Anti-Pollution Activity, Antioxidant and Anti-Inflammatory Effects of Fermented Extract from Smilax china Leaf in Macrophages and Keratinocytes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Standardized SCL Fermented Extracts
2.3. Cytotoxicity Assay
2.4. The DPPH Radical Scavenging Activity
2.5. XRE-Luciferase Activity
2.6. Measurement of Prostaglandin E2 (PGE2) Production
2.7. Statistical Analysis
3. Results
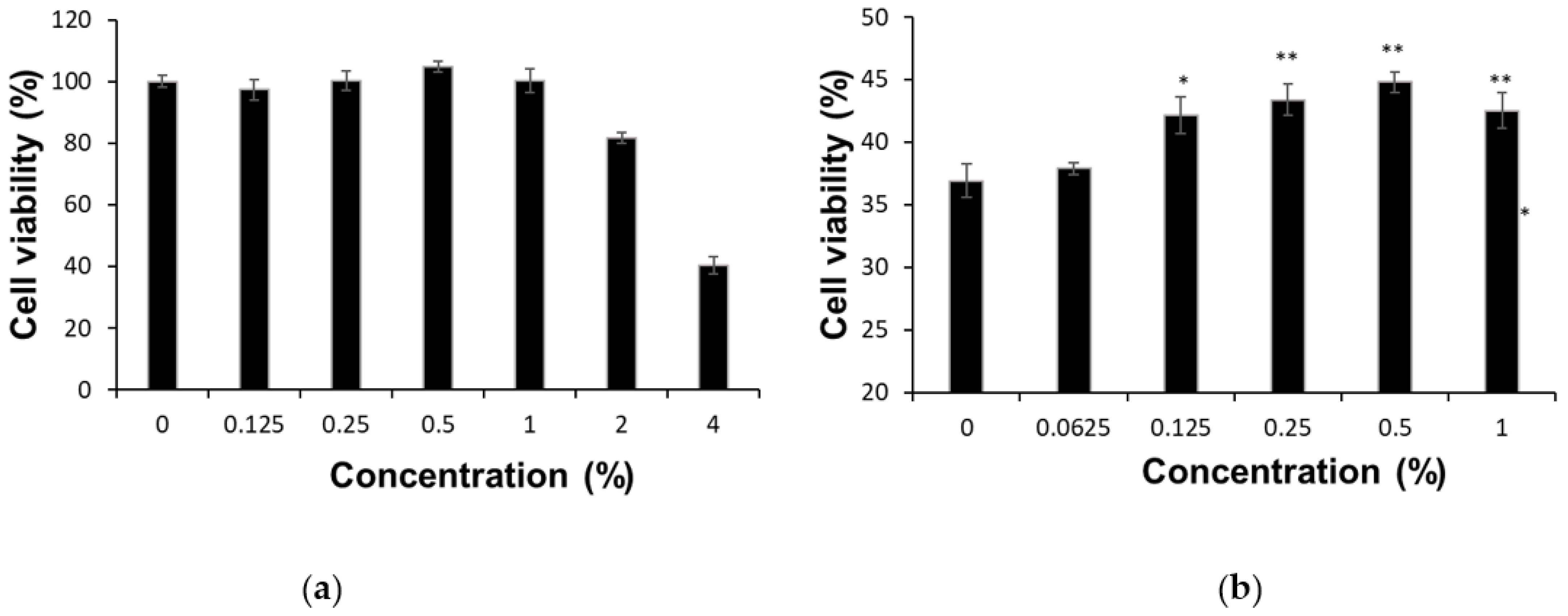
3.1. Cytotoxicity of FESCL in Heavy Metals Exposure
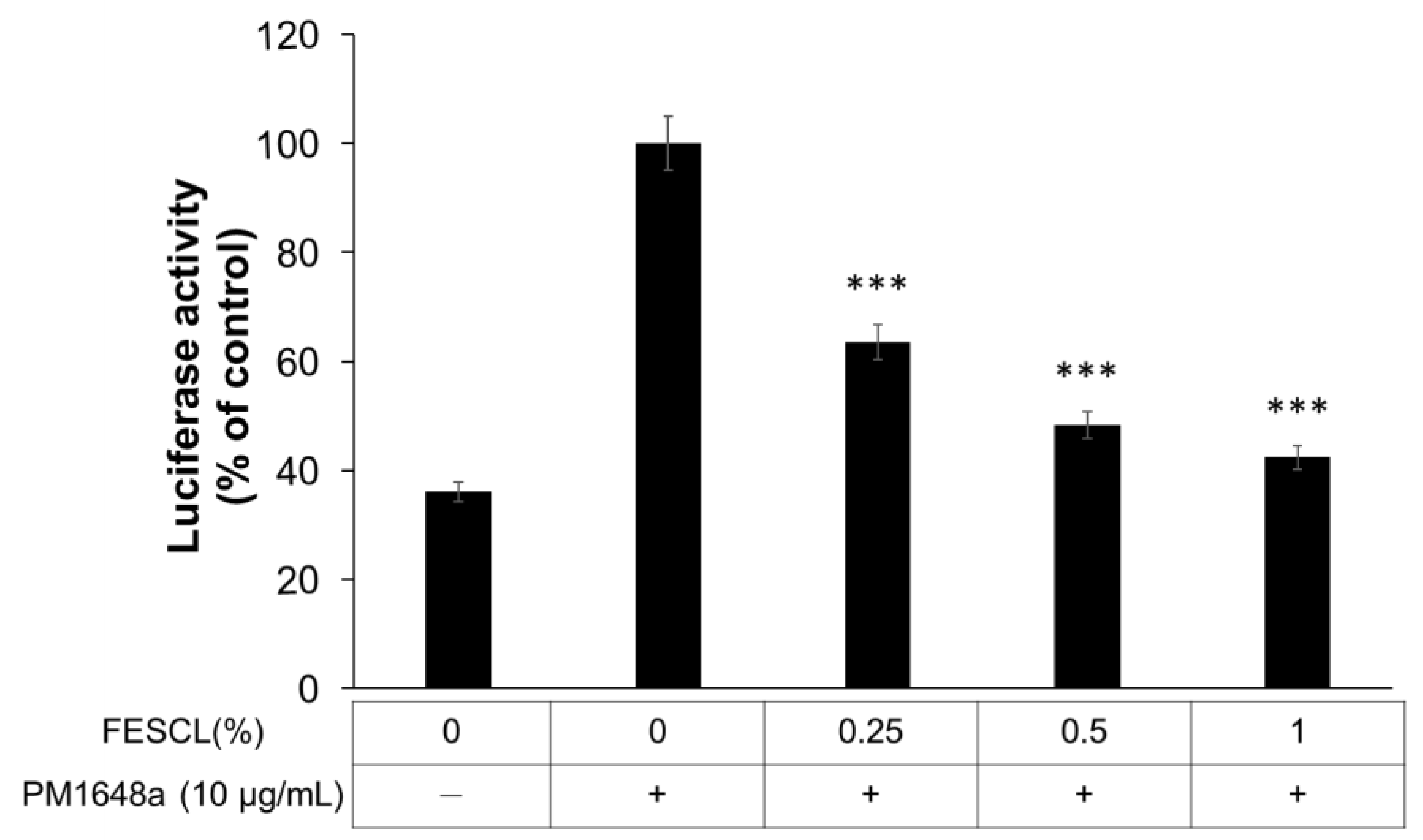
3.2. Inhibitory Effect of FESCL on Air Pollutants
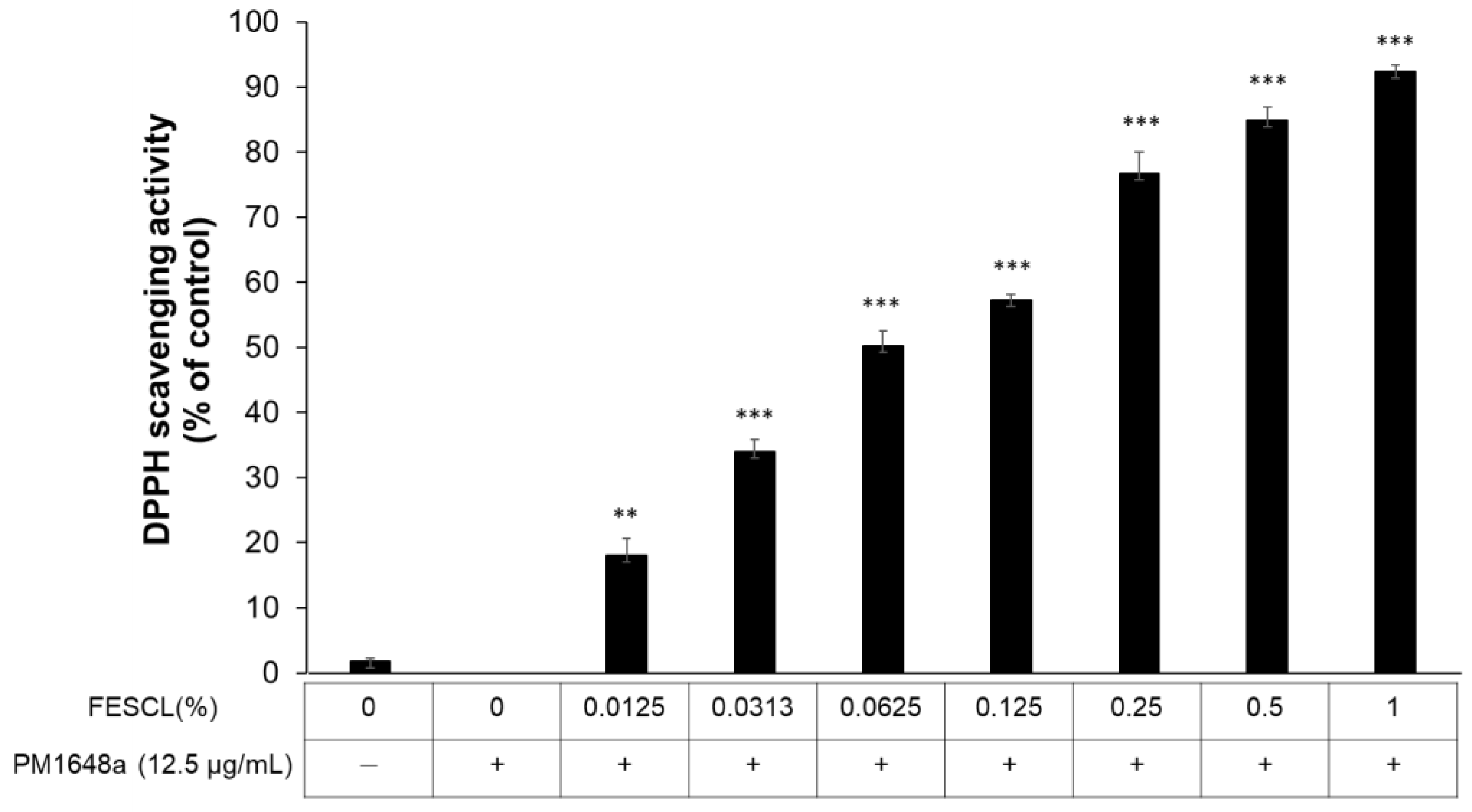
3.3. Free Radical Scavenging Activity of FESCL on Air Pollutants
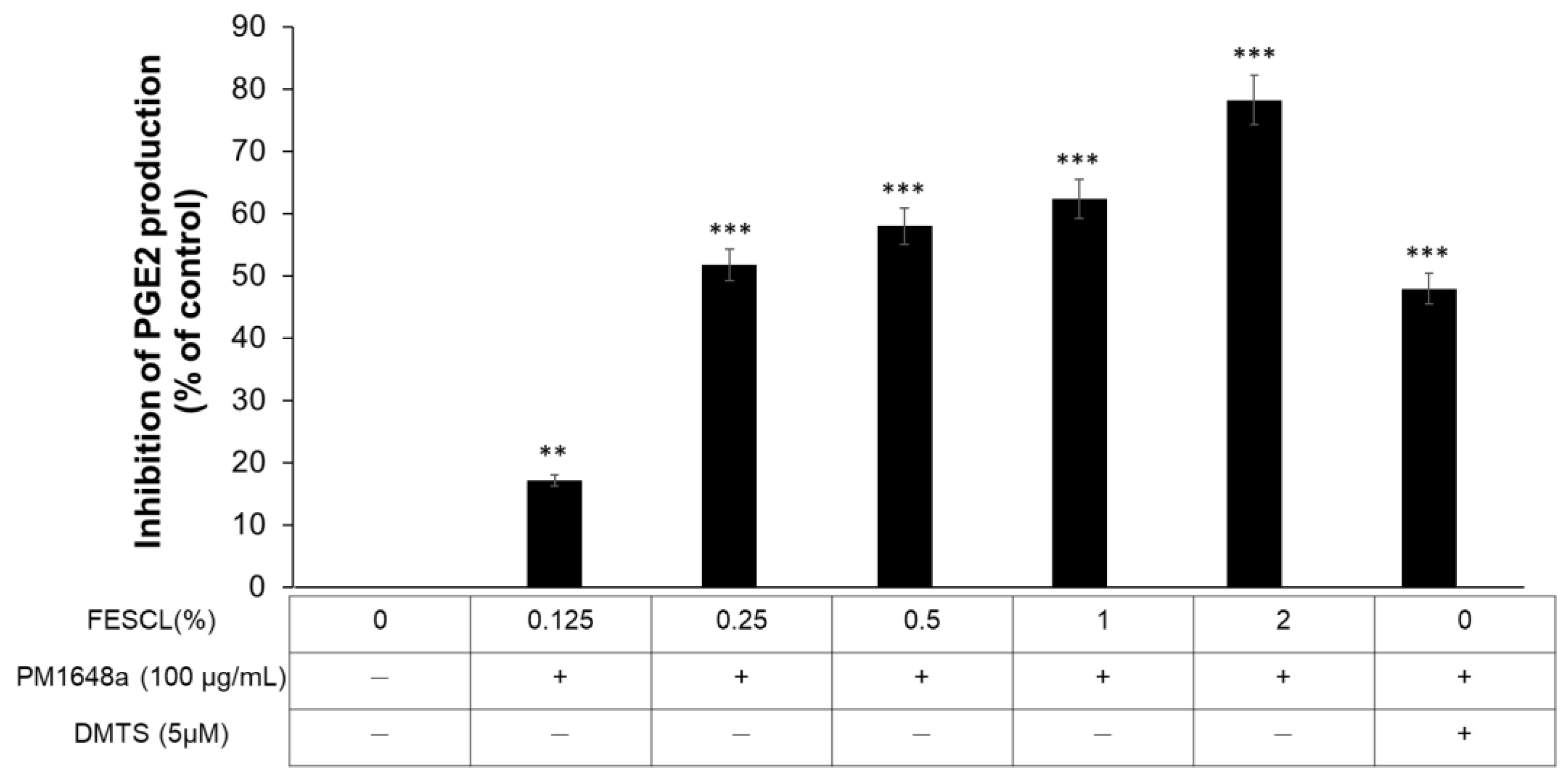
3.4. Anti-Inflammatory Activity of FESCL on Air Pollutants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Juliano, C.; Magrini, G.A. Cosmetic Functional Ingredients from Botanical Sources for Anti-Pollution Skincare Products. Cosmetics 2018, 5, 19. [Google Scholar] [CrossRef]
- Poljšak, B.; Dahmane, R. Free Radicals and Extrinsic Skin Aging. Dermatol. Res. Pr. 2012, 2012, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Araviiskaia, E.; Berardesca, E.; Bieber, T.; Gontijo, G.; Viera, M.S.; Marrot, L.; Chuberre, B.; Dreno, B. The impact of airborne pollution on skin. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1496–1505. [Google Scholar] [CrossRef]
- Ali, A.; Khan, H.; Bahadar, R.; Riaz, A.; Bin Asad, M.H.H. The impact of airborne pollution and exposure to solar ultraviolet radiation on skin: Mechanistic and physiological insight. Environ. Sci. Pollut. Res. 2020, 27, 28730–28736. [Google Scholar] [CrossRef] [PubMed]
