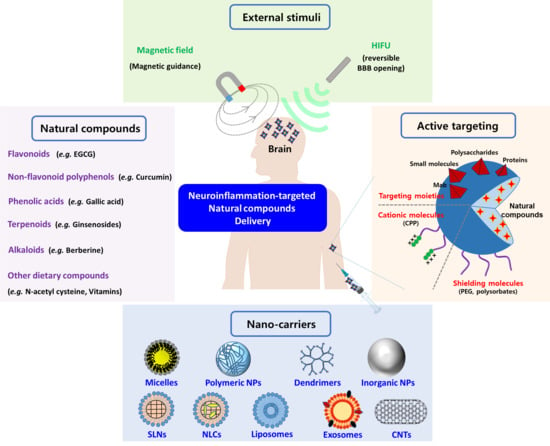
The Role of Natural Compounds and their Nanocarriers in the Treatment of CNS Inflammation
Abstract
1. Introduction
2. Neuroinflammation and CNS Degenerative Diseases
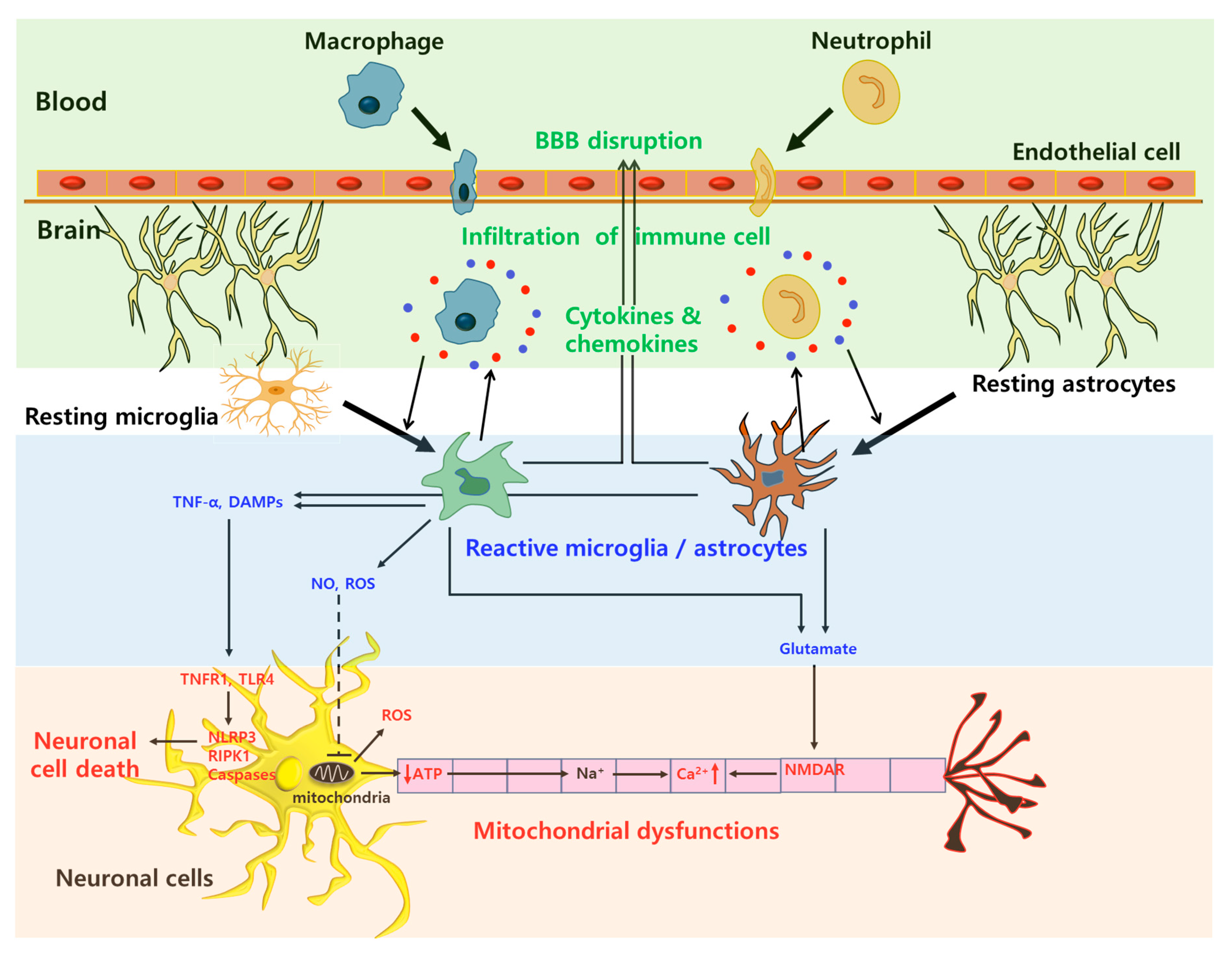
2.1. Molecular Mechanisms of Neuroinflammation and Neurodegeneration
2.1.1. Reactive Microglia and Astrocytes
2.1.2. Mitochondrial Dysfunction
2.1.3. BBB Disruption
2.1.4. Neuronal Apoptosis
2.1.5. Pyroptosis
2.1.6. Necroptosis
2.1.7. Neuronal Autophagy
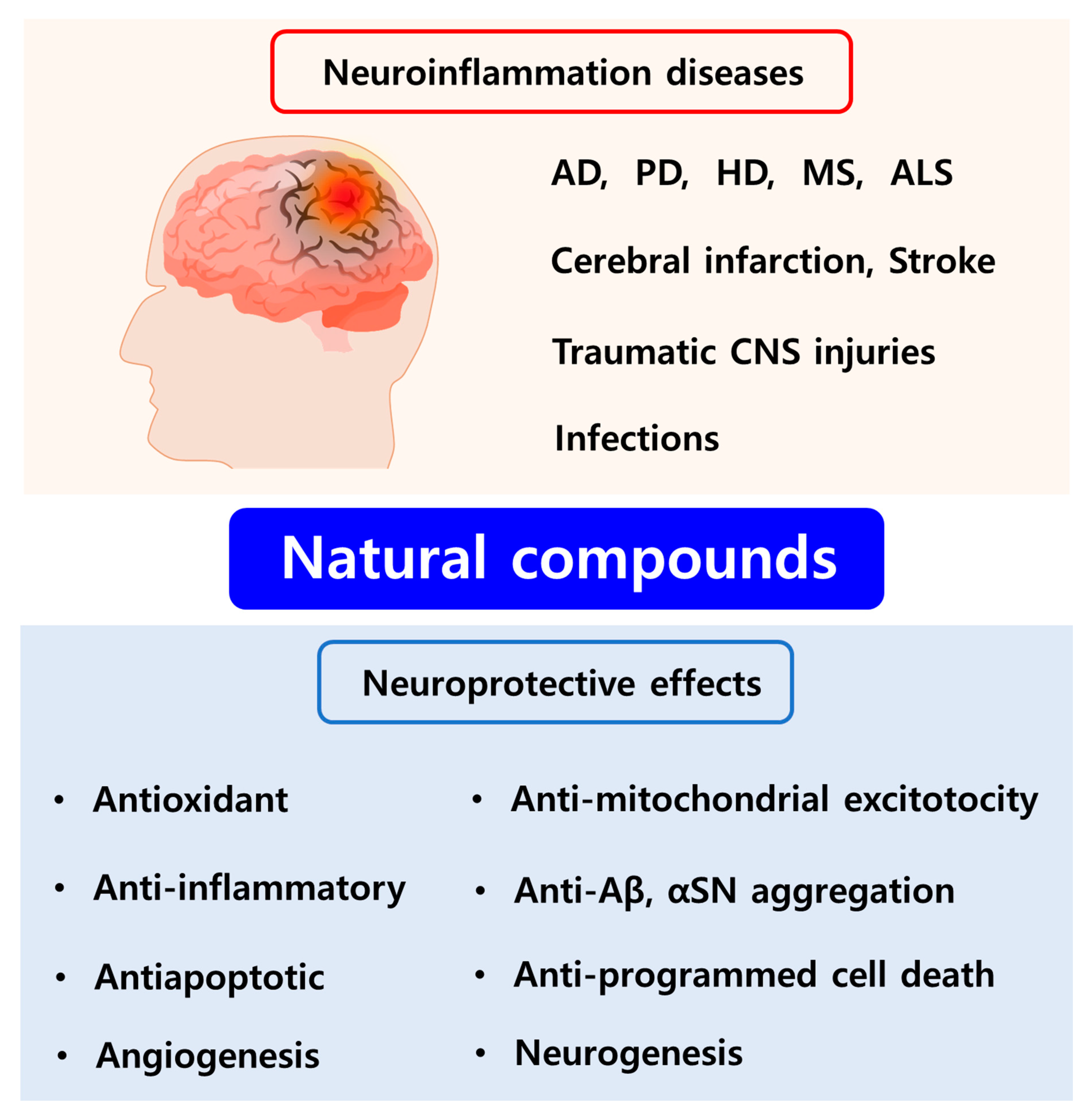
2.1.8. CNS Disorders with Neurodegeneration
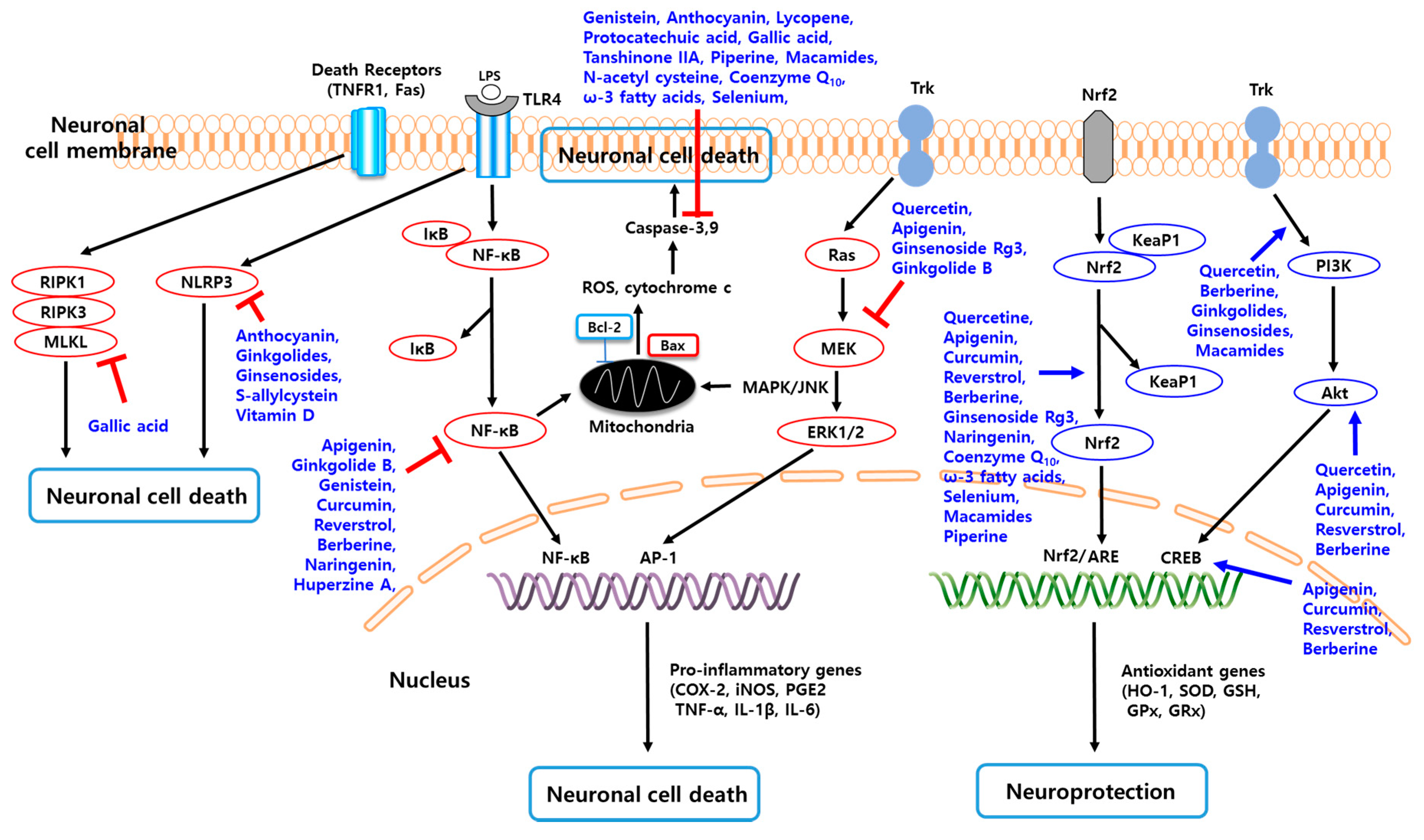
3. Neuroprotective Effects of Potential Natural Compounds and Their Limitation
3.1. Various Natural Compounds for the Treatment of Neuroinflammation
3.1.1. Flavonoid Polyphenols
3.1.2. Non-Flavonoid Polyphenols
3.1.3. Phenolic Acids
3.1.4. Terpenoids
3.1.5. Alkaloids
3.1.6. Other Dietary Compounds
3.2. Physico-Chemical and Pharmacokinetic Limitations
4. Various Nanocarriers Containing Natural Compounds for the Treatment of Neuroinflammation
4.1. Polymer-Based NPs
4.1.1. Polymeric Micelles
4.1.2. Synthetic Polymer NPs
4.1.3. Natural Polymer NPs
4.1.4. Dendrimers
4.2. Lipid-Based NPs
4.2.1. Lipid NPs (Solid Lipid NPs; SLNs and Nanostructured Lipid Carriers; NLCs)
4.2.2. Liposomes
4.3. Inorganic NPs
4.3.1. Se NPs
4.3.2. Gold NPs
4.3.3. Iron Oxide NPs
4.4. Other NPs
4.4.1. Carbon-Based NPs
4.4.2. Albumin NPs
4.4.3. Exosomes
4.5. Advanced NPs for Active Targeting of CNS Inflammation
4.5.1. Targeting Ligand-Functionalized NPs
4.5.2. Nanovalve Systems
4.5.3. Biomimetic NPs
5. Conclusions and Future Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Chitnis, T.; Weiner, H.L. CNS inflammation and neurodegeneration. J. Clin. Investig. 2017, 127, 3577–3587. [Google Scholar] [CrossRef] [PubMed]
- Guzman-Martinez, L.; Maccioni, R.B.; Andrade, V.; Navarrete, L.P.; Pastor, M.G.; Ramos-Escobar, N. Neuroinflammation as a common feature of neurodegenerative disorders. Front. Pharmacol. 2019, 10, 1008. [Google Scholar] [CrossRef] [PubMed]
- Picca, A.; Calvani, R.; Coelgo-Junior, H.J.; Landi, F.; Bernabei, R.; Marzetti, E. Mitochondrial dysfunction, oxidative stress, and neuroinflammation: Intertwined roads to neurodegeneration. Antioxidants 2020, 9, 647. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Gómez, J.A.; Kavanagh, E.; Engskog-Vlachos, P.; Engskog, M.K.R.; Herrera, A.J.; Espinosa-Oliva, A.M.; Joseph, B.; Hajji, N.; Venero, J.L.; Burguillos, M.A. Microglia: Agents of the CNS pro-inflammatory response. Cells 2020, 9, 1717. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative stress: A key modulator in neurodegenerative diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef]
- Aguilera, G.; Colín-González, A.L.; Rangel-López, E.; Chavarría, A.; Santamaría, A. Redox signaling, neuroinflammation, and neurodegeneration. Antioxid. Redox Signal. 2018, 28, 1626–1651. [Google Scholar] [CrossRef]
- Lee, K.H.; Kang, T.B. The molecular links between cell death and inflammasome. Cells 2019, 8, 1057. [Google Scholar] [CrossRef]
- Makkar, R.; Behl, T.; Bungau, S.; Zengin, G.; Mehta, V.; Kumar, A.; Uddin, M.S.; Ashraf, G.M.; Abdel-Daim, M.M.; Arora, S.; et al. Nutraceuticals in neurological disorders. Int. J. Mol. Sci. 2020, 21, 4424. [Google Scholar] [CrossRef]
- Uddin, M.S.; Hossain, M.F.; Mamun, A.A.; Shah, M.A.; Hasana, S.; Bulbul, I.J.; Sarwar, M.S.; Mansouri, R.A.; Ashraf, G.M.; Rauf, A.; et al. Exploring the multimodal role of phytochemicals in the modulation of cellular signaling pathways to combat age-related neurodegeneration. Sci. Total Environ. 2020, 725, 138313. [Google Scholar] [CrossRef]
- Maheshwari, R.K.; Singh, A.K.; Gaddipati, J.; Srimal, R.C. Multiple biological activities of curcumin: A short review. Life Sci. 2006, 78, 2081–2087. [Google Scholar] [CrossRef]
- Bertoncello, K.T.; Aguiar, G.P.S.; Oliveira, J.V.; Siebal, A.M. Micronization potentiates curcumin’s anti-seizure effect and brings an important advance in epilepsy treatment. Sci. Rep. 2018, 8, 2645. [Google Scholar] [CrossRef] [PubMed]
- Qneibi, M.; Hamed, O.; Fares, O.; Jaradat, N.; Natsheh, A.R.; AbuHasan, Q.; Emwas, N.; Al-Kerm, R.; Al-Kerm, R. The inhibitory role of curcumin derivatives on AMPA receptor subunits and their effect on the gating biophysical properties. Eur. J. Pharm. Sci. 2019, 136, 104951. [Google Scholar] [CrossRef] [PubMed]
- Qneibi, M.; Jaradat, N.; Emwas, N. Effect of geraniol and citronellol essential oils on the biophysical gating properties of AMPA receptors. Appl. Sci. 2019, 9, 4693. [Google Scholar] [CrossRef]
- Liu, Z.; Ying, Y. The inhibitory effect of curcumin on virus-induced cytokine storm and its potential use in the associated severe pneumonia. Front. Cell Dev. Biol. 2020, 8, 479. [Google Scholar] [CrossRef]
- Castejón-Vega, B.; Giampieri, R.; Alvarez-Suarez, J.M. Nutraceutical compounds targeting inflammasomes in human diseases. Int. J. Mol. Sci. 2020, 21, 4829. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Bramanti, P.; Mazzon, E. Activation of Nrf2 by natural bioactive compounds: A promising approach for stroke? Int. J. Mol. Sci. 2020, 21, 4875. [Google Scholar] [CrossRef]
- Niu, X.; Chen, J.; Gao, J. Nanocarriers as a powerful vehicle to overcome blood-brain barrier in treating neurodegenerative diseases: Focus on recent advances. Asian J. Pharm. Sci. 2019, 14, 480–496. [Google Scholar] [CrossRef]
- Moradi, S.Z.; Momtaz, S.; Bayrami, Z.; Farzaei, M.H.; Abdollahi, M. Nanoformulations of herbal extracts in treatment of neurodegenerative disorders. Front. Bioeng. Biotechnol. 2020, 8, 238. [Google Scholar] [CrossRef]
- DiSabato, D.J.; Quan, N.; Godbout, J.P. Neuroinflammation: The devil is in the details. J. Neurochem. 2016, 139, 136–153. [Google Scholar] [CrossRef]
- Chen, W.W.; Zhang, X.; Huang, W.J. Role of neuroinflammation in neurodegenerative dieases (Review). Mol. Med. Rep. 2016, 13, 3391–3396. [Google Scholar] [CrossRef]
- Sochocka, M.; Diniz, B.S.; Leszek, J. Inflammatory response in the CNS: Friend of foe? Mol. Neurobiol. 2017, 54, 8071–8089. [Google Scholar] [CrossRef] [PubMed]
- Tjalkens, R.B.; Popichak, K.A.; Kirkley, K.A. Inflammatory activation of microglia and astrocytes in manganese neurotoxicity. Adv. Neurobiol. 2017, 18, 159–181. [Google Scholar] [PubMed]
- Palpagama, T.H.; Waldvogel, H.J.; Faull, R.L.M.; Kwakowsky, A. The role of microglia and astrocytes in Huntington’s disease. Front. Mol. Neurosci. 2019, 12, 258. [Google Scholar] [CrossRef] [PubMed]
- Skowronska, K.; Obara-Michlewska, M.; Zielinska, M.; Albrecht, J. NMDA receptors in astrocytes: In search for roles in neurotransmission and astrocytic homeostasis. Int. J. Mol. Sci. 2019, 20, 309. [Google Scholar] [CrossRef]
- Olloquequi, J.; Cornejo-Córdova, E.; Verdaguer, E.; Soriano, F.X.; Binvignat, O.; Auladell, C.; Camins, A. Excitotoxicity in the pathogenesis of neurological and psychiatric disorders: Therapeutic implications. J. Psychopharmacol. 2018, 32, 265–275. [Google Scholar] [CrossRef]
- Sahel, D.K.; Kaira, M.; Raj, K.; Sharma, S.; Singh, S. Mitochondrial dysfunctioning and neuroinflammation: Recent highlights on the possible mechanisms involved in traumatic brain injury. Neurosci. Lett. 2019, 170, 134347. [Google Scholar] [CrossRef]
- Kim, K.T.; Lee, H.S.; Lee, J.J.; Park, E.K.; Lee, B.S.; Lee, J.Y.; Bae, J.S. Nanodelivery systems for overcoming limited transportation of therapeutic molecules through the blood-brain barrier. Future Med. Chem. 2018, 10, 2659–2674. [Google Scholar] [CrossRef]
- Sulfan, S.; Lyon, K.A.; Shapiro, L.A.; Huang, J.H. Neuroinflammation and blood-brain barrier disruption following traumatic brain injury: Pathophysiology and potential therapeutic targets. J. Neuro. Res. 2020, 98, 19–28. [Google Scholar]
- Haruwaka, K.; Ikegami, A.; Tachibana, Y.; Ohno, N.; Konishi, H.; Hashimoto, A.; Matsumoto, M.; Kato, D.; Ono, R.; Kiyama, H.; et al. Dual microglia effects on blood brain barrier permeability induced by systemic inflammation. Nat. Commun. 2019, 10, 5816. [Google Scholar] [CrossRef]
