Metabolic Fingerprint of Chronic Obstructive Lung Diseases: A New Diagnostic Perspective
Abstract
1. Introduction
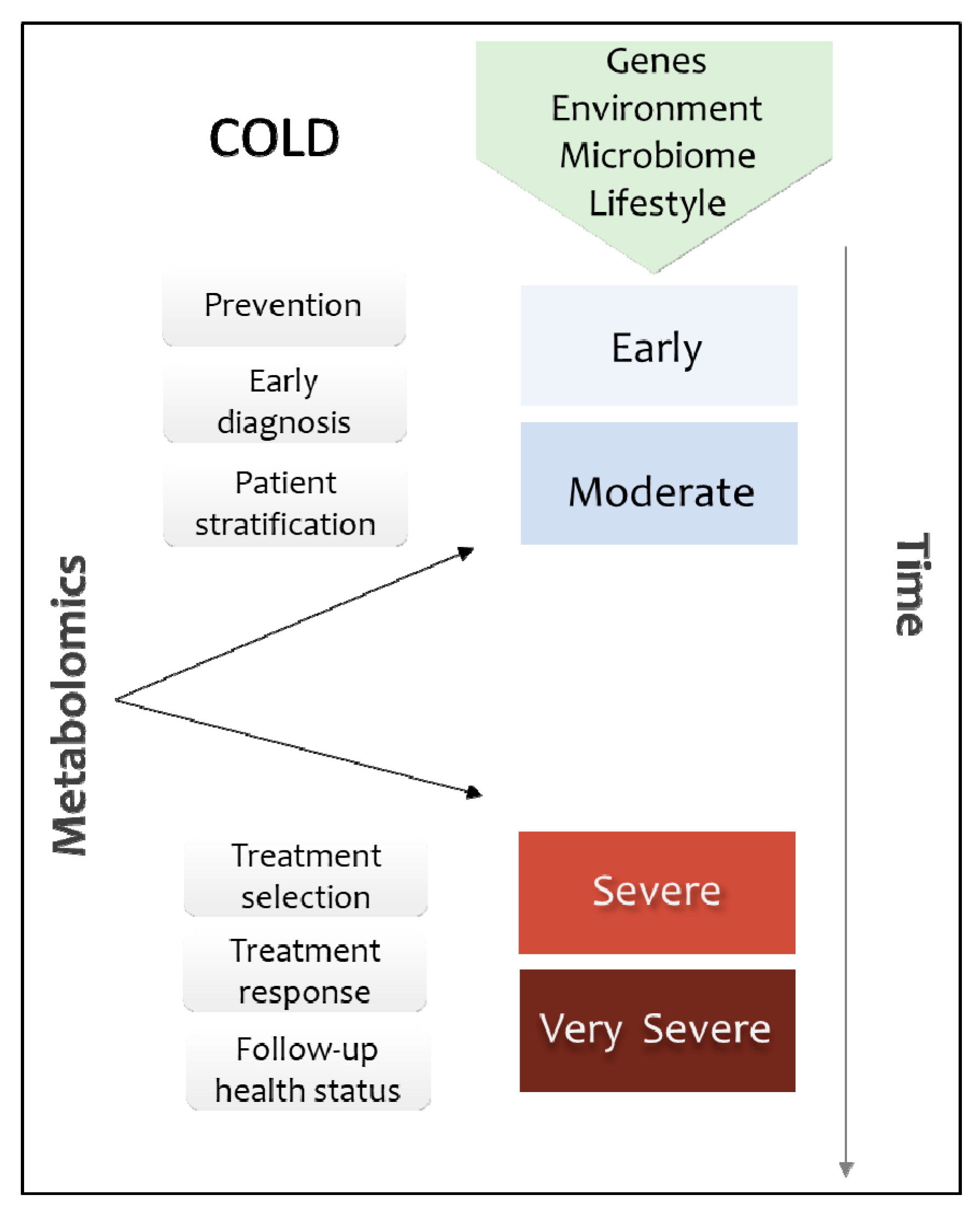
2. The Diagnostic Value of Metabolome in COLD
2.1. COLD Versus Healthy
2.2. COLD Classification Based onGenetic and Environmental Factors
3. Matching the Right COLD Treatment to the Right Patient
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ito, S.; Ishimori, K.; Ishikawa, S. Effects of repeated cigarette smoke extract exposure over one month on human bronchial epithelial organotypic culture. Toxicol. Rep. 2018, 5, 864–870. [Google Scholar] [CrossRef]
- Ungureanu, A.; Zlatian, O.; Mitroi, G.; Drocaş, A.; Ţîrcã, T.; Cǎlina, D.; Dehelean, C.; Docea, A.O.; Izotov, B.N.; Rakitskii, V.N.; et al. Staphylococcus aureus colonisation in patients from a primary regional hospital. Mol. Med. Rep. 2017, 16, 8771–8780. [Google Scholar] [CrossRef]
- Zlatian, O.; Balasoiu, A.T.; Balasoiu, M.; Cristea, O.; Docea, A.O.; Mitrut, R.; Spandidos, D.A.; Tsatsakis, A.M.; Bancescu, G.; Calina, D. Antimicrobial resistance in bacterial pathogens among hospitalised patients with severe invasive infections. Exp. Ther. Med. 2018, 16, 4499–4510. [Google Scholar] [CrossRef]
- Calina, D.; Docea, A.O.; Rosu, L.; Zlatian, O.; Rosu, A.F.; Anghelina, F.; Rogoveanu, O.; Arsene, A.L.; Nicolae, A.C.; Dragoi, C.M.; et al. Antimicrobial resistance development following surgical site infections. Mol. Med. Rep. 2017, 15, 681–688. [Google Scholar] [CrossRef] [PubMed]
- Călina, D.; Roșu, L.; Roșu, A.F.; Ianoşi, G.; Ianoşi, S.; Zlatian, O.; Mitruț, R.; Docea, A.O.; Rogoveanu, O.; Mitruț, P.; et al. Etiological diagnosis and pharmacotherapeutic management of parapneumonic pleurisy. Farmacia 2016, 64, 946–952. [Google Scholar]
- Huangfu, Q.; Li, M.; Xiao, L.; Tao, H.; Wang, W.; Fei, X. Effect of inhaled glucocorticoids on chronic obstructive pulmonary disease in male patients with osteoporosis. Farmacia 2018, 66, 70–77. [Google Scholar]
- Diaz-Guzman, E.; Mannino, D.M. Epidemiology and prevalence of chronic obstructive pulmonary disease. Clin. Chest Med. 2014, 35, 7–16. [Google Scholar] [CrossRef]
- Han, M.K.; Agusti, A.; Calverley, P.M.; Celli, B.R.; Criner, G.; Curtis, J.L.; Fabbri, L.M.; Goldin, J.G.; Jones, P.W.; MacNee, W.; et al. Chronic Obstructive Pulmonary Disease Phenotypes. The Future of COPD. Am. J. Respir. Crit. Care Med. 2010, 182, 598–604. [Google Scholar] [CrossRef]
- Lange, P.; Halpin, D.M.; O’Donnell, D.E.; MacNee, W. Diagnosis, assessment, and phenotyping of COPD: beyond FEV1. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11 Spec Is, 3–12. [Google Scholar]
- Brightling, C.; Greening, N. Airway inflammation in COPD- progress to precision medicine. Eur. Respir. J. 2019, 1900651. [Google Scholar] [CrossRef]
- Ran, N.; Pang, Z.; Gu, Y.; Pan, H.; Zuo, X.; Guan, X.; Yuan, Y. An Updated Overview of Metabolomic Profile Changes in Chronic Obstructive Pulmonary Disease. Metabolites 2019, 9, 111. [Google Scholar] [CrossRef]
- Singh, B.; Jana, S.K.; Ghosh, N.; Das, S.K.; Joshi, M.; Bhattacharyya, P.; Chaudhury, K. Metabolomic profiling of doxycycline treatment in chronic obstructive pulmonary disease. J. Pharm. Biomed. Anal. 2017, 132, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Sidhaye, V.K.; Nishida, K.; Martinez, F.J. Precision medicine in COPD: where are we and where do we need to go? Eur. Respir. Rev. 2018, 27, 180022. [Google Scholar] [CrossRef] [PubMed]
