Metabolomic Analysis of Small Extracellular Vesicles Derived from Pancreatic Cancer Cells Cultured under Normoxia and Hypoxia
Abstract
1. Introduction
2. Results
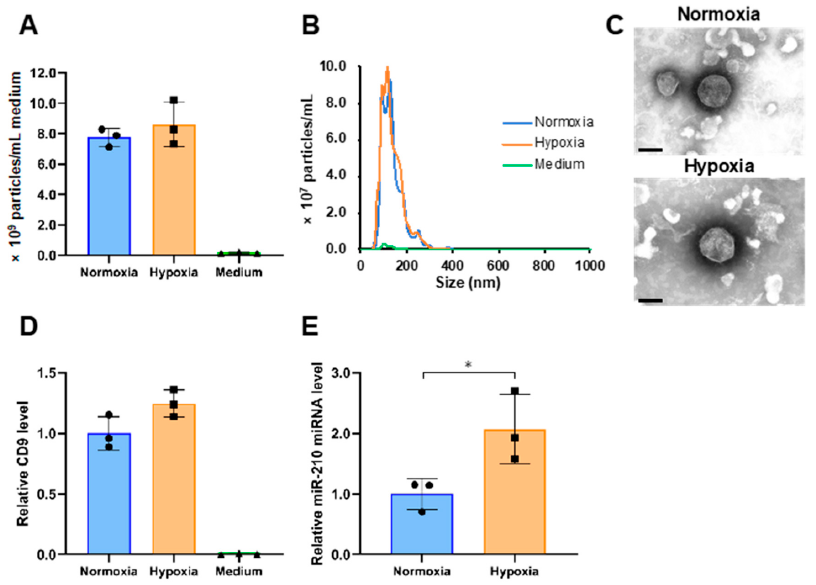
2.1. Isolation of sEVs Released from PANC-1 Cells Cultured under Normoxia and Hypoxia
2.2. Analysis of Hydrophilic Metabolites
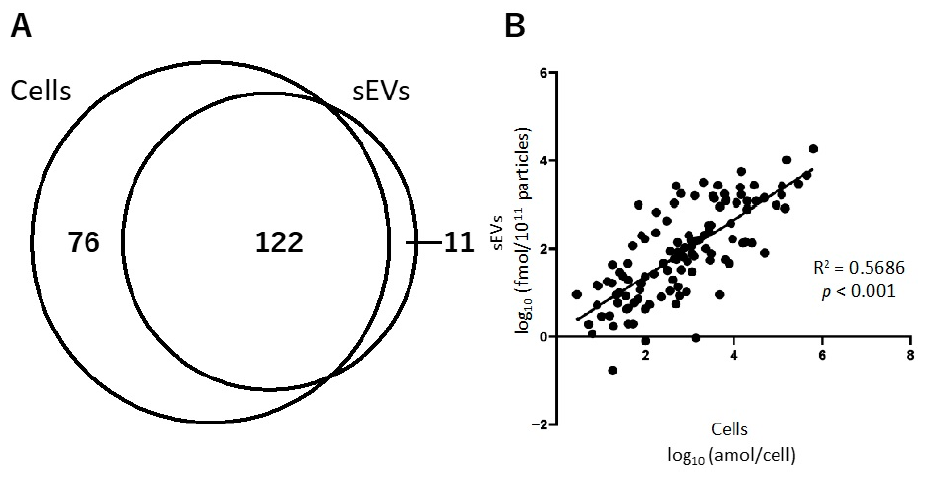
2.2.1. Relationship of Hydrophilic Metabolites in Cells and sEVs
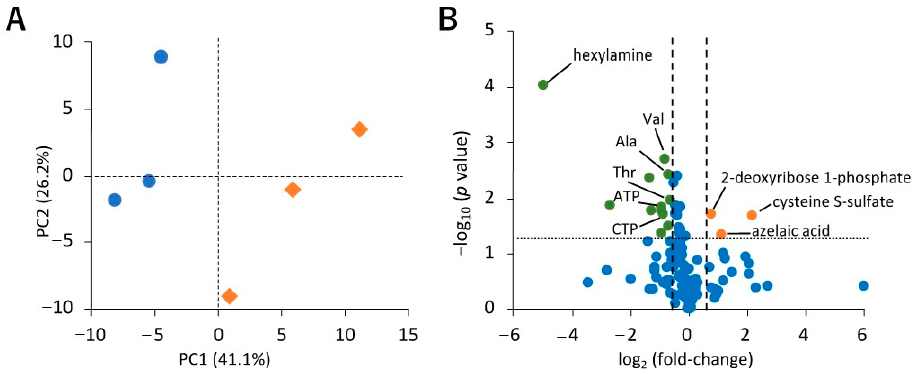
2.2.2. Effect of Hypoxic Stress on the Level of Hydrophilic Metabolites in sEVs
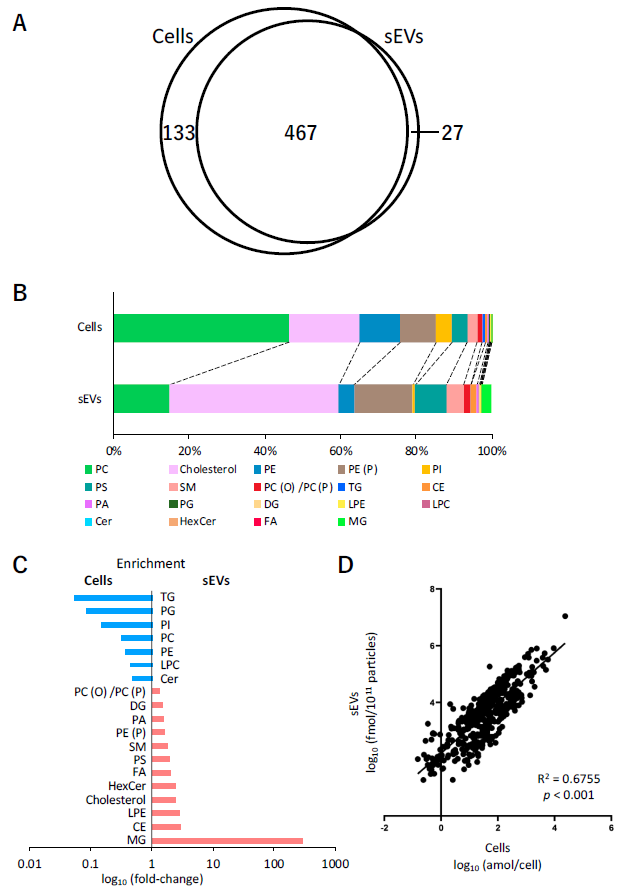
2.3. Lipid Analysis
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Isolation of sEVs
4.3. Extraction of Hydrophilic Metabolites from Cells
4.4. Extraction of Hydrophilic Metabolites from sEVs
4.5. Extraction of Lipids from Cells
4.6. Extraction of Lipids from sEVs
4.7. Analysis of Hydrophilic Metabolites
4.8. Lipidomic Analyses
4.9. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thery, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, A.; Costa-Silva, B.; Shen, T.L.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef] [PubMed]
- McCready, J.; Sims, J.D.; Chan, D.; Jay, D.G. Secretion of extracellular hsp90alpha via exosomes increases cancer cell motility: A role for plasminogen activation. BMC Cancer 2010, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Tadokoro, H.; Umezu, T.; Ohyashiki, K.; Hirano, T.; Ohyashiki, J.H. Exosomes derived from hypoxic leukemia cells enhance tube formation in endothelial cells. J. Biol. Chem. 2013, 288, 34343–34351. [Google Scholar] [CrossRef]
- Liu, W.; Glunde, K.; Bhujwalla, Z.M.; Raman, V.; Sharma, A.; Phang, J.M. Proline oxidase promotes tumor cell survival in hypoxic tumor microenvironments. Cancer Res. 2012, 72, 3677–3686. [Google Scholar] [CrossRef]
- Jeppesen, D.K.; Fenix, A.M.; Franklin, J.L.; Higginbotham, J.N.; Zhang, Q.; Zimmerman, L.J.; Liebler, D.C.; Ping, J.; Liu, Q.; Evans, R.; et al. Reassessment of Exosome Composition. Cell 2019, 177, 428–445.e418. [Google Scholar] [CrossRef]
- Laulagnier, K.; Motta, C.; Hamdi, S.; Roy, S.; Fauvelle, F.; Pageaux, J.F.; Kobayashi, T.; Salles, J.P.; Perret, B.; Bonnerot, C.; et al. Mast cell- and dendritic cell-derived exosomes display a specific lipid composition and an unusual membrane organization. Biochem. J. 2004, 380, 161–171. [Google Scholar] [CrossRef]
- Trajkovic, K.; Hsu, C.; Chiantia, S.; Rajendran, L.; Wenzel, D.; Wieland, F.; Schwille, P.; Brügger, B.; Simons, M. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 2008, 319, 1244–1247. [Google Scholar] [CrossRef]
