Alteration of Metabolic Pathways in Osteoarthritis
Abstract
1. Introduction
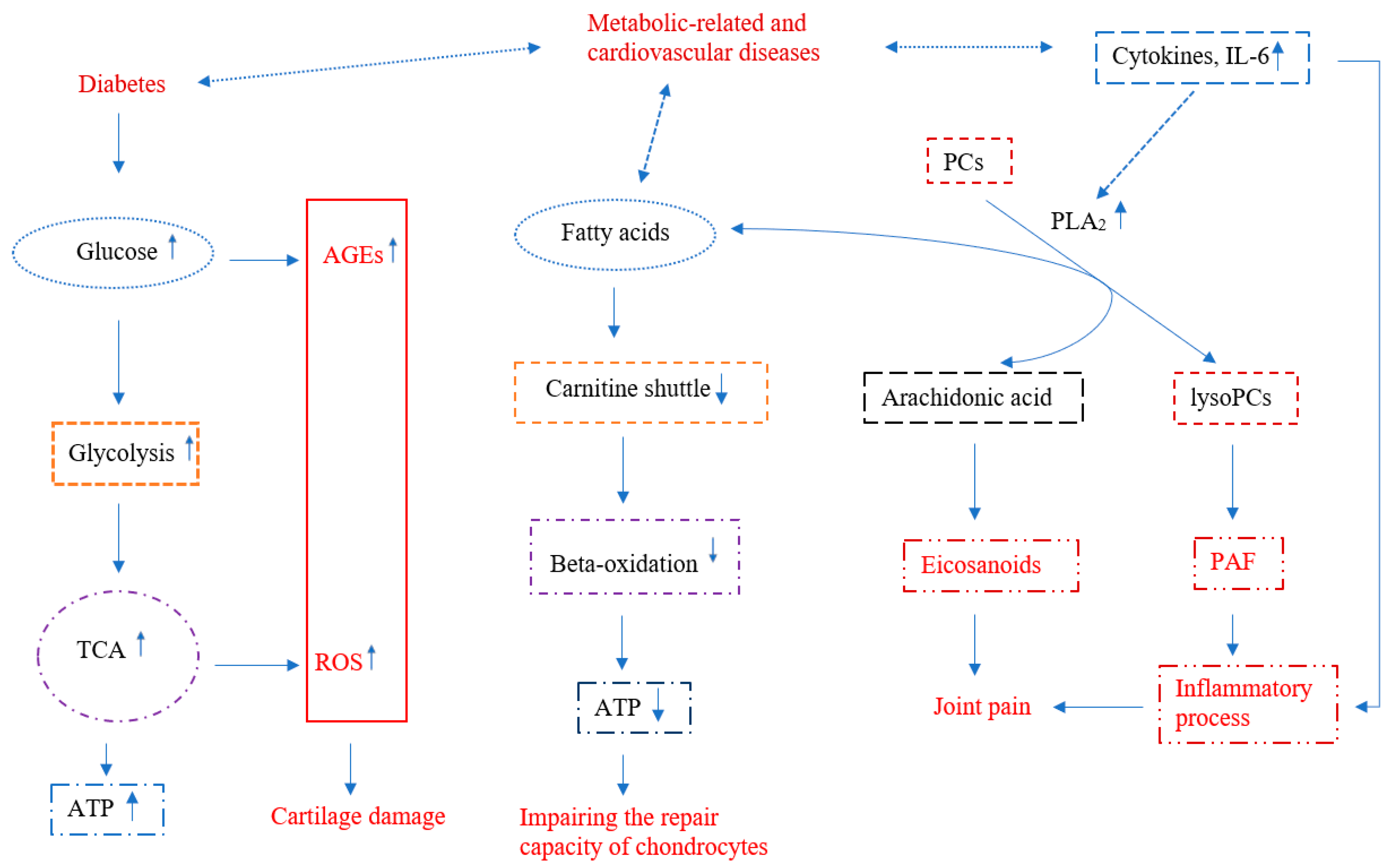
2. Energy Metabolic Pathways
2.1. Glucose Metabolism
2.2. Tricarboxylic Acid Cycle (TCA)
2.3. ß-Oxidation Pathway
3. Lipid Metabolism
Phospholipid Metabolism
4. Eicosanoid Pathway
5. Amino Acid Metabolism
5.1. Branched Chain Amino Acid (BCAA)
5.2. Arginine
6. Other Metabolic Factors
7. Summary
Funding
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. The Burden of Musculoskeletal Conditions at the Start of the New Millennium: Report of a WHO Scientific Group; World Health Organisation: Geneva, Switzerland, 2003. [Google Scholar]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Hiligsmann, M.; Cooper, C.; Arden, N.; Boers, M.; Branco, J.C.; Luisa Brandi, M.; Bruyere, O.; Guillemin, F.; Hochberg, M.C.; Hunter, D.J.; et al. Health economics in the field of osteoarthritis: An expert’s consensus paper from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin. Arthritis Rheum. 2013, 43, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Culliford, D.J.; Maskell, J.; Kiran, A.; Judge, A.; Javaid, M.K.; Cooper, C.; Arden, N.K. The lifetime risk of total hip and knee arthroplasty: Results from the UK general practice research database. Osteoarthr. Cartil. 2012, 20, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Beswick, A.D.; Wylde, V.; Gooberman-Hill, R.; Blom, A.; Dieppe, P. What proportion of patients report long-term pain after total hip or knee replacement for osteoarthritis? A systematic review of prospective studies in unselected patients. BMJ Open 2012, 2, e000435. [Google Scholar] [CrossRef] [PubMed]
- Goldring, S.R.; Goldring, M.B. Changes in the osteochondral unit during osteoarthritis: Structure, function and cartilage-bone crosstalk. Nat. Rev. Rheumatol. 2016, 12, 632–644. [Google Scholar] [CrossRef] [PubMed]
- Mobasheri, A.; Rayman, M.P.; Gualillo, O.; Sellam, J.; van der Kraan, P.; Fearon, U. The role of metabolism in the pathogenesis of osteoarthritis. Nat. Rev. Rheumatol. 2017, 13, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Guilak, F. Biomechanical factors in osteoarthritis. Best Pract. Res. Clin. Rheumatol. 2011, 25, 815–823. [Google Scholar] [CrossRef]
- Croucher, L.J.; Crawford, A.; Hatton, P.V.; Russell, R.G.; Buttle, D.J. Extracellular ATP and UTP stimulate cartilage proteoglycan and collagen accumulation in bovine articular chondrocyte pellet cultures. Biochim. Biophys. Acta 2000, 1502, 297–306. [Google Scholar] [CrossRef]
- Millward-Sadler, S.J.; Wright, M.O.; Flatman, P.W.; Salter, D.M. ATP in the mechanotransduction pathway of normal human chondrocytes. Biorheology 2004, 41, 567–575. [Google Scholar]
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The basic science of articular cartilage: Structure, composition, and function. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef]
