A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part II
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Solutions and Sample Preparation
2.3. Fluorescence and the UV-VIS Spectra
2.4. Circular Dichroism
2.5. Statistical Analysis
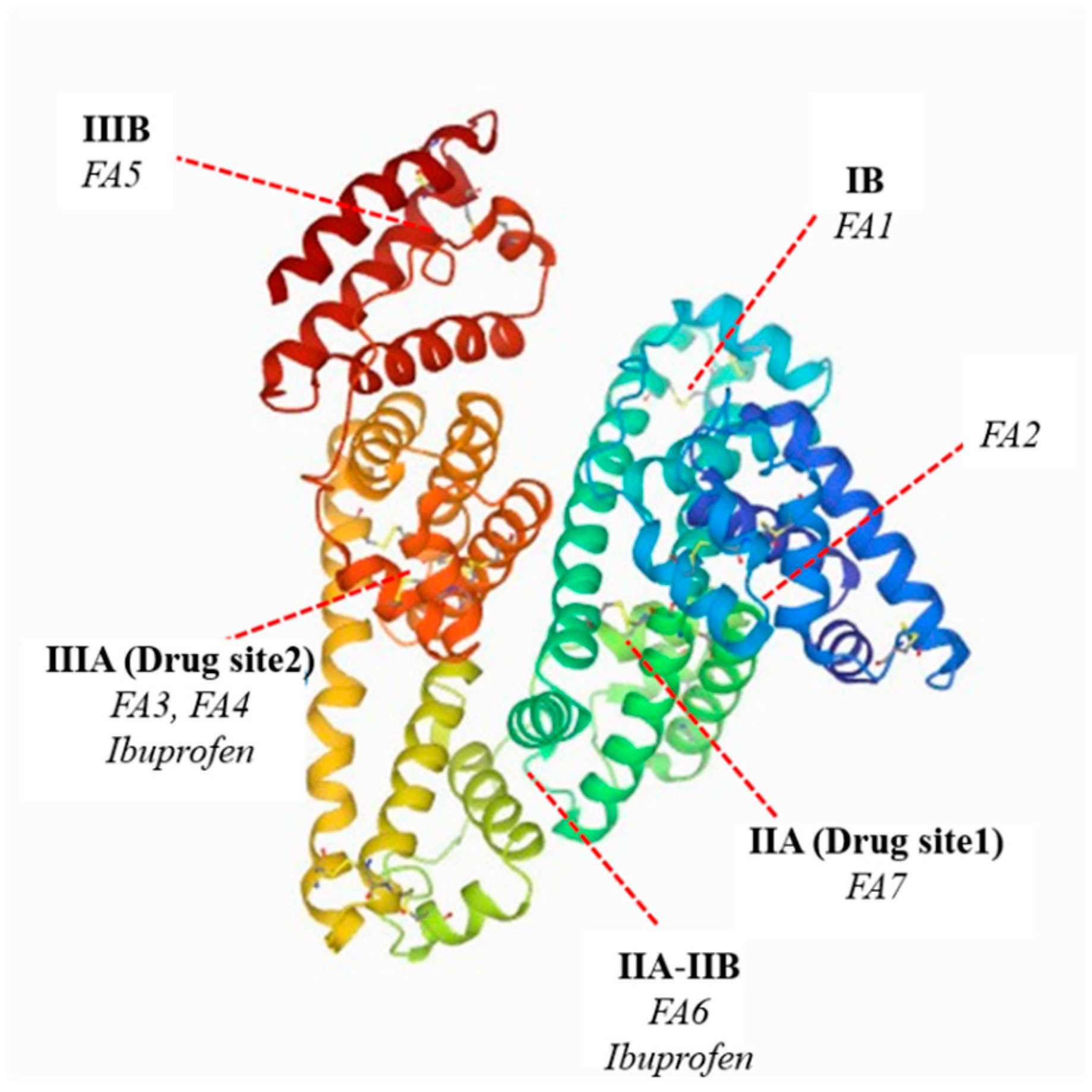
3. Results and Discussion
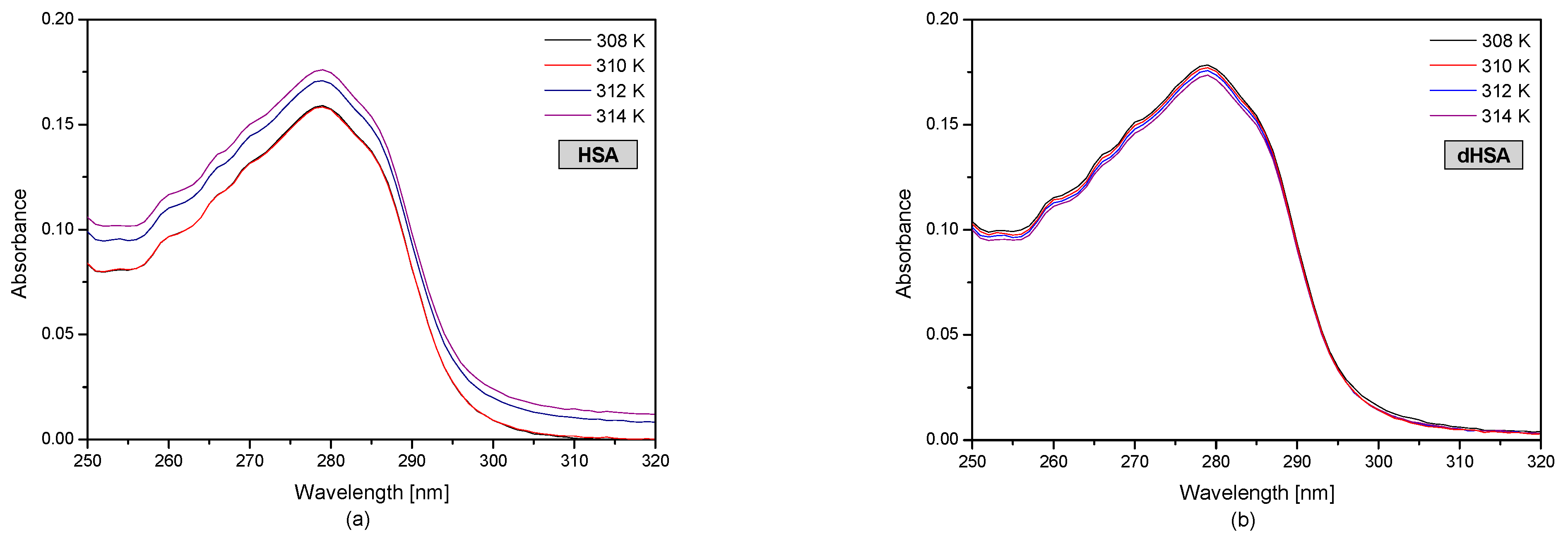
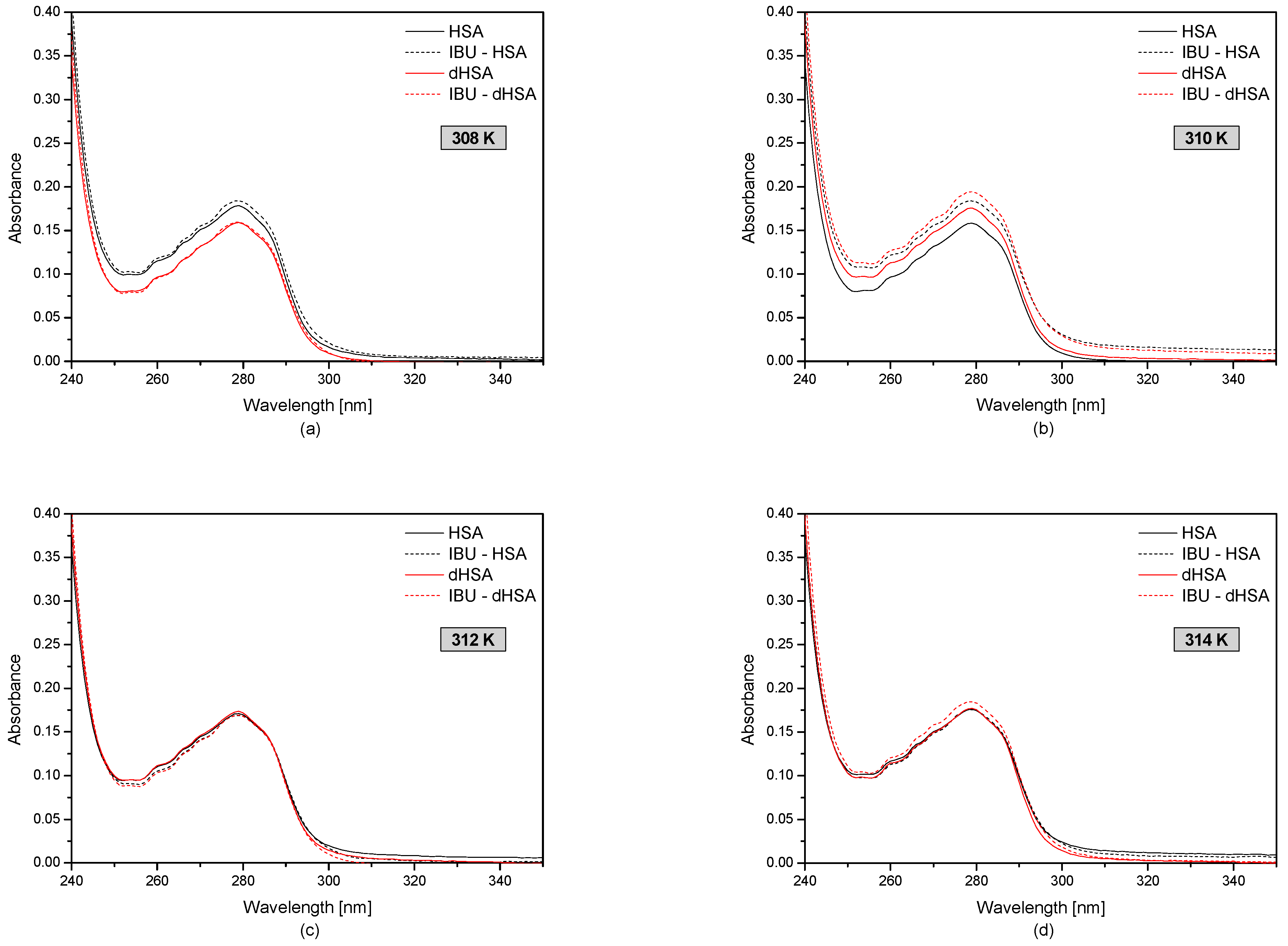
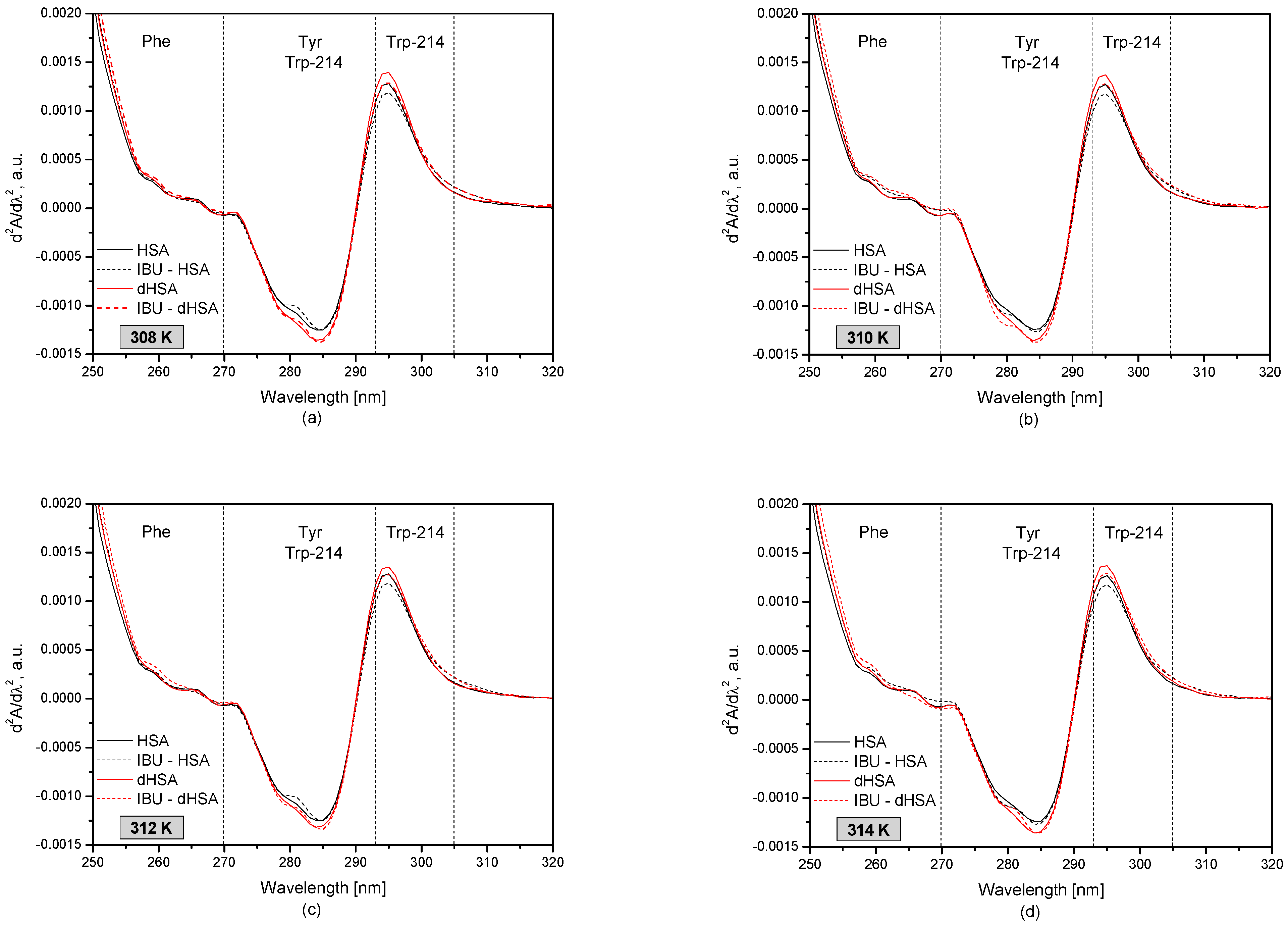
3.1. The UV-VIS Analysis of Effect of Fatty Acids and Temperature on the Binding of Human Serum Albumin with Ibuprofen
3.2. The Fluorescence Analysis of Effect of Fatty Acids and Temperature on the Binding of Human Serum Albumin to Ibuprofen
3.3. The Circular Dichroism Analysis of Effect of Fatty Acids and Temperature on the Binding of Human Serum Albumin with Ibuprofen
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Anand, U.; Mukherjee, S. Binding, unfolding and refolding dynamics of serum albumins. Biochim. Biophys. Acta 2013, 1830, 5394–5404. [Google Scholar] [CrossRef]
- Kragh-Hansen, U.; Minchiotti, L.; Galiano, M.; Peters, T., Jr. Human serum albumin isoforms: Genetic and molecular aspects and functional consequences. Biochim. Biophys. Acta 2013, 1830, 5405–5417. [Google Scholar] [CrossRef]
- Peng, X.; Sun, Y.; Qi, W.; Su, R.; He, Z. Study of the Interaction between Coenzyme Q10 and Human Serum Albumin: Spec-troscopic Approach. J. Solut. Chem. 2014, 43, 585–607. [Google Scholar] [CrossRef]
- He, X.M.; Carter, D.C. Atomic structure and chemistry of human serum albumin. Nature 1992, 358, 209–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peters, T. All about Albumin: Biochemistry, Genetics and Medical Applications; Academic Press: San Diego, CA, USA, 1995. [Google Scholar]
- Sugio, S.; Kashima, A.; Mochizuki, S.; Noda, M.; Kobayashi, K. Crystal structure of human serum albumin at 2.5 A resolution. Protein Eng. 1999, 12, 439–446. [Google Scholar] [CrossRef]
- Carter, D.C.; Ho, J.X. Structure of Serum Albumin. Adv. Protein Chem. 1994, 45, 153–203. [Google Scholar] [CrossRef]