- Yang, C.; Hawkins, K.E.; Doré, S.; Candelario-Jalil, E. Neuroinflammatory mechanisms of blood-brain barrier damage in ischemic stroke. Am. J. Physiol. Cell Phsysiol. 2019, 316, 135–153. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Voet, S.; Srinivasan, S.; Lamkanfi, M.; van Loo, G. Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol. Med. 2019, 11, e10248. [Google Scholar] [CrossRef] [PubMed]
- Mckenzie, B.A.; Dixit, V.M.; Power, C. Fiery cell death: Pyroptosis in the central nervous system. Trends Neurosci. 2020, 43, 55–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Tang, M.B.; Luo, H.Y.; Shi, C.H.; Xu, Y.M. Necroptosis in neurodegenerative diseases: A potential therapeutic target. Cell Death Dis. 2017, 8, e2905. [Google Scholar] [CrossRef]
- Yuan, J.; Amin, P.; Ofengeim, D. Necroptosis and RIPK1-mediated neuroinflammation in CNS diseases. Nat. Rev. Neurosci. 2019, 20, 19–33. [Google Scholar] [CrossRef]
- Menzies, F.M.; Fleming, A.; Caricasole, A.; Bento, C.F.; Andrews, S.P.; Ashkenazi, A.; Füllgrabe, J.; Jackson, A.; Sanchéz, M.J.; Karabiyik, C.; et al. Autophagy and neurodegeneration: Pathogenic mechanisms and therapeutic opportunities. Neuron 2017, 93, 1015–1034. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, C.J. Astrocytes autophagy in aging and neurodegenerative disorders. Biomed. Pharmacother. 2020, 122, 109691. [Google Scholar] [CrossRef]
- Filomeni, G.; De Zio, D.; Cecconi, F. Oxidative stress and autophagy: The clash between damage and metabolic needs. Cell Death Differ. 2015, 22, 377–388. [Google Scholar] [CrossRef]
- Periyasamy, P.; Guo, M.L.; Buch, S. Cocaine induces astrocytosis through ER stress-mediated activation of autophagy. Autophagy 2016, 12, 1310–1329. [Google Scholar] [CrossRef]
- Ransohoff, R.M. How neuroinflammation contributes to neurodegeneration. Science 2016, 353, 777–783. [Google Scholar] [CrossRef]
- Leyns, C.E.G.; Holtzman, D.M. Glial contributions to neurodegeneration in tauopathies. Mol. Neurodegener. 2017, 12, 50. [Google Scholar] [CrossRef]
- Cortes, N.; Andrade, V.; Guzman-Martinez, L.; Estrella, M.; Maccioni, R.B. Neuroimmune tau mechanisms: Their role in the progression of neuronal degeneration. Int. J. Mol. Sci. 2018, 19, 956. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, T.; Mochizuki, H. The regulatory role of α-synuclein and parkin in neuronal cell apoptosis: Possible implications for the pathogenesis of Parkinson’s disease. Apoptosis 2010, 15, 1312–1321. [Google Scholar] [CrossRef] [PubMed]
- Collins, L.M.; Toulouse, A.; Connor, T.J.; Nolan, Y.M. Contribution of central and systemic inflammation to the pathophysiology of Parkinson’s disease. Neuropharmacology 2012, 62, 2154–2168. [Google Scholar] [CrossRef] [PubMed]
- Ross, C.A.; Aylward, E.H.; Wild, E.J.; Langbehn, D.R.; Long, J.D.; Warner, J.H.; Scahill, R.I.; Leavitt, B.R.; Stout, J.C.; Paulsen, J.S.; et al. Huntington disease: Natural history, biomarkers and prospects for therapeutics. Nat. Rev. Neurol. 2014, 10, 204–216. [Google Scholar] [CrossRef]
- Bates, G.P.; Dorsey, R.; Gusella, J.F.; Hayden, M.R.; Kay, C.; Leavitt, B.R.; Nance, M.; Ross, C.A.; Scahill, R.I.; Wetzel, R.; et al. Huntington disease. Nat. Rev. Dis. Primers 2015, 1, 15005. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujiharar, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef]
- Hooten, K.G.; Beers, D.R.; Zhao, W.; Appel, S.H. Protective and toxic neuroinflammation in amyotrophic lateral sclerosis. Neurotherapeutics 2015, 12, 364–375. [Google Scholar] [CrossRef]
- Liu, J.; Wang, F. Role of neuroinflammation in amyotrophic lateral sclerosis: Cellular mechanisms and therapeutic implications. Front. Immunol. 2017, 8, 1005. [Google Scholar] [CrossRef]
- Lee, R.H.C.; Lee, M.H.H.; Wu, C.Y.C.; Silva, A.C.E.; Possoit, H.E.; Hsieh, T.H.; Minagar, A.; Lin, H.W. Cerebral ischemia and neurodegeneration. Neural Regen. Res. 2018, 13, 373–385. [Google Scholar]
- Jayaraj, R.L.; Azimullah, S.; Beiram, R.; Jalal, F.Y.; Rosenberg, G.A. Neuroinflammation: Friend and foe for ischemic stroke. J. Neuroinflamm. 2019, 16, 142. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Ryu, J.S.; Vig, P.J. Current strategies for therapeutic drug delivery after traumatic CNS injury. Ther. Deliv. 2019, 10, 251–263. [Google Scholar] [CrossRef] [PubMed]
- Csobonyeiova, M.; Polak, S.; Zamborsky, R.; Danisovic, L. Recent progress in the regulation of spinal cord injuries by induced pluripotent stem cells. Int. J. Mol. Sci. 2019, 20, 3838. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.Y.; Lee, A.Y.W. Traumatic brain injuries: Pathophysiology and potential therapeutic targets. Front. Cell Neurosci. 2019, 13, 528. [Google Scholar] [CrossRef] [PubMed]
- Conte, R.; Marturano, V.; Peluso, G.; Calarco, A.; Cerruti, P. Recent advances in nanoparticle-mediated delivery of anti-inflammatory phytocompounds. Int. J. Mol. Sci. 2017, 18, 709. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.R.; Grosso, C.; Delerue-Matos, C.; Rocha, J.M. Comprehensive review on the interaction between natural compounds and brain receptors: Benefits and toxicity. Eur. J. Med. Chem. 2019, 174, 87–115. [Google Scholar] [CrossRef]
- Paolo, M.D.; Papi, L.; Gori, F.; Turillazzi, E. Natural products in neurodegenerative diseases: A great promise but an ethical challenge. Int. J. Mol. Sci. 2019, 20, 5170. [Google Scholar] [CrossRef]
- Spencer, J.P.E.; Vafeiadou, K.; Williams, R.J.; Vauzour, D. Neuroinflammation: Modulation by flavonoids and mechanisms of action. Mol. Aspects Med. 2012, 33, 83–97. [Google Scholar] [CrossRef]
- Calis, Z.; Mogulkoc, R.; Baltaci, A.K. The roles of flavonoles/flavonoids in neurodegeneration and neuroinflammation. Mini Rev. Med. Chem. 2020, 20, 1475–1488. [Google Scholar] [CrossRef]
- Ginwala, R.; Bhavsar, R.; Chigbu, D.I.; Jain, P.; Khan, Z.K. Potential role of flavonoids in treating chronic inflammatory diseases with a special focus on the anti-inflammatory activity of apigenin. Antioxidants 2019, 8, 35. [Google Scholar] [CrossRef]
- Ali, F.; Rahul; Naz, F.; Jyoti, S.; Siddique, Y.H. Health functionality of apigenin: A review. Int. J. Food Prop. 2017, 20, 1197–1238. [Google Scholar] [CrossRef]
- Zhang, T.; Su, J.; Guo, B.; Wang, K.; Li, X.; Liang, G. Apigenin protects blood-brain barrier and ameliorates early brain injury by inhibiting TLR4-mediated inflammatory pathway in subarachnoid hemorrhage rats. Int. Immunopharmacol. 2015, 28, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wang, J.L.; Liu, R.; Li, X.X.; Li, J.F.; Zhang, L. Neuroprotective, Anti-amyloidogenic and neurotrophic effects of apigenin in an Alzheimer’s disease mouse model. Molecules 2013, 18, 9949. [Google Scholar] [CrossRef] [PubMed]
- Balez, R.; Steiner, N.; Engel, M.; Muñoz, S.S.; Lum, J.S.; Wu, Y.; Wang, D.; Vallotton, P.; Sachdev, P.; O’Connor, M.; et al. Neuroprotective effects of apigenin against inflammation, neuronal excitability and apoptosis in an induced pluripotent stem cell model of Alzheimer’s disease. Sci. Rep. 2016, 6, 31450. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Pourbagher-Shahri, A.M.; Ashrafizadeh, M.; Folgado, S.L.; Rajabpour-Sanati, A.; Khazdair, M.R.; Samarghandian, S. Green tea catechins inhibit microglial activation which prevents the development of neurological disorders. Neural. Regen. Res. 2020, 15, 1792–1798. [Google Scholar]
- Li, Q.; Gordon, M.; Tan, J.; Morgan, D. Oral administration of green tea epigallocatechin-3-gallate (EGCG) reduces amyloid beta deposition in transgenic mouse model of Alzheimer’s disease. Exp. Neurol. 2006, 198, 576. [Google Scholar] [CrossRef]
- Lee, Y.J.; Choi, D.Y.; Yun, Y.P.; Han, S.B.; Oh, K.W.; Hong, J.T. Epigallocatechin-3-gallate prevents systemic inflammation-induced memory deficiency and amyloidogenesis via its anti-neuroinflammatory properties. J. Nutr. Biochem. 2013, 24, 298–310. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Sunphenon EGCG (Epigallocatechin-Gallate) in the Early Stage of Alzheimer’s Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT00951834 (accessed on 10 August 2020).