- Maneiro, E.; Martin, M.A.; de Andres, M.C.; Lopez-Armada, M.J.; Fernandez-Sueiro, J.L.; del Hoyo, P.; Galdo, F.; Arenas, J.; Blanco, F.J. Mitochondrial respiratory activity is altered in osteoarthritic human articular chondrocytes. Arthritis Rheumatol. 2003, 48, 700–708. [Google Scholar] [CrossRef]
- Kudirka, J.C.; Panupinthu, N.; Tesseyman, M.A.; Dixon, S.J.; Bernier, S.M. P2Y nucleotide receptor signaling through MAPK/ERK is regulated by extracellular matrix: Involvement of beta3 integrins. J. Cell Physiol. 2007, 213, 54–64. [Google Scholar] [CrossRef]
- Yang, X.; Chen, W.; Zhao, X.; Chen, L.; Li, W.; Ran, J.; Wu, L. Pyruvate Kinase M2 Modulates the Glycolysis of Chondrocyte and Extracellular Matrix in Osteoarthritis. DNA Cell Biol. 2018, 37, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Cimmino, M.A.; Cutolo, M. Plasma glucose concentration in symptomatic osteoarthritis: A clinical and epidemiological survey. Clin. Exp. Rheumatol. 1990, 8, 251–257. [Google Scholar] [PubMed]
- Garessus, E.D.; de Mutsert, R.; Visser, A.W.; Rosendaal, F.R.; Kloppenburg, M. No association between impaired glucose metabolism and osteoarthritis. Osteoarthr. Cartil. 2016, 24, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Schett, G.; Kleyer, A.; Perricone, C.; Sahinbegovic, E.; Iagnocco, A.; Zwerina, J.; Lorenzini, R.; Aschenbrenner, F.; Berenbaum, F.; D’Agostino, M.A.; et al. Diabetes is an independent predictor for severe osteoarthritis: Results from a longitudinal cohort study. Diabetes Care 2013, 36, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Dubey, N.K.; Ningrum, D.N.A.; Dubey, R.; Deng, Y.H.; Li, Y.C.; Wang, P.D.; Wang, J.R.; Syed-Abdul, S.; Deng, W.P. Correlation between Diabetes Mellitus and Knee Osteoarthritis: A Dry-To-Wet Lab Approach. Int. J. Mol. Sci. 2018, 19, 3021. [Google Scholar] [CrossRef]
- Mobasheri, A. Glucose: An energy currency and structural precursor in articular cartilage and bone with emerging roles as an extracellular signaling molecule and metabolic regulator. Front. Endocrinol. 2012, 3, 153. [Google Scholar] [CrossRef] [PubMed]
- Rosa, S.C.; Goncalves, J.; Judas, F.; Mobasheri, A.; Lopes, C.; Mendes, A.F. Impaired glucose transporter-1 degradation and increased glucose transport and oxidative stress in response to high glucose in chondrocytes from osteoarthritic versus normal human cartilage. Arthritis Res. 2009, 11, R80. [Google Scholar] [CrossRef]
- June, R.K.; Liu-Bryan, R.; Long, F.; Griffin, T.M. Emerging role of metabolic signaling in synovial joint remodeling and osteoarthritis. J. Orthop. Res. 2016, 34, 2048–2058. [Google Scholar] [CrossRef] [PubMed]
- Lepetsos, P.; Papavassiliou, A.G. ROS/oxidative stress signaling in osteoarthritis. Biochim. Biophys. Acta 2016, 1862, 576–591. [Google Scholar] [CrossRef]