- Ubhi, B.K.; Riley, J.H.; Shaw, P.A.; Lomas, D.A.; Tal-Singers, R.; MacNeef, W.; Griffin, J.L.; Connor, S.C. Metabolic profiling detects biomarkers of protein degradation in COPD patients. Eur. Respir. J. 2012, 40, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Ubhi, B.K.; Cheng, K.K.; Dong, J.; Janowitz, T.; Jodrell, D.; Tal-Singer, R.; MacNee, W.; Lomas, D.A.; Riley, J.H.; Griffin, J.L.; et al. Targeted metabolomics identifies perturbations in amino acid metabolism that sub-classify patients with COPD. Mol. Biosyst. 2012, 8, 3125–3133. [Google Scholar] [CrossRef] [PubMed]
- Kilk, K.; Aug, A.; Ottas, A.; Soomets, U.; Altraja, S.; Altraja, A. Phenotyping of chronic obstructive pulmonary disease based on the integration of metabolomes and clinical characteristics. Int. J. Mol. Sci. 2018, 19, 666. [Google Scholar] [CrossRef]
- Novotna, B.; Abdel-Hamid, M.; Koblizek, V.; Svoboda, M.; Hejduk, K.; Rehacek, V.; Bis, J.; Salajka, F. A pilot data analysis of a metabolomic HPLC-MS/MS study of patients with COPD. Adv. Clin. Exp. Med. 2018, 27, 531–539. [Google Scholar] [CrossRef]
- Wang, L.; Tang, Y.; Liu, S.; Mao, S.; Ling, Y.; Liu, D.; He, X.; Wang, X. Metabonomic Profiling of Serum and Urine by 1 H NMR-Based Spectroscopy Discriminates Patients with Chronic Obstructive Pulmonary Disease and Healthy Individuals. PLoS ONE 2013, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Deeb, R.S.; Ma, Y.; Staudt, M.R.; Crystal, R.G.; Gross, S.S. Serum metabolite biomarkers discriminate healthy smokers from COPD smokers. PLoS ONE 2015, 10, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Naz, S.; Kolmert, J.; Yang, M.; Reinke, S.N.; Kamleh, M.A.; Snowden, S.; Heyder, T.; Levänen, B.; Erle, D.J.; Sköld, C.M.; et al. Metabolomics analysis identifies sex-associated metabotypes of oxidative stress and the autotaxin-lysoPA axis in COPD. Eur. Respir. J. 2017, 49, 1602322. [Google Scholar] [CrossRef]
- De Benedetto, F.; Pastorelli, R.; Ferrario, M.; de Blasio, F.; Marinari, S.; Brunelli, L.; Wouters, E.F.M.; Polverino, F.; Celli, B.R. Supplementation with Qter ® and Creatine improves functional performance in COPD patients on long term oxygen therapy. Respir. Med. 2018, 142, 86–93. [Google Scholar] [CrossRef]
- Rodríguez, D.A.; Alcarraz-Vizán, G.; Díaz-Moralli, S.; Reed, M.; Gómez, F.P.; Falciani, F.; Günther, U.; Roca, J.; Cascante, M. Plasma metabolic profile in COPD patients: Effects of exercise and endurance training. Metabolomics 2012, 8, 508–516. [Google Scholar] [CrossRef]
- Hodgson, S.; Griffin, T.J.; Reilly, C.; Harvey, S.; Witthuhn, B.A.; Sandri, B.J.; Kunisaki, K.M.; Wendt, C.H. Plasma sphingolipids in HIV-associated chronic obstructive pulmonary disease. BMJ Open Respir. Res. 2017, 4, 1–8. [Google Scholar] [CrossRef]
- Fortis, S.; Lusczek, E.R.; Weinert, C.R.; Beilman, G.J. Metabolomics in COPD Acute Respiratory Failure Requiring Noninvasive Positive Pressure Ventilation. Can. Respir. J. 2017, 2017, 1–9. [Google Scholar] [CrossRef]
- Tan, L.C.; Yang, W.J.; Fu, W.P.; Su, P.; Shu, J.K.; Dai, L.M. 1 H-NMR-based metabolic profiling of healthy individuals and high-resolution CT-classified phenotypes of COPD with treatment of tiotropium bromide. Int. J. COPD 2018, 13, 2985–2997. [Google Scholar] [CrossRef]
