Metabolically Healthy Obesity—Heterogeneity in Definitions and Unconventional Factors
Abstract
1. Introduction
2. The Diverse Weight-Metabolic Phenotypes
2.1. The Metabolically Unhealthy Normal-Weight Phenotype
2.2. The Metabolically Healthy Obese Phenotype
The Multiple Definitions of Metabolically Healthy Obesity
2.3. The Metabolically Unhealthy Obese Phenotype
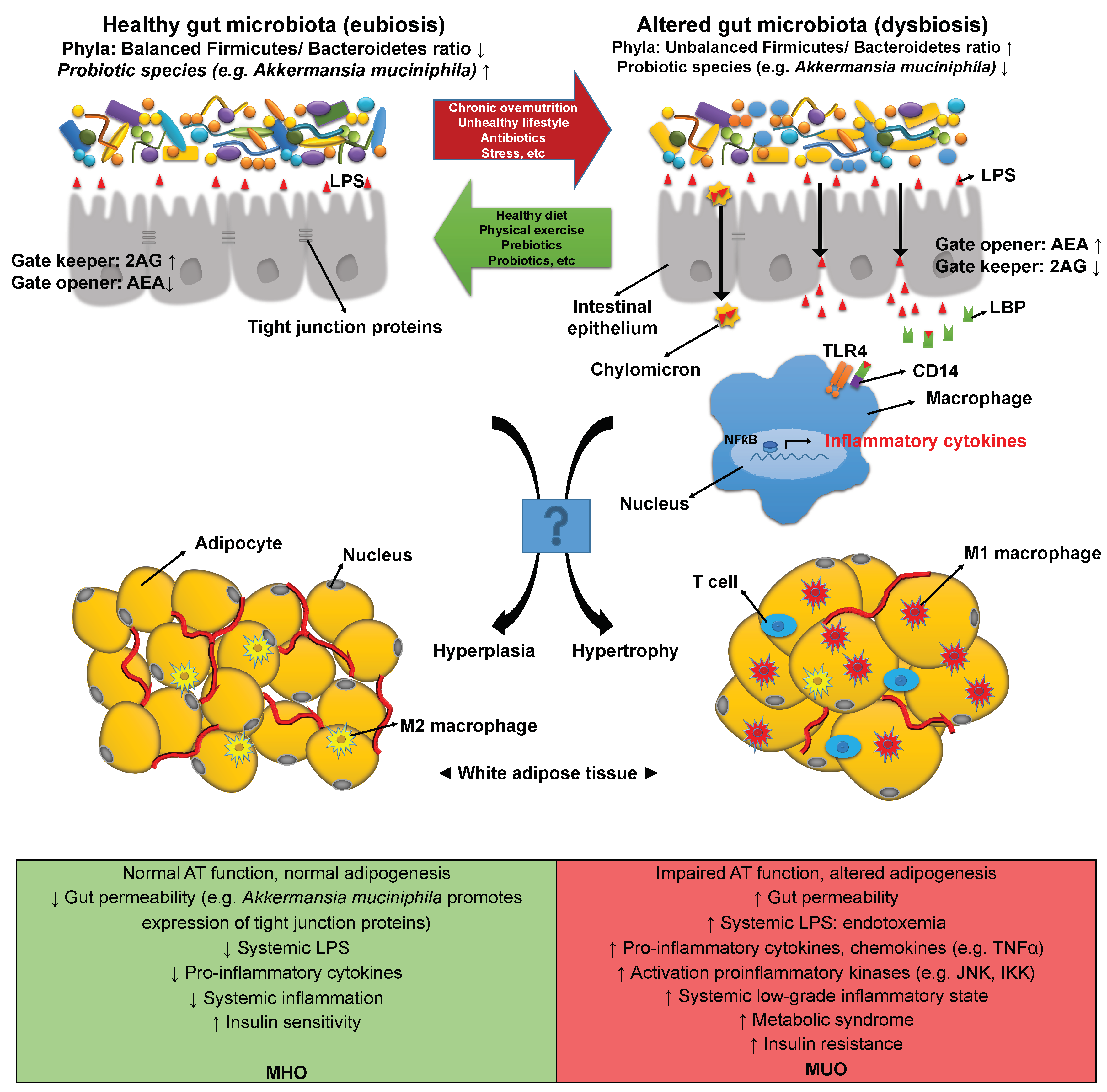
3. Metabolic Inflammation
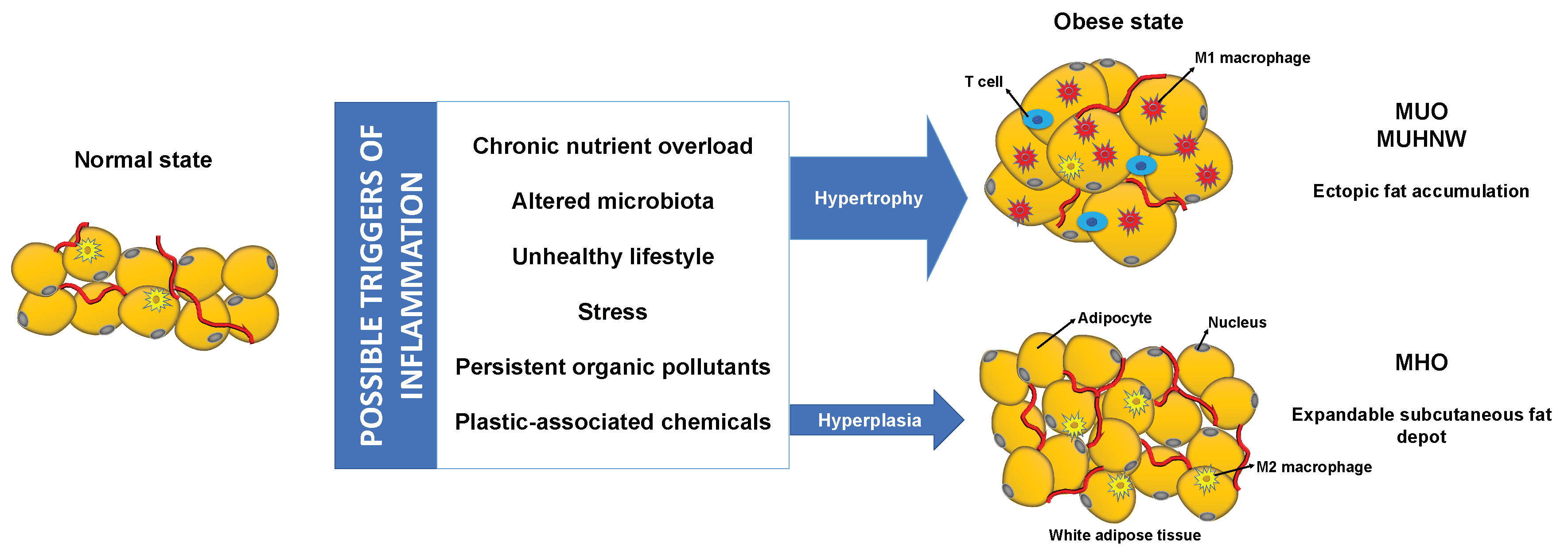
Adipose Tissue Inflammation and Pathogenesis of Obesity
Adipose Tissue Cellularity, Remodeling, and Inflammation
Adipose Tissue Macrophages
Patterns of Adipose Tissue Distribution
Endocrine Disrupting Chemicals: Adipose Tissue Persistent Organic Pollutants and Plastic-Associated Chemicals
Gut Microbiota, Endocannabinoid System, and Inflammasome
Nutrient Intake and Dietary Pattern
4. Conclusions
Funding
Conflicts of Interest
References
- WHO. Obesity and Overweight; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Mokdad, A.H.; Ford, E.S.; Bowman, B.A.; Dietz, W.H.; Vinicor, F.; Bales, V.S.; Marks, J.S. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA 2003, 289, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.-J.; Hyun, Y.J.; Kim, O.Y.; Kim, J.Y.; Jang, Y.; Lee, J.H. Weight loss effect on inflammation and LDL oxidation in metabolically healthy but obese (MHO) individuals: low inflammation and LDL oxidation in MHO women. Int. J. Obes. 2006, 30, 1529–1534. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-L.; Lin, W.-Y.; Lee, L.-T.; Wang, H.-H.; Lee, W.-J.; Huang, K.-C. Metabolic syndrome is related to nonalcoholic steatohepatitis in severely obese subjects. Obes. Surg. 2007, 17, 1457–1463. [Google Scholar] [CrossRef] [PubMed]
- Arnlov, J.; Ingelsson, E.; Sundstrom, J.; Lind, L. Impact of Body Mass Index and the Metabolic Syndrome on the Risk of Cardiovascular Disease and Death in Middle-Aged Men. Circulation 2010, 121, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.M. Metabolically healthy obesity: Definitions, determinants and clinical implications. Rev. Endocr. Metab. Disord. 2013, 14, 219–227. [Google Scholar] [CrossRef]
- Blüher, M. Are metabolically healthy obese individuals really healthy? Eur. J. Endocrinol. 2014, 171, R209–R219. [Google Scholar] [CrossRef]
- Phillips, C.M.; Perry, I.J. Does Inflammation Determine Metabolic Health Status in Obese and Nonobese Adults? J. Clin. Endocrinol. Metab. 2013, 98, E1610–E1619. [Google Scholar] [CrossRef]
- Ortega, F.B.; Lee, D.; Katzmarzyk, P.T.; Ruiz, J.R.; Sui, X.; Church, T.S.; Blair, S.N. The intriguing metabolically healthy but obese phenotype: cardiovascular prognosis and role of fitness. Eur. Heart J. 2013, 34, 389–397. [Google Scholar] [CrossRef]
- Manu, P.; Ionescu-Tirgoviste, C.; Tsang, J.; Napolitano, B.A.; Lesser, M.L.; Correll, C.U. Dysmetabolic Signals in “Metabolically Healthy” Obesity. Obes. Res. Clin. Pract. 2012, 6, e9–e20. [Google Scholar] [CrossRef]
- Phillips, C.M.; Dillon, C.; Harrington, J.M.; McCarthy, V.J.C.; Kearney, P.M.; Fitzgerald, A.P.; Perry, I.J. Defining Metabolically Healthy Obesity: Role of Dietary and Lifestyle Factors. PLoS ONE 2013, 8, e76188. [Google Scholar] [CrossRef]
- Iacobellis, G.; Ribaudo, M.C.; Zappaterreno, A.; Iannucci, C.V.; Leonetti, F. Prevalence of Uncomplicated Obesity in an Italian Obese Population. Obes. Res. 2005, 13, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Blüher, M. The distinction of metabolically ‘healthy’ from ‘unhealthy’ obese individuals. Curr. Opin. Lipidol. 2010, 21, 38–43. [Google Scholar] [CrossRef]
- Karelis, A.D. Obesity: To be obese—does it matter if you are metabolically healthy? Nat. Rev. Endocrinol. 2011, 7, 699–700. [Google Scholar] [CrossRef] [PubMed]
- Calori, G.; Lattuada, G.; Piemonti, L.; Garancini, M.P.; Ragogna, F.; Villa, M.; Mannino, S.; Crosignani, P.; Bosi, E.; Luzi, L.; et al. Prevalence, Metabolic Features, and Prognosis of Metabolically Healthy Obese Italian Individuals: The Cremona Study. Diabetes Care 2011, 34, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Rey-López, J.P.; de Rezende, L.F.; Pastor-Valero, M.; Tess, B.H. The prevalence of metabolically healthy obesity: a systematic review and critical evaluation of the definitions used. Obes. Rev. 2014, 15, 781–790. [Google Scholar] [CrossRef]
- Mongraw-Chaffin, M.; Foster, M.C.; Anderson, C.A.M.; Burke, G.L.; Haq, N.; Kalyani, R.R.; Ouyang, P.; Sibley, C.T.; Tracy, R.; Woodward, M.; et al. Metabolically Healthy Obesity, Transition to Metabolic Syndrome, and Cardiovascular Risk. J. Am. Coll. Cardiol. 2018, 71, 1857–1865. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Schneider, S.H.; Berchtold, P. The “metabolically-obese” normal-weight individual. Am. J. Clin. Nutr. 1981, 34, 1617–1621. [Google Scholar] [CrossRef]
- Tremmel, M.; Gerdtham, U.-G.; Nilsson, P.; Saha, S. Economic Burden of Obesity: A Systematic Literature Review. Int. J. Environ. Res. Public Health 2017, 14, 435. [Google Scholar] [CrossRef]
- Blüher, M. Are there still healthy obese patients? Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 341–346. [Google Scholar] [CrossRef]
- Mutie, P.M.; Giordano, G.N.; Franks, P.W. Lifestyle precision medicine: the next generation in type 2 diabetes prevention? BMC Med. 2017, 15, 171. [Google Scholar] [CrossRef]
- Monteiro, R.; Azevedo, I. Chronic inflammation in obesity and the metabolic syndrome. Mediat. Inflamm. 2010, 2010, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P.; Wood, I.S. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br. J. Nutr. 2004, 92, 347. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.; Teixeira, D.; Calhau, C. Estrogen signaling in metabolic inflammation. Mediat. Inflamm. 2014, 2014, 615917. [Google Scholar] [CrossRef]
- Gregor, M.F.; Hotamisligil, G.S. Inflammatory Mechanisms in Obesity. Annu. Rev. Immunol. 2011, 29, 415–445. [Google Scholar] [CrossRef]
- González, A.S.; Guerrero, D.B.; Soto, M.B.; Díaz, S.P.; Martinez-Olmos, M.; Vidal, O. Metabolic syndrome, insulin resistance and the inflammation markers C-reactive protein and ferritin. Eur. J. Clin. Nutr. 2006, 60, 802–809. [Google Scholar] [CrossRef]
- Wannamethee, S.G.; Lowe, G.D.O.; Shaper, A.G.; Rumley, A.; Lennon, L.; Whincup, P.H. The metabolic syndrome and insulin resistance: relationship to haemostatic and inflammatory markers in older non-diabetic men. Atherosclerosis 2005, 181, 101–108. [Google Scholar] [CrossRef]
- Goossens, G.H.; Blaak, E.E. Adipose tissue dysfunction and impaired metabolic health in human obesity: a matter of oxygen? Front. Endocrinol. (Lausanne) 2015, 6, 55. [Google Scholar] [CrossRef]
- Bremer, A.A.; Jialal, I. Adipose Tissue Dysfunction in Nascent Metabolic Syndrome. J. Obes. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Durward, C.M.; Hartman, T.J.; Nickols-Richardson, S.M. All-Cause Mortality Risk of Metabolically Healthy Obese Individuals in NHANES III. J. Obes. 2012, 2012, 460321. [Google Scholar] [CrossRef]
- Boonchaya-anant, P.; Apovian, C.M. Metabolically Healthy Obesity—Does it Exist? Curr. Atheroscler. Rep. 2014, 16, 441. [Google Scholar] [CrossRef]
