang, L.; Chen, W.; Sun, Q.; Zhang, Q.; Yang, F.; Sun, Z.; Cao, L.; et al. Endothelial cells promote triple-negative breast cancer cell metastasis via PAI-1 and CCL5 signaling. FASEB J. 2017, 32, 276–288. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Nico, B.; Ruggieri, S.; Tamma, R.; Simone, G.; Mangia, A. Angiogenesis and Antiangiogenesis in Triple-Negative Breast cancer. Transl. Oncol. 2016, 9, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Morales-Ruiz, M.; Fulton, D.; Sowa, G.; Languino Lucia, R.; Fujio, Y.; Walsh, K.; Sessa William, C. Vascular Endothelial Growth Factor–Stimulated Actin Reorganization and Migration of Endothelial Cells Is Regulated via the Serine/Threonine Kinase Akt. Circul. Res. 2000, 86, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Downward, J. PI 3-kinase, Akt and cell survival. Semin. Cell Dev. Biol. 2004, 15, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Jadeski, L.C.; Hum, K.O.; Chakraborty, C.; Lala, P.K. Nitric oxide promotes murine mammary tumour growth and metastasis by stimulating tumour cell migration, invasiveness and angiogenesis. Int. J. Cancer 2000, 86, 30–39. [Google Scholar] [CrossRef]
- Turashvili, G.; Bouchal, J.; Burkadze, G.; Kolar, Z. Wnt Signaling Pathway in Mammary Gland Development and Carcinogenesis. Pathobiology 2006, 73, 213–223. [Google Scholar] [CrossRef]
- Olsen, J.J.; Pohl, S.Ö.-G.; Deshmukh, A.; Visweswaran, M.; Ward, N.C.; Arfuso, F.; Agostino, M.; Dharmarajan, A. The Role of Wnt Signalling in Angiogenesis. Clin. Biochem. Rev. 2017, 38, 131–142. [Google Scholar]
- Mailhos, C.; Modlich, U.; Lewis, J.; Harris, A.; Bicknell, R.; Ish-Horowicz, D. Delta4, an endothelial specific notch ligand expressed at sites of physiological and tumor angiogenesis. Differentiation 2001, 69, 135–144. [Google Scholar] [CrossRef]
- Dufraine, J.; Funahashi, Y.; Kitajewski, J. Notch signaling regulates tumor angiogenesis by diverse mechanisms. Oncogene 2008, 27, 5132–5137. [Google Scholar] [CrossRef]
- Ghiabi, P.; Jiang, J.; Pasquier, J.; Maleki, M.; Abu-Kaoud, N.; Halabi, N.; Guerrouahen, B.S.; Rafii, S.; Rafii, A. Breast cancer cells promote a notch-dependent mesenchymal phenotype in endothelial cells participating to a pro-tumoral niche. J. Transl. Med. 2015, 13, 27. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Lum, J.J.; Hatzivassiliou, G.; Thompson, C.B. The Biology of Cancer: Metabolic Reprogramming Fuels Cell Growth and Proliferation. Cell Metab. 2008, 7, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. über den Stoffwechsel der Carcinomzelle. Klinische Wochenschrift 1925, 4, 534–536. [Google Scholar] [CrossRef]
- Khalid, O.A.; Claudiu, T.S.; Laurent, S. The Warburg Effect and the Hallmarks of Cancer. Anti-Cancer Agents Med. Chem. 2017, 17, 164–170. [Google Scholar]
- Teuwen, L.-A.; Geldhof, V.; Carmeliet, P. How glucose, glutamine and fatty acid metabolism shape blood and lymph vessel development. Dev. Biol. 2019, 447, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Kuosmanen, S.M.; Kansanen, E.; Kaikkonen, M.U.; Sihvola, V.; Pulkkinen, K.; Jyrkkänen, H.-K.; Tuoresmäki, P.; Hartikainen, J.; Hippeläinen, M.; Kokki, H.; et al. NRF2 regulates endothelial glycolysis and proliferation with miR-93 and mediates the effects of oxidized phospholipids on endothelial activation. Nucleic Acids Res. 2017, 46, 1124–1138. [Google Scholar] [CrossRef] [PubMed]
- Zecchin, A.; Kalucka, J.; Dubois, C.; Carmeliet, P. How Endothelial Cells Adapt Their Metabolism to Form Vessels in Tumors. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Yetkin-Arik, B.; Vogels, I.M.C.; Neyazi, N.; van Duinen, V.; Houtkooper, R.H.; van Noorden, C.J.F.; Klaassen, I.; Schlingemann, R.O. Endothelial tip cells in vitro are less glycolytic and have a more flexible response to metabolic stress than non-tip cells. Sci. Rep. 2019, 9, 10414. [Google Scholar] [CrossRef]
- Cantelmo, A.R.; Conradi, L.-C.; Brajic, A.; Goveia, J.; Kalucka, J.; Pircher, A.; Chaturvedi, P.; Hol, J.; Thienpont, B.; Teuwen, L.-A.; et al. Inhibition of the Glycolytic Activator PFKFB3 in Endothelium Induces Tumor Vessel Normalization, Impairs Metastasis, and Improves Chemotherapy. Cancer Cell 2016, 30, 968–985. [Google Scholar] [CrossRef]
- Xu, Y.; An, X.; Guo, X.; Habtetsion, T.G.; Wang, Y.; Xu, X.; Kandala, S.; Li, Q.; Li, H.; Zhang, C.; et al. Endothelial PFKFB3 plays a critical role in angiogenesis. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1231–1239. [Google Scholar] [CrossRef]
- Schoors, S.; De Bock, K.; Cantelmo, A.R.; Georgiadou, M.; Ghesquière, B.; Cauwenberghs, S.; Kuchnio, A.; Wong, B.W.; Quaegebeur, A.; Goveia, J.; et al. Partial and Transient Reduction of Glycolysis by PFKFB3 Blockade Reduces Pathological Angiogenesis. Cell Metab. 2014, 19, 37–48. [Google Scholar] [CrossRef]
- Kim, B.; Li, J.; Jang, C.; Arany, Z. Glutamine fuels proliferation but not migration of endothelial cells. EMBO J. 2017, 36, 2321–2333. [Google Scholar] [CrossRef] [PubMed]
- Harjes, U.; Kalucka, J.; Carmeliet, P. Targeting fatty acid metabolism in cancer and endothelial cells. Crit. Rev. Oncol. Hematol. 2016, 97, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Foldi, M.; Stickeler, E.; Bau, L.; Kretz, O.; Watermann, D.; Gitsch, G.; Kayser, G.; Zur Hausen, A.; Coy, J.F. Transketolase protein TKTL1 overexpression: A potential biomarker and therapeutic target in breast cancer. Oncol. Rep. 2007, 17, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Langbein, S.; Zerilli, M.; zur Hausen, A.; Staiger, W.; Rensch-Boschert, K.; Lukan, N.; Popa, J.; Ternullo, M.P.; Steidler, A.; Weiss, C.; et al. Expression of transketolase TKTL1 predicts colon and urothelial cancer patient survival: Warburg effect reinterpreted. Br. J. Cancer 2006, 94, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, K.C.; Cunha, I.W.; Rocha, R.M.; Ayala, F.R.; Cajaíba, M.M.; Begnami, M.D.; Vilela, R.S.; Paiva, G.R.; Andrade, R.G.; Soares, F.A. GLUT1 expression in malignant tumors and its use as an immunodiagnostic marker. Clinics 2011, 66, 965–972. [Google Scholar] [CrossRef]