- Llorente, A.; Skotland, T.; Sylvanne, T.; Kauhanen, D.; Rog, T.; Orlowski, A.; Vattulainen, I.; Ekroos, K.; Sandvig, K. Molecular lipidomics of exosomes released by PC-3 prostate cancer cells. Biochim. Biophys. Acta 2013, 1831, 1302–1309. [Google Scholar] [CrossRef]
- Chapuy-Regaud, S.; Dubois, M.; Plisson-Chastang, C.; Bonnefois, T.; Lhomme, S.; Bertrand-Michel, J.; You, B.; Simoneau, S.; Gleizes, P.E.; Flan, B.; et al. Characterization of the lipid envelope of exosome encapsulated HEV particles protected from the immune response. Biochimie 2017, 141, 70–79. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, L.; Baddour, J.; Achreja, A.; Bernard, V.; Moss, T.; Marini, J.C.; Tudawe, T.; Seviour, E.G.; San Lucas, F.A.; et al. Tumor microenvironment derived exosomes pleiotropically modulate cancer cell metabolism. eLife 2016, 5, 1–27. [Google Scholar] [CrossRef]
- Satoh, K.; Yachida, S.; Sugimoto, M.; Oshima, M.; Nakagawa, T.; Akamoto, S.; Tabata, S.; Saitoh, K.; Kato, K.; Sato, S.; et al. Global metabolic reprogramming of colorectal cancer occurs at adenoma stage and is induced by MYC. Proc. Natl. Acad. Sci. USA 2017, 114, E7697–E7706. [Google Scholar] [CrossRef]
- Sugimoto, M.; Wong, D.T.; Hirayama, A.; Soga, T.; Tomita, M. Capillary electrophoresis mass spectrometry-based saliva metabolomics identified oral, breast and pancreatic cancer-specific profiles. Metabolomics 2010, 6, 78–95. [Google Scholar] [CrossRef]
- Hirayama, A.; Kami, K.; Sugimoto, M.; Sugawara, M.; Toki, N.; Onozuka, H.; Kinoshita, T.; Saito, N.; Ochiai, A.; Tomita, M.; et al. Quantitative metabolome profiling of colon and stomach cancer microenvironment by capillary electrophoresis time-of-flight mass spectrometry. Cancer Res. 2009, 69, 4918–4925. [Google Scholar] [CrossRef]
- Wang, J.; Christison, T.T.; Misuno, K.; Lopez, L.; Huhmer, A.F.; Huang, Y.; Hu, S. Metabolomic profiling of anionic metabolites in head and neck cancer cells by capillary ion chromatography with Orbitrap mass spectrometry. Anal. Chem. 2014, 86, 5116–5124. [Google Scholar] [CrossRef]
- Hu, S.; Wang, J.; Ji, E.H.; Christison, T.; Lopez, L.; Huang, Y. Targeted Metabolomic Analysis of Head and Neck Cancer Cells Using High Performance Ion Chromatography Coupled with a Q Exactive HF Mass Spectrometer. Anal. Chem. 2015, 87, 6371–6379. [Google Scholar] [CrossRef]
- Petucci, C.; Zelenin, A.; Culver, J.A.; Gabriel, M.; Kirkbride, K.; Christison, T.T.; Gardell, S.J. Use of Ion Chromatography/Mass Spectrometry for Targeted Metabolite Profiling of Polar Organic Acids. Anal. Chem. 2016, 88, 11799–11803. [Google Scholar] [CrossRef]
- Cui, L.; Liu, J.; Yan, X.; Hu, S. Identification of Metabolite Biomarkers for Gout Using Capillary Ion Chromatography with Mass Spectrometry. Anal. Chem. 2017, 89, 11737–11743. [Google Scholar] [CrossRef]
