Parameters of the Endocannabinoid System as Novel Biomarkers in Sepsis and Septic Shock
Abstract
1. Introduction
1.1. Sepsis
1.2. State of Sepsis-Specific Treatments & Considerations with Immunomodulators
1.3. Biomarkers in Sepsis
1.3.1. Overview
1.3.2. As Diagnostic Markers
1.3.3. Goals in Biomarker Selection & Multi-Biomarker Panels
1.3.4. Key Markers in Clinical Practice
Procalcitonin (PCT) & C-Reactive Protein (CRP)
Lactate
Assessing Immune Status & Function in Sepsis: A Guide for Immunomodulatory Therapy
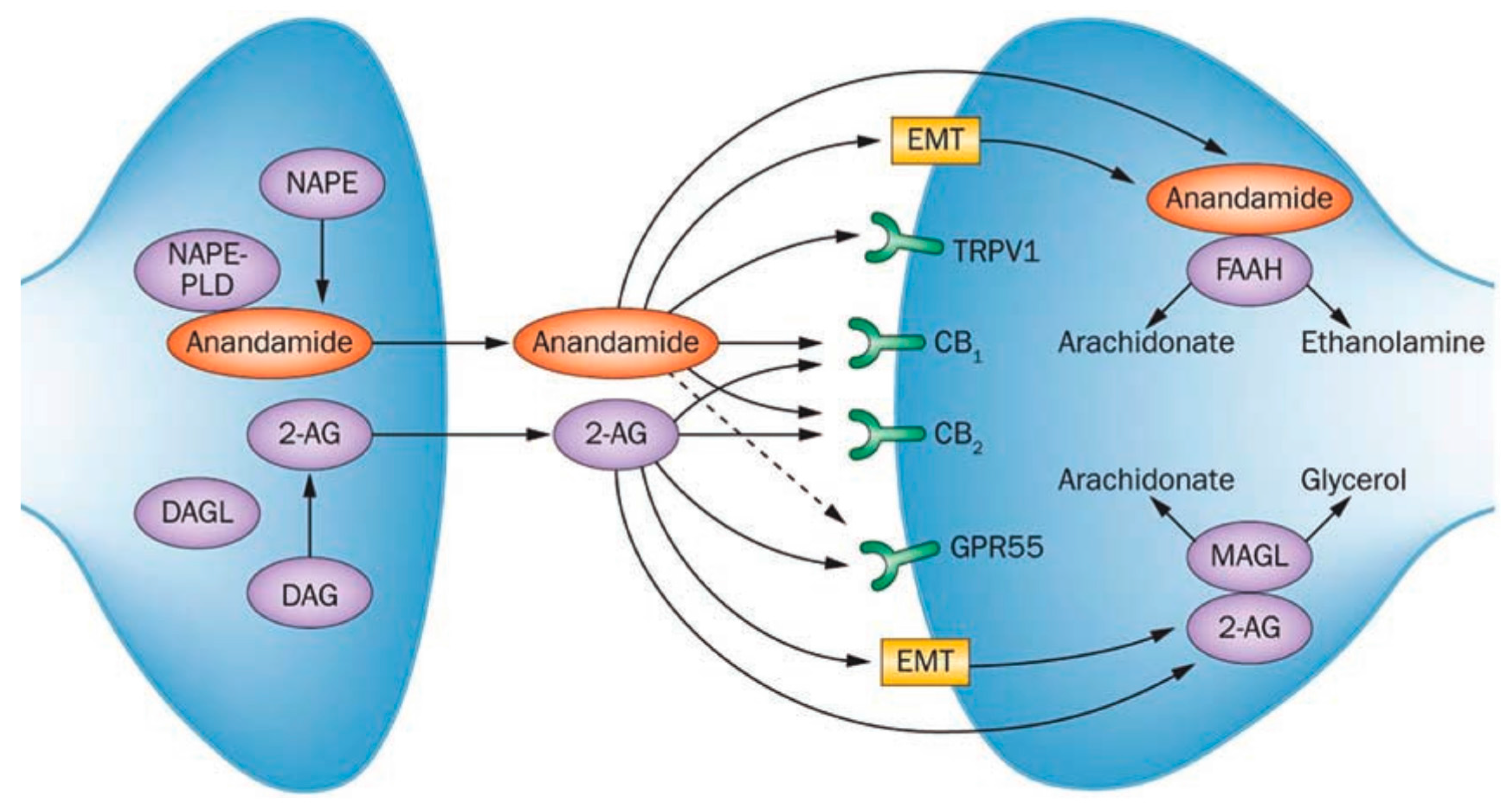
2. The Endocannabinoid System (ECS)
3. Immune System & the ECS in Sepsis
3.1. The ECS as a Tool in Sepsis
3.1.1. Biomarkers of the ECS
Endocannabinoids
Enzymes
Expression of Cannabinoid Receptors
3.1.2. Theoretical Mechanisms of ECS-Based Immunomodulatory Therapy in Sepsis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, R.S.; Monneret, G.; Payen, D. Immunosuppression in sepsis: A novel understanding of the disorder and a new therapeutic approach. Lancet Infect. Dis. 2013, 13, 260–268. [Google Scholar] [CrossRef]
- Reber, A.J.; Chirkova, T.; Kim, J.H.; Cao, W.; Biber, R.; Shay, D.K.; Sambhara, S. Immunosenescence and challenges of vaccination against influenza in the aging population. Aging Dis. 2012, 3, 68–90. [Google Scholar] [PubMed]
- Mayr, F.B.; Yende, S.; Angus, D.C. Epidemiology of severe sepsis. Virulence 2014, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Dombrovskiy, V.Y.; Martin, A.A.; Sunderram, J.; Paz, H.L. Rapid increase in hospitalization and mortality rates for severe sepsis in the United States: A trend analysis from 1993 to 2003. Crit. Care Med. 2007, 35, 1244–1250. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.L.; Rello, J.; Marshall, J.; Silva, E.; Anzueto, A.; Martin, C.D.; Moreno, R.; Lipman, J.; Gomersall, C.; Sakr, Y.; et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009, 302, 2323–2329. [Google Scholar] [CrossRef] [PubMed]
