1. Introduction
In general, optical frequency measurements of
–
transition in hydrogen are of significant interest in high-precision spectroscopy [
1,
2,
3,
4]. For the interpretation of experimental data, the pressure shift experienced by atoms inside the atomic beam is of particular interest [
5,
6] and its description requires considerable theoretical effort. This is because the pressure shift is generated by interatomic long-range (van-der-Waals) interactions. The evaluation of the interaction potentials constitutes a highly nontrivial task in the case of excited-state atoms [
7,
8,
9,
10,
11,
12] in view of the nontrivial placement of the infinitesimal imaginary parts in the propagator denominators [
10], as well as due to the necessity of employing hyperfine resolution for the evaluation of the long-range interaction in the presence of quasi-degenerate states.
The leading-order van-der-Waals interaction between two excited electrically neutral hydrogen atoms, which are in spherically symmetric states, has been studied in Refs. [
7,
8,
9], with a full account of the hyperfine sub-manifolds of states. Long-range interactions among excited-state atoms are characterized by the presence of quasi-degenerate states. Therefore, the treatment of the long-range (van-der-Waals) interaction of two excited atoms proceeds differently from that of ground-state atoms and from that of the excited-ground-state system. This statement applies in particular to atoms where the virtual transitions to the quasi-degenerate states are of low multipole order, e.g., in the case of dipole-allowed transitions.
For two atoms in reference
S states, virtual dipole transitions to
P states lead to interaction potentials proportional to
, where
R is the interatomic distance. The so-called
coefficients multiplying the second-order interaction potential can assume large numerical values in the presence of quasi-degenerate states (see Refs. [
6,
8,
9,
12,
13]). For hydrogen, two sources of quasi-degeneracy need to be distinguished. The first of these is the Lamb shift. For example, in the
manifold of states, the
state is removed from
only by the Lamb shift frequency interval of
, which is very small on the scale of atomic energy. The latter is governed by the Hartree energy
(here,
is the fine-structure constant,
is the electron mass, and
c is the speed of light). The other source of quasi-degeneracy lies in the hyperfine structure itself. For example, in the case of two hydrogen atoms in metastable
states, the hyperfine splitting (together with the Lamb shift) enters the propagator denominators in the coefficients of the
interaction [
8]. (Note that first-order interactions, proportional to
, average out to zero in the calculation of pressure shifts in atomic beams [
5,
6] and are therefore not considered here. Higher orders of perturbation theory, by contrast, lead to more powers of
R in the interaction energy and thus to parametric suppression.) Due to the extremely long wavelength of hyperfine and fine-structure transitions (when measured on the atomic scale), the dominant contribution to the long-range interaction remains nonretarded on all experimentally relevant length scales.
A prime example is the interaction of metastable hydrogen atoms in the state with other hydrogen atoms in excited states. We here put special emphasis on the cases and . There are dipole-allowed virtual transitions to and , as well as states (the latter are present for ). The presence of these quasi-degenerate states makes a full diagonalization of the van-der-Waals Hamiltonian in the hyperfine-resolved basis necessary. Since for D states, there are possible projections of orbital angular momentum along the axis of quantization, in addition to the spin projections. This aspect enhances the dimensionality of the Hamiltonian matrices in the hyperfine-resolved basis, which describes the virtual transitions among the energetically quasi-degenerate states. The second complicating aspect is that, within the hyperfine-resolved manifolds, we also have fine-structure-hyperfine-structure mixing matrix elements, which couple states with different j values (but the same F), where j is the total angular momentum of the electronic part and F is the overall total angular momentum quantum number (which includes the nuclear spin).
SI mksA units are used throughout this paper, which is organized as follows. Details of the Hamiltonian of our system are discussed in
Section 2. In
Section 3, for the
system, we look into how the Hamiltonian matrix decomposes into nine hyperfine manifolds, namely,
, where
is the sum of the projections of
F for both atoms. We devote
Section 4 to the analysis of the second-order van-der-Waals shifts in the
system. Conclusions are drawn in
Section 5.