- Yoneda, T.; Yoshikawa, M.; Fu, A.; Tsukaguchi, K.; Okamoto, Y.; Takenaka, H. Plasma levels of amino acids and hypermetabolism in patients with chronic obstructive pulmonary disease. Nutrition 2001, 17, 95–99. [Google Scholar] [CrossRef]
- Engelen, M.P.K.J.; Schols, A.M.W.J.; Does, J.D.; Gosker, H.R.; Deutz, N.E.P.; Wouters, E.F.M. Exercise-induced lactate increase in relation to muscle substrates in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2000, 162, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Airoldi, C.; Ciaramelli, C.; Fumagalli, M.; Bussei, R.; Mazzoni, V.; Viglio, S.; Iadarola, P.; Stolk, J. 1 H NMR to Explore the Metabolome of Exhaled Breath Condensate in α 1 -Antitrypsin Deficient Patients: A Pilot Study. J. Proteome Res. 2016, 15, 4569–4578. [Google Scholar] [CrossRef] [PubMed]
- Ribbenstedt, A.; Ziarrusta, H.; Benskin, J.P. Development, characterization and comparisons of targeted and non-targeted metabolomics methods. PLoS ONE 2018, 13, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.D.; Souza, A.L.; Gerszten, R.E.; Clish, C.B. Targeted metabolomics. Curr. Protoc. Mol. Biol. 2012, 98, 30–32. [Google Scholar] [CrossRef]
- Gertsman, I.; Barshop, B.A. Promises and Pitfalls of Untargeted Metabolomics. J. Inherit. Metab. Dis. 2018, 41, 355–366. [Google Scholar] [CrossRef]
- Wang, C.; Li, J.X.; Tang, D.; Zhang, J.Q.; Fang, L.Z.; Fu, W.P.; Liu, L.; Dai, L.M. Metabolic changes of different high-resolution computed tomography phenotypes of COPD after budesonide-formoterol treatment. Int. J. COPD 2017, 12, 3511–3521. [Google Scholar] [CrossRef][Green Version]
- De Rosa, V.; La Cava, A.; Matarese, G. Metabolic pressure and the breach of immunological self-tolerance. Nat. Immunol. 2017, 18, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Clish, C.B. Metabolomics: an emerging but powerful tool for precision medicine. Mol. Case Stud. 2015, 1, a000588. [Google Scholar] [CrossRef] [PubMed]
- Remels, A.H.V.; Gosker, H.R.; Langen, R.C.J.; Schols, A.M.W.J. The mechanisms of cachexia underlying muscle dysfunction in COPD. J. Appl. Physiol. 2013, 114, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Schols, A.M.W.J.; Fredrix, E.W.H.M.; Soeters, P.B.; Westerterp, K.R.; Wouters, E.F.M. Resting energy expenditure in patients with chronic obstructive pulmonary disease. Am. J. Clin. Nutr. 1991, 54, 983–987. [Google Scholar] [CrossRef] [PubMed]
- Langen, R.C.J.; Gosker, H.R.; Remels, A.H.V.; Schols, A.M.W.J. Triggers and mechanisms of skeletal muscle wasting in chronic obstructive pulmonary disease. Int. J. Biochem. Cell Biol. 2013, 45, 2245–2256. [Google Scholar] [CrossRef]
- Marchand, C.R.; Farshidfar, F.; Rattner, J.; Bathe, O.F. A Framework for Development of Useful Metabolomic Biomarkers and Their Effective Knowledge Translation. Metabolites 2018, 8, 59. [Google Scholar] [CrossRef]
- Kohler, I.; Hankemeier, T.; van der Graaf, P.H.; Knibbe, C.A.J.; van Hasselt, J.G.C. Integrating clinical metabolomics-based biomarker discovery and clinical pharmacology to enable precision medicine. Eur. J. Pharm. Sci. 2017, 109, S15–S21. [Google Scholar] [CrossRef]
- Wendt, C.H.; Nelsestuen, G.; Harvey, S.; Gulcev, M.; Stone, M.; Reilly, C. Peptides in bronchoalveolar lavage in chronic obstructive pulmonary disease. PLoS ONE 2016, 11, 1–11. [Google Scholar] [CrossRef]