- Roberson, L.L.; Aneni, E.C.; Maziak, W.; Agatston, A.; Feldman, T.; Rouseff, M.; Tran, T.; Blaha, M.J.; Santos, R.D.; Sposito, A.; et al. Beyond BMI: The “Metabolically healthy obese” phenotype & its association with clinical/subclinical cardiovascular disease and all-cause mortality—A systematic review. BMC Public Health 2014, 14, 14. [Google Scholar]
- Dobson, R.; Burgess, M.I.; Sprung, V.S.; Irwin, A.; Hamer, M.; Jones, J.; Daousi, C.; Adams, V.; Kemp, G.J.; Shojaee-Moradie, F.; et al. Metabolically healthy and unhealthy obesity: differential effects on myocardial function according to metabolic syndrome, rather than obesity. Int. J. Obes. 2016, 40, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-M.M.; White, A.J.; Nichols, H.B.; O’brien, K.M.; Weinberg, C.R.; Sandler, D.P. The association between metabolic health, obesity phenotype and the risk of breast cancer. UICC Int. J. Cancer IJC 2017, 140, 2657–2666. [Google Scholar] [CrossRef] [PubMed]
- Meigs, J.B.; Wilson, P.W.F.; Fox, C.S.; Vasan, R.S.; Nathan, D.M.; Sullivan, L.M.; D’Agostino, R.B. Body Mass Index, Metabolic Syndrome, and Risk of Type 2 Diabetes or Cardiovascular Disease. J. Clin. Endocrinol. Metab. 2006, 91, 2906–2912. [Google Scholar] [CrossRef] [PubMed]
- Cembrowska, P.; Stefańska, A.; Odrowąż-Sypniewska, G. Obesity phenotypes: normal-weight individuals with metabolic disorders versus metabolically healthy obese. Med. Res. J. 2017, 1, 95–99. [Google Scholar] [CrossRef]
- Mathew, H.; Farr, O.M.; Mantzoros, C.S. Metabolic health and weight: Understanding metabolically unhealthy normal weight or metabolically healthy obese patients. Metabolism. 2016, 65, 73–80. [Google Scholar] [CrossRef]
- Aung, K.; Lorenzo, C.; Hinojosa, M.A.; Haffner, S.M. Risk of Developing Diabetes and Cardiovascular Disease in Metabolically Unhealthy Normal-Weight and Metabolically Healthy Obese Individuals. J. Clin. Endocrinol. Metab. 2014, 99, 462–468. [Google Scholar] [CrossRef]
- Dvorak, R.V.; DeNino, W.F.; Ades, P.A.; Poehlman, E.T. Phenotypic characteristics associated with insulin resistance in metabolically obese but normal-weight young women. Diabetes 1999, 48, 2210–2214. [Google Scholar] [CrossRef]
- Marini, M.A.; Succurro, E.; Frontoni, S.; Hribal, M.L.; Andreozzi, F.; Lauro, R.; Perticone, F.; Sesti, G.; Pedro-Botet, J. Metabolically healthy but obese women have an intermediate cardiovascular risk profile between healthy nonobese women and obese insulin-resistant women. Diabetes Care 2007, 30, 2145–2147. [Google Scholar] [CrossRef]
- Lee, S.-H.; Han, K.; Yang, H.K.; Kim, H.-S.; Cho, J.-H.; Kwon, H.-S.; Park, Y.-M.; Cha, B.-Y.; Yoon, K.-H. A novel criterion for identifying metabolically obese but normal weight individuals using the product of triglycerides and glucose. Nutr. Diabetes 2015, 5, e149. [Google Scholar] [CrossRef]
- Muñoz-Garach, A.; Cornejo-Pareja, I.; Tinahones, F.J. Does Metabolically Healthy Obesity Exist? Nutrients 2016, 8, 320. [Google Scholar] [CrossRef] [PubMed]
- Andres, R. Effect of obesity on total mortality. Int. J. Obes. 1980, 4, 381–386. [Google Scholar] [PubMed]
- Sims, E.A.H. Are there persons who are obese, but metabolically healthy? Metabolism 2001, 50, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Zhang, L.; Zheng, R.; Zheng, Y. The prevalence, metabolic risk and effects of lifestyle intervention for metabolically healthy obesity: a systematic review and meta-analysis: A PRISMA-compliant article. Medicine (Baltimore) 2017, 96, e8838. [Google Scholar] [CrossRef] [PubMed]
- Kuk, J.L.; Ardern, C.I. Are metabolically normal but obese individuals at lower risk for all-cause mortality? Diabetes Care 2009, 32, 2297–2299. [Google Scholar] [CrossRef] [PubMed]
- Marques-Vidal, P.; Velho, S.; Waterworth, D.; Waeber, G.; von Känel, R.; Vollenweider, P. The association between inflammatory biomarkers and metabolically healthy obesity depends of the definition used. Eur. J. Clin. Nutr. 2012, 66, 426–435. [Google Scholar] [CrossRef]
- Bobbioni-Harsch, E.; Pataky, Z.; Makoundou, V.; Laville, M.; Disse, E.; Anderwald, C.; Konrad, T.; Golay, A. RISC Investigators From Metabolic Normality to Cardiometabolic Risk Factors in Subjects With Obesity. Obesity 2012, 20, 2063–2069. [Google Scholar] [CrossRef]
- Arnlov, J.; Sundstrom, J.; Ingelsson, E.; Lind, L. Impact of BMI and the Metabolic Syndrome on the Risk of Diabetes in Middle-Aged Men. Diabetes Care 2011, 34, 61–65. [Google Scholar] [CrossRef]
- Oflaz, H.; Ozbey, N.; Mantar, F.; Genchellac, H.; Mercanoglu, F.; Sencer, E.; Molvalilar, S.; Orhan, Y. Determination of endothelial function and early atherosclerotic changes in healthy obese women. Diabetes Nutr. Metab. 2003, 16, 176–181. [Google Scholar]
- Espinosa De Ycaza, A.; Donegan, D.; Jensen, M.D. Long-term metabolic risk for the metabolically healthy overweight/obese phenotype. Int. J. Obes. 2018, 42, 302–309. [Google Scholar] [CrossRef]
- Hamer, M.; Stamatakis, E. Metabolically healthy obesity and risk of all-cause and cardiovascular disease mortality. J. Clin. Endocrinol. Metab. 2012, 97, 2482–2488. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.H.; Lee, W.J.; Song, K.-H. Metabolically healthy obesity: a friend or foe? Korean J. Intern. Med. 2017, 32, 611–621. [Google Scholar] [CrossRef] [PubMed]
- St-Pierre, A.C.; Cantin, B.; Mauriège, P.; Bergeron, J.; Dagenais, G.R.; Després, J.-P.; Lamarche, B. Insulin resistance syndrome, body mass index and the risk of ischemic heart disease. Can. Med. Assoc. J. 2005, 172, 1301–1305. [Google Scholar] [CrossRef]
- van Vliet-Ostaptchouk, J.V.; Nuotio, M.-L.; Slagter, S.N.; Doiron, D.; Fischer, K.; Foco, L.; Gaye, A.; Gögele, M.; Heier, M.; Hiekkalinna, T.; et al. The prevalence of metabolic syndrome and metabolically healthy obesity in Europe: a collaborative analysis of ten large cohort studies. BMC Endocr. Disord. 2014, 14, 9. [Google Scholar] [CrossRef]
- Eckel, N.; Meidtner, K.; Kalle-Uhlmann, T.; Stefan, N.; Schulze, M.B. Metabolically healthy obesity and cardiovascular events: A systematic review and meta-analysis. Eur. J. Prev. Cardiol. 2016, 23, 956–966. [Google Scholar] [CrossRef]
- Ortega, F.B.; Lavie, C.J.; Blair, S.N. Obesity and Cardiovascular Disease. Circ. Res. 2016, 118, 1752–1770. [Google Scholar] [CrossRef]
- Al-Khalidi, B.; Kimball, S.M.; Kuk, J.L.; Ardern, C.I. Metabolically healthy obesity, vitamin D, and all-cause and cardiometabolic mortality risk in NHANES III. Clin. Nutr. 2018, 38, 820–828. [Google Scholar] [CrossRef]
- Ogorodnikova, A.D.; Kim, M.; McGinn, A.P.; Muntner, P.; Khan, U.; Wildman, R.P. Incident Cardiovascular Disease Events in Metabolically Benign Obese Individuals. Obesity 2012, 20, 651–659. [Google Scholar] [CrossRef]
- Gonçalves, C.G.; Glade, M.J.; Meguid, M.M. Metabolically healthy obese individuals: Key protective factors. Nutrition 2016, 32, 14–20. [Google Scholar] [CrossRef]
- Ridker, P.M.; Buring, J.E.; Cook, N.R.; Rifai, N. C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: an 8-year follow-up of 14 719 initially healthy American women. Circulation 2003, 107, 391–397. [Google Scholar] [CrossRef]
- Robson, E.M.; Costa, S.; Hamer, M.; Johnson, W. Life course factors associated with metabolically healthy obesity: A protocol for the systematic review of longitudinal studies. Syst. Rev. 2018, 7, 50. [Google Scholar] [CrossRef] [PubMed]
