Protective Effects of Epigallocatechin Gallate (EGCG) on Endometrial, Breast, and Ovarian Cancers
Abstract
1. Introduction
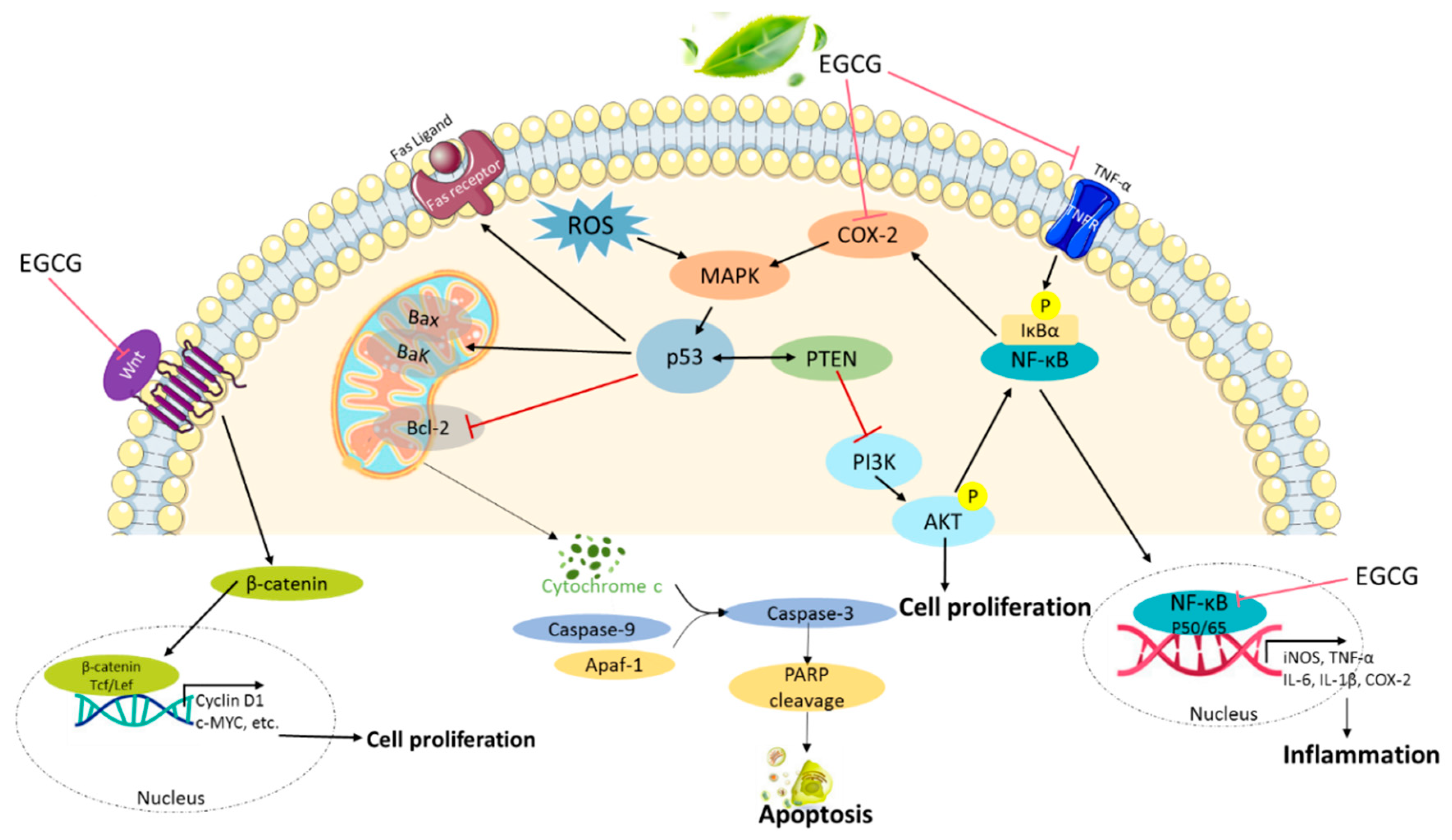
2. Cancer-Protective Mechanisms of EGCG
2.1. Antioxidant Activity
2.2. Anti-Inflammatory Activity
2.3. Antiproliferative Activity
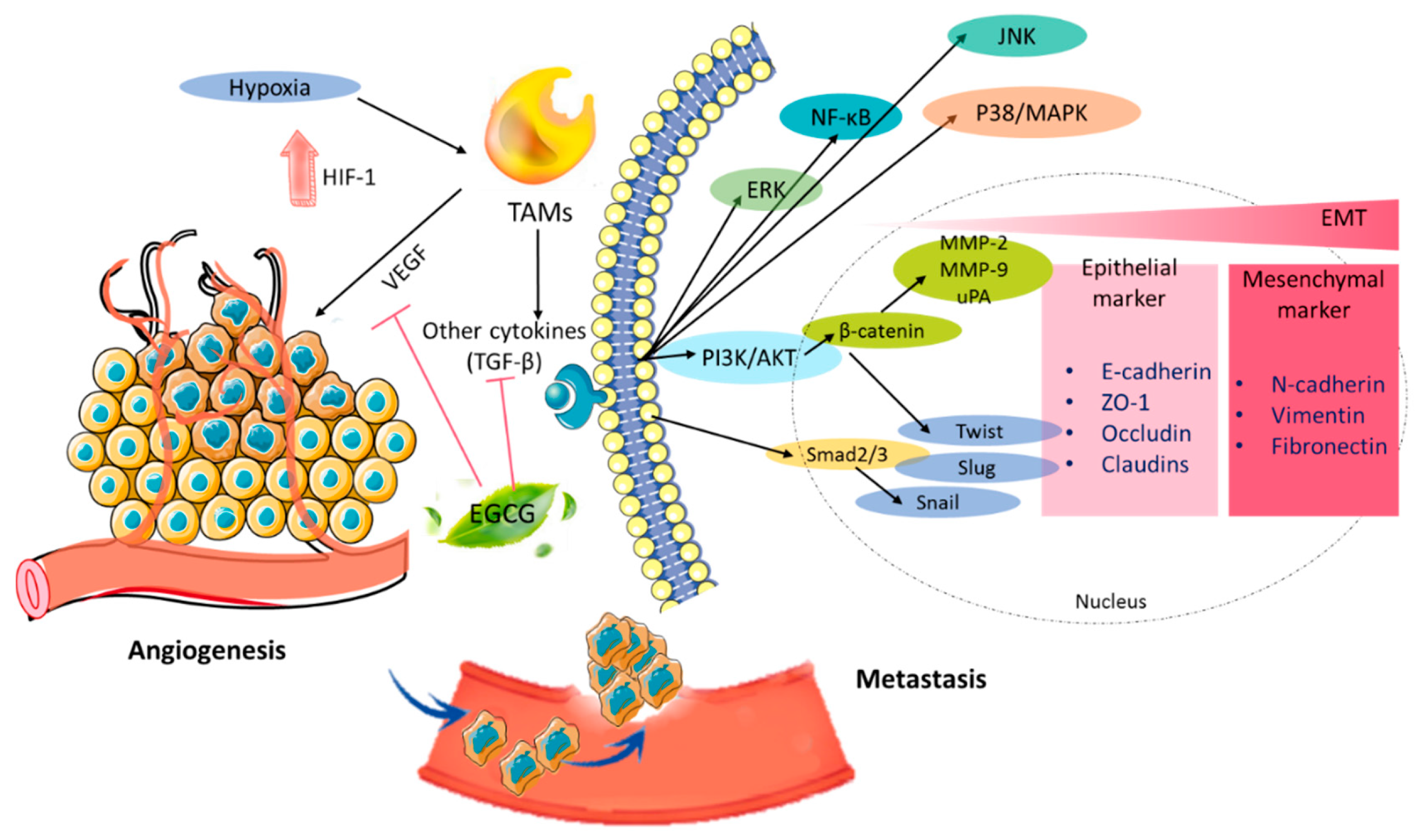
2.4. Antiangiogenesis Effect
2.5. Antimetastatic Activity
2.6. Induction of Apoptosis
2.7. Autophagy and Exosome
2.8. Bioavailability
2.9. Pharmaceutical Synergistic Effect
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EGCG | (−)-epigallocatechin gallate |
| Nrf2 | nuclear factor erythroid 2-related factor 2 |
| NF-Κb | nuclear factor-κB |
| EMT | epithelial–mesenchymal transition |
| DNMTs | DNA methyltransferases |
| HDACs | histone deacetylases |
| EGC | (−)-epigallocatechin |
| ECG | (−)-epicatechin-3-gallate |
| EC | (−)-epicatechin |
| siRNA | small interfering RNA |
| FIGO | Federation of Gynecology and Obstetrics |
| PTEN | phosphatase and tensin homolog |
| TAH-BSO | total abdominal hysterectomy and bilateral salpingo-oophorectomy |
| ER | estrogen receptor |
| PR | progesterone receptor |
| HER2 | human epidermal growth factor receptor 2 |
| COX-2 | cyclooxygenase-2 |
| ROS | reactive oxygen species |
| GPx | glutathione peroxidase |
| GST | glutathione-S-transferase |
| SOD | superoxide dismutase |
| ARE | antioxidant response element |
| HO-1 | heme oxygenase-1 |
| MnSOD | manganese superoxide dismutase |
| ERK1/2 | extracellular signal-regulated kinases ½ |
| KEAP1 | Kelch-like ECH-associated protein |
| HUVEC | human umbilical vein endothelial cells |
| MAP | mitogen-activated protein |
| NO | nitric oxide |
| TNF-α | tumor necrosis factor alpha |
| LPS | lipopolysaccharide |
| AMPK | AMP-activated protein kinase |
| 4-OHT | 4-hydroxytamoxifen |
| HSP90 | heat shock protein 90 |
| NGAL | neutrophil gelatinase-associated lipocalin |
| CRP | C-reactive protein |
| EGFR | epidermal growth factor receptor |
| shRNA | short hairpin RNA |
| GSK3β | glycogen synthase kinase-3β |
| PCNA | proliferating-cell nuclear antigen |
| PARP-1 | poly (ADP-ribose) polymerase 1 |
| Skp2 | S-phase kinase protein 2 |
| SAHA | suberoylanilide hydroxamic acid |
| AQP5 | abnormal expression of aquaporin 5 |
| ERα | estrogen receptor α |
| VEGF | vascular endothelial growth factor |
| HIF-1 | hypoxia-inducible factor |
| TAMs | tumor-associated macrophages |
| TGF-β | transforming growth factor-beta |
| MMPs | metalloproteinases |
| MT1-MMP | type-1-matrix metalloproteinase |
| FAK | focal adhesion kinase |
| JNK | c-jun N-terminal kinase |
| uPA | urokinase-type plasminogen activator |
| ETAR/ET-1 | endothelin A receptor/endothlelin-1 |
| PKC | protein kinase C |
| GLUT-1 | glucose transporter 1 |
| TNBC | triple-negative breast cancer |
| cIAP2 | cellular inhibitor of apoptosis 2 |
| hTERT | human telomerase reverse transcriptase |
| FASN | fatty acid synthase |
| HIF1α | hypoxia-inducible factor 1α |
| MDR1 | multidrug resistance 1 |
| TRAIL | tumor necrosis factor (TNF)-related apoptosis-inducing ligand |
| DNMT2 | DNA methyltransferase-2 |
| DNMT | DNA methyltransferase |
| COMT | catechol-O-methyltransferase |
| VEGFA | vascular endothelial growth factor A |
| PS–PSyox–NPs | polystyrene–polysoyaoil–diethanol amine nanoparticles |
| NLC-RGDs | nanostructured lipid carriers–arginyl-glycyl-aspartic acid |
| CTR1 | copper transporter 1 |
| 5-aza 2′dC | decitabine or 5-aza-2′-deoxycytidine |
References
- Di Chen, S.B.W.; Yang, H.; Yuan, J.; Chan, T.H.; Dou, Q.P. EGCG, green tea polyphenols and their synthetic analogs and prodrugs for human cancer prevention and treatment. Adv. Clin. Chem. 2011, 53, 155. [Google Scholar]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (EGCG): Mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jie, G.; Zhang, J.; Zhao, B. Significant longevity-extending effects of EGCG on Caenorhabditis elegans under stress. Free Radic. Biol. Med. 2009, 46, 414–421. [Google Scholar] [CrossRef]
- Kaya, Z.; Yayla, M.; Cinar, I.; Atila, N.E.; Ozmen, S.; Bayraktutan, Z.; Bilici, D. Epigallocatechin-3-gallate (EGCG) exert therapeutic effect on acute inflammatory otitis media in rats. Int. J. Pediatr. Otorhinolaryngol. 2019, 124, 106–110. [Google Scholar] [CrossRef]
- Avramovich-Tirosh, Y.; Reznichenko, L.; Amit, T.; Zheng, H.; Fridkin, M.; Weinreb, O.; Mandel, S.; Youdim, M.B. Neurorescue activity, APP regulation and amyloid-β peptide reduction by novel multi-functional brain permeable iron-chelating-antioxidants, M-30 and green tea polyphenol, EGCG. Curr. Alzheimer Res. 2007, 4, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Xu, L.; Liang, Q.; Sun, Q.; Chen, C.; Zhang, Y.; Ding, Y.; Zhou, P. Metal chelator EGCG attenuates Fe (III)-induced conformational transition of α-synuclein and protects AS-PC 12 cells against Fe (III)-induced death. J. Neurochem. 2017, 143, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Stuart, E.C.; Scandlyn, M.J.; Rosengren, R.J. Role of epigallocatechin gallate (EGCG) in the treatment of breast and prostate cancer. Life Sci. 2006, 79, 2329–2336. [Google Scholar] [CrossRef] [PubMed]