- Johnson, J.M.; Cotzia, P.; Fratamico, R.; Mikkilineni, L.; Chen, J.; Colombo, D.; Mollaee, M.; Whitaker-Menezes, D.; Domingo-Vidal, M.; Lin, Z.; et al. MCT1 in Invasive Ductal Carcinoma: Monocarboxylate Metabolism and Aggressive Breast Cancer. Front. Cell Dev. Biol. 2017, 5, 27. [Google Scholar] [CrossRef]
- Treps, L.; Conradi, L.-C.; Harjes, U.; Carmeliet, P. Manipulating Angiogenesis by Targeting Endothelial Metabolism: Hitting the Engine Rather than the Drivers—A New Perspective? Pharmacol.Rev. 2016, 68, 872. [Google Scholar] [CrossRef]
- Castelló, A.; Pollán, M.; Buijsse, B.; Ruiz, A.; Casas, A.M.; Baena-Cañada, J.M.; Lope, V.; Antolín, S.; Ramos, M.; Muñoz, M.; et al. Spanish Mediterranean diet and other dietary patterns and breast cancer risk: Case-control EpiGEICAM study. Br. J. Cancer 2014, 111, 1454–1462. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Arora, S.; Averett, C.; Singh, S.; Singh, A.P. Modulation of MicroRNAs by Phytochemicals in Cancer: Underlying Mechanisms and Translational Significance. Biomed Res. Int. 2015, 2015, 9. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Z.; Chen, F.; Hu, T.; Peng, W.; Gu, Q.; Sun, Y. The Diverse Oncogenic and Tumor Suppressor Roles of microRNA-105 in Cancer. Front. Oncol. 2019, 9, 518. [Google Scholar] [CrossRef]
- Shah, N.R.; Chen, H. MicroRNAs in pathogenesis of breast cancer: Implications in diagnosis and treatment. World J. Clin. Oncol. 2014, 5, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Dews, M.; Homayouni, A.; Yu, D.; Murphy, D.; Sevignani, C.; Wentzel, E.; Furth, E.E.; Lee, W.M.; Enders, G.H.; Mendell, J.T.; et al. Augmentation of tumor angiogenesis by a Myc-activated microRNA cluster. Nat. Genet. 2006, 38, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Poliseno, L.; Tuccoli, A.; Mariani, L.; Evangelista, M.; Citti, L.; Woods, K.; Mercatanti, A.; Hammond, S.; Rainaldi, G. MicroRNAs modulate the angiogenic properties of HUVECs. Blood 2006, 108, 3068–3071. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, A.; Mukherjee, B.; Dixit, M. MicroRNA Key to Angiogenesis Regulation: MiRNA Biology and Therapy. Curr. Cancer Drug Targ. 2018, 18, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Olson, E.N. AngiomiRs--key regulators of angiogenesis. Curr. Opin. Genet. Dev. 2009, 19, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Hunter, S.; Nault, B.; Ugwuagbo, C.K.; Maiti, S.; Majumder, M. Mir526b and Mir655 Promote Tumour Associated Angiogenesis and Lymphangiogenesis in Breast Cancer. Cancers 2019, 11, 938. [Google Scholar] [CrossRef] [PubMed]
- Salinas-Vera, Y.M.; Marchat, L.A.; Gallardo-Rincon, D.; Ruiz-Garcia, E.; Astudillo-De La Vega, H.; Echavarria-Zepeda, R.; Lopez-Camarillo, C. AngiomiRs: MicroRNAs driving angiogenesis in cancer (Review). Int. J. Mol. Med. 2019, 43, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, L.; Chen, C.; Chu, X. New insights into the regulatory role of microRNA in tumor angiogenesis and clinical implications. Mol. Cancer 2018, 17, 22. [Google Scholar] [CrossRef]
- Wen, S.; Stolarov, J.; Myers, M.P.; Su, J.D.; Wigler, M.H.; Tonks, N.K.; Durden, D.L. PTEN controls tumor-induced angiogenesis. Proc. Natl. Acad. Sci. USA 2001, 98, 4622–4627. [Google Scholar] [CrossRef]
- Ma, J.; Sawai, H.; Ochi, N.; Matsuo, Y.; Xu, D.; Yasuda, A.; Takahashi, H.; Wakasugi, T.; Takeyama, H. PTEN regulates angiogenesis through PI3K/Akt/VEGF signaling pathway in human pancreatic cancer cells. Mol. Cell. Biochem. 2009, 331, 161–171. [Google Scholar] [CrossRef]
- Niu, G.; Wright, K.L.; Huang, M.; Song, L.; Haura, E.; Turkson, J.; Zhang, S.; Wang, T.; Sinibaldi, D.; Coppola, D.; et al. Constitutive Stat3 activity up-regulates VEGF expression and tumor angiogenesis. Oncogene 2002, 21, 2000–2008. [Google Scholar] [CrossRef] [PubMed]
- Chin, K.; Kurashima, Y.; Ogura, T.; Tajiri, H.; Yoshida, S.; Esumi, H. Induction of vascular endothelial growth factor by nitric oxide in human glioblastoma and hepatocellular carcinoma cells. Oncogene 1997, 15, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, G.; Soro-Arnaiz, I.; De Bock, K. The Warburg Effect in Endothelial Cells and its Potential as an Anti-angiogenic Target in Cancer. Front. Cell Dev. Biol. 2018, 6, 100. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-B.; Tang, Y.-L.; Liang, X.-H. Targeting VEGF pathway to normalize the vasculature: An emerging insight in cancer therapy. Onco. Targets Ther. 2018, 11, 6901–6909. [Google Scholar] [CrossRef] [PubMed]
- Cascio, S.; D’Andrea, A.; Ferla, R.; Surmacz, E.; Gulotta, E.; Amodeo, V.; Bazan, V.; Gebbia, N.; Russo, A. miR-20b modulates VEGF expression by targeting HIF-1α and STAT3 in MCF-7 breast cancer cells. J. Cell. Physiol. 2010, 224, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Curtarello, M.; Zulato, E.; Nardo, G.; Valtorta, S.; Guzzo, G.; Rossi, E.; Esposito, G.; Msaki, A.; Pastò, A.; Rasola, A.; et al. VEGF-Targeted Therapy Stably Modulates the Glycolytic Phenotype of Tumor Cells. Cancer Res. 2015, 75, 120. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Qiu, Y.; Yagüe, E.; Ji, W.; Liu, J.; Zhang, J. miRNA-205 targets VEGFA and FGF2 and regulates resistance to chemotherapeutics in breast cancer. Cell Death Dis. 2016, 7, e2291. [Google Scholar] [CrossRef]
- Wang, S.; Aurora, A.B.; Johnson, B.A.; Qi, X.; McAnally, J.; Hill, J.A.; Richardson, J.A.; Bassel-Duby, R.; Olson, E.N. The Endothelial-Specific MicroRNA miR-126 Governs Vascular Integrity and Angiogenesis. Dev. Cell 2008, 15, 261–271. [Google Scholar] [CrossRef]
- Alhasan, L. MiR-126 Modulates Angiogenesis in Breast Cancer by Targeting VEGF-A -mRNA. Asian Pac. J. Cancer Prev. 2019, 20, 193–197. [Google Scholar] [CrossRef]
- Lu, Y.Y.; Sweredoski, M.J.; Huss, D.; Lansford, R.; Hess, S.; Tirrell, D.A. Prometastatic GPCR CD97 is a direct target of tumor suppressor microRNA-126. ACS Chem. Biol. 2014, 9, 334–338. [Google Scholar] [CrossRef]