- Holmes, E.; Wilson, I.D.; Nicholson, J.K. Metabolic Phenotyping in Health and Disease. Cell 2008, 134, 714–717. [Google Scholar] [CrossRef] [PubMed]
- Baum, S.J. A Survey of Internists and Cardiologists: Are Discoveries in Fatty Acids Truly being translated into Clinical Practice? Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Tsoukalas, D.; Alegakis, A.; Fragkiadaki, P.; Papakonstantinou, E.; Nikitovic, D.; Karataraki, A.; Nosyrev, A.E.; Papadakis, E.G.; Spandidos, D.A.; Drakoulis, N.; et al. Application of metabolomics: Focus on the quantification of organic acids in healthy adults. Int. J. Mol. Med. 2017, 40, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Tsoukalas, D.; Alegakis, A.K.; Fragkiadaki, P.; Papakonstantinou, E.; Tsilimidos, G.; Geraci, F.; Sarandi, E.; Nikitovic, D.; Spandidos, D.A.; Tsatsakis, A. Application of metabolomics part II: Focus on fatty acids and their metabolites in healthy adults. Int. J. Mol. Med. 2019, 43, 233–242. [Google Scholar] [CrossRef]
- Tsoukalas, D.; Fragoulakis, V.; Sarandi, E.; Docea, A.O.; Papakonstantinou, E.; Tsilimidos, G.; Anamaterou, C.; Fragkiadaki, P.; Aschner, M.; Tsatsakis, A.; et al. Targeted Metabolomic Analysis of Serum Fatty Acids for the Prediction of Autoimmune Diseases. Front. Mol. Biosci. 2019, 6, 1–14. [Google Scholar] [CrossRef]
- Dias, D.A.; Jones, O.A.H.; Beale, D.J.; Boughton, B.A.; Benheim, D.; Kouremenos, K.A.; Wolfender, J.L.; Wishart, D.S. Current and future perspectives on the structural identification of small molecules in biological systems. Metabolites 2016, 6, 46. [Google Scholar] [CrossRef]
- van Rijswijk, M.; Beirnaert, C.; Caron, C.; Cascante, M.; Dominguez, V.; Dunn, W.B.; Ebbels, T.M.D.; Giacomoni, F.; Gonzalez-beltran, A.; Hankemeier, T.; et al. The future of metabolomics in ELIXIR. F1000 Res. 2017, 6, 1–17. [Google Scholar] [CrossRef]
- Sanchon-Lopez, B.; Everett, J.R. New Methodology for Known Metabolite Identification in Metabonomics/Metabolomics: Topological Metabolite Identification Carbon Efficiency (tMICE). J. Proteome Res. 2016, 15, 3405–3419. [Google Scholar] [CrossRef]
- Clayton, T.A.; Lindon, J.C.; Cloarec, O.; Antti, H.; Charuel, C.; Hanton, G.; Provost, J.P.; Le Net, J.L.; Baker, D.; Walley, R.J.; et al. Pharmaco-metabonomic phenotyping and personalized drug treatment. Nature 2006, 440, 1073–1077. [Google Scholar] [CrossRef]
- Engelen MP, S.A. Altered amino acid metabolism in chronic obstructive pulmonary disease: new therapeutic perspective? Curr Opin Clin Nutr Metab Care 2003, 6, 73–78. [Google Scholar] [CrossRef]
- Papamichael, M.M.; Shrestha, S.K.; Itsiopoulos, C.; Erbas, B. The role of fish intake on asthma in children: A meta-analysis of observational studies. Pediatric Allergy Immunol. 2018, 29, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, M.; Katsardis, C.; Lamber, K.; Tsoukalas, D.; Koutsilieris, M.; Erbas, B.; Itsiopoulos, C. Efficacy of a Mediterranean diet supplemented with fatty fish in ameliorating inflammation in paediatric asthma: A randomised controlled trial. J. Hum. Nutr. Diet. 2018, 24, 184. [Google Scholar] [CrossRef] [PubMed]