- Yeh, T.-L.; Chen, H.-H.; Tsai, S.-Y.; Lin, C.-Y.; Liu, S.-J.; Chien, K.-L. The Relationship between Metabolically Healthy Obesity and the Risk of Cardiovascular Disease: A Systematic Review and Meta-Analysis. J. Clin. Med. 2019, 8, 1228. [Google Scholar] [CrossRef] [PubMed]
- Ortega, F.B.; Cadenas-Sanchez, C.; Migueles, J.H.; Labayen, I.; Ruiz, J.R.; Sui, X.; Blair, S.N.; Martínez-Vizcaino, V.; Lavie, C.J. Role of Physical Activity and Fitness in the Characterization and Prognosis of the Metabolically Healthy Obesity Phenotype: A Systematic Review and Meta-analysis. Prog. Cardiovasc. Dis. 2018, 61, 190–205. [Google Scholar] [CrossRef]
- Mirzababaei, A.; Djafarian, K.; Mozafari, H.; Shab-Bidar, S. The long-term prognosis of heart diseases for different metabolic phenotypes: a systematic review and meta-analysis of prospective cohort studies. Endocrine 2019, 63, 439–462. [Google Scholar] [CrossRef]
- Vekic, J.; Zeljkovic, A.; Stefanovic, A.; Jelic-Ivanovic, Z.; Spasojevic-Kalimanovska, V. Obesity and dyslipidemia. Metabolism 2019, 92, 71–81. [Google Scholar] [CrossRef]
- Iacobini, C.; Pugliese, G.; Blasetti Fantauzzi, C.; Federici, M.; Menini, S. Metabolically healthy versus metabolically unhealthy obesity. Metabolism 2019, 92, 51–60. [Google Scholar] [CrossRef]
- Beh, S. Is metabolically healthy obesity a useful concept? Diabet. Med. 2019, 36, 539–545. [Google Scholar] [CrossRef]
- Katzmarzyk, P.T.; Church, T.S.; Janssen, I.; Ross, R.; Blair, S.N. Metabolic syndrome, obesity, and mortality: impact of cardiorespiratory fitness. Diabetes Care 2005, 28, 391–397. [Google Scholar] [CrossRef]
- Cadenas-Sanchez, C.; Ruiz, J.R.; Labayen, I.; Huybrechts, I.; Manios, Y.; González-Gross, M.; Breidenassel, C.; Kafatos, A.; De Henauw, S.; Vanhelst, J.; et al. Prevalence of Metabolically Healthy but Overweight/Obese Phenotype and Its Association With Sedentary Time, Physical Activity, and Fitness. J. Adolesc. Health 2017, 61, 107–114. [Google Scholar] [CrossRef]
- Karelis, A.D.; Brochu, M.; Rabasa-Lhoret, R. Can we identify metabolically healthy but obese individuals (MHO)? Diabetes Metab. 2004, 30, 569–572. [Google Scholar] [CrossRef]
- Karelis, A.D.; Faraj, M.; Bastard, J.-P.; St-Pierre, D.H.; Brochu, M.; Prud’homme, D.; Rabasa-Lhoret, R. The Metabolically Healthy but Obese Individual Presents a Favorable Inflammation Profile. J. Clin. Endocrinol. Metab. 2005, 90, 4145–4150. [Google Scholar] [CrossRef] [PubMed]
- Daly, C.A.; Hildebrandt, P.; Bertrand, M.; Ferrari, R.; Remme, W.; Simoons, M.; Fox, K.M. EUROPA investigators Adverse prognosis associated with the metabolic syndrome in established coronary artery disease: data from the EUROPA trial. Heart 2007, 93, 1406–1411. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, T.; Sherman, A.; Tsao, P.; Gonzalez, O.; Yee, G.; Lamendola, C.; Reaven, G.M.; Cushman, S.W. Enhanced proportion of small adipose cells in insulin-resistant vs insulin-sensitive obese individuals implicates impaired adipogenesis. Diabetologia 2007, 50, 1707–1715. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Manson, J.E.; Meigs, J.B.; Ridker, P.M.; Buring, J.E.; Liu, S. Comparison of usefulness of body mass index versus metabolic risk factors in predicting 10-year risk of cardiovascular events in women. Am. J. Cardiol. 2007, 100, 1654–1658. [Google Scholar] [CrossRef]
- Aguilar-Salinas, C.A.; García, E.G.; Robles, L.; Riaño, D.; Ruiz-Gomez, D.G.; García-Ulloa, A.C.; Melgarejo, M.A.; Zamora, M.; Guillen-Pineda, L.E.; Mehta, R.; et al. High Adiponectin Concentrations Are Associated with the Metabolically Healthy Obese Phenotype. J. Clin. Endocrinol. Metab. 2008, 93, 4075–4079. [Google Scholar] [CrossRef]
- Karelis, A.D.; Rabasa-Lhoret, R. Inclusion of C-reactive protein in the identification of metabolically healthy but obese (MHO) individuals. Diabetes Metab. 2008, 34, 183–184. [Google Scholar] [CrossRef]
- Stefan, N.; Kantartzis, K.; Machann, J.; Schick, F.; Thamer, C.; Rittig, K.; Balletshofer, B.; Machicao, F.; Fritsche, A.; Häring, H.-U. Identification and Characterization of Metabolically Benign Obesity in Humans. Arch. Intern. Med. 2008, 168, 1609. [Google Scholar] [CrossRef]
- Hosseinpanah, F.; Barzin, M.; Sheikholeslami, F.; Azizi, F. Effect of Different Obesity Phenotypes on Cardiovascular Events in Tehran Lipid and Glucose Study (TLGS). Am. J. Cardiol. 2011, 107, 412–416. [Google Scholar] [CrossRef]
- Khan, U.I.; Wang, D.; Thurston, R.C.; Sowers, M.; Sutton-Tyrrell, K.; Matthews, K.A.; Barinas-Mitchell, E.; Wildman, R.P. Burden of subclinical cardiovascular disease in “metabolically benign” and “at-risk” overweight and obese women: the Study of Women’s Health Across the Nation (SWAN). Atherosclerosis 2011, 217, 179–186. [Google Scholar] [CrossRef]
- Park, J.; Kim, S.H.; Cho, G.-Y.; Baik, I.; Kim, N.H.; Lim, H.E.; Kim, E.J.; Park, C.G.; Lim, S.Y.; Kim, Y.H.; et al. Obesity phenotype and cardiovascular changes. J. Hypertens. 2011, 29, 1765–1772. [Google Scholar] [CrossRef]
- Voulgari, C.; Tentolouris, N.; Dilaveris, P.; Tousoulis, D.; Katsilambros, N.; Stefanadis, C. Increased Heart Failure Risk in Normal-Weight People With Metabolic Syndrome Compared With Metabolically Healthy Obese Individuals. J. Am. Coll. Cardiol. 2011, 58, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Batty, G.D.; Kivimaki, M. Risk of future depression in people who are obese but metabolically healthy: the English longitudinal study of ageing. Mol. Psychiatry 2012, 17, 940–945. [Google Scholar] [CrossRef] [PubMed]
- Bervoets, L.; Massa, G. Classification and clinical characterization of metabolically “healthy” obese children and adolescents. J. Pediatr. Endocrinol. Metab. 2016, 29, 553–560. [Google Scholar] [CrossRef]
- Al Masri, M.; Romain, A.J.; Boegner, C.; Maimoun, L.; Mariano-Goulart, D.; Attalin, V.; Leprieur, E.; Picandet, M.; Avignon, A.; Sultan, A. Vitamin D status is not related to insulin resistance in different phenotypes of moderate obesity. Appl. Physiol. Nutr. Metab. 2017, 42, 438–442. [Google Scholar] [CrossRef]
- Wildman, R.P.; Muntner, P.; Reynolds, K.; McGinn, A.P.; Rajpathak, S.; Wylie-Rosett, J.; Sowers, M.R. The Obese Without Cardiometabolic Risk Factor Clustering and the Normal Weight With Cardiometabolic Risk Factor Clustering. Arch. Intern. Med. 2008, 168, 1617. [Google Scholar] [CrossRef]
- Sánchez-Iñigo, L.; Navarro-González, D.; Fernández-Montero, A.; Pastrana-Delgado, J.; Martínez, J.A. Risk of incident ischemic stroke according to the metabolic health and obesity states in the Vascular-Metabolic CUN cohort. Int. J. Stroke 2017, 12, 187–191. [Google Scholar] [CrossRef]
- Lassale, C.; Tzoulaki, I.; Moons, K.G.M.; Sweeting, M.; Boer, J.; Johnson, L.; Huerta, J.M.; Agnoli, C.; Freisling, H.; Weiderpass, E.; et al. Separate and combined associations of obesity and metabolic health with coronary heart disease: A pan-European case-cohort analysis. Eur. Heart J. 2018, 39, 397–406. [Google Scholar] [CrossRef]
- Stefan, N.; Häring, H.-U.; Hu, F.B.; Schulze, M.B. Metabolically healthy obesity: epidemiology, mechanisms, and clinical implications. Lancet Diabetes Endocrinol. 2013, 1, 152–162. [Google Scholar] [CrossRef]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421.