- Li, H.Y.; Liang, J.L.; Kuo, Y.L.; Lee, H.H.; Calkins, M.J.; Chang, H.T.; Lin, F.C.; Chen, Y.C.; Hsu, T.I.; Hsiao, M.; et al. miR-105/93-3p promotes chemoresistance and circulating miR-105/93-3p acts as a diagnostic biomarker for triple negative breast cancer. Breast Cancer Res. 2017, 19, 133. [Google Scholar] [CrossRef] [PubMed]
- Lokody, I. Cancer genetics: Exosomally derived miR-105 destroys tight junctions. Nat. Rev. Genet. 2014, 15, 362. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Wu, X.; Zhou, W.; Fong, M.Y.; Cao, M.; Liu, J.; Liu, X.; Chen, C.H.; Fadare, O.; Pizzo, D.P.; et al. Cancer-cell-secreted exosomal miR-105 promotes tumour growth through the MYC-dependent metabolic reprogramming of stromal cells. Nat. Cell Biol. 2018, 20, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Fong, M.Y.; Min, Y.; Somlo, G.; Liu, L.; Palomares, M.R.; Yu, Y.; Chow, A.; O’Connor, S.T.; Chin, A.R.; et al. Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell 2014, 25, 501–515. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Du, W.W.; Yang, W.; Rutnam, Z.J.; Peng, C.; Li, H.; O’Malley, Y.Q.; Askeland, R.W.; Sugg, S.; Liu, M.; et al. MiR-93 enhances angiogenesis and metastasis by targeting LATS2. Cell Cycle 2012, 11, 4352–4365. [Google Scholar] [CrossRef] [PubMed]
- Plummer, P.N.; Freeman, R.; Taft, R.J.; Vider, J.; Sax, M.; Umer, B.A.; Gao, D.; Johns, C.; Mattick, J.S.; Wilton, S.D.; et al. MicroRNAs Regulate Tumor Angiogenesis Modulated by Endothelial Progenitor Cells. Cancer Res. 2013, 73, 341. [Google Scholar] [CrossRef]
- Kong, W.; He, L.; Richards, E.J.; Challa, S.; Xu, C.X.; Permuth-Wey, J.; Lancaster, J.M.; Coppola, D.; Sellers, T.A.; Djeu, J.Y.; et al. Upregulation of miRNA-155 promotes tumour angiogenesis by targeting VHL and is associated with poor prognosis and triple-negative breast cancer. Oncogene 2014, 33, 679–689. [Google Scholar] [CrossRef]
- Kim, S.; Lee, E.; Jung, J.; Lee, J.W.; Kim, H.J.; Kim, J.; Yoo, H.J.; Lee, H.J.; Chae, S.Y.; Jeon, S.M.; et al. microRNA-155 positively regulates glucose metabolism via PIK3R1-FOXO3a-cMYC axis in breast cancer. Oncogene 2018, 37, 2982–2991. [Google Scholar] [CrossRef]
- Zhang, T.; Jing, L.; Li, H.; Ding, L.; Ai, D.; Lyu, J.; Zhong, L. MicroRNA-4530 promotes angiogenesis by targeting VASH1 in breast carcinoma cells. Oncol. Lett. 2017, 14, 111–118. [Google Scholar] [CrossRef]
- Liang, H.; Xiao, J.; Zhou, Z.; Wu, J.; Ge, F.; Li, Z.; Zhang, H.; Sun, J.; Li, F.; Liu, R.; et al. Hypoxia induces miR-153 through the IRE1α-XBP1 pathway to fine tune the HIF1α/VEGFA axis in breast cancer angiogenesis. Oncogene 2018, 37, 1961–1975. [Google Scholar] [CrossRef]
- Litchfield, L.M.; Mukherjee, A.; Eckert, M.A.; Johnson, A.; Mills, K.A.; Pan, S.; Shridhar, V.; Lengyel, E.; Romero, I.L. Hyperglycemia-induced metabolic compensation inhibits metformin sensitivity in ovarian cancer. Oncotarget 2015, 6, 23548–23560. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Bian, X.; Shim, H. Downregulation of microRNA-206 promotes invasion and angiogenesis of triple negative breast cancer. Biochem. Biophys Res. Commun. 2016, 477, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Lyu, P.; Cao, Z.; Li, J.; Guo, G.; Xia, W.; Gu, Y. Overexpression of miR-206 suppresses glycolysis, proliferation and migration in breast cancer cells via PFKFB3 targeting. Biochem. Biophys Res. Commun. 2015, 463, 1115–1121. [Google Scholar] [CrossRef]
- Chen, W.-X.; Hu, Q.; Qiu, M.-T.; Zhong, S.-L.; Xu, J.-J.; Tang, J.-H.; Zhao, J.-H. miR-221/222: Promising biomarkers for breast cancer. Tumor Biol. 2013, 34, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.; Ding, H. Differential role of microRNAs miR-221/222 and miR-103/107 in type 2 diabetes and effects of metformin (851.2). FASEB J. 2014, 28, 851–852. [Google Scholar] [CrossRef]
- Pakravan, K.; Babashah, S.; Sadeghizadeh, M.; Mowla, S.J.; Mossahebi-Mohammadi, M.; Ataei, F.; Dana, N.; Javan, M. MicroRNA-100 shuttled by mesenchymal stem cell-derived exosomes suppresses in vitro angiogenesis through modulating the mTOR/HIF-1α/VEGF signaling axis in breast cancer cells. Cell. Oncol. 2017, 40, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cai, B.; Shen, L.; Dong, Y.; Lu, Q.; Sun, S.; Liu, S.; Ma, S.; Ma, P.X.; Chen, J. MiRNA-29b suppresses tumor growth through simultaneously inhibiting angiogenesis and tumorigenesis by targeting Akt3. Cancer Lett. 2017, 397, 111–119. [Google Scholar] [CrossRef]
- Wang, N.; Tan, H.-Y.; Feng, Y.-G.; Zhang, C.; Chen, F.; Feng, Y. microRNA-23a in Human Cancer: Its Roles, Mechanisms and Therapeutic Relevance. Cancers 2018, 11, 7. [Google Scholar] [CrossRef]
- Wu, X.-D.; Guo, T.; Liu, L.; Wang, C.; Zhang, K.; Liu, H.-Q.; Wang, F.; Bai, W.-D.; Zhang, M.-Y. MiR-23a targets RUNX2 and suppresses ginsenoside Rg1-induced angiogenesis in endothelial cells. Oncotarget 2017, 6, 35. [Google Scholar] [CrossRef]
- Hannafon, B.N.; Cai, A.; Calloway, C.L.; Xu, Y.-F.; Zhang, R.; Fung, K.-M.; Ding, W.-Q. miR-23b and miR-27b are oncogenic microRNAs in breast cancer: Evidence from a CRISPR/Cas9 deletion study. BMC Cancer 2019, 19, 642. [Google Scholar] [CrossRef]
- Pellegrino, L.; Stebbing, J.; Braga, V.M.; Frampton, A.E.; Jacob, J.; Buluwela, L.; Jiao, L.R.; Periyasamy, M.; Madsen, C.D.; Caley, M.P.; et al. miR-23b regulates cytoskeletal remodeling, motility and metastasis by directly targeting multiple transcripts. Nucleic Acids Res. 2013, 41, 5400–5412. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, Y.; Lu, Y.; Ritchie, W.; Grau, G.; Vadas, M.A.; Gamble, J.R. The Poly-cistronic miR-23-27-24 Complexes Target Endothelial Cell Junctions: Differential Functional and Molecular Effects of miR-23a and miR-23b. Mol. Ther. Nucleic Acids 2016, 5, e354. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.-H.; Chu, P.-Y.; Hou, M.-F.; Hung, W.-C. MiR-182 promotes proliferation and invasion and elevates the HIF-1α-VEGF-A axis in breast cancer cells by targeting FBXW7. Am. J. Cancer Res. 2016, 6, 1785–1798. [Google Scholar] [PubMed]