- Saudek, D.M.; Kay, J. Advanced glycation endproducts and osteoarthritis. Curr. Rheumatol. Rep. 2003, 5, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Randell, E.W.; Sun, G.; Likhodii, S.; Liu, M.; Furey, A.; Zhai, G. Hyperglycemia-related advanced glycation end-products is associated with the altered phosphatidylcholine metabolism in osteoarthritis patients with diabetes. PLoS ONE 2017, 12, e0184105. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Sun, G.; Aitken, D.; Likhodii, S.; Liu, M.; Martin, G.; Furey, A.; Randell, E.; Rahman, P.; Jones, G.; et al. Lysophosphatidylcholines to phosphatidylcholines ratio predicts advanced knee osteoarthritis. Rheumatology 2016, 55, 1566–1574. [Google Scholar] [CrossRef]
- Adams, S.B., Jr.; Setton, L.A.; Kensicki, E.; Bolognesi, M.P.; Toth, A.P.; Nettles, D.L. Global metabolic profiling of human osteoarthritic synovium. Osteoarthr. Cartil. 2012, 20, 64–67. [Google Scholar] [CrossRef]
- Anderson, J.R.; Chokesuwattanaskul, S.; Phelan, M.M.; Welting, T.J.M.; Lian, L.Y.; Peffers, M.J.; Wright, H.L. 1H NMR Metabolomics Identifies Underlying Inflammatory Pathology in Osteoarthritis and Rheumatoid Arthritis Synovial Joints. J. Proteome Res. 2018. [Google Scholar] [CrossRef]
- Kim, S.; Hwang, J.; Kim, J.; Ahn, J.K.; Cha, H.S.; Kim, K.H. Metabolite profiles of synovial fluid change with the radiographic severity of knee osteoarthritis. Jt. Bone Spine 2017, 84, 605–610. [Google Scholar] [CrossRef]
- Muraki, S.; Akune, T.; En-yo, Y.; Yoshida, M.; Tanaka, S.; Kawaguchi, H.; Nakamura, K.; Oka, H.; Yoshimura, N. Association of dietary intake with joint space narrowing and osteophytosis at the knee in Japanese men and women: The ROAD study. Mod. Rheumatol. 2014, 24, 236–242. [Google Scholar] [CrossRef]
- Dehghan, M. Comparative effectiveness of B and e vitamins with diclofenac in reducing pain due to osteoarthritis of the knee. Med. Arch. 2015, 69, 103–106. [Google Scholar] [CrossRef]
- Wang, J.; Sun, H.; Fu, Z.; Liu, M. Chondroprotective effects of alpha-lipoic acid in a rat model of osteoarthritis. Free Radic. Res. 2016, 50, 767–780. [Google Scholar] [CrossRef]
- Hah, Y.S.; Sung, M.J.; Lim, H.S.; Jun, J.S.; Jeong, Y.G.; Kim, H.O.; Kim, J.; Hur, H.J.; Davaatseren, M.; Kwon, D.Y.; et al. Dietary alpha lipoic acid supplementation prevents synovial inflammation and bone destruction in collagen-induced arthritic mice. Rheumatol. Int. 2011, 31, 1583–1590. [Google Scholar] [CrossRef] [PubMed]
- Tootsi, K.; Kals, J.; Zilmer, M.; Paapstel, K.; Ottas, A.; Martson, A. Medium- and long-chain acylcarnitines are associated with osteoarthritis severity and arterial stiffness in end-stage osteoarthritis patients: A case-control study. Int. J. Rheum. Dis. 2018, 21, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Likhodii, S.; Zhang, Y.; Aref-Eshghi, E.; Harper, P.E.; Randell, E.; Green, R.; Martin, G.; Furey, A.; Sun, G.; et al. Classification of osteoarthritis phenotypes by metabolomics analysis. BMJ Open 2014, 4, e006286. [Google Scholar] [CrossRef] [PubMed]