- Hornikx, M.; Van Remoortel, H.; Lehouck, A.; Mathieu, C.; Maes, K.; Gayan-Ramirez, G.; Decramer, M.; Troosters, T.; Janssens, W. Vitamin D supplementation during rehabilitation in COPD: a secondary analysis of a randomized trial. Respir. Res. 2012, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Bellocchia, M.; Boita, M.; Patrucco, F.; Ferrero, C.; Verri, G.; Libertucci, D.; Coni, F.; Tabbia, G.; Mattei, A.; Bucca, C. Vitamin D deficiency and COPD exacerbations: Effect of vitamin D supplementation. Eur. Respir. J. 2015, 46, PA3961. [Google Scholar]
- Hotamislikil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
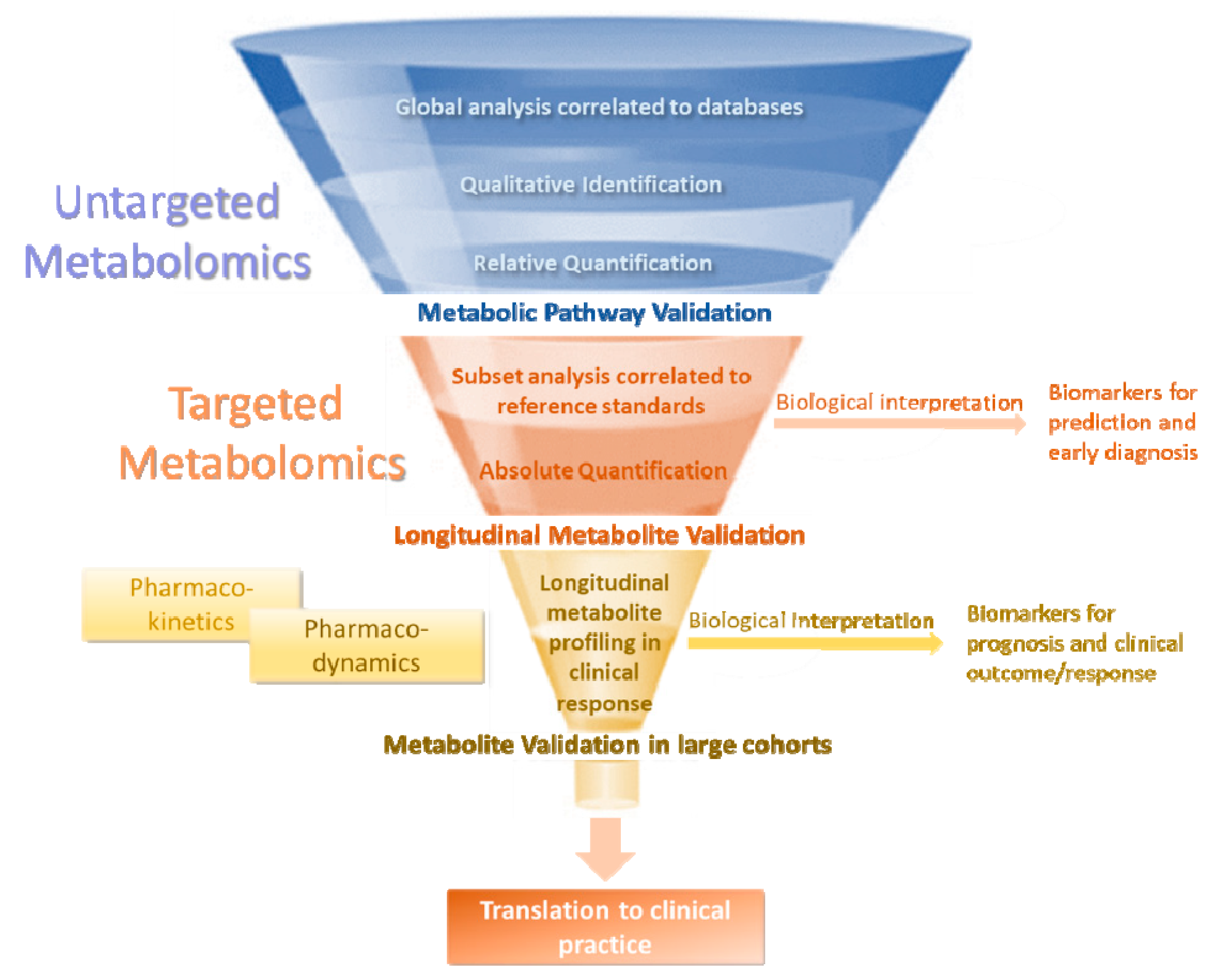
| Study | Subjects | Criteria | Sample | Method | Metabolites | Confounders |
|---|---|---|---|---|---|---|
| Ubhi et al., [14] | Control: n = 66: non-smokers: n = 15 (8M/7F), Age: 61, BMI:27.7 smokers: n = 53, (34M/19F), Age: 57, BMI: 28.6 Patients: n =163 GOLD II: n =69, (46M/23F), Age: 65, BMI:27.9 GOLD III: n = 63 (43M/20F), Age: 64, BMI: 26.9 GOLD IV: n = 31, (18M/13F), Age: 63, BMI: 25.8 | Controls and COLD patients were matched for sex, age, and smoking history | Serum | Untargeted NMR/LC-MS/MS | glutamine, phenylalanine, creatine, glycine, methionine, glycerol, monoglyceride, trimethylamine BCAA degradation: isobutyrate, 3-hydroxyisobutyrate, isoleucine, leucine, valine Lipid metabolism: HDL,LDL/VLDL, monoglyceride, glycerol Ketone bodies: acetoacetate, ascorbate, 3-hydroxybutyrate | Analysis based on GOLD stage, cachexia, emphysema, diabetes, patient location, age, sex, and comorbidities |
| Ubhi et al., [15] | Control: n =30 (30 M), Age:57, BMI: 29.9 Patients: n = 30 GOLD IV (30 M), Age:65, BMI: 26.2 | Inclusion Control: aged 40–75, current or ex-smokers with >10 pack–year history, postbronchodilator FEV1 < 80% of predicted normal and FEV1/FVC ratio < 0.7. Patients: smoking (≥ 10 pack–years) and non-smoking (<1 pack–year). Control subjects: aged 40–75 years with normal lung function (post-bronchodilator FEV1>85% predicted and FEV1/FVC >0.7). | Serum | Targeted LC-MS/MS | glutamine, arginine, aspartate, aminoadipic acid, proline, leucine, valine, isoleucine, g-aminobutyric acid, a-aminobutyric acid, 4-hydroxyproline | Aminoacids profile analysis based on weight, BMI, age, and sex |
| Kilk et al., 2018 [16] | Control: n =21 (9M/12F), Age: 37, BMI: 24 Patients: n =25 (25M), Age: 67, BMI: 26 | De novo phenotyping according to characteristics, medication, and co-morbidities pulmonary function | Blood/ EBC | Untargeted HPLC-MS | carnitine, glutamine, histidine, lysine, kynurenine, putrescine, lysoPC | Analysis based on clinical parameters and metabolomics |
| Novotna et al., 2018 [17] | Control: n = 10 (5M/5F), Age: 61.5, BMI: 25.3 Patients: n = 10 (5M/5F), Age:55, BMI: 27.1 | Inclusion: Patients: non-smokers or ex-smokers >6 months, patients without acute exposition to carbon monoxide, COLD patients with post-bronchodilator values FEV1 < 60%. Exclusion: current smokers or ex-smokers <6 months, with a known metabolic disease or kidney disease, or presence of coronary artery disease. | Blood | Untargeted HPLC-MS/MS | carnitine, phenylalanine, tyrosine, carnitine/ acycarnitine, valine, methionine, glycine, leucine, isoleucine, | Analysis of different metabolic profiles based on age, sex, and BMI |