- Hirayama, A.; Tabata, S.; Kudo, R.; Hasebe, M.; Suzuki, K.; Tomita, M.; Soga, T. The use of a double coaxial electrospray ionization sprayer improves the peak resolutions of anionic metabolites in capillary ion chromatography-mass spectrometry. J. Chromatogr. A 2020, 1619, 460914. [Google Scholar] [CrossRef]
- Takeda, H.; Izumi, Y.; Takahashi, M.; Paxton, T.; Tamura, S.; Koike, T.; Yu, Y.; Kato, N.; Nagase, K.; Shiomi, M.; et al. Widely-targeted quantitative lipidomics method by supercritical fluid chromatography triple quadrupole mass spectrometry. J. Lipid Res. 2018, 59, 1283–1293. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Hori, M.; Matsuda, T.; Shibata, A.; Katanoda, K.; Sobue, T.; Nishimoto, H.; Japan Cancer Surveillance Research Group. Cancer incidence and incidence rates in Japan in 2009: A study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn. J. Clin. Oncol. 2015, 45, 884–891. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.K.; Zhou, Y.; Cheng, C.; Zhong, S.; Wu, H.Q.; Wang, B.; Fan, P.; Xiong, J.X.; Yang, H.J.; Wu, H.S. Overexpression of membrane-type 2 matrix metalloproteinase induced by hypoxia-inducible factor-1alpha in pancreatic cancer: Implications for tumor progression and prognosis. Mol. Clin. Oncol. 2014, 2, 973–981. [Google Scholar] [CrossRef] [PubMed]
- Schodel, J.; Oikonomopoulos, S.; Ragoussis, J.; Pugh, C.W.; Ratcliffe, P.J.; Mole, D.R. High-resolution genome-wide mapping of HIF-binding sites by ChIP-seq. Blood 2011, 117, e207–e217. [Google Scholar] [CrossRef]
- Eales, K.L.; Hollinshead, K.E.; Tennant, D.A. Hypoxia and metabolic adaptation of cancer cells. Oncogenesis 2016, 5, e190. [Google Scholar] [CrossRef]
- Jung, K.O.; Youn, H.; Lee, C.H.; Kang, K.W.; Chung, J.K. Visualization of exosome-mediated miR-210 transfer from hypoxic tumor cells. Oncotarget 2017, 8, 9899–9910. [Google Scholar] [CrossRef]
- Berchem, G.; Noman, M.Z.; Bosseler, M.; Paggetti, J.; Baconnais, S.; Le Cam, E.; Nanbakhsh, A.; Moussay, E.; Mami-Chouaib, F.; Janji, B.; et al. Hypoxic tumor-derived microvesicles negatively regulate NK cell function by a mechanism involving TGF-beta and miR23a transfer. Oncoimmunology 2016, 5, e1062968. [Google Scholar] [CrossRef]
- Chen, X.; Ying, X.; Wang, X.; Wu, X.; Zhu, Q.; Wang, X. Exosomes derived from hypoxic epithelial ovarian cancer deliver microRNA-940 to induce macrophage M2 polarization. Oncol. Rep. 2017, 38, 522–528. [Google Scholar] [CrossRef]
- Wang, X.; Luo, G.; Zhang, K.; Cao, J.; Huang, C.; Jiang, T.; Liu, B.; Su, L.; Qiu, Z. Hypoxic Tumor-Derived Exosomal miR-301a Mediates M2 Macrophage Polarization via PTEN/PI3Kgamma to Promote Pancreatic Cancer Metastasis. Cancer Res. 2018, 78, 4586–4598. [Google Scholar] [CrossRef]
