Metabolomics Contributions to the Discovery of Prostate Cancer Biomarkers
Abstract
1. Introduction
2. Cancer and Metabolic Reprogramming: Metabolomics Opportunities
3. Metabolomics and PCa
4. PCa Metabolic Biomarkers in Biofluids
4.1. Urine Biomarkers
4.2. Serum Biomarkers
4.3. Seminal Fluid Biomarkers
5. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 1H-NMR | Proton nuclear magnetic resonance spectroscopy |
| 2D-DIGE-MS | Two dimensional–difference gel electrophoresis–mass spectrometry |
| AUC | Area under the curve |
| BCAA | Branched-chain amino acids |
| BPH | Benign prostatic hyperplasia |
| CRPC | Castration-resistant prostate cancer |
| CS | Citrate synthase |
| DRE | Digital rectal examination |
| ELISA | Enzyme-linked immunosorbent assay |
| EPS | Expressed prostatic secretions |
| EV | Extracellular vesicles |
| FBP | Fructose-bisphosphatase |
| FIA-MS/MS | Flow injection analysis–tandem mass spectrometry |
| FPLC-MS | Fast ultra-high-performance liquid chromatography–mass spectrometry |
| GAA | Guanidinoacetate |
| GABA | Gamma-aminobutyric acid |
| GPI | Glucose-6-phosphate isomerase |
| GS | Gleason Score |
| GC-MS | Gas chromatography–mass spectrometry |
| GC-QqQ-MS | Gas chromatography–triple quadrupole–mass spectrometry |
| HG | High-grade (GS ≥ 8) |
| HK2 | Hexokinase 2 |
| HPLC-ESI-QTOF-MS | High performance liquid chromatography–electrospray ionization–quadrupole time of flight–mass spectrometry |
| HPLC-TOF-MS | High performance liquid chromatography–time of flight–mass spectrometry |
| HV | Healthy Volunteers |
| iTRAQ | Isobaric tag for relative and absolute quantification |
| LC-MS | Liquid chromatography–mass spectrometry |
| LC-MS/MS | Liquid chromatography–tandem mass spectrometry |
| LDH | Lactate dehydrogenase |
| LG | Low-grade (GS ≤ 7) |
| MALDI-TOF-MS | Matrix-assisted laser desorption ionization–time of flight–mass spectrometry |
| MS | Mass spectroscopy |
| NMR | Nuclear magnetic resonance |
| QqQ-MS: | Triple quadrupole–mass spectrometry |
| PCa | Prostate cancer |
| PDH | Pyruvate dehydrogenase |
| PEP | Phosphoenolpyruvate |
| PFK | Phosphofructokinase |
| PK | Pyruvate kinase |
| PM | Prostatic massage |
| PSA | Prostate specific antigen |
| SAM | S-Adenosyl methionine |
| T | Stage |
| TCA | Tricarboxylic acid |
| TMAO | Trimethylamine N-oxide |
| TRUS | Trans-rectal ultrasound |
| UHPLC-MS | Ultra-high-performance liquid chromatography–mass spectrometry |
| UPLC-MS/MS | Ultra performance liquid chromatography–tandem mass spectrometry |
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Schoenfield, L.; Jones, J.S.; Zippe, C.D.; Reuther, A.M.; Klein, E.; Zhou, M.; Magi-Galluzzi, C. The incidence of high-grade prostatic intraepithelial neoplasia and atypical glands suspicious for carcinoma on first-time saturation needle biopsy, and the subsequent risk of cancer. BJU Int. 2007, 99, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Offermann, A.; Hohensteiner, S.; Kuempers, C.; Ribbat-Idel, J.; Schneider, F.; Becker, F.; Hupe, M.C.; Duensing, S.; Merseburger, A.S.; Kirfel, J.; et al. Prognostic Value of the New Prostate Cancer International Society of Urological Pathology Grade Groups. Front. Med. 2017, 4, 157. [Google Scholar] [CrossRef] [PubMed]
- Chistiakov, D.A.; Myasoedova, V.A.; Grechko, A.V.; Melnichenko, A.A.; Orekhov, A.N. New biomarkers for diagnosis and prognosis of localized prostate cancer. Semin. Cancer Biol. 2018, 52, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Gordetsky, J.; Epstein, J. Grading of prostatic adenocarcinoma: Current state and prognostic implications. Diagn. Pathol. 2016, 11, 25. [Google Scholar] [CrossRef] [PubMed]
- Foley, R.W.; Maweni, R.M.; Gorman, L.; Murphy, K.; Lundon, D.J.; Durkan, G.; Power, R.; O’Brien, F.; O’Malley, K.J.; Galvin, D.J.; et al. European Randomised Study of Screening for Prostate Cancer (ERSPC) risk calculators significantly outperform the Prostate Cancer Prevention Trial (PCPT) 2.0 in the prediction of prostate cancer: A multi-institutional study. BJU Int. 2016, 118, 706–713. [Google Scholar] [CrossRef]