- Kandil, R.; Merkel, O.M. Recent progress of polymeric nanogels for gene delivery. Curr. Opin. Colloid Interface Sci. 2019, 39, 11–23. [Google Scholar] [CrossRef]
- Fujiki, H.; Sueoka, E.; Watanabe, T.; Suganuma, M. Synergistic enhancement of anticancer effects on numerous human cancer cell lines treated with the combination of EGCG, other green tea catechins, and anticancer compounds. J. Cancer Res. Clin. Oncol. 2015, 141, 1511–1522. [Google Scholar] [CrossRef]
- Lee, C.M.; Szabo, A.; Shrieve, D.C.; Macdonald, O.K.; Gaffney, D.K. Frequency and effect of adjuvant radiation therapy among women with stage I endometrial adenocarcinoma. JAMA 2006, 295, 389–397. [Google Scholar] [CrossRef]
- Kübler, K.; Ayub, T.H.; Weber, S.K.; Zivanovic, O.; Abramian, A.; Keyver-Paik, M.D.; Mallmann, M.R.; Kaiser, C.; Serçe, N.B.; Kuhn, W. Prognostic significance of tumor-associated macrophages in endometrial adenocarcinoma. Gynecol. Oncol. 2014, 135, 176–183. [Google Scholar] [CrossRef]
- Chen, J.; Wang, Y.; McMonechy, M.K.; Anglesio, M.S.; Yang, W.; Senz, J.; Maines-Bandiera, S.; Rosner, J.; Trigo-Gonzalez, G.; Grace Cheng, S. Recurrent DICER1 hotspot mutations in endometrial tumours and their impact on microRNA biogenesis. J. Pathol. 2015, 237, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wendel, J.R.; Emerson, R.E.; Broaddus, R.R.; Creighton, C.J.; Rusch, D.B.; Buechlein, A.; DeMayo, F.J.; Lydon, J.P.; Hawkins, S.M. Pten and Dicer1 loss in the mouse uterus causes poorly differentiated endometrial adenocarcinoma. Oncogene 2020, 39, 6286–6299. [Google Scholar] [CrossRef] [PubMed]
- Kohler, M.F.; Carney, P.; Dodge, R.; Soper, J.T.; Clarke-Pearson, D.L.; Marks, J.R.; Berchuck, A. p53 overexpression in advanced-stage endometrial adenocarcinoma. Am. J. Obstet. Gynecol. 1996, 175, 1246–1252. [Google Scholar] [CrossRef]
- Buchynska, L.; Nesina, I. Expression of the cell cycle regulators p53, p21 (WAF1/CIP1) and p16 (INK4a) in human endometrial adenocarcinoma. Exp. Oncol. 2006, 28, 125. [Google Scholar]
- Wallace, A.E.; Gibson, D.A.; Saunders, P.; Jabbour, H.N. Inflammatory events in endometrial adenocarcinoma. J. Endocrinol. 2010, 206, 141–157. [Google Scholar] [CrossRef]
- Yang, X.R.; Chang-Claude, J.; Goode, E.L.; Couch, F.J.; Nevanlinna, H.; Milne, R.L.; Gaudet, M.; Schmidt, M.K.; Broeks, A.; Cox, A. Associations of breast cancer risk factors with tumor subtypes: A pooled analysis from the Breast Cancer Association Consortium studies. J. Natl. Cancer Inst. 2011, 103, 250–263. [Google Scholar] [CrossRef]
- Bernstein, L. Epidemiology of endocrine-related risk factors for breast cancer. J. Mammary Gland Biol. Neoplasia 2002, 7, 3–15. [Google Scholar] [CrossRef]
- Arun, B.; Goss, P. The role of COX-2 inhibition in breast cancer treatment and prevention. Semin. Oncol. 2004, 31, 22–29. [Google Scholar] [CrossRef]
- Schneider, B.P.; Winer, E.P.; Foulkes, W.D.; Garber, J.; Perou, C.M.; Richardson, A.; Sledge, G.W.; Carey, L.A. Triple-negative breast cancer: Risk factors to potential targets. Clin. Cancer Res. 2008, 14, 8010–8018. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast cancer treatment: A review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef] [PubMed]
- Salehi, F.; Dunfield, L.; Phillips, K.P.; Krewski, D.; Vanderhyden, B.C. Risk factors for ovarian cancer: An overview with emphasis on hormonal factors. J. Toxicol. Environ. Health B Crit. Rev. 2008, 11, 301–321. [Google Scholar] [CrossRef] [PubMed]
- Wentzensen, N.; Poole, E.M.; Trabert, B.; White, E.; Arslan, A.A.; Patel, A.V.; Setiawan, V.W.; Visvanathan, K.; Weiderpass, E.; Adami, H.O. Ovarian cancer risk factors by histologic subtype: An analysis from the ovarian cancer cohort consortium. Int. J. Clin. Oncol. 2016, 34, 2888. [Google Scholar] [CrossRef] [PubMed]
- Sueblinvong, T.; Carney, M.E. Current understanding of risk factors for ovarian cancer. Curr. Treat. Options Oncol. 2009, 10, 67–81. [Google Scholar] [CrossRef]
- Jelovac, D.; Armstrong, D.K. Recent progress in the diagnosis and treatment of ovarian cancer. CA Cancer J. Clin. 2011, 61, 183–203. [Google Scholar] [CrossRef]
- El-Mowafy, A.; Al-Gayyar, M.; Salem, H.; El-Mesery, M.; Darweish, M. Novel chemotherapeutic and renal protective effects for the green tea (EGCG): Role of oxidative stress and inflammatory-cytokine signaling. Phytomedicine 2010, 17, 1067–1075. [Google Scholar] [CrossRef]
- Zhou, H.; Chen, J.X.; Yang, C.S.; Yang, M.Q.; Deng, Y.; Wang, H. Gene regulation mediated by microRNAs in response to green tea polyphenol EGCG in mouse lung cancer. BMC Genom. 2014, 15, S3. [Google Scholar] [CrossRef]