- Ye, S.B.; Zhang, H.; Cai, T.T.; Liu, Y.N.; Ni, J.J.; He, J.; Peng, J.Y.; Chen, Q.Y.; Mo, H.Y.; Jun, C.; et al. Exosomal miR-24-3p impedes T-cell function by targeting FGF11 and serves as a potential prognostic biomarker for nasopharyngeal carcinoma. J. Pathol. 2016, 240, 329–340. [Google Scholar] [CrossRef]
- Shao, C.; Yang, F.; Miao, S.; Liu, W.; Wang, C.; Shu, Y.; Shen, H. Role of hypoxia-induced exosomes in tumor biology. Mol. Cancer 2018, 17, 120. [Google Scholar] [CrossRef]
- Patton, M.C.; Zubair, H.; Khan, M.A.; Singh, S.; Singh, A.P. Hypoxia alters the release and size distribution of extracellular vesicles in pancreatic cancer cells to support their adaptive survival. J. Cell Biochem. 2020, 121, 828–839. [Google Scholar] [CrossRef]
- King, H.W.; Michael, M.Z.; Gleadle, J.M. Hypoxic enhancement of exosome release by breast cancer cells. BMC Cancer 2012, 12, 421. [Google Scholar] [CrossRef]
- Luo, P.; Mao, K.; Xu, J.; Wu, F.; Wang, X.; Wang, S.; Zhou, M.; Duan, L.; Tan, Q.; Ma, G.; et al. Metabolic characteristics of large and small extracellular vesicles from pleural effusion reveal biomarker candidates for the diagnosis of tuberculosis and malignancy. J. Extracell. Vesicles 2020, 9, 1790158. [Google Scholar] [CrossRef]
- Puhka, M.; Takatalo, M.; Nordberg, M.E.; Valkonen, S.; Nandania, J.; Aatonen, M.; Yliperttula, M.; Laitinen, S.; Velagapudi, V.; Mirtti, T.; et al. Metabolomic Profiling of Extracellular Vesicles and Alternative Normalization Methods Reveal Enriched Metabolites and Strategies to Study Prostate Cancer-Related Changes. Theranostics 2017, 7, 3824–3841. [Google Scholar] [CrossRef]
- Vallabhaneni, K.C.; Penfornis, P.; Dhule, S.; Guillonneau, F.; Adams, K.V.; Mo, Y.Y.; Xu, R.; Liu, Y.; Watabe, K.; Vemuri, M.C.; et al. Extracellular vesicles from bone marrow mesenchymal stem/stromal cells transport tumor regulatory microRNA, proteins, and metabolites. Oncotarget 2015, 6, 4953–4967. [Google Scholar] [CrossRef]
- Tadokoro, H.; Hirayama, A.; Kudo, R.; Hasebe, M.; Yoshioka, Y.; Matsuzaki, J.; Yamamoto, Y.; Sugimoto, M.; Soga, T.; Ochiya, T. Adenosine leakage from perforin-burst extracellular vesicles inhibits perforin secretion by cytotoxic T-lymphocytes. PLoS ONE 2020, 15, e0231430. [Google Scholar] [CrossRef]
- Ludwig, N.; Gillespie, D.G.; Reichert, T.E.; Jackson, E.K.; Whiteside, T.L. Purine Metabolites in Tumor-Derived Exosomes May Facilitate Immune Escape of Head and Neck Squamous Cell Carcinoma. Cancers 2020, 12, 1602. [Google Scholar] [CrossRef]
- Beccard, I.J.; Hofmann, L.; Schroeder, J.C.; Ludwig, S.; Laban, S.; Brunner, C.; Lotfi, R.; Hoffmann, T.K.; Jackson, E.K.; Schuler, P.J.; et al. Immune Suppressive Effects of Plasma-Derived Exosome Populations in Head and Neck Cancer. Cancers 2020, 12, 1997. [Google Scholar] [CrossRef]