- Denke, M.A.; Pasternak, R.C. Defining and Treating the Metabolic Syndrome: A Primer from the Adult Treatment Panel III. Curr. Treat. Options Cardiovasc. Med. 2001, 3, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Brewer, H.B.; Cleeman, J.I.; Smith, S.C.; Lenfant, C. American Heart Association; National Heart, Lung, and Blood Institute Definition of Metabolic Syndrome: Report of the National Heart, Lung, and Blood Institute/American Heart Association Conference on Scientific Issues Related to Definition. Circulation 2004, 109, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Cleeman, J.I.; Daniels, S.R.; Donato, K.A.; Eckel, R.H.; Franklin, B.A.; Gordon, D.J.; Krauss, R.M.; Savage, P.J.; Smith, S.C.; et al. Diagnosis and Management of the Metabolic Syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 2005, 112, 2735–2752. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J. Metabolic syndrome-a new world-wide definition. A Consensus Statement from the International Diabetes Federation. Diabet. Med. 2006, 23, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Zimmet, P.; Alberti, K.G.M.; Kaufman, F.; Tajima, N.; Silink, M.; Arslanian, S.; Wong, G.; Bennett, P.; Shaw, J.; Caprio, S.; et al. The metabolic syndrome in children and adolescents? an IDF consensus report. Pediatr. Diabetes 2007, 8, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, C.J.; Janssen, I. Development of Age-Specific Adolescent Metabolic Syndrome Criteria That Are Linked to the Adult Treatment Panel III and International Diabetes Federation Criteria. J. Am. Coll. Cardiol. 2007, 49, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.-C.; James, W.P.T.; Loria, C.M.; Smith, S.C.; et al. Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Delavari, A.; Forouzanfar, M.H.; Alikhani, S.; Sharifian, A.; Kelishadi, R. First Nationwide Study of the Prevalence of the Metabolic Syndrome and Optimal Cutoff Points of Waist Circumference in the Middle East: The National Survey of Risk Factors for Noncommunicable Diseases of Iran. Diabetes Care 2009, 32, 1092–1097. [Google Scholar] [CrossRef]
- Ford, E.S.; Giles, W.H.; Dietz, W.H. Prevalence of the metabolic syndrome among US adults: Findings from the Third National Health and Nutrition Examination Survey. J. Am. Med. Assoc. 2002, 287, 356–359. [Google Scholar] [CrossRef]
- Grundy, S.M.; Cleeman, J.I.; Bairey Merz, C.N.; Brewer, H.B.; Clark, L.T.; Hunninghake, D.B.; Pasternak, R.C.; Smith, S.C.; Stone, N.J. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004, 110, 227–239. [Google Scholar] [CrossRef]
- Grundy Erratum: Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines (Circulation (July 13, 2004) 110 (227-239)). Circulation 2004, 110, 763.
- Alberti, K.G.M.; Zimmet, P.; Shaw, J. IDF Epidemiology Task Force Consensus Group The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Despres, J.-P. Body Fat Distribution and Risk of Cardiovascular Disease: An Update. Circulation 2012, 126, 1301–1313. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, A.; Soldati, L.; Sarlo, F.; Calvani, M.; Di Lorenzo, N.; Di Renzo, L. New obesity classification criteria as a tool for bariatric surgery indication. World J. Gastroenterol. 2016, 22, 681–703. [Google Scholar] [CrossRef] [PubMed]
- Pajunen, P.; Kotronen, A.; Korpi-Hyövälti, E.; Keinänen-Kiukaanniemi, S.; Oksa, H.; Niskanen, L.; Saaristo, T.; Saltevo, J.T.; Sundvall, J.; Vanhala, M.; et al. Metabolically healthy and unhealthy obesity phenotypes in the general population: the FIN-D2D Survey. BMC Public Health 2011, 11, 754. [Google Scholar] [CrossRef]
- Björntorp, P. Metabolic implications of body fat distribution. Diabetes Care 1991, 14, 1132–1143. [Google Scholar] [CrossRef]
- Manolopoulos, K.N.; Karpe, F.; Frayn, K.N. Gluteofemoral body fat as a determinant of metabolic health. Int. J. Obes. 2010, 34, 949–959. [Google Scholar] [CrossRef]
- Jensen, M.D. Role of body fat distribution and the metabolic complications of obesity. J. Clin. Endocrinol. Metab. 2008, 93, S57–S63. [Google Scholar] [CrossRef]
- Makki, K.; Froguel, P.; Wolowczuk, I. Adipose tissue in obesity-related inflammation and insulin resistance: cells, cytokines, and chemokines. ISRN Inflamm. 2013, 2013, 1–12. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Invest. 1995, 95, 2409–2415. [Google Scholar] [CrossRef]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.M.; Weschenfelder, J.; Sander, C.; Minkwitz, J.; Thormann, J.; Chittka, T.; Mergl, R.; Kirkby, K.C.; Faßhauer, M.; Stumvoll, M.; et al. Inflammatory cytokines in general and central obesity and modulating effects of physical activity. PLoS ONE 2015, 10, e0121971. [Google Scholar] [CrossRef]
- Sun, S.; Ji, Y.; Kersten, S.; Qi, L. Mechanisms of inflammatory responses in obese adipose tissue. Annu. Rev. Nutr. 2012, 32, 261–286. [Google Scholar] [CrossRef] [PubMed]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Wellen, K.E.; Hotamisligil, G.S. Inflammation, stress, and diabetes. J. Clin. Invest. 2005, 115, 1111–1119. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, E.E.; Flier, J.S. Adipose Tissue as an Endocrine Organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Prins, J.B. Adipose tissue as an endocrine organ. Best Pract. Res. Clin. Endocrinol. Metab. 2002, 16, 639–651. [Google Scholar] [CrossRef]
- Kim, J.I.; Huh, J.Y.; Sohn, J.H.; Choe, S.S.; Lee, Y.S.; Lim, C.Y.; Jo, A.; Park, S.B.; Han, W.; Kim, J.B. Lipid-overloaded enlarged adipocytes provoke insulin resistance independent of inflammation. Mol. Cell. Biol. 2015, 35, 1686–1699. [Google Scholar] [CrossRef]
- Xu, H.; Barnes, G.T.; Yang, Q.; Tan, G.; Yang, D.; Chou, C.J.; Sole, J.; Nichols, A.; Ross, J.S.; Tartaglia, L.A.; et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J. Clin. Invest. 2003, 112, 1821–1830. [Google Scholar] [CrossRef]
- Weisberg, S.P.; McCann, D.; Desai, M.; Rosenbaum, M.; Leibel, R.L.; Ferrante, A.W., Jr. Obesity is associated with macrophage accumulation in adipose tissue. J. Clin. Invest. 2003, 112, 1796–1808. [Google Scholar] [CrossRef]
- Rudich, A.; Kanety, H.; Bashan, N. Adipose stress-sensing kinases: linking obesity to malfunction. Trends Endocrinol. Metab. 2007, 18, 291–299. [Google Scholar] [CrossRef]
- Qatanani, M.; Lazar, M.A. Mechanisms of obesity-associated insulin resistance: many choices on the menu. Genes Dev. 2007, 21, 1443–1455. [Google Scholar] [CrossRef]
- Mulder, P.; Morrison, M.C.; Verschuren, L.; Liang, W.; van Bockel, J.H.; Kooistra, T.; Wielinga, P.Y.; Kleemann, R. Reduction of obesity-associated white adipose tissue inflammation by rosiglitazone is associated with reduced non-alcoholic fatty liver disease in LDLr-deficient mice. Sci. Rep. 2016, 6, 31542. [Google Scholar] [CrossRef] [PubMed]
- Badoud, F.; Perreault, M.; Zulyniak, M.A.; Mutch, D.M. Molecular insights into the role of white adipose tissue in metabolically unhealthy normal weight and metabolically healthy obese individuals. FASEB J. 2015, 29, 748–758. [Google Scholar] [CrossRef] [PubMed]
- Srdić, B.; Stokić, E.; Korać, A.; Ukropina, M.; Veličković, K.; Breberina, M. Morphological Characteristics of Abdominal Adipose Tissue in Normal-Weight and Obese Women of Different Metabolic Profiles. Exp. Clin. Endocrinol. Diabetes 2010, 118, 713–718. [Google Scholar]
- O’Connell, J.; Lynch, L.; Cawood, T.J.; Kwasnik, A.; Nolan, N.; Geoghegan, J.; McCormick, A.; O’Farrelly, C.; O’Shea, D. The Relationship of Omental and Subcutaneous Adipocyte Size to Metabolic Disease in Severe Obesity. PLoS ONE 2010, 5, e9997. [Google Scholar] [CrossRef]
- O’Connell, J.; Lynch, L.; Hogan, A.; Cawood, T.J.; O’Shea, D. Preadipocyte Factor-1 Is Associated with Metabolic Profile in Severe Obesity. J. Clin. Endocrinol. Metab. 2011, 96, E680–E684. [Google Scholar] [CrossRef] [PubMed]