- Nam, R.K.; Satkunasivam, R.; Chin, J.L.; Izawa, J.; Trachtenberg, J.; Rendon, R.; Bell, D.; Singal, R.; Sherman, C.; Sugar, L.; et al. Next-generation prostate cancer risk calculator for primary care physicians. Can. Urol. Assoc. J. 2017, 12, E64–E70. [Google Scholar] [CrossRef]
- Loeb, S.; Partin, A.W. Review of the Literature: PCA3 for Prostate Cancer Risk Assessment and Prognostication. Rev. Urol. 2011, 13, 191–195. [Google Scholar] [CrossRef]
- Sanhueza, C.; Kohli, M. Clinical and Novel Biomarkers in the Management of Prostate Cancer. Curr. Treat. Options Oncol. 2018, 19. [Google Scholar] [CrossRef]
- Biomarkers PCA3 and TMPRSS2-ERG: Better together: Prostate cancer. Nat. Rev. Urol. 2014, 11, 129. [CrossRef]
- Perner, S.; Mosquera, J.-M.; Demichelis, F.; Hofer, M.D.; Paris, P.L.; Simko, J.; Collins, C.; Bismar, T.A.; Chinnaiyan, A.M.; De Marzo, A.M.; et al. TMPRSS2-ERG Fusion Prostate Cancer: An Early Molecular Event Associated with Invasion. Am. J. Surg. Pathol. 2007, 31, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, C.E.; Demichelis, F.; Rubin, M.A. Molecular genetics of prostate cancer: Emerging appreciation of genetic complexity. Histopathology 2012, 60, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Tomlins, S.A. Recurrent Fusion of TMPRSS2 and ETS Transcription Factor Genes in Prostate. Cancer Sci. 2005, 310, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Buonerba, C.; Terracciano, D.; Lucarelli, G.; Cosimato, V.; Bottero, D.; Deliu, V.M.; Ditonno, P.; Perdonà, S.; Autorino, R.; et al. Biomarkers in localized prostate cancer. Future Oncol. 2016, 12, 399–411. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, R.J.; van Oort, I.M.; Schalken, J.A. Blood-based and urinary prostate cancer biomarkers: A review and comparison of novel biomarkers for detection and treatment decisions. Prostate Cancer Prostatic Dis. 2017, 20, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Choi, S.A.; Na, J.; Pamungkas, A.D.; Jung, K.J.; Jee, S.H.; Park, Y.H. Non-invasive Serum Metabolomic Profiling Reveals Elevated Kynurenine Pathway’s Metabolites in Humans with Prostate Cancer. J. Proteome Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.K.; Rise, K.; Giskeødegård, G.F.; Richardsen, E.; Bertilsson, H.; Størkersen, Ø.; Bathen, T.F.; Rye, M.; Tessem, M.-B. Integrative metabolic and transcriptomic profiling of prostate cancer tissue containing reactive stroma. Sci. Rep. 2018, 8, 14269. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Nonomura, N. Urinary biomarkers of prostate cancer. Int. J. Urol. 2018, 25, 770–779. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Gupta, A.; Nath, K. NMR-based metabolomics of prostate cancer: A protagonist in clinical diagnostics. Expert Rev. Mol. Diagn. 2016, 16, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.; Wilson, I.D.; Nicholson, J.K. Metabolic phenotyping in health and disease. Cell 2008, 134, 714–717. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; DeBerardinis, R.J. Understanding the Intersections between Metabolism and Cancer Biology. Cell 2017, 168, 657–669. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, N.N.; Thompson, C.B. The Emerging Hallmarks of Cancer Metabolism. Cell Metab. 2016, 23, 27–47. [Google Scholar] [CrossRef]
- Levine, A.J.; Puzio-Kuter, A.M. The control of the metabolic switch in cancers by oncogenes and tumor suppressor genes. Science 2010, 330, 1340–1344. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.S.; Patel, J.; Wise, D.R.; Abdel-Wahab, O.; Bennett, B.D.; Coller, H.A.; Cross, J.R.; Fantin, V.R.; Hedvat, C.V.; Perl, A.E.; et al. The common feature of leukemia-associated IDH1 and IDH2 mutations is a neomorphic enzyme activity converting alpha-ketoglutarate to 2-hydroxyglutarate. Cancer Cell 2010, 17, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Hipp, S.J.; Steffen-Smith, E.A.; Patronas, N.; Herscovitch, P.; Solomon, J.M.; Bent, R.S.; Steinberg, S.M.; Warren, K.E. Molecular imaging of pediatric brain tumors: Comparison of tumor metabolism using 18F-FDG-PET and MRSI. J. Neurooncol. 2012, 109, 521–527. [Google Scholar] [CrossRef]
- Zhan, H.; Ciano, K.; Dong, K.; Zucker, S. Targeting glutamine metabolism in myeloproliferative neoplasms. Blood Cells Mol. Dis. 2015, 55, 241–247. [Google Scholar] [CrossRef]