- Pugmire, M.J.; Ealick, S.E. Structural analyses reveal two distinct families of nucleoside phosphorylases. Biochem. J. 2002, 361, 1–25. [Google Scholar] [CrossRef]
- Kitazono, M.; Takebayashi, Y.; Ishitsuka, K.; Takao, S.; Tani, A.; Furukawa, T.; Miyadera, K.; Yamada, Y.; Aikou, T.; Akiyama, S. Prevention of hypoxia-induced apoptosis by the angiogenic factor thymidine phosphorylase. Biochem. Biophys. Res. Commun. 1998, 253, 797–803. [Google Scholar] [CrossRef]
- Vara, D.; Watt, J.M.; Fortunato, T.M.; Mellor, H.; Burgess, M.; Wicks, K.; Mace, K.; Reeksting, S.; Lubben, A.; Wheeler-Jones, C.P.D.; et al. Direct Activation of NADPH Oxidase 2 by 2-Deoxyribose-1-Phosphate Triggers Nuclear Factor Kappa B-Dependent Angiogenesis. Antioxid. Redox Signal. 2018, 28, 110–130. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, X.; Yang, Y.; Chen, W.; Zhang, K.; Teng, B.; Huang, C.; Zhao, Q.; Qiu, Z. Hypoxic Tumor-Derived Exosomal Long Noncoding RNA UCA1 Promotes Angiogenesis via miR-96-5p/AMOTL2 in Pancreatic Cancer. Mol. Ther. Nucleic Acids 2020, 22, 179–195. [Google Scholar] [CrossRef]
- Huang, J.; Weinstein, S.J.; Kitahara, C.M.; Karoly, E.D.; Sampson, J.N.; Albanes, D. A prospective study of serum metabolites and glioma risk. Oncotarget 2017, 8, 70366–70377. [Google Scholar] [CrossRef]
- Raghavamenon, A.; Garelnabi, M.; Babu, S.; Aldrich, A.; Litvinov, D.; Parthasarathy, S. Alpha-tocopherol is ineffective in preventing the decomposition of preformed lipid peroxides and may promote the accumulation of toxic aldehydes: A potential explanation for the failure of antioxidants to affect human atherosclerosis. Antioxid. Redox Signal. 2009, 11, 1237–1248. [Google Scholar] [CrossRef]
- Yu, L.; Aa, J.; Xu, J.; Sun, M.; Qian, S.; Cheng, L.; Yang, S.; Shi, R. Metabolomic phenotype of gastric cancer and precancerous stages based on gas chromatography time-of-flight mass spectrometry. J. Gastroenterol. Hepatol. 2011, 26, 1290–1297. [Google Scholar] [CrossRef]
- Brown, D.G.; Rao, S.; Weir, T.L.; O’Malia, J.; Bazan, M.; Brown, R.J.; Ryan, E.P. Metabolomics and metabolic pathway networks from human colorectal cancers, adjacent mucosa, and stool. Cancer Metab. 2016, 4, 11. [Google Scholar] [CrossRef]
- Warburg, O.; Negelein, E.; Posener, K. Versuche an Überlebendem Carcinomgewebe. Klinische Wochenschrift 1924, 3, 1062–1064. [Google Scholar] [CrossRef]
- Biancur, D.E.; Paulo, J.A.; Malachowska, B.; Quiles Del Rey, M.; Sousa, C.M.; Wang, X.; Sohn, A.S.W.; Chu, G.C.; Gygi, S.P.; Harper, J.W.; et al. Compensatory metabolic networks in pancreatic cancers upon perturbation of glutamine metabolism. Nat. Commun. 2017, 8, 15965. [Google Scholar] [CrossRef]
