Abstract
Methylcellulose and chitosan served as promising ingredients for a thermoresponsive hair styling gel after successful application in the medical industry. Both ingredients uphold the clean beauty standard without infringing on performance. By combining these two ingredients, a hair gel can be created that promises an extended hold of style once a heated external stimulus, such as a curling wand, is applied to the hair. Chitosan serves as the cationic biopolymer to adhere the gel to the hair, whereas the methylcellulose acts as the smart biopolymer to lock the desired hairstyle in place. Various ranges of chitosan and methylcellulose concentrations were explored for formulation optimization with rheology and curl drop testing. The rheology testing included a flow sweep test to understand the shear-thinning behavior of the sample as well as the effect of concentration on viscosity. Another rheology test completed was a temperature ramp test from room temperature (25 °C) to 60 °C to study the effect of heat on the various concentrations within the samples. A curl drop test was performed as well, over a 48-h period in which the different samples were applied to wet hair tresses, dried, curled, and hung vertically to see how the style held up over a long period of time with the influence of gravity.
1. Introduction
Smart materials are taking over the beauty industry for their personalization, differentiation, and device combination abilities after being used for years in other industries like fashion, automotive, and athletics [1]. Methylcellulose is a great example of one of the many smart materials on the market today. This ingredient has been used throughout the medical industry in numerous ways but is being implemented here for hair gel application. In this application, methylcellulose reaches its critical temperature after the use of an external stimulus (a heated curling wand), which initiates its sol-gel transformation [2]. This same sol-gel transformation is triggered by human body temperature in medical utilization [2,3,4,5]. The methylcellulose hydrogels are loaded with delivery components such as drugs or cells, which in the in-situ system go through the sol-gel transition once they are injected into the human body [5], another example of its smart abilities. Products that are formulated with smart materials tend to feel more personalized to the consumer, appealing to a much larger market.
Consumers love the personalization that smart materials add to a product. When an external stimulus is used, the product feels as though it has been revamped [1]. Throughout the beauty industry, this external stimulus is the adoption of device combinations that also support the profits of the brand. The thermosensitive ingredient within this formulation is methylcellulose, which is heat-activated once blow-dried after wet application, locking in any style when a heat-activated styling tool is used. As you apply the high-temperature styling tools to hair coated with these smart gels, the storage modulus (G’) increases with temperature, which in hand builds the crystalline structure that makes that gel-like consistency lock into place [6,7]. There is a consumer-driven desire for device combinations already in the industry, as Neutrogena has a Light Therapy Acne Mask [8] and Clarisonic [9] has a line of ultrasonic brushes that they recommend in combination with their cleansers for maximum results, as the LED light would penetrate the skin’s dermis or the Clarisonic would provide a “better-than-just-your-fingers clean.” [10]. Consumers are not only looking to purchase these personalized products, but they are often looking for more sustainable alternatives to products they already love.
The potential of formulations that use both methylcellulose and chitosan as possible natural ingredients that bring in smart gel benefits is yet to be explored. The consumers of the beauty industry are driving the clean beauty initiative as customers would rather have naturally derived ingredients in their products than synthetic ones. In the consumer industry today, customers are prioritizing sustainability in their purchases. The raw materials must be derived from environmentally and labor ethical means while upholding the original quality and performance of the initial product. These replacement ingredients must also uphold the necessary shelf-life expectancy of cosmetics or general consumer goods [11].
While there has been plenty of research done on both chitosan and methylcellulose separately, there has been little done on the combination of both ingredients. In this paper, both methylcellulose and chitosan are combined to form smart gels for hairstyling applications. Hair itself has a negative charge, so there is a need for a cationic polymer when creating this formulation. Chitosan is the cationic polymer used to ensure that the gel itself sticks to the hair, while methylcellulose is used as the thermoresponsive polymer that will lock the hairstyle in place. Frizz could make hair more difficult to manage and is often a result of hair having a negative charge as static increases and the charges start to repel each other. A temporary fix for this is coating the hair with a cationic polymer [12]. For maximum benefit, proper spreadability is required for any hair product. According to Pingali et al. [13], varying chitosan concentrations have a ranging viscosity between 10−1 and 101 Pa.s, which is an ideal range for spreadability, pumping, and rubbing. Chitosan is commercially produced from chitin. Various concentrations of salt are used to deacetylate the chitin into chitosan. Chitosan is unique in its polysaccharide chemical structure as it has an amino group attached to the C2 position in place of a hydroxyl group [14,15] (Figure 1) [16]. Even though chitosan has a comparable chemical structure to cellulose, it differs from the latter by an additional acetyl amine group that gives the ingredient entirely different attributes than cellulose [14]. Since chitosan is cellulose-based on chemical structure, it tends to have a greater intrinsic viscosity compared to other biopolymers with similar molecular weights [14]. Chitosan, as mentioned before, has a positive charge that makes it ideal for hair application. The strength of this positive charge is dependent on the concentration of sodium hydroxide that was used to deacetylate the chitin in production. Chitosan is an ingredient of surging interest in the consumer goods industry for its clean beauty aspects: biocompatibility, biodegradability, film-forming technology, and chelating agent ability [17,18]. Chitosan is also easily produced because it is the second most abundant naturally derived biopolymer behind cellulose due to its plentiful resources like crustaceans, fungi, algae, and insects [19,20]. Not only is chitosan abundant in nature, but it is also the only cationic polysaccharide abundantly found in nature. The overwhelming majority of polysaccharides found in nature are either neutral or negatively charged. [14] Any other polysaccharide would not support the attachment to hair and skin needed by cosmetic or personal care goods [21]. The second ingredient used in this research, methylcellulose, is also a biodegradable polymer and falls ahead of chitosan as the most abundant biopolymer in nature. Not only is methylcellulose largely available, but it is also a very affordable option for a biopolymer [22]. There has been a large amount of research conducted surrounding chitosan and its ability to build up viscosity [13,14]. Several different articles have been used to understand how chitosan concentrations affect the viscosity of the sample [13,14,17,18].