- Manohar, M.; Fatima, I.; Saxena, R.; Chandra, V.; Sankhwar, P.L.; Dwivedi, A. Epigallocatechin-3-gallate induces apoptosis in human endometrial adenocarcinoma cells via ROS generation and p38 MAP kinase activation. J. Nutr. Biochem. 2013, 24, 940–947. [Google Scholar] [CrossRef]
- Zhou, P.; Yu, J.F.; Zhao, C.G.; Sui, F.X.; Teng, X.; Wu, Y.B. Therapeutic potential of EGCG on acute renal damage in a rat model of obstructive nephropathy. Mol. Med. Rep. 2013, 7, 1096–1102. [Google Scholar] [CrossRef]
- Na, H.K.; Surh, Y.J. Modulation of Nrf2-mediated antioxidant and detoxifying enzyme induction by the green tea polyphenol EGCG. Food Chem. Toxicol. 2008, 46, 1271–1278. [Google Scholar] [CrossRef]
- Wang, C.C.; Xu, H.; Man, G.C.W.; Zhang, T.; Chu, K.O.; Chu, C.Y.; Cheng, J.T.Y.; Li, G.; He, Y.X.; Qin, L. Prodrug of green tea epigallocatechin-3-gallate (Pro-EGCG) as a potent anti-angiogenesis agent for endometriosis in mice. Angiogenesis 2013, 16, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Na, H.K.; Kim, E.H.; Jung, J.H.; Lee, H.H.; Hyun, J.W.; Surh, Y.J. (−)-Epigallocatechin gallate induces Nrf2-mediated antioxidant enzyme expression via activation of PI3K and ERK in human mammary epithelial cells. Arch. Biochem. Biophys. 2008, 476, 171–177. [Google Scholar] [CrossRef]
- Sun, W.; Liu, X.; Zhang, H.; Song, Y.; Li, T.; Liu, X.; Liu, Y.; Guo, L.; Wang, F.; Yang, T. Epigallocatechin gallate upregulates NRF2 to prevent diabetic nephropathy via disabling KEAP1. Free Radic. Biol. Med. 2017, 108, 840–857. [Google Scholar] [CrossRef] [PubMed]
- Han, X.D.; Zhang, Y.Y.; Wang, K.L.; Huang, Y.P.; Yang, Z.B.; Liu, Z. The involvement of Nrf2 in the protective effects of (-)-Epigallocatechin-3-gallate (EGCG) on NaAsO2-induced hepatotoxicity. Oncotarget 2017, 8, 65302. [Google Scholar] [CrossRef]
- Xie, H.; Sun, J.; Chen, Y.; Zong, M.; Li, S.; Wang, Y. EGCG attenuates uric acid-induced inflammatory and oxidative stress responses by medicating the NOTCH pathway. Oxid. Med. Cell. Longev. 2015, 2015, 214836. [Google Scholar] [CrossRef]
- Jatuworapruk, K.; Srichairatanakool, S.; Ounjaijean, S.; Kasitanon, N.; Wangkaew, S.; Louthrenoo, W. Effects of green tea extract on serum uric acid and urate clearance in healthy individuals. J. Clin. Rheumatol. 2014, 20, 310–313. [Google Scholar] [CrossRef]
- Park, S.B.; Bae, J.W.; Kim, J.M.; Lee, S.G.; Han, M. Antiproliferative and apoptotic effect of epigallocatechin-3-gallate on Ishikawa cells is accompanied by sex steroid receptor downregulation. Int. J. Mol. Med. 2012, 30, 1211–1218. [Google Scholar] [CrossRef]
- Wang, J.; Man, G.C.W.; Chan, T.H.; Kwong, J.; Wang, C.C. A prodrug of green tea polyphenol (–)-epigallocatechin-3-gallate (Pro-EGCG) serves as a novel angiogenesis inhibitor in endometrial cancer. Cancer Lett. 2018, 412, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Chisholm, K.; Bray, B.; Rosengren, R. Tamoxifen and epigallocatechin gallate are synergistically cytotoxic to MDA-MB-231 human breast cancer cells. Anti-Cancer Drugs 2004, 15, 889–897. [Google Scholar] [CrossRef]
- Kim, J.; Zhang, X.; Rieger-Christ, K.M.; Summerhayes, I.C.; Wazer, D.E.; Paulson, K.E.; Yee, A.S. Suppression of Wnt signaling by the green tea compound (–)-epigallocatechin 3-gallate (EGCG) in invasive breast cancer cells requirement of the transcriptional repressor HBP1. J. Biol. Chem. 2006, 281, 10865–10875. [Google Scholar] [CrossRef]
- Zan, L.; Chen, Q.; Zhang, L.; Li, X. Epigallocatechin gallate (EGCG) suppresses growth and tumorigenicity in breast cancer cells by downregulation of miR-25. Bioengineered 2019, 10, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.A.; Jordan, H.R.; Tollefsbol, T.O. Effects of SAHA and EGCG on growth potentiation of triple-negative breast cancer cells. Cancers 2019, 11, 23. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.Q.; Xu, M.; Zhong, W.T.; Cui, Z.Y.; Liu, F.M.; Zhou, K.Y.; Li, X.Y. EGCG decreases the expression of HIF-1α and VEGF and cell growth in MCF-7 breast cancer cells. J. BUON 2014, 19, 435–439. [Google Scholar]
- Gu, J.W.; Makey, K.L.; Tucker, K.B.; Chinchar, E.; Mao, X.; Pei, I.; Thomas, E.Y.; Miele, L. EGCG, a major green tea catechin suppresses breast tumor angiogenesis and growth via inhibiting the activation of HIF-1α and NFκB, and VEGF expression. Vasc. Cell 2013, 5, 9. [Google Scholar] [CrossRef]
- Sen, T.; Moulik, S.; Dutta, A.; Choudhury, P.R.; Banerji, A.; Das, S.; Roy, M.; Chatterjee, A. Multifunctional effect of epigallocatechin-3-gallate (EGCG) in downregulation of gelatinase-A (MMP-2) in human breast cancer cell line MCF-7. Life Sci. 2009, 84, 194–204. [Google Scholar] [CrossRef]
- Sen, T.; Dutta, A.; Chatterjee, A. Epigallocatechin-3-gallate (EGCG) downregulates gelatinase-B (MMP-9) by involvement of FAK/ERK/NFκB and AP-1 in the human breast cancer cell line MDA-MB-231. Anticancer Drugs 2010, 21, 632–644. [Google Scholar] [CrossRef]