- Qin, C.; Yang, G.; Yang, J.; Ren, B.; Wang, H.; Chen, G.; Zhao, F.; You, L.; Wang, W.; Zhao, Y. Metabolism of pancreatic cancer: Paving the way to better anticancer strategies. Mol. Cancer 2020, 19, 50. [Google Scholar] [CrossRef]
- Yoo, H.C.; Park, S.J.; Nam, M.; Kang, J.; Kim, K.; Yeo, J.H.; Kim, J.K.; Heo, Y.; Lee, H.S.; Lee, M.Y.; et al. A Variant of SLC1A5 Is a Mitochondrial Glutamine Transporter for Metabolic Reprogramming in Cancer Cells. Cell Metab. 2020, 31, 267–283.e212. [Google Scholar] [CrossRef] [PubMed]
- Vidal, M.; Sainte-Marie, J.; Philippot, J.R.; Bienvenue, A. Asymmetric distribution of phospholipids in the membrane of vesicles released during in vitro maturation of guinea pig reticulocytes: Evidence precluding a role for “aminophospholipid translocase”. J. Cell Physiol. 1989, 140, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Lydic, T.A.; Townsend, S.; Adda, C.G.; Collins, C.; Mathivanan, S.; Reid, G.E. Rapid and comprehensive ’shotgun’ lipidome profiling of colorectal cancer cell derived exosomes. Methods 2015, 87, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Nishida-Aoki, N.; Izumi, Y.; Takeda, H.; Takahashi, M.; Ochiya, T.; Bamba, T. Lipidomic Analysis of Cells and Extracellular Vesicles from High- and Low-Metastatic Triple-Negative Breast Cancer. Metabolites 2020, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Skotland, T.; Sagini, K.; Sandvig, K.; Llorente, A. An emerging focus on lipids in extracellular vesicles. Adv. Drug Deliv. Rev. 2020, 159, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Sagar, G.; Sah, R.P.; Javeed, N.; Dutta, S.K.; Smyrk, T.C.; Lau, J.S.; Giorgadze, N.; Tchkonia, T.; Kirkland, J.L.; Chari, S.T.; et al. Pathogenesis of pancreatic cancer exosome-induced lipolysis in adipose tissue. Gut 2016, 65, 1165–1174. [Google Scholar] [CrossRef]
- Zhang, H.; Freitas, D.; Kim, H.S.; Fabijanic, K.; Li, Z.; Chen, H.; Mark, M.T.; Molina, H.; Martin, A.B.; Bojmar, L.; et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat. Cell Biol. 2018, 20, 332–343. [Google Scholar] [CrossRef] [PubMed]
| Cells (fmol/cell) | sEVs (pmol/1011 Particles) | ||||
|---|---|---|---|---|---|
| Rank | Metabolite | Amount | Metabolite | Amount | Cell Rank |
| 1 | Phosphorylcholine | 628 | Phosphorylcholine | 18.0 | 1 |
| 2 | Glutathione (reduced) | 617 | Glycerophosphorylcholine | 10.2 | 5 |
| 3 | Glu | 447 | Arg | 5.7 | 24 |
| 4 | Ethanolamine phosphate | 294 | Glu | 4.7 | 3 |
| 5 | Glycerophosphorylcholine | 159 | Lys | 3.2 | 56 |
| 6 | Asp | 148 | Ethanolamine phosphate | 2.9 | 4 |
| 7 | Gln | 124 | Inosine | 2.8 | − |
| 8 | Gly | 122 | UDP-N-acetylglucosamine | 2.7 | 15 |
| 9 | Pro | 92.1 | ADP | 2.7 | 40 |
| 10 | Lactic acid | 77.0 | Gln | 2.6 | 7 |
| 11 | ATP | 49.9 | Glucose 1-phosphate | 2.6 | 95 |
