Curcumin against Prostate Cancer: Current Evidence
Abstract
1. Introduction
1.1. Prostate Cancer
1.2. Curcumin
2. Effects of Curcumin on Prostate Cancer Cells In Vitro
2.1. Androgen-Sensitive Prostate Cancer Cells
2.2. Androgen-Insensitive Prostate Cancer Cells
2.3. Prostate Cancer Stem Cells
3. Effects of Curcumin on Prostate Cancer In Vivo
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Proto-Oncogenes and Tumor-Suppressor Genes. In Molecular Cell Biology, 4th ed.; W. H. Freeman and Company: New York, NY, USA, 2000. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Sarkar, S.; Horn, G.; Moulton, K.; Oza, A.; Byler, S.; Kokolus, S.; Longacre, M. Cancer Development, Progression, and Therapy: An Epigenetic Overview. Int. J. Mol. Sci. 2013, 14, 21087–21113. [Google Scholar] [CrossRef] [PubMed]
- Mattiuzzi, C.; Lippi, G. Current Cancer Epidemiology. J. Epidemiol. Glob. Health 2019, 9, 217–222. [Google Scholar] [CrossRef]
- Leslie, S.W.; Soon-Sutton, T.L.; Sajjad, H.; Siref, L.E. Prostate Cancer. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef] [PubMed]
- Davey, R.A.; Grossmann, M. Androgen Receptor Structure, Function and Biology: From Bench to Bedside. Clin. Biochem. Rev. 2016, 37, 3–15. [Google Scholar] [PubMed]
- Yang, Q.; Fung, K.-M.; Day, W.V.; Kropp, B.P.; Lin, H.-K. Androgen receptor signaling is required for androgen-sensitive human prostate cancer cell proliferation and survival. Cancer Cell Int. 2005, 5, 8–18. [Google Scholar] [CrossRef]
- Jenster, G. The role of the androgen receptor in the development and progression of prostate cancer. Semin. Oncol. 1999, 26, 407–421. [Google Scholar]
- Ramsay, A.K.; Leung, H.Y. Signalling pathways in prostate carcinogenesis: Potentials for molecular-targeted therapy. Clin. Sci. 2009, 117, 209–228. [Google Scholar] [CrossRef]
- Sharifi, N.; Gulley, J.L.; Dahut, W.L. Androgen Deprivation Therapy for Prostate Cancer. JAMA 2005, 294, 238–244. [Google Scholar] [CrossRef]
- Feldman, B.J.; Feldman, D. The development of androgen-independent prostate cancer. Nat. Rev. Cancer 2001, 1, 34–45. [Google Scholar] [CrossRef]
- Lawrentschuk, N.; Trottier, G.; Kuk, C.; Zlotta, A.R. Role of surgery in high-risk localized prostate cancer. Curr. Oncol. 2010, 17, 25–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bill-Axelson, A.; Holmberg, L.; Ruutu, M.; Garmo, H.; Stark, J.R.; Busch, C.; Nordling, S.; Häggman, M.; Andersson, S.-O.; Bratell, S.; et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N. Engl. J. Med. 2011, 364, 1708–1717. [Google Scholar] [CrossRef] [PubMed]
- Hayden, A.J.; Catton, C.; Pickles, T. Radiation therapy in prostate cancer: A risk-adapted strategy. Curr. Oncol. 2010, 17 (Suppl. S2), 18–24. [Google Scholar] [CrossRef] [PubMed]
- Morton, G.C.; Hoskin, P.J. Brachytherapy: Current status and future strategies—Can high dose rate replace low dose rate and external beam radiotherapy? Clin. Oncol. R Coll. Radiol. 2013, 25, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.; Hodges, C.V. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. 1941. J. Urol. 2002, 168, 9–12. [Google Scholar] [CrossRef]
- Perlmutter, M.A.; Lepor, H. Androgen deprivation therapy in the treatment of advanced prostate cancer. Rev. Urol 2007, 9 (Suppl. S1), 3–8. [Google Scholar]
- Isbarn, H.; Boccon-Gibod, L.; Carroll, P.R.; Montorsi, F.; Schulman, C.; Smith, M.R.; Sternberg, C.N.; Studer, U.E. Androgen Deprivation Therapy for the Treatment of Prostate Cancer: Consider Both Benefits and Risks. Eur. Urol. 2009, 55, 62–75. [Google Scholar] [CrossRef]
- Picard, J.C.; Golshayan, A.-R.; Marshall, D.T.; Opfermann, K.J.; Keane, T.E. The multi-disciplinary management of high-risk prostate cancer. Urol. Oncol. 2012, 30, 3–15. [Google Scholar] [CrossRef]
- Tannock, I.F.; de Witt, R.; Berry, W.R.; Horty, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.C.; Théodore, C.; James, N.D.; Turesson, I.; et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef]
- Paller, C.J.; Antonarakis, E.S. Management of Biochemically Recurrent Prostate Cancer After Local Therapy: Evolving Standards of Care and New Directions. Clin. Adv. Hematol. Oncol. 2013, 11, 14–23. [Google Scholar]
- Karantanos, T.; Corn, P.G.; Thompson, T.C. Prostate cancer progression after androgen deprivation therapy: Mechanisms of castrate resistance and novel therapeutic approaches. Oncogene 2013, 32, 5501–5511. [Google Scholar] [CrossRef] [PubMed]
- Baskar, R.; Lee, K.A.; Yeo, R.; Yeoh, K.-W. Cancer and Radiation Therapy: Current Advances and Future Directions. Int J. Med. Sci. 2012, 9, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Nepple, K.G.; Stephenson, A.J.; Kallogjeri, D.; Michalski, J.; Grubb, R.L.; Strope, S.A.; Haslag-Minoff, J.; Piccirillo, J.F.; Ciezki, J.P.; Klein, E.A.; et al. Mortality after prostate cancer treatment with radical prostatectomy, external-beam radiation therapy, or brachytherapy in men without comorbidity. Eur. Urol. 2013, 64, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Nader, R.; El Amm, J.; Aragon-Ching, J.B. Role of chemotherapy in prostate cancer. Asian J. Androl. 2018, 20, 221–229. [Google Scholar] [CrossRef]
- Chen, F.; Zhao, X. Prostate Cancer: Current Treatment and Prevention Strategies. Iran. Red Crescent Med. J. 2013, 15, 279–284. [Google Scholar] [CrossRef]
- Litwin, M.S.; Tan, H.-J. The Diagnosis and Treatment of Prostate Cancer: A Review. JAMA 2017, 317, 2532–2542. [Google Scholar] [CrossRef]
- Gillet, J.-P.; Varma, S.; Gottesman, M.M. The Clinical Relevance of Cancer Cell Lines. J. Natl. Cancer Inst. 2013, 105, 452–458. [Google Scholar] [CrossRef]
- Mirabelli, P.; Coppola, L.; Salvatore, M. Cancer Cell Lines Are Useful Model Systems for Medical Research. Cancers 2019, 11, 1098. [Google Scholar] [CrossRef]
- Niu, N.; Wang, L. In vitro human cell line models to predict clinical response to anticancer drugs. Pharmacogenomics 2015, 16, 273–285. [Google Scholar] [CrossRef]
- Kaighn, M.E.; Narayan, K.S.; Ohnuki, Y.; Lechner, J.F.; Jones, L.W. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest. Urol. 1979, 17, 16–23. [Google Scholar]
- Seim, I.; Jeffery, P.L.; Thomas, P.B.; Nelson, C.C.; Chopin, L.K. Whole-Genome Sequence of the Metastatic PC3 and LNCaP Human Prostate Cancer Cell Lines. G3 (Bethesda) 2017, 7, 1731–1741. [Google Scholar] [CrossRef]
- Tararova, N.D.; Narizhneva, N.; Krivokrisenko, V.; Gudkov, A.V.; Gurova, K.V. Prostate Cancer Cells Tolerate a Narrow Range of Androgen Receptor Expression and Activity. Prostate 2007, 67, 1801–1815. [Google Scholar] [CrossRef]