- Unger, R.H. Lipid overload and overflow: metabolic trauma and the metabolic syndrome. Trends Endocrinol. Metab. 2003, 14, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Sniderman, A.D.; Bhopal, R.; Prabhakaran, D.; Sarrafzadegan, N.; Tchernof, A. Why might South Asians be so susceptible to central obesity and its atherogenic consequences? The adipose tissue overflow hypothesis. Int. J. Epidemiol. 2007, 36, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Neeland, I.J.; Poirier, P.; Després, J.-P. Cardiovascular and Metabolic Heterogeneity of Obesity. Circulation 2018, 137, 1391–1406. [Google Scholar] [CrossRef]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 2007, 117, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Cancello, R.; Tordjman, J.; Poitou, C.; Guilhem, G.; Bouillot, J.L.; Hugol, D.; Coussieu, C.; Basdevant, A.; Hen, A.B.; Bedossa, P.; et al. Increased Infiltration of Macrophages in Omental Adipose Tissue Is Associated With Marked Hepatic Lesions in Morbid Human Obesity. Diabetes 2006, 55, 1554–1561. [Google Scholar] [CrossRef] [PubMed]
- Tatsumi, Y.; Nakao, Y.M.; Masuda, I.; Higashiyama, A.; Takegami, M.; Nishimura, K.; Watanabe, M.; Ohkubo, T.; Okamura, T.; Miyamoto, Y. Risk for metabolic diseases in normal weight individuals with visceral fat accumulation: a cross-sectional study in Japan. BMJ Open 2017, 7, e013831. [Google Scholar] [CrossRef] [PubMed]
- Goday, A.; Calvo, E.; Vázquez, L.A.; Caveda, E.; Margallo, T.; Catalina-Romero, C.; Reviriego, J. Prevalence and clinical characteristics of metabolically healthy obese individuals and other obese/non-obese metabolic phenotypes in a working population: results from the Icaria study. BMC Public Health 2016, 16, 248. [Google Scholar] [CrossRef]
- Bastien, M.; Poirier, P.; Lemieux, I.; Després, J.-P. Overview of Epidemiology and Contribution of Obesity to Cardiovascular Disease. Prog. Cardiovasc. Dis. 2014, 56, 369–381. [Google Scholar] [CrossRef]
- Goossens, G.H. The Metabolic Phenotype in Obesity: Fat Mass, Body Fat Distribution, and Adipose Tissue Function. Obes. Facts 2017, 10, 207–215. [Google Scholar] [CrossRef]
- Vague, J. Sexual differentiation. A determinant factor of the forms of obesity. 1947. Obes. Res. 1996, 4, 201–203. [Google Scholar] [CrossRef]
- Vague, J. The degree of masculine differentiation of obesities: a factor determining predisposition to diabetes, atherosclerosis, gout, and uric calculous disease. Am. J. Clin. Nutr. 1956, 4, 20–34. [Google Scholar] [CrossRef]
- Kyrou, I.; Randeva, H.S.; Weickert, M.O. Clinical Problems Caused by Obesity; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Lumsden, M.A.; Hor, K. Impact of obesity on the health of women in midlife. Obstet. Gynaecol. 2015, 17, 201–208. [Google Scholar] [CrossRef]
- Okura, T.; Nakata, Y.; Yamabuki, K.; Tanaka, K. Regional Body Composition Changes Exhibit Opposing Effects on Coronary Heart Disease Risk Factors. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 923–929. [Google Scholar] [CrossRef]
- Kang, S.M.; Yoon, J.W.; Ahn, H.Y.; Kim, S.Y.; Lee, K.H.; Shin, H.; Choi, S.H.; Park, K.S.; Jang, H.C.; Lim, S. Android fat depot is more closely associated with metabolic syndrome than abdominal visceral fat in elderly people. PLoS ONE 2011, 6, e27694. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Song, A.; Zhou, Y.; Ma, X.; Jiao, J.; Yang, M.; Zhu, S. Association of regional body fat with metabolic risks in Chinese women. Public Health Nutr. 2014, 17, 2316–2324. [Google Scholar] [CrossRef] [PubMed]
- Samsell, L.; Regier, M.; Walton, C.; Cottrell, L. Importance of android/gynoid fat ratio in predicting metabolic and cardiovascular disease risk in normal weight as well as overweight and obese children. J. Obes. 2014, 2014, 846578. [Google Scholar] [CrossRef]
- Messier, V.; Karelis, A.D.; Robillard, M.-È.; Bellefeuille, P.; Brochu, M.; Lavoie, J.-M.; Rabasa-Lhoret, R. Metabolically healthy but obese individuals: relationship with hepatic enzymes. Metabolism 2010, 59, 20–24. [Google Scholar] [CrossRef]
- Brochu, M.; Tchernof, A.; Dionne, I.J.; Sites, C.K.; Eltabbakh, G.H.; Sims, E.A.H.; Poehlman, E.T. What Are the Physical Characteristics Associated with a Normal Metabolic Profile Despite a High Level of Obesity in Postmenopausal Women? J. Clin. Endocrinol. Metab. 2001, 86, 1020–1025. [Google Scholar]
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.-Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A.; et al. Abdominal Visceral and Subcutaneous Adipose Tissue Compartments: Association With Metabolic Risk Factors in the Framingham Heart Study. Circulation 2007, 116, 39–48. [Google Scholar] [CrossRef]
- Basat, O.; Ucak, S.; Ozkurt, H.; Basak, M.; Seber, S.; Altuntas, Y. Visceral Adipose Tissue as an Indicator of Insulin Resistance in Nonobese Patients with New Onset Type 2 Diabetes Mellitus. Exp. Clin. Endocrinol. Diabetes 2006, 114, 58–62. [Google Scholar] [CrossRef]
- Sam, S.; Haffner, S.; Davidson, M.H.; D’Agostino, R.B.; Feinstein, S.; Kondos, G.; Perez, A.; Mazzone, T.; Mazzone, T. Relationship of abdominal visceral and subcutaneous adipose tissue with lipoprotein particle number and size in type 2 diabetes. Diabetes 2008, 57, 2022–2027. [Google Scholar] [CrossRef]
- Montague, C.T.; O’Rahilly, S. The perils of portliness: causes and consequences of visceral adiposity. Diabetes 2000, 49, 883–888. [Google Scholar] [CrossRef]
- Snijder, M.B.; Visser, M.; Dekker, J.M.; Goodpaster, B.H.; Harris, T.B.; Kritchevsky, S.B.; De Rekeneire, N.; Kanaya, A.M.; Newman, A.B.; Tylavsky, F.A.; et al. Low subcutaneous thigh fat is a risk factor for unfavourable glucose and lipid levels, independently of high abdominal fat. The Health ABC Study. Diabetologia 2005, 48, 301–308. [Google Scholar] [CrossRef]
- Pinnick, K.E.; Nicholson, G.; Manolopoulos, K.N.; McQuaid, S.E.; Valet, P.; Frayn, K.N.; Denton, N.; Min, J.L.; Zondervan, K.T.; Fleckner, J.; et al. Distinct Developmental Profile of Lower-Body Adipose Tissue Defines Resistance Against Obesity-Associated Metabolic Complications. Diabetes 2014, 63, 3785–3797. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.M.; Jung, C.H.; Cho, Y.K.; Jang, J.E.; Hwang, J.Y.; Kim, E.H.; Lee, W.J.; Park, J.-Y.; Kim, H.-K. Visceral adiposity index predicts the conversion of metabolically healthy obesity to an unhealthy phenotype. PLoS ONE 2017, 12, e0179635. [Google Scholar] [CrossRef]
- Mouly, T.A.; Toms, L.-M.L. Breast cancer and persistent organic pollutants (excluding DDT): a systematic literature review. Environ. Sci. Pollut. Res. 2016, 23, 22385–22407. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, M.-S.; Rabasa-Lhoret, R.; Prud’homme, D.; Karelis, A.D.; Geng, D.; van Bavel, B.; Ruzzin, J. The Metabolically Healthy But Obese Phenotype Is Associated With Lower Plasma Levels of Persistent Organic Pollutants as Compared to the Metabolically Abnormal Obese Phenotype. J. Clin. Endocrinol. Metab. 2014, 99, E1061–E1066. [Google Scholar] [CrossRef] [PubMed]
- Pestana, D.; Faria, G.; Sá, C.; Fernandes, V.C.; Teixeira, D.; Norberto, S.; Faria, A.; Meireles, M.; Marques, C.; Correia-Sá, L.; et al. Persistent organic pollutant levels in human visceral and subcutaneous adipose tissue in obese individuals—Depot differences and dysmetabolism implications. Environ. Res. 2014, 133, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, D.; Pestana, D.; Calhau, C.; Monteiro, R. Letter to the Editor: Adipose tissue persistent organic pollutants as possible markers of dysmetabolic obesity. J. Clin. Endocrinol. Metab. 2014. Available online: http://press.endocrine.org/e-letters/10.1210/jc.20 (accessed on 22 December 2019).