| Wang et al., 2013 [18] | Patients Phenotype E: n =22 (20M/2F), Age: 73.64, BMI: 21.21 Phenotype M: n =28 (25M/3F), Age:70.18, BMI: 19.65 | Exclusion: respiratory tract infection, exacerbation of an airway disease in the previous 3 weeks, associated respiratory diseases, serious cardiovascular disease, cancer, cognitive impairment, immunodeficiency, or unable to complete protocol+D10 | Serum | Untargeted NMR | ADP, guanosine, tyrosine, uridine, maltose, sucrose, L-threonine, D-glucose, glycine, proline, betaine, choline, malonate, L-lysine, creatine, asparagine, aspartate, succinate, pyruvic acid, acetone, ornithine, L-alanine, lactate, isopropyl alcohol, L-valine, leucine | No information provided |
| Chen et al., 2015 [19] | Control: Non-smokers: n =37 (19M/18F), Age: 39.5, BMI: 26.6 Smokers: n =40 (35M/5F), Age: 41.8, BMI: 26.9 Patients Smokers: n =41 (38M/3F), Age: 53.2, BMI: 25.6 | Exclusion: non-smokers with no prior exposure to cigarette smoking and no detectable nicotine metabolites | Serum | Untargeted LC-MS | cotinine, 3-hydroxycotinin, Quinic acid, glycochenodeoxycholic acid 3-glucuronide, cysteinsulfonic acid, glycerophosphoinositol, phosphatidylinositol, creatinine, myoinositol, fibrinogen peptide B, hydrophobic unknowns | Analysis based on smoking status and clinical lung function parameters |
| Naz et al., 2017 [20] | Control: Non-smokers: n =38 (20M/18F), Age M: 62, Age F: 55.5, BMI M: 25.6, BMI F: 26.5 Smokers: n =40 (20M/20F), Age M: 52.5, Age F: 54, BMI M: 25, BM1 F: 24.2 Patients: Smokers: n =27 (15M/12F), Age M: 61, Age F: 59, BM1 M: 24.2, BMI F: 23.5 Ex-smokers: n =11 (5 M/6 F), Age M: 64, Age F: 58, BMI M: 29.1, BMI F: 27.6 | Inclusion: Patients: no allergy or asthma history, no use of inhaled or oral corticosteroids, and no exacerbations for at least 3 months prior to study COLD patients and smokers matched for smoking history and current smoking habits | Serum | Untargeted LC-MS | Both sexes: citrate cycle, glycerophospholipid metabolism, pyruvate metabolism Sex-enhanced - female COLD: Fatty acid biosynthesis, sphingolipid metabolism Sex-enhanced - male COLD: cAMP signaling pathway, retrograde endocannabinoid signaling, tryptophan metabolism | Sex-specific metabolomic analysis |
| De Benedetto et al., 2018 [21] | Patients: Active Coenzyme Q10(QTer) n =45(34M/11F), Age: 73, BMI: 31.2 Placebo: n =45 (34M/11F), Age: 73, BMI: 29.6 | Inclusion: clinically stable, no COLD exacerbation or hospitalization 4 weeks prior to enrolment, or receiving bronchodilator treatment Exclusion: mechanical ventilation, uncontrolled diabetes mellitus, severe heart, renal or hepatic failure and current or pre-existing malignant disease within the 3 years, persistent infections, chronic oral steroid or immunosuppressive therapy, or inability to complete tests and use of statins or amino acid supplements | Plasma | Untargeted LC-MS | lysophosphatidyicholine, phosphatidylcholine, sphingomyelins | No information provided |
| Rodríguez et al., 2012 [22] | Controls: n =12 (10M/2F), Age: 65, BMI: 26 Patients: n =18 (17M/1F), Age: 68, BMI: 24 | Inclusion: no COLD exacerbations, no oral steroid treatment in the previous 4 months, all on bronchodilators and inhaled corticosteroids, and no major co-morbidities | Plasma | Untargeted NMR | glutamine, tyrosine, alanine, valine, isoleucine, creatine, creatinine, citrate, glucose, lactate, succinate, pyruvate | No information provided |