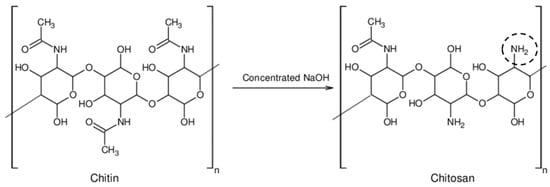
Figure 1.
Deacetylation of chitin (left) into chitosan (right).
Previous articles that have been studied prior to beginning this research have shown that ranging chitosan concentrations from 0.25% to 2.5% had consistent behavior where at low shear rates it remained a constant zero viscosity since the rate of disentanglement was the same as the rate of fresh entanglements. Meanwhile, in the power-law region, this zero-viscosity behavior changed since the rate of entanglement occurred faster under higher shear rates than the new entanglements could develop. Hwang et al. [14] observed a decrease in the viscosity of chitosan due to this behavior. Their article also observed a consistent trend where the chitosan concentration increased with shear-rate-dependent viscosity. As the number of entanglements increased, the space for movement for the current ones decreased. Therefore, as the shear rate was increased and disentanglement occurred, it then took longer to produce fresh entanglements again. Due to this lack of space, as the concentration of chitosan increased, the Newtonian behavior disappeared at lower shear rates [13]. There have not been detailed rheological studies on chitosan to the best of our knowledge.
With chitosan being used as the natural thickener for this hair gel system at room temperature, a smart polymer was needed to add the personalization aspect to this gel. As the smart gel of choice, methylcellulose was used due to its prior research within various industries for its smart applications. Methylcellulose is used across the food, ceramic, and pharmaceutical industries over the cosmetic industry and is most popularly found in gluten-free food products. This polymer is also newly introduced to the cosmetics world for its thickening and emulsifying capabilities [23]. Its rheology is dependent on a multitude of environmental and conditional factors: additives, temperature, pH level, molecular weight, polymer concentration, and ionic strength [23,24].
R. Moreira et al. [23] completed a study on the steady and dynamic rheology of methylcellulose at a temperature below gelation. They examined methylcellulose gum at 25 °C with varying concentrations of polymers at 7.5, 10, 15, and 20 g/L. During their experiment, the increased polymer concentration made the viscosity increase as well as expected at each shear rate. In their shear rate versus viscosity plots of the varying methylcellulose concentrations, there was one region with a Newtonian plateau and another with shear thinning behavior. At low and in-between shear rates, they observed the Newtonian plateau, whereas at high shear rates, the shear thinning behavior was noticed. Following this, there was a proportional increase in methylcellulose concentration and zero-shear rate viscosity. The zero-shear rate viscosity increased exponentially with the increased addition of polymer concentration. The effect of temperature on the rheology of methylcellulose in their study was only completed at a polymer concentration of 20 g/L. The originally Newtonian plateau at the low and in-between shear rates increased with temperature towards high shear rates. As the temperature increased from 5 to 35 °C, the viscosity decreased. Ultimately, their results were shear-thinning behavior at low temperatures. When they monitored pH as an effect on the rheology of the methylcellulose, there was no change in the viscoelastic behavior [23].
Smart gels have been explored for cosmetic applications, but research based on the combination of chitosan and methylcellulose is extremely limited. The application of thermoresponsive smart gels was investigated in mascara-based research by Chen and Amin [25]. The same thermoresponsive and shape memory polymer technology that emphasized the curvature of eyelashes was applied to the formulation of this hair gel for maximum hairstyle hold. Hairstyling is a part of everyday life, for some it is more demanding than others. The motivation to improve this aspect of everyday life through sensory aspects, time management, innovative technology, and so forth drives the cosmetic industry. Since there has been no work done with this technology in hairstyling, this hair gel was batched with the call-out ingredients methylcellulose and chitosan. The objective of this was to study the combination of methylcellulose and chitosan together to study long-term styling benefits. This paper explores the effect of both concentrations and further optimizes smart gel response and spreadability.
2. Materials and Methods
The smart hair gel was formulated with deacetylated chitosan, methylcellulose (400 cPs viscosity), and acetic acid (glacial). Chitosan is water-insoluble and can only dissolve in acidic solutions; in this case, a diluted acetic acid mixture was used [26]. This dilution was created by combining 0.574 g of acetic acid and filling the beaker up to the 100 mL line with deionized water, creating the 0.1 M dilution needed to dissolve chitosan. Cellulose is also water-insoluble due to its strong hydrogen bonds [27].