- Kragh-Hansen, U. Molecular aspects of ligand binding to serum albumin. Pharmacol. Rev. 1981, 33, 17–53. [Google Scholar] [PubMed]
- Sudlow, G.; Birkett, D.J.; Wade, D.N. The characterization of two specific drug binding sites on human serum albumin. Mol. Pharmacol. 1975, 11, 824–832. [Google Scholar] [PubMed]
- Sudlow, G.; Birkett, D.J.; Wade, D.N. Further characterization of specific drug binding sites on human serum albumin. Mol. Pharmacol. 1976, 12, 1052–1061. [Google Scholar]
- Kragh-Hansen, U.; Chuang, V.; Otagiri, M. Practical Aspects of the Ligand-Binding and Enzymatic Properties of Human Serum Albumin. Biol. Pharm. Bull. 2002, 25, 695–704. [Google Scholar] [CrossRef] [Green Version]
- Curry, S.; Mandelkow, H.; Brick, P.; Franks, N. Crystal structure of human serum albumin complexed with fatty acid reveals an asymmetric distribution of binding sites. Nat. Struct. Biol. 1998, 5, 827–835. [Google Scholar] [CrossRef]
- Bushra, R.; Aslam, N. An Overview of Clinical Pharmacology of Ibuprofen. Oman. Med. J. 2010, 25, 155–161. [Google Scholar] [CrossRef]
- Amirimoghadam, P.; Zihayat, B.; Dabaghzadeh, F.; Kiani, K.; Ebrahimi, J.; Ghazanfari, M. Evaluation and awareness of over the counter use of non-steroidal anti-inflammatory drugs? J. Appl. Pharm. Sci. 2017, 7, 154–159. [Google Scholar]
- Varrassi, G.; Pergolizzi, J.V.; Dowling, P.; Paladini, A. Ibuprofen Safety at the Golden Anniversary: Are all NSAIDs the Same? A Narrative Review. Adv. Ther. 2020, 37, 61–82. [Google Scholar] [CrossRef] [Green Version]
- Halford, G.M.; Lordkipanidzé, M.; Watson, S.P. 50th anniversary of the discovery of ibuprofen: An interview with Dr Stewart Adams. Platelets 2012, 23, 415–422. [Google Scholar] [CrossRef]
- Ngo, V.T.H.; Bajaj, T. Ibuprofen; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Van der Vusse, G.J. Albumin as fatty acid transporter. Drug Metab. Pharmacokinet. 2009, 24, 300–307. [Google Scholar] [CrossRef]