- Torgersen, C.; Moser, P.; Luckner, G.; Mayr, V.; Jochberger, S.; Hasibeder, W.R.; Dünser, M.W. Macroscopic postmortem findings in 235 surgical intensive care patients with sepsis. Anesth. Analg. 2009, 108, 1841–1847. [Google Scholar] [CrossRef] [PubMed]
- Webster, N.R.; Galley, H.F. Immunomodulation in the critically ill. Br. J. Anaesth. 2009, 103, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Bernard, G.R.; Vincent, J.L.; Laterre, P.F.; LaRosa, S.P.; Dhainaut, J.F.; Lopez-Rodriguez, A.; Steingrub, J.S.; Garber, G.E.; Helterbrand, J.D.; Ely, E.W.; et al. Efficacy and safety of recombinant human activated protein C for severe sepsis. N. Engl. J. Med. 2001, 344, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, V.M.; Thompson, B.T.; Barie, P.S.; Dhainaut, J.F.; Douglas, I.S.; Finfer, S.; Gårdlund, B.; Marshall, J.C.; Rhodes, A.; Artigas, A.; et al. Drotrecogin alfa (activated) in adults with septic shock. N. Engl. J. Med. 2012, 366, 2055–2064. [Google Scholar] [CrossRef] [PubMed]
- Opal, S.M.; Fisher, C.J., Jr.; Dhainaut, J.F.; Vincent, J.L.; Brase, R.; Lowry, S.F.; Sadoff, J.C.; Slotman, G.J.; Levy, H.; Balk, R.A.; et al. Confirmatory interleukin-1 receptor antagonist trial in severe sepsis: A phase III, randomized, double-blind, placebo-controlled, multicenter trial. The Interleukin-1 Receptor Antagonist Sepsis Investigator Group. Crit. Care Med. 1997, 25, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Hotchkiss, R.S.; Karl, I.E. The pathophysiology and treatment of sepsis. N. Engl. J. Med. 2003, 348, 138–150. [Google Scholar] [CrossRef] [PubMed]
- Pierrakos, C.; Vincent, J.L. Sepsis biomarkers: A review. Crit. Care 2010, 14, R15. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Mandel, J. Evaluation and Management of Suspected Sepsis and Septic Shock in Adults. UpToDate. Available online: https://www.uptodate.com/contents/evaluation-and-management-of-suspected-sepsis-and-septic-shock-in-adults (accessed on 29 September 2017).