- Khan, H.; Ulah, H.; Aschner, M.; Cheang, W.S.; Akkol, E.K. Neuroprotective effects of quercetin in Alzheimer’s disease. Biomolecules 2020, 10, 59. [Google Scholar] [CrossRef]
- Wang, D.M.; Li, S.Q.; Wu, W.L.; Zhu, X.Y.; Wang, Y.; Yuan, H.Y. Effects of long-term treatment with quercetin on cognition and mitochondrial function in a mouse model of Alzheimer’s disease. Neurochem. Res. 2014, 39, 1533–1543. [Google Scholar] [CrossRef]
- Kim, B.W.; Koppula, S.; Park, S.Y.; Hwang, J.W.; Park, P.J.; Lim, J.H.; Choi, D.K. Attenuation of inflammatory-mediated neurotoxicity by Saururus chinensis extract in LPS-induced BV-2 microglia cells via regulation of NF-κB signaling and anti-oxidant properties. BMC Complement. Altern. Med. 2014, 12, 502. [Google Scholar] [CrossRef]
- Nouri, A.; Fakhri, S.; El-Senduny, F.F.; Sanadgol, N.; Abd-ElGhani, G.E.; Farzaei, M.H.; Chen, J.T. On the neuroprotective effects of naringenin: Pharmacological targets, signaling pathways, molecular mechanisms, and clinical perspective. Biomolecules 2019, 9, 690. [Google Scholar] [CrossRef] [PubMed]
- Mani, S.; Sekar, S.; Chidambaram, S.B.; Sevanan, M. Naringenin protects against 1-methyl-4-phenylpyridinium-induced neuroinflammation and resulting reactive oxygen species production in SH-SY5Y cell line: An in vitro model of Parkinson’s disease. Pharmacogn. Mag. 2018, 14, 458–464. [Google Scholar]
- Park, H.Y.; Kim, G.Y.; Choi, Y.H. Naringenin attenuates the release of pro-inflammatory mediators from lipopolysaccharide-stimulated BV2 microglia by inactivating nuclear factor-kappaB and inhibiting mitogen-activated protein kinases. Int. J. Mol. Med. 2012, 30, 204–210. [Google Scholar] [PubMed]
- Wu, L.H.; Lin, C.; Lin, H.Y.; Liu, Y.S.; Wu, C.Y.; Tsai, C.F.; Chang, P.C.; Yeh, W.L.; Lu, D.Y. Naringenin suppresses neuroinflammatory responses through inducing suppressor of cytokine signaling 3 expression. Mol. Neurobiol. 2016, 53, 1080–1091. [Google Scholar] [CrossRef]
- Uddin, M.S.; Kabir, M.T. Emerging signal regulating potential of genistein against Alzheimer’s disease: A promising molecule of interest. Front. Cell Dev. Biol. 2019, 7, 197. [Google Scholar] [CrossRef]
- Xi, Y.D.; Yu, H.L.; Ding, J.; Ma, W.W.; Yuan, L.H.; Feng, J.F.; Xiao, Y.X.; Xiao, R. Flavonoids protect cerebrovascular endothelial cells through Nrf2 and PI3K from β-amyloid peptide-induced oxidative damage. Curr. Neurovasc. Res. 2012, 9, 32–41. [Google Scholar] [CrossRef]
- Ma, W.; Ding, B.; Yu, H.; Yuan, L.; Xi, Y.; Xiao, R. Genistein alleviates β-amyloid-induced inflammatory damage through regulating toll-like receptor 4/nuclear factor κ B. J. Med. Food 2015, 18, 273–279. [Google Scholar] [CrossRef]
- Bonet-Costa, V.; Herranz-Pérez, V.; Blanco-Gandía, M.; Mas-Bargues, C.; Inglés, M.; Garcia-Tarraga, R.; Rodriguez-Arias, M.; Miñarro, J.; Borras, C.; Garcia-Verdugo, J.M.; et al. Clearing amyloid-β through PPARγ/ApoE activation by genistein is a treatment of experimental Alzheimer’s disease. J. Alzheimers Dis. 2016, 51, 701–711. [Google Scholar] [CrossRef]
- Winter, A.N.; Bickford, P.C. Anthocyanins and their metabolites as therapeutic agents for neurodegenerative disease. Antioxidants 2019, 8, 333. [Google Scholar] [CrossRef]
- Poulose, S.M.; Bielinski, D.F.; Carey, A.; Schauss, A.G.; Shukitt-Hale, B. Modulation of oxidative stress, inflammation, autophagy and expression of Nrf2 in hippocampus and frontal cortex of rats fed with acai-enriched diets. Nutr. Neurosci. 2017, 20, 305–315. [Google Scholar] [CrossRef]
- Pan, Z.; Cui, M.; Dai, G.; Yuan, T.; Li, Y.; Ji, T.; Pan, Y. Protective effect of anthocyanin on neurovascular unit in cerebral ischemia/reperfusion injury in rats. Front. Neurosci. 2018, 12, 947. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Calina, D.; Docea, A.O.; Koirala, N.; Aryal, S.; Lombardo, D.; Pasqua, L.; Taheri, Y.; Castillo, C.M.S.; Martorell, M.; et al. Curcumin’s nanomedicine formulations for therapeutic application in neurological diseases. J. Clin. Med. 2020, 9, 430. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.J.; Li, Z.H.; Liu, L.; Tang, W.X.; Wang, Y.; Dong, M.R.; Xiao, C. Curcumin attenuates beta-amyloid-induced neuroinflammation via activation of peroxisome proliferator-activated receptor-gamma function in a rat model of Alzheimer’s disease. Front. Pharmacol. 2016, 7, 261. [Google Scholar] [CrossRef] [PubMed]
- Porro, C.; Cianciulli, A.; Trotta, T.; Lofrumento, D.D.; Panaro, M.A. Curcumin regulates anti-inflammatory responses by JAK/STAT/SOCS signaling pathway in BV-2 microglial cells. Biology 2019, 8, 51. [Google Scholar] [CrossRef]
- Ullah, F.; Asgarov, R.; Venigalla, M.; Liang, H.; Niedermayer, G.; Münch, G.; Gyengesi, E. Effects of a solid lipid curcumin particle formulation on chronic activation of microglia and astroglia in the GFAP-IL6 mouse model. Sci. Rep. 2020, 10, 2365. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Efficacy and Safety of Curcumin Formulation in Alzheimer’s Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT01001637 (accessed on 10 August 2020).
- Bastianetto, S.; Ménard, C.; Quirion, R. Neuroprotective action of resveratrol. Biochim. Biophys. Acta 2015, 1852, 1195–1201. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, J.; Shi, J.S. Anti-inflammatory activities of resveratrol in the brain: Role of resveratrol in microglial activation. Eur. J. Pharmacol. 2010, 636, 1–7. [Google Scholar] [CrossRef]
- Zhang, J.; Feng, X.; Wu, J.; Xu, H.; Li, G.; Zhu, D.; Yue, Q.; Liu, H.; Zhang, Y.; Sun, D.; et al. Neuroprotective effects of resveratrol on damages of mouse cortical neurons induced by beta-amyloid through activation of SIRT1/Akt1 pathway. BioFactors 2014, 40, 258–267. [Google Scholar] [CrossRef]
- Quincozes-Santos, A.; Bobermin, L.D.; Latini, A.; Wajner, M.; Souza, D.O.; Goncalves, C.A.; Gottfried, C. Resveratrol protects C6 astrocyte cell line against hydrogen peroxide-induced oxidative stress through heme oxygenase 1. PLoS ONE 2013, 8, e64372. [Google Scholar] [CrossRef]
- ClinicalTrials.gov. Resveratrol for Alzheimer’s Disease. Available online: https://clinicaltrials.gov/ct2/show/NCT01504854 (accessed on 10 August 2020).