- Dijkhoff, I.M.; Drasler, B.; Karakocak, B.B.; Petri-Fink, A.; Valacchi, G.; Eeman, M.; Rothen-Rutishauser, B. Impact of airborne particulate matter on skin: A systematic review from epidemiology to in vitro studies. Part. Fibre Toxicol. 2020, 17, 1–28. [Google Scholar] [CrossRef]
- Araujo, J.A. Particulate air pollution, systemic oxidative stress, inflammation, and atherosclerosis. Air Qual. Atmosphere Health 2010, 4, 79–93. [Google Scholar] [CrossRef]
- Kim, K.E.; Cho, D.; Park, H.J. Air pollution and skin diseases: Adverse effects of airborne particulate matter on various skin diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, E.-H.; Oh, I.; Jung, K.; Han, Y.; Cheong, H.-K.; Ahn, K. Symptoms of atopic dermatitis are influenced by outdoor air pollution. J. Allergy Clin. Immunol. 2013, 132, 495–498.e1. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K. The role of air pollutants in atopic dermatitis. J. Allergy Clin. Immunol. 2014, 134, 993–999. [Google Scholar] [CrossRef]
- Kim, K. Influences of Environmental Chemicals on Atopic Dermatitis. Toxicol. Res. 2015, 31, 89–96. [Google Scholar] [CrossRef]
- Tian, L.-W.; Zhang, Z.; Long, H.-L.; Zhang, Y.-J. Steroidal Saponins from the Genus Smilax and Their Biological Activities. Nat. Prod. Bioprospect. 2017, 7, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-L.; Gan, G.-P.; Zhang, H.-Z.; Wu, H.-Z.; Li, C.-L.; Huang, Y.-P.; Liu, Y.-W.; Liu, J.-W. A flavonoid glycoside isolated from Smilax china L. rhizome in vitro anticancer effects on human cancer cell lines. J. Ethnopharmacol. 2007, 113, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Lü, Y.; Chen, D.; Deng, J.; Tian, L. Effect of Smilax china on adjunctive arthritis mouse. J. Chin. Med. Mater. 2003, 26, 344–346. [Google Scholar]