- Tai, S.; Sun, Y.; Squires, J.M.; Zhang, H.; Oh, W.K.; Liang, C.-Z.; Huang, J. PC3 is a cell line characteristic of prostatic small cell carcinoma. Prostate 2011, 71, 1668–1679. [Google Scholar] [CrossRef]
- Gaupel, A.-C.; Wang, W.-L.W.; Mordan-McCombs, S.; Yu Lee, E.C.; Tenniswood, M. Xenograft, Transgenic, and Knockout Models of Prostate Cancer. In Animal Models for the Study of Human Disease; Chapter 39; Conn, P.M., Ed.; Academic Press: Boston, MA, USA, 2013; pp. 973–995. ISBN 978-0-12-415894-8. [Google Scholar]
- Stone, K.R.; Mickey, D.D.; Wunderli, H.; Mickey, G.H.; Paulson, D.F. Isolation of a human prostate carcinoma cell line (DU 145). Int. J. Cancer 1978, 21, 274–281. [Google Scholar] [CrossRef]
- Carroll, A.G.; Voeller, H.J.; Sugars, L.; Gelmann, E.P. p53 oncogene mutations in three human prostate cancer cell lines. Prostate 1993, 23, 123–134. [Google Scholar] [CrossRef]
- Pretlow, T.G.; Wolman, S.R.; Micale, M.A.; Pelley, R.J.; Kursh, E.D.; Resnick, M.I.; Bodner, D.R.; Jacobberger, J.W.; Delmoro, C.M.; Giaconia, J.M.; et al. Xenografts of Primary Human Prostatic Carcinoma. J. Natl. Cancer. Inst. 1993, 85, 394–398. [Google Scholar] [CrossRef]
- Sramkoski, R.M.; Pretlow, T.G.; Giaconia, J.M.; Pretlow, T.P.; Schwartz, S.; Sy, M.S.; Marengo, S.R.; Rhim, J.S.; Zhang, D.; Jacobberger, J.W. A new human prostate carcinoma cell line, 22Rv1. In Vitro Cell. Dev. Biol. Anim. 1999, 35, 403–409. [Google Scholar] [CrossRef]
- Hartel, A.; Didier, A.; Ulbrich, S.E.; Wierer, M.; Meyer, H.H.D. Characterisation of steroid receptor expression in the human prostate carcinoma cell line 22RV1 and quantification of androgen effects on mRNA regulation of prostate-specific genes. J. Steroid Biochem. Mol. Biol. 2004, 92, 187–197. [Google Scholar] [CrossRef]
- Wang, X.; Tryndyak, V.; Apostolov, E.O.; Yin, X.; Shah, S.V.; Pogribny, I.P.; Basnakian, A.G. Sensitivity of human prostate cancer cells to chemotherapeutic drugs depends on EndoG expression regulated by promoter methylation. Cancer Lett. 2008, 270, 132–143. [Google Scholar] [CrossRef][Green Version]
- Horoszewicz, J.S.; Leong, S.S.; Kawinski, E.; Karr, J.P.; Rosenthal, H.; Chu, T.M.; Mirand, E.A.; Murphy, G.P. LNCaP model of human prostatic carcinoma. Cancer Res. 1983, 43, 1809–1818. [Google Scholar]
- Spans, L.; Atak, Z.K.; Van Nieuwerburgh, F.; Deforce, D.; Lerut, E.; Aerts, S.; Claessens, F. Variations in the exome of the LNCaP prostate cancer cell line. Prostate 2012, 72, 1317–1327. [Google Scholar] [CrossRef] [PubMed]
- Veldscholte, J.; Ris-Stalpers, C.; Kuiper, G.G.; Jenster, G.; Berrevoets, C.; Claassen, E.; van Rooij, H.C.; Trapman, J.; Brinkmann, A.O.; Mulder, E. A mutation in the ligand binding domain of the androgen receptor of human LNCaP cells affects steroid binding characteristics and response to anti-androgens. Biochem. Biophys. Res. Commun. 1990, 173, 534–540. [Google Scholar] [CrossRef]
- Thalmann, G.N.; Sikes, R.A.; Wu, T.T.; Degeorges, A.; Chang, S.M.; Ozen, M.; Pathak, S.; Chung, L.W. LNCaP progression model of human prostate cancer: Androgen-independence and osseous metastasis. Prostate 2000, 44, 91–103. [Google Scholar] [CrossRef]
- Jiang, X.; Chen, S.; Asara, J.M.; Balk, S.P. Phosphoinositide 3-kinase pathway activation in phosphate and tensin homolog (PTEN)-deficient prostate cancer cells is independent of receptor tyrosine kinases and mediated by the p110beta and p110delta catalytic subunits. J. Biol. Chem. 2010, 285, 14980–14989. [Google Scholar] [CrossRef]
- Dorai, T.; Dutcher, J.P.; Dempster, D.W.; Wiernik, P.H. Therapeutic potential of curcumin in prostate cancer--V: Interference with the osteomimetic properties of hormone refractory C4-2B prostate cancer cells. Prostate 2004, 60, 1–17. [Google Scholar] [CrossRef]
- Vlachojannis, J.; Magora, F.; Chrubasik, S. Willow species and aspirin: Different mechanism of actions. Phytother. Res. 2011, 25, 1102–1104. [Google Scholar] [CrossRef]
- Ziegler, J.; Facchini, P.J.; Geissler, R.; Schmidt, J.; Ammer, C.; Kramell, R.; Voigtländer, S.; Gesell, A.; Pienkny, S.; Brandt, W. Evolution of morphine biosynthesis in opium poppy. Phytochemistry 2009, 70, 1696–1707. [Google Scholar] [CrossRef]
- Kampan, N.C.; Madondo, M.T.; McNally, O.M.; Quinn, M.; Plebanski, M. Paclitaxel and Its Evolving Role in the Management of Ovarian Cancer. Biomed. Res. Int. 2015. [Google Scholar] [CrossRef]
- Demain, A.L.; Vaishnav, P. Natural products for cancer chemotherapy. Microb. Biotechnol. 2011, 4, 687–699. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Dorai, T.; Gehani, N.; Katz, A. Therapeutic potential of curcumin in human prostate cancer-I. curcumin induces apoptosis in both androgen-dependent and androgen-independent prostate cancer cells. Prostate Cancer Prostatic Dis. 2000, 3, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, A.; Bueso-Ramos, C.; Chatterjee, D.; Pantazis, P.; Aggarwal, B.B. Curcumin downregulates cell survival mechanisms in human prostate cancer cell lines. Oncogene 2001, 20, 7597–7609. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Yasunaga, Y.; Segawa, T.; Ko, D.; Moul, J.W.; Srivastava, S.; Rhim, J.S. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int. J. Oncol. 2002, 21, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, A.; Banerjee, S.; Stafford, L.J.; Xia, C.; Liu, M.; Aggarwal, B.B. Curcumin-induced suppression of cell proliferation correlates with down-regulation of cyclin D1 expression and CDK4-mediated retinoblastoma protein phosphorylation. Oncogene 2002, 21, 8852–8861. [Google Scholar] [CrossRef]
- Deeb, D.; Xu, Y.X.; Jiang, H.; Gao, X.; Janakiraman, N.; Chapman, R.A.; Gautam, S.C. Curcumin (diferuloyl-methane) enhances tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis in LNCaP prostate cancer cells. Mol. Cancer Ther. 2003, 2, 95–103. [Google Scholar]
- Chaudhary, L.R.; Hruska, K.A. Inhibition of cell survival signal protein kinase B/Akt by curcumin in human prostate cancer cells. J. Cell. Biochem. 2003, 89, 1–5. [Google Scholar] [CrossRef]
- Deeb, D.; Jiang, H.; Gao, X.; Hafner, M.S.; Wong, H.; Divine, G.; Chapman, R.A.; Dulchavsky, S.A.; Gautam, S.C. Curcumin sensitizes prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L by inhibiting nuclear factor-kappaB through suppression of IkappaBalpha phosphorylation. Mol. Cancer Ther. 2004, 3, 803–812. [Google Scholar]
- Shankar, S.; Srivastava, R.K. Involvement of Bcl-2 family members, phosphatidylinositol 3’-kinase/AKT and mitochondrial p53 in curcumin (diferulolylmethane)-induced apoptosis in prostate cancer. Int. J. Oncol. 2007, 30, 905–918. [Google Scholar] [CrossRef]
- Shankar, S.; Chen, Q.; Sarva, K.; Siddiqui, I.; Srivastava, R.K. Curcumin enhances the apoptosis-inducing potential of TRAIL in prostate cancer cells: Molecular mechanisms of apoptosis, migration and angiogenesis. J. Mol. Signal. 2007, 10, 10–14. [Google Scholar] [CrossRef]