- Sen, T.; Chatterjee, A. Epigallocatechin-3-gallate (EGCG) downregulates EGF-induced MMP-9 in breast cancer cells: Involvement of integrin receptor α5β1 in the process. Eur. J. Nutr. 2011, 50, 465–478. [Google Scholar] [CrossRef]
- Moradzadeh, M.; Hosseini, A.; Erfanian, S.; Rezaei, H. Epigallocatechin-3-gallate promotes apoptosis in human breast cancer T47D cells through down-regulation of PI3K/AKT and Telomerase. Pharmacol. Rep. 2017, 69, 924–928. [Google Scholar] [CrossRef]
- Wei, R.; Mao, L.; Xu, P.; Zheng, X.; Hackman, R.M.; Mackenzie, G.G.; Wang, Y. Suppressing glucose metabolism with epigallocatechin-3-gallate (EGCG) reduces breast cancer cell growth in preclinical models. Food Funct. 2018, 9, 5682–5696. [Google Scholar] [CrossRef]
- Steed, K.L.; Jordan, H.R.; Tollefsbol, T.O. SAHA and EGCG promote apoptosis in triple-negative breast cancer cells, possibly through the modulation of cIAP2. Anticancer Res. 2020, 40, 9–26. [Google Scholar] [CrossRef]
- Puig, T.; Vázquez-Martín, A.; Relat, J.; Pétriz, J.; Menéndez, J.A.; Porta, R.; Casals, G.; Marrero, P.F.; Haro, D.; Brunet, J. Fatty acid metabolism in breast cancer cells: Differential inhibitory effects of epigallocatechin gallate (EGCG) and C75. Breast Cancer Res. Treat. 2008, 109, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Han, Z.; Li, X.; Xie, H.H.; Zhu, S.S. Mechanism of EGCG promoting apoptosis of MCF-7 cell line in human breast cancer. Oncol. Lett. 2017, 14, 3623–3627. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Pileczki, V.; Pop, L.; Petric, R.C.; Chira, S.; Pointiere, E.; Achimas-Cadariu, P.; Berindan-Neagoe, I. Dual targeted therapy with p53 siRNA and Epigallocatechingallate in a triple negative breast cancer cell model. PLoS ONE 2015, 10, e0120936. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Yan, J.; Luo, L.; Ma, M.; Zhu, H. Preparation and characterization of (−)-Epigallocatechin-3-gallate (EGCG)-loaded nanoparticles and their inhibitory effects on Human breast cancer MCF-7 cells. Sci. Rep. 2017, 7, 45521. [Google Scholar] [CrossRef]
- Huang, H.C.; Way, T.D.; Lin, C.L.; Lin, J.K. EGCG stabilizes p27kip1 in E2-stimulated MCF-7 cells through down-regulation of the Skp2 protein. Endocrinology 2008, 149, 5972–5983. [Google Scholar] [CrossRef]
- Guo, S.; Yang, S.; Taylor, C.; Sonenshein, G.E. Green tea polyphenol epigallocatechin-3 gallate (EGCG) affects gene expression of breast cancer cells transformed by the carcinogen 7, 12-dimethylbenz [a] anthracene. J. Nutr. 2005, 135, 2978S–2986S. [Google Scholar] [CrossRef]
- Yan, C.; Yang, J.; Shen, L.; Chen, X. Inhibitory effect of Epigallocatechin gallate on ovarian cancer cell proliferation associated with aquaporin 5 expression. Arch. Gynecol. Obstet. 2012, 285, 459–467. [Google Scholar] [CrossRef]
- Huh, S.W.; Bae, S.M.; Kim, Y.W.; Lee, J.M.; Namkoong, S.E.; Lee, I.P.; Kim, S.H.; Kim, C.K.; Ahn, W.S. Anticancer effects of (−)-epigallocatechin-3-gallate on ovarian carcinoma cell lines. Gynecol. Oncol. 2004, 94, 760–768. [Google Scholar] [CrossRef]
- Qin, J.; Fu, M.; Wang, J.; Huang, F.; Liu, H.; Huangfu, M.; Yu, D.; Liu, H.; Li, X.; Guan, X. PTEN/AKT/mTOR signaling mediates anticancer effects of epigallocatechin-3-gallate in ovarian cancer. Oncol. Rep. 2020, 43, 1885–1896. [Google Scholar] [CrossRef]
- Spinella, F.; Rosano, L.; Di Castro, V.; Decandia, S.; Albini, A.; Nicotra, M.R.; Natali, P.G.; Bagnato, A. Green tea polyphenol epigallocatechin-3-gallate inhibits the endothelin axis and downstream signaling pathways in ovarian carcinoma. Mol. Cancer Ther. 2006, 5, 1483–1492. [Google Scholar] [CrossRef]
- Laschke, M.W.; Schwender, C.; Scheuer, C.; Vollmar, B.; Menger, M.D. Epigallocatechin-3-gallate inhibits estrogen-induced activation of endometrial cells in vitro and causes regression of endometriotic lesions in vivo. Hum. Reprod. 2008, 23, 2308–2318. [Google Scholar] [CrossRef]
- Khalil, H.; Tazi, M.; Caution, K.; Ahmed, A.; Kanneganti, A.; Assani, K.; Kopp, B.; Marsh, C.; Dakhlallah, D.; Amer, A.O. Aging is associated with hypermethylation of autophagy genes in macrophages. Epigenetics 2016, 11, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Novilla, A.; Djamhuri, D.S.; Nurhayati, B.; Rihibiha, D.D.; Afifah, E.; Widowati, W. Anti-inflammatory properties of oolong tea (Camellia sinensis) ethanol extract and epigallocatechin gallate in LPS-induced RAW 264.7 cells. Asian Pac. J. Trop. Biomed. 2017, 7, 1005–1009. [Google Scholar] [CrossRef]
- Liu, J.B.; Zhou, L.; Wang, Y.Z.; Wang, X.; Zhou, Y.; Ho, W.Z.; Li, J.L. Neuroprotective Activity of ()-Epigallocatechin Gallate against Lipopolysaccharide-Mediated Cytotoxicity. J. Immunol. Res. 2016, 2016, 4962351. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Jing, D.; Shi, M.; Liu, Y.; Lin, T.; Xie, Z.; Zhu, Y.; Zhao, H.; Shi, X.; Du, F. Epigallocatechin gallate (EGCG) attenuates infrasound-induced neuronal impairment by inhibiting microglia-mediated inflammation. J. Nutr. Biochem. 2014, 25, 716–725. [Google Scholar] [CrossRef] [PubMed]