- Teixeira, D.; Pestana, D.; Santos, C.; Correia-Sá, L.; Marques, C.; Norberto, S.; Meireles, M.; Faria, A.; Silva, R.; Faria, G.; et al. Inflammatory and cardiometabolic risk on obesity: role of environmental xenoestrogens. J. Clin. Endocrinol. Metab. 2015, 100, 1792–1801. [Google Scholar] [CrossRef][Green Version]
- Teixeira, D.; Marques, C.; Pestana, D.; Faria, A.; Norberto, S.; Calhau, C.; Monteiro, R. Effects of xenoestrogens in human M1 and M2 macrophage migration, cytokine release, and estrogen-related signaling pathways. Environ. Toxicol. 2016, 31, 1496–1509. [Google Scholar] [CrossRef]
- Pestana, D.; Teixeira, D.; Meireles, M.; Marques, C.; Norberto, S.; Sá, C.; Fernandes, V.C.; Correia-Sá, L.; Faria, A.; Guardão, L.; et al. Adipose tissue dysfunction as a central mechanism leading to dysmetabolic obesity triggered by chronic exposure to p,p′-DDE. Sci. Rep. 2017, 7, 2738. [Google Scholar] [CrossRef]
- Smink, A.; Ribas-Fito, N.; Garcia, R.; Torrent, M.; Mendez, M.A.; Grimalt, J.O.; Sunyer, J. Exposure to hexachlorobenzene during pregnancy increases the risk of overweight in children aged 6 years. Acta Paediatr. 2008, 97, 1465–1469. [Google Scholar] [CrossRef]
- Lee, D.-H.; Steffes, M.W.; Sjödin, A.; Jones, R.S.; Needham, L.L.; Jacobs, D.R. Low Dose Organochlorine Pesticides and Polychlorinated Biphenyls Predict Obesity, Dyslipidemia, and Insulin Resistance among People Free of Diabetes. PLoS ONE 2011, 6, e15977. [Google Scholar] [CrossRef] [PubMed]
- Cano-Sancho, G.; Salmon, A.G.; La Merrill, M.A. Association between Exposure to p,p′-DDT and Its Metabolite p,p′-DDE with Obesity: Integrated Systematic Review and Meta-Analysis. Environ. Health Perspect. 2017, 125, 096002. [Google Scholar] [CrossRef] [PubMed]
- Daniels, S.I.; Chambers, J.C.; Sanchez, S.S.; La Merrill, M.A.; Hubbard, A.E.; Macherone, A.; McMullin, M.; Zhang, L.; Elliott, P.; Smith, M.T.; et al. Elevated Levels of Organochlorine Pesticides in South Asian Immigrants Are Associated With an Increased Risk of Diabetes. J. Endocr. Soc. 2018, 2, 832–841. [Google Scholar] [CrossRef] [PubMed]
- Warner, G.R.; Flaws, J.A. Bisphenol A and Phthalates: How Environmental Chemicals Are Reshaping Toxicology. Toxicol. Sci. 2018, 166, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Johns, L.E.; Cooper, G.S.; Galizia, A.; Meeker, J.D. Exposure assessment issues in epidemiology studies of phthalates. Environ. Int. 2015, 85, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Hauser, R.; Calafat, A.M. Phthalates and human health. Occup. Environ. Med. 2005, 62, 806–818. [Google Scholar] [CrossRef]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, 1–150. [Google Scholar] [CrossRef]
- Stojanoska, M.M.; Milosevic, N.; Milic, N.; Abenavoli, L. The influence of phthalates and bisphenol A on the obesity development and glucose metabolism disorders. Endocrine 2017, 55, 666–681. [Google Scholar] [CrossRef]
- Mansouri, V.; Ebrahimpour, K.; Poursafa, P.; Riahi, R.; Shoshtari-Yeganeh, B.; Hystad, P.; Kelishadi, R. Exposure to phthalates and bisphenol A is associated with higher risk of cardiometabolic impairment in normal weight children. Environ. Sci. Pollut. Res. 2019, 26, 18604–18614. [Google Scholar] [CrossRef]
- Sargis, R.M.; Simmons, R.A. Environmental neglect: endocrine disruptors as underappreciated but potentially modifiable diabetes risk factors. Diabetologia 2019, 62, 1811–1822. [Google Scholar] [CrossRef]
- Melzer, D.; Rice, N.E.; Lewis, C.; Henley, W.E.; Galloway, T.S. Association of urinary bisphenol A concentration with heart disease: Evidence from NHANES 2003/06. PLoS ONE 2010, 5, e8673. [Google Scholar] [CrossRef] [PubMed]
- Carwile, J.L.; Michels, K.B. Urinary bisphenol A and obesity: NHANES 2003-2006. Environ. Res. 2011, 111, 825–830. [Google Scholar] [CrossRef] [PubMed]
- James-Todd, T.; Stahlhut, R.; Meeker, J.D.; Powell, S.G.; Hauser, R.; Huang, T.; Rich-Edwards, J. Urinary phthalate metabolite concentrations and diabetes among women in the national health and nutrition examination survey (NHANES) 2001-2008. Environ. Health Perspect. 2012, 120, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.H.; Sung, Y.A.; Hong, Y.S.; Ha, E.; Jeong, K.; Chung, H.; Lee, H. Urinary bisphenol A is associated with insulin resistance and obesity in reproductive-aged women. Clin. Endocrinol. (Oxf.) 2017, 86, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Piecha, R.; Svačina, Š.; Malý, M.; Vrbík, K.; Lacinová, Z.; Haluzík, M.; Pavloušková, J.; Vavrouš, A.; Matějková, D.; Müllerová, D.; et al. Urine levels of phthalate metabolites and bisphenol a in relation to main metabolic syndrome components: Dyslipidemia, hypertension and type 2 diabetes a pilot study. Cent. Eur. J. Public Health 2016, 24, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Shim, Y.H.; Ock, J.W.; Kim, Y.J.; Kim, Y.; Kim, S.Y.; Kang, D. Association between Heavy Metals, Bisphenol A, volatile organic compounds and phthalates and metabolic syndrome. Int. J. Environ. Res. Public Health 2019, 16, 671. [Google Scholar] [CrossRef] [PubMed]
- Milošević, N.; Milanović, M.; Sudji, J.; Bosić Živanović, D.; Stojanoski, S.; Vuković, B.; Milić, N.; Medić Stojanoska, M. Could phthalates exposure contribute to the development of metabolic syndrome and liver disease in humans? Environ. Sci. Pollut. Res. 2019, 27, 1–13. [Google Scholar] [CrossRef]
- Martínez-Ibarra, A.; Martínez-Razo, L.D.; Vázquez-Martínez, E.R.; Martínez-Cruz, N.; Flores-Ramírez, R.; García-Gómez, E.; López-López, M.; Ortega-González, C.; Camacho-Arroyo, I.; Cerbón, M. Unhealthy levels of phthalates and bisphenol a in mexican pregnant women with gestational diabetes and its association to altered expression of miRNAs involved with metabolic disease. Int. J. Mol. Sci. 2019, 20, 3343. [Google Scholar] [CrossRef]
- Festi, D.; Schiumerini, R.; Eusebi, L.H.; Marasco, G.; Taddia, M.; Colecchia, A. Gut microbiota and metabolic syndrome. World J. Gastroenterol. 2014, 20, 16079–16094. [Google Scholar] [CrossRef]
- Brown, J.M.; Hazen, S.L. The gut microbial endocrine organ: bacterially derived signals driving cardiometabolic diseases. Annu. Rev. Med. 2015, 66, 343–359. [Google Scholar] [CrossRef]
- Ley, R.E.; Turnbaugh, P.J.; Klein, S.; Gordon, J.I. Microbial ecology: Human gut microbes associated with obesity. Nature 2006, 444, 1022–1023. [Google Scholar] [CrossRef] [PubMed]
- Santacruz, A.; Collado, M.C.; García-Valdés, L.; Segura, M.T.; Martín-Lagos, J.A.; Anjos, T.; Martí-Romero, M.; Lopez, R.M.; Florido, J.; Campoy, C.; et al. Gut microbiota composition is associated with body weight, weight gain and biochemical parameters in pregnant women. Br. J. Nutr. 2010, 104, 83–92. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1131. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef]
- Million, M.; Maraninchi, M.; Henry, M.; Armougom, F.; Richet, H.; Carrieri, P.; Valero, R.; Raccah, D.; Vialettes, B.; Raoult, D. Obesity-associated gut microbiota is enriched in Lactobacillus reuteri and depleted in Bifidobacterium animalis and Methanobrevibacter smithii. Int. J. Obes. 2012, 36, 817–825. [Google Scholar] [CrossRef]
- Jumpertz, R.; Le, D.S.; Turnbaugh, P.J.; Trinidad, C.; Bogardus, C.; Gordon, J.I.; Krakoff, J. Energy-balance studies reveal associations between gut microbes, caloric load, and nutrient absorption in humans. Am. J. Clin. Nutr. 2011, 94, 58–65. [Google Scholar] [CrossRef]
- Karlsson, C.L.J.; Önnerfält, J.; Xu, J.; Molin, G.; Ahrné, S.; Thorngren-Jerneck, K. The Microbiota of the Gut in Preschool Children With Normal and Excessive Body Weight. Obesity 2012, 20, 2257–2261. [Google Scholar] [CrossRef]
- Hu, H.-J.; Park, S.-G.; Jang, H.B.; Choi, M.-G.; Park, K.-H.; Kang, J.H.; Park, S.I.; Lee, H.-J.; Cho, S.-H. Obesity Alters the Microbial Community Profile in Korean Adolescents. PLoS ONE 2015, 10, e0134333. [Google Scholar] [CrossRef]
- Zhang, H.; DiBaise, J.K.; Zuccolo, A.; Kudrna, D.; Braidotti, M.; Yu, Y.; Parameswaran, P.; Crowell, M.D.; Wing, R.; Rittmann, B.E.; et al. Human gut microbiota in obesity and after gastric bypass. Proc. Natl. Acad. Sci. USA 2009, 106, 2365–2370. [Google Scholar] [CrossRef]