- Tu, Y.; Liu, L.; Zhao, D.; Liu, Y.; Ma, X.; Fan, Y.; Wan, L.; Huang, T.; Cheng, Z.; Shen, B. Overexpression of miRNA-497 inhibits tumor angiogenesis by targeting VEGFR2. Sci. Rep. 2015, 5, 13827. [Google Scholar] [CrossRef]
- Wu, Z.; Cai, X.; Huang, C.; Xu, J.; Liu, A. miR-497 suppresses angiogenesis in breast carcinoma by targeting HIF-1alpha. Oncol. Rep. 2016, 35, 1696–1702. [Google Scholar] [CrossRef]
- Wu, Z.; Li, X.; Cai, X.; Huang, C.; Zheng, M. miR-497 inhibits epithelial mesenchymal transition in breast carcinoma by targeting Slug. Tumor Biol. 2016, 37, 7939–7950. [Google Scholar] [CrossRef]
- Stenina, O.; Sul, K.; Krukovets, I. Abstract 57: Hyperglycemia-Induced miR-467 Regulates Angiogenesis and Tumor Growth in a Tissue-Specific Manner. Arterioscler. Thromb. Vasc. Biol. 2012, 32, A57. [Google Scholar] [CrossRef]
- Bhattacharyya, S.; Sul, K.; Krukovets, I.; Nestor, C.; Li, J.; Adognravi, O.S. Novel tissue-specific mechanism of regulation of angiogenesis and cancer growth in response to hyperglycemia. J. Am. Heart Assoc. 2012, 1, e005967. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Xie, H.; Zhou, Z.; Chen, H.; Hu, T.; Bai, Y.; Shen, Y.; Yuan, W.; Jing, Q.; et al. Endothelial-specific intron-derived miR-126 is down-regulated in human breast cancer and targets both VEGFA and PIK3R2. Mol. Cell. Biochem. 2011, 351, 157–164. [Google Scholar] [CrossRef]
- Tang, W.; Yu, F.; Yao, H.; Cui, X.; Jiao, Y.; Lin, L.; Chen, J.; Yin, D.; Song, E.; Liu, Q. miR-27a regulates endothelial differentiation of breast cancer stem like cells. Oncogene 2014, 33, 2629–2638. [Google Scholar] [CrossRef]
- Luo, M.; Tan, X.; Mu, L.; Luo, Y.; Li, R.; Deng, X.; Chen, N.; Ren, M.; Li, Y.; Wang, L.; et al. MiRNA-21 mediates the antiangiogenic activity of metformin through targeting PTEN and SMAD7 expression and PI3K/AKT pathway. Sci. Rep. 2017, 7, 43427. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-Z.; Li, C.; Chen, Q.; Jing, Y.; Carpenter, R.; Jiang, Y.; Kung, H.-F.; Lai, L.; Jiang, B.-H. MiR-21 induced angiogenesis through AKT and ERK activation and HIF-1α expression. PLoS ONE 2011, 6, e19139. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Xiong, Y.; Li, G.; Liu, M.; He, T.; Tang, Y.; Chen, Y.; Cai, L.; Jiang, R.; Tao, J. MiR-21 is overexpressed in response to high glucose and protects endothelial cells from apoptosis. Exp. Clin. Endocrinology Diab. 2013, 121, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Frankel, L.B.; Christoffersen, N.R.; Jacobsen, A.; Lindow, M.; Krogh, A.; Lund, A.H. Programmed Cell Death 4 (PDCD4) Is an Important Functional Target of the MicroRNA miR-21 in Breast Cancer Cells. J. Biol. Chem. 2008, 283, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ng, W.-L.; Wang, P.; Tian, L.; Werner, E.; Wang, H.; Doetsch, P.; Wang, Y. MicroRNA-21 modulates the levels of reactive oxygen species by targeting SOD3 and TNFα. Cancer Res. 2012, 72, 4707. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tan, Z.; Hu, H.; Liu, H.; Wu, T.; Zheng, C.; Wang, X.; Luo, Z.; Wang, J.; Liu, S.; et al. microRNA-21 promotes breast cancer proliferation and metastasis by targeting LZTFL1. BMC Cancer 2019, 19, 738. [Google Scholar] [CrossRef]
- Si, M.L.; Zhu, S.; Wu, H.; Lu, Z.; Wu, F.; Mo, Y.Y. miR-21-mediated tumor growth. Oncogene 2007, 26, 2799–2803. [Google Scholar] [CrossRef]
- Zhu, S.; Wu, H.; Wu, F.; Nie, D.; Sheng, S.; Mo, Y.-Y. MicroRNA-21 targets tumor suppressor genes in invasion and metastasis. Cell Res. 2008, 18, 350–359. [Google Scholar] [CrossRef]
- Zhou, B.; Ma, R.; Si, W.; Li, S.; Xu, Y.; Tu, X.; Wang, Q. MicroRNA-503 targets FGF2 and VEGFA and inhibits tumor angiogenesis and growth. Cancer Lett. 2013, 333, 159–169. [Google Scholar] [CrossRef]
- Long, J.; Ou, C.; Xia, H.; Zhu, Y.; Liu, D. MiR-503 inhibited cell proliferation of human breast cancer cells by suppressing CCND1 expression. Tumor Biol. 2015, 36, 8697–8702. [Google Scholar] [CrossRef]
- Arunachalam, G.; Lakshmanan, A.P.; Samuel, S.M.; Triggle, C.R.; Ding, H. Molecular Interplay between microRNA-34a and Sirtuin1 in Hyperglycemia-Mediated Impaired Angiogenesis in Endothelial Cells: Effects of Metformin. J. Pharmacol. Exp. Ther. 2016, 356, 314. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yuan, L.; Luo, J.; Gao, J.; Guo, J.; Xie, X. MiR-34a inhibits proliferation and migration of breast cancer through down-regulation of Bcl-2 and SIRT1. Clin. Exp. Med. 2013, 13, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Xiao, G.G.; Mao, J.; Lu, Y.; Song, B.; Wang, L.; Fan, S.; Fan, P.; Hou, Z.; Li, J.; et al. Dysregulation of the miR-34a-SIRT1 axis inhibits breast cancer stemness. Oncotarget 2015, 6, 10432–10444. [Google Scholar] [CrossRef] [PubMed]
- Jo, H.-N.; Kang, H.; Lee, A.; Choi, J.; Chang, W.; Lee, M.-S.; Kim, J. Endothelial miR-26a regulates VEGF-Nogo-B receptor-mediated angiogenesis. BMB Rep. 2017, 50, 384–389. [Google Scholar] [CrossRef]
- Gao, J.; Li, L.; Wu, M.; Liu, M.; Xie, X.; Guo, J.; Tang, H.; Xie, X. MiR-26a inhibits proliferation and migration of breast cancer through repression of MCL-1. PLoS ONE 2013, 8, e65138. [Google Scholar] [CrossRef]
- Siragam, V.; Rutnam, Z.J.; Yang, W.; Fang, L.; Luo, L.; Yan, X.; Li, M.; Deng, Z.; Qian, J.; Peng, C.; et al. MicroRNA miR-98 inhibits tumor angiogenesis and invasion by targeting activin receptor-like kinase-4 and matrix metalloproteinase-11. Oncotarget 2012, 3, 11. [Google Scholar] [CrossRef]
- Zampetaki, A.; Kiechl, S.; Drozdov, I.; Willeit, P.; Mayr, U.; Prokopi, M.; Mayr, A.; Weger, S.; Oberhollenzer, F.; Bonora, E.; et al. Plasma MicroRNA Profiling Reveals Loss of Endothelial MiR-126 and Other MicroRNAs in Type 2 Diabetes. Circul. Res. 2010, 107, 810–817. [Google Scholar] [CrossRef]
- Zou, C.; Xu, Q.; Mao, F.; Li, D.; Bian, C.; Liu, L.-Z.; Jiang, Y.; Chen, X.; Qi, Y.; Zhang, X.; et al. MiR-145 inhibits tumor angiogenesis and growth by N-RAS and VEGF. Cell Cycle 2012, 11, 2137–2145. [Google Scholar] [CrossRef]