- Huang, B.X.; Dass, C.; Kim, H.Y. Probing conformational changes of human serum albumin due to unsaturated fatty acid binding by chemical cross-linking and mass spectrometry. Biochem. J. 2005, 387, 695–702. [Google Scholar] [CrossRef]
- Høstmark, A.T. Serum albumin and prevalence of coronary heart disease: A population-based, cross-sectional study. Nor. Epidemiol. 2003, 13, 107–113. [Google Scholar] [CrossRef]
- Ghuman, J.; Zunszain, P.A.; Petitpas, I.; Bhattacharya, A.A.; Otagiri, M.; Curry, S. Structural Basis of the Drug-binding Spec-ificity of Human Serum Albumin. J. Mol. Biol. 2005, 353, 38–52. [Google Scholar] [CrossRef]
- Ashrafi-Kooshk, M.R.; Ebrahimi, F.; Ranjbar, S.; Ghobadi, S.; Moradi, N.; Khodarahmi, R. Comparative studies on drug binding to the purified and pharmaceutical grade human serum albumin. Biologicals 2015, 43, 333–343. [Google Scholar] [CrossRef]
- Salahuddin, P. Urea and Acid Induced Unfolding of Fatted and Defatted Human Serum Albumin. Protein Pept. Lett. 2008, 15, 826–833. [Google Scholar] [CrossRef]
- Yamasaki, K.; Hyodo, S.; Taguchi, K.; Nishi, K.; Yamaotsu, N.; Hirono, S.; Chuang, V.T.G.; Seo, H.; Maruyama, T.; Otagiri, M. Long chain fatty acids alter the interactive binding of ligands to the two principal drug binding sites of human serum albumin. PLoS ONE 2017, 12, e0180404. [Google Scholar] [CrossRef] [Green Version]
- Krenzel, E.S.; Chen, Z.; Hamilton, A. Correspondence of Fatty Acid and Drug Binding Sites on Human Serum Albumin: A Two-Dimensional Nuclear Magnetic Resonance Study. Biochemistry 2013, 52, 1559–1567. [Google Scholar] [CrossRef]
- Yamazaki, E.; Inagaki, M.; Kurita, O.; Inoue, T. Kinetics of fatty acid binding ability of glycated human serum albumin. J. Biosci. 2005, 30, 475–481. [Google Scholar] [CrossRef]
- Spector, A.A. Fatty acid binding to plasma albumin. J. Lipid Res. 1975, 16, 165–179. [Google Scholar] [CrossRef]