| 12 | Gly Gly | 49.2 | Ala | 2.5 | 26 |
| 13 | Asn | 48.4 | GDP | 1.7 | 86 |
| 14 | N-Acetylaspartate | 32.2 | UMP | 1.7 | 35 |
| 15 | UDP-N-acetylglucosamine | 28.7 | N,N-dimethylglycine | 1.7 | − |
| 16 | UTP | 26.0 | UDP-glucose | 1.7 | 25 |
| 17 | Citric acid | 20.3 | Gly | 1.6 | 8 |
| 18 | Creatine | 20.0 | Cytidine | 1.6 | − |
| 19 | beta-Ala | 19.2 | Uridine | 1.6 | − |
| 20 | Malic acid | 18.4 | UDP | 1.6 | 63 |
| Lipid Class | Abbreviation | Number of Lipids Detected | ||||
|---|---|---|---|---|---|---|
| Cells | sEVs | Common | Only Cells | Only sEVs | ||
| Free Fatty Acid | FA | 3 | 3 | 1 | 2 | 2 |
| Lysophosphatidylcholine | LPC | 19 | 9 | 9 | 10 | 0 |
| Lysophosphatidylethanolamine | LPE | 14 | 15 | 14 | 0 | 1 |
| Phosphatidylcholine | PC | 80 | 50 | 48 | 32 | 2 |
| Alkyl-Acyl Phosphatidylcholine/ Alkenyl-Acyl Phosphatidylcholine | PC (O)/ PC (P) | 27 | 27 | 26 | 1 | 1 |
| Phosphatidylethanolamine | PE | 90 | 60 | 60 | 30 | 0 |
| Alkenyl-Acyl Phosphatidylethanolamine | PE (P) | 79 | 73 | 72 | 7 | 1 |
| Phosphatidylglycerol | PG | 16 | 9 | 9 | 7 | 0 |
| Phosphatidic Acid | PA | 17 | 15 | 15 | 2 | 0 |
| Phosphatidylinositol | PI | 82 | 53 | 53 | 29 | 0 |
| Phosphatidylserine | PS | 51 | 51 | 51 | 0 | 0 |
| Sphingomyelin | SM | 15 | 13 | 13 | 2 | 0 |
| Ceramide | Cer | 13 | 12 | 12 | 1 | 0 |
| Hexosylceramides | HexCer | 8 | 8 | 8 | 0 | 0 |
| Cholesterol | Cholesterol | 1 | 1 | 1 | 0 | 0 |
| Cholesterol Ester | CE | 5 | 6 | 5 | 0 | 1 |
| Monoacylglycerol | MG | 4 | 9 | 2 | 2 | 7 |
| Diacylglycerol | DG | 43 | 51 | 39 | 4 | 12 |
| Triacylglycerol | TG | 33 | 29 | 29 | 4 | 0 |
| Total | 600 | 494 | 467 | 133 | 27 | |
| Lipid Class | Cells (amol/cell) | sEVs (pmol/1011 Particles) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Normoxia | Hypoxia | p Value | Normoxia | Hypoxia | p Value | |||||||||
| Mean | ± | SD | Mean | ± | SD | Mean | ± | SD | Mean | ± | SD | |||
| Free Fatty Acid (FA) | 18.2 | ± | 6.7 | 9.4 | ± | 12.9 | 0.3538 | 7.8 | ± | 6.5 | 3.6 | ± | 3.2 | 0.3631 |
| Lysophosphatidylcholine (LPC) | 83.2 | ± | 10.4 | 151 | ± | 20.9 | 0.0074 | 6.6 | ± | 3.4 | 5.7 | ± | 2.1 | 0.7302 |
| Lysophosphatidylethanolamine (LPE) | 104 | ± | 17.9 | 193 | ± | 25.9 | 0.0082 | 56.8 | ± | 23.8 | 42.5 | ± | 16.7 | 0.4420 |
| Phosphatidylcholine (PC) | 59338 | ± | 8356 | 93637 | ± | 11729 | 0.0146 | 3608 | ± | 715 | 3388 | ± | 860 | 0.7506 |
| Alkyl-Acyl Phosphatidylcholine (PC (O))/ Alkenyl-Acyl Phosphatidylcholine (PC (P)) | 1689 | ± | 213 | 2870 | ± | 247 | 0.0033 | 452 | ± | 105 | 379 | ± | 91.5 | 0.4192 |
| Phosphatidylethanolamine (PE) | 13766 | ± | 706 | 21431 | ± | 279 | 0.0001 | 999 | ± | 230 | 899 | ± | 214 | 0.6105 |