- Luengo-Gil, G.; Gonzalez-Billalabeitia, E.; Perez-Henarejos, S.A.; Navarro Manzano, E.; Chaves-Benito, A.; Garcia-Martinez, E.; Garcia-Garre, E.; Vicente, V.; Ayala de la Peña, F. Angiogenic role of miR-20a in breast cancer. PLoS ONE 2018, 13, e0194638. [Google Scholar] [CrossRef]
- Serguienko, A.; Grad, I.; Wennerstrøm, A.B.; Meza-Zepeda, L.A.; Thiede, B.; Stratford, E.W.; Myklebost, O.; Munthe, E. Metabolic reprogramming of metastatic breast cancer and melanoma by let-7a microRNA. Oncotarget 2015, 6, 2451–2465. [Google Scholar] [CrossRef]
- Parveen, A.; Subedi, L.; Kim, H.W.; Khan, Z.; Zahra, Z.; Farooqi, M.Q.; Kim, S.Y. Phytochemicals Targeting VEGF and VEGF-Related Multifactors as Anticancer Therapy. J. Clin Med. 2019, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, L.M.; Valente, I.M.; Rodrigues, J.A. An overview on cardamonin. J. Med. Food 2014, 17, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Lin, C.; Cheng, X.; Hua, H.; Xiang, T.; Huang, Y.; Huang, X. Cardamonin reduces chemotherapy resistance of colon cancer cells via the TSP50/NF-κB pathway in vitro. Oncol. Lett. 2018, 15, 9641–9646. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.-S.; Tian, S.-S.; Lu, J.-J.; Ding, X.-H.; Qian, C.-D.; Ding, B.; Ding, Z.-S.; Jin, B. Cardamonin Regulates miR-21 Expression and Suppresses Angiogenesis Induced by Vascular Endothelial Growth Factor. Biomed Res. Int. 2015, 2015, 501581. [Google Scholar] [CrossRef]
- Dong, G.; Liang, X.; Wang, D.; Gao, H.; Wang, L.; Wang, L.; Liu, J.; Du, Z. High expression of miR-21 in triple-negative breast cancers was correlated with a poor prognosis and promoted tumor cell in vitro proliferation. Med. Oncol. 2014, 31, 57. [Google Scholar] [CrossRef] [PubMed]
- Mirzaaghaei, S.; Foroughmand, A.M.; Saki, G.; Shafiei, M. Combination of Epigallocatechin-3-gallate and Silibinin: A Novel Approach for Targeting Both Tumor and Endothelial Cells. ACS Omega 2019, 4, 8421–8430. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Jo, H.; Cho, J.H.; Dhanasekaran, D.N.; Song, Y.S. Resveratrol as a Tumor-Suppressive Nutraceutical Modulating Tumor Microenvironment and Malignant Behaviors of Cancer. Int. J. Mol. Sci. 2019, 20, 925. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Liu, Q.; Liu, K.; Yagasaki, K.; Wu, E.; Zhang, G. Matrine suppresses breast cancer cell proliferation and invasion via VEGF-Akt-NF-κB signaling. Cytotechnology 2009, 59, 219–229. [Google Scholar] [CrossRef]
- Wang, P.S.; Yu, C.-H.; Su, J.-C. Effects of Evodiamine on the Expression and Secretion of Vascular Endothelial Growth Factor in Human Breast Cancer Cells. Biol. Reprod. 2008, 78, 215. [Google Scholar] [CrossRef]
- Li, P.; Zhang, M.; Ma, W.-j.; Sun, X.; Jin, F.-p. Effects of brucine on vascular endothelial growth factor expression and microvessel density in a nude mouse model of bone metastasis due to breast cancer. Chin. J. Integr. Med. 2012, 18, 605–609. [Google Scholar] [CrossRef]
- Siddiqui, F.A.; Prakasam, G.; Chattopadhyay, S.; Rehman, A.U.; Padder, R.A.; Ansari, M.A.; Irshad, R.; Mangalhara, K.; Bamezai, R.N.K.; Husain, M.; et al. Curcumin decreases Warburg effect in cancer cells by down-regulating pyruvate kinase M2 via mTOR-HIF1α inhibition. Sci. Rep. 2018, 8, 8323. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, K.M.; Anderson, N.G. The protein kinase B/Akt signalling pathway in human malignancy. Cell. Signal. 2002, 14, 381–395. [Google Scholar] [CrossRef]
- Tili, E.; Michaille, J.-J.; Adair, B.; Alder, H.; Limagne, E.; Taccioli, C.; Ferracin, M.; Delmas, D.; Latruffe, N.; Croce, C.M. Resveratrol decreases the levels of miR-155 by upregulating miR-663, a microRNA targeting JunB and JunD. Carcinogenesis 2010, 31, 1561–1566. [Google Scholar] [CrossRef] [PubMed]
- Talcott, S.U.; Li, X.; Chintharlapalli, S.; Safe, S. The Effects of betulinic acid on microRNA-27a regulated target genes in MDA-MB-231 breast cancer cells. FASEB J. 2008, 22, 158.158. [Google Scholar] [CrossRef]
- Du, G.-J.; Zhang, Z.; Wen, X.-D.; Yu, C.; Calway, T.; Yuan, C.-S.; Wang, C.-Z. Epigallocatechin Gallate (EGCG) is the most effective cancer chemopreventive polyphenol in green tea. Nutrients 2012, 4, 1679–1691. [Google Scholar] [CrossRef]
- Wei, R.; Mao, L.; Xu, P.; Zheng, X.; Hackman, R.M.; Mackenzie, G.G.; Wang, Y. Suppressing glucose metabolism with epigallocatechin-3-gallate (EGCG) reduces breast cancer cell growth in preclinical models. Food Function 2018, 9, 5682–5696. [Google Scholar] [CrossRef]
- de la Parra, C.; Castillo-Pichardo, L.; Cruz-Collazo, A.; Cubano, L.; Redis, R.; Calin, G.A.; Dharmawardhane, S. Soy Isoflavone Genistein-Mediated Downregulation of miR-155 Contributes to the Anticancer Effects of Genistein. Nutr. Cancer 2016, 68, 154–164. [Google Scholar] [CrossRef]
- Maxwell, P.H.; Pugh, C.W.; Ratcliffe, P.J. Activation of the HIF pathway in cancer. Curr Opin Genet. Dev. 2001, 11, 293–299. [Google Scholar] [CrossRef]
- Wu, X.; Li, L.; Li, Y.; Liu, Z. MiR-153 promotes breast cancer cell apoptosis by targeting HECTD3. Am. J. Cancer Res. 2016, 6, 1563–1571. [Google Scholar]
- Song, X.; Yao, J.; Wang, F.; Zhou, M.; Zhou, Y.; Wang, H.; Wei, L.; Zhao, L.; Li, Z.; Lu, N.; et al. Wogonin inhibits tumor angiogenesis via degradation of HIF-1α protein. Toxicol. Appl. Pharmacol. 2013, 271, 144–155. [Google Scholar] [CrossRef]
- Gu, J.-W.; Makey, K.L.; Tucker, K.B.; Chinchar, E.; Mao, X.; Pei, I.; Thomas, E.Y.; Miele, L. EGCG, a major green tea catechin suppresses breast tumor angiogenesis and growth via inhibiting the activation of HIF-1α and NFκB, and VEGF expression. Vasc. Cell 2013, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Tang, L.; Li, X.; Li, H. Triptolide inhibits vascular endothelial growth factor-mediated angiogenesis in human breast cancer cells. Exp. Ther. Med. 2018, 16, 830–836. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.J.; Poellinger, L. The dietary flavonoid quercetin modulates HIF-1α activity in endothelial cells. Biochem Biophys Res. Commun. 2002, 293, 446–450. [Google Scholar] [CrossRef]