- Miyamoto, H.; Matsueda, S.; Moritsuka, A.; Shimokawa, K.; Hirata, H.; Nakashima, M.; Sasaki, H.; Fumoto, S.; Nishida, K. Evaluation of hypothermia on the in vitro metabolism and binding and in vivo disposition of midazolam in rats: Hypothermic effects on midazolam disposition in rats. Biopharm. Drug Dispos. 2015, 36, 481–489. [Google Scholar] [CrossRef] [Green Version]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: New York, NY, USA, 2006. [Google Scholar] [CrossRef]
- Galley, W.C.; Bouvier, M.; Clas, S.D.; Brown, G.R. A simplified analysis of scatchard plots for systems with two interacting binding sites. Biopolymers 1988, 27, 79–86. [Google Scholar] [CrossRef]
- Zhao, P.; Zhu, G.; Zhang, W.; Zhang, L.; Liang, Z.; Zhang, Y. Study of multiple binding constants of dexamethasone with human serum albumin by capillary electrophoresis–frontal analysis and multivariate regression. Anal. Bioanal. Chem. 2008, 393, 257–261. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, H.; Niu, Q.; Ye, D.; Liang, G. Binding between Saikosaponin C and Human Serum Albumin by Fluorescence Spectroscopy and Molecular Docking. Molecules 2016, 21, 153. [Google Scholar] [CrossRef]
- Hill, A.V. The possible effects of the aggregation of the molecules of haemoglobin on its dissociation curves. J. Physiol. 1910, 40, 4–7. [Google Scholar]
- Venyaminov, S.Y.; Yang, J.T. Circular Dichroism and the Conformational Analysis of Biomolecules; Fasman, G.D., Ed.; Plenum Press: New York, NY, USA, 1996; pp. 69–109. [Google Scholar]
- Liu, J.; Tian, J.N.; Zhang, J.; Hu, Z.; Chen, X. Interaction of magnolol with bovine serum albumin: A fluorescence quenching study. Anal. Bioanal. Chem. 2003, 376, 864–867. [Google Scholar] [CrossRef]
- Yue, Y.; Chen, X.; Qin, J.; Yao, X. Characterization of the mangiferin-human serum albumin complex by spectroscopic and molecular modeling approaches. J. Pharm. Biomed. 2009, 49, 753–759. [Google Scholar] [CrossRef]
- Oliveira, A.F.; Cunha, D.A.; Ladriere, L.; Igoillo-Esteve, M.; Bugliani, M.; Marchetti, P.; Cnop, M. In vitro use of free fatty acids bound to albumin: A comparison of protocols. Biotechniques 2015, 58, 228–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuś, S.; Marczenko, Z.; Obarski, N. Derivative UV-Vis Spectrophotometry in Analytical Chemistry. Chem. Anal. 1996, 41, 899–927. [Google Scholar]