- Peairs, A.; Dai, R.; Gan, L.; Shimp, S.; Rylander, M.N.; Li, L.; Reilly, C.M. Epigallocatechin-3-gallate (EGCG) attenuates inflammation in MRL/lpr mouse mesangial cells. Cell. Mol. Immunol. 2010, 7, 123–132. [Google Scholar] [CrossRef]
- Rasheed, N.O.A.; Ahmed, L.A.; Abdallah, D.M.; El-Sayeh, B.M. Nephro-toxic effects of intraperitoneally injected EGCG in diabetic mice: Involvement of oxidative stress, inflammation and apoptosis. Sci. Rep. 2017, 7, 40617. [Google Scholar] [CrossRef]
- Stuart, E.C.; Rosengren, R.J. The combination of raloxifene and epigallocatechin gallate suppresses growth and induces apoptosis in MDA-MB-231 cells. Life Sci. 2008, 82, 943–948. [Google Scholar] [CrossRef]
- Niu, Y.; Na, L.; Feng, R.; Gong, L.; Zhao, Y.; Li, Q.; Li, Y.; Sun, C. The phytochemical, EGCG, extends lifespan by reducing liver and kidney function damage and improving age-associated inflammation and oxidative stress in healthy rats. Aging Cell 2013, 12, 1041–1049. [Google Scholar] [CrossRef]
- Wei, R.; Wirkus, J.; Yang, Z.; Machuca, J.; Esparza, Y.; Mackenzie, G.G. EGCG sensitizes chemotherapeutic-induced cytotoxicity by targeting the ERK pathway in multiple cancer cell lines. Arch. Biochem. Biophys. 2020, 692, 108546. [Google Scholar] [CrossRef]
- Zhang, L.; Xie, J.; Gan, R.; Wu, Z.; Luo, H.; Chen, X.; Lu, Y.; Wu, L.; Zheng, D. Synergistic inhibition of lung cancer cells by EGCG and NF-κB inhibitor BAY11-7082. J. Cancer 2019, 10, 6543. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lei, Z.; Huang, Z.; Zhang, X.; Zhou, Y.; Luo, Z.; Zeng, W.; Su, J.; Peng, C.; Chen, X. Epigallocatechin-3-gallate (EGCG) suppresses melanoma cell growth and metastasis by targeting TRAF6 activity. Oncotarget 2016, 7, 79557. [Google Scholar] [CrossRef] [PubMed]
- Harakeh, S.; Diab-Assaf, M.; Azar, R.; Hassan, H.; Tayeb, S.; Abou-El-Ardat, K.; Damanhouri, G.A.; Qadri, I.; Abuzenadah, A.; Chaudhary, A. Epigallocatechin-3-gallate inhibits tax-dependent activation of nuclear factor kappa B and of matrix metalloproteinase 9 in human T-cell lymphotropic virus-1 positive leukemia cells. Asian Pac. J. Cancer Prev. 2014, 15, 1219–1225. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Pezzani, R.; Redaelli, M.; Zorzan, M.; Imran, M.; Ahmed Khalil, A.; Salehi, B.; Sharopov, F.; Cho, W.C.; Sharifi-Rad, J. Preclinical Pharmacological Activities of Epigallocatechin-3-gallate in Signaling Pathways: An Update on Cancer. Molecules 2020, 25, 467. [Google Scholar] [CrossRef]
- Yang, C.; Du, W.; Yang, D. Inhibition of green tea polyphenol EGCG ((−)-epigallocatechin-3-gallate) on the proliferation of gastric cancer cells by suppressing canonical wnt/β-catenin signalling pathway. Int. J. Food Sci. Nutr. 2016, 67, 818–827. [Google Scholar] [CrossRef]
- Pahlke, G.; Ngiewih, Y.; Kern, M.; Jakobs, S.; Marko, D.; Eisenbrand, G. Impact of quercetin and EGCG on key elements of the Wnt pathway in human colon carcinoma cells. J. Agric. Food Chem. 2006, 54, 7075–7082. [Google Scholar] [CrossRef]
- Rao, X.; Di Leva, G.; Li, M.; Fang, F.; Devlin, C.; Hartman-Frey, C.; Burow, M.; Ivan, M.; Croce, C.M.; Nephew, K. MicroRNA-221/222 confers breast cancer fulvestrant resistance by regulating multiple signaling pathways. Oncogene 2011, 30, 1082–1097. [Google Scholar] [CrossRef]
- Siddiqui, I.A.; Bharali, D.J.; Nihal, M.; Adhami, V.M.; Khan, N.; Chamcheu, J.C.; Khan, M.I.; Shabana, S.; Mousa, S.A.; Mukhtar, H. Excellent anti-proliferative and pro-apoptotic effects of (−)-epigallocatechin-3-gallate encapsulated in chitosan nanoparticles on human melanoma cell growth both in vitro and in vivo. Nanomedicine 2014, 10, 1619–1626. [Google Scholar] [CrossRef]
- Lazzeroni, M.; Guerrieri-Gonzaga, A.; Gandini, S.; Johansson, H.; Serrano, D.; Cazzaniga, M.; Aristarco, V.; Macis, D.; Mora, S.; Caldarella, P. A presurgical study of lecithin formulation of green tea extract in women with early breast cancer. Cancer Prev. Res. 2017, 10, 363–370. [Google Scholar] [CrossRef]
- Chen, H.; Landen, C.N.; Li, Y.; Alvarez, R.D.; Tollefsbol, T.O. Epigallocatechin gallate and sulforaphane combination treatment induce apoptosis in paclitaxel-resistant ovarian cancer cells through hTERT and Bcl-2 down-regulation. Exp. Cell Res. 2013, 319, 697–706. [Google Scholar] [CrossRef]
- Kiselev, V.I.; Ashrafyan, L.A.; Muyzhnek, E.L.; Gerfanova, E.V.; Antonova, I.B.; Aleshikova, O.I.; Sarkar, F.H. A new promising way of maintenance therapy in advanced ovarian cancer: A comparative clinical study. BMC Cancer 2018, 18, 904. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Li, C.; Xu, Y.; Wang, L.; Liu, J.; Wang, D.; Hong, C.; Jiang, Z.; Ma, Y.; Chen, Q. Epigallocatechin gallate promotes p53 accumulation and activity via the inhibition of MDM2-mediated p53 ubiquitination in human lung cancer cells. Oncol. Rep. 2013, 29, 1983–1990. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.; Wang, F.; Wan, J. MicroRNA-381 inhibits cell proliferation and invasion in endometrial carcinoma by targeting the IGF-1R. Mol. Med. Rep. 2018, 17, 4090–4098. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.; Johnson, R.S. Hypoxia: A key regulator of angiogenesis in cancer. Cancer Metastasis Rev. 2007, 26, 281–290. [Google Scholar] [CrossRef]