2. Mathematical Formalism
The total Hamiltonian of a system, in which two neutral hydrogen atoms interact with each other, can be written as
where the perturbation
is the van-der-Waals Hamiltonian of the system. The unperturbed Hamiltonian
is the sum of the Lamb shift Hamiltonian
, the fine-structure Hamiltonian
, and the hyperfine-structure Hamiltonian
,
Here
enumerates the two atoms involved in the interaction. In SI mksA units adapted to the atomic scale (using the Hartree energy
as the energy scale), the Schrödinger and the fine-structure Hamiltonians, respectively, can be written as follows:
Here,
is the Hartree energy,
is the fine-structure constant,
is the position vector of the electron with respect to its nucleus for the
ith atom,
is the Bohr’s radius, and
c is the speed of light. We also add the Lamb shift Hamiltonian, which has the matrix elements
In the above form, the Lamb shift Hamiltonian is taken in leading order (self energy and vacuum polarization) within the non-recoil approximation. The self-energy operator and the Uehling potential, are diagonal in the angular-momentum basis [
14,
15], while the van-der-Waals interaction couples states with different angular momenta. Note that the numerical coefficient
in Equation (
5) is, quite famously, the result of a numerical coefficient
from the self energy and
from vacuum polarization [
14,
15]. The Bethe logarithm is denoted as
, and the Dirac angular momentum number
is equal to
, where we recall that
ℓ is the orbital and
j is the total electron angular momentum. In regard to the principal quantum numbers, we take only the diagonal matrix elements of the Lamb shift operator into account. Off-diagonal elements lead to tiny, higher-order corrections in the calculation of the van-der-Waals interaction and can be ignored.
The
ith momentum operator is
and
is the orbital angular momentum operator for electron
i. Furthermore,
e is the elementary charge,
is the electron mass, and
is the reduced mass of the system;
is the spin operator for electron
i. The Hamiltonians in Equations (
5) and (
4) are given for reference only. In the course of our investigations below, we use the full theoretical values for the fine-structure splitting and Lamb shift [
16].
The hyperfine Hamiltonian is
where
is the spin operators for proton
i. The proton mass is denoted as
, while
and