| Alkenyl-Acyl Phosphatidylethanolamine (PE (P)) | 11795 | ± | 1200 | 22803 | ± | 5034 | 0.0211 | 3792 | ± | 910 | 2927 | ± | 472 | 0.2172 |
| Phosphatidylglycerol (PG) | 243 | ± | 20.7 | 397 | ± | 39.5 | 0.0040 | 4.1 | ± | 1.1 | 2.8 | ± | 0.7 | 0.1570 |
| Phosphatidic Acid (PA) | 524 | ± | 28.1 | 953 | ± | 125 | 0.0043 | 161 | ± | 30.8 | 153 | ± | 37.1 | 0.8030 |
| Phosphatidylinositol (PI) | 5381 | ± | 464 | 8851 | ± | 468 | 0.0008 | 162 | ± | 36.7 | 139 | ± | 32.8 | 0.4580 |
| Phosphatidylserine (PS) | 5257 | ± | 377 | 8850 | ± | 491 | 0.0006 | 2038 | ± | 352 | 1970 | ± | 444 | 0.8456 |
| Sphingomyelin (SM) | 3279 | ± | 220 | 5346 | ± | 274 | 0.0005 | 1134 | ± | 250 | 938 | ± | 202 | 0.3501 |
| Ceramide (Cer) | 77.3 | ± | 8.1 | 98.6 | ± | 4.1 | 0.0152 | 7.2 | ± | 2.1 | 5.2 | ± | 1.2 | 0.2197 |
| Hexosylceramides (Hexcer) | 68.9 | ± | 4.3 | 125 | ± | 7.0 | 0.0003 | 32.3 | ± | 5.9 | 31.6 | ± | 6.5 | 0.8995 |
| Cholesterol | 23504 | ± | 2817 | 36016 | ± | 3118 | 0.0067 | 11080 | ± | 2242 | 9346 | ± | 2925 | 0.4608 |
| Cholesterol Ester (CE) | 704 | ± | 172 | 739 | ± | 152 | 0.8063 | 388 | ± | 16.1 | 492 | ± | 147 | 0.2923 |
| Monoacylglycerol (MG) | 10.5 | ± | 4.5 | 21.6 | ± | 8.6 | 0.1174 | 654 | ± | 384 | 524 | ± | 252 | 0.6509 |
| Diacylglycerol (DG) | 174 | ± | 22.1 | 275 | ± | 38.7 | 0.0172 | 49.9 | ± | 14.5 | 42.1 | ± | 10.1 | 0.4855 |
| Triacylglycerol (TG) | 1104 | ± | 308 | 1755 | ± | 457 | 0.1102 | 10.7 | ± | 5.9 | 7.6 | ± | 5.1 | 0.5210 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayasaka, R.; Tabata, S.; Hasebe, M.; Ikeda, S.; Ohnuma, S.; Mori, M.; Soga, T.; Tomita, M.; Hirayama, A. Metabolomic Analysis of Small Extracellular Vesicles Derived from Pancreatic Cancer Cells Cultured under Normoxia and Hypoxia. Metabolites 2021, 11, 215. https://doi.org/10.3390/metabo11040215
Hayasaka R, Tabata S, Hasebe M, Ikeda S, Ohnuma S, Mori M, Soga T, Tomita M, Hirayama A. Metabolomic Analysis of Small Extracellular Vesicles Derived from Pancreatic Cancer Cells Cultured under Normoxia and Hypoxia. Metabolites. 2021; 11(4):215. https://doi.org/10.3390/metabo11040215
Chicago/Turabian StyleHayasaka, Ryosuke, Sho Tabata, Masako Hasebe, Satsuki Ikeda, Sumiko Ohnuma, Masaru Mori, Tomoyoshi Soga, Masaru Tomita, and Akiyoshi Hirayama. 2021. "Metabolomic Analysis of Small Extracellular Vesicles Derived from Pancreatic Cancer Cells Cultured under Normoxia and Hypoxia" Metabolites 11, no. 4: 215. https://doi.org/10.3390/metabo11040215
APA StyleHayasaka, R., Tabata, S., Hasebe, M., Ikeda, S., Ohnuma, S., Mori, M., Soga, T., Tomita, M., & Hirayama, A. (2021). Metabolomic Analysis of Small Extracellular Vesicles Derived from Pancreatic Cancer Cells Cultured under Normoxia and Hypoxia. Metabolites, 11(4), 215. https://doi.org/10.3390/metabo11040215