2.1. Formulation Design
Ten different formulations (10 g each) were batched with varying methylcellulose levels and constant chitosan levels or constant methylcellulose levels and varying chitosan levels as seen in Table 1.

Table 1.
Smart Gel Sample Formulations.
Formulations 1 through 5 held chitosan constant at 25% but varied the methylcellulose at 0%, 0.1%, 0.5%, 1%, and 2.75%. The following 6 through 10 samples held methylcellulose constant at 1% and varied the chitosan levels at 0%, 0.25%, 0.50%, 0.75%, and 1%. All three ingredients were weighed out for each sample and mixed in separate labeled vials on an unheated stirring plate for four hours, or until the gels were fully mixed through. Each gel was left on the bench, capped off for 24 h after mixing to reach equilibrium before testing. pH values were measured and not varied. They were kept constant at a value of 4.67 for every sample formulated. A low pH value is ideal for safe application to hair, with no frizzing or damage applied to the hair itself.
2.2. Rheology
The varying concentrations of methylcellulose and chitosan were evaluated with rheology using the Discovery Hybrid Rheometer (DHR-3) from TA Instruments [Delaware, USA]. For each of the rheological tests, a 40 mm cone plate with a 2-degree angle was used. Rheological tests were performed to understand the spreadability of each sample as well as the shear thinning behavior that each sample exerts and finally its response to thermal activity. The spreadability and shear thinning behavior of each sample were tested by running a flow sweep test at room temperature (25 °C) with an increasing shear rate range from 1 to 1000 s−1. To better understand how each sample reacts to a thermal stimulus, a temperature sweep test was performed from room temperature (25 °C) to 60 °C. As the gel heats up, the structure of the gel will change from fully amorphous to fully crystalline, locking in any hairstyle or shape [1]. Finally, an amplitude sweep test was also performed at a constant angular frequency of 10 rad/s to understand the linear viscoelastic region of each sample. All measurements were taken under steady-state conditions.
2.3. Curl Drop Test
A visual hair curl drop test was completed with each of the ten smart gel formulations. Eleven hair tresses that were the same thickness, color, and style of hair were selected for testing. Each hair tress was washed with sodium lauryl sulfate (SLS) until the hair was fully clean of any impurities. One gram of formulated smart gel was applied to each wet hair tress, immediately followed by hair drying. A control piece of hair was washed and dried without any gel on it for each of the two 48-h tests that were run. After drying was completed, each hair tress was held around a Foxy Bae 25mm ceramic curling wand for 15 s at 410 °F (210 °C). The hair tresses were then hung up vertically in either increasing chitosan concentration or increasing methylcellulose concentration next to a labeled control piece. Each hair tress post styling was monitored by a curl drop test at 0, 24, and 48 h to see the effect of either concentration. The curl drop test was performed in a 75°F (approximately 24 °C) room with little to no humidity.
3. Results and Discussion
3.1. Rheological Testing: Flow Sweep (Viscosity vs. Shear Rate)
One type of rheological test that was performed was a flow sweep to understand the relationship between varying amounts of methylcellulose and chitosan and their viscosity. This data gives more insight into the spreadability factor of the hair gel and how it will perform while being applied to hair. As the sample is subjected to more shear, it will exhibit typical shear thinning behavior. Ideally, the faster the sample loses viscosity at higher shear rates, the better the product will spread once applied to the hair. Figure 2 and Figure 3 show the results of methylcellulose and chitosan concentration on the viscosity of the hair gel as the shear rate increases from 1 to 1000 s−1.
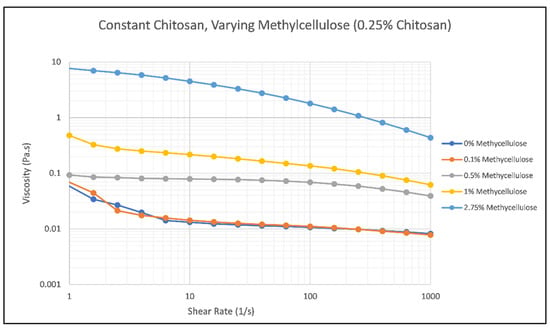
Figure 2.
Effect of Methylcellulose Concentration on the viscosity of Samples 1 (blue), 2 (orange), 3 (gray), 4 (yellow), and 5 (green) as the shear rate increases from 1 to 1000 s−1.
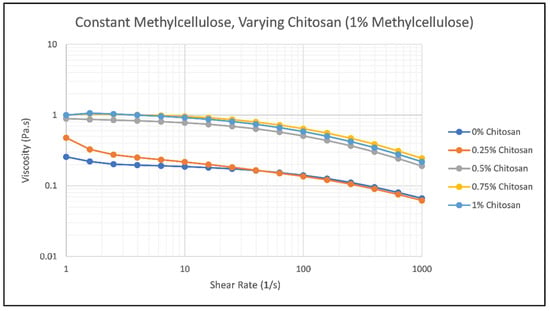
Figure 3.
Effect of Chitosan Concentration on the viscosity of Samples 6 (blue), 7 (orange), 8 (gray), 9 (yellow), and 10 (green) as the shear rate increases from 1 to 1000 s−1.