- Dann, J.M.; Sykes, P.H.; Mason, D.R.; Evans, J.J. Regulation of vascular endothelial growth factor in endometrial tumour cells by resveratrol and EGCG. Gynecol. Oncol. 2009, 113, 374–378. [Google Scholar] [CrossRef]
- Evans, J.; Chitcholtan, K.; Dann, J.; Guilford, P.; Harris, G.; Lewis, L.; Nagase, J.; Welkamp, A.; Zwerus, R.; Sykes, P. Adrenomedullin interacts with VEGF in endometrial cancer and has varied modulation in tumours of different grades. Gynecol. Oncol. 2012, 125, 214–219. [Google Scholar] [CrossRef]
- McConkey, D.J.; Choi, W.; Marquis, L.; Martin, F.; Williams, M.B.; Shah, J.; Svatek, R.; Das, A.; Adam, L.; Kamat, A. Role of epithelial-to-mesenchymal transition (EMT) in drug sensitivity and metastasis in bladder cancer. Cancer Metastasis Rev. 2009, 28, 335–344. [Google Scholar] [CrossRef]
- Adam, L.; Zhong, M.; Choi, W.; Qi, W.; Nicoloso, M.; Arora, A.; Calin, G.; Wang, H.; Siefker-Radtke, A.; McConkey, D. miR-200 expression regulates epithelial-to-mesenchymal transition in bladder cancer cells and reverses resistance to epidermal growth factor receptor therapy. Clin. Cancer Res. 2009, 15, 5060–5072. [Google Scholar] [CrossRef]
- Shankar, S.; Marsh, L.; Srivastava, R.K. EGCG inhibits growth of human pancreatic tumors orthotopically implanted in Balb C nude mice through modulation of FKHRL1/FOXO3a and neuropilin. Mol. Cell. Biochem. 2013, 372, 83–94. [Google Scholar] [CrossRef]
- Liu, J.D.; Chen, S.H.; Lin, C.L.; Tsai, S.H.; Liang, Y.C. Inhibition of melanoma growth and metastasis by combination with (−)-epigallocatechin-3-gallate and dacarbazine in mice. J. Cell. Biochem. 2001, 83, 631–642. [Google Scholar] [CrossRef]
- Deng, Y.T.; Lin, J.K. EGCG inhibits the invasion of highly invasive CL1-5 lung cancer cells through suppressing MMP-2 expression via JNK signaling and induces G2/M arrest. J. Agric. Food Chem. 2011, 59, 13318–13327. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Ganapathy, S.; Hingorani, S.R.; Srivastava, R.K. EGCG inhibits growth, invasion, angiogenesis and metastasis of pancreatic cancer. Front. Biosci. 2008, 13, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Banys-Paluchowski, M.; Witzel, I.; Aktas, B.; Fasching, P.A.; Hartkopf, A.; Janni, W.; Kasimir-Bauer, S.; Pantel, K.; Schön, G.; Rack, B. The prognostic relevance of urokinase-type plasminogen activator (uPA) in the blood of patients with metastatic breast cancer. Sci. Rep. 2019, 9, 2318. [Google Scholar] [CrossRef]
- Lim, Y.C.; Park, H.Y.; Hwang, H.S.; Kang, S.U.; Pyun, J.H.; Lee, M.H.; Choi, E.C.; Kim, C.H. (−)-Epigallocatechin-3-gallate (EGCG) inhibits HGF-induced invasion and metastasis in hypopharyngeal carcinoma cells. Cancer Lett. 2008, 271, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Cortez Penso, N.E.; Hackman, R.M.; Wang, Y.; Mackenzie, G.G. Epigallocatechin-3-Gallate (EGCG) Suppresses Pancreatic Cancer Cell Growth, Invasion, and Migration partly through the Inhibition of Akt Pathway and Epithelial–Mesenchymal Transition: Enhanced Efficacy When Combined with Gemcitabine. Nutrients 2019, 11, 1856. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.T.; Ha, J.; Park, I.J.; Lee, S.K.; Baik, H.W.; Kim, Y.M.; Park, O.J. Apoptotic effect of EGCG in HT-29 colon cancer cells via AMPK signal pathway. Cancer Lett. 2007, 247, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Meeran, S.M.; Patel, S.N.; Chan, T.H.; Tollefsbol, T.O. A novel prodrug of epigallocatechin-3-gallate: Differential epigenetic hTERT repression in human breast cancer cells. Cancer Prev. Res. 2011, 4, 1243–1254. [Google Scholar] [CrossRef] [PubMed]
- Kang, Q.; Zhang, X.; Cao, N.; Chen, C.; Yi, J.; Hao, L.; Ji, Y.; Liu, X.; Lu, J. EGCG enhances cancer cells sensitivity under 60Coγ radiation based on miR-34a/Sirt1/p53. Food Chem. Toxicol. 2019, 133, 110807. [Google Scholar] [CrossRef]
- Mittal, A.; Pate, M.S.; Wylie, R.C.; Tollefsbol, T.O.; Katiyar, S.K. EGCG down-regulates telomerase in human breast carcinoma MCF-7 cells, leading to suppression of cell viability and induction of apoptosis. Int. J. Oncol. 2004, 24, 703–710. [Google Scholar] [CrossRef]
- Wang, X.; Hao, M.W.; Dong, K.; Lin, F.; Ren, J.H.; Zhang, H.Z. Apoptosis induction effects of EGCG in laryngeal squamous cell carcinoma cells through telomerase repression. Arch. Pharm. Res. 2009, 32, 1263–1269. [Google Scholar] [CrossRef]
- Zhang, X.; Mar, V.; Zhou, W.; Harrington, L.; Robinson, M.O. Telomere shortening and apoptosis in telomerase-inhibited human tumor cells. Genes Dev. 1999, 13, 2388–2399. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, I.; Malik, A.; Adhami, V.; Asim, M.; Hafeez, B.; Sarfaraz, S.; Mukhtar, H. Green tea polyphenol EGCG sensitizes human prostate carcinoma LNCaP cells to TRAIL-mediated apoptosis and synergistically inhibits biomarkers associated with angiogenesis and metastasis. Oncogene 2008, 27, 2055–2063. [Google Scholar] [CrossRef] [PubMed]