- Stoppoloni, D.; Politi, L.; Dalla Vedova, P.; Messano, M.; Koverech, A.; Scandurra, R.; Scotto d’Abusco, A. l-carnitine enhances extracellular matrix synthesis in human primary chondrocytes. Rheumatol. Int. 2013, 33, 2399–2403. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, E.; Di Cesare Mannelli, L.; Menicacci, C.; Lorenzoni, P.; Agliano, M.; Ghelardini, C. Prophylactic role of acetyl-l-carnitine on knee lesions and associated pain in a rat model of osteoarthritis. Life Sci. 2014, 106, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Malek Mahdavi, A.; Mahdavi, R.; Kolahi, S. Effects of l-Carnitine Supplementation on Serum Inflammatory Factors and Matrix Metalloproteinase Enzymes in Females with Knee Osteoarthritis: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. J. Am. Coll. Nutr. 2016, 35, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Bliddal, H.; Leeds, A.R.; Christensen, R. Osteoarthritis, obesity and weight loss: Evidence, hypotheses and horizons—A scoping review. Obes. Rev. 2014, 15, 578–586. [Google Scholar] [CrossRef]
- Pottie, P.; Presle, N.; Terlain, B.; Netter, P.; Mainard, D.; Berenbaum, F. Obesity and osteoarthritis: More complex than predicted! Ann. Rheum. Dis. 2006, 65, 1403–1405. [Google Scholar] [CrossRef]
- Sellam, J.; Berenbaum, F. Is osteoarthritis a metabolic disease? Jt. Bone Spine 2013, 80, 568–573. [Google Scholar] [CrossRef]
- Yoshimura, N.; Muraki, S.; Oka, H.; Tanaka, S.; Kawaguchi, H.; Nakamura, K.; Akune, T. Accumulation of metabolic risk factors such as overweight, hypertension, dyslipidaemia, and impaired glucose tolerance raises the risk of occurrence and progression of knee osteoarthritis: A 3-year follow-up of the ROAD study. Osteoarthr. Cartil. 2012, 20, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.X.; Wei, J.; Zeng, C.; Yang, T.; Li, H.; Wang, Y.L.; Long, H.Z.; Wu, Z.Y.; Qian, Y.X.; Li, K.H.; et al. Association between metabolic syndrome and knee osteoarthritis: A cross-sectional study. BMC Musculoskelet. Disord. 2017, 18, 533. [Google Scholar] [CrossRef]
- Garcia-Gil, M.; Reyes, C.; Ramos, R.; Sanchez-Santos, M.T.; Prieto-Alhambra, D.; Spector, T.D.; Hart, D.J.; Arden, N.K. Serum Lipid Levels and Risk of Hand Osteoarthritis: The Chingford Prospective Cohort Study. Sci. Rep. 2017, 7, 3147. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.P.; Humm, G.; Crisp, A.J. 1H nuclear magnetic resonance investigation of synovial fluid components in osteoarthritis, rheumatoid arthritis and traumatic effusions. Br. J. Rheumatol. 1989, 28, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Sun, G.; Likhodii, S.; Aref-Eshghi, E.; Harper, P.E.; Randell, E.; Green, R.; Martin, G.; Furey, A.; Rahman, P.; et al. Metabolomic analysis of human synovial fluid and plasma reveals that phosphatidylcholine metabolism is associated with both osteoarthritis and diabetes mellitus. Metabolomics 2015, 12, 24. [Google Scholar] [CrossRef]