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef]
- Chen, D.; Huang, C.; Chen, Z. A review for the pharmacological effect of lycopene in central nervous system disorders. Biomed. Pharmacother. 2019, 111, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Prakash, A.; Kumar, A. Lycopene protects against memory impairment and mito-oxidative damage induced by colchicine in rats: An evidence of nitric oxide signaling. Eur. J. Pharmacol. 2014, 721, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.Y.; Huang, B.R.; Yeh, W.L.; Lee, C.H.; Huang, S.S.; Lai, C.H.; Lin, H.; Lu, D.Y. Antineuroinflammation effects of lycopene via activation of adenosine monophosphate-activated protein kinase-α1/heme oxygenase-1 pathways. Neurobiol. Aging 2014, 35, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.; Lim, J.W.; Kim, H. Inhibitory effect of lycopene on amyloid-β-induced apoptosis in neuronal cells. Nutrients 2017, 9, 883. [Google Scholar] [CrossRef] [PubMed]
- Krzysztoforska, K.; Mirowska-Guzel, D.; Widy-Tyszkiewicz, E. Pharmacological effects of protocatechuic acid and its therapeutic potential in neurodegenerative diseases: Review on the basis of in vitro and in vivo studies in rodents and humans. Nutr. Neurosci. 2019, 22, 72–82. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, X.; Lv, C.; Li, C.; Yu, Y.; Wang, X.; Han, F. Protocatechuic acid ameliorates neurocognitive functions impairment induced by chronic intermittent hypoxia. Sci. Rep. 2015, 5, 14507. [Google Scholar] [CrossRef]
- Kaewmool, C.; Kongtawelert, P.; Phitak, T.; Pothacharoen, P.; Udomruk, S. Protocatechuic acid inhibits inflammatory responses in LPS-activated BV2 microglia via regulating SIRT1/NF-κB pathway contributed to the suppression of microglial activation-induced PC12 cell apoptosis. J. Neuroimmunol. 2020, 341, 577164. [Google Scholar] [CrossRef]
- Shabani, S.; Rabiei, Z.; Amini-Khoei, H. Exploring the multifaceted neuroprotective actions of gallic acid: A review. Int. J. Food Prop. 2020, 23, 736–752. [Google Scholar] [CrossRef]
- Chandrasekhar, Y.; Kumar, G.P.; Ramya, E.M.; Anilakumar, K.R. Gallic acid protects 6-OHDA induced neurotoxicity by attenuating oxidative stress in human dopaminergic cell line. Neurochem. Res. 2018, 43, 1150–1160. [Google Scholar] [CrossRef]
- Liu, Y.L.; Hsu, C.C.; Huang, H.J.; Chang, C.J.; Sun, S.H.; Lin, A.M.Y. Gallic acid attenuated LPS-induced neuroinflammation: Protein aggregation and necroptosis. Mol. Neurobiol. 2020, 57, 96–104. [Google Scholar] [CrossRef]
- Feng, Z.; Sun, Q.; Chen, W.; Bai, Y.; Hu, D.; Xei, X. The neuroprotective mechanisms of ginkgolides and bilobalide in cerebral ischemic injury: A literature review. Mol. Med. 2019, 25, 57. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Xu, Y.; Yuan, J. Ginkgolide B ameliorates NLRP3 inflammasome activation after hypoxic-ischemic brain injury in the neonatal male rat. Int. J. Dev. Neurosci. 2018, 69, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.H.; Ge, J.B.; Li, M.; Wu, F.; Zhang, W.; Qin, Z.H. Inhibition of NF-κB activation is associated with anti-inflammatory and anti-apoptotic effects of ginkgolide B in a mouse model of cerebral ischemia/reperfusion injury. Eur. J. Pharm. Sci. 2012, 47, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Li, J.; Peng, Q.; Liu, Y.; Liu, W.; Luo, C.; Peng, J.; Li, J.; Yung, K.K.L.; Mo, Z. Neuroprotective effects of bilobalide on cerebral ischemia and reperfusion injury are associated with inhibition of pro-inflammatory mediator production and down-regulation of JNK1/2 and p38 MAPK activation. J. Neuroinflamm. 2014, 11, 167. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wu, X.; Yu, S.; Lin, X.; Wu, J.; Li, L.; Zhao, J.; Zhao, Y. Neuroprotection of tanshinone IIA against cerebral ischemia/reperfusion injury through inhibition of macrophage migration inhibitory factor in rats. PLoS ONE 2012, 7, e41065. [Google Scholar] [CrossRef]
- Feng, Y.; Feng, F.; Zheng, C.; Zhou, Z.; Jiang, M.; Liu, Z.; Xie, F.; Sun, X.; Wu, Z. Tanshinone IIA attenuates demyelination and promotes remyelination in A. cantonensis-infected BALB/c mice. Int. J. Biol. Sci. 2019, 15, 2211–2223. [Google Scholar] [CrossRef] [PubMed]
- Geng, L.; Liu, W.; Chen, Y. Tanshinone IIA attenuates Aβ-induced neurotoxicity by down-regulating COX-2 expression and PGE2 synthesis via inactivation of NF-κB pathway in SH-SY5Y cells. J. Biol. Res-Thessalon. 2019, 26, 15. [Google Scholar] [CrossRef]
- Huang, X.; Li, N.; Pu, Y.; Zhang, T.; Wang, B. Neuroprotective effects of ginseng phytochemicals: Recent perspectives. Molecules 2019, 24, 2939. [Google Scholar] [CrossRef]
- Wu, C.F.; Bi, X.L.; Yang, J.Y.; Zhan, J.Y.; Dong, Y.X.; Wang, J.H.; Wang, J.M.; Zhang, R.; Li, X. Differential effects of ginsenosides on NO and TNF-α production by LPS-activated N9 microglia. Int. Immunopharmacol. 2007, 7, 313–320. [Google Scholar] [CrossRef]
- Liu, Z.; Qi, Y.; Cheng, Z.; Zhu, X.; Fan, C.; Yu, S.Y. The effects of ginsenoside Rg1 on chronic stress induced depression-like behaviors, BDNF expression and the phosphorylation of PKA and CREB in rats. Neuroscience 2016, 322, 358–369. [Google Scholar] [CrossRef]
- Zhou, T.; Zu, G.; Zhang, X.; Wang, X.; Li, S.; Gong, X.; Liang, Z.; Zhao, J. Neuroprotective effects of ginsenoside Rg1 through the Wnt/beta-catenin signaling pathway in both in vivo and in vitro models of Parkinson’s disease. Neuropharmacology 2016, 101, 480–489. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Efficacy and safety of ginsenoside-Rd for acute ischemic stroke. Available online: https://clinicaltrials.gov/ct2/show/NCT00815763 (accessed on 10 August 2020).
- Liu, X.; Wang, L.; Wen, A.; Yang, J.; Yan, Y.; Song, Y.; Liu, X.; Ren, H.; Wu, Y.; Li, Z.; et al. Ginsenoside-Rd improves outcome of acute ischemic stroke—A randomized, double-blind, placebo-controlled, multicenter trial. Eur. J. Neurol. 2012, 19, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.; Liu, L.; Wu, Z.; Cao, M. Combating neurodegenerative diseases with the plant alkaloid berberine: Molecular mechanisms and therapeutic potential. Curr. Neuropharmacol. 2019, 17, 563–579. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.Y.; Chen, C.S.; Wu, S.N.; Jong, Y.J.; Lo, Y.C. Berberine activates Nrf2 nuclear translocation and protects against oxidative damage via a phosphatidylinositol 3-kinase/Akt-dependent mechanism in NSC34 motor neuron-like cells. Eur. J. Pharm. Sci. 2012, 46, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Jia, L.; Liu, J.; Song, Z.; Pan, X.; Chen, L.; Cui, X.; Wang, M. Berberine suppresses amyloid-beta-induced inflammatory response in microglia by inhibiting nuclear factor-kappaB and mitogen-activated protein kinase signalling pathways. J. Pharm. Pharmacol. 2012, 64, 1510–1521. [Google Scholar] [CrossRef]
- Yang, W.; Chen, Y.H.; Liu, H.; Qu, H.D. Neuroprotective effects of piperine on the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease mouse model. Int. J. Mol. Med. 2015, 36, 1369–1376. [Google Scholar] [CrossRef]
- Shrivastava, P.; Vaibhav, K.; Tabassum, R.; Khan, A.; Ishrat, T.; Khan, M.M.; Ahmad, A.; Islam, F.; Safhi, M.M.; Islam, F. Anti-apoptotic and anti-inflammatory effect of piperine on 6-OHDA induced Parkinson’s rat model. J. Nutr. Biochem. 2013, 24, 680–687. [Google Scholar] [CrossRef]
- Wang-Sheng, C.; Jie, A.; Jian-Jun, L.; Lan, H.; Zeng-Bao, X.; Chang-Qing, L. Piperine attenuates lipopolysaccharide (LPS)-induced inflammatory responses in BV2 microglia. Int. Immunopharmacol. 2017, 42, 44–48. [Google Scholar] [CrossRef]
- Yu, Z.; Jin, W.; Cui, Y.; Ao, M.; Liu, H.; Xu, H.; Yu, L. Protective effects of macamides from Lepidium meyenii Walp. Against corticosterone-induced neurotoxicity in PC12 cells. RSC Adv. 2019, 9, 23096. [Google Scholar] [CrossRef]
- Yang, Q.; Jin, W.; Lv, X.; Dai, P.; Ao, Y.; Wu, M.; Deng, W.; Yu, L. Effects of macamides on endurance capacity and anti-fatigue property in prolonged swimming mice. Pharm. Biol. 2016, 54, 827–834. [Google Scholar] [CrossRef]
- Gugnani, K.S.; Vu, N.; Rondón-Ortiz, A.N.; Böhlke, M.; Maher, T.J.; Pino-Figueroa, A.J. Neuroprotective activity of macamides on manganese-induced mitochondrial disruption in U-87 MG glioblastoma cells. Toxicol. Appl. Pharm. 2018, 340, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Colín-González, A.L.; Ali, S.F.; Túnez, I.; Santamaría, A. On the antioxidant, neuroprotective and anti-inflammatory properties of S-allyl cysteine: An update. Neurochem. Int. 2015, 89, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Ashafaq, M.; Khan, M.M.; Raza, S.S.; Ahmad, A.; Khuwaja, G.; Javed, H.; Khan, A.; Islam, F.; Siddiqui, M.S.; Safhi, M.M.; et al. S-allyl cysteine mitigates oxidative damage and improves neurologic deficit in a rat model of focal cerebral ischemia. Nutr. Res. 2012, 32, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Zarezadeh, M.; Baluchnejadmojarad, T.; Kiasalari, Z.; Afshin-Majd, S.; Roghani, M. Garlic active constituent s-allyl cysteine protects against lipopolysaccharide-induced cognitive deficits in the rats: Possible involved mechanisms. Eur. J. Pharmacol. 2017, 795, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Šalamon, Š.; Kramar, B.; Marolt, T.P.; Poljšak, B.; Milisav, I. Medical and dietary use of N-acetylcysteine. Antioxidants 2019, 8, 111. [Google Scholar] [CrossRef]
- Sandhir, R.; Sood, A.; Mehrotra, A.; Kamboj, S.S. N-acetylcysteine reverses mitochondrial dysfunctions and behavioral abnormalities in 3-nitropropionic acid-induced Huntington’s disease. Neurodegener. Dis. 2012, 9, 145–157. [Google Scholar] [CrossRef]
- Zhou, J.; Terluk, M.R.; Basso, L.; Mishra, U.R.; Orchard, P.J.; Cloyd, J.C.; Schröder, H.; Kartha, R.V. N-acetylcysteine provides cytoprotection in murine oligodendrocytes through heme oxygenase-1 activity. Biomedicines 2020, 8, 240. [Google Scholar] [CrossRef]
- Sabetghadam, M.; Mazdeh, M.; Abolfathi, P.; Mohammadi, Y.; Mehrpooya, M. Evidence for a beneficial effect of oral N-acetylcysteine on functional outcomes and inflammatory biomarkers in patients with acute ischemic stroke. Neuropsychiatr. Dis. Treat. 2020, 16, 1265–1278. [Google Scholar] [CrossRef]
- Lima, L.A.R.; Lopes, M.J.P.; Costa, R.O.; Lima, F.A.V.; Neves, K.R.T.; Calou, I.B.F.; Andrade, G.M.; Viana, G.S.B. Vitamin D protects dopaminergic neurons against neuroinflammation and oxidative stress in hemiParkinsonian rats. J. Neuroinflamm. 2018, 15, 249. [Google Scholar] [CrossRef]
- Calvello, R.; Cianciulli, A.; Nicolardi, G.; De Nuccio, F.; Giannotti, L.; Salvatore, R.; Porro, C.; Trotta, T.; Panaro, M.A.; Lofrumento, D.D. Vitamin D treatment attenuates neuroinflammation and dopaminergic neurodegeneration in an animal model of Parkinson’s disease, shifting M1 to M2 microglia responses. J. Neuroimmune Pharmacol. 2017, 12, 327–339. [Google Scholar] [CrossRef]
- Hashemi, R.; Hosseini-Asl, S.S.; Arefhosseini, S.R.; Morshedi, M. The impact of vitamin D3 intake on inflammatory markers in multiple sclerosis patients and their first-degree relatives. PLoS ONE 2020, 15, e0231145. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, L.R.C.; Mimura, L.A.N.; Fraga-Silva, T.F.dC.; Ishikawa, L.L.W.; Fernandes, A.A.H.; Zorzella-Pezavento, S.F.G.; Sartori, A. Calcitriol prevents neuroinflammation and reduces blood-brain barrier disruption and local macrophage/microglia activation. Front. Pharmacol. 2020, 11, 161. [Google Scholar] [CrossRef] [PubMed]
- Rodick, T.C.; Seibels, D.R.; Babu, R.; Huggins, K.W.; Ren, G.; Mathews, S.T. Potential role of coenzyme q10 in health and disease conditions. Nutr. Diet. Suppl. 2018, 10, 1. [Google Scholar] [CrossRef]
- Yousef, A.O.S.; Fahad, A.A.; Moneim, A.E.A.; Metwally, D.M.; El-Khadragy, M.F.; Kassab, R.B. The neuroprotective role of coenzyme Q10 against lead acetate-induced neurotoxicity is mediated by antioxidant, anti-inflammatory and anti-apoptotic activities. Int. J. Environ. Res. Public Health 2019, 16, 2895. [Google Scholar] [CrossRef]