- Duncan, S.H.; Lobley, G.E.; Holtrop, G.; Ince, J.; Johnstone, A.M.; Louis, P.; Flint, H.J. Human colonic microbiota associated with diet, obesity and weight loss. Int. J. Obes. 2008, 32, 1720–1724. [Google Scholar] [CrossRef]
- Ramakrishna, B.S. Role of the gut microbiota in human nutrition and metabolism. J. Gastroenterol. Hepatol. 2013, 28, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.J.; Gerasimidis, K.; Edwards, C.A.; Shaikh, M.G. Role of Gut Microbiota in the Aetiology of Obesity: Proposed Mechanisms and Review of the Literature. J. Obes. 2016, 2016, 7353642. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-A.; Gu, W.; Lee, I.-A.; Joh, E.-H.; Kim, D.-H. High Fat Diet-Induced Gut Microbiota Exacerbates Inflammation and Obesity in Mice via the TLR4 Signaling Pathway. PLoS ONE 2012, 7, e47713. [Google Scholar] [CrossRef] [PubMed]
- Boutagy, N.E.; McMillan, R.P.; Frisard, M.I.; Hulver, M.W. Metabolic endotoxemia with obesity: Is it real and is it relevant? Biochimie 2016, 124, 11–20. [Google Scholar] [CrossRef]
- Erridge, C.; Attina, T.; Spickett, C.M.; Webb, D.J. A high-fat meal induces low-grade endotoxemia: evidence of a novel mechanism of postprandial inflammation. Am. J. Clin. Nutr. 2007, 86, 1286–1292. [Google Scholar] [CrossRef]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic Endotoxemia Initiates Obesity and Insulin Resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef]
- Creely, S.J.; McTernan, P.G.; Kusminski, C.M.; Fisherff, M.; Da Silva, N.F.; Khanolkar, M.; Evans, M.; Harte, A.L.; Kumar, S. Lipopolysaccharide activates an innate immune system response in human adipose tissue in obesity and type 2 diabetes. AJP Endocrinol. Metab. 2006, 292, E740–E747. [Google Scholar] [CrossRef]
- Pussinen, P.J.; Havulinna, A.S.; Lehto, M.; Sundvall, J.; Salomaa, V. Endotoxemia Is Associated With an Increased Risk of Incident Diabetes. Diabetes Care 2011, 34, 392–397. [Google Scholar] [CrossRef]
- Liu, X.; Lu, L.; Yao, P.; Ma, Y.; Wang, F.; Jin, Q.; Ye, X.; Li, H.; Hu, F.B.; Sun, L.; et al. Lipopolysaccharide binding protein, obesity status and incidence of metabolic syndrome: a prospective study among middle-aged and older Chinese. Diabetologia 2014, 57, 1834–1841. [Google Scholar] [CrossRef]
- Collado, M.C.; Derrien, M.; Isolauri, E.; de Vos, W.M.; Salminen, S. Intestinal Integrity and Akkermansia muciniphila, a Mucin-Degrading Member of the Intestinal Microbiota Present in Infants, Adults, and the Elderly. Appl. Environ. Microbiol. 2007, 73, 7767–7770. [Google Scholar] [CrossRef]
- Cani, P.D.; de Vos, W.M. Next-Generation Beneficial Microbes: The Case of Akkermansia muciniphila. Front. Microbiol. 2017, 8, 1765. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Choi, Y.; Kim, D.-K.; Park, H.T.; Ghim, J.; Kwon, Y.; Jeon, J.; Kim, M.-S.; Jee, Y.-K.; Gho, Y.S.; et al. Akkermansia muciniphila-derived extracellular vesicles influence gut permeability through the regulation of tight junctions. Exp. Mol. Med. 2018, 50, e450. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Belzer, C.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; Delzenne, N.M.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Derrien, M.; Van Baarlen, P.; Hooiveld, G.; Norin, E.; Müller, M.; de Vos, W.M. Modulation of Mucosal Immune Response, Tolerance, and Proliferation in Mice Colonized by the Mucin-Degrader Akkermansia muciniphila. Front. Microbiol. 2011, 2, 166. [Google Scholar] [CrossRef]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef]
- Yan, M.; Song, M.-M.; Bai, R.-X.; Cheng, S.; Yan, W.-M. Effect of Roux-en-Y gastric bypass surgery on intestinal Akkermansia muciniphila. World J. Gastrointest. Surg. 2016, 8, 301. [Google Scholar] [CrossRef]
- de la Cuesta-Zuluaga, J.; Mueller, N.T.; Corrales-Agudelo, V.; Velásquez-Mejía, E.P.; Carmona, J.A.; Abad, J.M.; Escobar, J.S. Metformin Is Associated With Higher Relative Abundance of Mucin-Degrading Akkermansia muciniphila and Several Short-Chain Fatty Acid–Producing Microbiota in the Gut. Diabetes Care 2017, 40, 54–62. [Google Scholar] [CrossRef]
- Côté, M.; Matias, I.; Lemieux, I.; Petrosino, S.; Alméras, N.; Després, J.-P.; Di Marzo, V. Circulating endocannabinoid levels, abdominal adiposity and related cardiometabolic risk factors in obese men. Int. J. Obes. 2007, 31, 692–699. [Google Scholar] [CrossRef]
- Bluher, M.; Engeli, S.; Kloting, N.; Berndt, J.; Fasshauer, M.; Batkai, S.; Pacher, P.; Schon, M.R.; Jordan, J.; Stumvoll, M. Dysregulation of the Peripheral and Adipose Tissue Endocannabinoid System in Human Abdominal Obesity. Diabetes 2006, 55, 3053–3060. [Google Scholar] [CrossRef]
- Engeli, S.; Böhnke, J.; Feldpausch, M.; Gorzelniak, K.; Janke, J.; Bátkai, S.; Pacher, P.; Harvey-White, J.; Luft, F.C.; Sharma, A.M.; et al. Activation of the peripheral endocannabinoid system in human obesity. Diabetes 2005, 54, 2838–2843. [Google Scholar] [CrossRef]
- Liu, J.; Bátkai, S.; Pacher, P.; Harvey-White, J.; Wagner, J.A.; Cravatt, B.F.; Gao, B.; Kunos, G. Lipopolysaccharide Induces Anandamide Synthesis in Macrophages via CD14/MAPK/Phosphoinositide 3-Kinase/NF-κB Independently of Platelet-activating Factor. J. Biol. Chem. 2003, 278, 45034–45039. [Google Scholar] [CrossRef] [PubMed]
- Nam, D.H.; Lee, M.H.; Kim, J.E.; Song, H.K.; Kang, Y.S.; Lee, J.E.; Kim, H.W.; Cha, J.J.; Hyun, Y.Y.; Kim, S.H.; et al. Blockade of Cannabinoid Receptor 1 Improves Insulin Resistance, Lipid Metabolism, and Diabetic Nephropathy in db/db Mice. Endocrinology 2012, 153, 1387–1396. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Dopart, R.; Kendall, D.A. Controlled downregulation of the cannabinoid CB1 receptor provides a promising approach for the treatment of obesity and obesity-derived type 2 diabetes. Cell Stress Chaperones 2016, 21, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mehrpouya-Bahrami, P.; Chitrala, K.N.; Ganewatta, M.S.; Tang, C.; Murphy, E.A.; Enos, R.T.; Velazquez, K.T.; McCellan, J.; Nagarkatti, M.; Nagarkatti, P. Blockade of CB1 cannabinoid receptor alters gut microbiota and attenuates inflammation and diet-induced obesity. Sci. Rep. 2017, 7, 15645. [Google Scholar] [CrossRef]
- Muccioli, G.G.; Naslain, D.; Bäckhed, F.; Reigstad, C.S.; Lambert, D.M.; Delzenne, N.M.; Cani, P.D. The endocannabinoid system links gut microbiota to adipogenesis. Mol. Syst. Biol. 2010, 6, 392. [Google Scholar] [CrossRef]
- Rial, S.A.; Karelis, A.D.; Bergeron, K.F.; Mounier, C. Gut microbiota and metabolic health: The potential beneficial effects of a medium chain triglyceride diet in obese individuals. Nutrients 2016, 8, 281. [Google Scholar] [CrossRef]
- Vandanmagsar, B.; Youm, Y.-H.; Ravussin, A.; Galgani, J.E.; Stadler, K.; Mynatt, R.L.; Ravussin, E.; Stephens, J.M.; Dixit, V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011, 17, 179–188. [Google Scholar] [CrossRef]
- Mori, M.A.; Bezy, O.; Kahn, C.R. Metabolic syndrome: is Nlrp3 inflammasome a trigger or a target of insulin resistance? Circ. Res. 2011, 108, 1160–1162. [Google Scholar] [CrossRef]
- Esser, N.; L’homme, L.; De Roover, A.; Kohnen, L.; Scheen, A.J.; Moutschen, M.; Piette, J.; Legrand-Poels, S.; Paquot, N. Obesity phenotype is related to NLRP3 inflammasome activity and immunological profile of visceral adipose tissue. Diabetologia 2013, 56, 2487–2497. [Google Scholar] [CrossRef]
- Murphy, A.J.; Kraakman, M.J.; Kammoun, H.L.; Dragoljevic, D.; Lee, M.K.S.; Lawlor, K.E.; Wentworth, J.M.; Vasanthakumar, A.; Gerlic, M.; Whitehead, L.W.; et al. IL-18 Production from the NLRP1 Inflammasome Prevents Obesity and Metabolic Syndrome. Cell Metab. 2016, 23, 155–164. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): a prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef]
- Timóteo, A.T. Diet in patients with metabolic syndrome: What is the ideal macronutrient composition? Rev. Port. Cardiol. 2018, 37, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Martin-Calvo, N. Mediterranean diet and life expectancy; Beyond olive oil, fruits, and vegetables. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 401–407. [Google Scholar] [CrossRef]