- Kosinska, M.K.; Liebisch, G.; Lochnit, G.; Wilhelm, J.; Klein, H.; Kaesser, U.; Lasczkowski, G.; Rickert, M.; Schmitz, G.; Steinmeyer, J. A lipidomic study of phospholipid classes and species in human synovial fluid. Arthritis Rheumatol. 2013, 65, 2323–2333. [Google Scholar] [CrossRef] [PubMed]
- Castro-Perez, J.M.; Kamphorst, J.; DeGroot, J.; Lafeber, F.; Goshawk, J.; Yu, K.; Shockcor, J.P.; Vreeken, R.J.; Hankemeier, T. Comprehensive LC-MS E lipidomic analysis using a shotgun approach and its application to biomarker detection and identification in osteoarthritis patients. J. Proteome Res. 2010, 9, 2377–2389. [Google Scholar] [CrossRef]
- Zhai, G.; Pelletier, J.P.; Liu, M.; Aitken, D.; Randell, E.; Rahman, P.; Jones, G.; Martel-Pelletier, J. Activation of the phosphatidylcholine to lysophosphatidylcholine pathway is associated with osteoarthritis knee cartilage volume loss over time: Data from a 2-year multicentre clinical trial. Sci. Rep. 2018. under review. [Google Scholar]
- Pruzanski, W.; Bogoch, E.; Stefanski, E.; Wloch, M.; Vadas, P. Enzymatic activity and distribution of phospholipase A2 in human cartilage. Life Sci. 1991, 48, 2457–2462. [Google Scholar] [CrossRef]
- Livshits, G.; Zhai, G.; Hart, D.J.; Kato, B.S.; Wang, H.; Williams, F.M.; Spector, T.D. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: The Chingford study. Arthritis Rheumatol. 2009, 60, 2037–2045. [Google Scholar] [CrossRef]
- Wang, D.; Dubois, R.N. Eicosanoids and cancer. Nat. Rev. Cancer 2010, 10, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Attur, M.; Krasnokutsky, S.; Statnikov, A.; Samuels, J.; Li, Z.; Friese, O.; Hellio Le Graverand-Gastineau, M.P.; Rybak, L.; Kraus, V.B.; Jordan, J.M.; et al. Low-grade inflammation in symptomatic knee osteoarthritis: Prognostic value of inflammatory plasma lipids and peripheral blood leukocyte biomarkers. Arthritis Rheumatol. 2015, 67, 2905–2915. [Google Scholar] [CrossRef] [PubMed]
- Valdes, A.M.; Ravipati, S.; Pousinis, P.; Menni, C.; Mangino, M.; Abhishek, A.; Chapman, V.; Barrett, D.A.; Doherty, M. Omega-6 oxylipins generated by soluble epoxide hydrolase are associated with knee osteoarthritis. J. Lipid Res. 2018, 59, 1763–1770. [Google Scholar] [CrossRef] [PubMed]
- Zhai, G.; Wang-Sattler, R.; Hart, D.J.; Arden, N.K.; Hakim, A.J.; Illig, T.; Spector, T.D. Serum branched-chain amino acid to histidine ratio: A novel metabolomic biomarker of knee osteoarthritis. Ann. Rheum. Dis. 2010, 69, 1227–1231. [Google Scholar] [CrossRef] [PubMed]
- Maher, A.D.; Coles, C.; White, J.; Bateman, J.F.; Fuller, E.S.; Burkhardt, D.; Little, C.B.; Cake, M.; Read, R.; McDonagh, M.B.; et al. 1H NMR spectroscopy of serum reveals unique metabolic fingerprints associated with subtypes of surgically induced osteoarthritis in sheep. J. Proteome Res. 2012, 11, 4261–4268. [Google Scholar] [CrossRef] [PubMed]