- Sutinen, E.; Nurmi, M.; Roivainen, A.; Varpula, M.; Tolvanen, T.; Lehikoinen, P.; Minn, H. Kinetics of [(11)C]choline uptake in prostate cancer: A PET study. Eur. J. Nuclear Med. Mol. Imaging 2004, 31, 317–324. [Google Scholar] [CrossRef]
- Srivastava, A.; Creek, D.J. Discovery and Validation of Clinical Biomarkers of Cancer: A Review Combining Metabolomics and Proteomics. Proteomics 2018, 1700448. [Google Scholar] [CrossRef]
- Zhang, A.; Sun, H.; Yan, G.; Wang, P.; Wang, X. Metabolomics for Biomarker Discovery: Moving to the Clinic. Biomed. Res. Int. 2015, 2015, 354671. [Google Scholar] [CrossRef] [PubMed]
- Mirnaghi, F.S.; Caudy, A.A. Challenges of analyzing different classes of metabolites by a single analytical method. Bioanalysis 2014, 6, 3393–3416. [Google Scholar] [CrossRef] [PubMed]
- Wolfender, J.-L.; Marti, G.; Thomas, A.; Bertrand, S. Current approaches and challenges for the metabolite profiling of complex natural extracts. J. Chromatogr. A 2015, 1382, 136–164. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Marsal, S.; Julià, A. Analytical methods in untargeted metabolomics: State of the art in 2015. Front. Bioeng. Biotechnol. 2015, 3, 23. [Google Scholar] [CrossRef] [PubMed]
- Bingol, K.; Brüschweiler, R. Two elephants in the room: New hybrid nuclear magnetic resonance and mass spectrometry approaches for metabolomics. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Fuhrer, T.; Zamboni, N. High-throughput discovery metabolomics. Curr. Opin. Biotechnol. 2015, 31, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Clos-Garcia, M.; Loizaga-Iriarte, A.; Zuñiga-Garcia, P.; Sánchez-Mosquera, P.; Rosa Cortazar, A.; González, E.; Torrano, V.; Alonso, C.; Pérez-Cormenzana, M.; Ugalde-Olano, A.; et al. Metabolic alterations in urine extracellular vesicles are associated to prostate cancer pathogenesis and progression. J. Extracell. Vesicles 2018, 7, 1470442. [Google Scholar] [CrossRef]
- Liang, Q.; Liu, H.; Xie, L.; Li, X.; Zhang, A.-H. High-throughput metabolomics enables biomarker discovery in prostate cancer. RSC Adv. 2017, 7, 2587–2593. [Google Scholar] [CrossRef]
- Gkotsos, G.; Virgiliou, C.; Lagoudaki, I.; Sardeli, C.; Raikos, N.; Theodoridis, G.; Dimitriadis, G. The Role of Sarcosine, Uracil, and Kynurenic Acid Metabolism in Urine for Diagnosis and Progression Monitoring of Prostate Cancer. Metabolites 2017, 7, 9. [Google Scholar] [CrossRef]
- Struck-Lewicka, W.; Kordalewska, M.; Bujak, R.; Yumba Mpanga, A.; Markuszewski, M.; Jacyna, J.; Matuszewski, M.; Kaliszan, R.; Markuszewski, M.J. Urine metabolic fingerprinting using LC–MS and GC–MS reveals metabolite changes in prostate cancer: A pilot study. J. Pharm. Biomed. Anal. 2015, 111, 351–361. [Google Scholar] [CrossRef]
- Fernández-Peralbo, M.A.; Gómez-Gómez, E.; Calderón-Santiago, M.; Carrasco-Valiente, J.; Ruiz-García, J.; Requena-Tapia, M.J.; Luque de Castro, M.D.; Priego-Capote, F. Prostate Cancer Patients–Negative Biopsy Controls Discrimination by Untargeted Metabolomics Analysis of Urine by LC-QTOF: Upstream Information on Other Omics. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Puhka, M.; Takatalo, M.; Nordberg, M.-E.; Valkonen, S.; Nandania, J.; Aatonen, M.; Yliperttula, M.; Laitinen, S.; Velagapudi, V.; Mirtti, T.; et al. Metabolomic Profiling of Extracellular Vesicles and Alternative Normalization Methods Reveal Enriched Metabolites and Strategies to Study Prostate Cancer-Related Changes. Theranostics 2017, 7, 3824–3841. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Kume, H.; Matsuzaki, K.; Kawashima, A.; Ujike, T.; Nagahara, A.; Uemura, M.; Miyagawa, Y.; Tomonaga, T.; Nonomura, N. Proteomic analysis of urinary extracellular vesicles from high Gleason score prostate cancer. Sci. Rep. 2017, 7, 42961. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rambla, C.; Puchades-Carrasco, L.; García-Flores, M.; Rubio-Briones, J.; López-Guerrero, J.A.; Pineda-Lucena, A. Non-invasive urinary metabolomic profiling discriminates prostate cancer from benign prostatic hyperplasia. Metabolomics 2017, 13. [Google Scholar] [CrossRef]