- Moccia, M.; Capacchione, A.; Lanzillo, R.; Carbone, F.; Micillo, T.; Perna, F.; De Rosa, A.; Carotenuto, A.; Albero, R.; Matarese, G.; et al. Coenzyme Q10 supplementation reduces peripheral oxidative stress and inflammation in interferon-β1a-treated multiple sclerosis. Ther. Adv. Neurol. Disord. 2019, 12, 1–12. [Google Scholar] [CrossRef]
- Park, H.W.; Park, C.G.; Park, M.; Lee, S.H.; Park, H.R.; Lim, J.; Paek, S.H.; Choy, Y.B. Intrastriatal administration of coenzyme Q10 enhances neuroprotection in a Parkinson’s disease rat model. Sci. Rep. 2020, 10, 9572. [Google Scholar] [CrossRef]
- Layé, S.; Nadjar, A.; Joffre, C.; Bazinet, R.P. Anti-inflammatory effects of omega-3 fatty acids in the brain: Physiological mechanisms and relevance to pharmacology. Pharmacol. Rev. 2018, 70, 12–38. [Google Scholar] [CrossRef]
- Chen, X.; Wu, S.; Chen, C.; Xie, B.; Fang, Z.; Hu, W.; Chen, J.; Fu, H.; He, H. Omega-3 polyunsaturated fatty acid supplementation attenuates microglial-induced inflammation by inhibiting the HMGB1/TLR4/NF-κB pathway following experimental traumatic brain injury. J. Neuroinflamm. 2017, 14, 143. [Google Scholar] [CrossRef]
- Peng, Z.; Zhang, C.; Yan, L.; Zhang, Y.; Yang, Z.; Wang, J.; Song, C. EPA is more effective than DHA to improve depression-like behavior, glia cell dysfunction and hippocampal apoptosis signaling in a chronic stress-induced rat model of depression. Int. J. Mol. Sci. 2020, 21, 1769. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Yeo, J.E.; Kang, S.K. Selenium effectively inhibits ROS-mediated apoptotic neural precursor cell death in vitro and in vivo in traumatic brain injury. Biochim. Biophys. Acta 2007, 1772, 1199–1210. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.L.; Kumari, S.; Mendelev, N.; Li, P.A. Selenium preserves mitochondrial function, stimulates mitochondrial biogenesis, and reduces infarct volume after focal cerebral ischemia. BMC Neurosci. 2012, 13, 79. [Google Scholar] [CrossRef] [PubMed]
- Demirci, K.; Nazıroğlu, M.; Övey, İ.S.; Balaban, H. Selenium attenuates apoptosis, inflammation and oxidative stress in the blood and brain of aged rats with scopolamine-induced dementia. Metab. Brain Dis. 2017, 32, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Lu, H.; Meng, X.; Ryu, J.H.; Hara, Y.; Yang, C.S. Stability, cellular uptake, biotransformation, and efflux of tea polyphenol (-)-epigallocatechin-3-gallate in HT-29 human colon adenocarcinoma. Cancer Res. 2002, 62, 7241–7246. [Google Scholar] [PubMed]
- Caritá, A.C.; Fonseca-Santos, B.; Shultz, J.D.; Michniak-Kohn, B.; Chorilli, M.; Leonardi, G.R. Vitamin C: One compound, several uses. Advances for delivery, efficiency and stability. Nanomed. NBM 2020, 24, 102117. [Google Scholar] [CrossRef] [PubMed]
- Byeon, J.C.; Ahn, J.B.; Jang, W.S.; Lee, S.E.; Choi, J.S.; Park, J.S. Recent formulation approaches to oral delivery of herbal medicines. J. Pharm. Investig. 2019, 49, 17–26. [Google Scholar] [CrossRef]
- Patil, S.C.; Tagalpallewar, A.A.; Kokare, C.R. Natural anti-proliferative agent loaded self-microemulsifying nanoparticles for potential therapy in oral squamous carcinoma. J. Pharm. Investig. 2019, 49, 527–541. [Google Scholar] [CrossRef]
- Xiong, J.; Guo, J.; Huang, L.; Meng, B.; Ping, Q. The use of lipid-based formulations to increase the oral bioavailability of Panax notoginseng saponins following a single oral gavage to rats. Drug Dev. Ind. Pharm. 2008, 34, 65–72. [Google Scholar] [CrossRef]
- Xu, H.; Kulkarni, K.H.; Singh, R.; Yang, Z.; Wang, S.W.; Tam, V.H.; Hu, M. Disposition of naringenin via glucuronidation pathway is affected by compensating efflux transporters of hydrophilic glucuronides. Mol. Pharm. 2009, 6, 1703–1715. [Google Scholar] [CrossRef]
- Lin, C.H.; Chen, C.H.; Lin, Z.C.; Fang, J.Y. Recent advances in oral delivery of drugs and bioactive natural products using solid lipid nanoparticles as the carriers. J. Food Drug Anal. 2017, 25, 219–234. [Google Scholar] [CrossRef]
- Cai, Z.Y.; Li, X.M.; Liang, J.P.; Xiang, L.P.; Wang, K.R.; Shi, Y.L.; Yang, R.; Shi, M.; Ye, J.H.; Lu, J.L.; et al. Bioavailability of tea catechins and its improvement. Molecules 2018, 23, 2346. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Chen, O.; Ray, S.D.; Ji, J.; Bucci, L.R.; Preuss, H.G. Highly bioavailable forms of curcumin and promising avenues for curcumin-based research and application: A review. Molecules 2020, 25, 1397. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Chokriwal, A.; Sharma, M.M.; Jain, D.; Saxena, J.; Stephen, B.J. Therapeutic role and drug delivery potential of neuroinflammation as a target in neurodegenerative disorders. ACS Chem. Neurosci. 2017, 8, 1645–1655. [Google Scholar] [CrossRef] [PubMed]
- Poupot, R.; Bergozza, D.; Fruchon, S. Nanoparticle-based strategies to treat neuro-inflammation. Materials 2018, 11, 270. [Google Scholar] [CrossRef] [PubMed]
- Bruinsmann, F.A.; Vaz, G.R.; Alves, A.dC.S.; Aguirre, T.; Pohlmann, A.R.; Guterres, S.S.; Sonvico, F. Nasal drug delivery of anticancer drugs for the treatment of glioblastoma: Preclinical and clinical trials. Molecules 2019, 24, 4312. [Google Scholar] [CrossRef] [PubMed]
- Bicker, J.; Fortuna, A.; Alves, G.; Falcão, A. Nose-to-brain delivery of natural compounds for the treatment of central nervous system disorders. Curr. Pharm. Des. 2020, 26, 594–619. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.S.; Oh, K.T.; Choi, H.G.; Lim, S.J. Liposomal formulations for nose-to-brain delivery: Recent advances and future perspectives. Pharmaceutics 2019, 11, 540. [Google Scholar] [CrossRef]
- Ovais, M.; Zia, N.; Ahmad, I.; Khalil, A.T.; Raza, A.; Ayaz, M.; Sadiq, A.; Ullah, F.; Shinwari, Z.K. Phyto-therapeutic and nanomedicinal approaches to cure Alzheimer’s disease: Present status and future opportunities. Front. Aging Neurosci. 2018, 10, 284. [Google Scholar] [CrossRef]
- Zeb, A.; Cha, J.H.; Noh, A.R.; Qureshi, O.S.; Kim, K.W.; Choe, Y.H.; Shin, D.; Shah, F.A.; Majid, A.; Bae, O.N.; et al. Neuroprotective effects of carnosine-loaded elastic liposomes in cerebral ischemia rat model. J. Pharm. Investig. 2020, 50, 373–381. [Google Scholar] [CrossRef]
- Gendelman, H.E.; Anantharam, V.; Bronich, T.; Ghaisas, S.; Jin, H.; Kanthasamy, A.G.; Liu, X.; McMillan, J.; Mosley, R.L.; Narasimhan, B.; et al. Nanoneuromedicines for degenerative, inflammatory, and infectious nervous system diseases. Nanomed. NBM 2015, 11, 751–767. [Google Scholar] [CrossRef]
- Watkins, R.; Wu, L.; Zhang, C.; Davis, R.M.; Xu, B. Natural products-based nanomedicine: Recent advances and issues. Int. J. Nanomed. 2015, 10, 6055–6074. [Google Scholar]
- Cho, H.J. Recent progresses in the development of hyaluronic acid-based nanosystems for tumor-targeted drug delivery and cancer imaging. J. Pharm. Investig. 2020, 50, 115–129. [Google Scholar] [CrossRef]
- Alkaff, S.A.; Radhakrishnan, K.; Nedumaran, A.M.; Liao, P.; Czarny, B. Nanocarriers for stroke therapy: Advances and obstacles in translating animal studies. Int. J. Nanomed. 2020, 15, 445–464. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, Y.; Li, S. Polymeric micelles: Nanocarriers for cancer-targeted drug delivery. AAPS PharmSciTech. 2014, 15, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Avramović, N.; Mandić, B.; Savić-Radojević, A.; Simić, T. Polymeric nanocarriers of drug delivery systems in cancer therapy. Pharmaceutics 2020, 12, 298. [Google Scholar] [CrossRef]
- Sikorska, M.; Lanthier, P.; Miller, H.; Beyers, M.; Sodja, C.; Zurakowski, B.; Gangaraju, S.; Pandey, S.; Sandhu, J.K. Nanomicellar formulation of coenzyme Q10 (Ubisol-Q10) effectively blocks ongoing neurodegeneration in the mouse 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model: Potential use as an adjuvant treatment in Parkinson’s disease. Neurobiol. Aging 2014, 35, 2329–2346. [Google Scholar] [CrossRef]
- Calzoni, E.; Cesaretti, A.; Polchi, A.; Michele, A.D.; Tancini, B.; Emiliani, C. Biocompatible polymer nanoparticles for drug delivery applications in cancer and neurodegenerative disorder therapies. J. Funct. Biomater. 2019, 10, 4. [Google Scholar] [CrossRef]
- Elmowafy, E.M.; Tiboni, M.; Soliman, M.E. Biocompatibility, biodegradation and biomedical applications of poly(lactic acid)/poly(lactic-co-glycolic acid) micro and nanoparticles. J. Pharm. Investig. 2019, 49, 347–380. [Google Scholar] [CrossRef]
- Liu, X.; Ye, M.; An, C.; Pan, L.; Ji, L. The effect of cationic albumin-conjugated PEGylated tanshinone IIA nanoparticles on neuronal signal pathways and neuroprotection in cerebral ischemia. Biomaterials 2013, 34, 6893–6905. [Google Scholar] [CrossRef]
- Aalinkeel, R.; Kutscher, H.L.; Singh, A.; Cwiklinski, K.; Khechen, N.; Schwartz, S.A.; Prasad, P.N.; Mahajan, S.D. Neuroprotective effects of a biodegradable poly(lactic-co-glycolic acid)-ginsenoside Rg3 nanoformulation: A potential nanotherapy for Alzheimer’s disease? J. Drug Target. 2018, 26, 182–193. [Google Scholar] [CrossRef]
- Shi, X.X.; Miao, W.M.; Pang, D.W.; Wu, J.S.; Tong, Q.S.; Li, J.X.; Luo, J.Q.; Li, W.Y.; Du, J.Z.; Wang, J. Angiopep-2 conjugated nanoparticles loaded with doxorubicin for the treatment of primary central nervous system lymphoma. Biomater. Sci. 2020, 8, 1290–1297. [Google Scholar] [CrossRef] [PubMed]
- Sonvico, F.; Clementino, A.; Buttini, F.; Colombo, G.; Pescina, S.; Guterres, S.S.; Pohlmann, A.R.; Nicoli, S. Surface-modified nanocarriers for nose-to-brain delivery: From bioadhesion to targeting. Pharmaceutics 2018, 10, 34. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Yan, Z.; Hu, K.; Pang, Z.; Cheng, X.; Guo, L.; Zhang, Q.; Jiang, X.; Fang, L.; Lai, R. Odorranalectin-conjugated nanoparticles: Preparation, brain delivery and pharmacodynamic study on Parkinson’s disease following intranasal administration. J. Control. Release 2011, 151, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Su, J.; Kamal, Z.; Guo, P.; Wu, X.; Lu, L.; Wu, H.; Qiu, M. Odorranalectin modified PEG-PLGA/PEG/PBLG curcumin-loaded nanoparticle for intranasal administration. Drug Dev. Ind. Pharm. 2020, 46, 899–909. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, S.; Ashrafizadeh, M.; Zarrabi, A.; Roghanian, R.; Afshar, E.G.; Pardakhty, A.; Mohammadinejad, R.; Kumar, A.; Thakur, V.K. Multifunctional polymeric nanoplatforms for brain diseases diagnosis, therapy and theranostics. Biomedicines 2020, 8, 13. [Google Scholar] [CrossRef]
- Peptu, C.A.; Ochiuz, L.; Alupei, L.; Peptu, C.; Popa, M. Carbohydrate based nanoparticles for drug delivery across biological barriers. J. Biomed. Nanotechnol. 2014, 10, 2107–2148. [Google Scholar] [CrossRef]
- Kang, B.; Opatz, T.; Landfester, K.; Wurm, F.R. Carbohydrate nanocarriers in biomedical applications: Functionalization and construction. Chem. Soc. Rev. 2015, 44, 8301–8325. [Google Scholar] [CrossRef]
- Aderibigbe, B.A.; Naki, T. Chitosan-based nanocarriers for nose to brain delivery. Appl. Sci. 2019, 9, 2219. [Google Scholar] [CrossRef]
- Nagpal, K.; Singh, S.K.; Mishra, D.N. Chitosan nanoparticles: A promising system in novel drug delivery. Chem. Pharm. Bull. 2010, 58, 1423–1430. [Google Scholar] [CrossRef]
- Mistry, A.; Stolnik, S.; Illum, L. Nanoparticles for direct nose-to-brain delivery of drugs. Int. J. Pharm. 2009, 379, 146–157. [Google Scholar] [CrossRef]
- Ngo, D.H.; Kim, S.K. Antioxidant effects of chitin, chitosan, and their derivatives. Adv. Food Nutr. Res. 2014, 73, 15–31. [Google Scholar] [PubMed]