| Hodgson et al., 2017 [23] | HIV(+)COLD(+): n =38 (27M/11F), Age: 38.97 HIV(+)COLD(-): n=40(29M/11F) Age: 38.93 HIV(-)COLD(+): n =20 (18M/2F) Age: 48.18 HIV(-) COLD(-): n =17 (15M/2F), Age: 55.91 | Inclusion HIV-positive controls: normal lung function, matched on age, sex, region, and smoking status HIV-negative controls: from the COPDGene study, matched on lung function, age, sex, and race | Plasma | Untargeted LC-MS/MS | ceramide, fatty acids, diacyglycerol, kynurenine/tryptophan ratio | HIV-associated metabolomic analysis |
| Fortis et al., 2017 [24] | Stable COLD: n =15 (6M/9F), Age: 68, BMI: 29.25 AECOLD: n =12 (4M/8F), Age: 73.1, BMI: 28.8 CHF: n =8 (3M/5F), Age: 78.5, BMI: 29.1 PNA: n =9 (6M/ 3F), Age: 65.7, BMI: 29.8 | Inclusion Stable COLD: COLD diagnosis, smoking history, FEV1/FVC<lower limit of normal, FEV1%predicted<60% on stable respiratory condition AECOLD:COLD exacerbation, >40 years old, smoking history>20 pack-years with COLD, or COLD confirmed with PFTs CHF: Acute decompensate (systolic or diastolic) heart failure, defined as change in baseline dyspnea with evidence of fluid overload, elevated natriuretic peptides, or known history of chronic systolic or diastolic heart failure PNA: Pneumonia, defined as new infiltrate on admission CXR and symptoms consistent with pneumonia: malaise, sputum production, fever (T > 38.0°C), and crackles in auscultation of lung Exclusion: History of both COLD and heart failure, admitted with acute respiratory failure due to more than one reason (e.g., COLD and CHF, COLD and PNA, or CHF and PNA), previously diagnosed with bronchial asthma, bronchiectasis, bronchiolitis related to systemic pathology, cystic fibrosis, obstructive sleep apnea, or upper airway obstruction | Serum/ urine | Untargeted NMR | glycine, glutamine, alanine, proline, glutamate, mannitol, citrate, histidine, formate, creatine phosphate | Metabolomic analysis based on different clinical characteristics of COLD patients |
| Tan et al., 2018 [25] | Control: n =24 (14M/10F), Age: 61.5, BMI: 20.1 Patients: Phenotype E: n =20 (9M/11F), Age: 60.6, BMI: 19.1 Phenotype M: n =22 (14M/8F), Age: 62, BMI: 19.8 | Exclusion: other diseases and use of other medication | Serum | Untargeted NMR | Phenotype E vs. control: lactate, fructose, glycine, creatine, asparagine, citric acid, pyruvic acid, pyruvate, proline, acetone, L-glutamine, L-proline, ornithine, lipid CH2CH2CO, 2-hydroxyisobutyrate, threonine, isopropyl alcohol, pyridoxine, maltose, L-threonine, L-valine, glutamic acid, beta-alanine, cyclopentane, 2-aminoisobutyric acid Phenotype M vs. control: fructose, glycine, pyruvic acid, pyruvate, proline, acetone, L-proline, ornithine, lipid CH2CH2CO, threonine, isopropyl alcohol, guanosine, betaine, N-Acetyl-Cysteine(NAC),lipoprotein, L-alanine Phenotype E vs. Phenotype M: L-glutamine, L-alanine | Analysis based on lung function, serum samples, medical history, age, sex, smoking, physical examination, and scores of COLD assessment test |
| Yoneda et al., 2001 [26] | Controls: n =30 (29M/1F), Age: 64 Patients: n =30 (29M/1F) Age: 64 | Exclusion: other causes of weight loss (diabetes, endocrine disorders, malabsorption syndrome, neoplastic, infectious or liver diseases). Inclusion: Patients: receiving anticholinergic drugs, no requirement of supplemental oxygen, and no treatment with glucocorticoids or theophylline. Controls and patients matched for smoking habits | Plasma | Untargeted LC-MS | threonine, valine, leucine, isoleucine, methionine, phenylalanine, lysine, taurine, aspartic acid, glutamic acid, glutamine, serine, proline, glycine, alanine, tyrosine, ornithine, cysteine, histidine, arginine, BCAA, AAA, BCAA/AAA | Aminoacid analysis and BCAA/AAA ratio |