- Davalieva, K.; Kostovska, I.M.; Kiprijanovska, S.; Markoska, K.; Kubelka-Sabit, K.; Filipovski, V.; Stavridis, S.; Stankov, O.; Komina, S.; Petrusevska, G.; et al. Proteomics analysis of malignant and benign prostate tissue by 2D DIGE/MS reveals new insights into proteins involved in prostate cancer: Proteomics Analysis of Prostate Cancer. Prostate 2015, 75, 1586–1600. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Gupta, A.; Mandhani, A.; Sankhwar, S.N. NMR spectroscopy of filtered serum of prostate cancer: A new frontier in metabolomics: Metabolomics of Prostate Cancer. Prostate 2016, 76, 1106–1119. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Gupta, A.; Mandhani, A.; Sankhwar, S.N. Metabolomics-Derived Prostate Cancer Biomarkers: Fact or Fiction? J. Proteome Res. 2015, 14, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Heger, Z.; Michalek, P.; Guran, R.; Cernei, N.; Duskova, K.; Vesely, S.; Anyz, J.; Stepankova, O.; Zitka, O.; Adam, V.; et al. Differences in urinary proteins related to surgical margin status after radical prostatectomy. Oncol. Rep. 2015, 34, 3247–3255. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Giskeødegård, G.F.; Hansen, A.F.; Bertilsson, H.; Gonzalez, S.V.; Kristiansen, K.A.; Bruheim, P.; Mjøs, S.A.; Angelsen, A.; Bathen, T.F.; Tessem, M.-B. Metabolic markers in blood can separate prostate cancer from benign prostatic hyperplasia. Br. J. Cancer 2015, 113, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Lv, H.; Qiu, S.; Gao, L.; Ai, H. Plasma metabolic profiling and novel metabolite biomarkers for diagnosing prostate cancer. RSC Adv. 2017, 7, 30060–30069. [Google Scholar] [CrossRef]
- Lin, H.-M.; Mahon, K.L.; Weir, J.M.; Mundra, P.A.; Spielman, C.; Briscoe, K.; Gurney, H.; Mallesara, G.; Marx, G.; Stockler, M.R.; et al. A distinct plasma lipid signature associated with poor prognosis in castration-resistant prostate cancer: Prognostic lipid signature in metastatic prostate cancer. Int. J. Cancer 2017, 141, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Mondul, A.M.; Moore, S.C.; Weinstein, S.J.; Karoly, E.D.; Sampson, J.N.; Albanes, D. Metabolomic analysis of prostate cancer risk in a prospective cohort: The alpha-tocopherol, beta-carotene cancer prevention (ATBC) study: Serum Metabolomics Profiling of Prostate Cancer Risk. Int. J. Cancer 2015, 137, 2124–2132. [Google Scholar] [CrossRef] [PubMed]
- Kühn, T.; Floegel, A.; Sookthai, D.; Johnson, T.; Rolle-Kampczyk, U.; Otto, W.; von Bergen, M.; Boeing, H.; Kaaks, R. Higher plasma levels of lysophosphatidylcholine 18:0 are related to a lower risk of common cancers in a prospective metabolomics study. BMC Med. 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.A.; Fensom, G.K.; Rinaldi, S.; Scalbert, A.; Appleby, P.N.; Achaintre, D.; Gicquiau, A.; Gunter, M.J.; Ferrari, P.; Kaaks, R.; et al. Pre-diagnostic metabolite concentrations and prostate cancer risk in 1077 cases and 1077 matched controls in the European Prospective Investigation into Cancer and Nutrition. BMC Med. 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Mondul, A.M.; Weinstein, S.J.; Karoly, E.D.; Sampson, J.N.; Albanes, D. Prospective serum metabolomic profile of prostate cancer by size and extent of primary tumor. Oncotarget 2017, 8. [Google Scholar] [CrossRef]
- Andras, I.; Crisan, N.; Vesa, S.; Rahota, R.; Romanciuc, F.; Lazar, A.; Socaciu, C.; Matei, D.-V.; de Cobelli, O.; Bocsan, I.-S.; et al. Serum metabolomics can predict the outcome of first systematic transrectal prostate biopsy in patients with PSA <10 ng/mL. Future Oncol. 2017, 13, 1793–1800. [Google Scholar] [CrossRef]
- Kline, E.E.; Treat, E.G.; Averna, T.A.; Davis, M.S.; Smith, A.Y.; Sillerud, L.O. Citrate Concentrations in Human Seminal Fluid and Expressed Prostatic Fluid Determined via 1H Nuclear Magnetic Resonance Spectroscopy Outperform Prostate Specific Antigen in Prostate Cancer Detection. J. Urol. 2006, 176, 2274–2279. [Google Scholar] [CrossRef]
- Etheridge, T.; Straus, J.; Ritter, M.A.; Jarrard, D.F.; Huang, W. Semen AMACR protein as a novel method for detecting prostate cancer. Urol. Oncol. 2018. [Google Scholar] [CrossRef]