- Faix, J.D. Biomarkers of sepsis. Crit. Rev. Clin. Lab. Sci. 2013, 50, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Garnacho-Montero, J.; Huici-Moreno, M.J.; Gutiérrez-Pizarraya, A.; López, I.; Márquez-Vácaro, J.; Macher, H.; Puppo-Moreno, A. Prognostic and diagnostic value of eosinopenia, C-reactive protein, procalcitonin, and circulating cell-free DNA in critically ill patients admitted with suspicion of sepsis. Crit. Care 2014, 18, R116. [Google Scholar] [CrossRef] [PubMed]
- Kushner, I.; Samols, D. Oswald Avery and the Pneumococcus. Pharos Alpha Omega Alpha Honor Med. Soc. 2011, 74, 14. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21615066 (accessed on 29 October 2017). [PubMed]
- Tillett, W.S.; Francis, T. Serological reactions in pneumonia with a non-protein somatic fraction of the pneumococcus. J. Exp. Med. 1930, 52, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Kushner, I. The phenomenon of the acute phase response. Ann. N. Y. Acad. Sci. 1982, 389, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Gabay, C.; Kushner, I. Acute-phase proteins and other systemic responses to inflammation. N. Engl. J. Med. 1999, 340, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Muller, B.; White, J.C.; Nylen, E.S.; Snider, R.H.; Becker, K.L.; Habener, J.F. Ubiquitous expression of the calcitonin-I gene in multiple tissues in response to sepsis. J. Clin. Endocrinol. Metab. 2001, 86, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Assicot, M.; Gendrel, D. High serum procalcitonin concentrations in patients with sepsis and infection. Lancet 1993, 341, 515–518. [Google Scholar] [CrossRef]
- Karzai, W.; Oberhoffer, M.; Meier-Hellmann, A.; Reinhart, K. Procalcitonin: A new indicator of the systemic response to severe infections. Infection 1997, 25, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Barati, M.; Alinejad, F.; Bahar, M.A.; Tabrisi, M.S.; Shamshiri, A.R.; Bodouhi, N.O.; Karimi, H. Comparison of WBC, ESR, CRP and PCT serum levels in septic and non-septic burn cases. Burns 2008, 34, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Brunkhorst, F.M.; Al-Nawas, B.; Krummenauer, F.; Forycki, Z.F.; Shah, P.M. Procalcitonin, C-reactive protein and APACHE II score for risk evaluation in patients with severe pneumonia. Clin. Microbiol. Infect. 2002, 8, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Bele, N.; Darmon, M.; Coquet, I.; Feugeas, J.P.; Legriel, S.; Adaoui, N.; Schlemmer, B.; Azoulay, E. Diagnostic accuracy of procalcitonin in critically ill immunocompromised patients. BMC Infect. Dis. 2011, 11, 224. [Google Scholar] [CrossRef] [PubMed]
- Simon, L.; Gauvin, F.; Amre, D.K.; Saint-Louis, P.; Lacroix, J. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: A systematic review and meta-analysis. Clin. Infect. Dis. 2004, 39, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Póvoa, P.; Coelho, L.; Almeida, E.; Fernandes, A.; Mealha, R.; Moreira, P.; Sabino, H. C-reactive protein as a marker of infection in critically ill patients. Clin. Microbiol. Infect. 2005, 11, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Hofer, N.; Zacharias, E.; Muller, W.; Resch, B. An update on the use of C-reactive protein in early-onset neonatal sepsis: Current insights and new tasks. Neonatology 2012, 102, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, N.I.; Howell, M.D.; Talmor, D.; Nathanson, L.A.; Lisbon, A.; Wolfe, R.E.; Weiss, J.W. Serum lactate as a predictor of mortality in emergency department patients with infection. Ann. Emerg. Med. 2005, 45, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Kraut, J.A.; Madias, N.E. Lactic acidosis. N. Engl. J. Med. 2014, 371, 2309–2319. [Google Scholar] [CrossRef] [PubMed]