are the electronic and protonic
g-factors.
The perturbation comes through the distance-dependent van-der-Waals Hamiltonian of the system
. The interaction Hamiltonian of atoms
A and
B can be written as
where
and
are the position vectors of atoms
A and
B, respectively, whose electrons are at positions
and
. We assume that the inter-nuclear distance,
is much larger than the distance between a nucleus and its respective electron, i.e.,
as well as
, where the components of
are given as
The leading-order contribution to the van-der Waals interaction is the dipole–dipole interaction, which reads
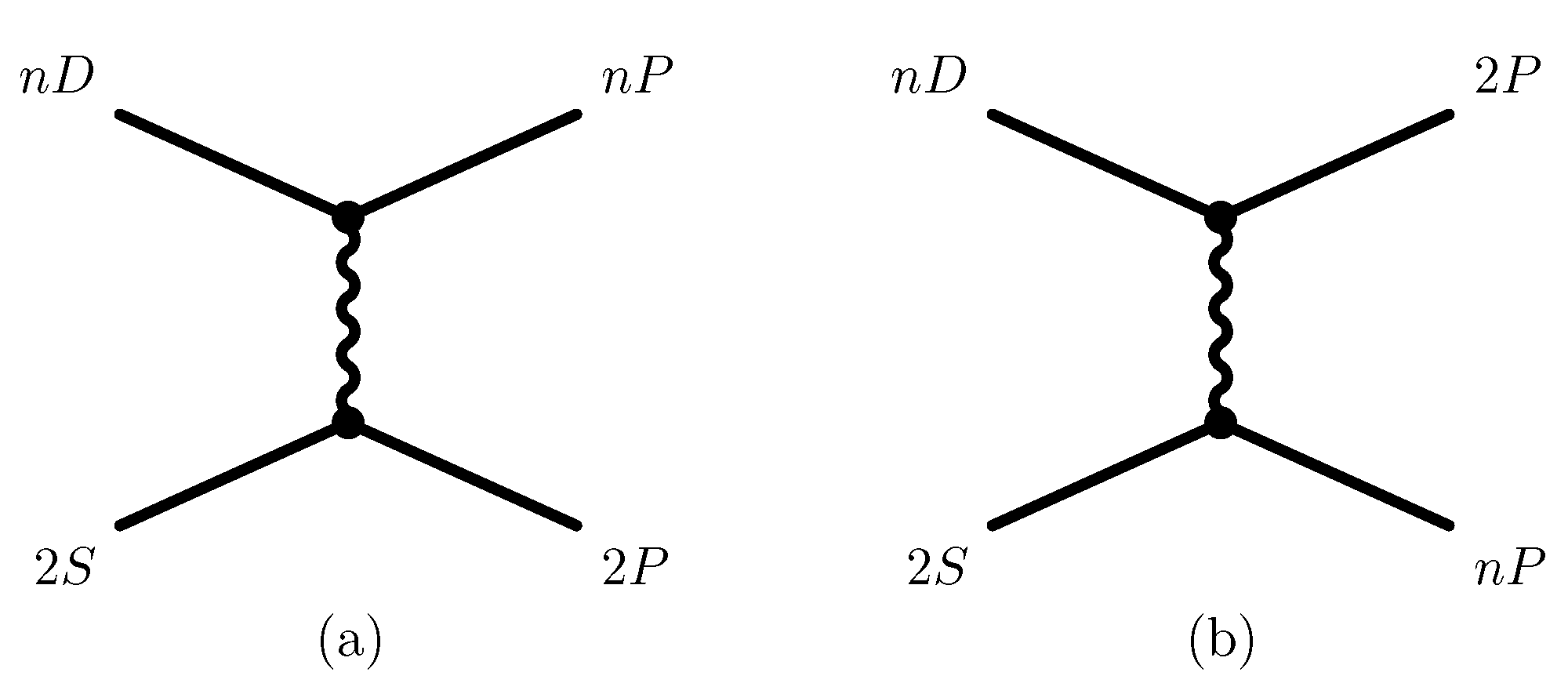
where we assume that the interatomic separation is aligned with the quantization axis,
. The corresponding Feynman diagram for one-photon exchange (for the
system, involving virtual
P states) is depicted in
Figure 1.
The role of the virtual
states is worthy of special attention. The
and
levels are displaced only by the classic Lamb shift,
, while the
levels are displaced by the fine structure which is larger than the classic Lamb shift by an order of magnitude (see
Figure 2). Because of this relatively large separation from the reference
level, we discard the
states. The theoretical accuracy of our results for the
coefficients can thus be estimated to be on the order of roughly
. We stress that the main aim of our calculations is the determination of estimates of the
coefficients for pressure shifts in atomic beams; the pressure shift is proportional to
(see Ref. [
6]). The resulting theoretical uncertainty of
in the pressure shift is perfectly acceptable, both because the effect is small overall and because its determination depends on other parameters such as the volume density of atoms in the beam, which can typically be estimated only with much less precision [
6]. Thus, here and in the following, whenever we discuss
levels, we do not mean
. The
and
levels are about
MHz apart, and the separation between
and
levels is only about
MHz. The spacing between
and
levels is about three orders of magnitude larger than the
–
splitting (see
Figure 2a). The same is true in the case of
–
and
–
spacings (see
Figure 2b). The role of virtual
states is thus suppressed in comparison to virtual
states. The energetically quasi-degenerate
–
transition plays a significant role in the long-range
–
interaction.