| Singh et al., 2017 [12] | COLD patients: Standard therapy: n =20 (20 M) Age: 64.2, BMI: 23.2 Standard+Doxy: n =30 (30M) Age: 67, BMI: 22.7 | Exclusion: significant cardiac and other co-morbidities, history of exacerbations in the preceding 6 weeks, and history of doxycycline intolerance or co-existing pulmonary condition affecting the assessment or intervention for COLD | Serum | Untargeted NMR | formate, citrate, imidazole, lactate, L-arginine, fatty acid | No information provided |
| Engelen et al., 2000 [27] | Control: Physically inactive: n= 15(10M/5F), Age: 67 Physically active: n =7(7M), Age: 63 Patients: EMPH+ (with macroscopic emphysema): n = 12 (10M/2F), Age: 64 EMPH- (without macroscopic emphysema): n =15 (11M/4F), Age: 64 | Inclusion: Patients: chronic airflow limitation (FEV1 < 70%),irreversible obstructive airway disease (<10% improvement of predicted baseline FEV1 after inhalation of b2antagonist), in clinically stable condition and without respiratory tract infection or exacerbation of their disease for at least 4 weeks before the study Exclusion: malignancy, cardiac failure, distal arteriopathy, recent surgery, severe endocrine, hepatic, or renal disorder and use of anticoagulant medication | Muscle biopsy/serum | Untargeted HPLC | glutamate, glycogen, glucose, pyruvate, lactate, lactate/pyruvate | Analysis of physical activity-dependent metabolic profiles |
| Airoldi et al., 2016 [28] | Controls: n= 11(4M/7F), Age: 55.27 Patients: n =11 (8M/3F), Age: 53 | Inclusion: protease inhibitor genotype ZZ-α1-antitrypsin deficient(PiZZ-AATD)patients with pulmonary emphysema recruited from the Department of Pulmonology of Leiden University Medical Center, The Netherlands Control group with non-smoking healthy volunteers, with normal spirometry results and no significant history of respiratory diseases | EBC | Untargeted NMR | acetate, 2,3-butanediol, propionic acid, lactate, butyrate, acetone, benzoate, fatty acid, formate, propylen glycol, alanine, ethanol, acetoion, isopropanol | No information provided |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsoukalas, D.; Sarandi, E.; Thanasoula, M.; Docea, A.O.; Tsilimidos, G.; Calina, D.; Tsatsakis, A. Metabolic Fingerprint of Chronic Obstructive Lung Diseases: A New Diagnostic Perspective. Metabolites 2019, 9, 290. https://doi.org/10.3390/metabo9120290
Tsoukalas D, Sarandi E, Thanasoula M, Docea AO, Tsilimidos G, Calina D, Tsatsakis A. Metabolic Fingerprint of Chronic Obstructive Lung Diseases: A New Diagnostic Perspective. Metabolites. 2019; 9(12):290. https://doi.org/10.3390/metabo9120290
Chicago/Turabian StyleTsoukalas, Dimitris, Evangelia Sarandi, Maria Thanasoula, Anca Oana Docea, Gerasimos Tsilimidos, Daniela Calina, and Aristides Tsatsakis. 2019. "Metabolic Fingerprint of Chronic Obstructive Lung Diseases: A New Diagnostic Perspective" Metabolites 9, no. 12: 290. https://doi.org/10.3390/metabo9120290
APA StyleTsoukalas, D., Sarandi, E., Thanasoula, M., Docea, A. O., Tsilimidos, G., Calina, D., & Tsatsakis, A. (2019). Metabolic Fingerprint of Chronic Obstructive Lung Diseases: A New Diagnostic Perspective. Metabolites, 9(12), 290. https://doi.org/10.3390/metabo9120290