- Qanungo, S.; Das, M.; Haldar, S.; Basu, A. Epigallocatechin-3-gallate induces mitochondrial membrane depolarization and caspase-dependent apoptosis in pancreatic cancer cells. Carcinogenesis 2005, 26, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Wei, F.; Wang, Y.; Wu, B.; Fang, Y.; Xiong, B. EGCG synergizes the therapeutic effect of cisplatin and oxaliplatin through autophagic pathway in human colorectal cancer cells. J. Pharmacol. Sci. 2015, 128, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Farah, B.L.; Sinha, R.A.; Wu, Y.; Singh, B.K.; Bay, B.H.; Yang, C.S.; Yen, P.M. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, stimulates hepatic autophagy and lipid clearance. PLoS ONE 2014, 9, e87161. [Google Scholar] [CrossRef]
- Niedzwiecki, A.; Bhanap, B.; Roomi, M.W.; Rath, M. Expanding Metabolic Targets in Cancer by Select Combinations of Vitamin C and EGCG with Different Natural Compounds. In Nutraceuticals in Veterinary Medicine; Springer: Berlin/Heidelberg, Germany, 2019; pp. 611–624. [Google Scholar]
- Huo, C.; Yang, H.; Cui, Q.C.; Dou, Q.P.; Chan, T.H. Proteasome inhibition in human breast cancer cells with high catechol-O-methyltransferase activity by green tea polyphenol EGCG analogs. Bioorg. Med. Chem. 2010, 18, 1252–1258. [Google Scholar] [CrossRef]
- Landis-Piwowar, K.; Chen, D.; Chan, T.H.; Dou, Q.P. Inhibition of catechol-O-methyltransferase activity in human breast cancer cells enhances the biological effect of the green tea polyphenol (-)-EGCG. Oncol. Rep. 2010, 24, 563–569. [Google Scholar]
- Karahaliloğlu, Z.; Kilicay, E.; Alpaslan, P.; Hazer, B.; Baki Denkbas, E. Enhanced antitumor activity of epigallocatechin gallate–conjugated dual-drug-loaded polystyrene–polysoyaoil–diethanol amine nanoparticles for breast cancer therapy. J. Bioact. Compat. Polym. 2018, 33, 38–62. [Google Scholar] [CrossRef]
- Hajipour, H.; Hamishehkar, H.; Nazari Soltan Ahmad, S.; Barghi, S.; Maroufi, N.F.; Taheri, R.A. Improved anticancer effects of epigallocatechin gallate using RGD-containing nanostructured lipid carriers. Artif. Cells Nanomed. Biotechnol. 2018, 46, 283–292. [Google Scholar] [CrossRef]
- Alizadeh, L.; Alizadeh, E.; Zarebkohan, A.; Ahmadi, E.; Rahmati-Yamchi, M.; Salehi, R. AS1411 aptamer-functionalized chitosan-silica nanoparticles for targeted delivery of epigallocatechin gallate to the SKOV-3 ovarian cancer cell lines. J. Nanopart. Res. 2020, 22, 5. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, P.; Wang, P.; Yang, C.S.; Wang, X.; Feng, Q. EGCG enhances cisplatin sensitivity by regulating expression of the copper and cisplatin influx transporter CTR1 in ovary cancer. PLoS ONE 2015, 10, e0125402. [Google Scholar]
- Chan, M.M.; Soprano, K.J.; Weinstein, K.; Fong, D. Epigallocatechin-3-gallate delivers hydrogen peroxide to induce death of ovarian cancer cells and enhances their cisplatin susceptibility. J. Cell. Physiol. 2006, 207, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, M.E.H.; Beale, P.; Chan, C.; Yu, J.Q.; Huq, F. Epigallocatechin gallate acts synergistically in combination with cisplatin and designed trans-palladiums in ovarian cancer cells. Anticancer Res. 2012, 32, 4851–4860. [Google Scholar]
- Tyagi, T.; Treas, J.N.; Mahalingaiah, P.K.S.; Singh, K.P. Potentiation of growth inhibition and epigenetic modulation by combination of green tea polyphenol and 5-aza-2′-deoxycytidine in human breast cancer cells. Breast Cancer Res. Treat. 2015, 149, 655–668. [Google Scholar] [CrossRef]
| Cancers | Cell lines | EGCG Concentrations | Anticancer Actions | Potential Molecular Mechanisms |
|---|---|---|---|---|
| Endometrial cancer | HEK-293 and Ishikawa cells | 100, 125, and 150 µM | Inhibit cell proliferation and induce apoptosis | Activation of the P38 MAP kinase [28] |
| Ishikawa cells | 100 μM | Inhibit cell proliferation and induces apoptosis | Inhibition of the AKT and MAPK signal pathways [37] | |
| AN3CA, RL95-2, THP-1, and PHES | Pro-(peracetate) 20, 40, and 60 µM | Inhibit tumor angiogenesis | Downregulation of HIF1α/VEGFA through the PI3K/AKT/mTOR/HIF1α pathway [38] | |
| Breast cancer | MCF-7 | 100 μM | Antioxidant Induce apoptosis | Inhibition of the Nrf2 signaling pathway [31] |
| MCF-7, T47D, MDA-MB-231, and HS578T | 5–25 μM | Evaluation of cytotoxicity [39] | ||
| MDA-MB-231 | 25–100 μM | Inhibit invasion | Inhibition of Wnt signaling and target gene c-MYC [40] | |
| MCF-7 | 5 and 20 μg/mL | Induce apoptosis | Upregulation of caspase 3, caspase 9, and PARP [41] | |
| MCF-7, MDA-MB-157, MDA-MB-231, and HCC1806 | 5 μM | Inhibit migration | Inhibition of N-cadherin and increase of E-cadherin [42] | |
| MCF-7 | 25, 50, and 100 mg/L | Inhibit proliferation | Downregulation of HIF-1α and VEGF [43] | |