When setting up the calculation, it is useful to define the following parameters:
The hyperfine parameter
is the hyperfine splitting between the
hyperfine singlet and the
hyperfine triplet of the
states;
is fine-structure splitting between
and
levels whereas
is the fine-structure splitting between
and
levels of hydrogen atom [
16]. Corresponding quantities for the
system are defined in
Figure 2b. Numerical values of fundamental physical constants are taken from Refs. [
17,
18]. One can calculate the ratios of energy level spacings given in
Figure 2 and find that the Lamb shift
and the fine-structure splittings
,
, and
approximately follow the
scaling law, where
n is the principal quantum number.
Due to the multitude of relevant levels in the
and
manifolds, it is alternatively possible to define the following parameters:
where
is a dimensionless parameter which expresses the energy displacement of the level with quantum numbers
n,
ℓ, and
j from the energetically lowest level
within the manifold of given
n in units of the Lamb shift scale
. One may use the database [
16] to obtain the following results:
The smaller parameters for illustrate that the higher excited states are energetically closer than for , which results in smaller propagator denominators and larger second-order long-range energy shifts.
To explain these ideas, a few remarks might be in order. An atom in a state has four hyperfine states, namely, a hyperfine singlet for and a hyperfine triplet for . The states have and fine structures. For , the hyperfine quantum number F takes the values and , while for states, one either has or . As a result, the and states further split into eight and twelve hyperfine states, respectively. For atomic states with , one has hyperfine manifolds with and . We here use the notation to denote the basis states, where n is the principal quantum number, ℓ is the orbital quantum number, and j is the total (orbital + spin) electronic angular momentum quantum number. Furthermore, we denote by F the overall total (orbital + electron spin + nuclear spin) angular momentum quantum number. The projection of the overall total angular momentum quantum number onto the quantization axis (z axis, aligned with the interatomic separation) of an individual atomic electron is denoted as (or, as with if the specification of the atom is not clear from the context). For the two atoms, we anticipate that the total overall angular momentum projection is as the sum of the projections for the electrons in atoms A and B. The quantum number is a conserved quantity under the long-range interaction and allows us to separate the Hamiltonian matrices into mutually noninteracting submanifolds.
The unperturbed states in our problem are thus the states
with both hydrogen atoms in well-defined hyperfine states. The hyperfine states are constructed from the states
via the addition of the nuclear angular momentum. Conversely, one establishes, with reference to Equation (
5), the diagonality of the Lamb shift Hamiltonian in the hyperfine basis:
In this context, it is useful to remember that hyperfine effects are excluded from the energy levels of states
given in the database [
16]. This observation applies to the definition of the
parameters in Equation (
14).
Let us now briefly discuss the evaluation of the diagonal elements of the hyperfine Hamiltonian, noting that we can temporarily drop the index
indicating the atom, since we are dealing with two identical hydrogen atoms. The expectation value of
measures the diagonal entries of the hyperfine splitting Hamiltonian, and it reads [
19]
where
is the proton mass. The quantity
depends only on the electronic part. In the nonrelativistic limit, one obtains the following results [
19]:
The proton spin is
. For all hydrogenic states of interest, Equations (
23) and (
24) define the hyperfine splitting uniquely. An example is the hyperfine energy shift of
states:
For
and
states, one obtains the results
It is well known that the hyperfine Hamiltonian has off-diagonal elements in the fine-structure resolved basis. The relevant off-diagonal elements of the hyperfine Hamiltonian for the
reference states under investigation are given by
The nontrivial off-diagonal matrix elements are incurred for
and
states with the same overall total angular momentum quantum number
and the same
. It is evident from the ratio of Equation (
30) to Equation (31), i.e.,
, that the off-diagonal hyperfine splitting follows the
scaling.
A rediagonalization of the hyperfine Hamiltonian in the basis of states
leads to the following second-order energy shifts:
Taking the ratio of the
s from Equations (
33) and (
34), we have
, consistent with the fact that
.