- He, J.; Jiang, B.-H. Interplay between Reactive oxygen Species and MicroRNAs in Cancer. Curr. Pharmacol. Rep. 2016, 2, 82–90. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Oxid. Med. Cell. Longev. 2016, 2016, 4350965. [Google Scholar] [CrossRef]
- Sarmiento-Salinas, F.L.; Delgado-Magallón, A.; Montes-Alvarado, J.B.; Ramírez-Ramírez, D.; Flores-Alonso, J.C.; Cortés-Hernández, P.; Reyes-Leyva, J.; Herrera-Camacho, I.; Anaya-Ruiz, M.; Pelayo, R.; et al. Breast Cancer Subtypes Present a Differential Production of Reactive Oxygen Species (ROS) and Susceptibility to Antioxidant Treatment. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef]
- Babu, K.R.; Tay, Y. The Yin-Yang Regulation of Reactive Oxygen Species and MicroRNAs in Cancer. Int. J. Mol. Sci. 2019, 20, 5335. [Google Scholar] [CrossRef]
- Yang, M.; Yao, Y.; Eades, G.; Zhang, Y.; Zhou, Q. MiR-28 regulates Nrf2 expression through a Keap1-independent mechanism. Breast Cancer Res. Treat. 2011, 129, 983–991. [Google Scholar] [CrossRef]
- Shin, B.; Feser, R.; Nault, B.; Hunter, S.; Maiti, S.; Ugwuagbo, K.C.; Majumder, M. miR526b and miR655 Induce Oxidative Stress in Breast Cancer. Int. J. Mol. Sci. 2019, 20, 4039. [Google Scholar] [CrossRef]
- La Sala, L.; Mrakic-Sposta, S.; Micheloni, S.; Prattichizzo, F.; Ceriello, A. Glucose-sensing microRNA-21 disrupts ROS homeostasis and impairs antioxidant responses in cellular glucose variability. Cardiovasc. Diabetol. 2018, 17, 105. [Google Scholar] [CrossRef]
- Yan, L.-X.; Huang, X.-F.; Shao, Q.; Huang, M.-Y.; Deng, L.; Wu, Q.-L.; Zeng, Y.-X.; Shao, J.-Y. MicroRNA miR-21 overexpression in human breast cancer is associated with advanced clinical stage, lymph node metastasis and patient poor prognosis. RNA 2008, 14, 2348–2360. [Google Scholar] [CrossRef] [PubMed]
- Robaszkiewicz, A.; Balcerczyk, A.; Bartosz, G. Antioxidative and prooxidative effects of quercetin on A549 cells. Cell Biol. Int. 2007, 31, 1245–1250. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Zhang, K.; Clarke, K.; Weiland, T.; Sauter, E.R. Methylation and miRNA Effects of Resveratrol on Mammary Tumors vs. Normal Tissue. Nutr. Cancer 2014, 66, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Phuah, N.H.; Nagoor, N.H. Regulation of MicroRNAs by Natural Agents: New Strategies in Cancer Therapies. Biomed Res. Int. 2014, 2014, 17. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, M.M.; Ranji, N.; Motamed, N. Deregulation of miR-21 and miR-155 and their putative targets after silibinin treatment in T47D breast cancer cells. Iran. J. Basic Med. Sci. 2015, 18, 1209–1214. [Google Scholar] [PubMed]
- Chen, J.; Xu, T.; Chen, C. The critical roles of miR-21 in anti-cancer effects of curcumin. Ann. Transl. Med. 2015, 3, 330. [Google Scholar] [CrossRef]
- Yang, J.; Cao, Y.; Sun, J.; Zhang, Y. Curcumin reduces the expression of Bcl-2 by upregulating miR-15a and miR-16 in MCF-7 cells. Med. Oncol. 2010, 27, 1114–1118. [Google Scholar] [CrossRef]
- Wang, T.-y.; Chen, J.-X. Effects of Curcumin on Vessel Formation Insight into the Pro- and Antiangiogenesis of Curcumin. Evid. Based Complementary Altern. Med. 2019, 2019, 9. [Google Scholar] [CrossRef]
- Cabello, P.; Pineda, B.; Tormo, E.; Lluch, A.; Eroles, P. The Antitumor Effect of Metformin Is Mediated by miR-26a in Breast Cancer. Int. J. Mol. Sci. 2016, 17, 1298. [Google Scholar] [CrossRef]
- Liu, B.; Fan, Z.; Edgerton, S.M.; Deng, X.-S.; Alimova, I.N.; Lind, S.E.; Thor, A.D. Metformin induces unique biological and molecular responses in triple negative breast cancer cells. Cell Cycle 2009, 8, 2031–2040. [Google Scholar] [CrossRef]
- Oliveras-Ferraros, C.; Cufí, S.; Vazquez-Martin, A.; Torres-Garcia, V.Z.; Del Barco, S.; Martin-Castillo, B.; Menendez, J.A. Micro(mi)RNA expression profile of breast cancer epithelial cells treated with the anti-diabetic drug metformin: Induction of the tumor suppressor miRNA let-7a and suppression of the TGFβ-induced oncomiR miRNA-181a. Cell Cycle 2011, 10, 1144–1151. [Google Scholar] [CrossRef] [PubMed]
- Avci, C.B.; Susluer, S.Y.; Caglar, H.O.; Balci, T.; Aygunes, D.; Dodurga, Y.; Gunduz, C. Genistein-induced mir-23b expression inhibits the growth of breast cancer cells. Contemp. Oncol. (Pozn) 2015, 19, 32–35. [Google Scholar] [CrossRef] [PubMed]
- Fix, L.N.; Shah, M.; Efferth, T.; Farwell, M.A.; Zhang, B. MicroRNA expression profile of MCF-7 human breast cancer cells and the effect of green tea polyphenon-60. Cancer Genom. Proteom. 2010, 7, 261–278. [Google Scholar]
- Sun, D.W.; Zhang, H.D.; Mao, L.; Mao, C.F.; Chen, W.; Cui, M.; Ma, R.; Cao, H.X.; Jing, C.W.; Wang, Z.; et al. Luteolin Inhibits Breast Cancer Development and Progression In Vitro and In Vivo by Suppressing Notch Signaling and Regulating MiRNAs. Mol. Cell. Biochem. 2015, 37, 1693–1711. [Google Scholar] [CrossRef]
- Lijuan, W.; Jing, W. Luteolin induced apoptosis in breast cancer cell by decreasing the expression of microRNA-21. Chongqing Med. 2013, 12. [Google Scholar]
- Gao, G.; Ge, R.; Li, Y.; Liu, S. Luteolin exhibits anti-breast cancer property through up-regulating miR-203. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3265–3271. [Google Scholar] [CrossRef]
- Zhang, H.; Ren, Y.; Tang, X.; Wang, K.; Liu, Y.; Zhang, L.; Li, X.; Liu, P.; Zhao, C.; He, J. Vascular normalization induced by sinomenine hydrochloride results in suppressed mammary tumor growth and metastasis. Sci. Rep. 2015, 5, 8888. [Google Scholar] [CrossRef]
- Gao, G.; Liang, X.; Ma, W. Sinomenine restrains breast cancer cells proliferation, migration and invasion via modulation of miR-29/PDCD-4 axis. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3839–3846. [Google Scholar] [CrossRef]
- Liu, Q.; Wang, W.; Li, F.; Yu, D.; Xu, C.; Hu, H. Triptolide Inhibits Breast Cancer Cell Metastasis Through Inducing the Expression of miR-146a, a Negative Regulator of Rho GTPase. Oncol. Res. 2019, 27, 1043–1050. [Google Scholar] [CrossRef]
- WHO. Breast cancer. 2018. Available online: https://www.who.int/cancer/prevention/diagnosis-screening/breast-cancer/en/ (accessed on 12 November 2019).