- Ronco, J.J.; Fenwick, J.C.; Tweeddale, M.G.; Wiggs, B.R.; Phang, P.T.; Cooper, D.J.; Cunningham, K.F.; Russell, J.A.; Walley, K.R. Identification of the critical oxygen delivery for anaerobic metabolism in critically ill septic and nonseptic humans. JAMA 1993, 270, 1724–1730. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Alvarez, M.; Marik, P.; Bellomo, R. Sepsis-associated hyperlactatemia. Crit. Care 2014, 18, 503. [Google Scholar] [CrossRef] [PubMed]
- Toffaletti, J.G. Blood lactate: Biochemistry, laboratory methods, and clinical interpretation. Crit. Rev. Clin. Lab. Sci. 1991, 28, 253–268. [Google Scholar] [CrossRef] [PubMed]
- Monneret, G.; Venet, F. Monocyte HLA-DR in sepsis: Shall we stop following the flow? Crit. Care 2014, 18, 102. [Google Scholar] [CrossRef] [PubMed]
- Schefold, J.C. Measurement of monocytic HLA-DR (mHLA-DR) expression in patients with severe sepsis and septic shock: Assessment of immune organ failure. Intensive Care Med. 2010, 36, 1810–1812. [Google Scholar] [CrossRef] [PubMed]
- Russo, E.B. History of cannabis and its preparations in saga, science, and sobriquet. Chem. Biodivers. 2007, 4, 1614–1648. [Google Scholar] [CrossRef] [PubMed]
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular characterization of a peripheral receptor for cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Howlett, A.C.; Barth, F.; Bonner, T.I.; Cabral, G.; Casellas, P.; Devane, W.A.; Felder, C.C.; Herkenham, M.; Mackie, K.; Martin, B.R.; et al. International Union of Pharmacology: XXVII. Classification of cannabinoid receptors. Pharmacol. Rev. 2002, 54, 161–202. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Receptors and channels targeted by synthetic cannabinoid receptor agonists and antagonists. Curr. Med. Chem. 2010, 17, 1360–1381. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J. The endocannabinoid signaling System in the CNS: A primer. Int. Rev. Neurobiol. 2015, 125, 1–47. [Google Scholar] [PubMed]
- Devane, W.A.; Dysarz, F.A., III; Johnson, M.R.; Melvin, L.S.; Howlett, A.C. Determination and Characterization of a cannabinoid receptor in rat brain. Mol. Pharmacol. 1998, 34, 605–613. [Google Scholar]
- Kreth, S.; Kaufmann, I.; Ledderose, C.; Luchting, B.; Thiel, M. Reduced ligand affinity leads to an impaired function of the adenosine A2A receptor of human granulocytes in sepsis. J. Cell. Mol. Med. 2009, 13, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Tschöp, J.; Kasten, K.R.; Nogueiras, R.; Goetzman, H.S.; Cave, C.M.; England, L.G.; Dattilo, J.; Lentsch, A.B.; Tschöp, M.H.; Caldwell, C.C. The cannabinoid receptor 2 is critical for the host response to sepsis. J. Immunol. 2009, 183, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.W.; Cabral, G.A. Cannabinoid-induced immune suppression and modulation of antigen-presenting cells. J. Neuroimmune Pharmacol. 2006, 1, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Goparaju, S.K.; Ueda, N.; Taniguchi, K.; Yamamoto, S. Enzymes of porcine brain hydrolyzing 2-arachidonoylglycerol, an endogenous ligand of cannabinoid receptors. Biochem. Pharmacol. 1999, 57, 417–423. [Google Scholar] [CrossRef]