- Srivastava, R.K.; Chen, Q.; Siddiqui, I.; Sarva, K.; Shankar, S. Linkage of curcumin-induced cell cycle arrest and apoptosis by cyclin-dependent kinase inhibitor p21(/WAF1/CIP1). Cell Cycle 2007, 6, 2953–2961. [Google Scholar] [CrossRef]
- Choi, H.Y.; Lim, J.E.; Hong, J.H. Curcumin interrupts the interaction between the androgen receptor and Wnt/β-catenin signaling pathway in LNCaP prostate cancer cells. Prostate Cancer Prostatic Dis. 2010, 13, 343–349. [Google Scholar] [CrossRef]
- Yu, S.; Shen, G.; Khor, T.O.; Kim, J.-H.; Kong, A.-N.T. Curcumin inhibits Akt/mammalian target of rapamycin signaling through protein phosphatase-dependent mechanism. Mol. Cancer Ther. 2008, 7, 2609–2620. [Google Scholar] [CrossRef] [PubMed]
- Hilchie, A.L.; Furlong, S.J.; Sutton, K.; Richardson, A.; Robichaud, M.R.J.; Giacomantonio, C.A.; Ridgway, N.D.; Hoskin, D.W. Curcumin-induced apoptosis in PC3 prostate carcinoma cells is caspase-independent and involves cellular ceramide accumulation and damage to mitochondria. Nutr. Cancer 2010, 62, 379–389. [Google Scholar] [CrossRef] [PubMed]
- Rivera, M.; Ramos, Y.; Rodríguez-Valentín, M.; López-Acevedo, S.; Cubano, L.A.; Zou, J.; Zhang, Q.; Wang, G.; Boukli, N.M. Targeting multiple pro-apoptotic signaling pathways with curcumin in prostate cancer cells. PLoS ONE 2017, 12, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Zheng, Z.; Huang, J.; Ma, X.; Huang, C.; Wu, R.; Li, X.; Liang, Z.; Deng, F.; Wu, J.; et al. Modulation of miR-34a in curcumin-induced antiproliferation of prostate cancer cells. J. Cell. Biochem. 2019, 120, 15616–15624. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-N.; Yu, C.-X.; Zhang, P.-J.; Chen, W.-W.; Jiang, A.-L.; Kong, F.; Deng, J.-T.; Zhang, J.-Y.; Young, C.Y. Curcumin downregulates homeobox gene NKX3.1 in prostate cancer cell LNCaP. Acta Pharm. Sin. 2007, 28, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, Z.; Hill, D.L.; Wang, H.; Zhang, R. Curcumin, a dietary component, has anticancer, chemosensitization, and radiosensitization effects by down-regulating the MDM2 oncogene through the PI3K/mTOR/ETS2 pathway. Cancer Res. 2007, 67, 1988–1996. [Google Scholar] [CrossRef]
- Deeb, D.; Jiang, H.; Gao, X.; Al-Holou, S.; Danyluk, A.L.; Dulchavsky, S.A.; Gautam, S.C. Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1-6-heptadine-3,5-dione; C21H20O6] sensitizes human prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L-induced apoptosis by suppressing nuclear factor-kappaB via inhibition of the prosurvival Akt signaling pathway. J. Pharm. Exp. Ther. 2007, 321, 616–625. [Google Scholar] [CrossRef]
- Nonn, L.; Duong, D.; Peehl, D.M. Chemopreventive anti-inflammatory activities of curcumin and other phytochemicals mediated by MAP kinase phosphatase-5 in prostate cells. Carcinogenesis 2007, 28, 1188–1196. [Google Scholar] [CrossRef]
- Thangapazham, R.L.; Shaheduzzaman, S.; Kim, K.-H.; Passi, N.; Tadese, A.; Vahey, M.; Dobi, A.; Srivastava, S.; Maheshwari, R.K. Androgen responsive and refractory prostate cancer cells exhibit distinct curcumin regulated transcriptome. Cancer Biol. Ther. 2008, 7, 1427–1435. [Google Scholar] [CrossRef]
- Tsui, K.-H.; Feng, T.-H.; Lin, C.-M.; Chang, P.-L.; Juang, H.-H. Curcumin blocks the activation of androgen and interlukin-6 on prostate-specific antigen expression in human prostatic carcinoma cells. J. Androl. 2008, 29, 661–668. [Google Scholar] [CrossRef]
- Piantino, C.B.; Salvadori, F.A.; Ayres, P.P.; Kato, R.B.; Srougi, V.; Leite, K.R.; Srougi, M. An evaluation of the anti-neoplastic activity of curcumin in prostate cancer cell lines. Int. Braz. J. Urol. 2009, 35, 354–360. [Google Scholar] [CrossRef]
- Slusarz, A.; Shenouda, N.S.; Sakla, M.S.; Drenkhahn, S.K.; Narula, A.S.; MacDonald, R.S.; Besch-Williford, C.L.; Lubahn, D.B. Common botanical compounds inhibit the hedgehog signaling pathway in prostate cancer. Cancer Res. 2010, 70, 3382–3390. [Google Scholar] [CrossRef]
- Teiten, M.-H.; Gaascht, F.; Cronauer, M.; Henry, E.; Dicato, M.; Diederich, M. Anti-proliferative potential of curcumin in androgen-dependent prostate cancer cells occurs through modulation of the Wingless signaling pathway. Int. J. Oncol. 2011, 38, 603–611. [Google Scholar] [CrossRef]
- Chung, L.-C.; Tsui, K.-H.; Feng, T.-H.; Lee, S.-L.; Chang, P.-L.; Juang, H.-H. Curcumin provides potential protection against the activation of hypoxia and prolyl 4-hydroxylase inhibitors on prostate-specific antigen expression in human prostate carcinoma cells. Mol. Nutr Food Res. 2011, 55, 1666–1676. [Google Scholar] [CrossRef]
- Khor, T.O.; Huang, Y.; Wu, T.-Y.; Shu, L.; Lee, J.; Kong, A.-N.T. Pharmacodynamics of curcumin as DNA hypomethylation agent in restoring the expression of Nrf2 via promoter CpGs demethylation. Biochem. Pharmacol. 2011, 82, 1073–1078. [Google Scholar] [CrossRef]
- Shu, L.; Khor, T.O.; Lee, J.-H.; Boyanapalli, S.S.S.; Huang, Y.; Wu, T.-Y.; Saw, C.L.-L.; Cheung, K.-L.; Kong, A.-N.T. Epigenetic CpG demethylation of the promoter and reactivation of the expression of Neurog1 by curcumin in prostate LNCaP cells. AAPS J. 2011, 13, 606–614. [Google Scholar] [CrossRef]
- Teiten, M.-H.; Gaigneaux, A.; Chateauvieux, S.; Billing, A.M.; Planchon, S.; Fack, F.; Renaut, J.; Mack, F.; Muller, C.P.; Dicato, M.; et al. Identification of differentially expressed proteins in curcumin-treated prostate cancer cell lines. OMICS 2012, 16, 289–300. [Google Scholar] [CrossRef]
- Sundram, V.; Chauhan, S.C.; Ebeling, M.; Jaggi, M. Curcumin attenuates β-catenin signaling in prostate cancer cells through activation of protein kinase D1. PLoS ONE 2012, 7, 1–13. [Google Scholar] [CrossRef]
- Cheng, T.-S.; Chen, W.-C.; Lin, Y.-Y.; Tsai, C.-H.; Liao, C.-I.; Shyu, H.-Y.; Ko, C.-J.; Tzeng, S.-F.; Huang, C.-Y.; Yang, P.-C.; et al. Curcumin-targeting pericellular serine protease matriptase role in suppression of prostate cancer cell invasion, tumor growth, and metastasis. Cancer Prev Res. (Phila) 2013, 6, 495–505. [Google Scholar] [CrossRef]
- Rodriguez-Garcia, A.; Hevia, D.; Mayo, J.C.; Gonzalez-Menendez, P.; Lu, J.; Holmgren, A.; Sainz, R.M. Thioredoxin 1 modulates apoptosis induced by bioactive compounds in prostate cancer cells. Redox Biol. 2017, 12, 634–647. [Google Scholar] [CrossRef]
- Ide, H.; Lu, Y.; Noguchi, T.; Muto, S.; Okada, H.; Kawato, S.; Horie, S. Modulation of AKR1C2 by curcumin decreases testosterone production in prostate cancer. Cancer Sci. 2018, 109, 1230–1238. [Google Scholar] [CrossRef]