- Serkova, N.J.; Gamito, E.J.; Jones, R.H.; O’Donnell, C.; Brown, J.L.; Green, S.; Sullivan, H.; Hedlund, T.; Crawford, E.D. The metabolites citrate, myo-inositol, and spermine are potential age-independent markers of prostate cancer in human expressed prostatic secretions. Prostate 2008, 68, 620–628. [Google Scholar] [CrossRef]
- Averna, T.; Kline, E.; Smith, A.; Sillerud, L. A decrease in 1h nuclear magnetic resonance spectroscopically determined citrate in human seminal fluid accompanies the development of prostate adenocarcinoma. J. Urol. 2005, 173, 433–438. [Google Scholar] [CrossRef]
- Roberts, M.J.; Richards, R.S.; Chow, C.W.K.; Buck, M.; Yaxley, J.; Lavin, M.F.; Schirra, H.J.; Gardiner, R.A. Seminal plasma enables selection and monitoring of active surveillance candidates using nuclear magnetic resonance-based metabolomics: A preliminary investigation. Prostate Int. 2017, 5, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Eidelman, E.; Twum-Ampofo, J.; Ansari, J.; Siddiqui, M.M. The Metabolic Phenotype of Prostate Cancer. Front. Oncol. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.; Araújo, A.; Pinto, J.; Jerónimo, C.; Henrique, R.; Bastos, M.; Carvalho, M.; Guedes de Pinho, P. GC-MS-Based Endometabolome Analysis Differentiates Prostate Cancer from Normal Prostate Cells. Metabolites 2018, 8, 23. [Google Scholar] [CrossRef] [PubMed]
- Giunchi, F.; Fiorentino, M.; Loda, M. The Metabolic Landscape of Prostate Cancer. Eur. Urol. Oncol. 2018, 2, 28–36. [Google Scholar] [CrossRef]
- Sadeghi, R.N.; Karami-Tehrani, F.; Salami, S. Targeting prostate cancer cell metabolism: Impact of hexokinase and CPT-1 enzymes. Tumour Biol. 2015, 36, 2893–2905. [Google Scholar] [CrossRef] [PubMed]
- Twum-Ampofo, J.; Fu, D.-X.; Passaniti, A.; Hussain, A.; Siddiqui, M.M. Metabolic targets for potential prostate cancer therapeutics. Curr. Opin. Oncol. 2016, 28, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Awwad, H.M.; Geisel, J.; Obeid, R. The role of choline in prostate cancer. Clin. Biochem. 2012, 45, 1548–1553. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, A.; Poisson, L.M.; Rajendiran, T.M.; Khan, A.P.; Cao, Q.; Yu, J.; Laxman, B.; Mehra, R.; Lonigro, R.J.; Li, Y.; et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 2009, 457, 910–914. [Google Scholar] [CrossRef] [PubMed]
- Giskeødegård, G.F.; Bertilsson, H.; Selnæs, K.M.; Wright, A.J.; Bathen, T.F.; Viset, T.; Halgunset, J.; Angelsen, A.; Gribbestad, I.S.; Tessem, M.-B. Spermine and Citrate as Metabolic Biomarkers for Assessing Prostate Cancer Aggressiveness. PLoS ONE 2013, 8, e62375. [Google Scholar] [CrossRef]
- Zabala-Letona, A.; Arruabarrena-Aristorena, A.; Martín-Martín, N.; Fernandez-Ruiz, S.; Sutherland, J.D.; Clasquin, M.; Tomas-Cortazar, J.; Jimenez, J.; Torres, I.; Quang, P.; et al. mTORC1-dependent AMD1 regulation sustains polyamine metabolism in prostate cancer. Nature 2017, 547, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Ni, J.; Beretov, J.; Cozzi, P.; Willcox, M.; Wasinger, V.; Walsh, B.; Graham, P.; Li, Y. Urinary biomarkers in prostate cancer detection and monitoring progression. Crit. Rev. Oncol. Hematol. 2017, 118, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Amobi, A.; Qian, F.; Lugade, A.A.; Odunsi, K. Tryptophan Catabolism and Cancer Immunotherapy Targeting IDO Mediated Immune Suppression. Adv. Exp. Med. Biol. 2017, 1036, 129–144. [Google Scholar] [PubMed]
- Santhanam, S.; Alvarado, D.M.; Ciorba, M.A. Therapeutic targeting of inflammation and tryptophan metabolism in colon and gastrointestinal cancer. Transl. Res. 2016, 167, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.P.; Rajendiran, T.M.; Bushra, A.; Asangani, I.A.; Athanikar, J.N.; Yocum, A.K.; Mehra, R.; Siddiqui, J.; Palapattu, G.; Wei, J.T.; et al. The Role of Sarcosine Metabolism in Prostate Cancer Progression. Neoplasia 2013, 15, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Ankerst, D.P.; Liss, M.; Zapata, D.; Hoefler, J.; Thompson, I.M.; Leach, R.J. A case control study of sarcosine as an early prostate cancer detection biomarker. BMC Urol. 2015, 15, 99. [Google Scholar] [CrossRef] [PubMed]