- Elnaggar, Y.S.R.; Etman, S.M.; Abdelmonsif, D.A.; Abdallah, O.Y. Intranasal piperine-loaded chitosan nanoparticles as brain-targeted therapy in Alzheimer’s disease: Optimization, biological efficacy, and potential toxicity. J. Pharm. Sci. 2015, 104, 3544–3556. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Wang, A.; Hua, H.; Jiang, Y.; Wang, Y.; Mu, H.; Wu, Z.; Sun, K. Intranasal delivery of huperzine A to the brain using lactoferrin-conjugated N-trimethylated chitosan surface-modified PLGA nanoparticles for treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 705–718. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.D.; Xavier, M.; Leite, D.M.; Moreira, D.A.; Custódio, B.; Torrado, M.; Castro, R.; Leiro, V.; Rodrigues, J.; Tomás, H.; et al. PAMAM dendrimers: Blood-brain barrier transport and neuronal uptake after focal brain ischemia. J. Control. Release 2018, 291, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Abedi-Gaballu, F.; Dehghan, G.; Ghaffari, M.; Yekta, R.; Abbaspour-Ravasjani, S.; Baradaran, B.; Dolatabadi, J.E.N.; Hamblin, M.R. PAMAM dendrimers as efficient drug and gene delivery nanosystems for cancer therapy. Appl. Mater. Today 2018, 12, 177–190. [Google Scholar] [CrossRef]
- Srinageshwar, B.; Dils, A.; Sturgis, J.; Wedster, A.; Kathirvelu, B.; Baiyasi, S.; Swanson, D.; Sharma, A.; Dunbar, G.L.; Rossignol, J. Surface-modified G4 PAMAM dendrimers cross the blood-brain barrier following multiple tail-vein injections in C57BL/6J mice. ACS Chem. Neurosci. 2019, 10, 4145–4150. [Google Scholar] [CrossRef]
- Nance, E.; Zhang, F.; Mishra, M.K.; Zhang, Z.; Kambhampati, S.P.; Kannan, R.M.; Kannan, S. Nanoscale effects in dendremer-mediated targeting of neuroinflammation. Biomaterials 2016, 101, 96–107. [Google Scholar] [CrossRef]
- Wang, B.; Navath, R.S.; Romero, R.; Kannan, S.; Kannan, R. Anti-inflammatory and anti-oxidant activity of anionic dendrimer-N-acetyl cysteine conjugates in activated microglial cells. Int. J. Pharm. 2009, 377, 159–168. [Google Scholar] [CrossRef]
- García-Pinel, B.; Porras-Alcalá, C.; Ortega-Rodríguez, A.; Sarabia, F.; Pardos, J.; Melguizo, C.; López-Romero, J.M. Lipid-based nanoparticles: Application and recent advances in cancer treatment. Nanomaterials 2019, 9, 638. [Google Scholar] [CrossRef]
- Subramaniam, B.; Siddik, Z.H.; Nagoor, N.H. Optimization of nanostructured lipid carriers: Understanding the types, designs, and parameters in the process of formulations. J. Nanoparticle Res. 2020, 22, 141. [Google Scholar] [CrossRef]
- Tapeinos, C.; Battaglini, M.; Ciofani, G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Control. Release 2017, 264, 306–332. [Google Scholar] [CrossRef] [PubMed]
- Gastaldi, L.; Battaglia, L.; Peira, E.; Chirio, D.; Muntoni, E.; Solazzi, I.; Gallarate, M.; Dosio, F. Solid lipid nanoparticles as vehicles of drugs to the brain: Current state of the art. Eur. J. Pharm. Biopharm. 2014, 87, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Magro, R.D.; Ornaghi, F.; Cambianica, I.; Beretta, S.; Re, F.; Musicanti, C.; Rigolio, R.; Donzelli, E.; Canta, A.; Ballarini, E.; et al. ApoE-modified solid lipid nanoparticles: A feasible strategy to cross the blood-brain barrier. J. Control. Release 2017, 249, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Graverini, G.; Piazzini, V.; Landucci, E.; Pantano, D.; Nardiello, P.; Casamenti, F.; Pellegrini-Giampietro, D.E.; Bilia, A.R.; Bergonzi, M.C. Solid lipid nanoparticles for delivery of andrographolide across the blood-brain barrier: In vitro and in vivo evaluation. Colloids Surf B. Biointerfaces 2018, 161, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Yasir, M.; Sara, U.V.S. Solid lipid nanoparticles for nose to brain delivery of haloperidol: In vitro drug release and pharmacokinetics evaluation. Acta Pharm. Sin. B 2014, 4, 454–463. [Google Scholar] [CrossRef]
- Bhatt, P.C.; Srivastava, P.; Pandey, P.; Khan, W.; Panda, B.P. Nose to brain delivery of astaxanthin-loaded solid lipid nanoparticles: Fabrication, radio labeling, optimization and biological studies. RSC Adv. 2016, 6, 10001. [Google Scholar] [CrossRef]
- Meng, F.; Asghar, S.; Gao, S.; Su, Z.; Song, J.; Huo, M.; Meng, W.; Ping, Q.; Xiao, Y. A novel LDL-mimic nanocarrier for the targeted delivery of curcumin into the brain to treat Alzheimer’s disease. Colloids Surf. B Biointerfaces 2015, 134, 88–97. [Google Scholar] [CrossRef]
- Lai, F.; Fadda, A.M.; Sinico, C. Liposomes for brain delivery. Expert Opin. Drug Deliv. 2013, 10, 1003–1022. [Google Scholar] [CrossRef]
- Vieira, D.B.; Gamarra, L.F. Getting into the brain: Liposome-based strategies for effective drug delivery across the blood-brain barrier. Int. J. Nanomed. 2016, 11, 5381–5414. [Google Scholar] [CrossRef]
- Wnag, Z.Y.; Sreenivasmurthy, S.G.; Song, J.X.; Liu, J.Y.; Li, M. Strategies for brain-targeting liposomal delivery of small hydrophobic molecules in the treatment of neurodegenerative diseases. Drug Discov. Today 2019, 24, 595–605. [Google Scholar] [CrossRef]
- Piazzini, V.; Landucci, E.; Graverini, G.; Pellegrini-Giampietro, D.E.; Bilia, A.R.; Bergonzi, M.C. Stealth and cationic nanoliposomes as drug delivery systems to increase andrographolide BBB permeability. Pharmaceutics 2018, 10, 128. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, M.; Ajazuddin; Tripathi, D.K.; Saraf, S.; Saraf, S.; Antimisiaris, S.G.; Mourtas, S.; Hammarlund-Udenaes, M.; Alexander, A. Recent advancements in liposomes targeting strategies to cross blood-brain barrier (BBB) for the treatment of Alzheimer’s disease. J. Control. Release 2017, 260, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Riaz, M.K.; Riaz, M.A.; Zhang, X.; Lin, C.; Wong, K.H.; Chen, X.; Zhang, G.; Lu, A.; Yang, Z. Surface functionalization and targeting strategies of liposomes in solid tumor therapy: A review. Int. J. Mol. Sci. 2018, 19, 195. [Google Scholar] [CrossRef] [PubMed]
- Papadia, K.; Giannou, A.D.; Markoutsa, E.; Bigot, C.; Vanhoute, G.; Mourtas, S.; Van der Linded, A.; Stathopoulos, G.T.; Antimisiaris, S.G. Multifunctional LUV liposomes decorated for BBB and amyloid targeting—B. In vivo brain targeting potential in wild-type and APP/PS1 mice. Eur. J. Pharm. Sci. 2017, 102, 180–187. [Google Scholar] [PubMed]
- Liu, C.; Liu, X.N.; Wang, G.L.; Hei, Y.; Meng, S.; Yang, L.F.; Yuan, L.; Xie, Y. A dual-mediated liposomal drug delivery system targeting the brain: Rational construction, integrity evaluation across the blood-brain barrier, and the transporting mechanism to glioma cells. Int. J. Nanomed. 2017, 12, 2407–2425. [Google Scholar] [CrossRef] [PubMed]
- Maiyo, F.; Singh, M. Selenium nanoparticles: Potential in cancer gene and drug delivery. Nanomedicine 2017, 12, 1075–1089. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Peng, Q.; Baron, M.; Melcova, M.; Opatrilova, R.; Zidkova, J.; et al. Nano-selenium and its nanomedicine applications: A critical review. Int. J. Nanomed. 2018, 13, 2107–2128. [Google Scholar] [CrossRef]
- Khurana, A.; Tekula, S.; Saifi, M.A.; Venkatesh, P.; Godugu, C. Therapeutic applications of selenium nanoparticles. Biomed. Pharmacother. 2019, 111, 802–812. [Google Scholar] [CrossRef]
- Amani, H.; Habibey, R.; Shokri, R.; Hajmiresmail, S.J.; Akhavan, O.; Mashaghi, A.; Pazoki-Toroudi, H. Selenium nanoparticles for targeted stroke therapy through modulation of inflammatory and metabolic signaling. Sci. Rep. 2019, 9, 6044. [Google Scholar] [CrossRef]
- Yin, T.; Yang, L.; Liu, Y.; Zhou, X.; Sun, J.; Liu, J. Sialic acid (SA)-modified selenium nanoparticles coated with a high blood-brain barrier permeability peptide-B6 peptide for potential use in Alzheimer’s disease. Acta Biomater. 2015, 25, 172–183. [Google Scholar] [CrossRef]
- Sela, H.; Cohen, H.; Elia, P.; Zach, R.; Karpas, Z.; Zeiri, Y. Spontaneous penetration of gold nanoparticles through the blood brain barrier (BBB). J. Nanobiotechnol. 2015, 13, 71. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.Y.; Zhang, J.W.; Li, R.F.; Wang, Z.X.; Wang, W.J.; Wang, W. Unique roles of gold nanoparticles in drug delivery, targeting and imaging applications. Molecules 2017, 22, 1445. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Cao, Z.; Liu, R.; Liu, L.; Li, H.; Li, X.; Chen, Y.; Lu, C.; Liu, Y. AuNPs as an important inorganic nanoparticle applied in drug carrier systems. Artif. Cells Nanomed. Biotechnol. 2019, 47, 4222–4233. [Google Scholar] [CrossRef] [PubMed]
- Khongkow, M.; Yata, T.; Boonrungsiman, S.; Ruktanonchai, U.R.; Graham, D.; Namdee, K. Surface modification of gold nanoparticles with neuron-targeted exosome for enhanced blood-brain barrier penetration. Sci. Rep. 2019, 9, 8278. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Rehman, S.U.; Amin, F.U.; Kim, M.O. Enhanced neuroprotection of anthocyanin-loaded PEG-gold nanoparticles against Aβ1-42-induced neuroinflammation and neurodegeneration via the NF-κB/JNK/GSK3β signaling pathway. Nanomed. NBM 2017, 13, 2533–2544. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Chen, A.; Zhang, Y.; Wang, J.; Shao, L.; Wei, L. Central nervous system toxicity of metallic nanoparticles. Int. J. Nanomed. 2015, 10, 4321–4340. [Google Scholar]
- El-Boubbou, K. Magnetic iron oxide nanoparticles as drug carriers: Preparation, conjugation and delivery. Nanomedicine 2018, 13, 929–952. [Google Scholar] [CrossRef]
- Israel, L.L.; Galstyan, A.; Holler, E.; Ljubimova, J.Y. Magnetic iron oxide nanoparticles for imaging, targeting and treatment of primary and metastatic tumors of the brain. J. Control. Release 2020, 320, 45–62. [Google Scholar] [CrossRef]
- Amin, F.U.; Hoshiar, A.K.; Do, T.D.; Noh, Y.; Shah, S.A.; Khan, M.S.; Yoon, J.; Kim, M.O. Osmotin-loaded magnetic nanoparticles with electromagnetic guidance for the treatment of Alzheimer’s disease. Nanoscale 2017, 9, 10619–10632. [Google Scholar] [CrossRef]
- D’Agata, F.; Ruffinatti, F.A.; Boschi, S.; Stura, I.; Rainero, I.; Abollino, O.; Cavalli, R.; Guiot, C. Magnetic nanoparticles in the central nervous system: Targeting principles, applications and safety issues. Molecules 2018, 23, 9. [Google Scholar] [CrossRef]
- Aguilera, G.; Berry, C.C.; West, R.M.; Gonzalez-Monterrubio, E.; Angulo-Molina, A.; Arias-Carrión, Ó.; Méndez-Rojas, M.Á. Carboxymethyl cellulose coated magnetic nanoparticles transport across a human lung microvascular endothelial cell model of the blood-brain barrier. Nanoscale Adv. 2019, 1, 671–685. [Google Scholar] [CrossRef]
- Wang, J.T.W.; Al-Jamal, K.T. Functionalized carbon nanotubes: Revolution in brain delivery. Nanomedicine 2015, 10, 2639–2642. [Google Scholar] [CrossRef] [PubMed]
- Teleanu, D.M.; Chircov, C.; Grumezescu, A.M.; Volceanov, A.; Teleanu, R.I. Blood-brain delivery methods using nanotechnology. Pharmaceutics 2018, 10, 269. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.; Saito, N. Biocompatibility and carcinogenicity of carbon nanotubes as biomaterials. Nanomaterials 2020, 10, 264. [Google Scholar] [CrossRef] [PubMed]
- Kafa, H.; Wang, J.T.W.; Rubio, N.; Venner, K.; Anderson, G.; Pach, E.; Ballesteros, B.; Preston, J.E.; Abbott, N.J.; Al-Jamal, K.T. The interaction of carbon nanotubes with an in vitro blood-brain barrier model and mouse brain in vivo. Biomaterials 2015, 53, 437–452. [Google Scholar] [CrossRef] [PubMed]
- Lohan, S.; Raza, K.; Mehta, S.K.; Bhatti, G.K.; Saini, S.; Singh, B. Anti-Alzheimer’s potential of barberine using surface decorated multi-walled carbon nanotubes: A preclinical evidence. Int. J. Pharm. 2017, 530, 263–278. [Google Scholar] [CrossRef] [PubMed]
- Hoosain, F.G.; Choonara, Y.E.; Tomar, L.K.; Kumar, P.; Tyagi, C.; du Toit, L.C.; Pillay, V. Bypassing P-glycoprotein drug efflux mechanisms: Possible applications in pharmacoresistant schizophrenia therapy. Biomed. Res. Int. 2015, 2015, 484963. [Google Scholar] [CrossRef]
- An, F.F.; Zhang, X.H. Strategies for preparing albumin-based nanoparticles for multifunctional bioimaging and drug delivery. Theranostics 2017, 7, 3667–3689. [Google Scholar] [CrossRef]
- Bilia, A.R.; Nardiello, P.; Piazzini, V.; Leri, M.; Bergonzi, M.C.; Bucciantini, M.; Casamenti, F. Successful brain delivery of andrographolide loaded in human serum albumin nanoparticles to TgCRND8 mice, an Alzheimer’s disease mouse model. Front. Pharmacol. 2019, 10, 910. [Google Scholar] [CrossRef]