- Zhao, W.; Zhou, X.; Qi, G.; Guo, Y. Curcumin suppressed the prostate cancer by inhibiting JNK pathways via epigenetic regulation. J. Biochem. Mol. Toxicol. 2018, 32, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Katta, S.; Srivastava, A.; Thangapazham, R.L.; Rosner, I.L.; Cullen, J.; Li, H.; Sharad, S. Curcumin-Gene Expression Response in Hormone Dependent and Independent Metastatic Prostate Cancer Cells. Int. J. Mol. Sci. 2019, 20, 4891. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.H.; Ahn, K.S.; Bae, E.; Jeon, S.S.; Choi, H.Y. The effects of curcumin on the invasiveness of prostate cancer in vitro and in vivo. Prostate Cancer Prostatic Dis. 2006, 9, 147–152. [Google Scholar] [CrossRef]
- Santel, T.; Pflug, G.; Hemdan, N.Y.A.; Schäfer, A.; Hollenbach, M.; Buchold, M.; Hintersdorf, A.; Lindner, I.; Otto, A.; Bigl, M.; et al. Curcumin inhibits glyoxalase 1: A possible link to its anti-inflammatory and anti-tumor activity. PLoS ONE 2008, 3, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Herman, J.G.; Stadelman, H.L.; Roselli, C.E. Curcumin blocks CCL2-induced adhesion, motility and invasion, in part, through down-regulation of CCL2 expression and proteolytic activity. Int. J. Oncol. 2009, 34, 1319–1327. [Google Scholar]
- Liu, S.; Wang, Z.; Hu, Z.; Zeng, X.; Li, Y.; Su, Y.; Zhang, C.; Ye, Z. Anti-tumor activity of curcumin against androgen-independent prostate cancer cells via inhibition of NF-κB and AP-1 pathway in vitro. J. Huazhong Univ. Sci. Technol. Med. Sci. 2011, 31, 530. [Google Scholar] [CrossRef]
- Killian, P.H.; Kronski, E.; Michalik, K.M.; Barbieri, O.; Astigiano, S.; Sommerhoff, C.P.; Pfeffer, U.; Nerlich, A.G.; Bachmeier, B.E. Curcumin inhibits prostate cancer metastasis in vivo by targeting the inflammatory cytokines CXCL1 and -2. Carcinogenesis 2012, 33, 2507–2519. [Google Scholar] [CrossRef]
- Yu, X.-L.; Jing, T.; Zhao, H.; Li, P.-J.; Xu, W.-H.; Shang, F.-F. Curcumin inhibits expression of inhibitor of DNA binding 1 in PC3 cells and xenografts. Asian Pac. J. Cancer Prev. 2014, 15, 1465–1470. [Google Scholar] [CrossRef]
- Li, J.; Xiang, S.; Zhang, Q.; Wu, J.; Tang, Q.; Zhou, J.; Yang, L.; Chen, Z.; Hann, S.S. Combination of curcumin and bicalutamide enhanced the growth inhibition of androgen-independent prostate cancer cells through SAPK/JNK and MEK/ERK1/2-mediated targeting NF-κB/p65 and MUC1-C. J. Exp. Clin. Cancer Res. 2015, 34, 46. [Google Scholar] [CrossRef]
- Du, Y.; Long, Q.; Zhang, L.; Shi, Y.; Liu, X.; Li, X.; Guan, B.; Tian, Y.; Wang, X.; Li, L.; et al. Curcumin inhibits cancer-associated fibroblast-driven prostate cancer invasion through MAOA/mTOR/HIF-1α signaling. Int. J. Oncol. 2015, 47, 2064–2072. [Google Scholar] [CrossRef]
- Hu, H.-J.; Lin, X.-L.; Liu, M.-H.; Fan, X.-J.; Zou, W.-W. Curcumin mediates reversion of HGF-induced epithelial-mesenchymal transition via inhibition of c-Met expression in DU145 cells. Oncol. Lett. 2016, 11, 1499–1505. [Google Scholar] [CrossRef]
- Sha, J.; Li, J.; Wang, W.; Pan, L.; Cheng, J.; Li, L.; Zhao, H.; Lin, W. Curcumin induces G0/G1 arrest and apoptosis in hormone independent prostate cancer DU-145 cells by down regulating Notch signaling. Biomed. Pharmacother. 2016, 84, 177–184. [Google Scholar] [CrossRef]
- Yang, C.; Ma, X.; Wang, Z.; Zeng, X.; Hu, Z.; Ye, Z.; Shen, G. Curcumin induces apoptosis and protective autophagy in castration-resistant prostate cancer cells through iron chelation. Drug Des. Devel. Ther. 2017, 11, 431–439. [Google Scholar] [CrossRef]
- Yang, J.; Wang, C.; Zhang, Z.; Chen, X.; Jia, Y.; Wang, B.; Kong, T. Curcumin inhibits the survival and metastasis of prostate cancer cells via the Notch-1 signaling pathway. APMIS 2017, 125, 134–140. [Google Scholar] [CrossRef]
- Cao, H.; Yu, H.; Feng, Y.; Chen, L.; Liang, F. Curcumin inhibits prostate cancer by targeting PGK1 in the FOXD3/miR-143 axis. Cancer Chemother. Pharmacol. 2017, 79, 985–994. [Google Scholar] [CrossRef]
- Liu, T.; Chi, H.; Chen, J.; Chen, C.; Huang, Y.; Xi, H.; Xue, J.; Si, Y. Curcumin suppresses proliferation and in vitro invasion of human prostate cancer stem cells by ceRNA effect of miR-145 and lncRNA-ROR. Gene 2017, 631, 29–38. [Google Scholar] [CrossRef]
- Zhang, H.; Zheng, J.; Shen, H.; Huang, Y.; Liu, T.; Xi, H.; Chen, C. Curcumin Suppresses In Vitro Proliferation and Invasion of Human Prostate Cancer Stem Cells by Modulating DLK1-DIO3 Imprinted Gene Cluster MicroRNAs. Genet. Test. Mol. Biomark. 2018, 22, 43–50. [Google Scholar] [CrossRef]
- Dorai, T.; Cao, Y.C.; Dorai, B.; Buttyan, R.; Katz, A.E. Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate 2001, 47, 293–303. [Google Scholar] [CrossRef]
- Khor, T.O.; Keum, Y.-S.; Lin, W.; Kim, J.-H.; Hu, R.; Shen, G.; Xu, C.; Gopalakrishnan, A.; Reddy, B.; Zheng, X.; et al. Combined inhibitory effects of curcumin and phenethyl isothiocyanate on the growth of human PC-3 prostate xenografts in immunodeficient mice. Cancer Res. 2006, 66, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Ganapathy, S.; Chen, Q.; Srivastava, R.K. Curcumin sensitizes TRAIL-resistant xenografts: Molecular mechanisms of apoptosis, metastasis and angiogenesis. Mol. Cancer 2008, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Barve, A.; Khor, T.O.; Hao, X.; Keum, Y.-S.; Yang, C.S.; Reddy, B.; Kong, A.-N.T. Murine prostate cancer inhibition by dietary phytochemicals—curcumin and phenyethylisothiocyanate. Pharm. Res. 2008, 25, 2181–2189. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, N.K.; Nargi, D.; Randolph, C.; Narayanan, B.A. Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in PTEN knockout mice. Int. J. Cancer 2009, 125, 1–8. [Google Scholar] [CrossRef]
- Fernández-Martínez, A.B.; Bajo, A.M.; Valdehita, A.; Isabel Arenas, M.; Sánchez-Chapado, M.; Carmena, M.J.; Prieto, J.C. Multifunctional role of VIP in prostate cancer progression in a xenograft model: Suppression by curcumin and COX-2 inhibitor NS-398. Peptides 2009, 30, 2357–2364. [Google Scholar] [CrossRef]
- Yallapu, M.M.; Khan, S.; Maher, D.M.; Ebeling, M.C.; Sundram, V.; Chauhan, N.; Ganju, A.; Balakrishna, S.; Gupta, B.K.; Zafar, N.; et al. Anti-cancer activity of curcumin loaded nanoparticles in prostate cancer. Biomaterials 2014, 35, 8635–8648. [Google Scholar] [CrossRef]
- Dorai, T.; Diouri, J.; O’Shea, O.; Doty, S.B. Curcumin Inhibits Prostate Cancer Bone Metastasis by Up-Regulating Bone Morphogenic Protein-7 in Vivo. J. Cancer Ther. 2014, 5, 369–386. [Google Scholar] [CrossRef]
- Yang, J.; Ning, J.; Peng, L.; He, D. Effect of curcumin on Bcl-2 and Bax expression in nude mice prostate cancer. Int. J. Clin. Exp. Pathol. 2015, 8, 9272–9278. [Google Scholar]