- Dereziński, P.; Klupczynska, A.; Sawicki, W.; Pałka, J.A.; Kokot, Z.J. Amino Acid Profiles of Serum and Urine in Search for Prostate Cancer Biomarkers: A Pilot Study. Int. J. Med. Sci. 2017, 14, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Locasale, J.W. Serine, glycine and one-carbon units: Cancer metabolism in full circle. Nat. Rev. Cancer 2013, 13, 572–583. [Google Scholar] [CrossRef] [PubMed]
- Koslowski, M.; Türeci, O.; Bell, C.; Krause, P.; Lehr, H.-A.; Brunner, J.; Seitz, G.; Nestle, F.O.; Huber, C.; Sahin, U. Multiple splice variants of lactate dehydrogenase C selectively expressed in human cancer. Cancer Res. 2002, 62, 6750–6755. [Google Scholar] [PubMed]
- Kong, L.; Du, W.; Cui, Z.; Wang, L.; Yang, Z.; Zhang, H.; Lin, D. Expression of lactate dehydrogenase C in MDA-MB-231 cells and its role in tumor invasion and migration. Mol. Med. Rep. 2016, 13, 3533–3538. [Google Scholar] [CrossRef]
- Merchant, M.L.; Rood, I.M.; Deegens, J.K.J.; Klein, J.B. Isolation and characterization of urinary extracellular vesicles: Implications for biomarker discovery. Nat. Rev. Nephrol. 2017, 13, 731–749. [Google Scholar] [CrossRef]
- Myers, J.S.; von Lersner, A.K.; Sang, Q.-X.A. Proteomic Upregulation of Fatty Acid Synthase and Fatty Acid Binding Protein 5 and Identification of Cancer- and Race-Specific Pathway Associations in Human Prostate Cancer Tissues. J. Cancer 2016, 7, 1452–1464. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Liu, W.-P.; Liu, X.-P.; Li, L.-Y.; Fang, Y.-Q.; Sun, Q.-P.; Liu, S.-J.; Li, M.-T.; Su, Z.-L.; Gao, X. Profiling protein markers associated with lymph node metastasis in prostate cancer by DIGE-based proteomics analysis. J. Proteome Res. 2010, 9, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liu, T.; Ma, C.; Xue, R.; Deng, C.; Zeng, H.; Shen, X. GC/MS-based metabolomic approach to validate the role of urinary sarcosine and target biomarkers for human prostate cancer by microwave-assisted derivatization. Anal. Bioanal. Chem. 2011, 401, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Kami, K.; Fujimori, T.; Sato, H.; Sato, M.; Yamamoto, H.; Ohashi, Y.; Sugiyama, N.; Ishihama, Y.; Onozuka, H.; Ochiai, A.; et al. Metabolomic profiling of lung and prostate tumor tissues by capillary electrophoresis time-of-flight mass spectrometry. Metabolomics 2013, 9, 444–453. [Google Scholar] [CrossRef] [PubMed]
- McDunn, J.E.; Li, Z.; Adam, K.-P.; Neri, B.P.; Wolfert, R.L.; Milburn, M.V.; Lotan, Y.; Wheeler, T.M. Metabolomic signatures of aggressive prostate cancer. Prostate 2013, 73, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Woda, B.A. Diagnostic utility of alpha-methylacyl CoA racemase (P504S) on prostate needle biopsy. Adv. Anat. Pathol. 2004, 11, 316–321. [Google Scholar] [CrossRef]
- Zhou, M.; Jiang, Z.; Epstein, J.I. Expression and diagnostic utility of alpha-methylacyl-CoA-racemase (P504S) in foamy gland and pseudohyperplastic prostate cancer. Am. J. Surg. Pathol. 2003, 27, 772–778. [Google Scholar] [CrossRef]
- Box, A.; Alshalalfa, M.; Hegazy, S.A.; Donnelly, B.; Bismar, T.A. High alpha-methylacyl-CoA racemase (AMACR) is associated with ERG expression and with adverse clinical outcome in patients with localized prostate cancer. Tumour Biol. 2016, 37, 12287–12299. [Google Scholar] [CrossRef]
- Alinezhad, S.; Väänänen, R.-M.; Ochoa, N.T.; Vertosick, E.A.; Bjartell, A.; Boström, P.J.; Taimen, P.; Pettersson, K. Global expression of AMACR transcripts predicts risk for prostate cancer—A systematic comparison of AMACR protein and mRNA expression in cancerous and noncancerous prostate. BMC Urol. 2016, 16, 10. [Google Scholar] [CrossRef]
- Mroz, A.; Kiedrowski, M.; Lewandowski, Z. α-Methylacyl-CoA racemase (AMACR) in gastric cancer: Correlation with clinicopathologic data and disease-free survival. Appl. Immunohistochem. Mol. Morphol. 2013, 21, 313–317. [Google Scholar] [CrossRef]