- Yang, X.; Wang, X.; Shen, H.; Deng, R.; Xue, K. Combination of miR-21 with Circulating Tumor Cells Markers Improve Diagnostic Specificity of Metastatic Breast Cancer. Cell Biochem. Biophysics 2015, 73, 87–91. [Google Scholar] [CrossRef]
- Hu, J.; Xu, J.; Wu, Y.; Chen, Q.; Zheng, W.; Lu, X.; Zhou, C.; Jiao, D. Identification of microRNA-93 as a functional dysregulated miRNA in triple-negative breast cancer. Tumor Biol. 2015, 36, 251–258. [Google Scholar] [CrossRef] [PubMed]
- McGuire, A.; Brown, J.A.L.; Kerin, M.J. Metastatic breast cancer: The potential of miRNA for diagnosis and treatment monitoring. Cancer Metastasis Rev. 2015, 34, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Büsselberg, D. Flavonoids in Cancer and Apoptosis. Cancers 2018, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Samec, M.; Liskova, A.; Kubatka, P.; Uramova, S.; Zubor, P.; Samuel, S.M.; Zulli, A.; Pec, M.; Bielik, T.; Biringer, K.; et al. The role of dietary phytochemicals in the carcinogenesis via the modulation of miRNA expression. J. Cancer Res. Clin. Oncol. 2019, 145, 1665–1679. [Google Scholar] [CrossRef]
- Flabouraris, G.; Karikas, G.A. Nutri-epigenetics and synthetic analogs in cancer chemoprevention. J. BUON 2016, 21, 4–16. [Google Scholar]
- Chuang, J.C.; Jones, P.A. Epigenetics and MicroRNAs. Pediatr. Res. 2007, 61, 24–29. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; Leapman, S.B.; Cotran, R.S.; Folkman, J. Tumor dormancy in vivo by prevention of neovascularization. J. Exp. Med. 1972, 136, 261–276. [Google Scholar] [CrossRef]
- Varghese, E.; Samuel, S.M.; Abotaleb, M.; Cheema, S.; Mamtani, R.; Busselberg, D. The “Yin and Yang” of Natural Compounds in Anticancer Therapy of Triple-Negative Breast Cancers. Cancers 2018, 10, 346. [Google Scholar] [CrossRef]
- Blenkiron, C.; Goldstein, L.D.; Thorne, N.P.; Spiteri, I.; Chin, S.-F.; Dunning, M.J.; Barbosa-Morais, N.L.; Teschendorff, A.E.; Green, A.R.; Ellis, I.O.; et al. MicroRNA expression profiling of human breast cancer identifies new markers of tumor subtype. Genome Biol. 2007, 8, R214. [Google Scholar] [CrossRef]
- Baldassari, F.; Zerbinati, C.; Galasso, M.; Corrà, F.; Minotti, L.; Agnoletto, C.; Previati, M.; Croce, C.M.; Volinia, S. Screen for MicroRNA and Drug Interactions in Breast Cancer Cell Lines Points to miR-126 as a Modulator of CDK4/6 and PIK3CA Inhibitors. Front. Genet. 2018, 9. [Google Scholar] [CrossRef]
- Valeri, N.; Gasparini, P.; Braconi, C.; Paone, A.; Lovat, F.; Fabbri, M.; Sumani, K.M.; Alder, H.; Amadori, D.; Patel, T.; et al. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2). Proc. Natl. Acad. Sci. USA 2010, 107, 21098–21103. [Google Scholar] [CrossRef] [PubMed]
- Harjes, U.; Bensaad, K.; Harris, A.L. Endothelial cell metabolism and implications for cancer therapy. Br. J. Cancer 2012, 107, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Arora, A.; Singh, S.; Bhatt, A.N.; Pandey, S.; Sandhir, R.; Dwarakanath, B.S. Interplay Between Metabolism and Oncogenic Process: Role of microRNAs. Transl. Oncogenomics 2015, 7, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Verdegem, D.; Moens, S.; Stapor, P.; Carmeliet, P. Endothelial cell metabolism: Parallels and divergences with cancer cell metabolism. Cancer Metab. 2014, 2, 19. [Google Scholar] [CrossRef]
- Bakhashab, S.; Ahmed, F.; Schulten, H.-J.; Ahmed, F.W.; Glanville, M.; Al-Qahtani, M.H.; Weaver, J.U. Proangiogenic Effect of Metformin in Endothelial Cells Is via Upregulation of VEGFR1/2 and Their Signaling under Hyperglycemia-Hypoxia. Int. J. Mol. Sci. 2018, 19, 293. [Google Scholar] [CrossRef]
- Abbasi, B.; Iqbal, J.; Mahmood, T.; Khalil, A.; Ali, B.; Kanwal, S.; Shah, S.; Ahmad, R. Role of dietary phytochemicals in modulation of miRNA expression: Natural swords combating breast cancer. Asian Pac. J. Trop. Med. 2018, 11, 501–509. [Google Scholar] [CrossRef]
- Caporali, A.; Emanueli, C. MicroRNA regulation in angiogenesis. Vasc. Pharmacol. 2011, 55, 79–86. [Google Scholar] [CrossRef]

| miRNA | Cellular Function | Target | Signaling Pathway | Cell Line | References | |
|---|---|---|---|---|---|---|
| 1 | miRNA-105 | Promote metastasis | ZO-1 (tight junction protein) MXI1 |
| MDA-MB-231 | [61,62,63,64] |
| 2 | miRNA-93 | EC glycolysis and EC proliferation | KLF2 and PFKFB3 (in glycolysis). FOXO1 and MYC (in proliferation). WNK lysine deficient protein kinase 1 (WNK1) ↓LATS2 |
| MT-1 MDA-MB-231 Breast carcinoma specimens. | [25,65] |
| 3 | miRNA-10b and miRNA-196b | Produced in response to tumor secreted VEGF and regulate EPC function and angiogenesis | ↓HOXD10 |
| IDC grade III tumors | [66] |
| 4 | miRNA-155 | Up-regulates glucose transporters and glycolytic enzymes. Associated with poor prognosis and metastasis. | ↓VHL |
| TNBC- late-stage (stage III/IV), lymph node metastasis | [67,68] |
| 5 | miRNA-4530 | Decreases proliferation, induces apoptosis, promotes angiogenesis | ↓VASH1 (endogenous angiogenesis inhibitor |
| MCF-7 MDA-MB-231 HUVEC | [69] |
| 6 | miRNA-153 | Response to high glucose Tumor suppressor EMT suppressor Down-regulated in BC | IRE1α-XBP1 ↓MTDH |
| MDA-MB-231 HCC1937 Luminal A and B, Basal, HER 2 positive | [70,71] |
| 7 | miRNA-205 | Enhances chemosensitivity of breast cancer cells to TAC chemotherapy (docetaxol, doxorubicin plus cyclophosphamide) | ↓VEGFA and FGF2 |
| MCF-7/A02 and CALDOX | [57] |
| 8 | miRNA-206 | Suppresses glycolysis | ↓VEGF PFKFB3 |
| MDA-MB-231, MDA-MB-435, and HCC1395 | [72,73] |
| 9 | miRNA-221/222 | Anti-angiogenic Anti-proliferative Increases senescence Response to high glucose | ↓eNOS ↓ZEB2 ↓P27 |
| Mouse microvascular endothelial cells (MMECs) | [74,75] |
| 10 | miRNA-100 | Mesenchymal stem cell derived exosomes | ↓VEGF |
| MDA-MB-231, MCF-7, T47D, HUVEC BM-MSC | [76] |