- Hong, J.H.; Lee, G.; Choi, H.Y. Effect of curcumin on the interaction between androgen receptor and Wnt/β-catenin in LNCaP xenografts. Korean J. Urol. 2015, 56, 656–665. [Google Scholar] [CrossRef]
- Lodi, A.; Saha, A.; Lu, X.; Wang, B.; Sentandreu, E.; Collins, M.; Kolonin, M.G.; DiGiovanni, J.; Tiziani, S. Combinatorial treatment with natural compounds in prostate cancer inhibits prostate tumor growth and leads to key modulations of cancer cell metabolism. Npj. Precis. Oncol. 2017, 1, 1–12. [Google Scholar] [CrossRef]
- Ma, Q.; Qian, W.; Tao, W.; Zhou, Y.; Xue, B. Delivery of Curcumin Nanoliposomes Using Surface Modified With CD133 Aptamers For Prostate Cancer. Drug Des. Devel. Ther. 2019, 13, 4021–4033. [Google Scholar] [CrossRef]
- Li, H.; Zhang, N.; Hao, Y.; Wang, Y.; Jia, S.; Zhang, H. Enhancement of curcumin antitumor efficacy and further photothermal ablation of tumor growth by single-walled carbon nanotubes delivery system in vivo. Drug Deliv. 2019, 26, 1017–1026. [Google Scholar] [CrossRef]
- Gracia, E.; Mancini, A.; Colapietro, A.; Mateo, C.; Gracia, I.; Festuccia, C.; Carmona, M. Impregnation of Curcumin into a Biodegradable (Poly-lactic-co-glycolic acid, PLGA) Support, to Transfer Its Well Known In Vitro Effect to an In Vivo Prostate Cancer Model. Nutrients 2019, 11, 2312. [Google Scholar] [CrossRef]
- De Velasco, M.A.; Lu, Y.; Kura, Y.; China, T.; Inoue, Y.; Nakayama, A.; Okada, H.; Horie, S.; Uemura, H.; Ide, H. Chemopreventive effects of nanoparticle curcumin in a mouse model of Pten-deficient prostate cancer. Hum. Cell 2020, 33, 730–736. [Google Scholar] [CrossRef]
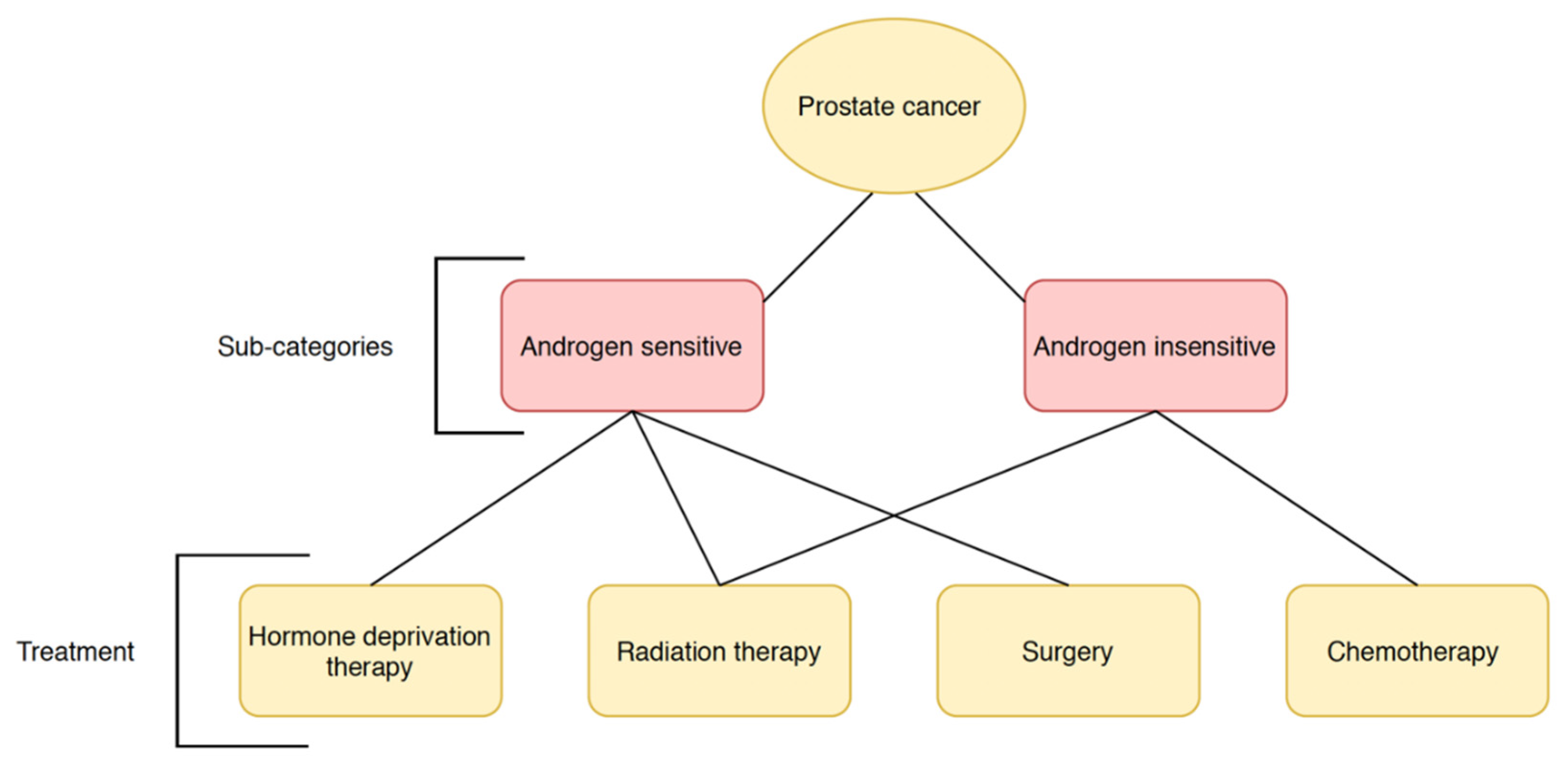
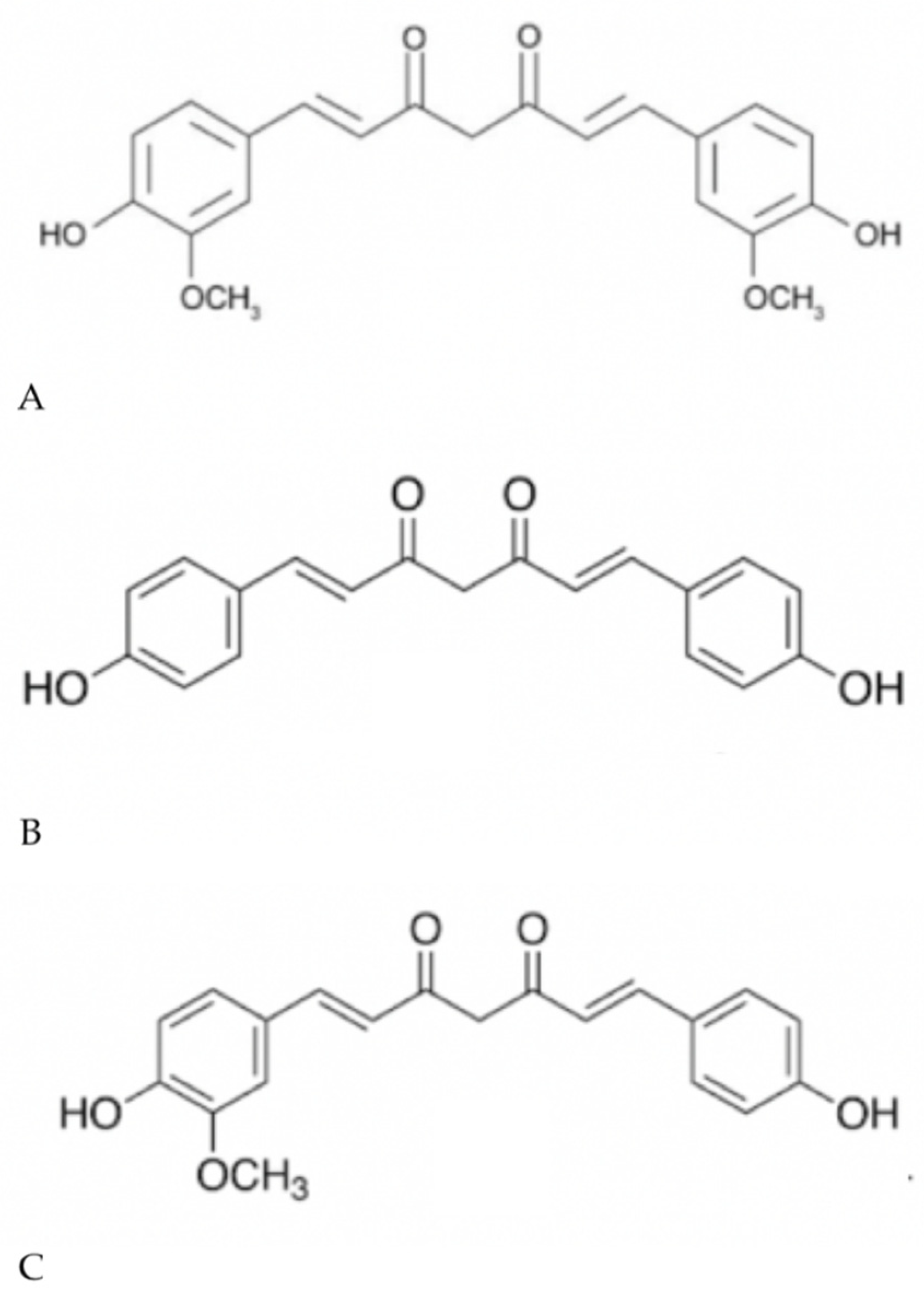
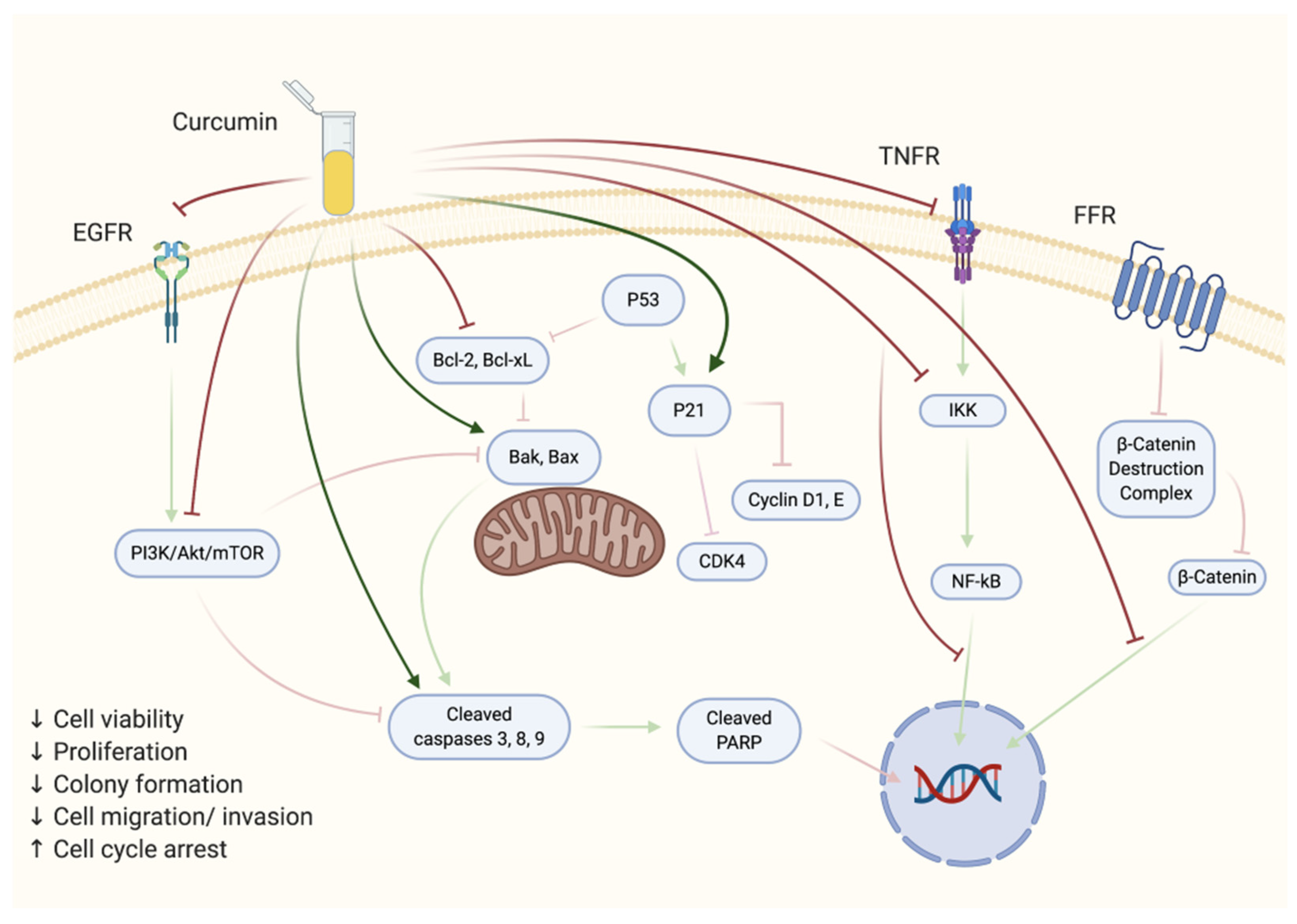
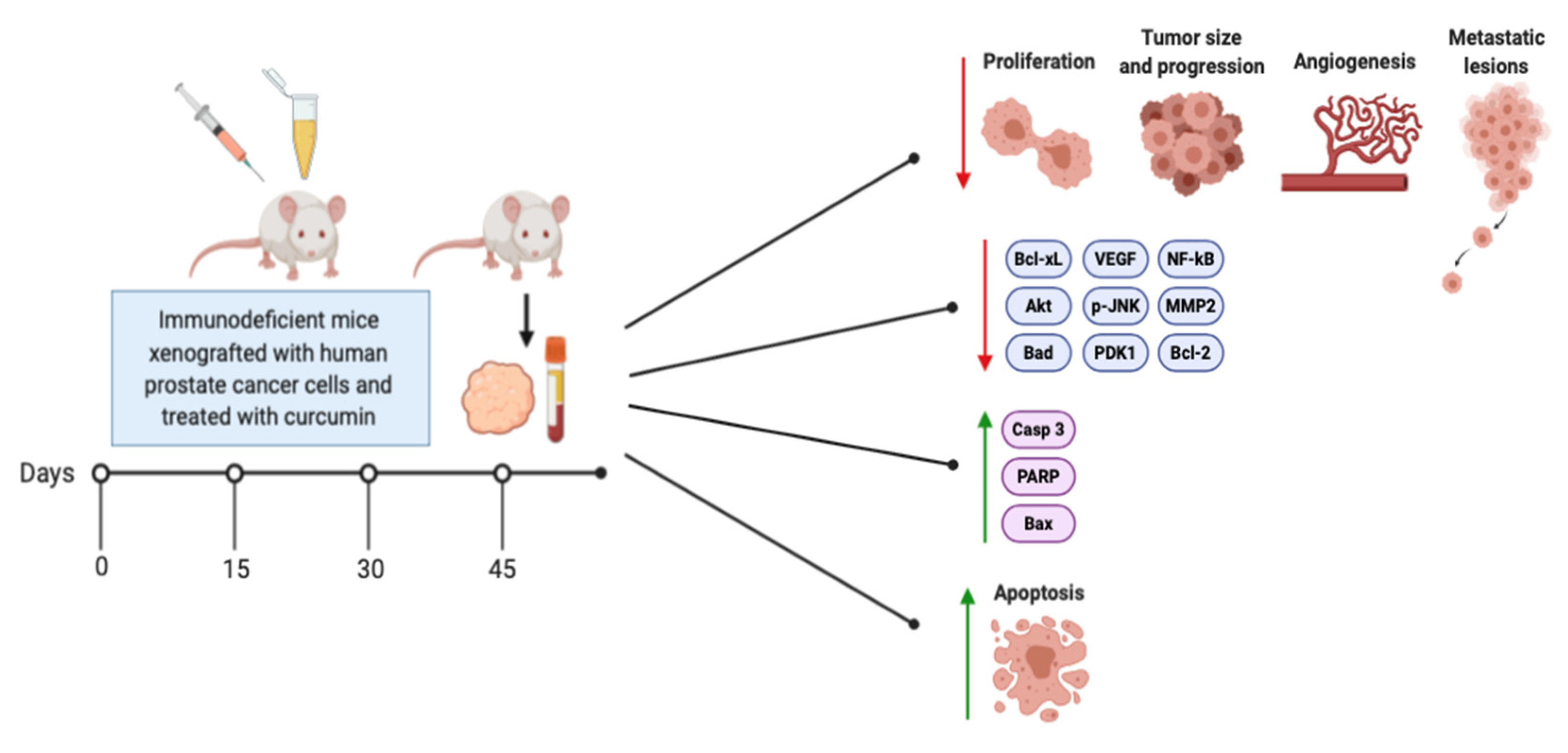
| Treatment | Target | Risks and Recurrence |
|---|---|---|
| Prostatectomy | Efficient against localized and early- or advanced-stage tumors [13] | Moderate biochemical recurrence rate in localized tumors, risk of spreading beyond original location [22] |
| Androgen deprivation therapy | Efficient against androgen- dependent carcinomas | Androgen insensitivity may develop leading to reoccurrence [23] |
| Radiation therapy | Efficient against localized tumors or minimally metastasized tumors [15] | Low efficacy if tumor has developed beyond early stages, requiring combination treatments; higher mortality compared to prostatectomy [24,25] |
| Chemotherapy | Primarily utilized to treat advanced-stage, metastatic, castration-resistant prostate cancer [26] | Improves quality of life and slows disease progression in high-risk prostate cancer, but unlikely to cure it [27,28] |
| Prostate Cancer Cell Line | Category | Gene Mutations | Representative Prostate Cancer Subtype |
|---|---|---|---|
| PC3 | Prostatic adenocarcinoma, derived from a bone metastasis of grade IV prostate cancer extracted from a 62 year old Caucasian male [32] | PTEN, p53 [33] PSA-negative Lost AR expression [34] | Androgen-insensitive, highly invasive, rare small-cell prostatic carcinoma [35,36] |
| DU-145 | Prostatic adenocarcinoma, derived from a metastasis in the CNS of a 69 year old Caucasian male originating from a primary prostate adenocarcinoma [37] | p53 [38] PSA-negative Lost AR expression [34] | Androgen-insensitive, invasive osteolytic tumor phenotype [36] |
| 22RV1 | Prostatic adenocarcinoma, derived from a human carcinoma xenograft (CWR22R) serially propagated in nude mice following the castration-induced regression and relapse of androgen dependent CWR22 xenograft [39,40] | p53 [38] AR mutation at codon 874 (His to Tyr) [41] Express endogenous AR PSA-positive [34] | Androgen-sensitive but androgen-independent, low invasiveness, tumors form primarily osteosclerotic lesions [36,42] |
| LNCaP | Prostatic adenocarcinoma, derived from a lymph node metastasis extracted from a 50 year old Caucasian male [43] | PTEN [44] AR gene mutation at codon 868 (Thr to Ala) [45] Express endogenous AR PSA-positive [34] | Androgen-sensitive, localized tumor, fast growth but low invasiveness [35,36,42] |
| C4-2B | Prostatic adenocarcinoma cell lines derived from co-inoculation of LNCaP androgen-dependent cell lines with fibroblasts derived from human osteosarcoma in nude athymic mice for 12 weeks after castration at 8 weeks, and then re-inoculating the extracted tumor with osteosarcoma fibroblasts in castrated mice for another 12 weeks [46] | PTEN [47] PSA-positive Express endogenous AR [34] | Androgen-sensitive but androgen-independent, highly metastatic, primarily form osteoblastic lesions [36,48]. |
| Cell Line | Curcumin Dosage | Effects | Reference |
|---|---|---|---|
| LNCaP | 0–50 μM; 72 h to assess cell proliferation and cell morphology 20 μM; 24 h to assess expression of Bcl-2, Bcl-xL and Bax 0–50 μM; 24 h to assess PARP cleavage, AR expression, and PSA levels. | ↓ Proliferation ↑ Lifted, round cells ↓ Bcl-2 protein ↓ Bcl-xL protein ↑ Phosphatidylserine translocation to outer plasma membrane ↑ PARP cleavage ↓ AR protein ↓ PSA secretion | [54] |
| LNCaP | 10 and 50 μM; 1–4 days to assess cell viability 0–100 μM; 5 h to assess NF-κB expression 50 μM; 0–72 h to assess the expression of Bcl-2 and Bcl-xL 100 μM; 0–72 h to assess the expression and activity of procaspases 3 and 8 50 μM; 0–4 days to assess cell proliferation, PARP cleavage, and apoptosis | ↓ Cell viability ↓ Proliferation ↓ NF-κB protein activation ↓ Bcl-2 protein ↓ Bcl- xL protein ↑ Procaspase-3 and -8 activity ↑ PARP cleavage ↑ Apoptosis | [55] |
| LNCaP | 40 μM; 21 days to assess colony formation efficiency 0–50 μM; 24 h to assess cell growth 40 μM; 24 h | ↓ Cell growth ↓ AR ↓ AR transcriptional activity ↓ AR transactivation ↓ c-Jun ↓ AP-1 ↓ CBP ↓ Colony formation efficiency | [56] |
| LNCaP | 0–100 μM; 24 h to assess cell proliferation and cyclin D1 protein expression 0–50 μM; 24 h to assess DNA synthesis 25 and 50 μM; 3 and 24 h to assess cell viability 50 μM; 0–180 min and 30–120 min to assess cyclin D1 mRNA expression and CDK4 activity 10 μM; 0–120 min to assess Cyclin D1 promoter activity | ↓ Proliferation ↓ DNA synthesis ↓ Cyclin D1 protein and mRNA ↓ CDK4 activity ↓ Cyclin D1 promoter activity | [57] |
| LNCaP | 5–40 μM; 48 h to assess cell viability, caspase activity, DNA fragmentation 10 μM; 20 h to assess cytochrome c release and protein expression of procaspases | ↓ Cell viability ↑ TRAIL sensitivity ↑ Cell-cycle arrest at the G2/M phase ↓ Bid protein ↓ Mitochondrial cytochrome c | [58] |
| LNCaP | 35 μM; 0–4 h | ↓ p-Akt | [59] |
| C4-2B | 0-15 μM; 1 h to assess EGFR autophosphorylation 1–10 μM; 1 h to assess CSF-1 phosphorylation 7.5 μM and 15 μM; 12 days to assess cell mineralization, IKK activity, and COX-2 expression | ↓ EGFR autophosphorylation ↓ CSF-1 phosphorylation ↓ Cell mineralization ↓ IKK activity ↓ COX-2 protein | [48] |