- Shu, X.-S.; Gao, Z.-H.; Yang, X.-L. Anti-inflammatory and anti-nociceptive activities of Smilax china L. aqueous extract. J. Ethnopharmacol. 2006, 103, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Nisar, M.; Ebad, F.; Nadeem, S.; Saeed, M.; Khan, H.; Samiullah; Khuda, F.; Karim, N.; Ahmad, Z. Anti-inflammatory activities of Sieboldogenin from Smilax china Linn.: Experimental and computational studies. J. Ethnopharmacol. 2009, 121, 175–177. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Pan, X.; Ran, S.; Wang, K. Purification, structural elucidation and anti-inflammatory activity in vitro of polysaccharides from Smilax china L. Int. J. Biol. Macromol. 2019, 139, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.; Yao, X. Protodioscin (NSC-698 796): Its Spectrum of Cytotoxicity Against Sixty Human Cancer Cell Lines in an Anticancer Drug Screen Panel. Planta Med. 2002, 68, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Challinor, V.L.; Parsons, P.G.; Chap, S.; White, E.F.; Blanchfield, J.T.; Lehmann, R.P.; De Voss, J.J. Steroidal saponins from the roots of Smilax sp.: Structure and bioactivity. Steroids 2012, 77, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.-K.; Lee, J.-H.; Kim, H.-S.; Lee, C.-K.; Lee, S.-C. Antioxidant and antimicrobial activities of Smilax china L. leaf extracts. Food Sci. Biotechnol. 2012, 21, 1723–1727. [Google Scholar] [CrossRef]