| 11 | miRNA-29b | Invasion, proliferation and migration | AKT3 SPIN1 |
| HUVEC MDA-MB-231 | [77] |
| 12 | miRNA-23a | Up/down-regulated in specific type of cancers. Aerobic glycolysis Anti-angiogenic | ↓LDHA and LDHB |
| HUVEC | [78,79] |
| 13 | miRNA-23b | Inversely correlated with metastasis | ↓PAK2, MLC II Phosphorylation, under the regulation of AP-1, directly target cytoskeleton genes. JAM-C and ZO-2 |
| MCF7 MDA-MB-231 HUVEC Xenograft | [80,81,82] |
| 14 | miRNA-182 | Oncogenic | ↓FBXW7 |
| hy926 MCF-7 ER and PR positive BC | [83] |
| 15 | miRNA-497 | Down-regulated EMT | ↓VEGFR2, ↓VEGF and ↓HIF-1α ↓ Slug |
| MCF-7 4T1 Xenograft Invasive ductal breast cancer | [84,85,86] |
| 16 | miRNA-140-5p | Tumor suppressor | ↓VEGFA, ↓MMP9 |
| MCF-7 MDA-MB-231 | [11] |
| 17 | miRNA-467 | Response to hyperglycemia Tissue specific | ↓TSP-1 |
| EMT6 (mu) C116 | [87,88] |
| 18 | miRNA-126 | Endothelial cell specific | ↓VEGFA ↓PIK3R2 ↓SPRED1 |
| MCF-7 | [89] |
| 19 | miRNA-27a | Pro-angiogenic | ↑ZBTB10 |
| MDA-MB-231 BCSLCs (SK-3rd) | [90] |
| 20 | miRNA-21 | Responsive to glucose level Pro-angiogenic Modulates ROS level Promotes metastasis | ↓PTEN and SMAD7 HIF-1α PDCD4, maspin LZTFL1 |
| MCF-7 MDA-MB-231 | [91,92,93,94,95,96,97,98] |
| 21 | miRNA-503 | Anti-angiogenic | ↓CCND1 |
| MCF-7, T47D, MDA-MB231, BT549, SKBR3, ZR-75-30 | [99,100] |
| 22 | miRNA-34a | Response to high glucose | ↓SIRT1 |
| BT-474, MDA-MB-231, MDA-MB-435, MDA-MB-468, SK-BR-3, EC | [101,102,103] |
| 23 | miRNA-26a | ↓ VEGF dependent migration and proliferation Anti-angiogenic | ↓NgBR MCL-1 |
| HUVECs MDA-MB-231, MCF-7, MDA-MB-435, MDA-MB-468 | [104,105] |
| 24 | miRNA- 98 | Anti-angiogenic | ↓MMP11, ↓ALK4 |
| 4T1, MT1, MDA-MB-231, MDA-MB-468, Xenograft | [106] |
| 25 | miRNA-126 | Response to high glucose ↓Type 2 diabetes | ↓VEGFA and PI3K regulatory subunit 2 (PIK3R2) CD97, GPCR |
| MDA-MB-231 MCF-7 plasma from DM patients | [59,60,107] |
| 26 | miRNA-145 | Anti-angiogenic | N-RAS and VEGF-A IGF-I/IRS |
| MCF-7 MDA-MB-231 | [108] |
| 27 | miRNA-20a | Predominantly in TNBC pro-angiogenic | ↑VEGFA and HIF-1α |
| MCF-7 MDA-MB-231 | [109] |
| 28 | miRNA-526b miRNA-655 | Tumor associated angiogenesis and lymphangiogenesis | EP4, ↓PTEN and PI3K/Akt ↑VEGFA |
| MCF-7 ER and PR positive HER 2 negative BC | [46] |
| 29 | let-7a | Regulates key anabolic enzymes ROS production | Stearol Co-A Desaturase (SCD) G6PD, FASN, BACH1 |
| MDA-MB-231 | [110] |
| Compound | Target miRNA | Effects | Cells | References | |
|---|---|---|---|---|---|
| 1 | Cardamonin (50 μM) | ↓miRNA-21 | ↓VEGF mediated angiogenesis, inhibits EC proliferation and migration | HUVECs | [114] |
| 2 | Resveratrol (50 μM) | miRNA-34a miRNA-424 miRNA-503 ↓miR-155 ↑miRNA-21 ↑miR129 and miR489 ↑miR-141 and miR-200c | ↓VEGF-↓glycolytic genes, ↓ERK 1/2, ↓NO ↓IL-8/CXCL8 ↓DNMT1, DNMT3b ↓Stemness | HUVEC, Estrogen dependent mammary carcinoma rat model MDA-MB-231 | [117,123,143,144] |
| 3 | Silibinin (in combination with EGCG) (91.22 μM and 68.07 μM) | ↓miRNA-21 ↓miR-17−92 | ↑CASP-9 and APAF-1 ↓VEGF−VEGFR2 axis | T47D, HUVEC | [145] |
| 4 | Curcumin (30–60 lmol l−1) | miRNA-29 ↓miRNA-21 ↑miR-15a and miR-16 | PDCD4, PTEN/PI3K/AKT and NF-κB ↓Bcl-2 Pro- or anti-angiogenic at different concentrations | MCF-7 | [146,147,148] |
| 5 | Metformin (anti-diabetic drug) (20 mM) | ↓miRNA-21 ↓miRNA-221 miRNA-34a ↑miRNA-26a ↓Let-7a | Anti-angiogenic via ↓TGF-β, PTEN, EHZ2 and SMAD7 eNOS signalling | HUVECs MDA-MB-231, MDA-MB-468, BT 549, MCF-7 | [75,91,101,149,150,151] |
| 6 | Genistein (10–25 μM) | ↓miRNA-155 ↑miRNA-23b ↓miRNA 221/222 | ↑FOXO3, PTEN Regulates viability and apoptosis via transcriptional regulation of miR-155 ↓Metastasis, enhances focal adhesion connections | MDA-MB-435, Hs578t | [127,152] |
| 7 | EGCG (20 µM) | ↓pro angiogenic ↑anti angiogenic miRNA ↓miRNA 27a ↓miRNA-21 | ↓HIF1α, GLUT1 ↓HK, PFK, LDH (glycolytic enzymes) ZBTB10 | 4T1, MCF-7 | [126] [116,153] |
| 8 | Luteolin (50 µmol/L) | ↓miRNA-155 ↓miRNA-21 ↑miR-34a ↑miRNA-181a, ↑miRNA-139-5p ↑miRNA-224 and ↑miRNA-246 ↑miRNA-203 | ↓Notch signaling, ↓PI3K/Akt, ↓VEGF, ↓Notch-1, ↓Hes-1, ↓Hey, ↓VEGF, ↓Cyclin D1 and MMP2/9 ↓Tumor growth, ↓Invasion ↓Angiogenesis Anti-oxidant | MDA-MB-231, HUVECs | [154,155,156] |
| 9 | Sinomenine (4 µM) | ↑miRNA-29 | miR-29/PDCD-4 axis ↓Tumor growth, ↓Metastasis, invasion, vascular normalization, improved tumor immunity | HUVEC 4T1(murine breast cancer model) MDA-MB-231, MCF-7 | [157,158] |
| 10 | Triptolide (15 ng/mL) | ↑miRNA-146a | ↓Rho GTPase - ↓Metastasis and invasion ↓ERK1/2-/HIF-1α/6666VEGFA axis | MDA-MB-231 | [132,159] |
| 11 | Betulinic acid (2.5–10 μmMol/L) | ↓miRNA-27a | ↑ZBTB10 and Myt-1 | MDA-MB-231, BT-549 | [124] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varghese, E.; Liskova, A.; Kubatka, P.; Samuel, S.M.; Büsselberg, D. Anti-Angiogenic Effects of Phytochemicals on miRNA Regulating Breast Cancer Progression. Biomolecules 2020, 10, 191. https://doi.org/10.3390/biom10020191
Varghese E, Liskova A, Kubatka P, Samuel SM, Büsselberg D. Anti-Angiogenic Effects of Phytochemicals on miRNA Regulating Breast Cancer Progression. Biomolecules. 2020; 10(2):191. https://doi.org/10.3390/biom10020191
Chicago/Turabian StyleVarghese, Elizabeth, Alena Liskova, Peter Kubatka, Samson Mathews Samuel, and Dietrich Büsselberg. 2020. "Anti-Angiogenic Effects of Phytochemicals on miRNA Regulating Breast Cancer Progression" Biomolecules 10, no. 2: 191. https://doi.org/10.3390/biom10020191
APA StyleVarghese, E., Liskova, A., Kubatka, P., Samuel, S. M., & Büsselberg, D. (2020). Anti-Angiogenic Effects of Phytochemicals on miRNA Regulating Breast Cancer Progression. Biomolecules, 10(2), 191. https://doi.org/10.3390/biom10020191