As the methylcellulose concentration increases, the viscosity of the hair gel increases very slightly when the chitosan concentration remains constant at 0.25%. This trend can be clearly seen in Figure 2 as the methylcellulose concentration increases from 0% to 2.75%, with Sample 5 having the highest viscosity out of Samples 1, 2, 3, and 4. The samples show typical shear-thinning behavior, with viscosity decreasing as the shear rate increases. Methylcellulose does not impact the viscosity of the samples as much as chitosan does at room temperature. The thinner samples (1–5) provide more difficulty when being applied to the hair versus the thicker samples (6–10). Figure 2 confirms that the thinner samples do not have the best spreadability due to them not losing as much viscosity over time as the samples in Figure 3.
As noted previously, increasing the overall concentration of chitosan within the solution at room temperature increases the viscosity of the sample, which can be seen in Figure 3. As the concentration of chitosan increases, the number of entanglements within the sample also increases, increasing viscosity and shear stress [28]. Sample 10, the sample containing the highest concentration of chitosan, has the highest viscosity and a very intense viscosity-shear rate relationship compared to any of the samples tested. The viscosities of these samples decrease rapidly, and the ending viscosities, especially of Sample 10, are quite different from their starting viscosities, changing by values as large as 7 Pa s., Sample 5 (which can be seen in Figure 2) also exhibits this behavior. With these substantial changes in viscosity over a shear rate ranging from 1 to 1000 s−1, these samples have better spreadability factors when compared to Samples 1 through 4. Thickness did become an issue during the curl drop test that was performed later in the research, especially when dealing with Sample 10.
3.2. Rheological Testing: Temperature Ramp (G’ vs. Temperature)
When the smart gels are applied to hair, they are activated via a heat source such as blow drying. Application testing was done on both wet and dry hair, concluding that application of the gel on wet hair results in a more evenly distributed product versus dry hair, which resulted in the separation and clumping of hair. As the temperature of the smart gel increases, the gel itself should start to thicken, indicating that its structure is becoming more crystalline and solid. This is due to the thermoresponsive behavior of methylcellulose, which gels as temperature increases due to highly substituted hydrophobic groups forming attractive complexes [29]. Figure 4 and Figure 5 show the results of methylcellulose and chitosan concentration effects on the elastic behavior (G’) of the smart gels versus increasing temperature.
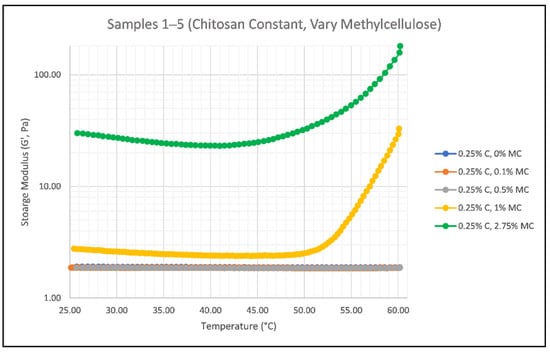
Figure 4.
Effect of Methylcellulose Concentration on the Storage Modulus (G’) of Samples 1 (blue), 2 (orange), 3 (gray), 4 (yellow), and 5 (green) as temperature increases from 25 °C to 60 °C; where C is chitosan and MC is methylcellulose.
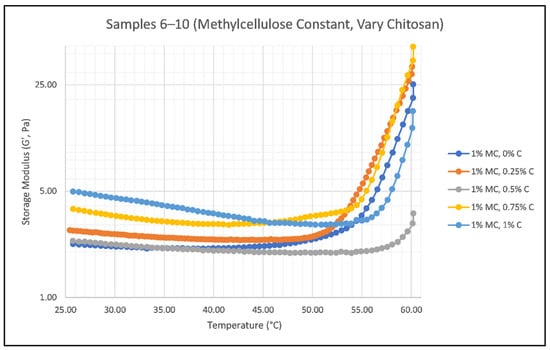
Figure 5.
Effect of Chitosan Concentration on the Storage Modulus (G’) of Samples 6 (blue), 7 (orange), 8 (gray), 9 (yellow), and 10 (green) as temperature increases from 25 °C to 60 °C; where C is chitosan and MC is methylcellulose.
As the temperature of the gel increases, gel formation should occur and the values of G’ should increase, telling us about the solid-like behavior of the gel. For increasing methylcellulose concentrations, this relationship is noted in Figure 4 by the sharp increase in G’ values for Sample 5 which has the highest concentration of methylcellulose (2.75%). This relationship is not noticeably clear between Samples 1, 2, and 3, which have low amounts of the thermosensitive ingredient. Sample 4 was created using twice the amount of methylcellulose used in Sample 3 and its linear relationship with G’ is clearly seen in Figure 4. The thermal responsiveness of the system is enhanced as the methylcellulose concentration increases [30]. This idea should coincide with the curl drop test, suggesting that higher methylcellulose concentrations should result in a longer-lasting curl due to its more elastic solution. The structure of the gel at high methylcellulose concentrations would “lock” the curl in place as its structure becomes more crystalline, granting a longer curl hold time.