- Stella, N.; Schweitzer, P.; Piomelli, D. A second endogenous cannabinoid that modulates long-term potentiation. Nature 1997, 388, 773–778. [Google Scholar] [CrossRef] [PubMed]
- Piomelli, D.; Beltramo, M.; Giuffrida, A.; Stella, N. Endogenous cannabinoid signaling. Neurobiol. Dis. 1998, 5, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Cravatt, B.F.; Demarest, K.; Patricelli, M.P.; Bracey, M.H.; Giang, D.K.; Martin, B.R.; Lichtman, A.H. Supersensitivity to anandamide and enhanced endogenous cannabinoid signaling in mice lacking fatty acid amide hydrolase. Proc. Natl. Acad. Sci. USA 2001, 98, 9371–9376. [Google Scholar] [CrossRef] [PubMed]
- Burstein, S.H.; Zurier, R.B. Cannabinoids, Endocannabinoids, and Related Analogs in Inflammation. AAPS J. 2009, 11, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Schicho, R.; Storr, M. Patients with IBD find symptom relief in the Cannabis field. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 142–143. [Google Scholar] [CrossRef] [PubMed]
- Kelly, M.E.M.; Lehmann, C.; Zhou, J. The Endocannabinoid System in Local and Systemic Inflammation; Morgan & Claypool: San Rafael, CA, USA, 2017. [Google Scholar]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; Le Fur, G.; Casellas, P. Expression of central and peripheral cannabinoid receptors in human immune tissues and leukocyte subpopulations. Eur. J. Biochem. 1995, 232, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Kasten, K.R.; Tschöp, J.; Tschöp, M.H.; Caldwell, C.C. The cannabinoid 2 receptor as a potential therapeutic target for sepsis. Endocr. Metab. Immune Disord. Drug Targets 2010, 10, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Orliac, M.L.; Peroni, R.; Celuch, S.M.; Adler-Graschinsky, E. Potentiation of anandamide effects in mesenteric beds isolated from endotoxemic rats. J. Pharmacol. Exp. Ther. 2003, 304, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Steffens, S.; Pacher, P. Targeting cannabinoid receptor CB2 in cardiovascular disorders: Promises and controversies. Br. J. Pharmacol. 2012, 167, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Rom, S.; Persidsky, Y. Cannabinoid receptor 2: Potential role in immunomodulation and neuroinflammation. J. Neuroimmune Pharmacol. 2013, 8, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Del Giudice, E.; Furegato, S.; Passarotto, M.; Leon, A. Cannabinoids ablate release of TNF-alpha in rat microglial cells stimulated with lypopolysaccharide. Glia 2003, 41, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.; Hwang, S.G.; Han, S.H.; Kaminski, N.E. Suppression of interleukin-2 by the putative endogenous cannabinoid 2-arachidonyl-glycerol is mediated through down-regulation of the nuclear factor of activated T cells. Mol. Pharmacol. 1998, 53, 676–683. [Google Scholar] [PubMed]
- Gallily, R.; Breuer, A.; Mechoulam, R. 2-Arachidonylglycerol, an endogenous cannabinoid, inhibits tumor necrosis factor-alpha production in murine macrophages, and in mice. Eur. J. Pharmacol. 2000, 406, R5–R7. [Google Scholar] [CrossRef]