| LNCaP | 10 μM; 48 h to assess cytotoxicity 10 μM; 20 h to assess cleavage of procaspase-3 12.5–50 μM; 20 h to assess NF-κB expression 12.5 μM; 4 and 20h to assess IκBα expression | ↑ Cytotoxicity ↑ Apoptosis ↓ NF-κB ↓ P-IκBα ↑ Cleaved caspase-3 | [60] |
| LNCaP | 0–40 μM; 24 h | ↓ NKX3.1 mRNA and protein ↓ AR mRNA and protein ↓ ARE binding activity | [69] |
| LNCaP | 0–50 μM; 0–72 h | ↓ MDM2 | [70] |
| LNCaP | 0–30 μM; 48 h to assess cell viability and the expression of Bcl-2, Bax, and Bak 0–30 μM; 3 weeks to assess colony formation efficiency 0–30 μM; 24 h to assess caspase-3 activation and PARP cleavage 0–20 μM; 6–24 h to assess the expression of Bim, Bax, Bak, PMMA, Noxa, Bcl-2, and Bcl-xL genes, and p110, p85, and p-Akt proteins 0–30 μM; 0–24 h to assess mitochondrial membrane potential, release of mitochondrial proteins, translocation of Bax and p53 to the mitochondria, and p53 acetylation and phosphorylation | ↓ Cell viability ↓ Colony formation ↑ Caspase-3 activation and nuclear translocation ↑ PARP cleavage ↓ Bcl-2 protein and mRNA ↑ Bax protein and mRNA ↑ Bak protein and mRNA ↑ Bim mRNA ↑ PΜMA mRNA ↑ Noxa mRNA ↓ Bcl-xL mRNA ↓ Mitochondrial membrane potential ↑ Release of Smac/ DIABLO, cytochrome c, and Omi/HtrA2 proteins from mitochondria ↑ Translocation of Bax and p53 to the mitochondria ↑ p53 acetylation and phosphorylation ↑ ROS ↓ p110 ↓ p85 ↓ p-Akt | [61] |
| LNCaP | 10 μM; 48 h to assess cell viability and NF-κB expression 10–30 μM; 24 h to assess Akt phosphorylation 10–30 μM; 24 h to assess protein expression | ↓ p-Akt | [71] |
| LNCaP | 25 μM; 17 h | ↑ MKP5 mRNA | [72] |
| LNCaP | 0–30 μM; 24 h to assess cell viability 5–40 μM; 21 days to assess colony formation efficiency and TRAIL-induced apoptosis 0–10 μM; 24 h to assess expression of DR4, DR5, DcR1, and DcR2 0–20 μM; 24 and 48 h to assess the expression of pro- and antiapoptotic proteins, caspases, cleaved PARP, and mitochondrial membrane potential | ↓ Cell viability ↓ Colony formation efficiency ↑ TRAIL-induced apoptosis ↑ DR4 death receptor ↑ DR5 death receptor ↑ Bak ↑ Bax ↑ PMMA ↑ Bim ↑ Noxa ↓ Bcl-2 ↓ Bcl-xL ↑ Bid cleavage to tBid ↓ IAPs ↓ XIAP ↓ Mitochondria membrane potential ↑ Caspase-3, -8, and -9 cleavage ↑ PARP cleavage | [62] |
| LNCaP | 20 μM; 24 h to assess cell-cycle progression 0–20 μM; 48 h and 20 μM; 0-48 h to assess apoptotic processes 0–20 μM; 0–48 h to assess protein expression | ↑ Cell cycle arrest at the G1/S phase ↑ Apoptosis ↑ p27 ↑ p21 ↓ Cyclin D1 ↓ Cyclin E | [63] |
| LNCaP, C4-2B | 0–100 μM; 24–74 h to assess cell proliferation 10 μM; 3–48 h to assess gene expression 5–20 μM; 12 h to assess protein and gene expression, and PSA ELISA | ↓ Proliferation ↓ Cell growth ↑ Methionine tRNA synthase ↑ Hemeoxygenase decyclizing ↑ Transcription corepressor activity ↓ Kallikrein 2, 3 ↓ NEDD4- binding protein ↓Transmembrane proteases ↓ Cyclin B1 ↓ AR protein ↓ NKX3.1 ↓ PSA ↓ ERBB2 ↓ EGFR | [73] |
| LNCaP | 0–80 μM; 24–72 h to assess cell proliferation 0–20 μM; 24 h | ↓ Proliferation ↓ PSA protein and mRNA ↓ AR mRNA ↓ IL-6 | [74] |
| LNCaP | 10 μM, 25 μM, or 50 μM; 24 h | ↓ Cell viability ↑ Apoptosis ↓ Necrosis | [75] |
| TRAMP-C2 | 0–100 μM; 24 and 72 h | ↓ Cell growth ↓ Gli1 (Hedgehog signaling) | [76] |
| LNCaP | 25, 50, and 100 μM; 24–72 h to assess apoptosis and DNA fragmentation | ↑ Apoptosis ↑ Ceramide ↑ ssDNA | [66] |
| LNCaP | 0–100 μM; 24 and 48 h to assess cell proliferation 0–30 μM; 24 h to assess AR expression 25 μM; 24 h to assess expression of proteins in the Wnt/β-catenin signaling pathway | ↓ Proliferation ↓ AR↓ Nuclear β-catenin ↓ GSK-3b ↓ c-myc ↓ Wnt/β-catenin pathway | [64] |
| 22RV1, LNCaP | 10–100 μM; 4 and 24 h 20 μM; 24 h in 22RV1 and DU-145 only for fluorescent microscopy and nuclear staining | ↑ Curcumin compartmentalization within cytoplasm and exclusion from nucleus ↑ Cytotoxicity ↑ Apoptosis ↑ Autophagy ↑ LC3B-II isoform ↑ Cell-cycle arrest at the G2 stage ↓ Cyclin B1 ↓ PCNA ↓ β-catenin signaling ↓ c-myc mRNA ↓ Survivin mRNA ↓ Cyclin D1 mRNA ↓ TCF-4 ↓ CREB binding protein ↓ P300 | [77] |
| LNCaP | 10 μM; 24 h | ↓ AR ↓ PSA | [78] |
| TRAMP-C1 | 2.5 and 5 μM; 5 days to assess protein and gene expression 5–100 μM; 1 h to assess methylation | ↓ Hypermethylation of CpG sites in Nrf2 ↑ Nrf2 ↑ NQO-1 ↓ CpG methylase (M.Sssl) activity | [79] |
| LNCaP | 5 μM; 7 days | ↑ Cytotoxicity ↓ Tri-methylation of H3K27 (H3K27me3) ↓ Methylation of Neurog1 gene ↑ Neurog1 ↑ HDAC1 ↑ HDAC4 ↑ HDAC5 ↑ HDAC8 ↓ HDAC3 ↓ HDAC activity | [80] |
| LNCaP, 22RV1 | 20 μM; 24 h 0–50 μM; 24 to assess AR expression | ↓ AR ↑ HSP90AA1, GJA1, PRKCE, XRCC6, MIR-141, MIR-183, HMGB1 | [81] |
| LNCaP, C4-2B | 5–40 μM; 48 h to assess cell proliferation 20 μM; 24 h for immunoblotting and PCR 15 μM; 1 h for aggregation assay 5 and 10 μM; 24 h for Boyden’s chamber assay | ↓ Proliferation ↓ β-catenin signaling ↓ Nuclear β-catenin ↑ β-catenin localization in membrane ↑ Cell–cell aggregation ↑ P-PKD1 ↑ Inactive p-cofilin | [82] |
| LNCaP, CW22RV1, C-33 | 0–50 μM; 16 h 25 and 50 μM; 30 min acute treatments | ↓ Cell migration/invasion ↓ Total and activated matripase | [83] |
| LNCaP | 25 μM; 48 h to evaluate apoptosis, gene and protein expression 25 μM; 24 h to assess H2O2 production 1.6–25 μM; 48 h to assess cell viability 10–50 μM; 30 min to assess ROS production | ↓ Cell viability ↑ Cytotoxicity ↑ Apoptosis ↓ Bcl-2 ↑ Bax ↑ ROS ↑ CuZnSOD ↑ TRX1 oxidation ↑ TRXR1 mRNA | [84] |
| LNCaP, 22RV1 | 0–50 μM; 0–72 h to assess cell proliferation 0–50 μM; 6 h for apoptosis assay 0–50 μM; 24h to assess protein expression and levels of DHT | ↓ Cell viability ↑ Caspase-3/7 ↓ DHT ↑ AKR1C2 ↑ SRD5A1 ↓ StAR ↓ CYP11A1 ↓ HSD3B2 | [85] |
| LNCaP | 10 U/mL 74 h to assess cell viability | ↑ Apoptosis ↓ p-JNK ↓ c-Jun ↓ Bcl-2 mRNA ↓ Tertiary methylation of H3K4 | [86] |
| LNCaP, C4-2B | 10 μM; 3–48 h to assess gene expression | ↑ Apoptosis ↑ Cell-cycle arrest ↓ Myc ↑ Heme Oxygenase-1 ↑ Cyclic AMP-dependent transcription factor ↓ RAF1 ↓ RAF1 ↓ IGF1R ↓ BCL6 ↑ PTEN ↑ EGFR1 ↑ SMAD ↑ FOXO3 ↑ Akt1 ↑ RAD51 ↓ SOX4 ↓ EGFR ↓ WT1 ↓ E2F2 ↓ MALAT1↑ BMP receptor signaling ↑ PTEN-regulated cell-cycle arrest ↓ TGF-b receptor signaling ↓ WNT signaling ↓ AP-1 ↓ NF-κB signaling ↓ PI3K/Akt/mTOR signaling ↓ FOXM1 ↑ IL-6 signaling ↑ FTH1 ↑ CPEB4 ↑ C6orf61 ↓ PMEPA1 | [87] |
| Cell Line | Curcumin Dosage | Effects | Reference |
|---|---|---|---|
| PC3 | 0–50 μM; 0–72 h IC50: 10–20 μM | ↓ Proliferation ↑ Lifted, round cells | [54] |