Chitosan itself does not have thermoresponsive properties, but when it is used in a system with an acid (in this case, acetic acid), it forms a thick gel at room temperature [31,32]. Therefore, in Figure 5, there is no clear trend between increasing chitosan concentrations and increasing temperatures. There is an increase in G’ values for all samples as temperature increases, but the chitosan concentration has no linear effect. The upward transition temperature is impacted by the concentration of chitosan due to the interactions between chitosan and methylcellulose as relative amounts of each are changed. While formulating each sample, the viscosity of the sample can be increased by increasing the concentration of chitosan [33]. The smart gel should ideally be thick enough to hold its shape once it is on the consumer’s hand without flowing off, but it should not be too thick that it is difficult to remove from its container. Samples 6 and 7 had little to no viscosity due to their exceptionally low levels of chitosan. This product would not sell very well on the market if it were labeled as a “gel” yet had an incredibly low viscosity. Sample 10 was the most viscous out of all ten samples, which rendered it almost impossible to remove from the sample vial to test.
3.3. Curl Drop Test
Eleven hair tresses were washed with SLS, had 1 g of their respective smart hair gel applied directly to the wet hair tresses, were immediately dried with a blow dryer, and curled with a Foxy Bae 25 mm ceramic curling wand for 15 s at 410 °F (210 °C). These hair tresses are labeled below: Figure 6a–c show a control next to samples 1, 2, 3, 4, and 5 from right to left, and Figure 7a–c show a control next to samples 6, 7, 8, 9, and 10 from right to left. Hair tress C (control) was washed with SLS, but no smart hair gel was applied, immediately dried with a blow dryer, and curled using the same method. Hair tresses 1 through 5 had a constant chitosan wt % at 0.25 and a varying methylcellulose wt % between 0 and 2.75. Hair tresses 6 through 10 had a constant methylcellulose concentration of 1% but a varying chitosan wt % between 0 and 1.

Figure 6.
(a): Control piece of hair (far left) next to Samples 1 (next to control) through 5 (far right) curl drop test. Photo was taken at 0 h right after gel application. (b): Control piece of hair (far left) next to Samples 1 (next to control) through 5 (far right) curl drop test. Photo was taken at 24 h right after gel application. (c): Control piece of hair (far left) next to Samples 1 (next to control) through 5 (far right) curl drop test. Photo was taken 48 h after gel application.


Figure 7.
(a): Control piece of hair (far left) next to Samples 6 (next to control) through 10 (far right) curl drop test. Photo was taken at 0 h right after gel application. (b): Control piece of hair (far left) next to Samples 6 (next to control) through 10 (far right) curl drop test. Photo was taken 24 h after gel application. (c): Control piece of hair (far left) next to Samples 6 (next to control) through 10 (far right) curl drop test. Photo was taken 48 h after gel application.
The curl drop test is a great way to picture how the smart gels will react after being applied to the hair. These hair gels are formulated using shape memory polymers (SMPs) that allow the hair to be deformed into a temporary style (in this test, a curl) due to the hard and soft segments within the SMPs [34,35]. Once an external stimulus is applied, in this case, both the hair dryer used to dry the wet tresses as well as the curling wand, the SMPs within the hair crosslink, which provided a longer-lasting styling effect [36]. Looking at Figure 6a–c, it can clearly be seen that the hair tress with Sample 5 has the longest-lasting curl over a 48-h period compared to the other samples. This sample has the highest methylcellulose concentration and when compared to Figure 4, it can be concluded that this sample has the highest thermal responsiveness out of all the samples formulated, which, in turn, results in a longer-lasting curl. Samples 1, 2, 3, and 4 all experienced a large drop in curl over the 48-h period compared to the control piece, which had no product on it. The control piece managed to sustain quite a curl over 2 days. Sample 2 had the most exaggerated drop in curl, showcasing the slightest bend at the 48-h mark. Samples 3, 4, and 5 all experienced a little bit of hair separation and clumping at the ends of the hair due to higher levels of methylcellulose causing strands of hair to stick together. This is very minimal and is only noticed up close, but it is something to account for when it comes to the aesthetic properties of the gel. This issue is also highlighted in Figure 7a–c.
The next part of this curl drop test studied the effects of chitosan on the longevity of the curls. Chitosan exhibits an excellent ability to form a film around the hair tresses, acting as a protectant between the hair itself and damaging heat [37,38]. As seen in Figure 7a–c, there is quite a bit of separation between the strands of hair as the chitosan concentration increases, especially in Samples 9 and 10. After a total of 48 h, Samples 8, 9, and 10 held the best curl compared to Samples 6 and 7. Although they provided the best curl, Samples 9 and 10 also created lots of separation and a “crunchy” sensation to the hair tresses, which would not be an ideal sensory aspect for the consumer. Formulations containing higher levels of chitosan often have significantly lower levels of acceptability when it comes to sensory testing [39]. Since each sample formulated only contained 10 g of sample, as more chitosan was added to the system, less acetic acid was added. A higher amount of acid promotes a smoother and less “crunchy” film [40]. Overall, Sample 8 had the best curl of this second batch of samples over a 48-h period.