- Xu, B.; Cai, Z.; Zeng, Y.; Chen, L.; Du, X.; Huang, A.; Liu, X.; Liu, J. α-Methylacyl-CoA racemase (AMACR) serves as a prognostic biomarker for the early recurrence/metastasis of HCC. J. Clin. Pathol. 2014, 67, 974–979. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-E.; He, H.-L.; Lee, S.-W.; Chen, T.-J.; Chang, K.-Y.; Hsing, C.-H.; Li, C.-F. AMACR overexpression as a poor prognostic factor in patients with nasopharyngeal carcinoma. Tumour Biol. 2014, 35, 7983–7991. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, I.A.; Hennenlotter, J.; Stühler, V.; Kühs, U.; Scharpf, M.; Todenhöfer, T.; Stenzl, A.; Bedke, J. Transketolase like 1 (TKTL1) expression alterations in prostate cancer tumorigenesis. Urol. Oncol. 2018, 36, 472.e21–472.e27. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Yoneyama, T.; Hatakeyama, S.; Mikami, J.; Sato, T.; Mori, K.; Hashimoto, Y.; Koie, T.; Ohyama, C.; Fukuda, M.; et al. Detection of Core2 β-1,6-N-Acetylglucosaminyltransferase in Post-Digital Rectal Examination Urine Is a Reliable Indicator for Extracapsular Extension of Prostate Cancer. PLoS ONE 2015, 10, e0138520. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Yoneyama, T.; Tobisawa, Y.; Hatakeyama, S.; Yamamoto, H.; Kojima, Y.; Mikami, J.; Mori, K.; Hashimoto, Y.; Koie, T.; et al. Core 2 β-1, 6-N-acetylglucosaminyltransferase-1 expression in prostate biopsy specimen is an indicator of prostate cancer aggressiveness. Biochem. Biophys. Res. Commun. 2016, 470, 150–156. [Google Scholar] [CrossRef] [PubMed]
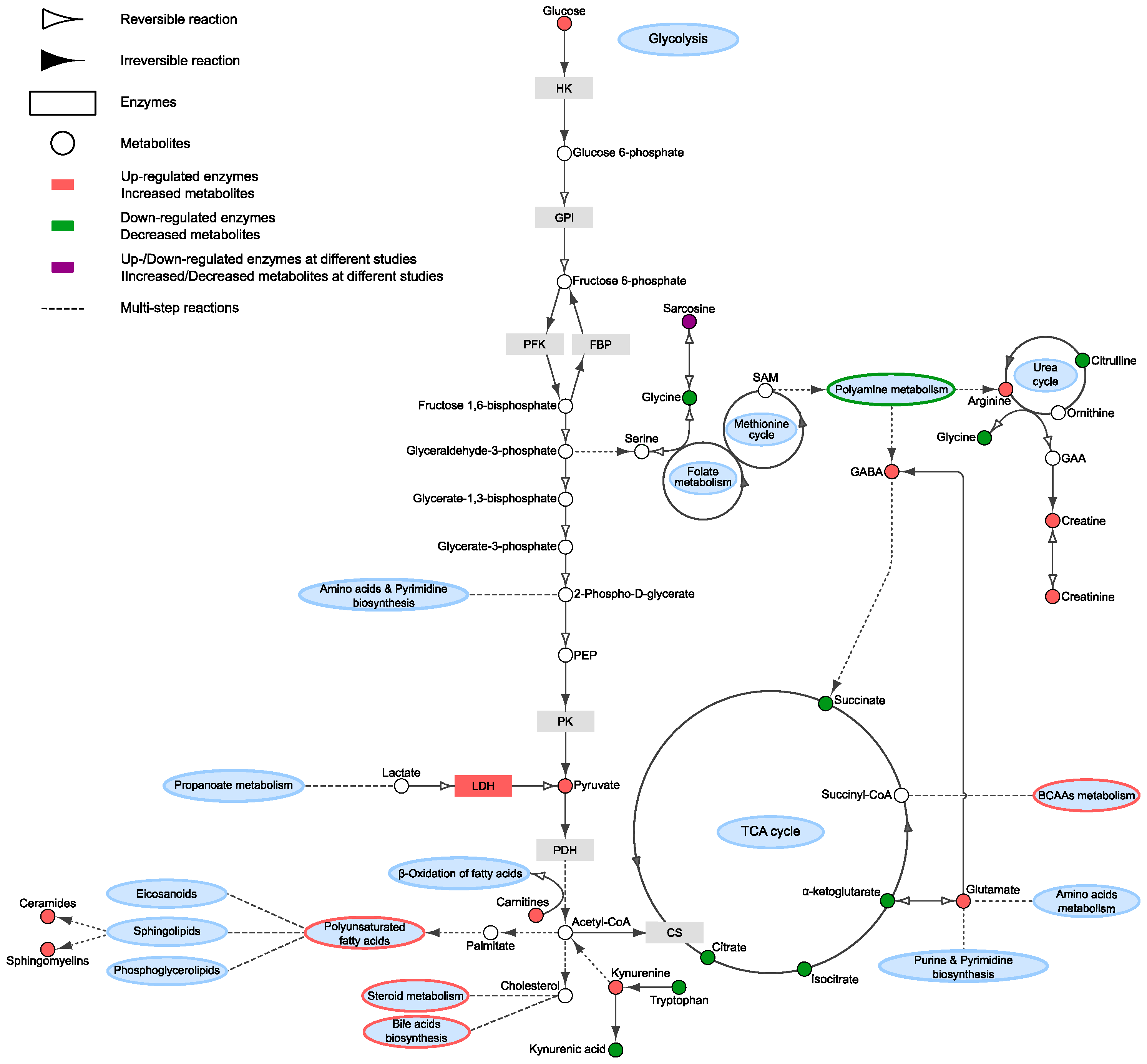
| Article | Sample | Experimental Approach | Research Aim | Sample Cohort | Main Findings | Validation Cohort |
|---|---|---|---|---|---|---|
| Clos-Garcia et al., 2018 [37] | Urine EVs | UHPLC-MS | Diagnosis | 31 × PCa; 14 × BPH | Statistically significant changes in 76 metabolites and 7 enzymes related to urea cycle, TCA cycle, and metabolism of steroid hormone biosynthesis, leukotriene, and prostaglandin, linoleate and purine, glycerophospholipid and tryptophan | No |
| Liang et al., 2017 [38] | Urine | FPLC-MS/MS | Diagnosis | 236 × PCa; 233 × HV | ↑ glycocholic acid; hippurate; chenodeoxycholic acid: PCa > HV ↓ taurocholic acid; 5-hydroxy-l-tryptophan: PCa < HV | No |
| Gkotsos et al., 2017 [39] | Urine | UPLC-MS/MS | Diagnosis | 52 × PCa, 49 × HV | ↓ kynurenic acid: PCa < HV | No |
| Struck-Lewicka et al., 2015 [40] | Urine | HPLC-TOF-MS; GC-QqQ-MS | Diagnosis | 32 × PCa; 32 × HV | Statistically significant changes in 82 metabolites related to amino acid, organic acids, sphingolipids, fatty acids, and carbohydrates metabolism | No |
| Fernández-Peralbo et al., 2016 [41] | Urine | LC-QTOF-MS/MS | Diagnosis | 43 × PCa; 29 × HV | ↑ 7-methylguanine: PCa > HV ↓ Statistically significant changes in 27 metabolites related to amino acid metabolism: PCa < HV | 19 × PCa; 13 × HV |
| Puhka et al., 2017 [42] | Urine EVs; Plasma EVs | UPLC-MS/MS | Diagnosis | 3 × PCa pre-prostatectomy; 3 × PCa post-prostatectomy; 3 × HV | ↓ glucoronate; isobutyryl-L-carnitine; D-Ribose-5-phosphate: pre- < post-prostatectomy and HV | No |
| Fujita et al., 2017 [43] | Urine EVs | iTRAQ; LC-MS/MS | Diagnosis and prognosis | 12 × PCa (6 × HG PCa; 6 × LG PCa); 6 × HV | ↑ FABP5: PCa > HV ↑ FABP5; GRN; AMBP; CHMP4A; CHMP4C associated with higher GS | 18 × PCa (6 × HG; 12 × LG); 11 × HV |
| Perez-Rambla et al., 2017 [44] | Urine | 1H-NMR | Diagnosis | 64 × PCa; 51 × BPH | ↑ BCAAs; glutamate; pseudouridine: PCa > BPH ↓ glycine; dimethylglycine; fumarate; 4-imidazole-acetate: PCa < BPH | No |
| Davalieva et al., 2015 [45] | Urine | 2D-DIGE-MS | Diagnosis | 8 × PCa; 16 × BPH | ↑ AMBP: PCa > BPH ↓ HP: PCa < BPH | 16 × PCa; 16 × BPH |
| Heger et al., 2015 [48] | Urine | 2D-DIGE; MALDI-TOF-MS | Diagnosis | 15 × HG PCa; 15 × LG PCa | ↑ CDK6; M2BP; LDHC: HG PCa > LG PCa | No |
| Kumar et al., 2016 [46] | Serum | 1H-NMR | Diagnosis | 75 × PCa; 70 × BPH; 65 x HV | ↑ alanine; sarcosine; creatine; creatinine: PCa > BPH and HV ↑ pyruvate; 3-methylhistidine; xanthine; hypoxanthine: BPH and PCa > HV ↓ glycine: PCa < HV ↓ citrate: PCa < BPH and HV | No |
| Kumar et al., 2015 [47] | Serum | 1H-NMR | Diagnosis and prognosis | 21 × HG PCa; 28 × LG PCa; 22 × HV | ↑ alanine; sarcosine: LG PCa > HG PCa and HV ↑ pyruvate: LG PCa and HG PCa > HV ↓ glycine: LG PCa and HG PCa < HV | 9 × HG PCa; 12 × LG PCa; 12 × HV |
| Giskeødegård et al., 2015 [49] | Plasma/Serum | 1H-NMR; UPLC-MS/MS; GC-MS | Diagnosis | 29 × PCa; 21 × BPH | ↑ decanoylcarnitine (c10); tetradecenoylcarnitine (c14:1); octanoyl-carnitine (c8); dimethylsulfone; phenylalanine; lysine: PCa > BPH ↓ phosphatidylcholine diacyl (c34:4); lipid -(CH2)n-CH2-CH2-CO: PCa < BPH | No |