| DU-145 | 10 and 50 μM; 1–4 days to assess cell viability 0–100 μM; 5 h to assess NF-κB expression 0-50 μM; 5 h to assess AP-1 expression 50 μM; 0–72 h to assess the expression of Bcl-2 and Bcl-xL 100 μM; 0–72 h to assess the expression and activity of procaspases 3 and 8 50 μM; 0–4 days to assess cell proliferation, PARP cleavage, and apoptosis | ↓ Cell viability ↓ Proliferation ↓ NF-κB protein activation ↓ AP-1 protein ↓ Bcl-2 protein ↓ Bcl- xL protein ↑ Procaspase-3 and -8 activity ↑ PARP cleavage ↑ Apoptosis | [55] |
| PC3 | 30 μM; 21 days to assess colony formation efficiency 0–50 μM; 24 h to assess cell growth 30 μM; 24 h | ↓ Cell growth ↓ Colony formation efficiency ↓ AR transcriptional activity ↓ c-Jun ↓ AP-1 ↓ CBP ↓ NF-κB mRNA | [56] |
| DU-145 | 5–40 μM; 48 h | ↓ Cell viability ↑ Cell cycle arrest at the G2/M phase | [58] |
| PC3, DU-145 | 35 μM; 30 min in PC3 14 μM; 30 min in DU-145 | ↓ p-Akt in PC3 only | [59] |
| DU-145 | 0–125 ug/mL; 24–72 h to assess cell proliferation 10 ug/mL; 0–48 h to assess apoptosis 0–100 ug/mL; 0–48 h to assess MMP-2 an MMP-9 secretion 1–15 ug/mL; 24 h to assess MMP-9 protein expression | ↓ Proliferation ↑ Apoptosis ↓ MMP-2 secretion ↓ MMP-9 secretion ↓ MMP-9 protein | [88] |
| PC3 | 0–50 μM; 0–72 h to assess the expression of MDM2, p21, Bax, and E2F1 0–30 μM; 24 h and 15 μM; 0–20 h to assess MDM2 mRNA expression 0–30 μM; 24 h to assess ETS2 and p-Akt expression 0–30 μM; 48 h to assess apoptosis, cell viability, and proliferation | ↓ Cell viability ↓ Proliferation ↓ MDM2 protein and mRNA ↑ P21 ↑ Bax ↓ E2F1 ↓ Bcl-2 ↓ ETS2 ↓ P-Akt ↑ Apoptosis | [70] |
| DU-145, PC3 | 0–30 μM; 48 h to assess cell viability 0–30 μM; 3 weeks to assess colony formation efficiency 0–30 μM; 24 h to assess caspase-3 activation and PARP cleavage | ↓ Cell viability ↓ Colony formation efficiency ↑ Caspase-3 activation and nuclear translocation ↑ PARP cleavage | [61] |
| PC3, DU-145 | 20 μM in DU-145, 30 μM in PC3; 48 h to assess cell viability and NF-κB expression 20–40 μM in PC3, 10–30 μM in DU-145; 24 h to assess Akt phosphorylation 10–30 μM; 24 h to assess protein expression | ↓ NF-κB ↓ IκBα ↓ Bcl-2 ↓ Bcl-xL ↓ XIAP | [71] |
| PC3, DU-145 | 25 μM; 17 h 10–50 μM; 17 h to assess IL-6 and IL-8 gene expression 10–25 μM; 17 h to assess NF-κB-luc activity | ↑ MKP5 mRNA ↓ TNFα ↓ P-p38 ↓ COX-2 ↓ COX-2 mRNA ↓ IL-6 mRNA ↓ IL-8 mRNA ↓ p38-mediated inflammatory signaling | [72] |
| PC3 | 0–30 μM; 24 h to assess cell viability 5–40 μM; 21 days to assess colony formation efficiency and TRAIL-induced apoptosis 0–10 μM; 24 h to assess expression of DR4, DR5, DcR1, an dDcR2 0–20 μM; 24 and 48 h to assess the expression of pro- and antiapoptotic proteins, caspases, cleaved PARP, and mitochondrial membrane potential | ↓ Cell viability ↓ Colony formation efficiency ↑ TRAIL-induced apoptosis ↑ DR4 death receptor ↑ DR5 death receptor ↑ Bak ↑ Bax ↑ PMMA ↑ Bim ↑ Noxa ↓ Bcl-2 ↓ Bcl-xL ↑ Bid cleavage to tBid ↓ IAPs ↓ XIAP ↓ Mitochondria membrane potential ↑ Caspase-3 activity ↑ Caspase-3, -8, -9 cleavage ↑ PARP cleavage | [62] |
| PC3 | 20 μM; 24 h to assess cell-cycle progression 0–20 μM; 48 h and 20 μM; 0–48 h to assess apoptotic processes 0–20 μM; 0–48 h to assess protein expression | ↑ Cell-cycle arrest at the G1/S phase ↑ Apoptosis ↑ p27 ↑ p21 ↑ p16 ↓ Rb hyperphosphorylation ↓ Cyclin D1 ↓ Cyclin E ↓ CDK4 | [63] |
| PC3 | 0–100 μM; 24 h | ↓ Cell proliferation ↓ Glyoxalase 1 activity ↑ Cytotoxicity ↑ Necrosis | [89] |
| PC3 | 0–50 μM; 24 h to assess cell viability, 8h to assess protein and DNA synthesis 0–50 μM; 1 h and 40 μM; 0–2 h to assess protein expression 40 μM; 1 h to assess the expression of p-Akt, p-mTOR, and p-S6 | ↓ Proliferation ↓ p-Akt ↓ p-mTOR ↓ p70S6K ↓ FOXO1 ↓ GSK3β ↑ p-AMPK ↑ MAPK ↓ Cyclin D1 ↑ Phosphatase activity | [65] |
| PC3 | 30 μM; 18 h | ↓ CCL2 triggered cell adhesion ↓ Cell invasion ↓ Cell motility ↓ Adhesion to fibronectin ↓ CCL2 mRNA ↓ CCL2 | [90] |
| Pc-Bra1 | 10 μM, 25 μM, or 50 μM; 24 h | ↓ Cell viability ↑ Apoptosis ↓ Necrosis | [75] |
| PC3 | 25, 50, and 100 μM; 24–72 h to assess apoptosis and DNA fragmentation 100 μM; 3 and 6 h to assess expression of JNK and p38 MAPK 100 μM; 48 h to assess expression of caspases and cytochrome c | ↑ Apoptosis ↑ Ceramide ↑ ssDNA ↑ JNK ↑ P38 MAPK ↓ Procaspases-3, -8, and -9 ↑ Accumulation of cytochrome c in cytoplasm | [66] |
| PC3, DU-145 | 10–100 μM; 4 and 24 h 20 μM; 24 h in 22RV1 and DU-145 only for fluorescent microscopy and nuclear staining | Curcumin compartmentalization within cytoplasm and exclusion from nucleus ↑ Cytotoxicity ↑ Apoptosis ↑ Autophagy | [77] |
| PC3 | 50 μM; 24 h | ↓ Proliferation Cell cycle arrest at G2/M phase ↑ Apoptosis ↓ NF-κB ↓ AP-1 | [91] |
| PC3 | 20 μM; 24 h | ↑ IL-6, INS, DDIT3, NDRG1, MIR-152 | [81] |
| PC3 | 15 μM; 24 h | ↓ Iκb kinase β ↑ IκBα ↓ CXCL1 ↓ CXCL2 ↓ NF-κB | [92] |
| PC3, DU-145, | 0–50 μM; 16 h, 24 h 25 and 50 μM; 1 h acute treatments | ↓ Cell growth ↓ Cell migration/invasion ↓ Total and activated matripase | [83] |
| PC3 | 0–20 μM; 48 h | ↓ Cell viability ↑ Apoptosis ↓ ld1 ↓ ld1 mRNA | [93] |
| PC3, DU-145, | 40 μM; 0–24 h to assess expression of ERK1/2, SAPK/JNK 0-60 μM; 24 h to assess expression of p65 and MUC1-C 10-100 μM; 24–72 h to assess cell viability | ↓ Cell viability ↑ P-ERK 1/2 ↑ P-SAPK/JNK in PC3 and DU-145 ↓ MUC1-C ↓ NF-κB subunit p65 | [94] |
| PC3 | 25 μM | ↓ EMT ↓ IL-6 ↓ ROS ↓ MAOA/mTOR/HIF-1α ↓ Cell invasion | [95] |
| DU-145 | 0–50 μM; 48 h to assess cell proliferation 15 μM; 48 h | ↑ Cell death ↓ HGF-induced cell scattering ↓ Wound closure ↓ Cell invasion ↑ E-cadherin ↓ Vimentin ↓ c-Met ↓ Snail mRNA ↓ p-ERK | [96] |
| DU-145 | 10–50 μM; 24–72 h to assess cell viability and apoptotic activity 25 μM; 48 h for immunoblotting and PCR 25 μM; 24–72 h to assess cell-cycle progression | ↓ Proliferation ↑ Apoptosis ↓ NOTCH1 ↓ Cell survival ↓ Cell growth Cell cycle arrest at the G0/G1 stage ↑ CDK inhibitors ↑ P21 ↑ P27 | [97] |
| PC3, DU-145 | 0–50 μM; 24h | ↑ Apoptosis ↑ Autophagy ↑ Cytotoxicity ↑ TFR1 ↑ IRP1 | [98] |
| DU-145, PC3 | 0–50 μM; 48 h to assess dose–response relationship 25 μM; 0–48 h to assess time–effect relationship 25 μM; 24 h for scratch assay 10 and 50 μM; 24 h for immunoblotting and PCR | ↓ Cell viability ↓ Proliferation ↓ Wound closure ↓ MT1-MMP mRNA ↓ MMP2 mRNA | [99] |
| DU145, PC3 | 10 μM; 1–5 days to assess cell viability and migration 10 μM; 0–24 h for PCR and immunoblotting 10 μM; 48 h to assess PGK1 expression | ↑ miR-143 ↓ Proliferation ↓ Cell migration ↓ KRAS signaling ↑ Docetaxel sensitivity ↓ PGK1 ↑ FOXD3 | [100] |