- Balestrieri, C.; Colonna, G.; Giovane, A.; Irace, G.; Servillo, L. Second-Derivative Spectroscopy of Proteins: A Method for the Quantitiative Determination of Aromatic Amino Acids in Proteins. Eur. J. Biol. Chem. 1978, 90, 433–440. [Google Scholar] [CrossRef]
- Levine, R.L.; Federici, M.M. Quantitation of aromatic residues in proteins: Model compounds for second-derivative spectros-copy. Biochemistry 1982, 21, 2600–2606. [Google Scholar] [CrossRef]
- Chadborn, N.; Bryant, J.; Bain, A.J.; O’Shea, P. Ligand-Dependent Conformational Equilibria of Serum Albumin Revealed by Tryptophan Fluorescence Quenching. Biophys. J. 1999, 76, 2198–2207. [Google Scholar] [CrossRef] [Green Version]
- Amézqueta, S.; Beltrán, J.L.; Bolioli, A.M.; Campos-Vicens, L.; Luque, F.J.; Ràfols, C. Evaluation of the Interactions between Human Serum Albumin (HSA) and Non-Steroidal Anti-Inflammatory (NSAIDs) Drugs by Multiwavelength Molecular Fluo-rescence, Structural and Computational Analysis. Pharmaceuticals 2021, 14, 214. [Google Scholar] [CrossRef]
- Ding, F.; Buldyrev, S.V.; Dokholyan, N.V. Folding Trp-Cage to NMR Resolution Native Structure Using a Coarse-Grained Protein Model. Biophys. J. 2005, 88, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Ploch-Jankowska, A.; Pentak, D. A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part I. Pharmaceuticals 2020, 13, 205. [Google Scholar] [CrossRef]
- Evoli, S.; Mobley, D.L.; Guzzi, R.; Rizzuti, B. Multiple binding modes of ibuprofen in human serum albumin identified by absolute binding free energy calculations. Phys. Chem. Chem. Phys. 2016, 18, 32358–32368. [Google Scholar] [CrossRef] [Green Version]
- Eftink, M.R.; Ghiron, C.A. Exposure of tryptophanyl residues in proteins. Quantitative determination by fluorescence quenching studies. Biochemistry 1976, 15, 672–680. [Google Scholar] [CrossRef]