- Li, Y.; Won, K.J.; Kim, D.Y.; Bin Kim, H.; Kang, H.M.; Lee, S.Y.; Lee, H.M. Positive Promoting Effects of Smilax China Flower Absolute on the Wound Healing/Skin Barrier Repair-Related Responses of HaCaT Human Skin Keratinocytes. Chem. Biodivers. 2021, 18, e2001051. [Google Scholar] [CrossRef] [PubMed]
- You, D.-H.; Park, J.-W.; Yuk, H.-G.; Lee, S.-C. Antioxidant and tyrosinase inhibitory activities of different parts of guava (Psidium guajava L.). Food Sci. Biotechnol. 2011, 20, 1095–1100. [Google Scholar] [CrossRef]
- Lee, S.E.; Kwon, K.; Oh, S.W.; Park, S.J.; Yu, E.; Kim, H.; Yang, S.; Park, J.Y.; Chung, W.-J.; Cho, J.Y.; et al. Mechanisms of Resorcinol Antagonism of Benzo[a]pyrene-Induced Damage to Human Keratinocytes. Biomol. Ther. 2021, 29, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.Y.; Wang, H.; Tsang, D.S.C.; Chen, Z.-Y.; Leung, L.K. Screening of Chemopreventive Tea Polyphenols Against PAH Genotoxicity in Breast Cancer Cells by a XRE-Luciferase ReporterConstruct. Nutr. Cancer 2003, 46, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Tsay, J.-C.J.; Tchou-Wong, K.-M.; Greenberg, A.K.; Pass, H.; Rom, W.N. Aryl hydrocarbon receptor and lung cancer. Anticancer. Res. 2013, 33, 1247–1256. [Google Scholar] [PubMed]
- Nam, J.-H.; Seo, J.-T.; Kim, Y.-H.; Kim, K.-D.; Yoo, D.-L.; Lee, J.-N.; Hong, S.-Y.; Kim, S.-J.; Sohn, H.-B.; Kim, H.-S.; et al. Inhibitory Effects of Extracts from Arabis glabra on Lipopolysaccharide Induced Nitric Oxide and Prostaglandin E2 Production in RAW264.7 Macrophages. Korean J. Plant Resour. 2015, 28, 568–573. [Google Scholar] [CrossRef][Green Version]