- Karamanos, B.; Thanopoulou, A.; Angelico, F.; Assaad-Khalil, S.; Barbato, A.; Del Ben, M.; Dimitrijevic-Sreckovic, V.; Djordjevic, P.; Gallotti, C.; Katsilambros, N.; et al. Nutritional habits in the Mediterranean Basin. The macronutrient composition of diet and its relation with the tradiational Mediterranean diet. Multi-centre study of the Mediterranean Group for the study of diabetes (MGSD). Eur. J. Clin. Nutr. 2002, 56, 983–991. [Google Scholar] [CrossRef]
- Di Daniele, N.D.; Noce, A.; Vidiri, M.F.; Moriconi, E.; Marrone, G.; Annicchiarico-Petruzzelli, M.; D’Urso, G.; Tesauro, M.; Rovella, V.; De Lorenzo, A.D. Impact of Mediterranean diet on metabolic syndrome, cancer and longevity. Oncotarget 2017, 8, 8947–8979. [Google Scholar] [CrossRef]
- Billingsley, H.E.; Carbone, S. The antioxidant potential of the Mediterranean diet in patients at high cardiovascular risk: An in-depth review of the PREDIMED. Nutr. Diabetes 2018, 8, 1–8. [Google Scholar] [CrossRef]
- Park, Y.-M.; Steck, S.E.; Fung, T.T.; Zhang, J.; Hazlett, L.J.; Han, K.; Merchant, A.T. Mediterranean diet and mortality risk in metabolically healthy obese and metabolically unhealthy obese phenotypes. Int. J. Obes. 2016, 40, 1541–1549. [Google Scholar] [CrossRef]
- Arenaza, L.; Huybrechts, I.; Ortega, F.B.; Ruiz, J.R.; De Henauw, S.; Manios, Y.; Marcos, A.; Julián, C.; Widhalm, K.; Bueno, G.; et al. Adherence to the Mediterranean diet in metabolically healthy and unhealthy overweight and obese European adolescents: the HELENA study. Eur. J. Nutr. 2019, 58, 2615–2623. [Google Scholar] [CrossRef]
- Bell, J.A.; Kivimaki, M.; Hamer, M. Metabolically healthy obesity and risk of incident type 2 diabetes: a meta-analysis of prospective cohort studies. Obes. Rev. 2014, 15, 504–515. [Google Scholar] [CrossRef]
| Parameters Used in the Multiple MHO Definitions | References |
|---|---|
| CVD diagnosis | [12,55] |
| Evaluation of insulin sensitivity (determined by euglycemic-hyperinsulinemic clamp, homeostatic model assessment-insulin resistance (HOMA-IR), Matsuda index, insulin suppression test, glucose disposal rate, triglyceride glucose index, and oral glucose tolerance test) | [3,5,6,7,10,11,12,15,16,30,31,32,35,38,40,42,46,48,53,56,59,64,68,71,72,74,77,78,79,84,85,86,87,89] |
| Determination of systolic and diastolic blood pressure (including information on antihypertensive drug treatment) | [5,6,7,9,10,11,12,16,30,31,32,35,38,42,46,48,52,53,54,55,56,57,58,59,61,64,68,69,70,73,75,76,79,80,81,82,83,84,86,87,88,89] |
| Circulating lipid profile (apolipoprotein B, triglycerides and total-, low density lipoprotein (LDL)-, and HDL-cholesterol as well as triglycerides/HDL-cholesterol, total-cholesterol/HDL-cholesterol, and % of LDL particles with diameter <255 Å; plus data on associated medication treatment) | [3,5,6,7,9,10,11,12,16,30,31,32,35,38,42,46,48,52,53,54,55,56,57,58,59,61,64,68,69,70,71,73,75,76,77,79,80,81,82,83,84,86,87,88,89] |
| Circulating glucose levels and related parameters (glycated hemoglobin and history/diagnosis of T2DM as well as use of blood glucose lowering agents/T2DM treatment) | [5,6,7,9,10,11,12,16,30,31,32,35,38,42,46,48,52,53,55,56,57,58,59,61,64,68,69,70,73,75,76,79,80,81,82,83,84,86,87,88,89] |
| Circulating insulin levels | [12,16,32,54,56,68] |
| Circulating inflammatory profile (C-reactive protein, fibrinogen and white blood cell count) | [6,11,12,16,31,32,42,52,53,54,56,59,68,75,77,80,83,86,89] |
| Uric acid levels | [12,16] |
| Waist circumference | [6,10,11,16,30,31,32,35,52,53,56,61,64,68,69,79,81,82,88,89] |
| Assessment of cardiorespiratory fitness | [56,89] |
| Ridker et al. 2003 [61] | Song et al. 2007 [75] | Khan et al. 2011 [80] | Hamer et al. 2012a [83] | Hamer et al. 2012b [52] | Iacobellis et al. 2005 [12] | St-Pierre et al. 2005 [54] | Karelis et al. 2008 [77] | Wildman et al. 2008 [86] | Ogorodnikova et al. 2012 [59] | |
|---|---|---|---|---|---|---|---|---|---|---|
| Glucose | ≥110 mg/dL | Diagnosis of incident T2DM during follow-up | ≥100 mg/dL or self-reported use of antidiabetic medications | HbA1c > 6.0% or doctor diagnosed DM | Doctor diagnosed DM | <100 mg/dL or 2-h glucose levels < 140 mg/dL during OGTT | - | - | ≥100 mg/dL or antidiabeticmedication use | ≥100 mg/dL or DM treatment |
| Blood pressure | SBP/DBP ≥ 135/85 mm Hg | SBP/DBP ≥135/85 mm Hg | SBP/DBP ≥ 130/85 mm Hg or antihypertensive medication use | SBP/DBP > 130/85 mm Hg or hypertension diagnosis or antihypertensive medication use | SBP/DBP > 130/85 mm Hg or hypertension diagnosis or antihypertensive medication use | SBP/DBP < 130/85 mm Hg | SBP/DBP ≥ 135/85 mm Hg | - | SBP/DBP ≥130/85 mm Hg or antihypertensive medication use | SBP/DBP ≥130/85 mm Hg or antihypertensive medication use |
| HDL-C | <50 mg/dL | <50 mg/dL | ≤50 mg/dL or lipid lowering medication use | <1.03 mmol/L in men and <1.30 mmol/L in women <40 mg/dL in men and <50 mg/dL in women | <1.03 mmol/L in men and <1.30 mmol/L in women <40 mg/dL in men and <50 mg/dL in women | >40 mg/dL in men and >50 mg/dL in women | <1.0 mmol/L <39 mg/dL | ≥1.3 mmol/L ≥50 mg/dL | <40 mg/dL in men, <50 mg/dL in women or lipid-lowering medication use | <40 mg/dL in men, <50 mg/dL in women or lipid-lowering treatment use |
| Triglycerides | ≥150 mg/dL | ≥150 mg/dL | ≥150 mg/dL | ≥1.7 mmol/L ≥150 mg/dL | - | <150 mg/dL | ≥1.7 mmol/L ≥150 mg/dL | ≤1.7 mmol/L ≤150 mg/dL | ≥150 mg/dL | ≥150 mg/dL |
| Other lipid parameters | - | - | - | - | LDL-C < 130 mg/dL, TC < 200 mg/dL, TG/HDL-C < 3.00 and TC/HDL-C < 4.4 | LDL% < 255 Å ≥54.5% ApoB ≥ 1.36 g/L | LDL-C ≤ 2.6 mmol/L LDL-C ≤ 100 mg/dL | - | - | |
| Insulin sensibility | - | - | - | - | Insulin < 15 microU/ mL | Insulin ≥ 85.2 pmol/L | HOMA index ≤ 2.7 | HOMA-IR > 90th percentile (>5.13) | HOMA-IR > 75th percentile (cut-off = 4.03) | |
| Inflammatory markers | (Distribution of CRP levels and stratification for CRP ≥ 3.0 mg/dL vs. ˂3.0 mg/dL) | (Additional stratification for CRP > 3.0 mg/dL vs. ≤3.0 mg/dL) | CRP ≥ 3.0 mg/dL | CRP ≥ 3.0 mg/L | CRP ≥ 3.0 mg/L | WBC < 10,000 cells/mm3 and plasma fibrinogen < 4.0 g/L | CRP ≥ 3.0 mg/L | hsCRP ≤ 3.0 mg/L | hsCRP > 90th percentile (>0.1 mg/L) | WBC > 75th percentile (cut-off = 7000 cells/mm3) |
| Other parameters | WC ˃ 88 cm | - | - | - | WC > 102 cm in men and >88 cm in women | Uric acid < 5.6 mg/dL in women and <7.0 mg/dL in men; no clinically significant abnormalities on physical examination, no lipid-lowering, hypoglycemic, or antihypertensive drugs, normal thyroid function, no history of metabolic, cardiovascular, respiratory, or other systemic diseases and normal ECG | Nondiabetic individuals free of ischemic heart | - | - | - |
| MHO definition | <3 cardiometabolic abnormalities | <3 cardiometabolic abnormalities | <3 cardiometabolic abnormalities | ˂2 cardiometabolic abnormalities | ˂2 cardiometabolic abnormalities | All these criteria | <3 cardiometabolic abnormalities | ≥4 cardiometabolic abnormalities | ˂2 cardiometabolic abnormalities | ≤1 cardiometabolic Abnormalities |
| Ridker et al. 2003 [61] | Song et al. 2007 [75] | Khan et al. 2011 [80] | Hamer et al. 2012a [83] | Hamer et al. 2012b [52] | Iacobellis et al. 2005 [12] | St-Pierre et al. 2005 [54] | Karelis et al. 2008 [77] | Wildman et al. 2008 [86] | Ogorodnikova et al. 2012 [59] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brandão, I.; Martins, M.J.; Monteiro, R. Metabolically Healthy Obesity—Heterogeneity in Definitions and Unconventional Factors. Metabolites 2020, 10, 48. https://doi.org/10.3390/metabo10020048
Brandão I, Martins MJ, Monteiro R. Metabolically Healthy Obesity—Heterogeneity in Definitions and Unconventional Factors. Metabolites. 2020; 10(2):48. https://doi.org/10.3390/metabo10020048
Chicago/Turabian StyleBrandão, Inês, Maria João Martins, and Rosário Monteiro. 2020. "Metabolically Healthy Obesity—Heterogeneity in Definitions and Unconventional Factors" Metabolites 10, no. 2: 48. https://doi.org/10.3390/metabo10020048
APA StyleBrandão, I., Martins, M. J., & Monteiro, R. (2020). Metabolically Healthy Obesity—Heterogeneity in Definitions and Unconventional Factors. Metabolites, 10(2), 48. https://doi.org/10.3390/metabo10020048