- Hashempour, S.; Shahabadi, N.; Adewoye, A.; Murphy, B.; Rouse, C.; Salvatore, B.A.; Stratton, C.; Mahdavian, E. Binding Studies of AICAR and Human Serum Albumin by Spectroscopic, Theoretical, and Computational Methodologies. Molecules 2020, 25, 5410. [Google Scholar] [CrossRef]
- Varshney, A.; Ahmad, B.; Khan, R.H. Comparative studies of unfolding and binding of ligands to human serum albumin in the presence of fatty acid: Spectroscopic approach. Int. J. Biol. Macromol. 2008, 42, 483–490. [Google Scholar] [CrossRef]
- Gesztelyi, R.; Zsuga, J.; Kemeny-Beke, A.; Varga, B.; Juhasz, B.; Tosaki, A. The Hill equation and the origin of quantitative pharmacology. Arch. Hist. Exact Sci. 2012, 66, 427–438. [Google Scholar] [CrossRef]
- Greenfield, N.J. Using circular dichroism spectra to estimate protein secondary structure. Nat. Protoc. 2006, 1, 2876–2890. [Google Scholar] [CrossRef]
- Whitmore, L.; Wallace, B.A. Protein secondary structure analyses from circular dichroism spectroscopy: Methods and refer-ence databases. Biopolymers 2008, 89, 392–400. [Google Scholar] [CrossRef]
- Kosa, T.; Maruyama, T.; Sakai, N.; Yonemura, N.; Yahara, S.; Otagiri, M. Species differences of serum albumins: III. Analysis of structural characteristics and ligand binding properties during N-B transitions. Pharm. Res. 1998, 15, 592–598. [Google Scholar] [CrossRef]
- Wang, W.; Nema, S.; Teagarden, D. Protein aggregation—Pathways and influencing factors. Int. J. Pharm. 2010, 390, 89–99. [Google Scholar] [CrossRef]

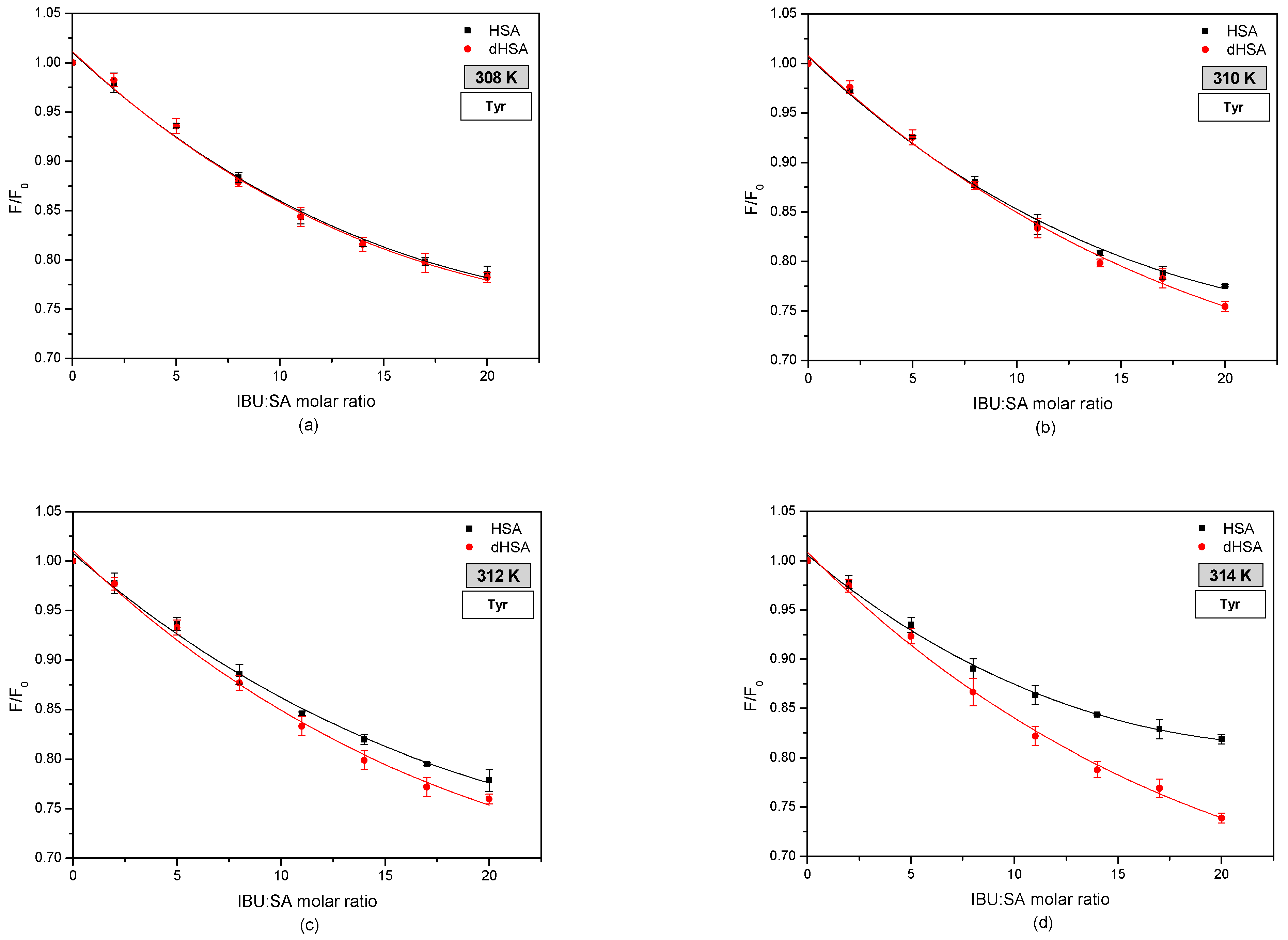
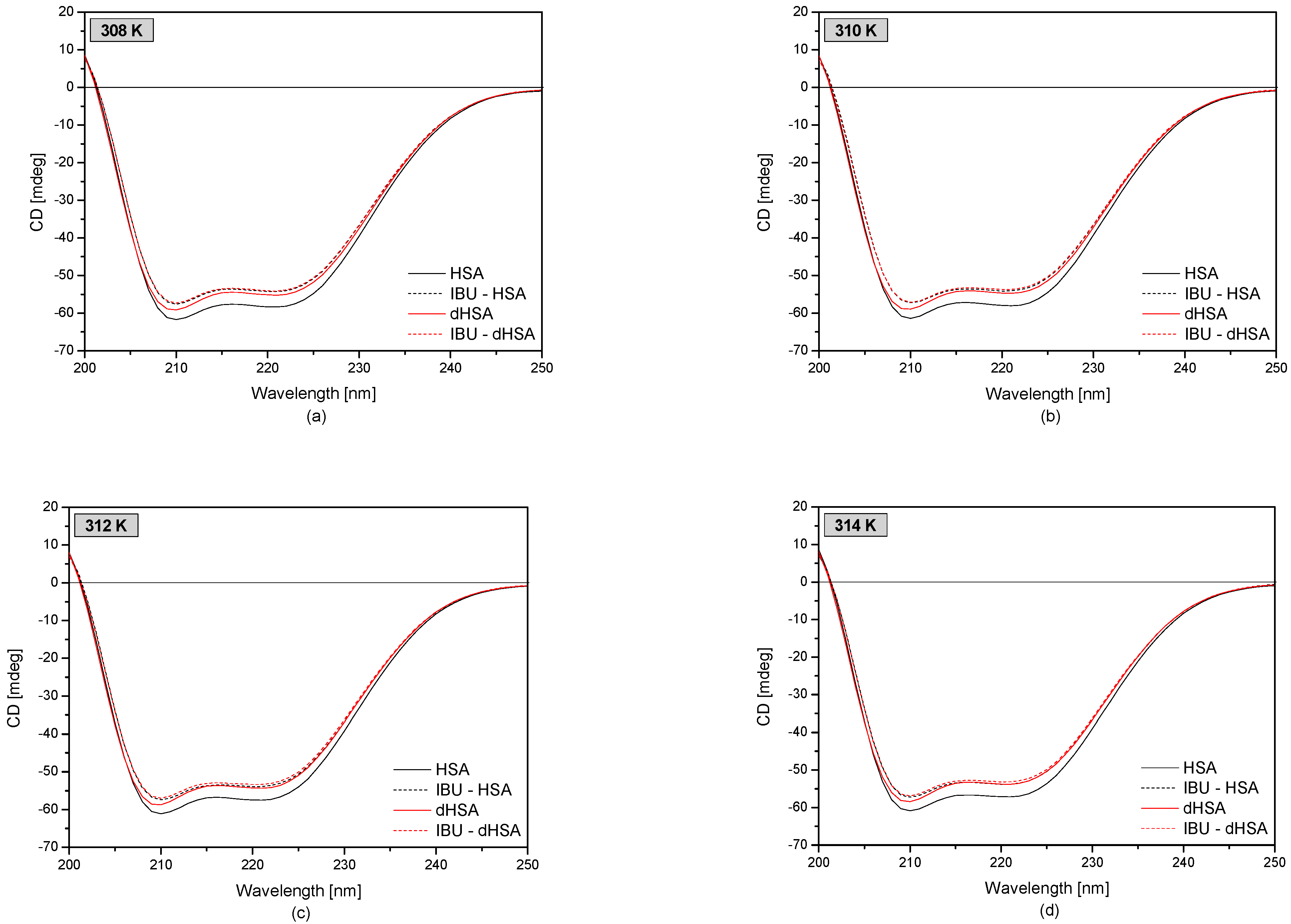
| KSV (M−1) ± SD × 10−4 (M−1) | ||||
|---|---|---|---|---|
| λex 275 nm | λex 295 nm | Tyr | ||
| 308 K | HSA | 2.83 ± 0.04 | 0.73 ± 0.11 | 0.23 ± 0.12 |
| dHSA | 4.09 ± 0.13 | 1.48 ± 0.01 | 0.83 ± 0.03 | |
| 310 K | HSA | 3.10 ± 0.08 | 0.88 ± 0.05 | 0.33 ± 0.09 |
| dHSA | 5.05 ± 0.48 | 1.24 ± 0.02 | 0.51 ± 0.07 | |
| 312 K | HSA | 3.95 ± 0.11 | 0.87 ± 0.06 | 0.38 ± 0.06 |
| dHSA | 5.02 ± 0.47 | 1.27 ± 0.02 | 0.47 ± 0.07 | |
| 314 K | HSA | 4.76 ± 0.17 | 1.26 ± 0.04 | 0.53 ± 0.12 |
| dHSA | 6.71 ± 0.17 | 1.66 ± 0.01 | 0.57 ± 0.05 | |