- Neuman, P.; Kostogiannis, I.; Friden, T.; Roos, H.; Dahlberg, L.E.; Englund, M. Patellofemoral osteoarthritis 15 years after anterior cruciate ligament injury—A prospective cohort study. Osteoarthr. Cartil. 2009, 17, 284–290. [Google Scholar] [CrossRef]
- Ikeda, T.; Jinno, T.; Masuda, T.; Aizawa, J.; Ninomiya, K.; Suzuki, K.; Hirakawa, K. Effect of exercise therapy combined with branched-chain amino acid supplementation on muscle strengthening in persons with osteoarthritis. Hong Kong Physiother. J. 2018, 38, 23–31. [Google Scholar] [CrossRef]
- Zhang, W.; Sun, G.; Likhodii, S.; Liu, M.; Aref-Eshghi, E.; Harper, P.E.; Randell, E.; Green, R.; Martin, G.; Furey, A.; et al. Metabolomic analysis of human plasma reveals that arginine is depleted in knee osteoarthritis patients. Osteoarthr. Cartil. 2016, 24, 827–834. [Google Scholar] [CrossRef]
- Morris, S.M., Jr. Arginine metabolism: Boundaries of our knowledge. J. Nutr. 2007, 137, 1602S–1609S. [Google Scholar] [CrossRef]
- Pascale, V.; Pascale, W.; Lavanga, V.; Sansone, V.; Ferrario, P.; De Gennaro Colonna, V. l-arginine, asymmetric dimethylarginine, and symmetric dimethylarginine in plasma and synovial fluid of patients with knee osteoarthritis. Med. Sci. Monit. 2013, 19, 1057–1062. [Google Scholar] [CrossRef]
- Morris, S.M., Jr. Arginine: Beyond protein. Am. J. Clin. Nutr. 2006, 83, 508S–512S. [Google Scholar] [CrossRef] [PubMed]
- Abramson, S.B. Nitric oxide in inflammation and pain associated with osteoarthritis. Arthritis Res. 2008, 10 (Suppl. 2), S2. [Google Scholar] [CrossRef]
- Ohnishi, A.; Osaki, T.; Matahira, Y.; Tsuka, T.; Imagawa, T.; Okamoto, Y.; Minami, S. Correlation of plasma amino acid concentrations and chondroprotective effects of glucosamine and fish collagen peptide on the development of osteoarthritis. J. Vet. Med. Sci. 2013, 75, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Loeser, R.F.; Pathmasiri, W.; Sumner, S.J.; McRitchie, S.; Beavers, D.; Saxena, P.; Nicklas, B.J.; Jordan, J.; Guermazi, A.; Hunter, D.J.; et al. Association of urinary metabolites with radiographic progression of knee osteoarthritis in overweight and obese adults: An exploratory study. Osteoarthr. Cartil. 2016, 24, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhai, G. Alteration of Metabolic Pathways in Osteoarthritis. Metabolites 2019, 9, 11. https://doi.org/10.3390/metabo9010011
Zhai G. Alteration of Metabolic Pathways in Osteoarthritis. Metabolites. 2019; 9(1):11. https://doi.org/10.3390/metabo9010011
Chicago/Turabian StyleZhai, Guangju. 2019. "Alteration of Metabolic Pathways in Osteoarthritis" Metabolites 9, no. 1: 11. https://doi.org/10.3390/metabo9010011
APA StyleZhai, G. (2019). Alteration of Metabolic Pathways in Osteoarthritis. Metabolites, 9(1), 11. https://doi.org/10.3390/metabo9010011