3. Second–Order van-der-Waals Shifts in the System
The calculation proceeds as follows. The reference states are
states, with one atom in the metastable
state and the other in the excited
state. One includes virtual
states that could be reached via electric dipole transitions and have nonvanishing transition matrix elements with the Hamiltonian (
9). Specifically, for the reasons outlined above, one investigates the manifolds composed of the product states of
,
,
,
,
, and
. Of the product states, only the
states could be interpreted as reference states. The total number of states is
. These states could in principle form
product states in hyperfine resolution, which all should be analyzed. However, we can select from the product states only those that are energetically quasi-degenerate with respect to the reference states. The physically relevant basis states have
for the
interaction and
. Namely, for
and
, one has a reference state, while for
, one has a virtual state composed of two
P states. Here,
and
are the principal and the orbital angular quantum numbers, respectively, of the
ith atom
. The van-der-Waals Hamiltonian (
9) is diagonal in the quantum number
which is the sum of the two projections of atoms
A and
B. One can easily find that the nine physically relevant hyperfine manifolds are those with
. The two reference states in the
hyperfine manifold are (see also
Table 1),
The
manifold does not involve any quasi-degenerate
states that could be reached via dipole transitions (remember that we exclude
states); hence, the second-order energy shift vanishes (see also
Table 2). The manifolds with
have 12 quasi-degenerate states, while for
, one has 32 quasi-degenerate states. For
, one encounters 52 states, while for
, one has 60 states. For an example of a matrix obtained for a given value of
in a related calculation for a different atomic-state configuration, we refer to Section 3.4 of Ref. [
5]. Our calculation reported here proceeds analogously, but with even higher-dimensional matrices for each
as compared to Ref. [
5].
For clarity, we thus present one particular case, namely, the
submanifold for the
system, in
Section 4. Note also that, as explained in the discussion following Equation (
9), the final results for the
coefficients have a theoretical uncertainty of roughly 10%. We still indicate the results in
Table 2 to five significant figures, in order to facilitate an accurate independent recalculation of the coefficients within the indicated, specified basis set of states. The quoted accuracy is thus nominal and does not imply that all indicated figures are physically significant.
One then selects from every
manifold the reference
states with the defined
eigenvalue and then calculates the second-order shifts due to all
hyperfine sublevels coupled to the reference level via the van-der-Waals Hamiltonian. One verifies that the first-order shifts vanish when the average is taken in a specific
manifold, which makes the first-order shifts physically irrelevant [
6]. One may average the second-order shifts in various ways. For a given
value, when averaging over the possible
j and
F values of the excited
D state, one obtains the the following results after the extensive use of computer algebra [
20],
When averaging over
for given
j and
F, one has
The fine-structure averages, keeping
j fixed but averaging over
F and
, are given as
Recall that
is given in Equation (
33).
4. Second–Order van-der-Waals Shifts in the System
One proceeds similar to the
case. With the reference
states, one includes virtual
and virtual
states that could be reached via electric dipole transitions and have nonvanishing transition matrix elements with the Hamiltonian (
9). Specifically, one investigates the manifolds composed of the product states of
,
,
,
,
, and
, as well as
and
. Only the hyperfine manifolds of the
states act as reference states. The total number of states is
states. Of the
product states, one can select only those which are energetically quasi-degenerate with respect to the reference states. The relevant basis states have
for the
interaction, and
and/or
(virtual
states). For
and
, one has a reference state, while for
,
, and
, one has virtual states. Here, again,
and
are the principal and the orbital angular quantum numbers of the
ith atom
. Since the van-der-Waals Hamiltonian (
9) is diagonal in the quantum number
, one can diagonalize the interaction in the submanifolds with
separately. For our purposes, the submanifold with
is physically irrelevant because it does not contain
reference states.