4. Conclusions
Inspiration for this research was drawn from an increasing need for more personalized products within the cosmetics industry. By employing ingredients that are labeled as “smart” materials, products can be formulated on a large scale yet feel like they were made specifically for the consumer who purchases it. There has been much research done on both chitosan and methylcellulose formulations, but not a ton of research done on formulations combining the two. Methylcellulose, a clean beauty ingredient with thermoresponsive behavior, was included in this smart hair gel after its success in medical technology and its sustainability aspects. Five various levels of methylcellulose (ranging from 0% to 2.75%) were used with a constant level of chitosan (0.25%) to see the effects of varying methylcellulose levels on the gels. This set of gels experienced an extremely low viscosity, making the application onto wet hair tresses quite difficult. Chitosan, a natural thickener derived from the shell of shrimp, was used as the viscosity builder in this gel. When combined with acetic acid, a gel-like base is created. Five various levels of chitosan (ranging from 0% to 1%) were used in a constant level of methylcellulose (1%) to see how the various levels of chitosan affect the gels. Adding elevated levels of chitosan resulted in very viscous products, some being too thick to flow out of the test vial for hair application. Combining all three ingredients together provides a thick, thermoresponsive gel that is sustainable.
Rheology was used as the main point of testing to understand how the gel would react once it was applied to hair. A flow sweep test was completed at room temperature with an increasing shear rate range from 1 to 1000 s−1 to understand the spreadability aspect of the gels. An ideal gel should be quite thick but should still flow out of the test vial without any extra force. If the sample is too thin, it will be exceedingly difficult to apply to the hair. On the other hand, if a sample is too thick, it may clump up on the hair and make it impossible to fully coat the hair. The more chitosan that was added to the samples, the higher the viscosity levels they experienced. These samples also experienced a much larger drop in viscosity over this shear rate range, concluding that their spreadability, when compared to the first set, is better. The samples ranging in methylcellulose concentration did not see a large decrease in viscosity after a flow sweep test was performed, relating to the impact of spreadability on the hair. These samples with varying methylcellulose levels resulted in thin samples that were too difficult to apply to the hair. A temperature sweep test was also completed on the rheometer at a temperature range increasing from 25 °C to 60 °C to study the thermoresponsive properties of the gels. In rheology testing, the storage modulus (G’) increased with temperature, which was highest for sample 5, with the greatest methylcellulose concentration at 2.75%. Since chitosan does not have a thermoresponsive element, its relationship with temperature would not be established based on storage modulus with rheology testing. Finally, a curl drop test was performed with all 10 samples over a 48-h period to see how the gels would apply and style. Increased chitosan concentration hinders positive sensory experience as it causes separation and crunchiness, sample 8 had the best curl without this negative consumer issue.
Creating a thermoresponsive hair gel is a great alternative to the many types of hair gels on the market that advertise holding a style in place for an extended period. This smart gel will enable hairstyles to truly be “locked” into place due to the addition of smart biopolymerssetting the hairstyle in place for hours on end. This idea was confirmed by performing a curl drop test to see how different methylcellulose levels lock the curls in place. The addition of chitosan is also a more sustainable option since methylcellulose is derived from shrimp skin. Methylcellulose is also another sustainable ingredient derived from plants, making this simple hair gel formulation a great sustainable alternative to the many different types of gels on the market. Applying this smart gel on hair acts as both a heat protectant due to the chitosan acting as a film-former on the hair, protecting it from any heat damage and acting as an ultra-hold hairspray on top. This, in turn, results in the need for fewer products on the hair, which can save costs in the end. It also provides the need to use less heat on the hair, since the hairstyles will hold for a longer period without the need for heat touchups.
5. Next Steps and Recommendations
Since chitosan is derived from shrimp, which is a widely known food allergen, the vegetable-derived chitosan should be applied and tested in this hair gel for greater consumer use. Moreover, since the cosmetic lab at Manhattan College does not have a controlled humidity chamber, a frizz test should be performed on the formulations for continued optimization. The final optimization of this formulation would include more ingredients to enhance the hair’s aesthetic beyond the baseline ingredients presently included. Due to the lack of financial resources, a large-scale consumer study for ideal sensory experience on differing formulations is also required. At this point, this step is beyond the scope of this study. Lastly, a differential scanning calorimeter (DCS) should be used to understand the temperature at which the crystallization may occur [41].
Author Contributions
Conceptualization and methodology, S.A.; investigation and validation, M.H. and C.C.; data curation and visualization, M.H. and C.C.; supervision and analysis, S.A.; writing, M.H. and C.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All of the data reported is in this publication and is not generated on any other platform.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hartson, M.; Coyle, C.; Amin, S. Smart Materials and Formulations Help Differentiate Beauty Products. Household and Personal Products Industry Magazine. 2021. Available online: https://www.happi.com/issues/2021-09-01/view_features/smart-materials-and-formulations-help-differentiate-beauty-products/ (accessed on 19 February 2022).