- Aziz, R.; Rafiq, M.T.; Yang, J.; Liu, D.; Lu, L.; He, Z.; Daud, M.K.; Li, T.; Yang, X. Impact Assessment of Cadmium Toxicity and Its Bioavailability in Human Cell Lines (Caco-2 and HL-7702). BioMed Res. Int. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Tian, C.; Yu, X.; Fang, Y.; Zhao, X.; Zhang, X.; Xia, D. Protective Effects of Smilax glabra Roxb. Against Lead-Induced Renal Oxidative Stress, Inflammation and Apoptosis in Weaning Rats and HEK-293 Cells. Front. Pharmacol. 2020, 11, 556248. [Google Scholar] [CrossRef]
- Huang, H.-L.; Lu, Z.-Q.; Chen, G.-T.; Zhang, J.-Q.; Wang, W.; Yang, M.; Guo, D.-A. Phenylpropanoid-Substituted Catechins and Epicatechins from Smilax china. Helvetica Chim. Acta 2007, 90, 1751–1757. [Google Scholar] [CrossRef]
- Feng, H.; He, Y.; La, L.; Hou, C.; Song, L.; Yang, Q.; Wu, F.; Liu, W.; Hou, L.; Li, Y.; et al. The flavonoid-enriched extract from the root of Smilax china L. inhibits inflammatory responses via the TLR-4-mediated signaling pathway. J. Ethnopharmacol. 2020, 256, 112785. [Google Scholar] [CrossRef]
- Chen, L.; Yin, H.; Lan, Z.; Ma, S.; Zhang, C.; Yang, Z.; Li, P.; Lin, B. Anti-hyperuricemic and nephroprotective effects of Smilax china L. J. Ethnopharmacol. 2011, 135, 399–405. [Google Scholar] [CrossRef]
- Hirota, B.C.K.; Paula, C.D.S.; de Oliveira, V.B.; da Cunha, J.M.; Schreiber, A.K.; Ocampos, F.M.M.; Barison, A.; Miguel, O.G.; Miguel, M.D. Phytochemical and Antinociceptive, Anti-Inflammatory, and Antioxidant Studies of Smilax larvata (Smilacaceae). Evid.-Based Complement. Altern. Med. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Adebo, O.A.; Medina-Meza, I.G. Impact of Fermentation on the Phenolic Compounds and Antioxidant Activity of Whole Cereal Grains: A Mini Review. Molecules 2020, 25, 927. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.; Guo, H.-Z.; Cui, Y.-J.; Liu, A.-H.; Yu, H.-L.; Guo, H.; Xu, M.; Guo, D.-A. Simultaneous determination of six major stilbenes and flavonoids in Smilax china by high performance liquid chromatography. J. Pharm. Biomed. Anal. 2007, 44, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N.T.; Van Camp, J.; Smagghe, G.; Raes, K. Improved Release and Metabolism of Flavonoids by Steered Fermentation Processes: A Review. Int. J. Mol. Sci. 2014, 15, 19369–19388. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-K.; Kang, D.-J. Anti-Pollution Activity, Antioxidant and Anti-Inflammatory Effects of Fermented Extract from Smilax china Leaf in Macrophages and Keratinocytes. Cosmetics 2022, 9, 120. https://doi.org/10.3390/cosmetics9060120
Kim Y-K, Kang D-J. Anti-Pollution Activity, Antioxidant and Anti-Inflammatory Effects of Fermented Extract from Smilax china Leaf in Macrophages and Keratinocytes. Cosmetics. 2022; 9(6):120. https://doi.org/10.3390/cosmetics9060120
Chicago/Turabian StyleKim, Yoo-Kyung, and Dae-Jung Kang. 2022. "Anti-Pollution Activity, Antioxidant and Anti-Inflammatory Effects of Fermented Extract from Smilax china Leaf in Macrophages and Keratinocytes" Cosmetics 9, no. 6: 120. https://doi.org/10.3390/cosmetics9060120
APA StyleKim, Y.-K., & Kang, D.-J. (2022). Anti-Pollution Activity, Antioxidant and Anti-Inflammatory Effects of Fermented Extract from Smilax china Leaf in Macrophages and Keratinocytes. Cosmetics, 9(6), 120. https://doi.org/10.3390/cosmetics9060120