For the
system, one needs to consider a complicated array of virtual
P and
F states. The manifold with
(which contains 12 quasi-degenerate states) provides us with an opportunity to illustrate the calculation by way of and example. To this end, we supplement the definition given in Equations (
10)–(13) as follows:
where
measures the characteristic scale of a first-order element of the nonretarded van-der-Waals interaction, and the interatomic distance, expressed in atomic units, is denoted as
. The first six states, given in the notation introduced in Equation (
21), in the manifold with
. are as follows,
They are complemented by six further states with
,
In the basis of states
, the matrix of the total Hamiltonian defined in Equation (
1) is
Here,
is a concise notation for the unperturbed energy levels without hyperfine effects. The energy level
enters the definition of the
parameter according to Equation (
14). Now, in the basis of states
, the Hamiltonian matrix of the total Hamiltonian defined in Equation (
1) can be calculated as
The Hamiltonian for
in in the basis of the twelve states listed in Equations (
49)—(54) is
where
is a 6-by-6 matrix with zero entries. For the manifolds with
, we already have 34 quasi-degenerate states and the presentation of the Hamiltonian matrix is not practical. For
, one already has 62 quasi-degenerate states. For
, one encounters 84 states, while for
, one has 92 states in the quasi-degenerate, hyperfine-resolved basis. One then proceeds as in the
case, selects the reference states, and calculates the second-order shifts in the quasi-degenerate bases. When averaging over
j and
F for a given
, one obtains
Of particular interest is the global hyperfine average of the second-order van-der-Waals shifts over all possible
values for given values of
j and
F (see also
Table 3):
The fine-structure averages, for fixed
j, but averaged over
F and
, are given as
The second-order shifts increase with the principal quantum number of the excited
D state, as would be expected. One consults Refs. [
5,
12] for analogous observations in the
systems, with
. We recall that
is given in Equation (34). One can now invoke the formalism introduced in Ref. [
6], and assume, for definiteness, a typical number density of
atoms/m
, which is 1% of the number density of the ground state hydrogen atoms used in Ref. [
21], at a temperature of
K (see Ref. [
21]). One can estimate the pressure shift from inside the atomic beam for the
transitions under the given conditions and on the basis of the data given in
Table 3, as well as Equations (
59)–(69), to be of the order of 100 Hz. Under different experimental conditions, the effect can be much larger.
5. Conclusions
We have studied leading-order (dipole–dipole) long-range interactions in the
and
hydrogen systems with hyperfine resolution. The Hamiltonian of the system is determined by Lamb shift, fine structure, hyperfine structure, and the long-range interaction, as discussed in
Section 2. The fine-structure-hyperfine-structure mixing term,
, couples the
and
states. We found that one must include the
,
and
states in the basis (
). However, one can exclude the
level while maintaining sufficient accuracy, because the
levels are comparatively closer to the reference
level than
(see also the discussion following Equation (
9)). The fine-structure splitting decreases with the principal quantum number. Analogous considerations are applied to the
interaction. However, for the
system, it is necessary to also consider virtual
F levels. We define the quantum number
as the sum of the total overall angular momentum projections
and
of both atoms. Discarding the irrelevant (for our investigations) subspace with
, we resolve the Hamiltonian matrix into nine hyperfine manifolds with dimensions of at most 92.
The hyperfine-averaged van-der-Waals
coefficients of the
system are of the order of
(in atomic units), while for the
system, they are of the order of
(in atomic units, see
Section 3 and
Section 4). These constitute very large numerical coefficients for van-der-Waals interactions. This confirms a trend of increasing
with the increasing quantum number, seen for
interactions in Refs. [
5,
12]. The van der Waals-type collisional shift and the collisional broadening both are linear to the number density of atoms
and proportional to
(see Ref. [
6]). For typical experimental parameters, the collisional shift in the
interaction can easily be as large as a few hundred Hz, if the number density of the atom in the
state is in the order of
atoms/m
. Collisional shifts inside the atomic beam are among the most challenging systematic effects in high-precision experiments and are hard to control. We hope to have shed some light on these effects for the experimentally interesting transitions of the metastable
state to the relatively long-lived excited
D states, which have been analyzed in a number of very remarkable experiments [
1,
2,
3,
4].