- Bonetti, L.; De Nardo, L.; Fare, S. Thermo-Responsive Methylcellulose Hydrogels: From Design to Applications as Smart Biomaterials. Tissue Eng. Part B Rev. 2021, 27, 486–513. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-J.; Matsunaga, Y.T. Thermo-Responsive Polymers and Their Application as Smart Biomaterials. J. Mater. Chem. B 2017, 5, 4307–4321. [Google Scholar] [CrossRef] [PubMed]
- Ruel-Gariépy, E.; Leroux, J.-C. In Situ-Forming Hydrogels—Review of Temperature-Sensitive Systems. Eur. J. Pharm. Biopharm. 2004, 58, 409–426. [Google Scholar] [CrossRef]
- Klouda, L. Thermoresponsive Hydrogels in Biomedical Applications: A Seven-Year Update. Eur. J. Pharm. Biopharm. 2015, 97, 338–349. [Google Scholar] [CrossRef]
- Fattahpour, S.; Shamanian, M.; Tavakoli, N.; Fathi, M.; Sadeghi-aliabadi, H.; Sheykhi, S.R.; Fesharaki, M.; Fattahpour, S. An Injectable Carboxymethyl Chitosan-Methylcellulose-Pluronic Hydrogel for the Encapsulation of Meloxicam Loaded Nanoparticles. Int. J. Biol. Macromol. 2020, 151, 220–229. [Google Scholar] [CrossRef]
- Tang, Y.-F.; Du, Y.-M.; Shi, X.-W.; Kennedy, J.F. Rheological Characterisation of a Novel Thermosensitive Chitosan/Poly (Vinyl Alcohol) Blend Hydrogel. Carbohydr. Polym. 2007, 67, 491–499. [Google Scholar] [CrossRef]
- Krieger, L. Acne Mask Could be Your New Go-to Zit Zapper; Johns. Johnson: New Brunswick, NJ, USA, 2016. [Google Scholar]
- Newsweek Amplify. Your Favorite Clarisonic Cleansing Brushes Are Back—Here’s Where to Buy Them Online. 2020. Available online: https://www.newsweek.com/amplify/your-favorite-clarisonic-cleansing-brushes-are-back-heres-where-buy-them-online (accessed on 27 February 2022).
- Fasanella, K.; Han, S. The Best Skin-Care Tools and Devices to Try at Home. Allure. 2020. Available online: https://www.allure.com/gallery/new-skin-care-tools-devices (accessed on 27 February 2022).
- Bom, S.; Jorge, J.; Ribeiro, H.M.; Marto, J. A Step Forward on Sustainability in the Cosmetics Industry: A Review. J. Clean. Prod. 2019, 225, 270–290. [Google Scholar] [CrossRef]
- Abrutyn, E.S. Deciphering Frizz Control Hair Care Formulas; Cosmetics & Toiletries: Carol Stream, IL, USA, 2013; Available online: https://www.cosmeticsandtoiletries.com/formulas-products/hair-care/blog/21837664/deciphering-frizz-control-hair-care-formulas (accessed on 2 May 2022).
- Pingali, S.; Benhur, A.M.; Amin, S. Engineering Rheological Response in Chitosan-Sophorolipid Systems through Controlled Interactions. Int. J. Cosmet. Sci. 2020, 42, 407–414. [Google Scholar] [CrossRef]
- Hwang, J.K.; Shin, H.H. Rheological Properties of Chitosan Solutions. Korea-Aust. Rheoogy J. 2000, 12, 175–179. [Google Scholar]
- Varum, K.M.; Ottoy, M.H.; Smidsrod, O. Water-Solubility of Partially N-Acetylated Chitosans as a Function of pH: Effect of Chemical Composition and Depolymerization. Carbohydr. Polym. 1994, 25, 65–70. [Google Scholar] [CrossRef]
- Szatkowski, T.; Kolodziejczak-Radzimska, A.; Zdarta, J.; Szwarc-Rzepka, K.; Paukszta, D.; Wysokowski, M.; Ehrlich, H.; Jesionowski, T. Synthesis and Characterization of Hydroxyapatite/Chitosan Composites. Physiochemical Probl. Miner. Process. 2015, 51, 575–585. [Google Scholar]
- Hossain, M.S.; Iqbal, A. Production and Characterization of Chitosan from Shrimp Waste. J. Bangladesh Agric. Eng. 2014, 12, 143–160. [Google Scholar] [CrossRef] [Green Version]
- Rout, S.K. Physicochemical, Functional and Spectroscopic Analysis of Crawfish Chitin and Chitosan as Affected by Process Modification; LSU and Agricultural and Mechanical College: Baton Rouge, LA, USA, 2001. [Google Scholar]
- Mo, H.K.; Meyers, S.P. Utilization of Crawfish Processing Wastes as Carotenoids, Chitin, and Chitosan Sources. J. Korean Soc. Food Nutr. 1992, 21, 319–326. [Google Scholar]
- Tolamiate, A.; Debrieres, J.; Rhazi, M.; Alagui, M.; Vincendon, M.; Vottero, P. On the Influence of Deacetylation Process on the Physiochemical Characteristics of Chitosan from Squid Chitin. Polymer 2000, 41, 2463–2469. [Google Scholar] [CrossRef]
- Muzzarelli, R. Chitosan-Based Dietary Foods. Carbohydr. Polym. 1996, 29, 309–316. [Google Scholar] [CrossRef]