| Klotz Method | Hill Method | |||
|---|---|---|---|---|
| Ka (M−1) ± SD × 10−4 (M−1) n ± SD | Ka (M−1) ± SD × 10−4 (M−1) nH ± SD | |||
| λex 275 nm | HSA | dHSA | HSA | dHSA |
| 308 K | 0.51 ± 0.14 0.99 ± 0.03 | 1.70 ± 0.05 1.19 ± 0.12 | 0.11 ± 0.05 0.80 ± 0.12 | 0.65 ± 0.12 0.90 ± 0.12 |
| 310 K | 1.14 ± 0.11 0.98 ± 0.07 | 2.01 ± 0.13 0.91 ± 0.08 | 0.87 ± 0.10 0.83 ± 0.10 | 0.87 ± 0.07 0.85 ± 0.10 |
| 312 K | 1.02 ± 0.09 1.02 ± 0.05 | 2.35 ± 0.07 0.85 ± 0.13 | 0.79 ± 0.12 0.81 ± 0.11 | 0.89 ± 0.12 0.82 ± 0.10 |
| 314 K | 2.59 ± 0.10 2.59 ± 0.08 | 2.89 ± 0.10 0.96 ± 0.08 | 2.57 ± 0.01 0.84 ± 0.08 | 2.53 ± 0.12 0.86 ± 0.08 |
| λex 295 nm | HSA | dHSA | HSA | dHSA |
| 308 K | 0.83 ± 0.09 2.02 ± 0.21 | 0.82 ± 0.11 2.04 ± 0.43 | 0.63 ± 0.04 0.84 ± 0.02 | 0.86 ± 0.06 1.25 ± 0.23 |
| 310 K | 0.12 ± 0.03 1.01 ± 0.15 | 0.89 ± 0.05 1.01 ± 0. 10 | 0.08 ± 0.02 0.89 ± 0.07 | 1.08 ± 0.05 0.89 ± 0.07 |
| 312 K | 0.10 ± 0.05 1.01 ± 0.09 | 0.98 ± 0.06 0.95 ± 0.12 | 0.06 ± 0.02 0.81 ± 0.07 | 1.32 ± 0.18 1.23 ± 0.13 |
| 314 K | 1.02 ± 0.14 0.98 ± 0.17 | 1.33 ± 0.03 0.97 ± 0.04 | 0.63 ± 0.04 0.92 ± 0.05 | 1.29 ± 0.02 0.92 ± 0.05 |
| Tyr | HSA | dHSA | HSA | dHSA |
| 308 K | 0.09 ± 0.01 1.03 ± 0.03 | 0.74 ± 0.03 0.96 ± 0.05 | 0.10 ± 0.02 1.07 ± 0.05 | 0.68 ± 0.02 1.01 ± 0.03 |
| 310 K | 0.34 ± 0.05 1.02 ± 0.05 | 0.49 ± 0.06 1.06 ± 0.11 | 0.34 ± 0.08 1.03 ± 0.05 | 0.53 ± 0.03 1.03 ± 0.06 |
| 312 K | 0.06 ± 0.01 1.02 ± 0.03 | 0.45 ± 0.07 1.02 ± 0.09 | 0.07 ± 0.03 1.03 ± 0.05 | 0.59 ± 0.03 1.06 ± 0.07 |
| 314 K | 0.51 ± 0.09 1.02 ± 0.02 | 0.55 ± 0.05 0.98 ± 0.13 | 0.57 ± 0.03 1.08 ± 0.07 | 0.53 ± 0.03 1.02 ± 0.05 |
| λmin (nm) | θMRE (deg·cm2·dmol−1) | λmin (nm) | θMRE (deg·cm2·dmol−1) | ||
|---|---|---|---|---|---|
| 308 K | HSA | 210 | −21,051.35 | 221 | −19,917.10 |
| IBU–HSA | 210 | −19,653.67 | 220 | −18,504.21 | |
| dHSA | 210 | −20,173.29 | 221 | −18,834.88 | |
| IBU-dHSA | 210 | −19,559.85 | 221 | −18,466.50 | |
| 310 K | HSA | 210 | −20,928.94 | 221 | −19,795.50 |