| PC3 | 5 μg/mL; 72 h | ↓ Cell viability Cell-cycle arrest ↑ Caspase-3 ↑ Uncleaved caspase-3 ↑ Uncleaved caspase-9 ↑ Caspase-12 ↑ PARP ↑ GRP78 ↑ Inositol-requiring enzyme 1 ↑ Calreticulin ↑ P-eIF2α ↑ Autophagy ↑ LC3B | [67] |
| PC3 | 25 μM; 48 h to evaluate apoptosis, gene and protein expression 25 μM; 24 h to assess H2O2 production 1.6–25 μM; 48 h to assess cell viability 10–50 μM; 30 min to assess ROS production | ↓ Cell viability ↑ Cytotoxicity ↑ Apoptosis ↑ ROS ↑ CuZnSOD ↑ TRX1 oxidation ↑ TRXR1 mRNA | [84] |
| PC3, DU-145 | 0–20 μM; 4 days to assess cell viability 0–10 μM; 4 days to assess proliferation, protein expression and miR-34a expression | ↓ Cell viability ↓ DNA synthesis ↓ Proliferation ↓ Cyclin D1 ↓ PCNA ↑ P21 ↑ miR-34α ↓ β-catenin ↓ c-myc | [68] |
| Cell Line | Curcumin Dosage | Effects | Reference |
|---|---|---|---|
| 22RV1 and DU-145 stem cells | 46.5 μM; 24 and 48h | ↓ Proliferation ↑ miR-145 ↓ lncRNA-ROR ↑ miR-3127 ↑ miR-3178 ↑ miR-1275 ↑ miR-3198 ↑ miR-1908 ↓ Ccnd1 ↓Cdk4 ↓Oct4 ↓CD44 ↓CD133 ↓CCND1 | [101] |
| 22RV1 and DU-145 stem cells | 46.5 μM; 24 and 48 h to assess proliferation | ↓ Proliferation ↓ Cell migration ↑ miR-770-5p ↑ miR-411 ↑ miR-1247 ↓ miR-382 ↓ miR-654-3p | [102] |
| Animal Model | Curcumin Dosage | Effects | Reference |
|---|---|---|---|
| Heterotopically implanted LNCaP cell tumors in athymic nude mice | 2% composition of a synthetic diet; 6 weeks | ↓ Proliferation ↓ Tumor growth ↑ Apoptosis ↑ Pycnotic brown staining nuclei ↓ Mitosis ↓ Nuclear cytoplasm ratio ↑ Fibrotic characteristics ↓ Angiogenesis ↓ Microvessel density ↓ CD31 | [103] |
| SCID mice implanted with DU-145 cells | 5 mg/kg b.w./3 times per week; 4 weeks | ↓ Tumor volume ↑ Apoptosis ↓ Cancer progression ↑ Caspase-3 activity ↓ MMP-2 | [88] |
| NCr nude male mice injected with PC3 cells | 6 µM three times per week; 4 weeks | ↑ Apoptosis ↓ Tumor growth ↓ Proliferation ↑ Caspase-3 ↑ PARP ↓ Akt ↓ GSK3Bα ↓ BAD ↓ IKKBα ↓ IκBα | [104] |
| PC3 tumor-bearing nude mice | 5 mg/kg b.w./5 times per week; 4 weeks | ↓ Tumor growth ↑ Gemcitabine effect ↑ Irradiation effect ↓ MDM2 | [70] |
| Balb c nude mice implanted with TRAIL-resistant LNCaP cells | 30 mg.kg b.w./3 times per week; 6 weeks | ↑ Apoptosis ↓ Proliferation ↑ TRAIL-R1/DR4 ↑ TRAIL-R2/DR5 ↑ Bax ↑ Bak ↑ p21/WAF1 ↑ p27/KIP1 ↓ Cyclin D1 ↓ VEGF ↓ uPA ↓ MMP-3 & -9 ↓ Bcl-2 ↓ Bcl-XL ↓ NF-κB activation ↓ Number of blood vessels ↓ Circulating EGFR-2-positive endothelial cells | [105] |
| TRAMP mice with prostate adenocarcinoma | 1–2% dietary composition; 10 or 16 weeks | ↓ Tumor formation ↓ Proliferation ↑ Apoptosis ↓ High-grade PIN ↓ Akt ↓ PDK1 ↓ FKHR | [106] |
| PTEN-KO mice | 250 µM; 7 weeks | ↓ Prostate adenocarcinomas ↓ Weight gain ↓ Prostate weight ↓ Epithelial cell proliferation ↓ mPIN lesions | [107] |
| Nude mice injected with PC3 cells | 10 µM; 24 h | ↓ PC3 cell proliferation ↓ Tumor growth ↓ Tumor progression ↓ VIP mRNA ↓ MMP-2 ↓ MMP-9 ↓ VEGF mRNA and protein ↓ VPAC1 receptor density | [108] |
| CD-1 Foxn1nu male mice injected with PC3 cells | 1% diet; 5 weeks | ↓ Lung metastasis ↓ Tumor size ↓ Human p53 protein ↓ Proliferating Ki-67 positive cells ~ Tumor morphology | [92] |
| Athymic nude mice inoculated with C4-3 cells | 25 µg; intratumoral injection once | ↓ Tumor growth ↑ β-catenin subcellular localization | [82] |
| Male nude mice inoculated with luciferase-expressed PC3 cells | 100 mg/kg b.w.; 3 weeks | ↓ Tumor growth ↓ Metastasis ↓ Metastatic lesions ↓ Activated matriptase | [83] |
| Athymic mice injected with C4-2 cells | 25 µg; intratumoral injection once | ↓ Tumor volume ↓ Tumor blood vessel density ↑ PLGA–CUR NP curcumin accumulation ↓ Developed vasculature ↓ Bcl-xL ↓ Nuclear AR ↑ β-catenin membrane staining ↓ Nuclear β-catenin activity ↓ CD31β ↓ miR-21 | [109] |
| SCID mice implanted with C4-2B cells | 1–2% diet; 4 weeks | ↓ Tumor progression ↓ Tumor growth ↓ Osteosclerotic lesions ↓ PSA levels ↑ Osteoblast markers ↑ Fatty globules ↓ TGF-β ↑ BMP-2 ↑ BMP-7 ↑ SMAD-1,-5,-8 ↑ UCP-1 | [110] |
| BALB/c mice injected with PC3 cells | 100 mg/kg b.w./day; 1 month | ↓ Tumor volume ↓ Id1 mRNA and protein ↑ Id1 cytoplasm locatization | [93] |
| Balb/c nude mice administered prostate PC3 cells | 25 mg/kg b.w./day; 30 days | ↑ Apoptosis ↓ PC3 cell growth ↓ Tumor volume and weight ↓ Bcl-2 ↑ Bax | [111] |
| Balb/c nude mice subcutaneously inoculated with LNCaP cells | 500 mg/kg b.w./3 times per week; 4 weeks | ↓ Prostate cancer tumor ↓ Tumor growth ↓ PSA level ↓ AR mRNA and protein levels | [112] |
| FVB/N mice injected with HMVP2 spheroids | 1.0% diet; 32 days | ↑ Apoptosis ↓ Tumor size ↓ Tumor weight ↑ Glutamine metabolism ↓ Glutamine uptake | [113] |
| BALB/c nude mice subcutaneously injected with CD44+/CD133+ HuPCaSCs pretreated with curcumin | IC50; 48 h | ↓ Tumor size ↓ Tumor development ↓ Oct4 ↓ Ki67 ↓ PSA ↓ Pap | [101] |
| Immunodeficient mice subcutaneously xenografted with LNCap cells | 30 mg/kg; 50 days | ↓ Tumor size ↑ Apoptosis ↓ Phospho-JNK ↓ Phospho-c-Jun ↓ Bcl-2 ↓ Bcl-xL ↓ H3K4me3 | [86] |
| Male TRAMP mice | 200 mg/kg b.w./day; 1 month | ↓ Testosterone level ↓ AKR1C2 surface expression | [85] |
| DU145 xenograft mice | 25 µg; 7 days or 12 h | ↓ DU145 cell solid tumors ↓ Tumor size and weight ↑ Tumor localization | [114] |
| Male Kumming mice injected with S180 cells | 18.8 mg/kg b.w./day; 10 days | ↑ Survival ↓ Tumor volume ↓ Tumor weight ↑ Necrosis ↑ Cell lysis ↑ Cell fragmentation | [115] |
| Prostate cancer CD1 mice xenografts (PC3, 22rv1, and DU145 cell-lines) | 800 mg/kg; days 1, 9, 18, 27, and 34 | ↑ Apoptosis ↑ Fibrosis ↓ Inflammation ↓ Tumor size ↓ Tumor weight ↓ Tumor progression ↑ Collagen deposition ↓ Monocyte infiltration ↑ TUNEL-positive cells | [116] |
| Pten-deficient mice | 76 or 380 mg/kg b.w./day; 16 weeks | ↓ Proliferation ↑ Liver weight ↓ High-grade PIN ↓ Incidence of atrophic glands ↑ Stroma thickness | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Termini, D.; Den Hartogh, D.J.; Jaglanian, A.; Tsiani, E. Curcumin against Prostate Cancer: Current Evidence. Biomolecules 2020, 10, 1536. https://doi.org/10.3390/biom10111536
Termini D, Den Hartogh DJ, Jaglanian A, Tsiani E. Curcumin against Prostate Cancer: Current Evidence. Biomolecules. 2020; 10(11):1536. https://doi.org/10.3390/biom10111536
Chicago/Turabian StyleTermini, Deborah, Danja J. Den Hartogh, Alina Jaglanian, and Evangelia Tsiani. 2020. "Curcumin against Prostate Cancer: Current Evidence" Biomolecules 10, no. 11: 1536. https://doi.org/10.3390/biom10111536
APA StyleTermini, D., Den Hartogh, D. J., Jaglanian, A., & Tsiani, E. (2020). Curcumin against Prostate Cancer: Current Evidence. Biomolecules, 10(11), 1536. https://doi.org/10.3390/biom10111536