- Rimdusit, S.; Jingjid, S.; Damrongsakkul, S.; Tiptipakorn, S.; Takeichi, T. Biodegradability and Property Characterizations of Methyl Cellulose: Effect of Nanocompositing and Chemical Crosslinking. Carbohydr. Polym. 2008, 72, 444–455. [Google Scholar] [CrossRef]
- Moreira, R.; Chenlo, F.; Silva, C.; Torres, M. Rheological Behaviour of Aqueous Methylcellulose Systems: Effect of Concentration, Temperature and Presence of Tragacanth. LWT 2017, 84, 764–770. [Google Scholar] [CrossRef]
- Clasen, C.; Kulicke, W.M. Determination of Viscoelastic and Rheology-Optical Material Functions of Water-Soluble Cellulose Derivates. Prog. Polym. Sci. 2001, 25, 1839–1919. [Google Scholar] [CrossRef]
- Chen, S.; Amin, S. Design of High Performance Curlin Mascara through Utilization of Smart Thermoresponsive Polymer. Int. J. Cosmet. Sci. 2020, 42, 557–563. [Google Scholar] [CrossRef]
- Silva, E.; Paula, A.; Silva, V.; Alvarenga, A.; Bertolucci, S. Biostimulating Effect of Chitosan and Acetic Acid on the Growth and Profile of the Essential Oil of Mentha arvensis L. Ind. Crops Prod. 2021, 171, 113987. [Google Scholar] [CrossRef]
- Mondal, M.I.H. Cellulose-Based Superabsorbent Hydrogels. Polym. Polym. Compos. Ref. Ser. 2019. [Google Scholar] [CrossRef]
- Mucha, M. Rheological Characteristics of Semi-Dilute Chitosan Solutions. Macromol. Chem. Phys. 1997, 198, 471–484. [Google Scholar] [CrossRef]
- Desbrières, J.; Hirrien, M.; Ross-Murphy, S.B. Thermogelation of Methylcellulose: Rheological Considerations. Polymer 2000, 41, 2451–2461. [Google Scholar] [CrossRef]
- Eskens, O.; Villani, G.; Amin, S. Rheological Investigation of Thermoresponsive Alginate-Methylcellulose Gels for Epidermal Growth Factor Formulation. Cosmetics 2020, 8, 3. [Google Scholar] [CrossRef]
- Speer, S.; Amin, S. Sustainable Thermoresponsive Whey Protein-and Chitosan-Based Oil-In-Water Emulsions for Cosmetic Applications. Int. J. Cosmet. Sci. 2021, 44, 30–41. [Google Scholar] [CrossRef]
- Argüelles-Monal, W.; Recillas-Mota, M.; Fernández-Quiroz, D. Chitosan-Based Thermosensitive Materials. Biol. Act. Appl. Mar. Polysacch. 2017. [Google Scholar] [CrossRef] [Green Version]
- Kienzle-Sterzer, C.A.; Rodriguez-Sanchez, D.; Rha, C.K. Flow Behavior of a Cationic Biopolymer: Chitosan. Polym. Bull. 1985, 13, 1–6. [Google Scholar] [CrossRef]
- Bai, Y.; Liu, Y.; Wang, Q. Cellulose Acetate for Shape Memory Polymer: Natural, Simple, High Performance, and Recyclable. Adv. Polym. Technol. 2018, 37, 869–877. [Google Scholar] [CrossRef]
- Hoogenboom, R. Temperature-Responsive Polymers: Properties, Synthesis, and Applications. Smart Polym. Appl. 2014. [Google Scholar] [CrossRef]
- Hu, J.; Zhu, Y.; Chen, S.; Luo, H.; Lu, J. Conditioning Agent and a Method for Making Hair Having a Shape Memory Effect. U.S. Patent No. 8,349,299, 8 January 2013. [Google Scholar]
- Thao, N.T.; Wijerathna, H.; Kumar, R.S.; Saravana; Choi, D.; Dananjaya, S.; Attanayake, A.P. Preparation and Characterization of Succinyl Chitosan and Succinyl Chitosan Nanoparticle Film: In Vitro and In Vivo Evaluation of Wound Healing activity. Int. J. Biol. Macromol. 2021, 193, 1823–1834. [Google Scholar] [CrossRef]
- Qin, C.; Li, H.; Xiao, Q.; Liu, Y.; Zhu, J.; Du, Y. Water-Solubility of Chitosan and its Antimicrobial Activity. Carbohydr. Polym. 2006, 63, 367–374. [Google Scholar] [CrossRef]
- Kumar, R.; Xavier, K.M.; Lekshmi, M.; Balange, A.; Gudipati, V. Fortification of Extruded Snacks with Chitosan: Effects on Techno Functional and Sensory Quality. Carbohydr. Polym. 2018, 194, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Hekiem, N.L.L.; Ralib, A.A.M.; Hatta, M.A.M.; Ahmad, F.B.; Nordin, A.N.; Ab Rahim, R.; Za’Bah, N.F. Effect of Chitosan Dissolved in Different Acetic Acid Concentration towards VOC Sensing Performance of Quartz Crystal Microbalance Overlay with Chitosan. Mater. Lett. 2021, 291, 129524. [Google Scholar] [CrossRef]
- Usmani, M.; Khan, I.; Haque, A.; Bhat, A.; Mondal, D.; Gazal, U. Biomass-Based Composites from Different Sources: Properties, Characterization, and Transforming Biomass with Ionic Liquids. Compos. Sci. Eng. 2017, 1, 45–76. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).