| IBU-HSA | 210 | −19,541.80 | 220 | −18,457.74 | |
| dHSA | 210 | −20,101.37 | 221 | −18,665.48 | |
| IBU-dHSA | 210 | −19,478.44 | 220 | −18,318.40 | |
| 312 K | HSA | 210 | −20,852.21 | 221 | −19,626.00 |
| IBU-HSA | 210 | −19,580.87 | 220 | −19,397.07 | |
| dHSA | 210 | −20,031.33 | 221 | −18,544.05 | |
| IBU-dHSA | 210 | −19,448.25 | 220 | −18,203.90 | |
| 314 K | HSA | 210 | −20,765.85 | 221 | −19,494.31 |
| IBU-HSA | 210 | −19,535.56 | 220 | −18,361.93 | |
| dHSA | 210 | −19,921.64 | 221 | −18,349.67 | |
| IBU-dHSA | 210 | −19,415.43 | 220 | −18,142.76 | |
| % α—Helix | % β—Sheet | % Other | ||
|---|---|---|---|---|
| 308 K | HSA | 55.6 | 9.2 | 35.1 |
| IBU-HSA | 59.0 | 1.2 | 39.9 | |
| dHSA | 58.7 | 1.8 | 39.5 | |
| IBU-dHSA | 56.1 | 8.5 | 35.4 | |
| 310 K | HSA | 55.8 | 8.7 | 35.4 |
| IBU-HSA | 59.0 | 1.7 | 39.3 | |
| dHSA | 58.8 | 1.8 | 39.4 | |
| IBU-dHSA | 56.5 | 8.0 | 35.6 | |
| 312 K | HSA | 55.9 | 8.3 | 35.8 |
| IBU-HSA | 58.9 | 1.4 | 39.7 | |
| dHSA | 59.1 | 1.3 | 39.5 | |
| IBU-dHSA | 56.6 | 8.1 | 35.3 | |
| 314 K | HSA | 56.1 | 8.0 | 35.9 |
| IBU-HSA | 58.9 | 1.1 | 40.0 | |
| dHSA | 59.1 | 1.2 | 39.8 | |
| IBU-dHSA | 56.7 | 7.0 | 36.3 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ploch-Jankowska, A.; Pentak, D.; Nycz, J.E. A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part II. Sci. Pharm. 2021, 89, 30. https://doi.org/10.3390/scipharm89030030
Ploch-Jankowska A, Pentak D, Nycz JE. A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part II. Scientia Pharmaceutica. 2021; 89(3):30. https://doi.org/10.3390/scipharm89030030
Chicago/Turabian StylePloch-Jankowska, Anna, Danuta Pentak, and Jacek E. Nycz. 2021. "A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part II" Scientia Pharmaceutica 89, no. 3: 30. https://doi.org/10.3390/scipharm89030030
APA StylePloch-Jankowska, A., Pentak, D., & Nycz, J. E. (2021). A Comprehensive Spectroscopic Analysis of the Ibuprofen Binding with Human Serum Albumin, Part II. Scientia Pharmaceutica, 89(3), 30. https://doi.org/10.3390/scipharm89030030