- Mohammad-Beigi, H.; Morshedi, D.; Shojaosadati, S.A.; Pedersen, J.N.; Marvian, A.T.; Aliakbari, F.; Christiansen, G.; Pedersen, J.S.; Otzen, D.E. Gallic acid loaded onto polyethylenimine-coated human serum albumin nanoparticles (PEI-HSA-GA NPs) stabilizes α-synuclein in the unfolded conformation and inhibits aggregation. RSC Adv. 2016, 6, 85312. [Google Scholar] [CrossRef]
- Na, D.; Yang, N.; Nadithe, V. Exosomes as therapeutic drug carriers and delivery vesicles across biological membranes: Current perspectives and future challenges. Acta Pharm. Sin. B 2016, 6, 287–296. [Google Scholar]
- Bunggulawa, E.J.; Wang, W.; Yin, T.; Wang, N.; Durkan, C.; Wang, Y.; Wang, G. Recent advancements in the use of exosomes as drug delivery systems. J. Nanobiotechol. 2018, 16, 81. [Google Scholar] [CrossRef] [PubMed]
- Pullan, J.E.; Confeld, M.I.; Osborn, J.K.; Kim, J.; Sarkar, K.; Mallik, S. Exosomes as drug carriers for cancer therapy. Mol. Pharm. 2019, 16, 1789–1798. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Sui, H.; Zheng, Y.; Jiang, Y.; Shi, Y.; Liang, J.; Zhao, L. Curcumin-primed exosomes potently ameliorate cognitive function in AD mice by inhibiting hyperphosphorylation of the tau protein through the AKT/GSK-3β pathway. Nanoscale 2019, 11, 7481–7496. [Google Scholar] [CrossRef]
- Qi, Y.; Guo, L.; Jiang, Y.; Shi, Y.; Sui, H.; Zhao, L. Brain delivery of quercetin-loaded exosomes improved cognitive function in AD mice by inhibiting phosphorylated tau-mediated neurofibrillary tangles. Drug Deliv. 2020, 27, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, J.J.; Cunnane, E.M.; Ross, A.M.; Duskey, J.T.; Giovanni, T.; Grabrucker, A.M. Drug delivery across the blood-brain barrier: Recent advances in the use of nanocarriers. Nanomedicine 2020, 15, 205–214. [Google Scholar] [CrossRef]
- Li, Y.; An, C.; Han, D.; Dang, Y.; Liu, X.; Zhang, F.; Xu, Y.; Zhong, H.; Sun, X. Neutrophil affinity for PGP and HAIYPRH (T7) peptide dual-ligand functionalized nanoformulation enhances the brain delivery of tanshinone IIA and exerts neuroprotective effects against ischemic stroke by inhibiting pro-inflammatory signaling pathways. New J. Chem. 2018, 42, 19043–19061. [Google Scholar] [CrossRef]
- Dang, Y.; An, C.; Li, Y.; Han, D.; Liu, X.; Zhang, F.; Xu, Y.; Zhong, H.; Khan, M.K.K.; Zou, F.; et al. Neutrophil-mediated and low density lipoprotein receptor-mediated dual-targeting nanoformulation enhances brain accumulation of scutellarin and exerts neuroprotective effects against ischemic stroke. RSC Adv. 2019, 9, 1299. [Google Scholar] [CrossRef]
- Debnath, K.; Pradhan, N.; Singh, B.K.; Jana, N.R.; Jana, N.R. Poly(trehalose) nanoparticles prevent amyloid aggregation and suppress polyglutamine aggregation in a Huntington’s disease model mouse. ACS Appl. Mater. Interfaces 2017, 9, 24126–24139. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, X.; Yu, Q.; Yang, L.; Sun, D.; Zhou, Y.; Liu, J. Epigallocatechin-3-gallate (EGCG)-stabilized selenium nanoparticles coated with Tet-1 peptide to reduce amyloid-β aggregation and cytotoxicity. ACS Appl. Mater. Interfaces 2014, 6, 8475–8487. [Google Scholar] [CrossRef]
- Tian, T.; Zhang, H.X.; He, C.P.; Fan, S.; Zhu, Y.L.; Qi, C.; Huang, N.P.; Xiao, Z.D.; Lu, Z.H.; Tannous, B.A.; et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials 2018, 150, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wei, C.; Liu, Y.; Xie, W.; Xu, M.; Zhou, H.; Liu, J. Progressive release of mesoporous nano-selenium delivery system for the multi-channel synergistic treatment of Alzheimer’s disease. Biomaterials 2019, 197, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Dehaini, D.; Fang, R.H.; Zhang, L. Biomimetic strategies for targeted nanoparticle delivery. Bioeng. Transl. Med. 2016, 1, 30–46. [Google Scholar] [CrossRef]
- Wu, M.; Le, W.; Mei, T.; Wang, Y.; Chen, B.; Liu, Z.; Xue, C. Cell membrane camouflaged nanoparticles: A new biomimetic platform for cancer photothermal therapy. Int. J. Nanomed. 2019, 14, 4431–4448. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, J.; Chen, J.; Liu, Y.; Cheng, X.; Yang, F.; Gu, N. Platelet membrane biomimetic magnetic nanocarriers for targeted delivery and in situ generation of nitric oxide in early ischemic stroke. ACS Nano 2020, 14, 2024–2035. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.X.; Wei, C.X.; Lyu, Y.Q.; Chen, H.Z.; Jiang, G.; Gao, X.L. Biomimetic drug-delivery systems for the management of brain diseases. Biomater. Sci. 2020, 8, 1073–1088. [Google Scholar] [CrossRef]

| Type of Natural Compounds | Therapeutic Agents | Mechanisms of Action | Ref. |
|---|---|---|---|
| Flavonoids | Apigenin | Direct radical scavenging action ↑, SOD ↑, GPx ↑, MDA ↓, ROS ↓, Ca2+ signaling ↓, NMDA receptor ↓, PKC ↓, BDNF ↑ Pro-inflammatory mediators (NO, iNOS, COX-2, IL-1β, IL-6, TNFα, GFAP) ↓, TLR4/NF-κB pathway ↓, p38 MAPK ↓, SAPK/JNK pathway ↓ Neuronal cell apoptosis ↓, caspase-3 and -7 ↓, cytochrome c ↓, Aβ levels ↓, BACE-1 ↓ | [62,63,64] |
| EGCG | ROS ↓, NO ↓, Nrf-2/ARE pathway ↑ Microglial activation ↓, iNOS ↓, COX-2 ↓, pro-inflammatory cytokines ↓, NF-κB pathway ↓, Aβ levels ↓, plaques formation ↓ | [66,67] | |
| Quercetin | Neuroinflammation ↓, pro-inflammatory cytokines and proteins ↓, BACE-1 ↓, NF-κB ↓, αSN fibrillization ↓, Aβ ↓ Direct radical scavenging action ↑, ROS ↓, SOD ↑, AMPK ↑, Nrf-2/ARE ↑ | [70,71] | |
| Naringenin | NO ↓, PGE2 ↓, iNOS ↓, COX-2 ↓, Pro-inflammatory cytokines and chemokine ↓, NF-κB ↓, AMPK ↑, SOCS3 pathways ↑ Nrf-2/ARE pathway ↑, ROS ↓, SOD ↑, GSH ↑, HO-1 ↑ Neuronal cell apoptosis ↓, cleaved caspase-3 ↓, Bax ↓, Bcl-2 ↑ | [73,74,75] | |
| Genistein | ROS ↓, Nrf-2/HO-1 ↑, Inflammatory mediators (iNOS, COX, TNFα, and IL-1β) ↓, PPAR-γ ↑ Neuronal cell apoptosis ↓, cleaved caspase-3 ↓ Aβ ↓, plaque formation ↓, TLR4/NF-κB signaling pathway ↓ | [77,78,79] | |
| Anthocyanins | Direct radical scavenging action ↑, intrinsic anti-oxidant (GSH, SOD, and Coenzyme Q10) ↑, Nrf-2 pathway ↑ Intracellular Ca2+ ↓, mitochondrial excitotoxicity ↓, Inflammatory mediators (iNOS, COX-2, TNFα, and IL-1β) ↓ JNK phosphorylation ↓, MAPK pathway ↓, NF-κB pathway ↓, inflammasome pathway (NLRP3) ↓ Neuronal cell apoptosis ↓, caspase-3 activity ↓, Bax ↓, Bcl-2 ↑ | [81,82] | |
| Non-flavonoid polyphenols | Curcumin | Direct radical scavenging action ↑, anti-oxidant proteins (CAT, GPx, SOD, HO-1, and GST) ↑, Nrf-2 pathway ↑ Inflammatory mediators ↓, pro-inflammatory cytokines ↓, anti-inflammatory cytokines ↑, PPAR-γ ↑, SOCS pathway ↑, NF-κB pathway ↓, STAT3 pathway ↓, Iba-1 (microglial activation) ↓, GFAP (astrocytes activation) ↓ Aβ ↓, plaques formation ↓, tau hyperphosphorylation ↓ | [84,85,86] |
| Resveratrol | Direct radical scavenging action ↑, GPx ↑, HO-1 ↑, NO ↓, ROS ↓, AMPK ↑ Pro-inflammatory factors (COX-1, COX-2, TNFα, NO) ↓, NF-κB pathway ↓, MAPK pathway ↓, SIRT-1 ↑ Programmed cell death ↓, Bax ↓, MMP-9 ↓, Aβ fibrillation and production ↓ | [89,90,91] | |
| Lycopene | Direct radical scavenging action ↑, GPx ↑, GSH ↑, SOD ↑, HO-1 ↑, ROS ↓, NO ↓, Nrf-2 pathway ↑ Neuronal cell apoptosis ↓, caspase-3 ↓, Bax ↓, Bcl-2 ↑, Nucling (apoptosome complex) ↓, pro-inflammatory cytokines (TNFα, IL-1β, IL-6) ↓, iNOS ↓, NF-κB ↓, MAPK/JNK pathway ↓ BACE-1 ↓, Aβ ↓, tau phosphorylation ↓ | [95,96,97] | |
| Phenolic acids | Protocatechuic acid | Glutamate release ↓, direct radical scavenging action ↑, ROS ↓ Microglial activation ↓, pro-inflammatory mediators (NO, iNOS, COX-2, TNFα, IL-1β, IL-6, PGE2) ↓, BDNF ↑, SIRT-1 ↑, NF-κB pathway ↓, MAPK/JNK pathway ↓ Neuronal cell apoptosis ↓, cleaved caspase-3 ↓, p53 pathway ↓, Aβ fibrillation ↓, APP ↓ | [99,100] |
| Gallic acid | Direct radical scavenging action ↑, lipid peroxidation ↓, MDA ↓, SOD ↑, CAT ↑, GPx ↑, ROS ↓, Nrf-2 pathway ↑ CSPG ↓, GFAP ↓, ED-1 ↓, pro-inflammatory mediators (COX-2, NO, iNOS, IL-1β, TNFα) ↓, BDNF ↑, NF-κB pathway ↓ Bax ↓, Bcl-2 ↑, caspase-3 (apoptosis) ↓, RIPK-1 and RIPK-3 (necroptosis) ↓, Aβ and αSN aggregation ↓ | [102,103] | |
| Terpenoids | Terpenes in Ginkgo biloba extracts (Ginkgolides, Bilobalide) | MDA ↓, SOD ↑, GSH ↑, HO-1 ↑, ROS ↓, NO ↓, hippocampal Ca2+ ↓, Akt signaling ↑, Nrf-2 pathway ↑, BDNF ↑, BBB integrity ↑ Pro-inflammatory mediators (GFAP, MMP-9, iNOS, IL-1β, IL-6, TNFα) ↓, p38 MAPK ↓, TLR/NF-κB pathway ↓, PAF-signaling pathway ↓ Inflammatory M1 microglial cells ↓, anti-inflammatory M2 microglial cells ↑, NLRP3 inflammasome ↓, caspase-1 ↓ Neuronal cell apoptosis ↓, caspase-3,7,8,9 ↓, cytochrome c ↓, PARP ↓, Bax ↓, Bcl-2 ↑, PI3K/Akt pathway ↑, tau phosphorylation ↓ | [105,106,107] |
| Tanshinone IIA | Pro-inflammatory mediators (MMP-2, iNOS, PGE2, COX-2, IL-1β, IL-6, TNFα, MIF) ↓, NF-κB pathway ↓ MPO ↓, neutrophil infiltration ↓, M1 microglial genes ↓, M2 microglial genes ↑ Neuronal cell apoptosis ↓, Bcl-xL pathway ↑, Aβ levels ↓, BACE-1 ↓ | [108,109,110] | |
| Ginsenosides | Direct radical scavenging action ↑, HO-1 ↑, SOD ↑, GPx ↑, MDA ↓, ROS ↓, Nrf-2 pathway ↑ cAMP/PKA/CREB pathway ↑, HIF-1α/VEGF pathway ↑, NSCs proliferation and differentiation ↑, BDNF ↑, IGF-1 ↑ Preservation of mitochondrial potential, PAR-1 ↓, BBB integrity ↑, immune cells infiltration ↓ NR2B ↓, glutamate signaling pathway ↓, glutamate- induced Ca2+ ↓, Iba-1 ↓, Pro-inflammatory mediators (GFAP, NO, iNOS, COX-2, IL-1β, IL-6, TNFα) ↓, NF-κB pathway ↓, STAT1 pathway ↓, p38 MAPK ↓, p-JNK ↓, PPAR-γ ↑ Neuronal cell apoptosis ↓, caspase-1, -3, -9 ↓, Bax ↓, Bcl-2 ↑, NLRP1 inflammasome ↓, Wnt signaling pathway ↑, PI3K/Akt pathway ↑ Aβ aggregation ↓, tau hyperphosphorylation ↓, BACE-1 ↓, αSN fibrillization ↓ | [112,113,114] | |
| Alkaloids | Berberine | MDA ↓, ROS ↓, SOD ↑, GSH ↑, HO-1 ↑, NMDA/glutamate signaling pathway ↓, Nrf-2 pathway ↑ Caspase-3, -9 ↓, cytochrome c ↓, Bax ↓, Bcl-2 ↑, PI3K/Akt pathway ↑ NGF ↑, cAMP/PKA/CREB pathway ↑, BBB integrity ↑, cerebral blood flow ↑ Pro-inflammatory mediators (NO, iNOS, COX-2, PGE2, IL-1β, IL-6, MCP-1, TNFα, TNFR1) ↓, NF-κB pathway ↓, p38 MAPK ↓, MAPK/ERK1/2 pathway ↓, AMPK pathway↑ Aβ accumulation and production ↓, APP ↓, BACE-1 ↓, tau phosphorylation ↓, GSK3 ↓ | [118,119] |
| Piperine | Lipid peroxidation ↓, MDA ↓, ROS ↓, SOD ↑, GSH ↑, HO-1 ↑, Nrf-2 pathway ↑ NMDA/glutamate signaling pathway ↓, BDNF ↑ Pro-inflammatory mediators (iNOS, COX-2, PGE2, IL-1β, IL-6, TNFα) ↓, NF-κB pathway ↓ Neuronal cell apoptosis ↓, caspase-3, -9 ↓, cytochrome c ↓, Bax ↓, Bcl-2 ↑, PARP ↓ | [121,122] | |
| Macamides | FAAH inhibitors, AchE inhibitors MDA ↓, ROS ↓, SOD ↑, GSH ↑, GPx ↑, Preservation of mitochondrial potential, PPARγ ↑, BDNF ↑, cAMP/CREB pathway ↑ Neuronal cell apoptosis ↓, cleaved caspase-3 ↓, cytochrome c ↓, Bax ↓, Bcl-2 ↑, cleaved PARP ↓, PI3K/Akt pathway ↑ | [123,124,125] | |
| Other dietary compounds | S-allylcysteine | MDA ↓, ROS ↓, SOD ↑, CAT ↑, GSH ↑, HO-1 ↑, Nrf-2 pathway ↑ Pro-inflammatory mediators (GFAP, iNOS, IL-1β) ↓, TLR4/NF-κB pathway ↓, PPARγ ↑, Iba-1 ↓ Neuronal cell apoptosis ↓, NLRP1 and 3 inflammasome ↓ | [127,128] |
| N-acetyl cysteine | MDA ↓, ROS ↓, SOD ↑, GSH ↑, GPx ↑, HO-1 ↑, NMDA/glutamate signaling pathway ↓ Pro-inflammatory mediators (NO, iNOS, IL-1β, IL-6, TNFα, NSE, MMP-9) ↓, NF-κB pathway ↓, ICAM-1 ↓ Neuronal cell apoptosis ↓, caspase-3 ↓, cytochrome c ↓, p53 ↓, mitochondrial complex I ↑ | [130,131,132] | |
| Vitamin D | Lipid peroxidation ↓, MDA ↓, ROS ↓, NGF ↑, GDNF ↑, NT3 ↑ Pro-inflammatory mediators (GFAP, iNOS, COX-2, IL-1β, IL-6, IL-17A, TNFα) ↓, anti-inflammatory cytokines (IL-4, IL-10, TGF-β) ↑, Iba-1 ↓, TLR4 ↓, SOCS3 pathways ↑, NLRP3 ↓, caspase-1 ↓, M1 microglia ↓, M2 microglia ↑ | [134,135,136] | |
| Coenzyme Q10 | Lipid peroxidation ↓, ROS ↓, SOD ↑, CAT ↑, GSH ↑, GPx ↑, HO-1 ↑, Nrf-2 pathway ↑ Laminin (angiogenesis) ↑, ATP ↑, glutamate ↓, GABA ↓, mitochondrial potential ↑ Pro-inflammatory mediators (NO, iNOS, IL-1β, TNFα) ↓, anti-inflammatory cytokines (IL-10) ↑, Neuronal cell apoptosis ↓, caspase-3 ↓, Bax ↓, Bcl-2 ↑, ubiquitin-proteasome ↓ | [138,140] | |