- Tham, C.-S.; Whitaker, J.; Luo, L.; Webb, M. Inhibition of microglial fatty acid amide hydrolase modulates LPS stimulated release of inflammatory mediators. FEBS. Lett. 2007, 581, 2899–2904. [Google Scholar] [CrossRef] [PubMed]
- Naidu, P.; Kinsey, S.; Guo, T.; Cravatt, B.; Lichtman, A.H. Regulation of Inflammatory Pain by Inhibition of Fatty Acid Amide Hydrolase. J. Pharmacol. Exp. Ther. 2010, 334, 182. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J. Circulating Endocannabinoids: From Whence Do They Come and Where are They Going? Neuropsychopharmacology 2017. [Google Scholar] [CrossRef] [PubMed]
- Gouveia-Figueira, S.; Nording, M.L. Development and validation of a sensitive UPLC-ESI-MS/MS method for the simultaneous quantification of 15 endocannabinoids and related compounds in milk and other biofluids. Anal. Chem. 2014, 86, 1186–1195. [Google Scholar] [CrossRef] [PubMed]
- Hillard, C.J.; Weinlander, K.M.; Stuhr, K.L. Contributions of endocannabinoid signaling to psychiatric disorders in humans: Genetic and biochemical evidence. Neuroscience 2012, 204, 207–229. [Google Scholar] [CrossRef] [PubMed]
- Lam, P.M.; Marczylo, T.H.; Konje, J.C. Simultaneous measurement of three. N-acylethanolamides in human bio-matrices using ultra performance liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2010, 398, 2089–2097. [Google Scholar] [CrossRef] [PubMed]
- Vogeser, M.; Hauer, D.; Christina Azad, S.; Huber, E.; Storr, M.; Schelling, G. Release of anandamide from blood cells. Clin. Chem. Lab. Med. 2006, 44, 488–491. [Google Scholar] [CrossRef] [PubMed]
- Boldrup, L.; Wilson, S.; Barbier, A.; Fowler, C. A simple stopped assay for fatty acid amide hydrolase avoiding the use of a chloroform extraction phase. J. Biochem. Biophys. Methods 2004, 60, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Cable, J.C.; Tan, G.D.; Alexander, S.P.; O’sullivan, S.E. The activity of the endocannabinoid metabolising enzyme fatty acid amide hydrolase in subcutaneous adipocytes correlates with BMI in metabolically healthy humans. Lipids Health Dis. 2011, 10, 129. [Google Scholar] [CrossRef] [PubMed]
- Van der Poll, T.; van de Veerdonk, F.L.; Scicluna, B.P.; Netea, M.G. The immunopathology of sepsis and potential therapeutic targets. Nat. Rev. Immunol. 2017, 17, 407–420. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lafreniere, J.D.; Lehmann, C. Parameters of the Endocannabinoid System as Novel Biomarkers in Sepsis and Septic Shock. Metabolites 2017, 7, 55. https://doi.org/10.3390/metabo7040055
Lafreniere JD, Lehmann C. Parameters of the Endocannabinoid System as Novel Biomarkers in Sepsis and Septic Shock. Metabolites. 2017; 7(4):55. https://doi.org/10.3390/metabo7040055
Chicago/Turabian StyleLafreniere, J. Daniel, and Christian Lehmann. 2017. "Parameters of the Endocannabinoid System as Novel Biomarkers in Sepsis and Septic Shock" Metabolites 7, no. 4: 55. https://doi.org/10.3390/metabo7040055
APA StyleLafreniere, J. D., & Lehmann, C. (2017). Parameters of the Endocannabinoid System as Novel Biomarkers in Sepsis and Septic Shock. Metabolites, 7(4), 55. https://doi.org/10.3390/metabo7040055