| Zhao et al., 2017 [50] | Plasma | UPLC-MS/MS | Diagnosis | 32 × PCa; 32 × HV | Statistically significant changes in 19 metabolites related to amino acid, nucleotide, butanoate and propionate metabolism | No |
| Lin et al., 2017 [51] | Plasma | LC-MS/MS | Prognosis | 96 × CRPC | ↑ ceramide d18:1/24:1; sphingomyelin d18:2/16:0; phosphatidylcholine 16:0/16:0 correlated with shorter overall survival | 63 × CRPC |
| Mondul et al., 2015 [52] | Serum | UHPLC-MS; GC-MS | PCa risk Prognosis | 100 × HG PCa; 100 × LG PCa; 200 × HV | Statistically significant changes in 22 metabolites related to lipid and amino acid metabolism associated with overall PCa risk Statistically significant changes in 14 metabolites related to TCA cycle and lipid metabolism associated with HG PCa Statistically significant changes in 34 metabolites related to lipid, amino acid and nucleotide metabolism associated with LG PCa | No |
| Kühn et al., 2016 [53] | Plasma | LC-MS/MS; FIA-MS/MS | PCa risk | 310 × PCa; 774 × HV | ↑ Phosphatidylcholine (PC) associated with higher risk of PCa ↑ lysoPC associated with lower risk of PCa | No |
| Schmidt et al., 2017 [54] | Plasma | QqQ-MS | PCa risk Prognosis | 1077 × PCa; 1077 × HV 208 × advanced PCa; 456 × localized PCa | Statistically significant changes in 14 metabolites related to lipid and amino acid metabolism associated with overall PCa risk 12 glycerophospholipids inversely associated with risk of advanced PCa | No No |
| Huang et al., 2017 [55] | Serum | UHPLC-MS; GC-MS | PCa risk | 71 × PCa T2 stage; 51 × PCa T3 stage; 15 × PCa T4 stage; 200 × HV | Statistically significant changes in 8 metabolites related to histidine and uridine metabolism associated with PCa T2 risk. Statistically significant changes in 12 metabolites related to fatty acid and primary bile acid metabolism associated with PCa T3 risk Statistically significant changes in 16 metabolites related to TCA, BCAA secondary bile acid, sex steroids and histamine metabolism associated with PCa T4 risk. | No |
| Andras et al., 2017 [56] | Serum | HPLC-ESI-QTOF-MS | Prediction | 59 × patients with high PSA levels | 6 metabolites involved in lipid, purine and tryptophan metabolism predictive for prostate biopsy outcome | 31 × patients with high PSA levels |
| Kline et al., 2006 [57] | Seminal fluid; Prostatic secretion | 1H-NMR | Diagnosis | 28 × PCa; 33 × HV | ↓ citrate: PCa < HV | No |
| Etheridge et al., 2018 [58] | Seminal fluid | ELISA | Diagnosis | 28 × PCa; 15 × HV | ↑ AMACR: PCa > HV | No |
| Serkova et al., 2008 [59] | Prostatic secretion | 1H-NMR | Prediction PCa risk | 52 × PCa; 26 × HV | ↓ citrate; myo-inositol; spermine shown highly predictive of PCa and inversely associated with PCa risk | No |
| Averna et al., 2005 [60] | Seminal fluid | 1H-NMR | Diagnosis | 3 × PCa; 1 × BPH; 4 × HV | ↓ citrate: PCa < BPH | No |
| Roberts et al., 2017 [61] | Seminal fluid | 1H-NMR | Prediction | 98 × PCa; 53 × HV | Statistically significant changes in choline, valine and leucine, | No |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Cebrián, N.; Rojas-Benedicto, A.; Albors-Vaquer, A.; López-Guerrero, J.A.; Pineda-Lucena, A.; Puchades-Carrasco, L. Metabolomics Contributions to the Discovery of Prostate Cancer Biomarkers. Metabolites 2019, 9, 48. https://doi.org/10.3390/metabo9030048
Gómez-Cebrián N, Rojas-Benedicto A, Albors-Vaquer A, López-Guerrero JA, Pineda-Lucena A, Puchades-Carrasco L. Metabolomics Contributions to the Discovery of Prostate Cancer Biomarkers. Metabolites. 2019; 9(3):48. https://doi.org/10.3390/metabo9030048
Chicago/Turabian StyleGómez-Cebrián, Nuria, Ayelén Rojas-Benedicto, Arturo Albors-Vaquer, José Antonio López-Guerrero, Antonio Pineda-Lucena, and Leonor Puchades-Carrasco. 2019. "Metabolomics Contributions to the Discovery of Prostate Cancer Biomarkers" Metabolites 9, no. 3: 48. https://doi.org/10.3390/metabo9030048
APA StyleGómez-Cebrián, N., Rojas-Benedicto, A., Albors-Vaquer, A., López-Guerrero, J. A., Pineda-Lucena, A., & Puchades-Carrasco, L. (2019). Metabolomics Contributions to the Discovery of Prostate Cancer Biomarkers. Metabolites, 9(3), 48. https://doi.org/10.3390/metabo9030048