| E0771, MCF-7, and MDA-MB-231 | 10, 20, and 50 ug/mL | Inhibit proliferation and migration | Downregulation of HIF-1α and NF-κB [44] | |
| MCF-7 | 5, 10, and 20 μM | Inhibit invasion | Downregulation of the PI3K/ERK/NF-κB pathway [45] | |
| MDA-MB-231 | 20 μM | Inhibit invasion | Downregulation of the FAK/ERK/NF-κB pathway [46] | |
| MDA-MB-231 | 10 and 20 μM | Inhibit invasion | Downregulation of the FAK/PI3K/AKT pathway [47] | |
| T47D | 0–80 μM | Induce apoptosis | Downregulation of the PI3K/AKT pathway [48] | |
| 4T1 | 10–320 μM | Induce apoptosis | Upregulation of caspase 3, caspase 8, and caspase 9 [49] | |
| MCF-7, MDA-MB-157, MDA-MB-231, and HCC1806 | 5 μM | Inhibit metastasis | Increase proapoptotic caspase 7 [50] | |
| MCF-7, MDA-MB-231, and SK-Br3 | 20–150 μM | Induce apoptosis | Inhibition of FASN activity and downregulation of the ERK/AKT pathway [51] | |
| MCF-7 | 20~120 μmol/L | Induce apoptosis | Downregulation of the P53/Bcl-2 signaling pathway [52] | |
| Hs578T | 40 nmol | Induce apoptosis Activation autophagy | Target apoptotic and angiogenic pathways [53] | |
| MCF-7 | Nanoparticle 200 μg/mL | Inhibit proliferation | Regulation of the PI3K-Akt pathway [54] | |
| MCF-7 | 10 μM | Inhibit proliferation | Downregulation of Skp2 [55] | |
| DMBA-transformed human D3–1 | 60 μg/mL | Inhibit angiopoietin | Alteration expression related to nuclear and cytoplasmic transport, transformation, and redox signaling [56] | |
| Ovarian cancer | SKOV3 | 20–100 μg/ mL | Inhibit proliferation and induce apoptosis | Downregulation of AQP5, NF-κB, p65, and IκB-α [57] |
| SKOV-3, OVCAR-3, and PA-1 | 25, 50, and 100 µM | Induce apoptosis | Upregulation of P21 and Bax and downregulation of BCL-XL and PCNA [58] | |
| SKOV3, CAOV-3, and NIH-OVCAR-3 | 5, 10, 20, 40, and 80 µg/mL | Inhibit proliferation and induce apoptosis | Upregulation of Bax and caspase-3 and downregulation of Bcl-2 [59] | |
| HEY and OVCA 433 | 20–40 μmol/L | Inhibit proliferation and induce apoptosis | Downregulation of ETAR-dependent signaling pathways [60] |
| Cancers | Animal models | EGCG treatments | Potential molecular mechanisms |
|---|---|---|---|
| Endometrial cancer | Transgenic luciferase-expressing mice (CMV-Luc) | EGCG and Pro-(EGCG octaacetate) 50 mg/kg | Inhibits tumor growth and angiogenesis [31] |
| Female Syrian golden hamsters | 65 mg/kg | Inhibits VEGF expression [61] | |
| Breast cancer | Female C57BL/6 mice | 50-100 mg/kg | Inhibits tumor VEGF expression [44] |
| C57BL/6 aged (62-64 weeks old) and young (8 weeks old) mice | 300 μg/30 μl DMSO | Inhibits DNA methyltransferase 2 (DNMT2) methylation activity [62] | |
| Ovarian cancer | Female BALB/c nude mice | 50 mg/kg | Inhibits tumor growth by regulating the PTEN/AKT/mTOR pathway [59] |
| Female athymic (nu+/nu+) mice | 12.4 g/L | Inhibition of tumor growth by the reduction of ETAR and ET-1 expression [60] |
| Cancer | Cell Lines | EGCG Treatments | Drugs | Cytotoxic Action |
|---|---|---|---|---|
| Breast cancer | MDA-MB-231 | 25 µM | raloxifene | Induce apoptosis [68] |
| MCF-7 and MDA-MB 231 | 50 µM | 5-aza 2′dC | Changes in DNA methylation and histone modifications [115] | |
| Ovarian cancer | SKOV3-ip1 and SKOV3TR-ip2 (paclitaxel-sensitive and -resistant) | 5, 10, 20, and 30 µM | Paclitaxel | Induce apoptosis by the downregulation of Bcl-2 [80] |
| OVCAR3, SKOV3, and HEK-293T cells | 10 μM | Cisplatin | Upregulation of CTR1 and increase cDDP accumulation [112] | |
| CAOV3, SKOV3, OVCAR3, OVCAR10, A2780, CP70, C30, and C200 | 6.3, 12, 25, and 50 µM | Cisplatin | Increase in intracellular oxidative stress [113] | |
| A2780 (cisplatin-sensitive, parental cell line), A2780cisR (cisplatin-resistant), and A2780ZD0473R (ZD0473-resistant) | 1.37–21.98 μM and 1.33–21.34 μM | Cisplatin | Accumulation of platinum and a level of platinum−DNA binding [114] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-J.; Wang, K.-L.; Chen, H.-Y.; Chiang, Y.-F.; Hsia, S.-M. Protective Effects of Epigallocatechin Gallate (EGCG) on Endometrial, Breast, and Ovarian Cancers. Biomolecules 2020, 10, 1481. https://doi.org/10.3390/biom10111481
Huang Y-J, Wang K-L, Chen H-Y, Chiang Y-F, Hsia S-M. Protective Effects of Epigallocatechin Gallate (EGCG) on Endometrial, Breast, and Ovarian Cancers. Biomolecules. 2020; 10(11):1481. https://doi.org/10.3390/biom10111481
Chicago/Turabian StyleHuang, Yun-Ju, Kai-Lee Wang, Hsin-Yuan Chen, Yi-Fen Chiang, and Shih-Min Hsia. 2020. "Protective Effects of Epigallocatechin Gallate (EGCG) on Endometrial, Breast, and Ovarian Cancers" Biomolecules 10, no. 11: 1481. https://doi.org/10.3390/biom10111481
APA StyleHuang, Y.-J., Wang, K.-L., Chen, H.-Y., Chiang, Y.-F., & Hsia, S.-M. (2020). Protective Effects of Epigallocatechin Gallate (EGCG) on Endometrial, Breast, and Ovarian Cancers. Biomolecules, 10(11), 1481. https://doi.org/10.3390/biom10111481