| ω-3 fatty acids | AA ↓, Pro-inflammatory mediators (GFAP, iNOS, COX-2, PGE2, IL-1β, IL-6, TNFα, IFN-γ) ↓, HMGB1/TLR4/NF-κB pathway ↓, SIRT1 ↑, p38 MAPK ↓, PPARγ ↑, Iba-1 ↓ CD11b (microglia marker) ↓, APP ↓, PLA2 ↓, BDNF ↑, NGF ↑, GDNF ↑, TrkB (BDNF receptor) ↑ Neuronal cell apoptosis ↓, cleaved caspase-3 ↓, Bax ↓, Bcl-2 ↑, p75NTR ↓ | [142,143] | |
| Se | ROS ↓, GSH ↑, GPx ↑, GDNF ↑, VEGF ↑, PPARγ ↑, Preservation of mitochondrial potential Pro-inflammatory mediators (IL-1β, TNFα) ↓, Ubiquitin-proteasome ↓, STAT3 pathway ↓, mTOR phosphorylation ↓, Wnt signaling pathway ↑, p38 MAPK ↓, SAPK/JNK pathway ↓ Neuronal cell apoptosis ↓, caspase-3, -9 ↓, cytochrome c ↓, Bax ↓, Bcl-2 ↑, Mst1 (pro-apoptotic kinase) ↓, PARP ↓ | [145,146,147] |
| Therapeutic Agents | Commercial Names /Clinical Phase | Distinctive Features | Type of Diseases | Ref. |
|---|---|---|---|---|
| EGCG | Phase II/III (NCT00951834) | Inhibition of amyloid aggregation | Early stage of AD | [68] |
| Curcumin | Longvida®, Phase II (NCT01001637) | Solid lipid formulation (higher BA and BBB penetration; half-life: 7.5 h) | Moderate to severe AD | [86,87] |
| Resveratrol | Phase II (NCT01504854) | Reduction of MMP-9, Aβ42 and Aβ40 levels in CSF, attenuation of pro-inflammatory cytokines (IL-1R4, IL-8, IL-12, TNF-α) production, and elevation of IL-4 and FGF-2 levels | Mild to moderate AD | [92,93] |
| Ginsenoside Rd | Phase III (NCT00815763) | Significant improvement in the disability scores and stroke scales compared to placebo group | Acute ischemic stroke | [115,116] |
| N-acetyl cysteine | Phase II (IRCT20150629022965N16) | Improvement of neurological functional outcomes, reduction of inflammatory biomarkers (IL-6, sICAM-1, NO, MDA, NSE), and elevation of antioxidant enzymes (SOD, GPx) levels by anti-oxidant and anti-inflammatory effects | Acute ischemic stroke | [132] |
| Vitamin D3 | Phase III (IRCT20100407003655N4) | Downregulation of IL-17A expression and upregulation of TGF-β expression | MS | [135] |
| Coenzyme Q10 with IFN-β | Phase IV (EudraCT200800744714) | Reduction of pro-inflammatory mediator (IL-1β, IL-2R, IL-9, IL-17F, TNFα, IFN-γ, MIP-1α, GM-CSF) levels and elevation of anti-inflammatory cytokine (IL-4, IL-13) levels | MS | [139] |
| Type of Nanocarriers | Nanocarriers (Administration Routes) | Therapeutic Agents | Role of Nanocarriers | Type of Diseases | Ref. |
|---|---|---|---|---|---|
| Polymer-based NPs | PEG-α-tocopherol micelles (oral) | Coenzyme Q10 | The micelles solubilized hydrophobic coenzyme Q10 and enhanced its stability. The micelles improved its BA and delivery to brain. | MPTP-induced mouse model of PD | [170] |
| CBSA-conjugated PEG-PLA NPs (intravenous) | Tanshinone IIA | Positive charge of CBSA allowed tanshinone IIA to be more accumulated to the brain tissue through adsorptive mediated transcytosis. The NPs improved drug exposure and prolonged blood circulation. | MCAO surgery-induced rat cerebral ischemic stroke model | [173] | |
| Angiopep-2-conjugated PLGA NPs | Rg3 and thioflavin T | Angiopep-2 ligand allowed the NPs to cross the BBB and reach glial cells. Thioflavin T, encapsulated into the NPs, exhibited targeting Aβ fibrils. | In vitro BBB model using Aβ1-42 -pretreated C6 glial cells | [174] | |
| OL-conjugated PEG-PLGA NPs (intranasal) | Urocortin | OL ligand allowed the NPs to be more accumulated to the brain by its mucoadhesive properties and specific binding to l-fucose expressed on the olfactory epithelium. | 6-OHDA-induced rat model of PD | [177] | |
| TPP-CS NPs (intranasal) | Piperine | Positive charge of CS can exhibit absorption-enhancing effect and mucoadhesive properties, thereby improving nose-to-brain delivery of piperine. Negative charge of TPP allowed high loading efficiency of piperine. | Colchicine-induced rat model of AD | [186] | |
| Lf-conjugated TMCS NPs (intranasal) | Huperzine A | Positive charge of TMCS can exhibit absorption-enhancing effect and mucoadhesive properties. Lf ligand facilitated transportation into the brain through receptor-mediated endocytosis. The NPs improved absorption and brain distribution of huperzine A. | KM mouse (model for age-related decline) | [187] | |
| Anionic PAMAM dendrimers | N-acetyl cysteine (conjugated with dendrimer) | The dendrimers rapidly entered the neuronal cells and localized in the cytoplasm despite of their anionic charge. Based on this enhanced intracellular uptake, anti-oxidant and anti-inflammatory effects of drug were improved. | LPS-induced neuroinflammation in BV-2 cells | [192] | |
| Lipid-based NPs | SLNs (intranasal) | Astaxanthin | SLNs showed high drug-loading capacity and controlled release patterns. SLNs enhanced localization of astaxanthin in the brain. | H2O2-induced neurodegeneration in PC12 cells | [200] |
| Lf-conjugated NLCs (intravenous) | Curcumin | Lf ligand facilitated transportation across the BBB through receptor-mediated endocytosis, resulting in higher accumulation and localization of curcumin into the brain with reduced systemic distribution. | Aβ1-42- and D-gal- induced rat model of AD | [201] | |
| TfR Mab- and ApoE-conjugated liposomes (intravenous) | Curcumin derivative (as lipid component) | Dual, BBB specific ligands transported liposomes across the BBB. Lipid-derivative of curcumin allowed the liposomes to be targeted to amyloid peptides in the brain. | APP/PS1 transgenic mouse model of AD | [208] | |
| Inorganic NPs | Peptide B6-coated SA-Se NPs | SA and Se | Peptide B6 ligand allowed the NPs to be more uptake into the brain tissue (as PC12 cells) across the BBB (as bEND.3 cells). | In vitro BBB model using Aβ monomer -pretreated bEND.3 cells and PC12 cells | [214] |
| PEG-coated gold NPs (intravenous) | Anthocyanin | The NPs allowed anthocyanin to be highly accumulated into the brain across the BBB without cytotoxic effect. | Aβ1-42-induced mouse model of AD | [219] | |
| Dextran-coated SPIONs (intravenous) | Osmotin | Dextran coating can diminish undesired brain toxicity of SPIONs. Application of external magnetic field allowed the SPIONs and osmotin to be accumulated into the brain specifically (magnetic targeting) without disrupting the BBB integrity. | Aβ1-42-induced mouse model of AD | [223] | |
| Carbon-based NPs | PL- and polysorbate 80-coated MWCNTs (intravenous) | Berberine | MWCNTs led to sustained release of berberine. PL and polysorbate 80 coating let to higher biocompatibility of MWCNTs. The MWCNTs allowed berberine to be more transported into neuronal cells, be more absorbed systemically, and be accumulated in the brain across the BBB. | Aβ-induced rat model of AD | [230] |
| Biomimetic NPs | PEI-coated HSA NPs | Gallic acid | The cationic NPs reduced neurodegeneration by inhibiting fibrillation of αSN and interaction between its oligomers and cell membrane. It might be attributed to enhanced drug transportation into the neuronal cells via adsorptive mediated transcytosis. | αSN aggregates-treated PC12 cells (HD model) | [234] |
| Exosomes (intraperitoneal) | Quercetin, Curcumin | Exosomes enhanced drug BA owing to improved solubility and stability and prolonged half-life. Exosomes accelerated drug accumulation into the brain owing to their inherited targeting moieties. | OA-induced mouse model of AD | [238,239] |
| Targeting Strategies | Nanocarriers | Therapeutic Agents | Role of Nanocarriers and Observed Effects | Type of Diseases | Ref. |
|---|---|---|---|---|---|
| Dual-ligand functionalization (Neutrophil-targeting) | T7- and PGP-conjugated PEG-PAMAM dendrimers, Angiopep-2- and PGP-conjugated PEG-PAMAM dendrimers | Tanshinone IIA, Scutellarin | Dual-targeting ligands transported the nanocarrier across the BBB and targeted neutrophil, resulting in enhancing drug accumulation in the brain. The dendrimer NPs suppressed the concentration of intracellular Ca2+ and production of pro-inflammatory cytokines by inhibiting the HMGB1/TLRs signaling pathway. | MCAO surgery-induced rat cerebral ischemic stroke model | [241,242] |
| Natural compounds functionalization (Huntingtin-targeting) | Iron oxide-corded zwitter ionic polyacrylate NSs | Trehalose (covalently linked with NSs) | Zwitter ionic NSs can interact with cell membrane, thereby crossing the BBB. Multiple terminal trehalose offered interaction with intracellular huntingtin peptides, which leads to enhanced brain targeting. The NSs efficiently inhibited mutant huntingtin aggregation and amyloid fibrillation, resulting in attenuation of neurodegeneration. | Mouse model of HD | [243] |
| Natural compounds functionalization (Aβ- and neuron- targeting) | Tet-1-coated EGCG-Se NPs | EGCG (conjugated on Se core) | Tet-1 and EGCG ligands allowed the NPs to specifically interact with neurons and Aβ, respectively, resulting in enhancement of brain delivery. The NPs improved inhibition of EGCG on Aβ aggregation and ROS production, thereby attenuating apoptotic response. | Aβ monomer or disaggregated Aβ fibrill -pretreated PC12 cells | [244] |
| Ligand-functionalized exosome (Reactive vascular endothelium-targeting) | Cyclic RGDyK-conjugated exosomes | Curcumin | The ligand conjugation enhanced specific uptake of exosomes into reactive endothelial cells and the exosomes migrated to the lesion region of the ischemic brain. The exosomes reduced production of pro-inflammatory cytokines and expression of cleaved caspase-3 and attenuated activation of microglia and NF-κB pathway. | MCAO surgery-induced mouse cerebral ischemic stroke model | [245] |
| β-CD nanovalves (H2O2 and Aβ- targeting) | Bor-β-CD/Fc complexes-conjugated MSe NPs | Resveratrol | Bor ligand can interact with cell membrane, thereby allowing the NPs to cross the BBB. β-CD/Fc exhibited H2O2-sensitive dissociation, followed by release of resveratrol into Aβ-induced lesion site. The NPs improved drug BA and prolonged its blood circulation. The NPs attenuated neuroinflammation via the downregulation of pro-inflammatory cytokines, NO, and ROS and upregulation of anti-inflammatory cytokines. The NPs inhibited the formation of Aβ and tau phosphorylation, thereby attenuating neuronal cell death. | APP/PS1 transgenic mouse model of AD | [246] |
| Biomimetic magnetic NP (Thrombus-targeting and magnetic guidance) | Iron oxide NPs-loaded PMVs | l-arginine | PMVs can recognize damaged blood vessels and specifically bind to thrombus in the lesion site of the ischemic brain. Application of an external magnetic field allowed the PMVs to be more quickly adhered and more accumulated in the lesion site owing to loaded iron oxide NPs. The PMVs modulated the production of NO, resulting in promotion of revascularization. The PMVs enhanced the expression of CD31, leading to the attenuation of prolonged inflammatory response. | Focal cerebral ischemia mouse model | [249] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khadka, B.; Lee, J.-Y.; Park, D.H.; Kim, K.-T.; Bae, J.-S. The Role of Natural Compounds and their Nanocarriers in the Treatment of CNS Inflammation. Biomolecules 2020, 10, 1401. https://doi.org/10.3390/biom10101401
Khadka B, Lee J-Y, Park DH, Kim K-T, Bae J-S. The Role of Natural Compounds and their Nanocarriers in the Treatment of CNS Inflammation. Biomolecules. 2020; 10(10):1401. https://doi.org/10.3390/biom10101401
Chicago/Turabian StyleKhadka, Bikram, Jae-Young Lee, Dong Ho Park, Ki-Taek Kim, and Jong-Sup Bae. 2020. "The Role of Natural Compounds and their Nanocarriers in the Treatment of CNS Inflammation" Biomolecules 10, no. 10: 1401. https://doi.org/10.3390/biom10101401
APA StyleKhadka, B., Lee, J.-Y., Park, D. H., Kim, K.-T., & Bae, J.-S. (2020). The Role of Natural Compounds and their Nanocarriers in the Treatment of CNS Inflammation. Biomolecules, 10(10), 1401. https://doi.org/10.3390/biom10101401